Quantification and Tissue Localization of Selenium in Rice (Oryza sativa L., Poaceae) Grains: A Perspective of Agronomic Biofortification
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Fields
2.2. Climate, Soil, and Irrigation Water Analysis
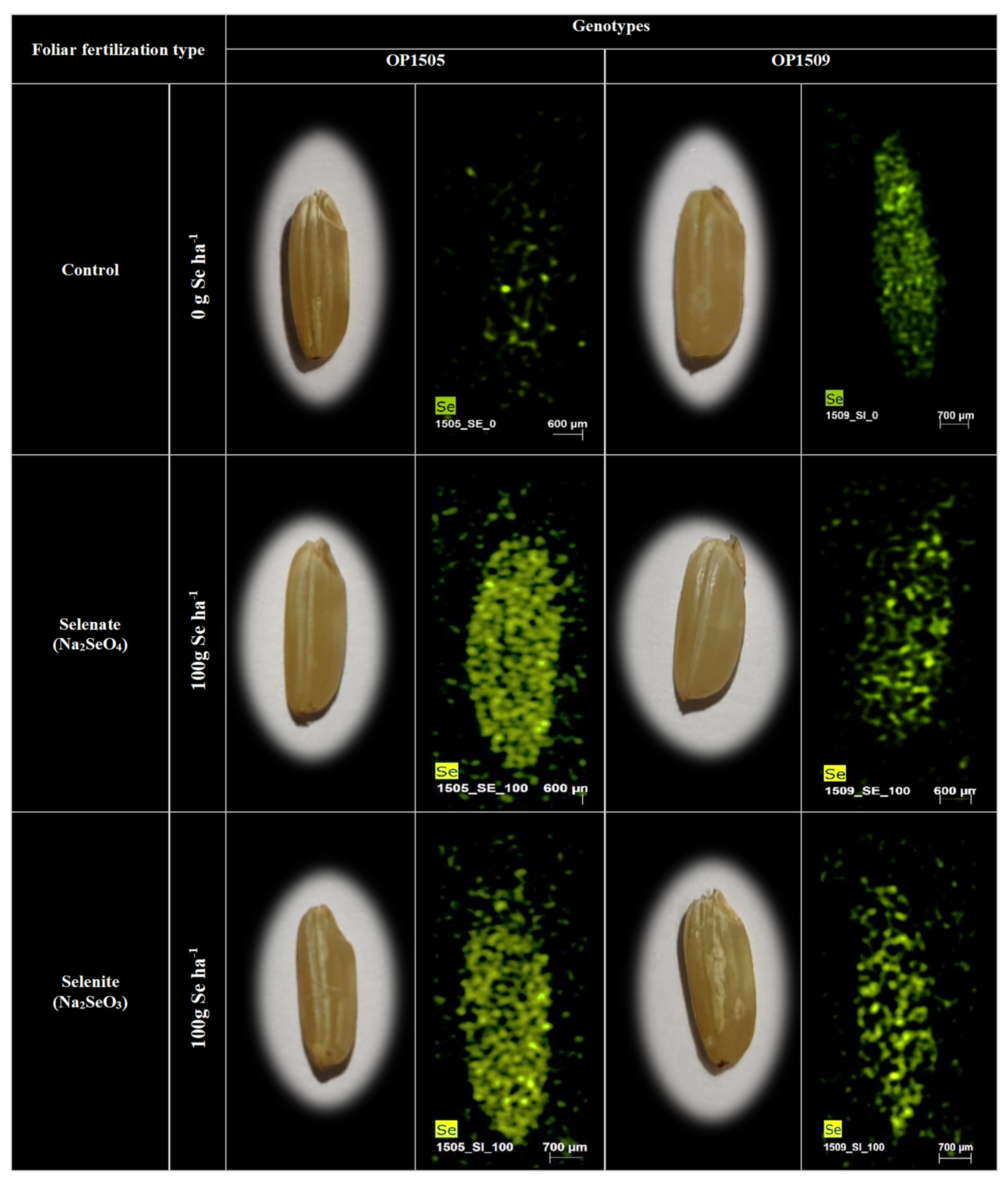
2.3. Analysis of Grain Selenium Content and Tissues Location
2.4. Thousand Grain Weight, Colorimetry and Crude Protein Content Analysis
2.5. Statistical Analysis
3. Results
3.1. Characterization of Rice Field and Yield
3.2. Selenium Accumulation in Rice Grains
3.3. Grain Weight, Colorimetry, and Crude Protein Content Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, Y.D.; Wang, X.; Wong, Y.S. Generation of selenium-enriched rice with enhanced grain yield, selenium content and bioavailability through fertilisation with selenite. Food Chem. 2013, 141, 2385–2393. [Google Scholar] [CrossRef] [PubMed]
- White, P.; Broadley, M. Biofortification of crops with seven mineral elements often lacking in human diets—Iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef] [PubMed]
- Sarwar, N.; Akhtar, M.; Kamran, M.A.; Imran, M.; Riaz, M.A.; Kamran, K.; Hussain, S. Selenium biofortification in food crops: Key mechanisms and future perspectives. J. Food Compos. Anal. 2020, 93, 103615. [Google Scholar] [CrossRef]
- Trippe, R.C., III; Pilon-Smits, E.A.H. Selenium transport and metabolism in plants: Phytoremediation and biofortification implications. J. Hazard. Mater. 2021, 404, 124–178. [Google Scholar] [CrossRef]
- Pérez-Corona, M.T.; Sanchez-Martınez, M.; Valderrama, M.J.; Rodriguez, M.E.; Camara, C.; Madrid, Y. Selenium biotransformation by Saccharomyces cerevisiae and Saccharomyces bayanus during white wine manufacture: Laboratory-scale experiments. Food Chem. 2011, 124, 1050–1055. [Google Scholar] [CrossRef]
- Andrade, F.; Da Silva, G.; Guimarães, K.; Barreto, H.; De Souza, K.; Guilherme, L.; Faquin, V.; Reis, A. Selenium protects rice plants from water deficit stress. Ecotoxicol. Environ. Saf. 2018, 164, 562–570. [Google Scholar] [CrossRef]
- Reis, A.; El-Ramady, H.; Santos, E.F.; Gratão, P.L.; Schomburg, L. Overview of selenium deficiency and toxicity worldwide: Affected areas, selenium-related health issues, and case studies. In Selenium in Plants; Plant Ecophysiology; Springer: Cham, Switzerland, 2017; Volume 11, pp. 209–230. [Google Scholar] [CrossRef]
- Lidon, F.; Oliveira, K.; Galhano, C.; Guerra, M.; Ribeiro, M.; Pelica, J.; Pataco, I.; Ramalho, J.; Leitão, A.; Almeida, A.; et al. Selenium biofortification of rice through foliar application with selenite and selenate. Exp. Agric. 2018, 55, 528–542. [Google Scholar] [CrossRef]
- Lidon, F.; Oliveira, K.; Ribeiro, M.; Pelica, J.; Pataco, I.; Ramalho, J.; Leitão, A.; Almeida, A.; Campos, P.; Ribeiro-Barros, A.; et al. Selenium biofortification of rice grains and implications on macronutrients quality. J. Cereal Sci. 2018, 81, 22–29. [Google Scholar] [CrossRef]
- Li, Y.; Liu, K.; Chen, F. Effect of selenium enrichment on the quality of germinated brown rice during storage. Food Chem. 2016, 207, 20–26. [Google Scholar] [CrossRef]
- Zhao, F.; McGrath, S. Biofortification and phytoremediation. Curr.Opin. Plant. Biol. 2009, 12, 373–380. [Google Scholar] [CrossRef]
- Boldrin, P.F.; Faquin, V.; Ramos, S.J.; Boldrin, K.V.F.; Ávila, F.W.; Guilherme, L.R.G. Soil and foliar application of selenium in rice biofortification. J. Food Compos. Anal. 2018, 31, 238–244. [Google Scholar] [CrossRef]
- Lyons, G.H.; Genc, Y.; Stangoulis, J.C.; Palmer, L.T.; Graham, R.D. Selenium distribution in wheat grain, and the effect of postharvest processing on wheat selenium content. Biol. Trace Elem. Res. 2005, 103, 155–168. [Google Scholar] [CrossRef]
- Hartikainen, H. Biogeochemistry of selenium and its impact of food chain quality and human health. J. Trace Elem. Med. Bio. 2005, 18, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsov, V.V.; Kholodova, V.P.; Kuznetsov, V.V.; Yagodin, B.A. Selenium regulates the water status of plants exposed to drought. Dokl. Bio. Sci. 2003, 390, 266–268. [Google Scholar] [CrossRef] [PubMed]
- Li, H.F.; McGrath, S.P.; Zhao, F.J. Selenium uptake, translocation and speciation in wheat supplied with selenate or selenite. New Phytol. 2008, 178, 92–102. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.; Lombi, E.; Sun, G.; Schecke, K.; Zhu, Y.; Feng, X.; Zhu, J.; Carey, A.; Adomako, E.; Lawgali, Y.; et al. Selenium Characterization in the Global Rice Supply Chain. Environ. Sci. Tech. 2009, 43, 6024–6030. [Google Scholar] [CrossRef]
- Yunus, M.; Rashid, S.; Chowdhury, S. Per Capita Rice Consumption in Bangladesh: Available Estimates and IFPRIs Validation Survey Results; IFPRP Working Paper 3; International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2019. [Google Scholar] [CrossRef]
- Gabinete de Planeamento, Políticas e Administração Geral (GPP). Rice. 2017. Available online: http://www.gpp.pt/images/gam/1/fi/ArrozFI.pdf (accessed on 1 October 2020).
- Instituto Nacional de Estatística (INE). 2019. Available online: https://www.ine.pt/xportal/xmain?xpid=INE&xpgid=ine_main (accessed on 1 October 2020).
- Pessoa, F.P.; Scotti-Campos, P.; Pais, I.P.; Feiteiro, A.; Canuto, D.; Simões, M.; Pelica, J.; Pataco, I.; Ribeiro, V.; Reboredo, F.H.; et al. Nutritional profile of the Portuguese cabbage (Brassica oleracea L. var. costata) and its relationship with the elemental soil analysis. Emir. J. Food Agric. 2016, 28, 381–388. [Google Scholar] [CrossRef]
- Pelica, J.; Barbosa, S.; Lidon, F.; Pessoa, M.; Reboredo, F.; Calvão, T. The paradigm of high concentration of metals of natural or anthropogenic origin in soils—The case of Neves-Corvo mine area (southern Portugal). J. Geochem. Explor. 2018, 186, 12–23. [Google Scholar] [CrossRef]
- Rodier, J.; Legube, B.; Merlet, N. L’Analyse de l’Eau, 9th ed.; Dunod: Paris, France, 2009; 1579p, ISBN 9782100072460. [Google Scholar]
- Cardoso, P.; Velu, G.; Singh, R.P.; Santos, J.P.; Carvalho, M.L.; Lourenço, V.M.; Lidon, F.C.; Reboredo, F.; Guerra, M. Localization and distribution of Zn and Fe in grains of biofortified bread wheat lines through micro- and triaxial- X-ray fluorescence spectrometry. Spectrochim. Acta B 2018, 141, 70–79. [Google Scholar] [CrossRef]
- Ramalho, J.C.; Pais, I.P.; Leitão, A.E.; Guerra, M.; Reboredo, F.H.; Máguas, C.M.; Carvalho, M.L.; Scotti-Campos, P.; Ribeiro-Barros, A.I.; Lidon, F.J.C.; et al. Can elevated air [CO2] conditions mitigate the predicted warming impact on the quality of coffee bean? Front. Plant Sci. 2018, 9. [Google Scholar] [CrossRef]
- ISO 20483. Cereals and Pulses—Determination of the Nitrogen Content and Calculation of the Crude Protein Content—Kjeldahl Method; International Organization for Standardization: Geneva, Switzerland, 2006. [Google Scholar]
- Nawaz, F.; Ahmad, R.; Ashraf, M.Y.; Waraich, E.A.; Khan, S.Z. Effect of selenium foliar spray on physiological and biochemical processes and chemical constituents of wheat under drought stress. Ecotoxicol. Environ. Saf. 2015, 113, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Kaur, N.; Kaur, S.; Nayyar, H. Selenium as a nutrient in biostimulation and biofortification of cereals. Indian J. Plant. Physiol. 2016, 22, 1–15. [Google Scholar] [CrossRef]
- Kolbert, Z.; Molnár, Á.; Feigl, G.; Van Hoewyk, D. Plant selenium toxicity: Proteome in the crosshairs. J. Plant Physiol. 2019, 232, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Feng, X.B.; Chan, H.M.; Larssen, T. New insights into traditional health risk assessments of mercury exposure: Implications of selenium. Environ. Sci. Technol. 2014, 48, 1206–1212. [Google Scholar] [CrossRef]
- Seregina, I.I.; Niloskaya, N.T.; Ostapenko, N.O. The role of selenium in the formation of the grain yield in spring wheat. J. Cereal Sci. 2001, 1, 44–50. [Google Scholar] [CrossRef]
- Reis, H.P.G.; de Queiroz Barcelos, J.P.; Furlani, E., Jr.; Santos, E.F.; Silva, V.M.; Moraes, M.F.; Putti, F.F.; Dos Reis, A.R. Agronomic biofortification of upland rice with selenium and nitrogen and its relation to grain quality. J. Cereal Sci. 2018, 79, 508–515. [Google Scholar] [CrossRef]
- Itani, T.; Tamaki, M.; Arai, E.; Horino, T.S. Distribution of amylose, nitrogen, and minerals in rice kernels with various character. J. Agric. Food Chem. 2002, 50, 5326–5332. [Google Scholar] [CrossRef]
- Mancebo, C.; Rodriguez, P.; Gomez, M. Assessing rice flour-starch- protein mixtures to produce gluten free sugar-snap cookies. Food Sci. Technol. 2016, 67, 127–132. [Google Scholar] [CrossRef]
- Puncha-Arnon, S.; Uttapap, D. Rice starch vs. rice flour: Differences in their properties when modified by heat–moisture treatment. Carbohydr. Polym. 2013, 91, 85–91. [Google Scholar] [CrossRef]
- Mangueze, A.; Pessoa, M.F.; Silva, M.J.; Ndayiragije, A.; Magaia, H.; Cossa, V.; Reboredo, F.; Carvalho, M.L.; Santos, J.P.; Guerra, M.; et al. Simultaneous zinc and selenium biofortification in rice accumulation, localization and implications on the overall mineral content of the flour. J. Cereal Sci. 2018, 82, 34–41. [Google Scholar] [CrossRef]
| A | |||||||||||
| Soil Analysis (0–30 cm Deep) | |||||||||||
| pH | Electrical Conductivity | Organic Matter | Ca | K | P | Fe | S | Mg | Pb | As | Se |
| dS/m | % | ppm | |||||||||
| 5.86 | 0.223 | 1.32 | 0.16 ± 0.01 | 2.48 ± 0.04 | 1.50 ± 0.06 | 0.28 ± 0.01 | 299 ± 114 | 682 ± 99 | 11.6 ± 0.61 | 13.1 ± 0.18 | 1< |
| B | |||||||||||
| Water Analysis | |||||||||||
| pH | Electrical Conductivity | Ca2+ | K+ | Mg2+ | Na+ | Cl− | HCO3− | SO42− | PO43− | ||
| dS/m | Mg.l−1 (meq.l−1) | ||||||||||
| 1 | 6.9 | 0.376 | 9.4 (0.5) | 8.1 (0.2) | 4.5 (0.4) | 30.2 (1.3) | 52 (1.4) | 67.1 (1.1) | 45 (0.9) | <1.5 (<0.04) | |
| 2 | 6.3 | 0.420 | 13.2 (0.7) | 2.3 (0.06) | 5.2 (0.4) | 39.1 (1.7) | 76 (2.1) | 54.9 (0.9) | 43 (0.8) | <1.5 (<0.04) | |
| 3 | 6.6 | 0.800 | 27.6 (1.4) | 14.4 (0.4) | 7.1 (0.6) | 62.5 (2.7) | 131 (3.7) | 85.4 (1.4) | 74 (1.5) | <1.5 (<0.04) | |
| Treatments (g Se·ha−1) | OP1505 | OP1509 |
|---|---|---|
| Na2SeO4 | ||
| Control | 2.43 ± 0.12as | 1.37 ± 0.07ar |
| 25 | 2.13 ± 0.11ar | 2.47 ± 0.12br |
| 50 | 6.98 ± 0.35cr | 6.72 ± 0.34cr |
| 75 | 4.75 ± 0.24br | 6.60 ± 0.33cr |
| 100 | 17.2 ± 0.86ds | 6.45 ± 0.32cr |
| Na2SeO3 | ||
| Control | 2.01 ± 0.10ar | 2.20 ± 0.11ar |
| 25 | 5.19 ± 0.26cr | 7.34 ± 0.37cs |
| 50 | 6.12 ± 0.31cr | 13.1 ± 0.65ds |
| 75 | 3.51 ± 0.18br | 5.32 ± 0.27br |
| 100 | 16.9 ± 0.84ds | 8.15 ± 0.41cr |
| Treatments (g Se·ha−1) | Paddy | Brown Rice | White Rice | |||
|---|---|---|---|---|---|---|
| OP1505 | OP1509 | OP1505 | OP1509 | OP1505 | OP1509 | |
| Na2SeO4 | ||||||
| Control | 26.7 ± 0.47a | 31.6 ± 0.67a | 24.2 ± 0.41a | 27.0 ± 0.36a | 21.7 ± 0.16a | 23.6 ± 0.19a |
| 25 | 27.2 ± 0.25a | 31.5 ± 0.61a | 24.1 ± 0.34a | 27.3 ± 0.57a | 20.9 ± 1.38a | 23.4 ± 0.40a |
| 50 | 28.5 ± 0.48a | 31.6 ± 0.32a | 24.0 ± 0.27a | 26.9 ± 0.31a | 21.4 ± 0.35a | 23.8 ± 0.48a |
| 75 | 27.6 ± 0.31a | 30.8 ± 0.80a | 24.4 ± 0.25a | 27.2 ± 0.16a | 20.9 ± 0.23a | 23.7 ± 0.47a |
| 100 | 27.7 ± 0.31a | 32.2 ± 1.71a | 24.3 ± 0.33a | 27.6 ± 0.69a | 21.5 ± 1.42a | 23.9 ± 0.44a |
| Na2SeO3 | ||||||
| Control | 26.4 ± 1.19a | 29.3 ± 0.52a | 24.2 ± 0.38a | 25.9 ± 0.63a | 21.0 ± 0.27a | 24.0 ± 0.81a |
| 25 | 26.7 ± 0.07a | 30.2 ± 0.51a | 24.3 ± 0.19a | 27.0 ± 0.27a | 21.1 ± 0.39a | 24.4 ± 0.14a |
| 50 | 26.6 ± 0.68a | 29.8 ± 0.67a | 24.1 ± 0.11a | 26.6 ± 0.17a | 21.5 ± 0.24a | 23.9 ± 0.31a |
| 75 | 28.3 ± 0.51a | 28.8 ± 1.01a | 23.6 ± 0.25a | 25.9 ± 0.38a | 21.3 ± 0.18a | 23.5 ± 0.37a |
| 100 | 27.2 ± 0.62a | 30.7 ± 1.38a | 23.9 ± 0.20a | 26.0 ± 0.38a | 20.6 ± 0.12a | 23.7 ± 0.17a |
| Treatments (g Se·ha−1) | Whole Flour | Refined Flour | ||
|---|---|---|---|---|
| OP1505 | OP1509 | OP1505 | OP1509 | |
| (%) | ||||
| Na2SeO4 | ||||
| Control | 6.39a,r | 6.24a,r | 5.53a,r | 5.47a,r |
| 25 | 6.05a,r | 5.98a,r | 5.16a,r | 5.15a,r |
| 50 | 6.62a,r | 6.49a,r | 5.83a,r | 4.63a,r |
| 75 | 5.82a,r | 6.06a,r | 5.40a,r | 5.00a,r |
| 100 | 6.62a,r | 6.16a,r | 5.24a,r | 4.99a,r |
| Na2SeO3 | ||||
| Control | 6.15a,r | 6.70a,r | 4.63a,r | 5.65a,r |
| 25 | 6.32a,r | 5.83a,r | 4.64a,r | 4.75a,r |
| 50 | 6.64a,r | 7.15a,r | 4.72a,r | 5.32a,r |
| 75 | 6.82a,r | 6.65a,r | 4.72a,r | 5.46a,r |
| 100 | 6.30a,r | 6.64a,r | 5.23a,r | 4.98a,r |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marques, A.C.; Lidon, F.C.; Coelho, A.R.F.; Pessoa, C.C.; Luís, I.C.; Scotti-Campos, P.; Simões, M.; Almeida, A.S.; Legoinha, P.; Pessoa, M.F.; et al. Quantification and Tissue Localization of Selenium in Rice (Oryza sativa L., Poaceae) Grains: A Perspective of Agronomic Biofortification. Plants 2020, 9, 1670. https://doi.org/10.3390/plants9121670
Marques AC, Lidon FC, Coelho ARF, Pessoa CC, Luís IC, Scotti-Campos P, Simões M, Almeida AS, Legoinha P, Pessoa MF, et al. Quantification and Tissue Localization of Selenium in Rice (Oryza sativa L., Poaceae) Grains: A Perspective of Agronomic Biofortification. Plants. 2020; 9(12):1670. https://doi.org/10.3390/plants9121670
Chicago/Turabian StyleMarques, Ana Coelho, Fernando C. Lidon, Ana Rita F. Coelho, Cláudia Campos Pessoa, Inês Carmo Luís, Paula Scotti-Campos, Manuela Simões, Ana Sofia Almeida, Paulo Legoinha, Maria Fernanda Pessoa, and et al. 2020. "Quantification and Tissue Localization of Selenium in Rice (Oryza sativa L., Poaceae) Grains: A Perspective of Agronomic Biofortification" Plants 9, no. 12: 1670. https://doi.org/10.3390/plants9121670
APA StyleMarques, A. C., Lidon, F. C., Coelho, A. R. F., Pessoa, C. C., Luís, I. C., Scotti-Campos, P., Simões, M., Almeida, A. S., Legoinha, P., Pessoa, M. F., Galhano, C., Guerra, M. A. M., Leitão, R. G., Ramalho, J. C., Semedo, J. M. N., Bagulho, A., Moreira, J., Rodrigues, A. P., Marques, P., ... Reboredo, F. H. (2020). Quantification and Tissue Localization of Selenium in Rice (Oryza sativa L., Poaceae) Grains: A Perspective of Agronomic Biofortification. Plants, 9(12), 1670. https://doi.org/10.3390/plants9121670

















