Carbon Transfer from the Host Diatom Enables Fast Growth and High Rate of N2 Fixation by Symbiotic Heterocystous Cyanobacteria
Abstract
:1. Introduction
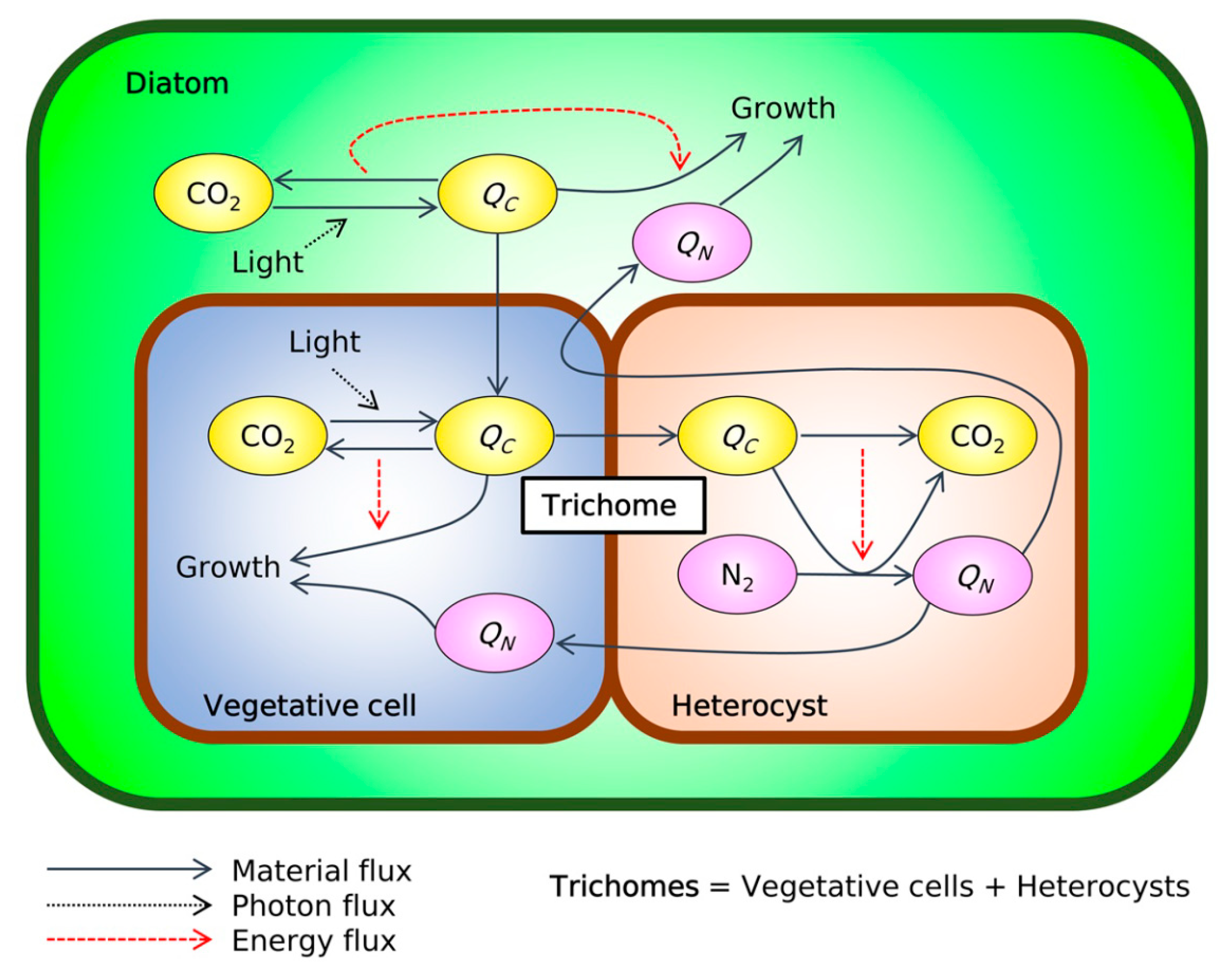
2. Results and Discussion
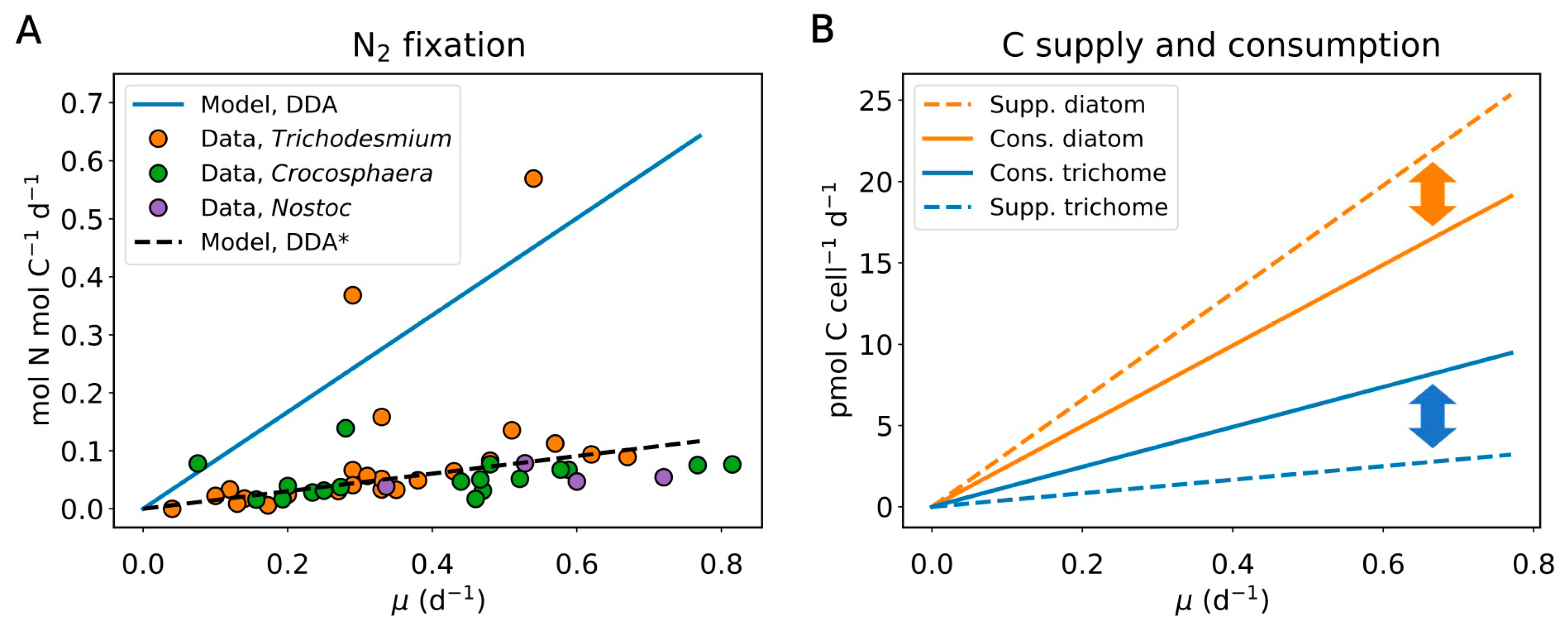
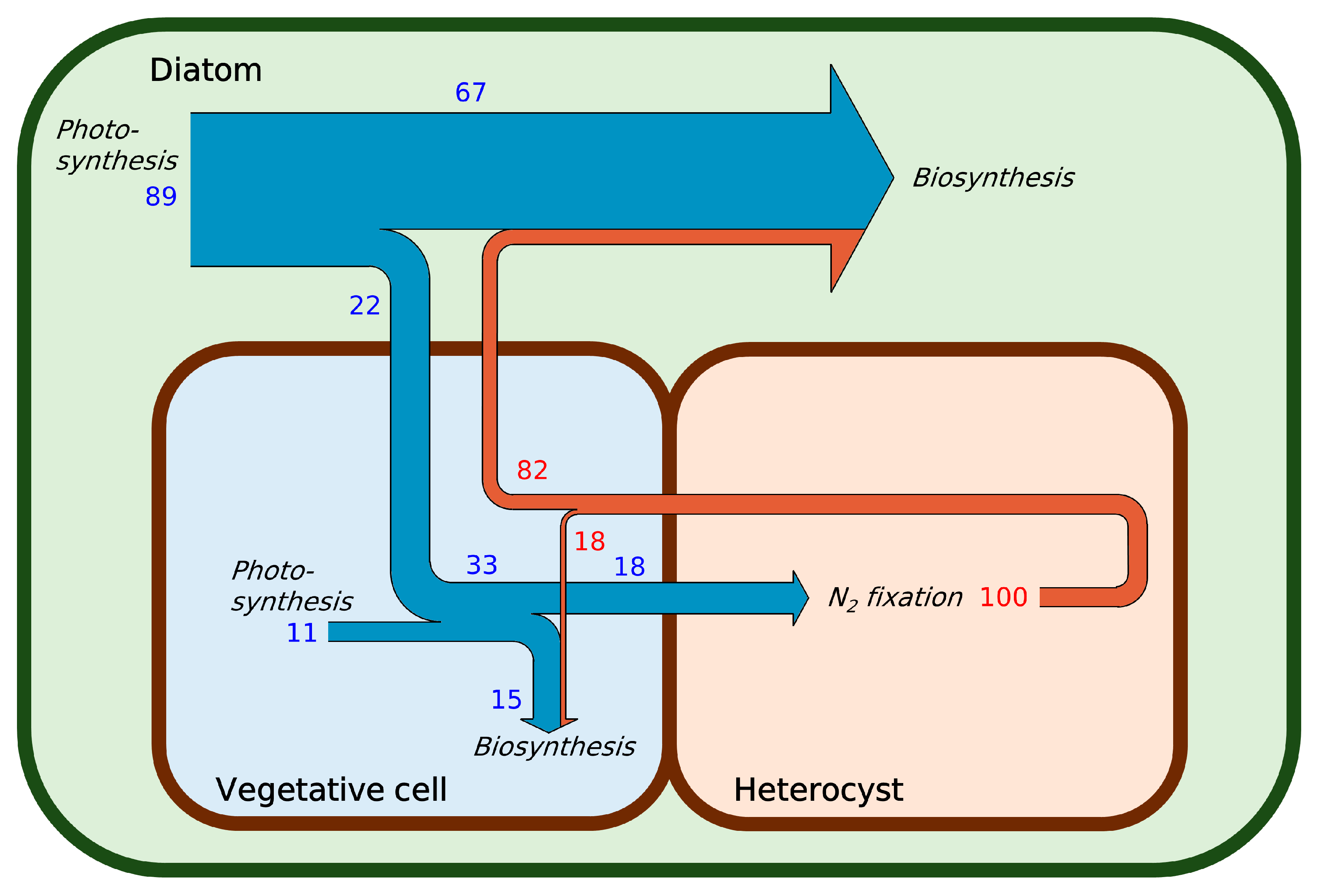
2.1. Nitrogen Budget
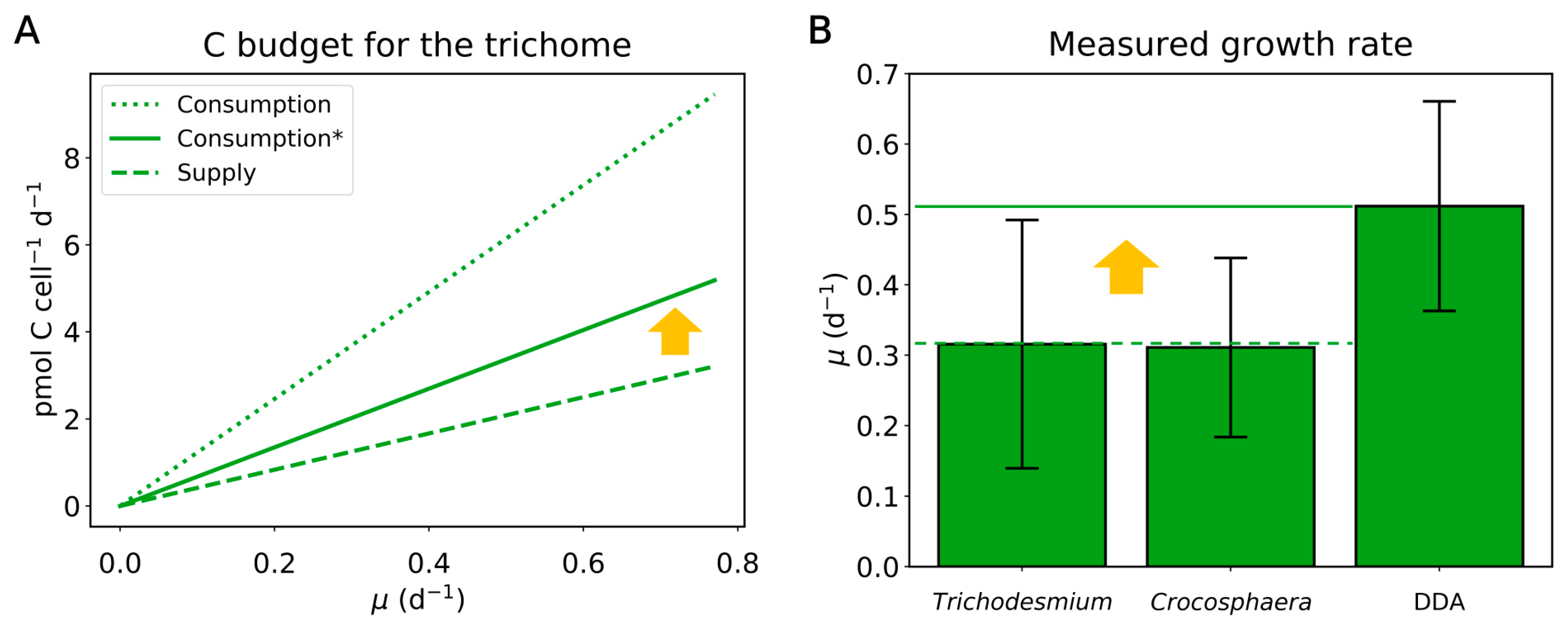
2.2. Carbon Budget
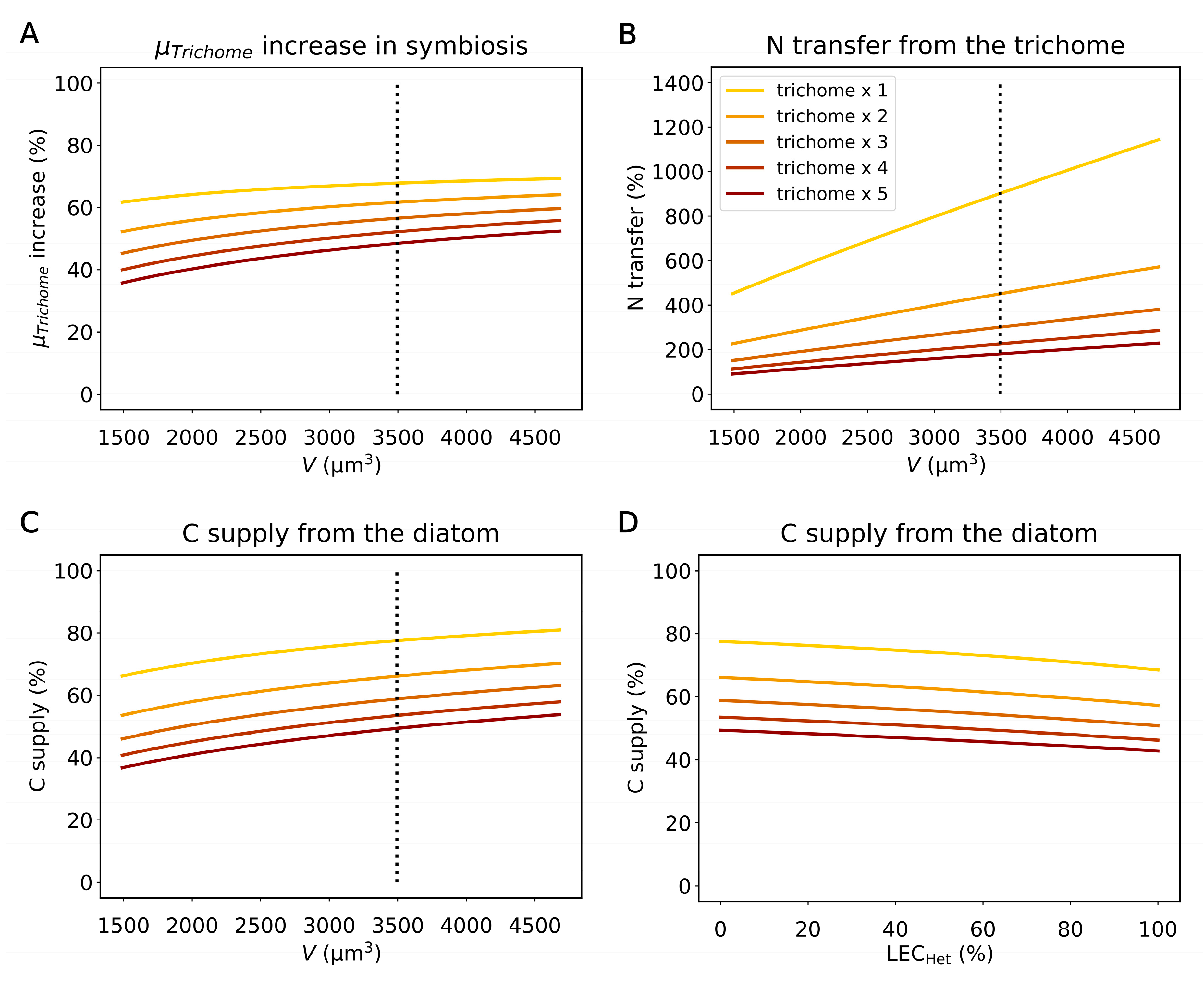
2.3. The Influence of the Diatom Size, Trichome Counts, and Light Harvesting by Heterocysts
2.4. Implication of the Model Results for Other DDAs
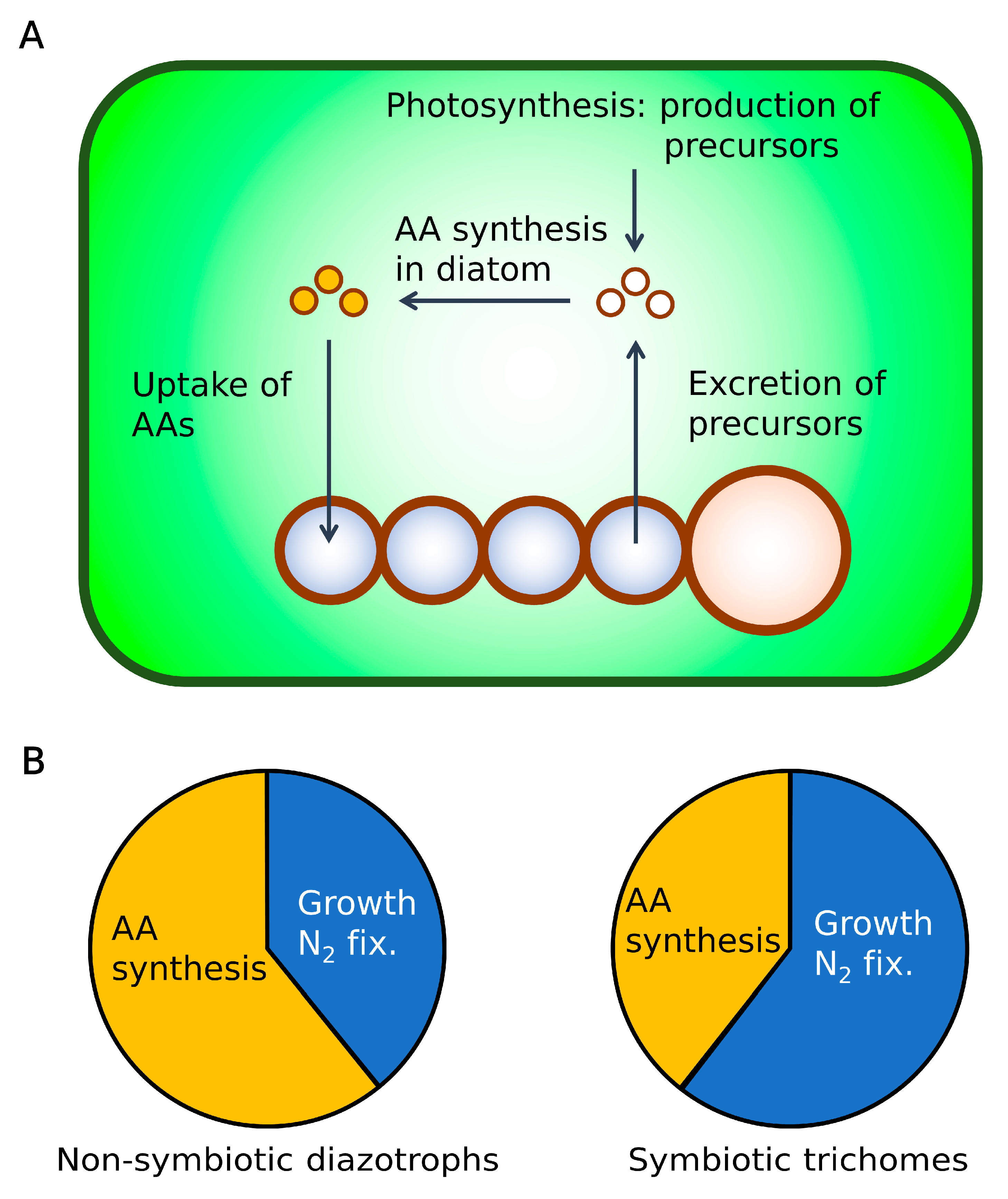
2.5. Hypothesis: Mechanism for High C Processing
3. Conclusions
4. Methods
Supplementary Materials
Author Contributions
Funding
Model Availability
Acknowledgments
Conflicts of Interest
References
- Villareal, T.A. Laboratory culture and Preliminary Characterization of the nitrogen-fixing Rhizosolenia-Richelia Symbiosis. Mar. Ecol. 1990, 11, 117–132. [Google Scholar] [CrossRef]
- Villareal, T.A. Marine nitrogen fixing diatom - cyanobacteria symbioses. In Marine Pelagic Cyanobacteria: Trichodesmium and Other Diazotrophs; Carpenter, E.J., Capone, D.G., Rueter, J.G., Eds.; Kluwer Academic Publishers: Dordrecht, The Neatherlands, 1992; pp. 163–175. [Google Scholar]
- Janson, S.; Wouters, J.; Bergman, B.; Carpenter, E.J. Host specificity in the Richelia-diatom symbiosis revealed by hetR gene sequence analysis. Environ. Microbiol. 1999, 1, 431–438. [Google Scholar] [CrossRef]
- Foster, R.A.; Zehr, J.P. Characterization of diatom-cyanobacteria symbioses on the basis of nifH, hetR and 16S rRNA sequences. Environ. Microbiol. 2006, 8, 1913–1925. [Google Scholar] [CrossRef]
- Goebel, N.L.; Turk, K.A.; Achilles, K.M.; Paerl, R.; Hewson, I.; Morrison, A.E.; Montoya, J.P.; Edwards, C.A.; Zehr, J.P. Abundance and distribution of major groups of diazotrophic cyanobacteria and their potential contribution to N2 fixation in the tropical Atlantic Ocean. Environ. Microbiol. 2010, 12, 3272–3289. [Google Scholar] [CrossRef]
- Foster, R.A.; Kuypers, M.M.M.; Vagner, T.; Paerl, R.W.; Musat, N.; Zehr, J.P. Nitrogen fixation and transfer in open ocean diatom– cyanobacterial symbioses. ISME J. 2011, 5, 1484–1493. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hilton, J.A.; Foster, R.A.; James Tripp, H.; Carter, B.J.; Zehr, J.P.; Villareal, T.A. Genomic deletions disrupt nitrogen metabolism pathways of a cyanobacterial diatom symbiont. Nat. Commun. 2013, 4, 1767. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carpenter, E.J.; Montoya, J.P.; Burns, J.; Mulholland, M.R.; Subramaniam, A.; Capone, D.G. Extensive bloom of a N2-fixing diatom/cyanobacterial association in the tropical Atlantic Ocean. Mar. Ecol. Prog. Ser. 1999, 185, 273–283. [Google Scholar] [CrossRef] [Green Version]
- Gómez, F.; Furuya, K.; Takeda, S. Distribution of the cyanobacterium Richelia intracellularis as an epiphyte of the diatom Chaetoceros compressus in the western Pacific Ocean. J. Plankton Res. 2005, 27, 323–330. [Google Scholar] [CrossRef] [Green Version]
- Church, M.J.; Mahaffey, C.; Letelier, R.M.; Lukas, R.; Zehr, J.P.; Karl, D.M. Physical forcing of nitrogen fixation and diazotroph community structure in the North Pacific subtropical gyre. Global Biogeochem. Cycles 2009, 23, GB2020. [Google Scholar] [CrossRef]
- Foster, R.A.; Subramaniam, A.; Zehr, J.P. Distribution and activity of diazotrophs in the Eastern. Environ. Microbiol. 2009, 11, 741–750. [Google Scholar] [CrossRef] [Green Version]
- Sohm, J.A.; Webb, E.A.; Capone, D.G. Emerging patterns of marine nitrogen fixation. Nature 2011, 9, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Villareal, T.A. Widespread occurrence of the Hemiaulus-cyanobacterial symbiosis in the Sourthwest North Atlantic Ocean. Bull. Mar. Sci. 1994, 54, 1–7. [Google Scholar]
- Subramaniam, A.; Yager, P.L.; Carpenter, E.J.; Mahaffey, C.; Björkman, K.; Kustka, A.B.; Montoya, J.P.; Sañudo-Wilhelmy, S.A.; Shipe, R.; Capone, D.G. Amazon river enhances diazotrophy and carbon sequestration in the tropical North Atlantic Ocean. Proc. Natl. Acad. Sci. USA 2008, 105, 10460–10465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villareal, T.A.; Brown, C.G.; Brzezinski, M.A.; Krause, J.W.; Wilson, C. Summer diatom blooms in the north Pacific subtropical gyre: 2008-2009. PLoS ONE 2012, 7, 2008–2009. [Google Scholar] [CrossRef] [PubMed]
- Shiozaki, T.; Kondo, Y.; Yuasa, D.; Takeda, S. Distribution of major diazotrophs in the surface water of the Kuroshio from northeastern Taiwan to south of mainland Japan. J. Plankton Res. 2018, 40, 407–419. [Google Scholar] [CrossRef]
- Monteiro, F.M.; Dutkiewicz, S.; Follows, M.J. Biogeographical controls on the marine nitrogen fixers. Global Biogeochem. Cycles 2011, 25, GB2003. [Google Scholar] [CrossRef] [Green Version]
- Monteiro, F.M.; Follows, M.J.; Dutkiewicz, S. Distribution of diverse nitrogen fixers in the global ocean. Global Biogeochem. Cycles 2010, 24, GB3017. [Google Scholar] [CrossRef] [Green Version]
- Stukel, M.R.; Coles, V.J.; Brooks, M.T.; Hood, R.R. Top-down, bottom-up and physical controls on diatom-diazotroph assemblage growth in the Amazon River plume. Biogeosciences 2014, 11, 3259–3278. [Google Scholar] [CrossRef] [Green Version]
- Staal, M.; Meysman, F.J.R.; Stal, L.J. Temperature excludes N2-fixing heterocystous cyanobacteria in the tropical oceans. Nature 2003, 425, 504–507. [Google Scholar] [CrossRef]
- Caputo, A.; Stenegren, M.; Pernice, M.C.; Foster, R.A. A short comparison of two marine planktonic diazotrophic symbioses highlights an un-quantified disparity. Front. Mar. Sci. 2018, 5, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Janson, S.; Rai, A.; B, B. Intracellular cyanobiont Richelia intracellularis: Ultrastructure and immunolocalization of phycoerythrin, nitrogenase, Rubisco and glutamine synthetase. Mar. Biol. 1995, 124, 1–8. [Google Scholar]
- Rai, A.N.; Söderbäck, E.; Bergman, B. Cyanobacterium-plant symbiosis. New Phytol. 2000, 147, 449–481. [Google Scholar] [CrossRef]
- Peters, G.A.; Meeks, J.C. The Azolla-Anabaena symbiosis: Basic biology. Annu. Rev. Plant. Physiol. Mol. Biol. 1989, 40, 193–210. [Google Scholar] [CrossRef]
- Rai, A.N.; Borthkur, M.; Singh, S.; Bergman, B. Anthoceros-Nostoc symbiosis: Immunoelectronmicroscopic localization of nitrogenase, glutamine synthetase, phycoerythrin and ribulose-1,5-bisphosphate carboxylase/oxygenase in the cyanobiont and the cultured (free-living) isolate Nostoc 7801. Microbiology 1989, 135, 385–395. [Google Scholar] [CrossRef] [Green Version]
- Rai, A.N. Handbook of Symbiotic Cyanobacteria; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Bergman, B.; Rai, A.N.; Johansson, C.; Söderbäck, E. Cyanobacterial-plant symbioses. Symbiosis 1992, 14, 61–81. [Google Scholar]
- Adams, D.G.; Bergman, B.; Nierzwicki-Bauer, S.A.; Rai, A.N.; Schüßler, A. Cyanobacterial-pldant symbioses. In Signalling and Communication in Plant Symbiosis; Dowrkin, S., Falkow, S., Rosenberg, E., Schleifer, K.H., Stackebrandt, E., Eds.; Springer: Berlin, Germany, 2006; Volume 1, pp. 331–363. [Google Scholar]
- Peters, G.A.; Kaplan, D.; Meeks, J.C.; Buzby, K.M.; Marsh, B.H.; Corbin, J.L. Aspects of nitrogen and carbon interchange in the Azolla-Anabaena symbiosis. In Nitrogen Fixation and CO2 Metabolism; Ludden, P.W., Burries, J.E., Eds.; Elsevier Science: New York, NY, USA, 1985; pp. 213–222. [Google Scholar]
- Kaplan, D.; Peters, G.A. Interaction of carbon metabolism in the Azolla-Anabaena symbiosis. Symbiosis 1988, 6, 53–68. [Google Scholar]
- Follett, C.L.; Dutkiewicz, S.; Karl, D.M.; Inomura, K.; Follows, M.J. Seasonal resource conditions favor a summertime increase in North Pacific diatom–diazotroph associations. ISME J. 2018, 12, 1543–1557. [Google Scholar] [CrossRef] [Green Version]
- Mulholland, M.R.; Bernhardt, P.W. The effect of growth rate, phosphorus concentration, and temperature on N2 fixation, carbon fixation, and nitrogen release in continuous cultures of Trichodesmium IMS101. Limnol. Oceanogr. 2005, 50, 839–849. [Google Scholar] [CrossRef]
- Hutchins, D.A.; Fu, F.-X.; Zhang, Y.; Warner, M.E.; Feng, Y.; Portune, K.; Bernhardt, P.W.; Mulholland, M.R. CO2 control of Trichodesmium N2 fixation, photosynthesis, growth rates, and elemental ratios: Implications for past, present, and future ocean biogeochemistry. Limnol. Oceanogr. 2007, 52, 1293–1304. [Google Scholar] [CrossRef] [Green Version]
- Fu, F.-X.; Mulholland, M.R.; Garcia, N.S.; Beck, A.; Bernhardt, P.W.; Warner, M.E.; Sañudo-Wilhelmy, S.A.; Hutchins, D.A. Interactions between changing pCO2, N2 fixation, and Fe limitation in the marine unicellular cyanobacterium Crocosphaera. Limnol. Oceanogr. 2008, 53, 2472–2484. [Google Scholar] [CrossRef] [Green Version]
- Großkopf, T.; LaRoche, J. Direct and indirect costs of dinitrogen fixation in Crocosphaera watsonii WH8501 and possible implications for the nitrogen cycle. Front. Microbiol. 2012, 3, 236. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sohm, J.A.; Edwards, B.R.; Wilson, B.G.; Webb, E.A. Constitutive extracellular polysaccharide (EPS) production by specific isolates of Crocosphaera watsonii. Front. Microbiol. 2011, 2, 229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inomura, K.; Bragg, J.; Follows, M.J. A quantitative analysis of the direct and indirect costs of nitrogen fixation: A model based on Azotobacter vinelandii. ISME J. 2017, 11, 166–175. [Google Scholar] [CrossRef] [PubMed]
- Inomura, K.; Bragg, J.; Riemann, L.; Follows, M.J. A quantitative model of nitrogen fixation in the presence of ammonium. PLoS ONE 2018, 13, e0208282. [Google Scholar] [CrossRef] [PubMed]
- Inomura, K.; Wilson, S.T.; Deutsch, C. Mechanistic model for the coexistence of nitrogen fixation and photosynthesis in marine Trichodesmium. mSystems 2019, 4, e00210-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inomura, K.; Deutsch, C.; Wilson, S.T.; Masuda, T.; Lawrenz, E.; Bučinská, L.; Sobotka, R.; Gauglitz, J.M.; Saito, M.A.; Prášil, O.; et al. Quantifying oxygen management and temperature and light dependencies of nitrogen fixation by Crocosphaera watsonii. mSphere 2019, 4, e00531-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inomura, K.; Masuda, T.; Gauglitz, J.M. Active nitrogen fixation by Crocosphaera expands their niche despite the presence of ammonium – A case study. Sci. Rep. 2019, 9, 15064. [Google Scholar] [CrossRef] [Green Version]
- Gallon, J.R. The oxygen sensitivity of nitrogenase: A problem for biochemists and micro-organisms. Trends Biochem. Sci. 1981, 6, 19–23. [Google Scholar] [CrossRef]
- Wang, Z.C.; Burns, A.; Watt, G.D. Complex formation and O2 sensitivity of Azotobacter vinelandii nitrogenase and its component proteins. Biochemistry 1985, 24, 214–221. [Google Scholar] [CrossRef]
- Gallon, J.R. Reconciling the incompatible: N2 fixation and O2. New Phytol. 1992, 122, 571–609. [Google Scholar] [CrossRef]
- Lang, N.J.; Fay, P. The heterocysts of blue-green algae II. Details of ultrastructure. Proc. R. Soc. B Biol. Sci. 1971, 178, 193–203. [Google Scholar]
- Walsby, A.E. The permeability of heterocysts to the gases nitrogen and oxygen. Proc. R. Soc. B Biol. Sci. 1985, 226, 345–366. [Google Scholar]
- Menden-deuer, S.; Lessard, E.J. Carbon to volume relationships for dinoflagellates, diatoms, and other protist plankton. Limnol. Oceanogr. 2000, 45, 569–579. [Google Scholar] [CrossRef] [Green Version]
- Redfield, A.C. The biological control of chemical factors in the environment. Am. Sci. 1958, 46, 205–221. [Google Scholar]
- Foster, R.A.; O’Mullan, G.D. Nitrogen-fixing and nitrifying symbioses in the marine environment. In Nitrogen in the Marine Environment; Capone, D.G., Bronk, D.A., Mulholland, M.R., Carpenter, E.J., Eds.; Academic Press: London, UK, 2008. [Google Scholar]
- Villareal, T.A. Nitrogen-fixation by the cyanobacterial symbiont of the diatom genus Hemiaulus. Mar. Ecol. Prog. Ser. 1991, 76, 201–204. [Google Scholar] [CrossRef]
- Harke, M.J.; Frischkorn, K.R.; Haley, S.T.; Aylward, F.O.; Zehr, J.P.; Dyhrman, S.T. Periodic and coordinated gene expression between a diazotroph and its diatom host. ISME J. 2019, 13, 118–131. [Google Scholar] [CrossRef]
- Foster, R.A.; Zehr, J.P. Diversity, genomics, and distribution of phytoplankton-cyanobacterium single-cell symbiotic associations. Annu. Rev. Microbiol. 2019, 73, 435–456. [Google Scholar] [CrossRef]
- Steinberg, N.A.; Meeks, J.C. Physiological sources of reductant for nitrogen fixation activity in Nostoc sp. strain UCD 7801 in symbiotic association with Anthoceros punctatus. J. Bacteriol. 1991, 173, 7324–7329. [Google Scholar] [CrossRef] [Green Version]
- Holl, C.M.; Montoya, J.P. Diazotrophic growth of the marine cyanobacterium Trichodesmium IMS101 in continuous culture: Effects of growth rate on N2-fixation rate, biomass, and C:N:P stoichiometry. J. Phycol. 2008, 44, 929–937. [Google Scholar] [CrossRef]
- Tuit, C.; Waterbury, J.; Ravizza, G. Diel variation of molybdenum and iron in marine diazotrophic cyanobacteria. Limnol. Oceanogr. 2004, 49, 978–990. [Google Scholar] [CrossRef]
- Masuda, T.; Furuya, K.; Kodama, T.; Takeda, S.; Harrison, P.J. Ammonium uptake and dinitrogen fixation by the unicellular nanocyanobacterium Crocosphaera watsonii in nitrogen-limited continuous cultures. Limnol. Oceanogr. 2013, 58, 2029–2036. [Google Scholar] [CrossRef]
- Lichtl, R.R.; Bazin, M.J.; Hall, D.O. The biotechnology of hydrogen production by Nostoc flagelliforme grown under chemostat conditions. Appl. Microbiol. Biotechnol. 1997, 47, 701–707. [Google Scholar] [CrossRef]
- Fay, P. Oxygen relations of nitrogen fixation in cyanobacteria. Microbiol. Rev. 1992, 56, 340–373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donze, M.; Haveman, J.; Schiereck, P. Absence of Photosystem 2 in heterocysts of the blue-green alga Anabaena. BBA Bioenerg. 1972, 256, 157–161. [Google Scholar] [CrossRef]
- Foster, R.A.; Goebel, N.L.; Zehr, J.P. Isolation of Calothrix rhizosoleniae (Cyanobacteria) strain SC01 from Chaetoceros (Bacillariophyta) spp. diatoms of the subtropical North Pacific Ocean. J. Phycol. 2010, 46, 1028–1037. [Google Scholar] [CrossRef]
- Letscher, R.T.; Villareal, T.A. Evaluation of the seasonal formation of subsurface negative preformed nitrate anomalies in the subtropical North Pacific and North Atlantic. Biogeosciences 2018, 15, 6461–6480. [Google Scholar] [CrossRef] [Green Version]
- Ostenfeld, C.H.; Schmidt, J. Plankton fra det Røde hav og Adenbugten, Vidensk. Meddel. Naturh. Forening i Kbhvn. In Marine Pelagic Cyanobacteria: Trichodesmium and other Diazotrophs; Carpenter, E.J., Capone, D.G., Eds.; Springer Science & Business Media: Berlin, German, 1901; pp. 141–182. [Google Scholar]
- Taylor, F.J.R. Symbioses in marine microplankton. Ann. Inst. Océanogr. 1982, 58, 61–90. [Google Scholar]
- Sundström, B.G. Observations on Rhizosolenia clevei Ostenfeld (Bacillariophyceae) and Richelia intracellularis Schmidt (Cyanophyceae). Bot. Mar. 1984, 27, 345–355. [Google Scholar] [CrossRef]
- Scott, M.; Gunderson, C.W.; Mateescu, E.M.; Zhang, Z.; Hwa, T. Interdependence of cell growth and gene expression: Origins and consequences. Science 2010, 330, 1099–1103. [Google Scholar] [CrossRef]
- You, C.; Okano, H.; Hui, S.; Zhang, Z.; Kim, M.; Gunderson, C.W.; Wang, Y.-P.; Lenz, P.; Yan, D.; Hwa, T. Coordination of bacterial proteome with metabolism by cyclic AMP signalling. Nature 2013, 500, 301–306. [Google Scholar] [CrossRef] [Green Version]
- Jahn, M.; Vialas, V.; Karlsen, J.; Maddalo, G.; Edfors, F.; Forsström, B.; Uhlén, M.; Käll, L.; Hudson, E.P. Growth of cyanobacteria is constrained by the abundance of light and carbon assimilation proteins. Cell Rep. 2018, 25, 478–486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zavřel, T.; Faizi, M.; Loureiro, C.; Poschmann, G.; Stühler, K.; Sinetova, M.; Zorina, A.; Steuer, R.; Červený, J. Quantitative insights into the cyanobacterial cell economy. Elife 2019, 8. [Google Scholar] [CrossRef] [PubMed]
- Ellis, R.J. Macromolecular crowding: Obvious but underappreciated. Trends Biochem. Sci. 2001, 26, 597–604. [Google Scholar] [CrossRef]
- Burnap, R.L. Systems and photosystems: Cellular limits of autotrophic productivity in cyanobacteria. Front. Bioeng. Biotechnol. 2015, 3, 1. [Google Scholar] [CrossRef]
- Lemmermann, E. Die Algenflora der Sandwich-Inseln. Ergebnisse einer Reise nach dem Pacific, H. Schauinsland 1896/97. Engler’s Bot. Jb. 1905, 34, 607–663. [Google Scholar]
- Yoon, H.S.; Golden, J.W. Heterocyst pattern formation controlled by a diffusible peptide. Science 1998, 282, 935–938. [Google Scholar] [CrossRef]
- Khudyakov, I.Y.; Golden, J.W. Different functions of HetR, a master regulator of heterocyst differentiation in Anabaena sp. PCC 7120, can be separated by mutation. Proc. Natl. Acad. Sci. USA 2004, 101, 16040–16045. [Google Scholar] [CrossRef] [Green Version]
- Yoon, H.S.; Golden, J.W. PatS and products of nitrogen fixation control heterocyst pattern. J. Bacteriol. 2001, 183, 2605–2613. [Google Scholar] [CrossRef] [Green Version]
- Risser, D.D.; Callahan, S.M. Genetic and cytological evidence that heterocyst patterning is regulated by inhibitor gradients that promote activator decay. Proc. Natl. Acad. Sci. USA 2009, 106, 19884–19888. [Google Scholar] [CrossRef] [Green Version]
- Borthakur, P.B.; Orozco, C.C.; Young-Robbins, S.S.; Haselkorn, R.; Callahan, S.M. Inactivation of patS and hetN causes lethal levels of heterocyst differentiation in the filamentous cyanobacterium Anabaena sp. PCC 7120. Mol. Microbiol. 2005, 57, 111–123. [Google Scholar] [CrossRef]
- Corrales-Guerrero, L.; Mariscal, V.; Nu¨rnberg, D.J.; Elhai, J.; Mullineaux, C.W.; Flores, E.; Herrero, A. Subcellular localization and clues for the function of the HetN factor influencing heterocyst distribution in Anabaena sp. strain PCC 7120. J. Bacteriol. 2014, 196, 3452–3460. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Muñoz-García, J.; Ares, S. Formation and maintenance of nitrogen-fixing cell patterns in filamentous cyanobacteria. Proc. Natl. Acad. Sci. USA 2016, 113, 6218–6223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berry, D.; Mader, E.; Lee, T.K.; Woebken, D.; Wang, Y.; Zhu, D.; Palatinszky, M.; Schintlmeister, A.; Schmid, M.C.; Hanson, B.T.; et al. Tracking heavy water (D2O) incorporation for identifying and sorting active microbial cells. Proc. Natl. Acad. Sci. USA 2015, 112, E194–E203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, C.; Fong, L.G.; Young, S.G.; Jiang, H. NanoSIMS imaging: An approach for visualizing and quantifying lipids in cells and tissues. J. Investig. Med. 2017, 65, 669–672. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.S.; Palatinszky, M.; Pereira, F.C.; Nguyen, J.; Fernandez, V.I.; Mueller, A.J.; Menolascina, F.; Daims, H.; Berry, D.; Wagner, M.; et al. An automated Raman-based platform for the sorting of live cells by functional properties. Nat. Microbiol. 2019, 4, 1035–1048. [Google Scholar] [CrossRef] [Green Version]
- Popa, R.; Weber, P.K.; Pett-Ridge, J.; Finzi, J.A.; Fallon, S.J.; Hutcheon, I.D.; Nealson, K.H.; Capone, D.G. Carbon and nitrogen fixation and metabolite exchange in and between individual cells of Anabaena oscillarioides. ISME J. 2007, 1, 354–360. [Google Scholar] [CrossRef]
- Sharkey, T.D.; Berry, J.A. Carbon isotope fractionation of algae influenced by an inducible CO2 concentrating mechanism. In Inorganic Carbon uptake by Aquatic Photosynthetic Organisms; Lucas, W.J., Berry, J.A., Eds.; American Society of Plant Physiologists: Rockville, MD, USA, 1985; pp. 389–401. [Google Scholar]
- Keller, K.; Morel, F.M.M. A model of carbon isotopic fractionation and active carbon uptake in phytoplankton. Mar. Ecol. Prog. Ser. 1999, 182, 295–298. [Google Scholar] [CrossRef] [Green Version]
- Rittmann, B.E.; McCarty, P.L. Environmental Biotechnology: Principles and Applications; McGraw-Hill: New York, NY, USA, 2001; pp. 126–164. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inomura, K.; Follett, C.L.; Masuda, T.; Eichner, M.; Prášil, O.; Deutsch, C. Carbon Transfer from the Host Diatom Enables Fast Growth and High Rate of N2 Fixation by Symbiotic Heterocystous Cyanobacteria. Plants 2020, 9, 192. https://doi.org/10.3390/plants9020192
Inomura K, Follett CL, Masuda T, Eichner M, Prášil O, Deutsch C. Carbon Transfer from the Host Diatom Enables Fast Growth and High Rate of N2 Fixation by Symbiotic Heterocystous Cyanobacteria. Plants. 2020; 9(2):192. https://doi.org/10.3390/plants9020192
Chicago/Turabian StyleInomura, Keisuke, Christopher L. Follett, Takako Masuda, Meri Eichner, Ondřej Prášil, and Curtis Deutsch. 2020. "Carbon Transfer from the Host Diatom Enables Fast Growth and High Rate of N2 Fixation by Symbiotic Heterocystous Cyanobacteria" Plants 9, no. 2: 192. https://doi.org/10.3390/plants9020192
APA StyleInomura, K., Follett, C. L., Masuda, T., Eichner, M., Prášil, O., & Deutsch, C. (2020). Carbon Transfer from the Host Diatom Enables Fast Growth and High Rate of N2 Fixation by Symbiotic Heterocystous Cyanobacteria. Plants, 9(2), 192. https://doi.org/10.3390/plants9020192




