Vacillantins A and B, New Anthrone C-glycosides, and a New Dihydroisocoumarin Glucoside from Aloe vacillans and Its Antioxidant Activities
Abstract
1. Introduction
2. Results and Discussion
2.1. Structure Elucidation of New Compounds
2.2. Antioxidant Activities Results
3. Materials and Methods
3.1. Apparatus and Chemicals
3.2. Plant Material
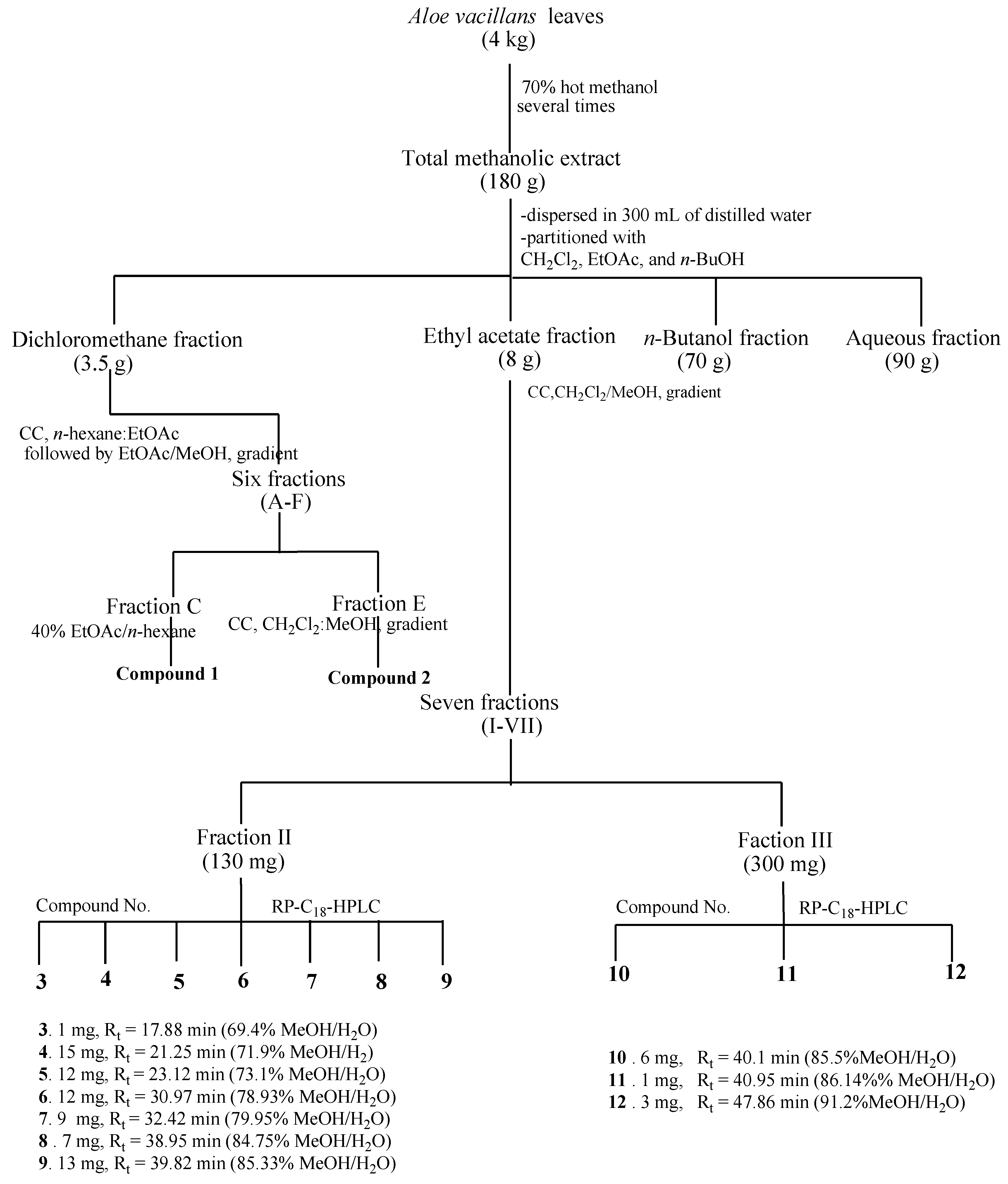
3.3. Extraction and Isolation
3.4. Antioxidant Activity
3.4.1. DPPH Radical Scavenging Assay
3.4.2. ABTS Radical Cation Scavenging Assay
3.4.3. Reducing Power Assay
3.4.4. Superoxide Radical Anion Scavenging Assay
3.4.5. Nitric Oxide Radical Scavenging Assay
3.4.6. Spectral Data of the New Compounds
Vacillanoside (3)
Vacillantin A (10)
Vacillantin B (11)
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Demmisew, S. Botanical aspects of Aloes of North East Africa. Bull. Chem. Soc. Ethiop. 1996, 10, 74–88. [Google Scholar]
- Gao, Y.; Kuok, K.I.; Jin, Y.; Wang, R. Biomedical applications of Aloe vera. Crit. Rev. Food Sci. Nutr. 2019, 59 (Suppl. S1), S244–S256. [Google Scholar] [CrossRef]
- Kojo, E.; Qian, H. Aloe Vera: A Valuable Ingredient for the Food, Pharmaceutical and Cosmetic Industries—A Review. Crit. Rev. Food Sci. Nutr. 2004, 44, 91–96. [Google Scholar]
- Dagne, E.; Bisrat, D.; Viljoen, A.; Van Wyk, B.E. Chemistry of Aloe Species. Curr. Org. Chem. 2000, 4, 1055–1078. [Google Scholar] [CrossRef]
- Steenkamp, V.; Stewart, M.J. Medicinal applications and toxicological activities of Aloe products. Pharmbiol. Biol. 2007, 45, 411–420. [Google Scholar] [CrossRef]
- Cock, I.E. The Genus Aloe: Phytochemistry and Therapeutic Uses Including Treatments for Gastrointestinal Conditions and Chronic Inflammation. Prog. Drug Res. 2015, 70, 179–235. [Google Scholar] [PubMed]
- Yagi, A.; Kabash, A.; Okamura, N.; Haraguchi, H.; Moustafa, S.M.; Khalifa, T.I. Antioxidant, free radical scavenging and anti-inflammatory effects of aloesin derivatives in Aloe vera. Planta Med. 2002, 68, 957–960. [Google Scholar] [CrossRef] [PubMed]
- Yagi, A.; Kabash, A.; Mizuno, K.; Moustafa, S.M.; Khalifa, T.I.; Tsuji, H. Radical scavenging glycoprotein inhibiting cyclooxygenase-2 and thromboxane A2 synthase from Aloe vera gel. Planta Med. 2003, 69, 269–271. [Google Scholar] [CrossRef]
- Lim, B.O.; Seong, N.S.; Choue, R.W.; Kim, J.D.; Lee, H.Y.; Kim, S.Y.; Yu, B.P.; Jeon, T.I.; Park, D.K. Efficacy of dietary Aloe vera supplementation on hepatic cholesterol and oxidative status in aged rats. J. Nutr. Sci. Vitaminol. 2003, 49, 292–296. [Google Scholar] [CrossRef]
- Su, Y.T.; Chang, H.L.; Shyue, S.K.; Hsu, S.L. Emodin induces apoptosis in human lung adenocarcinoma cells through a reactive oxygen species-dependent mitochondrial signaling pathway. Biochem. Pharmacol. 2005, 70, 229–241. [Google Scholar] [CrossRef] [PubMed]
- Im, S.A.; Oh, S.T.; Song, S.; Kim, M.R.; Kim, D.S.; Woo, S.S.; Jo, T.H.; Park, Y.I.; Lee, C.K. Identification of optimal molecular size of modified Aloe polysaccharides with maximum immunomodulatory activity. Int. Immunopharmacol. 2005, 5, 271–279. [Google Scholar] [CrossRef]
- Wang, Z.W.; Wang, Y.; Huang, Z.S. The radio-protective effect of Aloe polysaccharides on irradiated mice. Chin. Trad. Herb. Drugs 2002, 33, 251–254. [Google Scholar]
- US Food and Drug Administration. Code of Federal Regulations (Food and Drugs), Title 21; US Government Printing Office: Washington, DC, USA, 1991; pp. 170–199.
- Council of Europe. Flavoring Substances and Natural Source of Flavorings, 3rd ed.; Maisonneuve: Moulins-les Metz, France, 1981; p. 376. [Google Scholar]
- Valverde, J.M.; Valero, D.; Martinez-Romera, D.; Guillén, A.; Castillo, S.; Serrano, M. Novel edible coating based on Aloe vera gel to maintain table grape quality and safety. J. Agric. Food Chem. 2005, 53, 7807–7813. [Google Scholar] [CrossRef]
- Abdalla, H.I.; Shaaban, M.; Shaaban, K.A.; Abu-Gabal, N.S.; Shalaby, N.M.; Laatsch, H. New bioactive compounds from Aloe hijazensis. Nat. Prod. Res. 2009, 23, 1035–1049. [Google Scholar] [CrossRef] [PubMed]
- Collenette, S. Wild Flowers of Saudi Arabia. Riyadh; National Commission for Wild Life Conservation and Development (NCWCD): Riyadh, Saudi Arabia, 1999; p. 18. [Google Scholar]
- Wood, J.R.I.; Thomas, H.H. A Handbook of the Yemen Flora; Royal Botanic Gardens, Kew: Richmond, UK, 1997. [Google Scholar]
- Zhong, J.; Huang, Y.; Ding, W.; Wu, X.; Wan, J.; Luo, H. Chemical constituents of Aloe barbadensis Miller and their inhibitory effects on phosphodiesterase-4D. Fitoterapia 2013, 91, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Speranza, G.; Manitto, P.; Monti, D. Feralolide, a dihydroisocoumarin from Cape aloe. Phytochemistry 1993, 3, 175–178. [Google Scholar] [CrossRef]
- Kurizaki, A.; Watanabe, T.; Devkota, H.P. Chemical constituents from the flowers of Aloe arborescens. Nat. Prod. Commun. 2019, 14, 1–4. [Google Scholar] [CrossRef]
- Yasuda, K.; Uehara, S.; Takano, I.; Shindo, T.; Nishijima, M. Stability of barbaloin in aqueous solution. Food Preserv. Sci. 2000, 26, 85–90. [Google Scholar] [CrossRef]
- Farah, M.H.; Andersson, R.; Samuelsson, G. Microdontin A and B: Two new aloin derivatives from Aloe microdonta. Planta Med. 1992, 58, 88–93. [Google Scholar] [CrossRef]
- Shin, K.H.; Woo, W.S.; Lim, S.S.; Shim, C.S.; Chung, H.S.; Kennelly, E.J.; Kinghorn, D. Elgonica-Dimers A and B, two potent alcohol metabolism inhibitory constituents of Aloe arborescens. J. Nat. Prod. 1997, 60, 1180–1182. [Google Scholar] [CrossRef]
- Conner, J.M.; Alexander, I.; Gray, A.I.; Peter, G.; Waterman, P.G.; Reynolds, T. Novel anthrone-anthraquinone dimers from Aloe elgonica. J. Nat. Prod. 1990, 53, 1362–1364. [Google Scholar] [CrossRef]
- Abd-Alrahman, S.H.; Salem-Bekhit, M.M.; Elhalwagy, M.E.A.; Abdel-Mageed, W.M.; Radwan, A.A. Phytochemical Screening and Antimicrobial Activity of EthOH/Water Ziziphus jujuba Seeds Extracts. J. Pure Appl. Microbiol. 2013, 7, 823–828. [Google Scholar]
- Veitch, N.C.; Simmonds, M.S.J.; Blaney, W.M.; Reynolds, T. A dihydroisocoumarin glucoside from Aloe hildebrandtii. Phytochemistry 1994, 35, 1163–1166. [Google Scholar] [CrossRef]
- Dagne, E.; Bisrat, D.; Van Wyk, B.E.; Viljoen, A.; Hellwig, V.; Steglich, W. Anthrones from Aloe microstigma. Phytochemistry 1997, 44, 1271–1274. [Google Scholar] [CrossRef]
- Rauwald, H.W.; Lohse, K. Structure revision of 4-hydroxyaloin: 10-hydroxyaloins A and B as main In Vitro-oxidation products of the diastereomeric aloins. Planta Med. 1992, 58, 259–262. [Google Scholar] [CrossRef]
- Loots, D.T.; van der Westhuizen, F.H.; Botes, L. Aloe ferox leaf gel phytochemical content, antioxidant capacity, and possible health benefits. J. Agric. Food Chem. 2007, 55, 6891–6896. [Google Scholar] [CrossRef]
- Hu, Y.; Xu, J.; Hu, Q. Evaluation of antioxidant potential of Aloe vera (Aloe barbadensis miller) extracts. J. Agric. Food Chem. 2003, 51, 7788–7791. [Google Scholar] [CrossRef]
- Beppu, H.; Koike, T.; Shimpo, K.; Chihara, T.; Hoshino, M.; Ida, C.; Kuzuya, H. Radical-scavenging effects of Aloe arborescens Miller on prevention of pancreatic islet B-cell destruction in rats. J. Ethnopharmacol. 2003, 89, 37–45. [Google Scholar] [CrossRef]
- Wintola, O.A.; Afolayan, A.J. Phytochemical constituents and antioxidant activities of the whole leaf extract of Aloe ferox mill. Pharmacogn. Mag. 2011, 7, 325–333. [Google Scholar]
- Botes, L.; van der Westhuizen, F.H.; Loots, D.T. Phytochemical contents and antioxidant capacities of two Aloe Greatheadii Var. Davyana extracts. Molecules 2008, 13, 2169–2180. [Google Scholar] [CrossRef]
- Asamenew, G.; Bisrat, D.; Mazumder, A.; Asres, K. In vitro antimicrobial and antioxidant activities of anthrone and chromone from the latex of Aloe harlana reynolds. Phytother. Res. 2011, 25, 1756–1760. [Google Scholar] [CrossRef] [PubMed]
- Yoo, E.A.; Kim, S.D.; Lee, W.M.; Park, H.J.; Kim, S.K.; Cho, J.Y.; Min, W.; Rhee, M.H. Evaluation of Antioxidant, antinociceptive, and anti-Inflammatory activities of ethanol extracts from Aloe Saponaria Haw. Phytother. Res. 2008, 22, 1389–1395. [Google Scholar] [CrossRef] [PubMed]
- Cardarellia, M.; Rouphaelb, Y.; Pellizzonic, M.; Collad, G.; Lucin, L. Profile of bioactive secondary metabolites and antioxidant capacity of leaf exudates from eighteen Aloe species. Ind. Crops Prod. 2017, 108, 44–51. [Google Scholar] [CrossRef]
- Salehi, B.; Albayrak, S.; Antolak, H.; Kręgiel, D.; Pawlikowska, E.; Sharifi-Rad, M.; Uprety, Y.; Tsouh Fokou, P.V.; Yousef, Z.; Amiruddin Zakaria, Z.; et al. Aloe genus plants: From farm to food applications and phytopharmacotherapy. Int. J. Mol. Sci. 2018, 19, 2843. [Google Scholar] [CrossRef] [PubMed]
- Abdul Qadir, M.; Shahzadi, S.K.; Bashir, A.; Munir, A.; Shahzad, S. Evaluation of phenolic compounds and antioxidant and antimicrobial activities of some common herbs. Int. J. Anal. Chem. 2017, 2017, 3475738. [Google Scholar] [CrossRef] [PubMed]
- Hęś, M.; Dziedzic, K.; Górecka, D.; Jędrusek-Golińska, A.; Gujska, E. Aloe vera (L.) Webb.: Natural Sources of Antioxidants—A Review. Plant. Foods Hum. Nutr. 2019, 74, 255–265. [Google Scholar]
- Teka, T.; Kassahun, H. Characterization and Evaluation of Antioxidant Activity of Aloe schelpei Reynolds. Drug Des. Dev. Ther. 2020, 14, 1003. [Google Scholar] [CrossRef] [PubMed]
- Braca, A.; Tommasi, N.D.; Bari, L.D.; Pizza, C.; Politi, M.; Morelli, I. Antioxidant principles from Bauhinia terapotensis. J. Nat. Prod. 2001, 64, 892–895. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Oyaizu, M. Studies on products of browning reactions. Antioxidative activities of products of browning reaction prepared from glucosamine. Jpn. J. Nutr. 1986, 44, 307–315. [Google Scholar] [CrossRef]
- Fontana, L.; Giagulli, C.; Minuz, P.; Lechi, A.; Laudanna, C. 8-Iso-PGF2 alpha induces beta 2-integrinmediated rapid adhesion of human polymorphonuclear neutrophils: A link between oxidative stress and ischemia/reperfusion injury. Arterioschler Thromb. Vasc. Biol. 2001, 21, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Nagmoti, D.M.; Khatri, D.K.; Juvekar, P.R.; Juvekar, A.R. Antioxidant activity and free radical scavenging potential of Pithecellobium dulce Benth seed extracts. Free Rad. Antiox. 2011, 2, 37–43. [Google Scholar] [CrossRef]
 ) and HMBC (H
) and HMBC (H C) correlations for compounds 3 and 10.
C) correlations for compounds 3 and 10.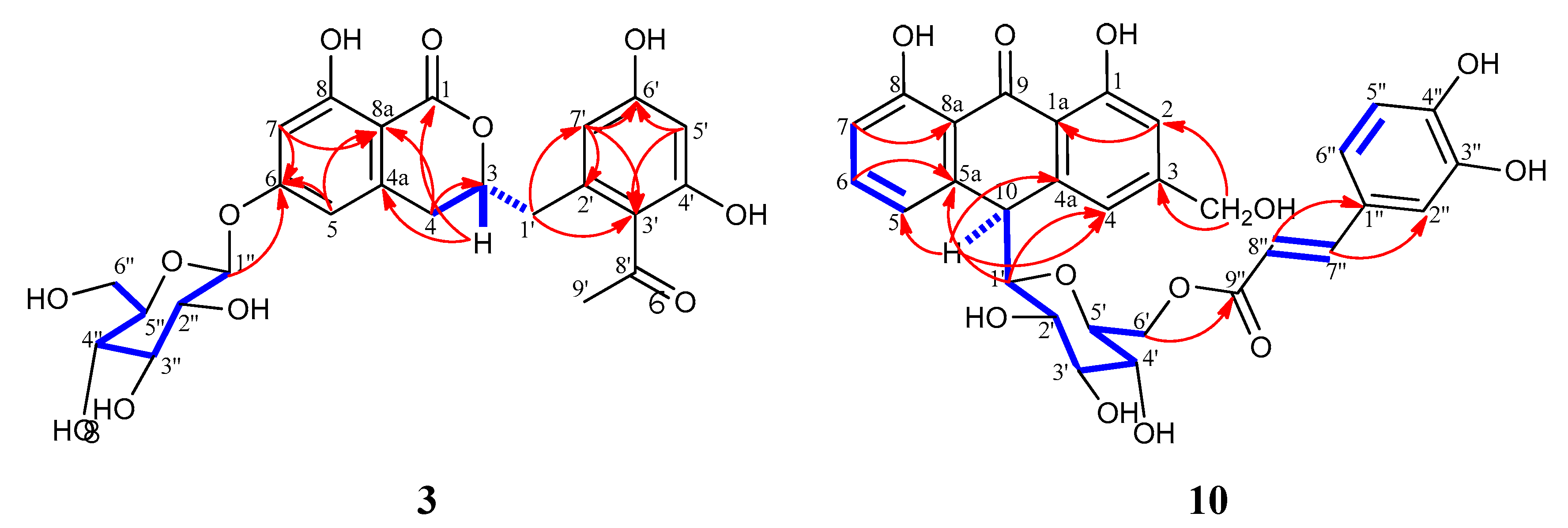
| No. | 2 | 3 | ||
|---|---|---|---|---|
| δHa | δCa | δHb | δCb | |
| 1 | -- | 171.3 | -- | 171.0 |
| 3 | 4.70, m | 81.5 | 4.76, m | 81.6 |
| 4 | 2.87, m | 33.5 | 2.92, m | 33.4 |
| 4a | -- | 143.3 | -- | 143.2 |
| 5 | 6.20, br s | 108.0 | 6.51, br s | 108.4 |
| 6 | -- | 166.3 | -- | 166.6 |
| 7 | 6.19, br s | 102.2 | 6.51, br s | 103.6 |
| 8 | -- | 165.6 | -- | 165.1 |
| 8a | -- | 101.6 | -- | 104.5 |
| 1′a 1′b | 2.95, dd (13.8, 5.2) 3.05, dd (13.8, 7.4) | 39.6 | 2.93, m 3.07, dd (13.6, 7.5) | 39.6 |
| 2′ | -- | 139.2 | -- | 139.2 |
| 3′ | -- | 121.8 | -- | 121.9 |
| 4′ | -- | 161.4 | -- | 161.4 |
| 5′ | 6.26, d (2.0) | 102.5 | 6.26, d (1.7) | 102.5 |
| 6′ | -- | 160.1 | -- | 160.1 |
| 7′ | 6.29, d (2.0) | 111.5 | 6.29, d (1.7) | 111.5 |
| 8′ | -- | 206.8 | -- | 206.8 |
| 9′ | 2.53, s | 32.9 | 2.55, s | 32.9 |
| 1″ | -- | -- | 4.99, d (7.2) | 101.2 |
| 2″ | -- | -- | 3.44, m | 74.7 |
| 3″ | -- | -- | 3.46–3.5 | 77.8 |
| 4″ | -- | -- | 3.38, m | 71.2 |
| 5″ | -- | -- | 3.44–3.47, m | 78.2 |
| 6″ | -- | -- | 3.68, dd (12.1, 5.7) 3.88, dd (12.2, 1.7) | 62.4 |
| No. | 10 | 11 | ||
|---|---|---|---|---|
| δHa | δCa | δHb | δCb | |
| 1 | - | 163.1 | - | 163.0 |
| 2 | 6.80, br s | 114.6 | 6.77, br d (2.2) | 114.2 |
| 3 | - | 150.9 | - | 152.2 |
| 4 | 6.97, br s | 119.1 | 6.97, br s | 117.2 |
| 5 | 6.90, d (8.0) | 119.4 | 7.03, d (7.9) | 121.3 |
| 6 | 7.39, t (8.0) | 136.9 | 7.43, br t (7.9) | 136.0 |
| 7 | 6.69, d (8.0) | 117.9 | 6.81, d (7.9) | 117.3 |
| 8 | - | 162.5 | - | 163.0 |
| 9 | - | 195.3 | - | 195.4 |
| 10 | 4.46, br s | 45.5 | 4.54, br s | 45.4 |
| 1a | - | 117.9 | - | 117.8 |
| 4a | - | 142.3 | 142.8 | |
| 5a | - | 142.3 | - | 142.9 |
| 8a | - | 118.8 | - | 119.2 |
| 11 | 4.62, br s | 64.5 | 4.60, br s | 64.5 |
| 1′ | 3.30, d (9.2) | 85.8 | 3.29, d (9.4) | 85.9 |
| 2′ | 3.07, t (9.2) | 71.5 | 3.10, t (9.3) | 71.5 |
| 3′ | 3.30, m | 79.7 | 3.30, m | 79.8 |
| 4′ | 2.87, t (9.3) | 71.7 | 2.88, t (9.3) | 71.8 |
| 5′ | 3.30, dd (8.1, 1.8) | 79.0 | 3.05, t (8.3) | 79.1 |
| 6′ | a. 3.83, dd (11.7, 6.9) b. 4.23, br d (10.2) | 64.4 | 3.84, dd (11.7, 6.7) 4.24, br d (11.7) | 64.5 |
| Caffeoyl moiety | ||||
| 1″ | - | 127.8 | - | 127.9 |
| 2″ | 7.09, br s | 115.6 | 7.09, br s | 115.6 |
| 3″ | - | 146.6 | - | 146.9 |
| 4″ | - | 149.4 | - | 149.4 |
| 5″ | 6.83, d (8.2) | 116.4 | 6.84, dd (8.2, 1.4) | 116.4 |
| 6″ | 6.97, br d (7.6) | 123.1 | 6.98, br d (8.5) | 123.1 |
| 7″ | 7.33, d (15.9) | 146.8 | 7.35, dd (15.9, 1.5) | 146.9 |
| 8″ | 6.06, d (15.9) | 114.8 | 6.08, dd (15.9, 1.5) | 114.9 |
| 9″ | - | 168.9 | - | 168.9 |
| Treatments | Mean % Scavenging ± SD | Mean % OD ± SD | ||
|---|---|---|---|---|
| Concentration (µg/mL) | DPPH | ABTS | FRAP | |
| MeOH | 10 | 9.03 ± 1.37 | 10.05 ± 3.04 | 0.24 ± 0.06 |
| 20 | 16.62 ± 4.28 | 24.40 ± 5.94 | 0.30 ± 0.04 | |
| 50 | 24.65 ± 4.12 | 35.10 ± 3.64 | 0.35 ± 0.02 | |
| 100 | 35.21 ± 3.07 | 45.12 ± 8.87 | 0.35 ± 0.02 | |
| Ascorbic Acid | 100 | 88.94 ± 2.29 | 88.20 ± 2.72 | 1.53 ± 0.13 |
| IC50 | 75.84 ± 8.51 | 59.24 ± 13.39 | 94.83 ± 5.44 | |
| CH2Cl2 | 10 | 29.62 ± 3.44 | 42.95 ± 3.22 | 0.57 ± 0.04 |
| 20 | 49.30 ± 5.30 | 60.07 ± 7.67 | 0.87 ± 0.03 | |
| 50 | 68.37 ± 3.40 | 72.54 ± 3.78 | 1.15 ± 0.15 | |
| 100 | 81.90 ± 1.43 | 83.49 ± 3.09 | 1.44 ± 0.07 | |
| Ascorbic Acid | 100 | 91.94 ± 0.92 | 90.51 ± 4.46 | 1.84 ± 0.11 |
| IC50 | 22.14 ± 2.25 | 13.51 ± 2.33 | 30.64 ± 2.46 | |
| EtOAc | 10 | 5.01 ± 0.75 | 5.43 ± 1.06 | 0.14 ± 0.02 |
| 20 | 12.04 ± 2.30 | 13.16 ± 4.20 | 0.25 ± 0.06 | |
| 50 | 23.59 ± 3.63 | 24.24 ± 5.11 | 0.32 ± 0.03 | |
| 100 | 32.71 ± 4.32 | 34.36 ± 2.99 | 0.46 ± 0.04 | |
| Ascorbic Acid | 100 | 92.98 ± 1.56 | 90.53 ± 2.63 | 1.75 ± 0.09 |
| IC50 | 79.92 ± 5.43 | 76.69 ± 7.53 | 118.3 ± 5.8 | |
| n-BuOH | 10 | 10.06 ± 4.40 | 38.10 ± 5.54 | 0.51 ± 0.02 |
| 20 | 33.71 ± 3.76 | 54.53 ± 4.77 | 0.69 ± 0.02 | |
| 50 | 55.32 ± 4.05 | 68.59 ± 4.97 | 0.73 ± 0.03 | |
| 100 | 67.83 ± 0.29 | 78.86 ± 5.84 | 0.86 ± 0.08 | |
| Ascorbic Acid | 100 | 93.47 ± 0.72 | 90.58 ± 2.56 | 1.83 ± 0.06 |
| IC50 | 37.07 ± 5.67 | 17.16 ± 3.03 | 44.93 ± 3.83 | |
| Sample (µg/mL) | % Inhibition ± SD | ||||
|---|---|---|---|---|---|
| MeOH | CH2Cl2 | EtOAc | n-BuOH | Ascorbic Acid | |
| 20 | 8.59 ± 1.87 | 36.33 ± 2.50 | 6.03 ± 0.13 | 23.72 ± 2.11 | 69.77 ± 1.76 |
| 40 | 15.19 ± 5.48 | 54.22 ± 2.72 | 14.12 ± 3.43 | 40.95 ± 5.83 | 77.84 ± 4.94 |
| 60 | 26.10 ± 10.64 | 68.79 ± 7.11 | 24.15 ± 3.08 | 52.59 ± 6.93 | 82.05 ± 4.63 |
| 80 | 38.43 ± 6.15 | 79.88 ± 3.23 | 33.74 ± 5.03 | 74.77 ± 2.26 | 86.40 ± 1.81 |
| 100 | 54.07 ± 4.20 | 87.90 ± 1.77 | 43.09 ± 4.81 | 85.86 ± 2.26 | 90.92 ± 1.70 |
| IC50 | 97.29 ± 11.13 | 32.92 ± 2.99 | 118.4 ± 11.4 | 46.86 ± 5.1 | 7.09 ± 3.09 |
| Sample (µg/mL) | % Inhibition ± SD | ||||
|---|---|---|---|---|---|
| MeOH | CH2Cl2 | EtOAc | n-BuOH | Ascorbic Acid | |
| 20 | 6.32 ± 3.50 | 34.83 ± 1.97 | 6.80 ± 4.49 | 42.09 ± 3.73 | 48.51 ± 7.92 |
| 40 | 15.21 ± 6.63 | 49.66 ± 3.85 | 16.69 ± 4.24 | 51.62 ± 3.73 | 64.26 ± 2.48 |
| 60 | 28.57 ± 2.06 | 60.80 ± 5.61 | 29.74 ± 1.28 | 61.16 ± 1.82 | 71.42 ± 4.81 |
| 80 | 42.05 ± 5.49 | 71.85 ± 3.23 | 35.08 ± 1.56 | 68.27 ± 2.84 | 79.82 ± 1.51 |
| 100 | 46.78 ± 7.61 | 80.03 ± 3.43 | 40.40 ± 2.10 | 72.29 ± 4.00 | 89.28 ± 2.02 |
| IC50 | 103.2 ± 12.13 | 37.23 ± 3.72 | 128.7 ± 16.3 | 32.44 ± 3.82 | 22.37 ± 3.82 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Tamimi, M.; Al-Massarani, S.M.; El-Gamal, A.A.; Basudan, O.A.; Abdel-Kader, M.S.; Abdel-Mageed, W.M. Vacillantins A and B, New Anthrone C-glycosides, and a New Dihydroisocoumarin Glucoside from Aloe vacillans and Its Antioxidant Activities. Plants 2020, 9, 1632. https://doi.org/10.3390/plants9121632
Al-Tamimi M, Al-Massarani SM, El-Gamal AA, Basudan OA, Abdel-Kader MS, Abdel-Mageed WM. Vacillantins A and B, New Anthrone C-glycosides, and a New Dihydroisocoumarin Glucoside from Aloe vacillans and Its Antioxidant Activities. Plants. 2020; 9(12):1632. https://doi.org/10.3390/plants9121632
Chicago/Turabian StyleAl-Tamimi, Maram, Shaza M. Al-Massarani, Ali A. El-Gamal, Omer A. Basudan, Maged S. Abdel-Kader, and Wael M. Abdel-Mageed. 2020. "Vacillantins A and B, New Anthrone C-glycosides, and a New Dihydroisocoumarin Glucoside from Aloe vacillans and Its Antioxidant Activities" Plants 9, no. 12: 1632. https://doi.org/10.3390/plants9121632
APA StyleAl-Tamimi, M., Al-Massarani, S. M., El-Gamal, A. A., Basudan, O. A., Abdel-Kader, M. S., & Abdel-Mageed, W. M. (2020). Vacillantins A and B, New Anthrone C-glycosides, and a New Dihydroisocoumarin Glucoside from Aloe vacillans and Its Antioxidant Activities. Plants, 9(12), 1632. https://doi.org/10.3390/plants9121632





