Phytochemical Profile, Antiradical Capacity and α-Glucosidase Inhibitory Potential of Wild Arbutus unedo L. Fruits from Central Italy: A Chemometric Approach
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material
2.3. Fourier–Transform Infrared Spectroscopy (FTIR) Analysis in Attenuated Total Reflectance (ATR) Mode
2.4. Extraction of Bioactive Compounds
2.5. Total Phenolic Content
2.6. Total Flavan-3-ol Content
2.7. Total Monomeric Anthocyanin Content
2.8. HPLC-DAD Profiles of Single Phenols and Anthocyanins
2.9. In Vitro Bioactivity of A. unedo Fruit Extracts
2.9.1. Antiradical Capacity (AC) Assays
2.9.2. α-Glucosidase Inhibition Assay
2.10. Statistical Analysis
3. Results and Discussion
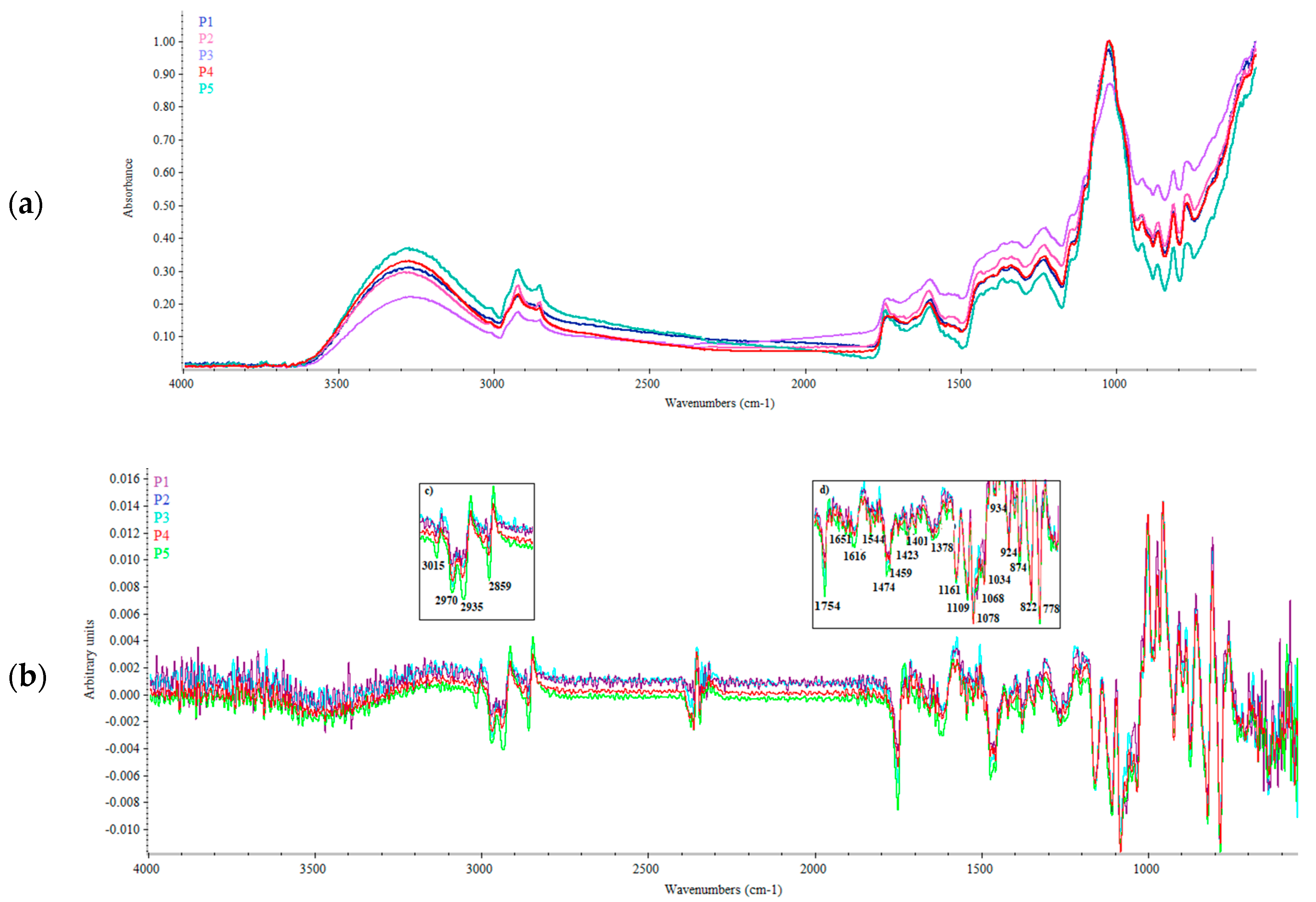
3.1. FTIR Spectra Analysis
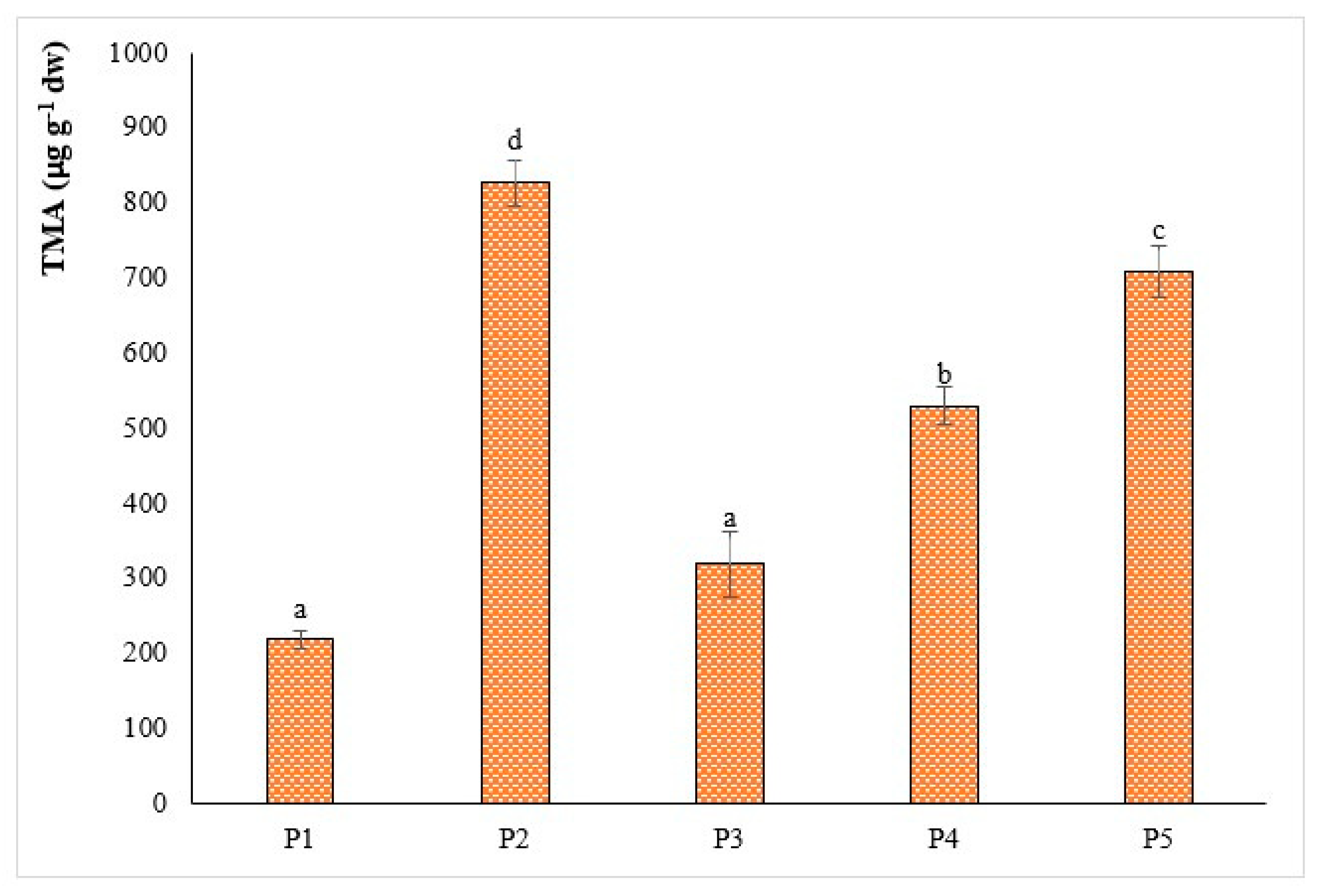
3.2. Phytochemical Composition of the Extracts Analyzed
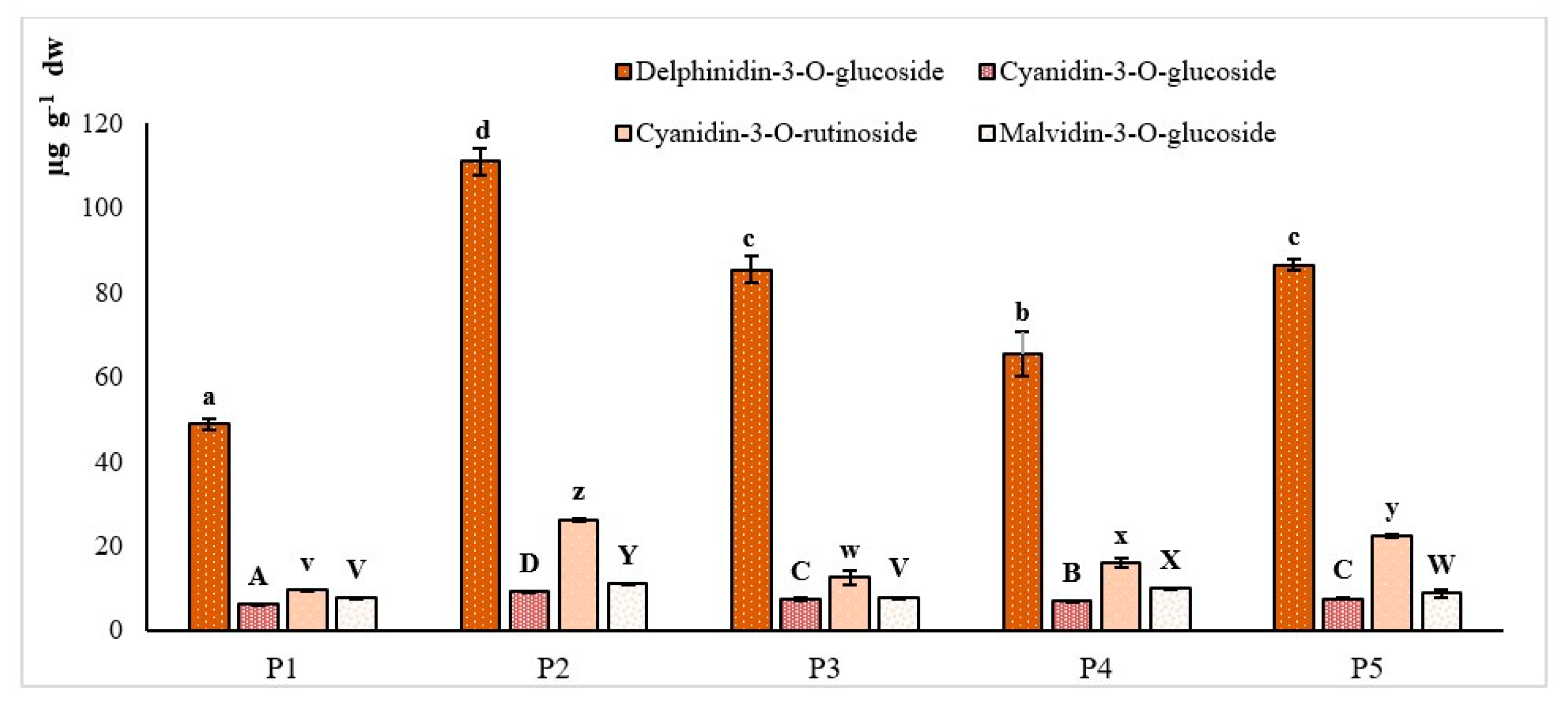
3.3. Chromatographic Profiling of Single Phenols
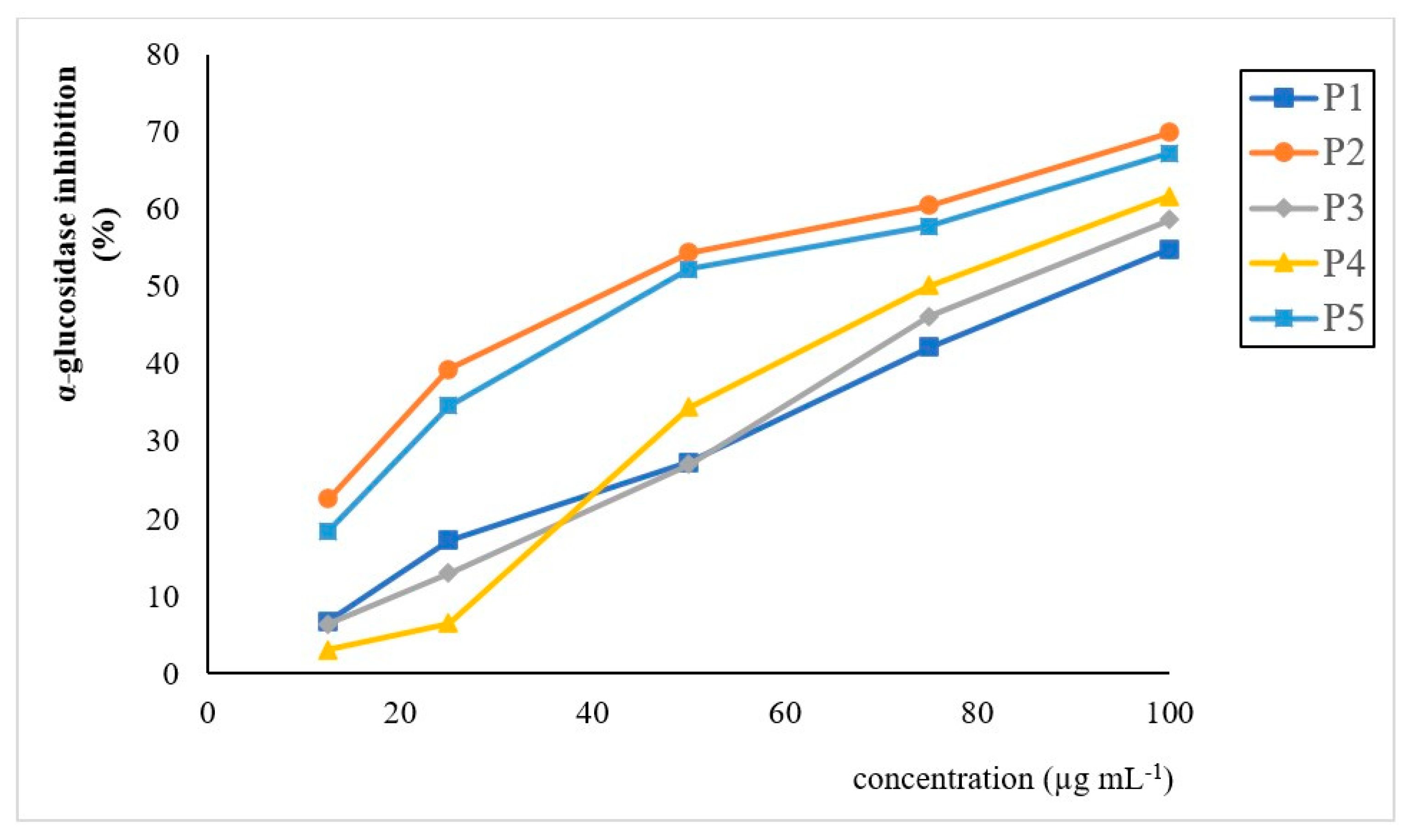
3.4. In Vitro Biological Activities
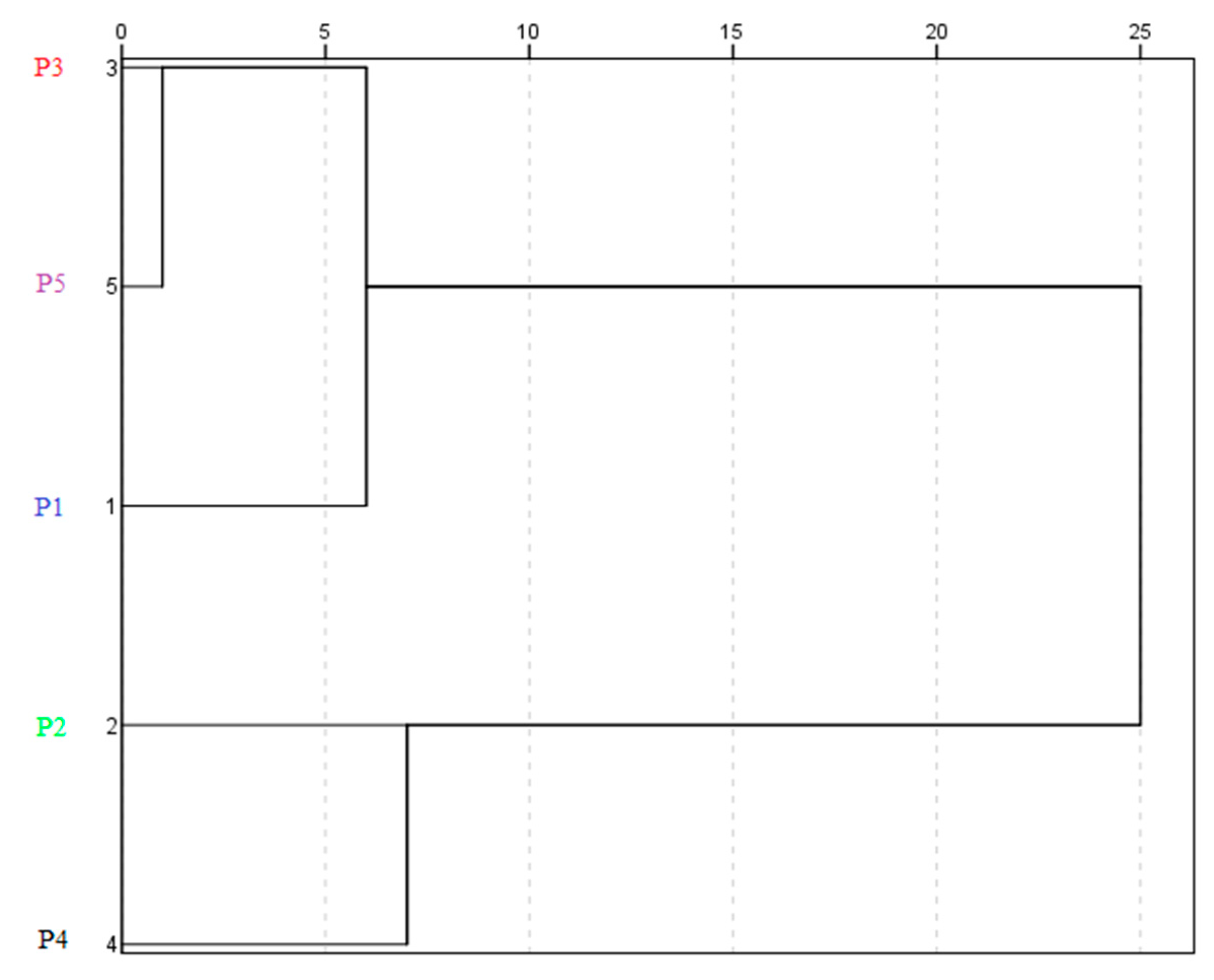
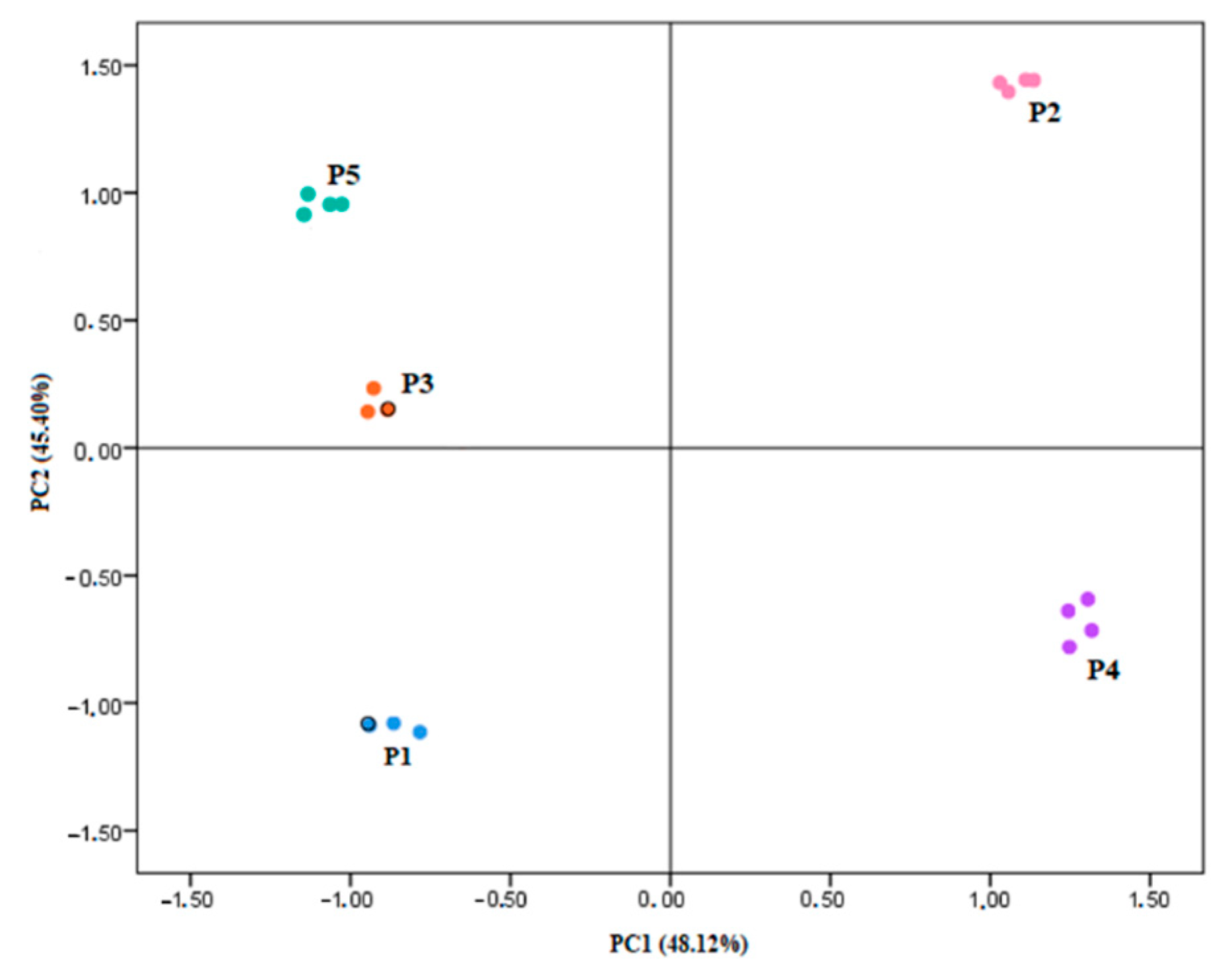
3.5. Exploratory Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Molina, M.; Pardo-de-Santayana, M.; Tardío, J. Natural production and cultivation of mediterranean wild edibles. In Mediterranean Wild Edible Plants; Sánchez-Mata, M.d.C., Tardío, J., Eds.; Springer International Publishing: New York, NY, USA, 2016; pp. 298–302. [Google Scholar]
- Schröter, D.; Cramer, W.; Leemans, R.; Prentice, I.C.; Araújo, M.B.; Arnell, N.W.; Anne, C. Ecosystem service supply and vulnerability to global change in Europe. Science 2005, 310, 1333–1337. [Google Scholar] [CrossRef]
- Miguel, M.G.; Faleiro, M.L.; Guerreiro, A.C.; Antunes, M.D. Arbutus unedo L.: Chemical and biological properties. Molecules 2014, 19, 15799–15823. [Google Scholar] [CrossRef]
- Alarcão-E-Silva, M.L.C.M.M.; Leitão, A.E.B.; Azinheira, H.G.; Leitão, M.C.A. The Arbutus berry: Studies on its color and chemical characteristics at two mature stages. J. Food Compos. Anal. 2001, 14, 27–35. [Google Scholar] [CrossRef]
- Morgado, S.; Morgado, M.; Plácido, A.I.; Roque, F.; Duarte, A.P. Arbutus unedo L.: From traditional medicine to potential uses in modern pharmacotherapy. J. Ethnopharmacol. 2018, 225, 90–102. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). The contribution of plant genetic resources for food and agriculture to food security and sustainable agricultural development. In The Second Report on the State of the World’s Plant Genetic Resources for Food and Agriculture; Food and Agriculture Organization (FAO): Rome, Italy, 2010; pp. 182–201. [Google Scholar]
- Şeker, M.; Toplu, C. Determination and comparison of chemical characteristics of Arbutus unedo L. and Arbutus andrachnae L. (family Ericaceae) fruits. J. Med. Food 2010, 13, 1013–1018. [Google Scholar] [CrossRef]
- McDougall, G.J.; Kulkarni, N.N.; Stewart, D. Current developments on the inhibitory efects of berry polyphenols on digestive enzymes. BioFactors 2008, 34, 73–80. [Google Scholar] [CrossRef]
- Montagut, G.; Onnockx, S.; Vaqué, M.; Bladé, C.; Blay, M.; Fernández-Larrea, J.; Ardévol, A. Oligomers of grape seed procyanidin extract activate the insulin receptor and key targets of the insulin signaling pathway diferently from insulin. J. Nutr. Biochem. 2010, 21, 476–481. [Google Scholar] [CrossRef]
- Ciccoritti, R.; Paliotta, M.; Centioni, L.; Mencarelli, F.; Carbone, K. The effect of genotype and drying condition on the bioactive compounds of sour cherry pomace. Eur. Food Res. Technol. 2018, 244, 635–645. [Google Scholar] [CrossRef]
- Garcia-Alonso, M.; Rimbach, G.; Sasai, M.; Nakahara, M.; Matsugo, S.; Uchida, Y.; Rivas-Gonzalo, J.C.; de Pascual-Teresa, S. Electron spin resonance spectroscopy studies on the free radical scavenging activity of wine anthocyanins and pyranoanthocyanins. Mol. Nutr. Food Res. 2005, 49, 1112–1119. [Google Scholar] [CrossRef]
- Tsuda, T.; Ueno, Y.; Yoshikawa, T.; Kojo, H.; Osawa, T. Microarray profiling of gene expression in human adipocytes in response to anthocyanins. Biochem. Pharmacol. 2006, 71, 1184–1197. [Google Scholar] [CrossRef]
- El Cadi, H.; el Cadi, A.; Kounnoun, A.; el Majdoub, Y.O.; Brigui, J.; Dugo, P.; Cacciola, F. Wild strawberry (Arbutus unedo): Phytochemical screening and antioxidant properties of fruits collected in northern Morocco. Arab. J. Chem. 2020, 13, 6299–6311. [Google Scholar] [CrossRef]
- Maldini, M.; D’Urso, G.; Pagliuca, G.; Petretto, G.L.; Foddai, M.; Gallo, F.R.; Pintore, G. HPTLC-PCA complementary to HRMS-PCA in the case study of Arbutus unedo antioxidant phenolic profiling. Foods 2019, 8, 294. [Google Scholar] [CrossRef] [PubMed]
- Amoriello, T.; Ciccoritti, R.; Carbone, K. Vibrational spectroscopy as a green technology for predicting nutraceutical properties and antiradical potential of early-to-late apricot genotypes. Postharvest Biol. Technol. 2019, 155, 156–166. [Google Scholar] [CrossRef]
- Carbone, K.; Giannini, B.; Picchi, V.; lo Scalzo, R.; Cecchini, F. Phenolic composition and free radical scavenging activity of different apple varieties in relation to the cultivar, tissue type and storage. Food Chem. 2011, 127, 493–500. [Google Scholar] [CrossRef]
- Carbone, K.; Mencarelli, F. Infuence of short-term postharvest ozone treatments in nitrogen or air atmosphere on the metabolic response of white wine grapes. Food Bioprocess. Technol. 2015, 8, 1739–1749. [Google Scholar] [CrossRef]
- Enzymatic Assay of α-glucosidase. Sigma-Aldrich Technical Bulletin. 1996. Available online: https://www.sigmaaldrich.com/content/dam/sigmaAldrich/docs/Sigma/General_Information/alpha_glucosidase_sed.pdf (accessed on 9 October 2019).
- Field, A. Discovering Statistics Using SPSS:(and Sex and Drugs and Rock’n’Roll); Sage Publications: Thousand Oaks, CA, USA, 2009. [Google Scholar]
- Umdale, S.; Ahire, M.; Aiwale, V.; Jadhav, A.; Mundada, P. Phytochemical investigation and antioxidant efficacy of wild, underutilized berries of economically important Indian Sandalwood (Santalum album L.). Biocatal. Agric. Biotechnol. 2020, 27, 101705. [Google Scholar] [CrossRef]
- Ayaz, F.A.; Kucukislamoglu, M.; Reunanen, M. Sugar, non-volatile and phenolic acids composition of strawberry tree (Arbutus unedo L. var. ellipsoidea) fruits. J. Food Compos. Anal. 2000, 13, 171–177. [Google Scholar] [CrossRef]
- Patle, T.K.; Shrivas, K.; Kurrey, R.; Upadhyay, S.; Jangde, R.; Chauhan, R. Phytochemical screening and determination of phenolics and flavonoids in Dillenia pentagyna using UV-vis and FTIR spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 242, 118717. [Google Scholar] [CrossRef]
- Heredia-Guerrero, J.A.; Benítez, J.J.; Domínguez, E.; Bayer, I.S.; Cingolani, R.; Athanassiou, A.; Heredia, A. Infrared and Raman spectroscopic features of plant cuticles: A review. Front. Plant Sci. 2014, 5, 305. [Google Scholar] [CrossRef]
- Alexandre, A.M.R.C.; Matias, A.; Duarte, C.M.M.; Bronze, M.R. High-pressure CO2 assisted extraction as a tool to increase phenolic content of strawberry-tree (Arbutus unedo) extracts. J. CO2 Util. 2018, 27, 73–80. [Google Scholar] [CrossRef]
- Doukani, K.; Tabak, S. Profil physicochimique du fruit “Lendj” (Arbutus unedo L.). Revue «Nature Technologie». B Sci. Agron. Biol. 2015, 12, 53–66. [Google Scholar]
- Zitouni, H.; Hssaini, L.; Zerhoune, M.; Ourradi, H.; Viuda-Martos, M.; Hernández, F.; Hanine, H. Phytochemical components and bioactivity assessment among twelve strawberry (Arbutus unedo L.) genotypes growing in Morocco using chemometrics. Foods 2020, 9, 1345. [Google Scholar] [CrossRef]
- López, C.J.; Caleja, C.; Prieto, M.A.; Barreiro, M.F.; Barros, L.; Ferreira, I.C. Optimization and comparison of heat and ultrasound assisted extraction techniques to obtain anthocyanin compounds from Arbutus unedo L. fruits. Food Chem. 2018, 264, 81–91. [Google Scholar] [CrossRef]
- Sang, J.; Sang, J.; Ma, Q.; Hou, X.F.; Li, C.Q. Extraction optimization and identification of anthocyanins from Nitraria tangutorun Bobr. seed meal and establishment of a green analytical method of anthocyanins. Food Chem. 2017, 218, 386–395. [Google Scholar] [CrossRef]
- Gironés-Vilaplana, A.; Baenas, N.; Villaño, D.; Speisky, H.; García-Viguera, C.; Moreno, D.A. Evaluation of Latin-American fruits rich in phytochemicals with biological effects. J. Funct. Foods 2014, 7, 599–608. [Google Scholar] [CrossRef]
- Pallauf, K.; Rivas-Gonzalo, J.C.; Del Castillo, M.D.; Cano, M.P.; de Pascual-Teresa, S. Characterization of the antioxidant composition of strawberry tree (Arbutus unedo L.) fruits. J. Food Compos. Anal. 2008, 21, 273–281. [Google Scholar] [CrossRef]
- Pawlowska, A.; Leo, M.; Braca, A. Phenolics of Arbutus unedo L. (Ericaceae) fruits: Identification of anthocyanins and gallic acid derivatives. J. Agric. Food Chem. 2006, 54, 10234–10238. [Google Scholar] [CrossRef]
- Rahman, M.M.; Ichiyanagi, T.; Komiyama, T.; Hatano, Y.; Konishi, T. Superoxide radical-and peroxynitrite-scavenging activity of anthocyanins; structure-activity relationship and their synergism. Free Radic. Res. 2006, 40, 993–1002. [Google Scholar] [CrossRef]
- Lee, D.Y.; Park, Y.J.; Song, M.G.; Kim, D.R.; Zada, S.; Kim, D.H. Cytoprotective effects of delphinidin for human chondrocytes against oxidative stress through activation of autophagy. Antioxidants 2020, 9, 83. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, Y.; Ren, G.; Yang, R.; Chen, J.; Xiang, X.; Chen, J. Inhibitory effect of delphinidin on oxidative stress induced by H2O2 in HepG2 cells. Oxid. Med. Cell. Longev. 2020. [Google Scholar] [CrossRef]
- Pimpão, R.C.; Dew, T.; Oliveira, P.B.; Williamson, G.; Ferreira, R.B.; Santos, C.N. Analysis of phenolic compounds in Portuguese wild and commercial berries after multienzyme hydrolysis. J. Agric. Food Chem. 2013, 61, 4053–4062. [Google Scholar] [CrossRef]
- Fritz, J.; Kern, M.; Pahlke, G.; Vatter, S.; Marko, D. Biological activities of malvidin, a red wine anthocyanidin. Mol. Nutr. Food Res. 2006, 50, 390–395. [Google Scholar] [CrossRef]
- Mosele, J.I.; Macià, A.; Motilva, M.J. Understanding of human metabolic pathways of different sub-classes of phenols from Arbutus unedo fruit after an acute intake. Food Funct. 2016, 7, 1700–1710. [Google Scholar] [CrossRef]
- Fortalezas, S.; Tavares, L.; Pimpão, R.; Tyagi, M.; Pontes, V.; Alves, P.M.; Santos, C.N. Antioxidant properties and neuroprotective capacity of strawberry tree fruit (Arbutus unedo). Nutrients 2010, 2, 214–229. [Google Scholar] [CrossRef]
- Albuquerque, B.R.; Prieto, M.A.; Barreiro, M.F.; Rodrigues, A.; Curran, T.P.; Barros, L.; Ferreira, I.C. Catechin-based extract optimization obtained from Arbutus unedo L. fruits using maceration/microwave/ultrasound extraction techniques. Ind. Crops Prod. 2017, 95, 404–415. [Google Scholar] [CrossRef]
- Tenuta, M.C.; Deguin, B.; Loizzo, M.R.; Dugay, A.; Acquaviva, R.; Malfa, G.A.; Tundis, R. Contribution of Flavonoids and Iridoids to the Hypoglycaemic, Antioxidant, and Nitric Oxide (NO) Inhibitory Activities of Arbutus unedo L. Antioxidants 2020, 9, 184. [Google Scholar] [CrossRef]



| Compound | P1 | P2 | P3 | P4 | P5 |
|---|---|---|---|---|---|
| Hydroxybenzoic acids (HBA) | |||||
| Gallic acid | 0.65 ± 0.01 b | 0.74 ± 0.01 b | 0.63 ± 0.01 b | 1.29 ± 0.02 c | 0.31 ± 0.03 a |
| Syringic acid | 0.09 ± 0.01 a | 0.14 ± 0.02 a | 0.11 ± 0.02 a | 0.26 ± 0.01 b | 0.47± 0.02 c |
| Protocatechuic acid | 0.35 ± 0.01 a | 0.45 ± 0.02 a | 0.31 ± 0.01 a | 0.59± 0.01 b | 0.44 ± 0.01 a |
| p-Hydroxybenzoic acid | 0.11 ± 0.02 a | 1.23 ± 0.02 d | 0.44 ± 0.01 b | 0.64 ± 0.02 c | 0.47 ± 0.02 b |
| Total HBA | 1.2 ± 0.1 | 2.56± 0.07 | 1.49 ± 0.05 | 2.78 ± 0.06 | 1.69 ± 0.08 |
| Hydroxycinnamic acids (HCA) | |||||
| p-Coumaric acid | n.d | n.d | n.d | n.d | n.d |
| Caffeic acid | n.d | n.d | n.d | n.d | n.d |
| Chlorogenic acid | 0.05 ± 0.01 b | 0.06 ± 0.01 c | 0.05 ± 0.02 a | 0.06 ± 0.01 d | 0.05 ± 0.01 a |
| Ferulic acid | n.d | n.d | n.d | n.d | n.d |
| Total HCA | 0.05 ± 0.01 | 0.06 ± 0.01 | 0.05 ± 0.02 | 0.06 ± 0.01 | 0.05 ± 0.01 |
| Flavan-3-ols | |||||
| Catechin | 0.82 ± 0.02 a | 1.72 ± 0.01 d | 1.04 ± 0.02 b | 1.34 ± 0.01 c | 0.87 ± 0.01 a |
| Epicatechin | 0.09 ± 0.01 a | 0.12 ± 0.02 a | 0.11 ± 0.02 a | 0.16 ± 0.02 b | 0.11 ± 0.01 a |
| Total flavans | 0.91 ± 0.03 | 1.84 ± 0.03 | 1.15 ± 0.04 | 1.5 ± 0.3 | 0.98 ± 0.02 b |
| Variable | Cluster 1 | Cluster 2 | Cluster 3 |
|---|---|---|---|
| TPC | 18 ± 2 ab | 23.9 ± 0.2 c | 18.1 ± 0.4 ba |
| FLC | 3.0 ± 0.4 ab | 8.4 ± 0.2 ca | 7.0 ± 0.4 ba |
| TMA | 30.1 ± 0.4 ab | 63.1 ± 0.4 ba | 48.6 ± 0.1 c |
| DEL | 49 ± 1 abc | 88 ± 3 ca | 86 ± 1 ba |
| CGC | 6.3 ± 0.1 ac | 8.1 ± 0.3 ca | 7.5 ± 0.1 b |
| CRC | 9.5 ± 0.4 ac | 21 ± 1 ca | 17.5 ± 0.3 ba |
| GA | 0.60 ± 0.06 b | 1.00 ± 0.04 cba | 0.50 ± 0.01 a |
| pOH-B | 0.10 ± 0.01 abc | 0.9 ± 0.1 cba | 0.40 ± 0.02 bac |
| SA | 0.08 ± 0.01 ac | 0.20 ± 0.01 ca | 0.160 ± 0.008 b |
| pCA | 0.35 ± 0.01 ac | 0.52 ± 0.03 ca | 0.38 ± 0.01 b |
| CAT | 0.82 ± 0.02 ac | 1.53 ± 0.06 cba | 0.95 ± 0.01 bc |
| CA | 1.6 ± 0.2 abc | 1.9 ± 0.5 cba | 1.7 ± 0.6 bac |
| ABTS | 3.0 ± 0.2 cba | 2.4 ± 0.1 bc | 2.0 ± 0.2 ac |
| DPPH | 109.6 ± 0.3 ca | 35 ± 1 abc | 100 ± 1 ba |
| Variables | ||
|---|---|---|
| 1 | 2 | |
| TPC | 0.974 | |
| TMA | 0.825 | |
| CAT | 0.862 | |
| CA | 0.848 | |
| MGC | 0.805 | 0.547 |
| CRC | 0.895 | |
| CGC | 0.914 | |
| DEL | 0.947 | |
| pOH-B | 0.679 | 0.721 |
| DPPH(EC50) | −0.918 | |
| Eigenvalues | 4.81 | 4.54 |
| % of variance | 48.12 | 45.40 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Macchioni, V.; Santarelli, V.; Carbone, K. Phytochemical Profile, Antiradical Capacity and α-Glucosidase Inhibitory Potential of Wild Arbutus unedo L. Fruits from Central Italy: A Chemometric Approach. Plants 2020, 9, 1785. https://doi.org/10.3390/plants9121785
Macchioni V, Santarelli V, Carbone K. Phytochemical Profile, Antiradical Capacity and α-Glucosidase Inhibitory Potential of Wild Arbutus unedo L. Fruits from Central Italy: A Chemometric Approach. Plants. 2020; 9(12):1785. https://doi.org/10.3390/plants9121785
Chicago/Turabian StyleMacchioni, Valentina, Veronica Santarelli, and Katya Carbone. 2020. "Phytochemical Profile, Antiradical Capacity and α-Glucosidase Inhibitory Potential of Wild Arbutus unedo L. Fruits from Central Italy: A Chemometric Approach" Plants 9, no. 12: 1785. https://doi.org/10.3390/plants9121785
APA StyleMacchioni, V., Santarelli, V., & Carbone, K. (2020). Phytochemical Profile, Antiradical Capacity and α-Glucosidase Inhibitory Potential of Wild Arbutus unedo L. Fruits from Central Italy: A Chemometric Approach. Plants, 9(12), 1785. https://doi.org/10.3390/plants9121785





