LED Lights Affecting Morphogenesis and Isosteroidal Alkaloid Contents in Fritillaria cirrhosa D. Don—An Important Chinese Medicinal Herb
Abstract
1. Introduction
2. Material and Methods
2.1. Callus Multiplication
2.2. Influence of Different Light Spectra on Morphogenesis in Embryogenic Calli and Contents of Isosteroidal Alkaloids
2.3. Development of Bulblets from Somatic Embryos
2.4. Chemicals and Other Materials
2.5. Preparation of Fe3O4@C18 Nanoparticle Composite
2.6. Extraction Procedure
2.7. LC-MS/MS Conditions
2.8. Statistical Analysis
3. Results and Discussion
3.1. Callus Proliferation
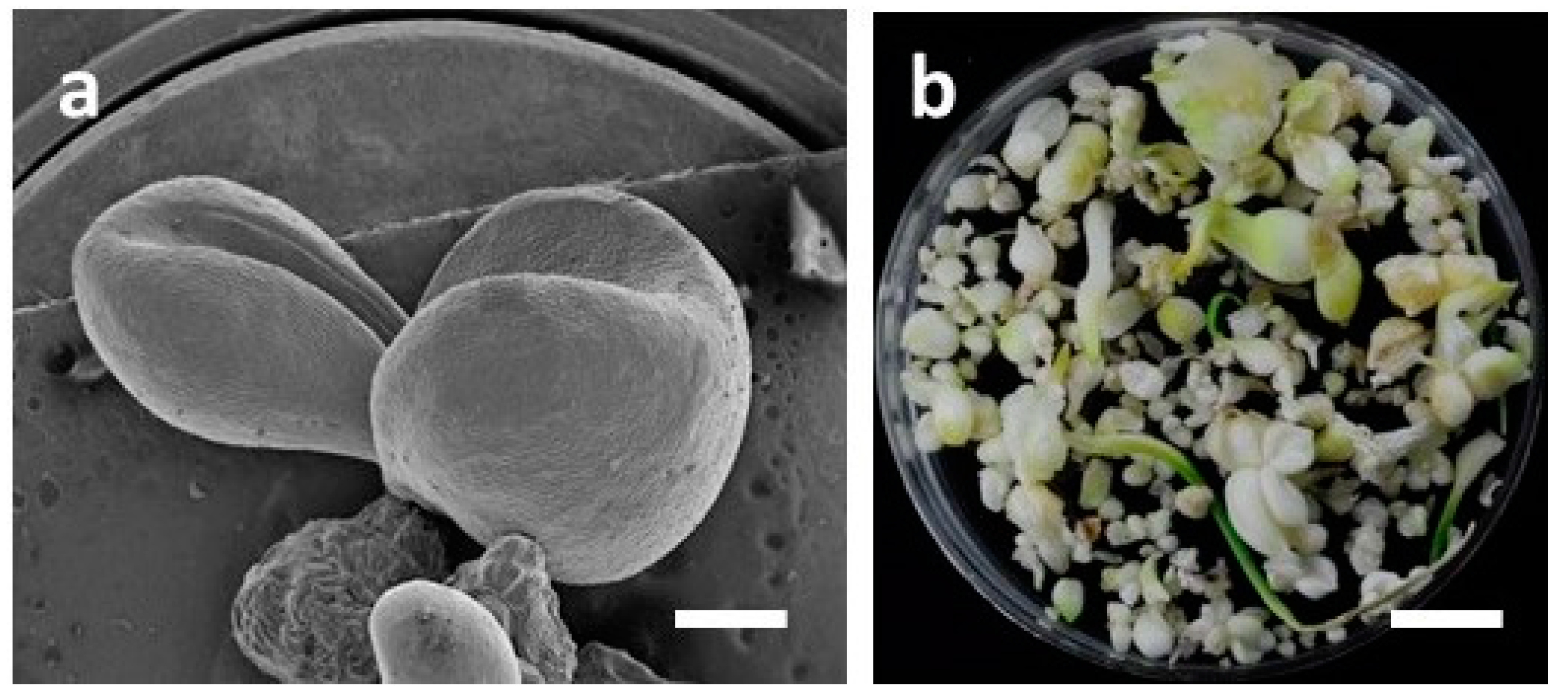
3.2. Influence of LED Lights on Morphogenesis of Embryogenic Calli (EC) of F. cirrhosa
3.3. Influence of LED Lights on the Contents of Isosteroidal Alkaloids in In Vitro Cultures of F. cirrhosa
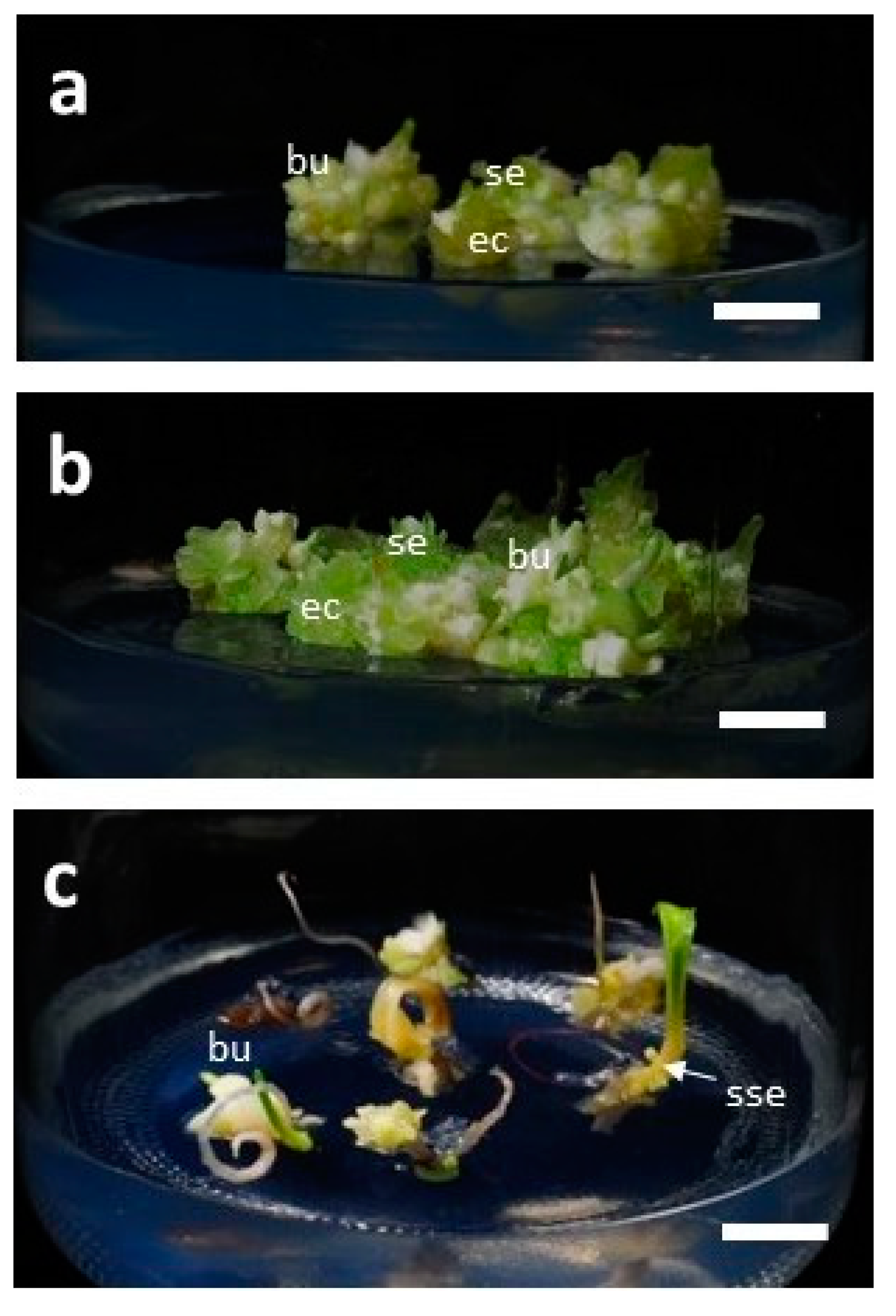
3.4. Development of Bulblets in Somatic Embryos of F. cirrhosa
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lin, G.; Li, P.; Li, S.L.; Chan, S.W. Chromatographic analysis of Fritillaria isosteroidal alkaloids, the active ingredients of Beimu, the antitussive traditional Chinese medicinal herb. J. Chromatogr. A 2001, 935, 321–338. [Google Scholar] [CrossRef]
- The State Pharmacopoeia Commission of P.R. China. Pharmacopoeia of the People’s Republic of China, 10th ed.; China Medical Science Press: Beijing, China, 2015; p. 37. [Google Scholar]
- Li, H.J.; Jiang, Y.; Li, P. Chemistry, bioactivity and geographical diversity of steroidal alkaloids from the Liliaceae family. Nat. Prod. Rep. 2006, 23, 735–752. [Google Scholar] [CrossRef]
- Li, K.; Wu, W.; Zheng, Y.; Dai, Y.; Xiang, L.; Liao, K. Genetic diversity of Fritillaria from Sichuan province based on ISSR. China J. Chin. Mater. Med. 2009, 34, 2149–2154. [Google Scholar]
- Ding, K.; Lin, G.; Ho, Y.P.; Cheng, T.Y.; Li, P. Prederivatization and high-performance liquid chromatographic analysis of alkaloids of bulbs of Fritillaria. J. Pharm. Sci. 1996, 85, 1174–1179. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zhao, W.; Pan, L.; Zhang, A.; Chen, Q.; Xu, K.; Lu, H.; Chen, Y. Peimine, a main active ingredient of Fritillaria, exhibits anti-inflammatory and pain suppression properties at the cellular level. Fitoterapia 2016, 111, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Yang, T.; Ming, T.W.; Gaun, T.K.W.; Zhou, T.; Wang, S.; Ye, B. Isosteroid alkaloids from Fritillaria cirrhosa bulbs as inhibitors of cigarette smoke-induced oxidative stress. Fitoterapia 2020, 140, 104434. [Google Scholar] [CrossRef]
- Cunningham, A.B.; Brinckmann, J.A.; Pei, S.J.; Luo, P.; Schippmann, U.; Long, X.; Bi, Y.F. High altitude species, high profits: Can the trade in wild harvested Fritillaria cirrhosa (Liliaceae) be sustained? J. Ethnopharmacol. 2018, 223, 142–151. [Google Scholar] [CrossRef]
- Zhang, D.; Gao, L.; Yang, Y. Genetic diversity and structure of a traditional Chinese medicinal plant species, Fritillaria cirrhosa (Liliaceae) in southwest China and implications for its conservation. Biochem. Syst. Ecol. 2010, 38, 236–242. [Google Scholar] [CrossRef]
- Chang, H.C.; Xie, H.M.; Lee, M.R.; Lin, C.Y.; Yip, M.K.; Agrawal, D.C.; Tsay, H.S. In vitro propagation of bulblets and LC–MS/MS analysis of isosteroidal alkaloids in tissue culture derived materials of Chinese medicinal herb Fritillaria cirrhosa D. Don. Bot. Stud. 2020, 61, 9. [Google Scholar] [CrossRef]
- Batista, D.S.; Felipe, S.H.S.; Silva, T.D.; de Castro, K.M.; Mamedes-Rodrigues, T.C.; Miranda, N.A.; Ríos-Ríos, A.M.; Faria, D.V.; Fortini, E.A.; Chagas, K.; et al. Light quality in plant tissue culture: Does it matter? In Vitro Cell. Dev. Biol.-Plant 2018, 54, 195–215. [Google Scholar] [CrossRef]
- Kozai, T. Why LED lighting for urban agriculture? In LED Lighting for Urban Agriculture, 1st ed.; Kozai, T., Fujiwara, K., Runkle, E., Eds.; Springer: Singapore, 2016; pp. 3–18. [Google Scholar]
- Xue, Y.; Gu, H.L. Determination of peimine and peiminine in Fritillaria thunbergii by HPLC-ELSD. Acta Pharm. Sin. 2005, 40, 550–552. [Google Scholar]
- Ruan, X.; Yang, L.; Cui, W.X.; Zhang, M.X.; Li, Z.H.; Liu, B.; Wan, Q. Optimization of supercritical fluid extraction of total Alkaloids, Peimisine, Peimine and Peiminine from the Bulb of Fritillaria thunbergii Miq, and evaluation of antioxidant activities of the extracts. Materials 2016, 9, 524. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Mo, C.; Xiao, H.; Jiang, Y.; Ye, B.; Wang, S. Imperialine and verticinone from bulbs of Fritillaria wabuensis inhibit pro-inflammatory mediators in LPS stimulated RAW 264.7 macrophages. Planta Med. 2015, 81, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Zheng, B.; Jiang, B.; Xue, X.; Xue, E.; Zhou, Y. Peiminine inhibits the IL-1β induced inflammatory response in mouse articular chondrocytes and ameliorates murine osteoarthritis. Food Funct. 2019, 10, 2198–2208. [Google Scholar] [CrossRef]
- Wang, D.; Zhu, J.; Wang, S.; Wang, X.; Ou, Y.; Wei, D.; Li, X. Antitussive, expectorant and anti-inflammatory alkaloids from Bulbus Fritillariae Cirrhosae. Fitoterapia 2011, 82, 1290–1294. [Google Scholar] [CrossRef]
- Lin, Q.; Qu, M.; Patra, H.K.; He, S.; Wang, L.; Hua, X.; Xiao, L.; Fu, Y.; Gong, T.; He, Q.; et al. Mechanistic and therapeutic study of novel anti-tumor function of natural compound imperialine for treating non-small cell lung cancer. J. Ethnopharmacol. 2020, 247, 112283. [Google Scholar] [CrossRef]
- Yin, Z.; Zhang, J.; Guo, Q.; Chen, L.; Zhang, W.; Kang, W. Pharmacological effects of verticine: Current status. Evid. Based Complement. Altern. Med. 2019, 2019, 2394605. [Google Scholar] [CrossRef]
- Zhang, L.; Cui, M.; Chen, S. Identification of the molecular mechanisms of peimine in the treatment of cough using computational target fishing. Molecules 2020, 25, 1105. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco culture. Physiol. Plant 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Chen, C.C.; Agrawal, D.C.; Lee, M.R.; Lee, R.J.; Kuo, C.L.; Wu, C.R.; Tsay, H.S.; Chang, H.C. Influence of LED light spectra on in vitro somatic embryogenesis and LC–MS analysis of chlorogenic acid and rutin in Peucedanum japonicum Thunb.: A medicinal herb. Bot. Stud. 2016, 57, 9. [Google Scholar] [CrossRef]
- Hsiao, H.H.; Hsieh, H.Y.; Chou, C.C.; Lin, S.Y.; Wang, A.H.J.; Khoo, K.H. Concerted experimental approach for sequential mapping of peptides and phosphopeptides using C18-functionalized magnetic nanoparticles. J. Proteome Res. 2007, 6, 1313–1324. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Liu, H.; Diao, J.; Sun, Y.; Wang, Y. Magnetic molecularly imprinted polymer nanoparticles for separating aromatic amines from azo dyes–Synthesis, characterization and application. Sep. Purif. Technol. 2018, 204, 213–219. [Google Scholar] [CrossRef]
- Agrawal, D.C.; Chang, H.C.; Chen, C.C.; Kuo, C.L.; Tsay, H.S. Biotechnology of medicinal plants and fungi in Taiwan—Production of bioactive secondary metabolites in vitro culture systems. In Medicinal Plants and Fungi: Recent Advances in Research and Development, 1st ed.; Agrawal, D.C., Chang, H.C., Chen, C.C., Kuo, C.L., Tsay, H.S., Eds.; Springer Nature: Singapore, 2017; pp. 459–483. [Google Scholar]
- Wu, C.T.; Vanisree, M.; Satish, M.N.; Chen, C.L.; Yang, T.F.; Tsay, H.S. Isolation and quantitative analysis of cryptotanshinone, an active quinoid diterpene formed in the callus of Salvia miltiorrhiza Bunge. Biol. Pharm. Bull. 2003, 26, 845–848. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.L.; Agrawal, D.C.; Chang, H.C.; Chiu, Y.T.; Huang, C.P.; Chen, Y.L.; Huang, S.H.; Tsay, H.S. In vitro culture and production of syringin and rutin in Saussurea involucrata (Kar. et Kir.)—An endangered medicinal plant. Bot. Stud. 2015, 56, 12. [Google Scholar] [CrossRef]
- Efferth, T. Biotechnology applications of plant callus cultures. Engineering 2019, 5, 50–59. [Google Scholar] [CrossRef]
- Higuchi, Y.; Hisamatsu, T. Light acts as a signal for regulation of growth and development. In LED Lighting for Urban Agriculture, 1st ed.; Kozai, T., Fujiwara, K., Runkle, E., Eds.; Springer: Singapore, 2016; pp. 57–73. [Google Scholar]
- Kim, S.J.; Hahn, E.J.; Heo, J.W.; Paek, K.Y. Effects of LEDs on net photosynthetic rate, growth and leaf stomata of chrysanthemum plantlets in vitro. Sci. Hortic. 2004, 101, 143–151. [Google Scholar] [CrossRef]
- Li, H.M.; Xu, Z.G.; Tang, C.M. Effect of light-emitting diodes on growth and morphogenesis of upland cotton (Gossypium hirsutum L.) plantlets in vitro. Plant Cell Tissue Organ Cult. 2010, 103, 155–163. [Google Scholar] [CrossRef]
- Nhut, D.T.; Takamura, T.; Watanabe, H.; Okamoto, K.; Tanaka, M. Responses of strawberry plantlets cultured in vitro under super bright red and blue light-emitting diodes (LEDs). Plant Cell Tissue Organ Cult. 2003, 73, 43–52. [Google Scholar] [CrossRef]
- Saebo, A.; Krekling, T.; Appelgren, M. Light quality affects photosynthesis and leaf anatomy of birch plantlets in vitro. Plant Cell Tissue Organ Cult. 1995, 41, 177–185. [Google Scholar] [CrossRef]
- Olle, M.; Viršile, A. The effects of light-emitting diode lighting on greenhouse plant growth and quality. Agric. Food Sci. 2013, 22, 223–234. [Google Scholar] [CrossRef]
- Pedmale, U.V.; Huang, S.C.; Zander, M.; Cole, B.J.; Hetzel, J.; Ljung, K.; Reis, P.A.B.; Sridevi, P.; Nito, K.; Nery, J.R.; et al. Cryptochromes interact directly with PIFs to control plant growth in limiting blue light. Cell 2016, 164, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Briggs, W.R.; Olney, M.A. Photoreceptors in plant photomorphogenesis to date, five phytochromes, two cryptochromes, one phototropin, and one superchrome. Plant Physiol. 2001, 125, 85–88. [Google Scholar] [CrossRef] [PubMed]
- Briggs, W.R.; Beck, C.F.; Cashmore, A.R.; Christie, J.M.; Hughes, J.; Jarillo, J.A.; Kagawa, T.; Kanegae, H.; Liscum, E.; Nagatani, A.; et al. The phototropin family of photoreceptors. Plant Cell 2001, 13, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Clouse, S.D. Integration of light and brassinosteroid signals in etiolated seedling growth. Trends Plant Sci. 2001, 6, 443–445. [Google Scholar] [CrossRef]
- Chung, J.P.; Huang, C.Y.; Dai, T.E. Spectral effects on embryogenesis and plantlet growth of Oncidium ‘Gower Ramsey’. Sci. Hortic. 2010, 124, 511–516. [Google Scholar] [CrossRef]
- Agarwal, A.; Gupta, S.D. Impact of light-emitting diodes (LEDs) and its potential on plant growth and development in controlled environment plant production system. Curr. Biotechnol. 2016, 5, 28–43. [Google Scholar] [CrossRef]
- Bello-Bello, J.J.; Pérez-Sato, J.A.; Cruz-Cruz, C.A.; Martínez-Estrada, E. Light-emitting diodes: Progress in plant micropropagation. In Chlorophyll; Jacob-Lopes, E., Zepka, L.Q., Queiroz, M.I., Eds.; InTech: Rijeka, Croatia, 2017; pp. 93–103. [Google Scholar]
- Fang, W.; Chen, C.C.; Lee, Y.Y.; Chang, M.Y. Development of LED lids for tissue culture lighting. Acta Hortic. 2011, 907, 397–402. [Google Scholar] [CrossRef]
- Cepauskas, D.; Miliute, I.; Staniene, G.; Gelvonauskiene, D.; Stanys, V.; Jesaitis, A.J.; Baniulis, D. Characterization of apple NADPH oxidase genes and their expression associated with oxidative stress in shoot culture in vitro. Plant Cell Tissue Organ Cult. 2016, 124, 621–633. [Google Scholar] [CrossRef]
- Li, C.X.; Xu, Z.G.; Dong, R.Q.; Chang, S.X.; Wang, L.Z.; Khalil-Ur-Rehman, M.; Tao, J.M. An RNA-Seq analysis of grape plantlets grown in vitro reveals different responses to blue, green, red led light, and white fluorescent light. Front. Plant Sci. 2017, 8, 78. [Google Scholar] [CrossRef]
- Neelakandan, A.K.; Wang, K. Recent progress in the understanding of tissue culture-induced genome level changes in plants and potential applications. Plant Cell Rep. 2012, 31, 597–620. [Google Scholar] [CrossRef]
- Ruan, X.; Cui, W.X.; Yang, L.; Li, Z.H.; Liu, B.; Wang, Q. Extraction of total alkaloids, peimine and peiminine from the flower of Fritillaria thunbergii Miq using supercritical carbon dioxide. J. CO2 Util. 2017, 18, 283–293. [Google Scholar] [CrossRef]
- Konchar, K.; Li, X.L.; Yang, Y.P.; Emshwiller, E. Phytochemical variation in Fritillaria cirrhosa D. Don (Chuan Bei Mu) in relation to plant reproductive stage and timing of harvest. Econ. Bot. 2011, 65, 283. [Google Scholar] [CrossRef]

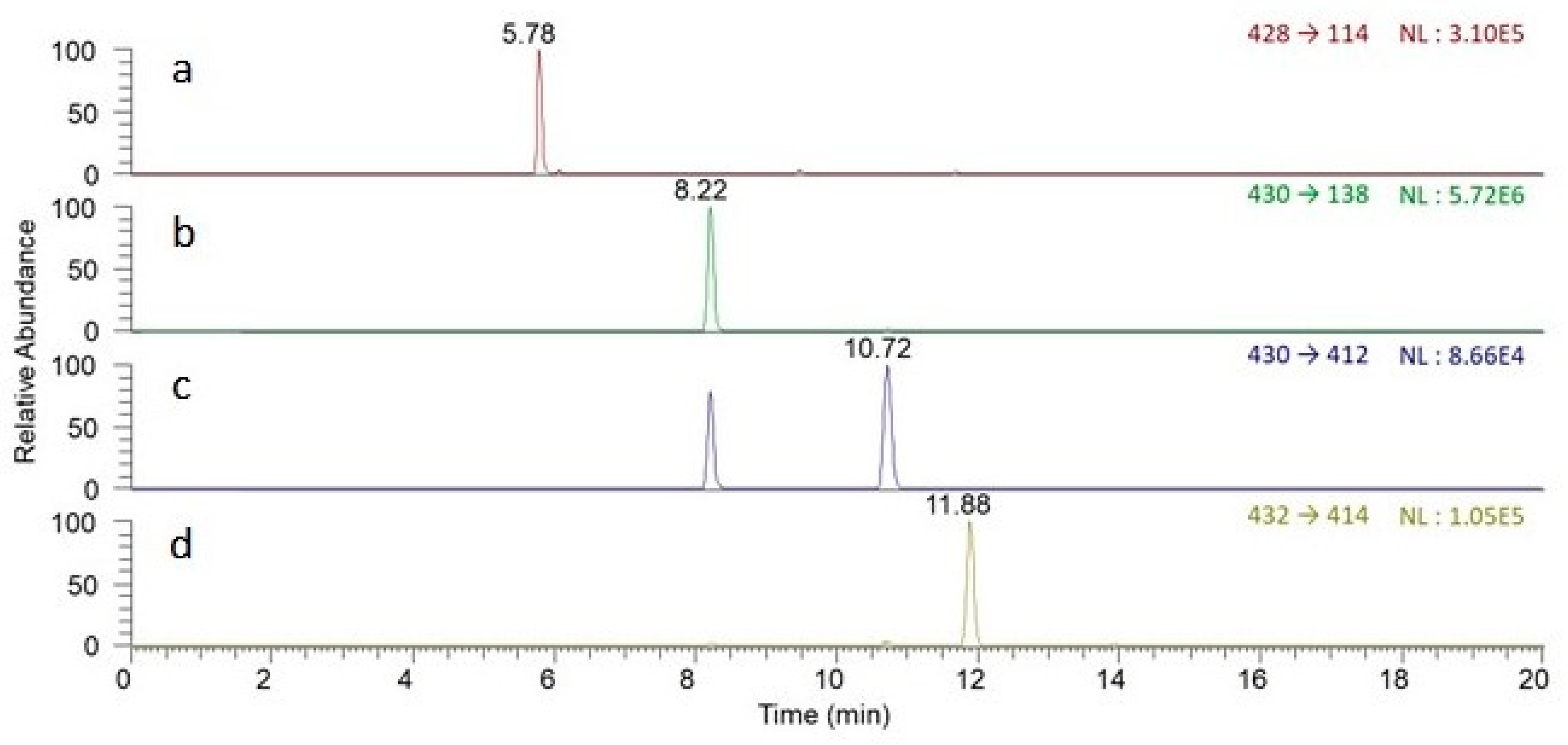



| Marker Compounds | tR a (min) | Mass/Charge (m/z) | Linearity and Range | Sensitivity c | |||
|---|---|---|---|---|---|---|---|
| Regression Equation b | Correlation Coefficient (r2) | Linear Range (μg/g) | LOD (ng/g) | LOQ (ng/g) | |||
| Peimisine | 5.78 | 428.316 | y = 0.0396x + 0.0630 | 0.9923 | 0.1–40 | 0.01 | 0.04 |
| Sipeimine | 8.22 | 430.332 | y = 0.5426x + 0.1531 | 0.9968 | 0.1–40 | 0.02 | 0.06 |
| Peiminine | 10.72 | 430.332 | y = 0.0047x + 0.0010 | 0.9967 | 0.1–40 | 0.97 | 3.23 |
| Peimine | 11.88 | 432.347 | y = 0.0067x + 0.0016 | 0.9977 | 0.1–40 | 0.56 | 1.88 |
| LED Light Treatment | Av No. of Somatic Embryos/Bottle | Av No. Of Somatic Embryos with Cotyledonary Leaves/Bottle | Av Total Fresh Weight of Cultures/Bottle (g) | Morphological Features of Cultures (Somatic Embryos = SEs) |
|---|---|---|---|---|
| CW-5000K * | 63.7 ± 7.6 c ** | 12.0 ± 6.0 abc | 12.82 ± 1.09 c | SEs at all stages from globular to mature, SEs with long cotyledonary leaves. Color of cultures is light green. |
| WW-2700K | 122.0 ± 65.0 bc | 22.3 ± 13.9 a | 13.64 ± 2.82 c | SE stages are similar to CW-5000K. Color of cultures is light green. |
| 7R1G1B | 108.0 ± 38.2 bc | 8.5 ± 3.5 bc | 12.99 ± 2.02 c | A majority of SEs in early stages, including globular shapes. Cotyledonary leaves absent. Color of cultures is light green to light brown. |
| 8R1B | 157.3 ± 9.3 ab | 16.7 ± 8.3 ab | 15.23 ± 0.97 abc | Stages similar to CW-5000K. Color of cultures is light green. |
| 9B | 169.0 ± 66.1 ab | 3.7 ± 2.3 c | 14.61 ± 1.02 bc | SE stages similar to CW-5000K, but cotyledonary leaves shorter in length. Color of cultures is light green to light yellow. |
| 9R | 223.7 ± 57.5 a | 5.3 ± 4.9 c | 17.92 ± 0.77 a | A majority of SEs in the early stages, including globular shapes. Cotyledonary leaves absent. Color of cultures is white. |
| 9IR | 231.3 ± 62.3 a | 4.7 ± 0.6 c | 16.67 ± 1.85 ab | SE stages and color of cultures similar to 9R. Only a few cotyledonary leaves are seen. Color of cultures is white. |
| 3R3B3IR | 230.7 ± 23.4 a | 4.3 ± 2.5 c | 17.56 ± 2.35 a | SE at all stages from globular to mature but cotyledonary leaves shorter in length. Color of cultures is dark green. |
| LED Light Treatment | Plant Material | Isosteroidal Alkaloids (µg/g/dw) | Total of Four Alkaloids (µg/g/dw) | |||
|---|---|---|---|---|---|---|
| Peimisine | Sipeimine | Peimine | Peiminine | |||
| CW-5000K | In vitro cultures | ND * | ND | ND | 0.12 ± 0.20 b ** | 0.12 ± 0.20 c ** |
| WW-2700K | In vitro cultures | ND | ND | ND | 0.28 ± 0.27 b | 0.28 ± 0.27 c |
| 7R1G1B | In vitro cultures | ND | ND | ND | 0.19 ± 0.32 b | 0.19 ± 0.32 c |
| 8R1B | In vitro cultures | ND | ND | ND | ND | 0.00 ± 0.00 c |
| 9B | In vitro cultures | ND | ND | ND | 0.60 ± 0.43 b | 0.65 ± 0.45 c |
| 9R | In vitro cultures | 3.65 ± 1.68 b ** | ND | 0.38 ± 0.11 a | 2.40 ± 0.30 a | 6.42 ± 2.06 b |
| 9IR | In vitro cultures | 3.22 ± 3.28 b | ND | 0.05 ± 0.09 b | 2.21 ± 0.87 a | 5.48 ± 3.21 b |
| 3R3B3IR | In vitro cultures | ND | ND | ND | 0.26 ± 0.24 b | 0.26 ± 0.24 c |
| Fluorescent tube | In vitro bulblets (3 months old) | 0.91 ± 0.97 b | ND | ND | 2.98 ± 1.09 a | 3.90 ± 1.51 bc |
| Natural habitat | Commercial bulbs (wild type, 3 years old) | 68.4 ± 7.8 a | 0.6 ± 0.4 a | ND | ND | 69.0 ± 7.4 a |
| Type of Somatic Embryo (SE) * | Percentage of Response (%) | Av No. of Bulblets/SE |
|---|---|---|
| Single embryo | 90.0 ± 10.0 a ** | 4.7 ± 1.3 a ** |
| Cluster of five embryos | 86.7 ± 12.0 a | 3.3 ± 1.5 ab |
| Embryo with the cotyledonary leaf | 43.6 ± 29.0 b | 1.1 ± 0.7 b |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, C.-C.; Lee, M.-R.; Wu, C.-R.; Ke, H.-J.; Xie, H.-M.; Tsay, H.-S.; Agrawal, D.C.; Chang, H.-C. LED Lights Affecting Morphogenesis and Isosteroidal Alkaloid Contents in Fritillaria cirrhosa D. Don—An Important Chinese Medicinal Herb. Plants 2020, 9, 1351. https://doi.org/10.3390/plants9101351
Chen C-C, Lee M-R, Wu C-R, Ke H-J, Xie H-M, Tsay H-S, Agrawal DC, Chang H-C. LED Lights Affecting Morphogenesis and Isosteroidal Alkaloid Contents in Fritillaria cirrhosa D. Don—An Important Chinese Medicinal Herb. Plants. 2020; 9(10):1351. https://doi.org/10.3390/plants9101351
Chicago/Turabian StyleChen, Chia-Chen, Maw-Rong Lee, Chi-Rei Wu, Hsin-Ju Ke, Hui-Min Xie, Hsin-Sheng Tsay, Dinesh Chandra Agrawal, and Hung-Chi Chang. 2020. "LED Lights Affecting Morphogenesis and Isosteroidal Alkaloid Contents in Fritillaria cirrhosa D. Don—An Important Chinese Medicinal Herb" Plants 9, no. 10: 1351. https://doi.org/10.3390/plants9101351
APA StyleChen, C.-C., Lee, M.-R., Wu, C.-R., Ke, H.-J., Xie, H.-M., Tsay, H.-S., Agrawal, D. C., & Chang, H.-C. (2020). LED Lights Affecting Morphogenesis and Isosteroidal Alkaloid Contents in Fritillaria cirrhosa D. Don—An Important Chinese Medicinal Herb. Plants, 9(10), 1351. https://doi.org/10.3390/plants9101351






