Crocus sativus L. Extract Containing Polyphenols Modulates Oxidative Stress and Inflammatory Response against Anti-Tuberculosis Drugs-Induced Liver Injury
Abstract
1. Introduction
2. Results
2.1. Chemical Profiling of CSP by LC–ESI-Q-TOF–MS
2.2. Estimation of TPC and TFC
2.3. In Vitro Antioxidant Activity
2.4. Elemental Analysis of the CS Petals Using ICP OES
2.5. Biochemical Analysis
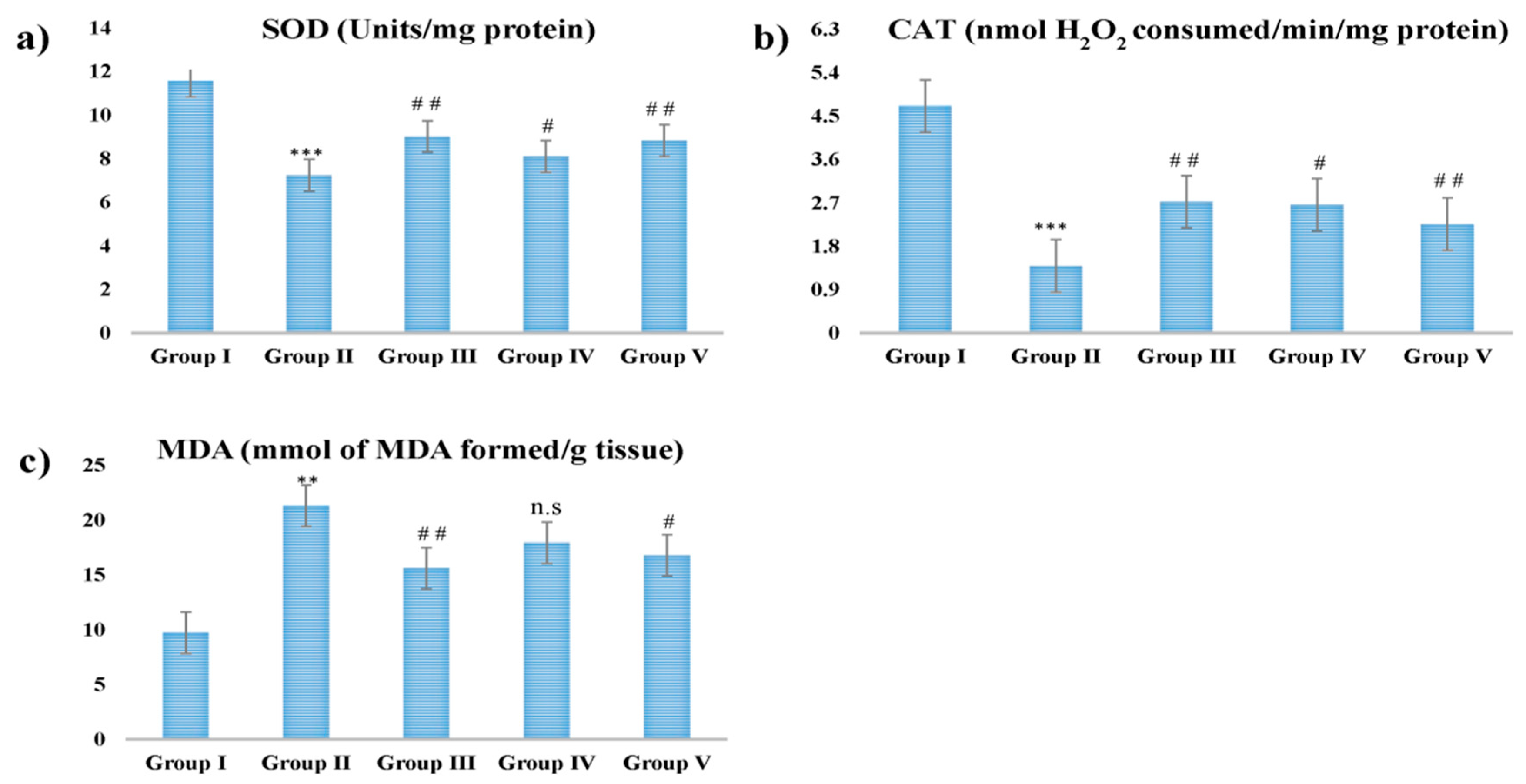
2.6. Estimation of CAT, SOD, and MDA
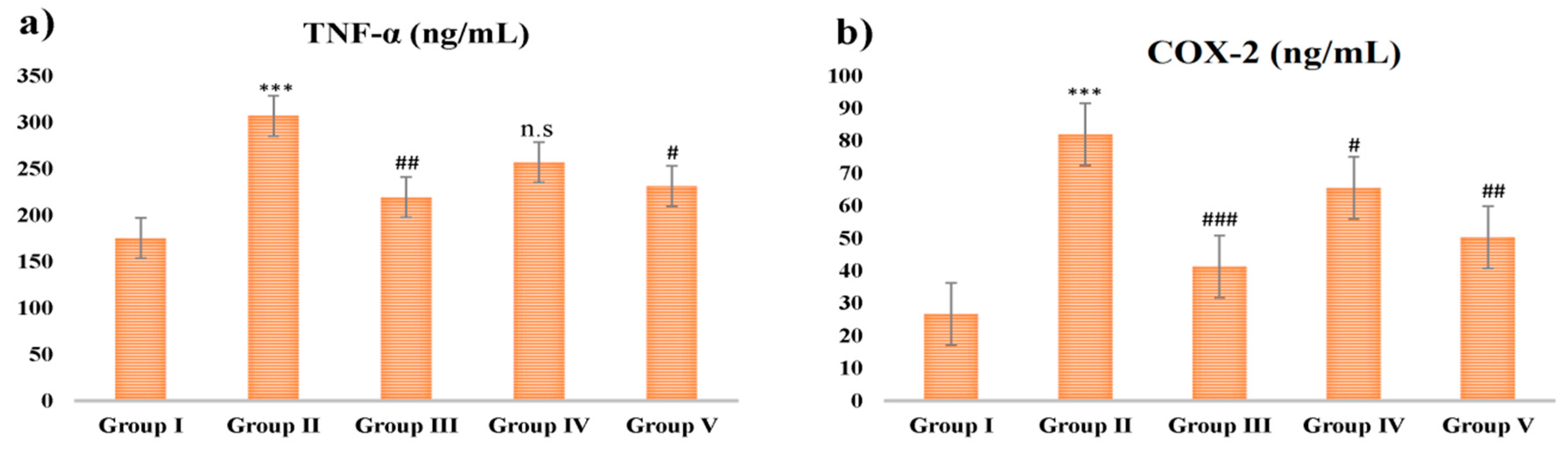
2.7. Proinflammatory Cytokine Analysis
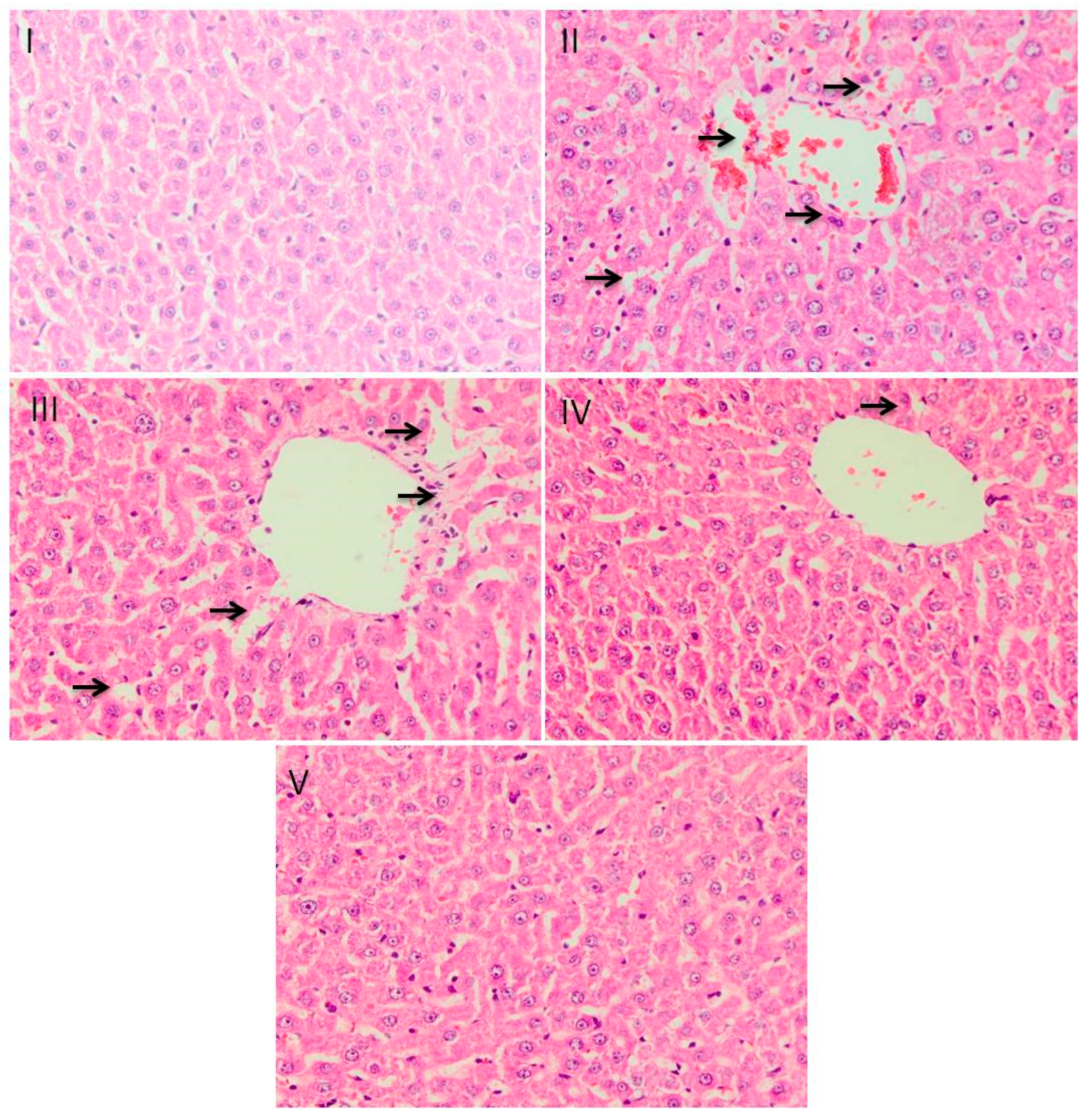
2.8. Histological Evaluation
3. Discussion
4. Materials and Methods
4.1. Plant Material and Extraction
4.2. Chemical Profiling of CSP by LC–ESI-Q-TOF–MS
4.3. Estimation of Total Phenolic Content (TPC)
4.4. Estimation Total Flavonoid Content (TFC)
4.5. In-Vitro Antioxidant Activity
4.6. Elemental Analysis of the CS Petals Using ICP OES
4.7. Induction of Hepatotoxicity in Wistar Rats
4.8. Biochemical Analysis
4.9. Estimation of Catalase (CAT), Superoxide Dismutase (SOD) and Malondialdehyde (MDA)
4.10. Proinflammatory Cytokine Analysis
4.11. Histological Evaluation
4.12. Statistical Analysis
4.13. Statement of Ethical Approval
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) |
| ALP | Alkaline phosphatase |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| ATC | Antituberculosis |
| CAT | Catalase |
| COX-2 | Cyclooxygenase |
| CS | Crocus sativus L. |
| CSP | Crocus sativus petals |
| DIH | Drug-induced hepatotoxicity |
| DPPH | 2,2-diphenyl-1-picry1hydrazy1 |
| EMB | Ethambutol |
| GAE/g | Gallic acid equivalent per gram |
| IC50 | Half maximal inhibitory concentration |
| ICP-OES | Inductively coupled plasma—optical emission spectrometry |
| INH | Isoniazid |
| LC-ESI-Q-TOFMS | Liquid chromatography coupled with electrospray ionization-quadrupole-time of flight-mass spectrometry |
| MDA | Malondialdehyde |
| PZA | Pymzinaide |
| QE/g | Quercetin equivalent per gram |
| R2 | Regression |
| RIF | Rifampicin |
| SOD | Superoxide dismutase |
| TB | Tuberculosis |
| TFC | Total flavonoid content |
| TNF-α | Tumor necrosis factor alpha |
| TP | Total protein |
| TPC | Total phenolic content |
| WHO | World Health Organization |
References
- Furin, J.; Cox, H.; Pai, M. Tuberculosis. Lancet 2019, 393, 1642–1656. [Google Scholar] [CrossRef]
- Gualano, G.; Capone, S.; Matteelli, A.; Palmieri, F. New antituberculosis drugs: From clinical trial to programmatic use. Infect. Dis. Rep. 2016, 8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y. The magic bullets and tuberculosis drug targets. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 529–564. [Google Scholar] [CrossRef] [PubMed]
- Hussain, T.; Gupta, R.K.; Sweety, K.; Khan, M.S.; Hussain, M.S.; Arif, M.; Hussain, A.; Faiyazuddin, M.; Rao, C.V. Evaluation of antihepatotoxic potential of Solanum xanthocarpum fruit extract against antitubercular drugs induced hepatopathy in experimental rodents. Asian Pac. J. Trop. Biomed. 2012, 2, 454–460. [Google Scholar] [CrossRef]
- Hong, M.; Li, S.; Tan, H.; Wang, N.; Tsao, S.-W.; Feng, Y. Current Status of Herbal Medicines in Chronic Liver Disease Therapy: The Biological Effects, Molecular Targets and Future Prospects. Int. J. Mol. Sci. 2015, 16, 28705–28745. [Google Scholar] [CrossRef]
- Sikander, M.; Malik, S.; Parveen, K.; Ahmad, M.; Yadav, D.; Hafeez, Z.B.; Bansal, M. Hepatoprotective effect of Origanum vulgare in Wistar rats against carbon tetrachloride-induced hepatotoxicity. Protoplasma 2012, 250, 483–493. [Google Scholar] [CrossRef]
- Rehman, M.U.; Wali, A.F.; Ahmad, A.; Shakeel, S.; Rasool, S.; Ali, R.; Rashid, S.M.; Madkhali, H.; Ganaie, M.A.; Khan, R. Neuroprotective Strategies for Neurological Disorders by Natural Products: An update. Curr. Neuropharmacol. 2019, 17, 247–267. [Google Scholar] [CrossRef]
- Moossavi, M.; Hoshyar, R.; Hemmati, M.; Farahi, A.; Javdani, H. An invivo study on the hepato-protective effects of Crocus sativus, Ziziphus jujuba and Berberis vulgaris against acute acetaminophen and rifampicin-induced hepatotoxicity. Clin. Phytoscience 2016, 2. [Google Scholar] [CrossRef]
- Alavizadeh, S.H.; Hosseinzadeh, H. Bioactivity assessment and toxicity of crocin: A comprehensive review. Food Chem. Toxicol. 2014, 64, 65–80. [Google Scholar] [CrossRef]
- Imenshahidi, M.; Hosseinzadeh, H.; Javadpour, Y. Hypotensive effect of aqueous saffron extract (Crocus sativus L.) and its constituents, safranal and crocin, in normotensive and hypertensive rats. Phytother. Res. 2009. [Google Scholar] [CrossRef]
- Nassiri-Asl, M.; Hosseinzadeh, H. Neuropharmacology Effects of Saffron (Crocus sativus) and Its Active Constituents. In Bioactive Nutraceuticals and Dietary Supplements in Neurological and Brain Disease; Elsevier: Amsterdam, The Netherlands, 2015; pp. 29–39. [Google Scholar] [CrossRef]
- Mollazadeh, H.; Emami, S.A.; Hosseinzadeh, H. Razi’s Al-Hawi and saffron (Crocus sativus): A review. Iran. J. Basic Med. Sci. 2015, 18, 1153. [Google Scholar] [PubMed]
- Srivastava, R.; Ahmed, H.; Dixit, R.K.; Saraf, S.A. Crocus sativus L.: A comprehensive review. Pharmacogn. Rev. 2010, 4, 200. [Google Scholar] [CrossRef] [PubMed]
- Rahaiee, S.; Moini, S.; Hashemi, M.; Shojaosadati, S.A. Evaluation of antioxidant activities of bioactive compounds and various extracts obtained from saffron (Crocus sativus L.): A review. J. Food Sci. Technol. 2014, 52, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Yousefsani, B.S.; Pourahmad, J.; Hosseinzadeh, H. The mechanism of protective effect of crocin against liver mitochondrial toxicity caused by arsenic III. Toxicol. Mech. Methods 2017, 28, 105–114. [Google Scholar] [CrossRef]
- Zeinali, M.; Zirak, M.R.; Rezaee, S.A.; Karimi, G.; Hosseinzadeh, H. Immunoregulatory and anti-inflammatory properties of Crocus sativus (Saffron) and its main active constituents: A review. Iran. J. Basic Med. Sci. 2019, 22, 334. [Google Scholar]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z.E. Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules 2010, 15, 6244–6256. [Google Scholar] [CrossRef]
- Bathaie, S.Z.; Mousavi, S.Z. New Applications and Mechanisms of Action of Saffron and its Important Ingredients. Crit. Rev. Food Sci. Nutr. 2010, 50, 761–786. [Google Scholar] [CrossRef]
- Khan, N.; Syed, D.N.; Ahmad, N.; Mukhtar, H. Fisetin: A Dietary Antioxidant for Health Promotion. Antioxid. Redox Signal. 2013, 19, 151–162. [Google Scholar] [CrossRef]
- Hosseini, A.; Razavi, B.M.; Hosseinzadeh, H. Saffron (Crocus sativus) petal as a new pharmacological target: A review. Iran. J. Basic Med. Sci. 2018, 21, 1091. [Google Scholar]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 1–16. [Google Scholar] [CrossRef]
- Nerdy, N.; Ritarwan, K. Hepatoprotective Activity and Nephroprotective Activity of Peel Extract from Three Varieties of the Passion Fruit (Passiflora Sp.) in the Albino Rat. Open Access Maced. J. Med. Sci. 2019, 7, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Rehman, M.U.; Ali, N.; Rashid, S.; Jain, T.; Nafees, S.; Tahir, M.; Khan, A.Q.; Lateef, A.; Khan, R.; Hamiza, O.O.; et al. Alleviation of hepatic injury by chrysin in cisplatin administered rats: Probable role of oxidative and inflammatory markers. Pharmacol. Rep. 2014, 66, 1050–1059. [Google Scholar] [CrossRef] [PubMed]
- Jeon, S.H.; Kuppusamy, S.; Yoon, Y.-E.; Kim, H.T.; Lee, Y.B. Are There as Many Essential and Non-essential Minerals in Hydroponic Strawberry (Fragaria ananassa L.) Compared to Those Grown in Soil? Biol. Trace Elem. Res. 2018, 187, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Wali, A.F.; Avula, B.; Ali, Z.; Khan, I.A.; Mushtaq, A.; Rehman, M.U.; Akbar, S.; Masoodi, M.H. Antioxidant, Hepatoprotective Potential and Chemical Profiling of Propolis Ethanolic Extract from Kashmir Himalaya Region Using UHPLC-DAD-QToF-MS. BioMed. Res. Int. 2015, 2015, 1–10. [Google Scholar] [CrossRef]
- Ranawat, L.; Bhatt, J.; Patel, J. Hepatoprotective activity of ethanolic extracts of bark of Zanthoxylum armatum DC in CCl4 induced hepatic damage in rats. J. Ethnopharmacol. 2010, 127, 777–780. [Google Scholar] [CrossRef]
- Soetan, K.; Olaiya, C.; Oyewole, O. The importance of mineral elements for humans, domestic animals and plants-A review. Afr. J. Food Sci. 2010, 4, 200–222. [Google Scholar]
- Huang, D.; Ou, B.; Prior, R.L. The Chemistry behind Antioxidant Capacity Assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Mujahid, M.; Siddiqui, H.H.; Hussain, A.; Hussain, M.S. Hepatoprotective effects of Adenanthera pavonina (Linn.) against anti-tubercular drugs-induced hepatotoxicity in rats. Pharmacogn. J. 2013, 5, 286–290. [Google Scholar] [CrossRef]
- Santhosh, S.; Sini, T.K.; Anandan, R.; Mathew, P.T. Effect of chitosan supplementation on antitubercular drugs-induced hepatotoxicity in rats. Toxicology 2006, 219, 53–59. [Google Scholar] [CrossRef]
- Lynch, T.; Price, A. The effect of cytochrome P450 metabolism on drug response, interactions, and adverse effects. Am. Fam. Physician 2007, 76, 391–396. [Google Scholar]
- Metushi, I.; Uetrecht, J.; Phillips, E. Mechanism of isoniazid-induced hepatotoxicity: Then and now. Br. J. Clin. Pharmacol. 2016, 81, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Tasduq, S.A.; Peerzada, K.; Koul, S.; Bhat, R.; Johri, R.K. Biochemical manifestations of anti-tuberculosis drugs induced hepatotoxicity and the effect of silymarin. Hepatol. Res. 2005, 31, 132–135. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Pradhan, K.; Zhong, X.-B.; Ma, X. Isoniazid metabolism and hepatotoxicity. Acta Pharm. Sin. B 2016, 6, 384–392. [Google Scholar] [CrossRef] [PubMed]
- Pauli-Magnus, C.; Meier, P.J. Hepatobiliary transporters and drug-induced cholestasis. Hepatology 2006, 44, 778–787. [Google Scholar] [CrossRef] [PubMed]
- Omidi, A.; Riahinia, N.; Torbati, M.B.M.; Behdani, M.-A. Hepatoprotective effect of Crocus sativus (saffron) petals extract against acetaminophen toxicity in male Wistar rats. Avicenna J. Phytomedicine 2014, 4, 330. [Google Scholar]
- Miltonprabu, S.; Tomczyk, M.; Skalicka-Woźniak, K.; Rastrelli, L.; Daglia, M.; Nabavi, S.F.; Alavian, S.M.; Nabavi, S.M. Hepatoprotective effect of quercetin: From chemistry to medicine. Food Chem. Toxicol. 2017, 108, 365–374. [Google Scholar] [CrossRef]
- Rizvi, F.; Mathur, A.; Kakkar, P. Morin mitigates acetaminophen-induced liver injury by potentiating Nrf2 regulated survival mechanism through molecular intervention in PHLPP2-Akt-Gsk3β axis. Apoptosis 2015, 20, 1296–1306. [Google Scholar] [CrossRef]
- Bansal, A.K.; Bansal, M.; Soni, G.; Bhatnagar, D. Protective role of Vitamin E pre-treatment on N-nitrosodiethylamine induced oxidative stress in rat liver. Chem. -Biol. Interact. 2005, 156, 101–111. [Google Scholar] [CrossRef]
- Kurutas, E.B. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress: Current state. Nutr. J. 2015, 15. [Google Scholar] [CrossRef]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. IJBS 2008, 4, 89. [Google Scholar]
- Tao, L.; Qu, X.; Zhang, Y.; Song, Y.; Zhang, S.-X. Prophylactic Therapy of Silymarin (Milk Thistle) on Antituberculosis Drug-Induced Liver Injury: A Meta-Analysis of Randomized Controlled Trials. Can. J. Gastroenterol. Hepatol. 2019, 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Zhu, R.; Wang, Y.; Zhang, L.; Guo, Q. Oxidative stress and liver disease. Hepatol. Res. 2012, 42, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Lykkesfeldt, J. Malondialdehyde as biomarker of oxidative damage to lipids caused by smoking. Clin. Chim. Acta 2007, 380, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Muriel, P. Role of free radicals in liver diseases. Hepatol. Int. 2009, 3, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Slater, T.F. Free-radical mechanisms in tissue injury. Biochem. J. 1984, 222, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kandimalla, R.; Kalita, S.; Choudhury, B.; Devi, D.; Kalita, D.; Kalita, K.; Dash, S.; Kotoky, J. Fiber from ramie plant (Boehmeria nivea): A novel suture biomaterial. Mater. Sci. Eng. C 2016, 62, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Luckey, S.W.; Petersen, D.R. Activation of Kupffer Cells During the Course of Carbon Tetrachloride-Induced Liver Injury and Fibrosis in Rats. Exp. Mol. Pathol. 2001, 71, 226–240. [Google Scholar] [CrossRef] [PubMed]
- Nguyen-Lefebvre, A.T.; Horuzsko, A. Kupffer cell metabolism and function. J. Enzymol. Metab. 2015, 1, 1. [Google Scholar]
- Sun, X.-F.; Zhang, H. NFKB and NFKBI polymorphisms in relation to susceptibility of tumour and other diseases. Histol. Histopathol. 2007, 12, 1387–1398. [Google Scholar]
- Zhang, H.Y.; Wang, H.L.; Zhong, G.Y.; Zhu, J.X. Molecular mechanism and research progress on pharmacology of traditional Chinese medicine in liver injury. Pharm. Biol. 2018, 56, 594–611. [Google Scholar] [CrossRef]
- Babaei, A.; Arshami, J.; Haghparast, A.; Mesgaran, M.D. Effects of saffron (Crocus sativus) petal ethanolic extract on hematology, antibody response, and spleen histology in rats. Avicenna J. Phytomedicine 2014, 4, 103. [Google Scholar]
- Nazima, B.; Manoharan, V.; Miltonprabu, S. Grape seed proanthocyanidins ameliorates cadmium-induced renal injury and oxidative stress in experimental rats through the up-regulation of nuclear related factor 2 and antioxidant responsive elements. Biochem. Cell Biol. 2015, 93, 210–226. [Google Scholar] [CrossRef] [PubMed]
- Domitrović, R.; Jakovac, H.; Vasiljev Marchesi, V.; Vladimir-Knežević, S.; Cvijanović, O.; Tadić, Ž.; Romić, Ž.; Rahelić, D. Differential hepatoprotective mechanisms of rutin and quercetin in CCl4-intoxicated BALB/cN mice. Acta Pharmacol. Sin. 2012, 33, 1260–1270. [Google Scholar] [CrossRef] [PubMed]
- Ayoub, N.; Nematallah, K.A.; Al-Gendy, A.A.; Zaghloul, S.S. Novel quercetin glycoside with promising hepatoprotective activity isolated from Lobularia libyca (viv). CfW (Brassicaceae). Eur. Sci. J. 2013, 9, 21. [Google Scholar]
- Chang, C.-C.; Yang, M.-H.; Wen, H.-M.; Chern, J.-C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 3. [Google Scholar]
- Ullah, F.; Ayaz, M.; Sadiq, A.; Hussain, A.; Ahmad, S.; Imran, M.; Zeb, A. Phenolic, flavonoid contents, anticholinesterase and antioxidant evaluation of Iris germanica var;florentina. Nat. Prod. Res. 2015, 30, 1440–1444. [Google Scholar] [CrossRef]
- Claiborne, A. Catalase activity. In CRC Handbook of Methods for Oxygen Radical Research; CRC Press: Boca Raton, FL, USA, 1985; pp. 283–284. [Google Scholar]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef]
- D’souza, D.; Babu, G.; Shetty, S.; Balan, P. Estimation of serum malondialdehyde in potentially malignant disorders and post-antioxidant treated patients: A biochemical study. Contemp. Clin. Dent. 2012, 3, 448. [Google Scholar] [CrossRef]
- Tahir, M.; Rehman, M.U.; Lateef, A.; Khan, R.; Khan, A.Q.; Qamar, W.; Ali, F.; O’Hamiza, O.; Sultana, S. Diosmin protects against ethanol-induced hepatic injury via alleviation of inflammation and regulation of TNF-α and NF-κB activation. Alcohol 2013, 47, 131–139. [Google Scholar] [CrossRef]
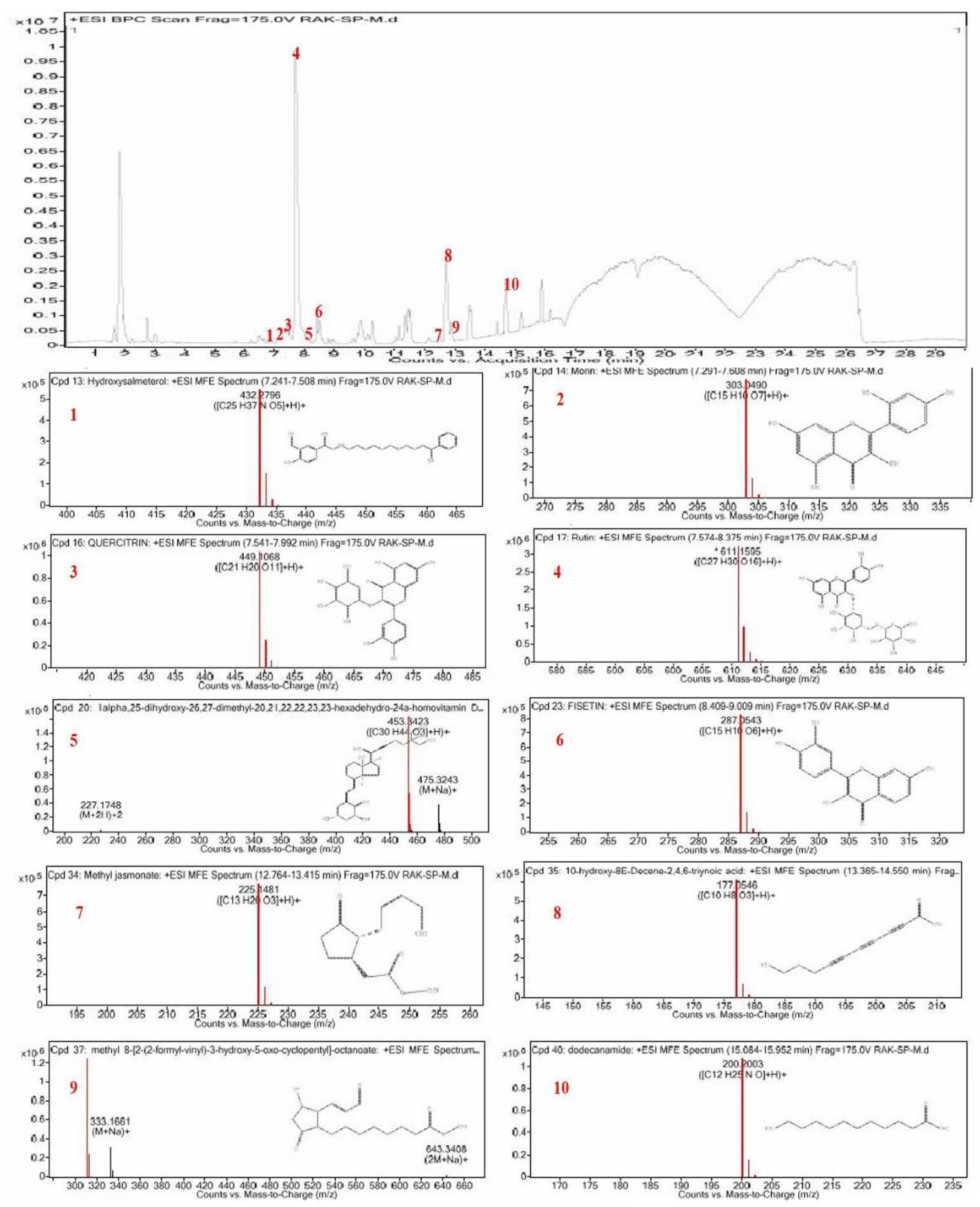
| Peak No. | Retention Time (min) | Molecular Formula | Theoretical (m/z) | Measured (m/z) | Compound Name |
|---|---|---|---|---|---|
| 1 | 7.241 | C25H37NO5 | 431.2722 | 432.2796 | Hydroxysalmeterol |
| 2 | 7.417 | C15H10O7 | 302.0417 | 303.0490 | Morin |
| 3 | 7.704 | C21H20O11 | 448.0995 | 449.1068 | Quercetin |
| 4 | 7.903 | C27H30O16 | 610.1523 | 611.1595 | Rutin |
| 5 | 8.107 | C30H44O3 | 452.335 | 454.3459 | 1alpha,25-dihydroxy-26,27-dimethyl-20,21,22,22,23,23-hexadehydro-24ahomovitaminD3 |
| 6 | 8.499 | C15H10O6 | 286.047 | 287.0543 | Fisetin |
| 7 | 12.887 | C13H20O3 | 224.1408 | 225.1481 | Methyl jasmonate |
| 8 | 13.477 | C10H8O3 | 176.0473 | 177.0546 | 10-Hydroxy-8E-Decene-2,4,6-triynoic acid |
| 9 | 13.526 | C17H26O5 | 310.1768 | 311.1841 | Methyl 8-[2-(2-formyl-vinyl)-3-hydroxy-5-oxo-cyclopentyl]-octanoate |
| 10 | 15.085 | C12H25NO | 200.2003 | 199.193 | Odecanamide |
| Methods | CSP | R2 |
|---|---|---|
| Total phenolic content (mg GAEa/g of extract) | 89.63 ± 0.99 | 0.992 |
| Total flavonoids content (μg QEb/g of extract) | 65 ± 1.09 | 0.989 |
| CSP IC50 (μg/mL) | R2 | BHA IC50 (μg/mL) | R2 | |
|---|---|---|---|---|
| DPPH Assay | 99.53 ± 0.63 | 0.992 | 65.46 ± 1.22 | 0.901 |
| ABTS Assay | 116.63 ± 1.93 | 0.990 | 87.42 ± 0.990 | 0.989 |
| S. No | Name of the Element | Concentration (mg/Kg) |
|---|---|---|
| 1. | Silver | 0.2 |
| 2. | Aluminum | 555 |
| 3. | Arsenic | 0.2 |
| 4. | Boron | 17.2 |
| 5. | Barium | 3.9 |
| 6. | Beryllium | <0.1 |
| 7. | Calcium | 4584 |
| 8. | Cadmium | <0.1 |
| 9. | Cobalt | 0.2 |
| 10. | Chromium | 1.6 |
| 11. | Copper | 9.6 |
| 12. | Iron | 1192 |
| 13. | Potassium | 22,256 |
| 14. | Magnesium | 1751 |
| 15. | Manganese | 33.9 |
| 16. | Molybdenum | 0.6 |
| 17. | Sodium | 211 |
| 18. | Nickel | 3.4 |
| 19. | Phosphorous | 3569 |
| 20. | Lead | 0.7 |
| 21. | Tin | 0.1 |
| 22. | Selenium | 0.1 |
| 23. | Strontium | 9.1 |
| 24. | Vanadium | 1.2 |
| 25. | Zinc | 60.2 |
| Groups | ALT (IUL−1) | AST (IUL−1) | ALP (IUL−1) | TP (g/dL) |
|---|---|---|---|---|
| Group I | 62.23 ± 5.32 | 87.21 ± 5.23 | 100.43 ± 5.98 | 9.34 ± 0.21 |
| Group II | 143.90 ± 3.54 *** | 302.43 ± 4.32 *** | 278.21 ± 3.65 *** | 3.18 ± 0.53 *** |
| Group III | 90.98 ± 2.98 ### | 95.01 ± 8.93 ### | 138.32 ± 2.43 ### | 7.02 ± 0.39 ### |
| Group IV | 132.76 ± 4.34 ## | 289.83 ± 8.93 # | 193.43 ± 3.64 ## | 3.98 ± 0.53 # |
| Group V | 99.21 ± 7.03 ## | 145.83 ± 6.83 ### | 143.29 ± 4.83 ### | 5.08 ± 0.98 ## |
| Time (min) | Function | Parameter |
|---|---|---|
| 2.00 | Change Solvent Composition | Solvent composition A: 95.00% B: 5.00% |
| 2.00 | Change Flow | Flow: 0.2 mL/min |
| 2.00 | Change Max. Pressure Limit | Max. Pressure Limit: 1200.00 bar |
| 15.00 | Change Solvent Composition | Solvent composition A: 5.00% B: 95.00% |
| 15.00 | Change Flow | Flow: 0.2 mL/min |
| 24.00 | Change Solvent Composition | Solvent composition A: 5.00% B: 95.00% |
| 24.00 | Change Flow | Flow: 0.2 mL/min |
| 25.00 | Change Solvent Composition | Solvent composition A: 95.00% B: 5.00% |
| 25.00 | Change Flow | Flow: 0.2 mL/min |
| 30.00 | Change Solvent Composition | Solvent composition A: 95.00% B: 5.00% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wali, A.F.; Pillai, J.R.; Al Dhaheri, Y.; Rehman, M.U.; Shoaib, A.; Sarheed, O.; Jabnoun, S.; Razmpoor, M.; Rasool, S.; Paray, B.A.; et al. Crocus sativus L. Extract Containing Polyphenols Modulates Oxidative Stress and Inflammatory Response against Anti-Tuberculosis Drugs-Induced Liver Injury. Plants 2020, 9, 167. https://doi.org/10.3390/plants9020167
Wali AF, Pillai JR, Al Dhaheri Y, Rehman MU, Shoaib A, Sarheed O, Jabnoun S, Razmpoor M, Rasool S, Paray BA, et al. Crocus sativus L. Extract Containing Polyphenols Modulates Oxidative Stress and Inflammatory Response against Anti-Tuberculosis Drugs-Induced Liver Injury. Plants. 2020; 9(2):167. https://doi.org/10.3390/plants9020167
Chicago/Turabian StyleWali, Adil Farooq, Jayachithra Ramakrishna Pillai, Yusra Al Dhaheri, Muneeb U. Rehman, Ambreen Shoaib, Omar Sarheed, Salma Jabnoun, Maryam Razmpoor, Saiema Rasool, Bilal Ahmad Paray, and et al. 2020. "Crocus sativus L. Extract Containing Polyphenols Modulates Oxidative Stress and Inflammatory Response against Anti-Tuberculosis Drugs-Induced Liver Injury" Plants 9, no. 2: 167. https://doi.org/10.3390/plants9020167
APA StyleWali, A. F., Pillai, J. R., Al Dhaheri, Y., Rehman, M. U., Shoaib, A., Sarheed, O., Jabnoun, S., Razmpoor, M., Rasool, S., Paray, B. A., & Ahmad, P. (2020). Crocus sativus L. Extract Containing Polyphenols Modulates Oxidative Stress and Inflammatory Response against Anti-Tuberculosis Drugs-Induced Liver Injury. Plants, 9(2), 167. https://doi.org/10.3390/plants9020167






