Regulation of Sulfate Uptake and Assimilation in Barley (Hordeum vulgare) as Affected by Rhizospheric and Atmospheric Sulfur Nutrition
Abstract
1. Introduction
2. Results and Discussion
2.1. Impact of Sulfate Deprivation and H2S Fumigation on Biomass Production and Mineral Nutrient Content
2.2. Impact of Sulfate Deprivation and H2S Fumigation on Sulfur and Nitrogen Metabolite Content
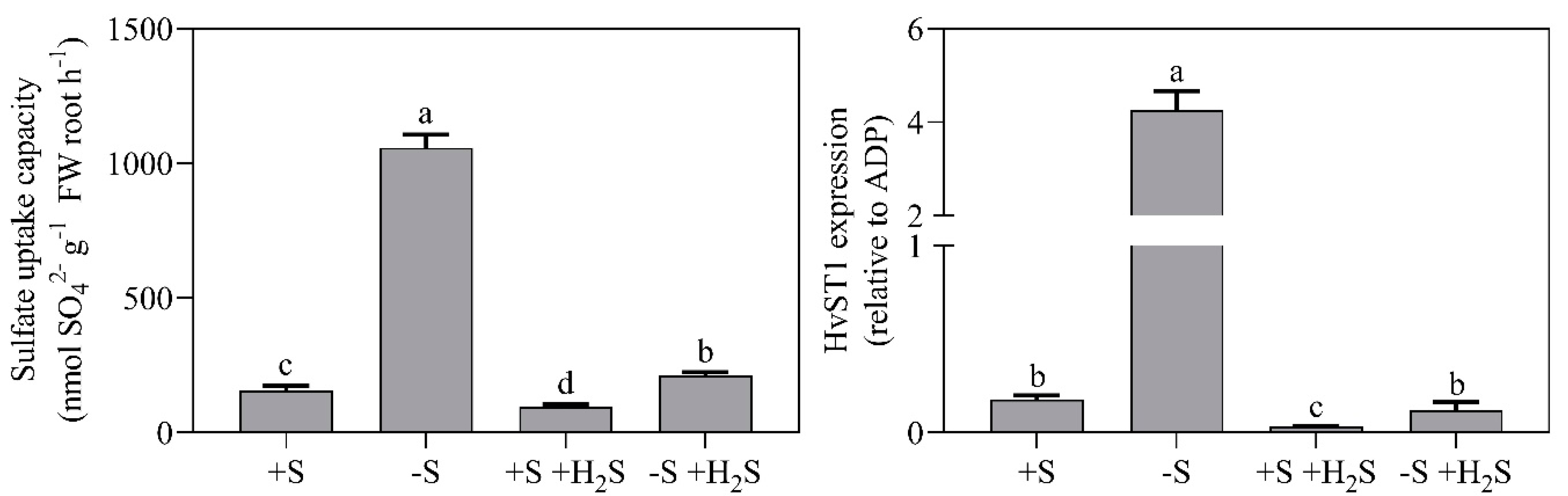
2.3. Impact of Sulfate Deprivation and H2S Fumigation on Sulfate Uptake and Reduction
2.4. Impact of Sulfate Deprivation and H2S Fumigation in Barley versus Other Species
3. Materials and Methods
3.1. Plant Material and H2S Fumigation
3.2. Mineral Nutrient Content
3.3. Sulfur and Nitrogen Metabolite Content
3.4. APR Activity and Sulfate Uptake Capacity
3.5. Expression of HvST1
3.6. Statistical Analyses
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhao, F.J.; Fortune, S.; Barbosa, V.L.; McGrath, S.P.; Stobart, R.; Bilsborrow, P.E.; Booth, E.J.; Brown, A.; Robson, P. Effects of sulphur on yield and malting quality of barley. J. Cereal Sci. 2006, 43, 369–377. [Google Scholar] [CrossRef]
- Holopainen, U.R.M.; Rajala, A.; Jauhiainen, L.; Wilhelmson, A.; Home, S.; Kaupilla, R.; Peltonen-Sainio, P. Influence of sulphur application on hordein composition and malting quality of barley (Hordeum vulgare L.) in Northern European growing conditions. J. Cereal Sci. 2015, 62, 151–158. [Google Scholar] [CrossRef]
- Hawkesford, M.J.; De Kok, L.J. Managing sulphur metabolism in plants. Plant Cell Environ. 2006, 29, 382–395. [Google Scholar] [CrossRef]
- Ausma, T.; De Kok, L.J. Atmospheric H2S: Impact on plant functioning. Front. Plant Sci. 2019, 10, 743. [Google Scholar] [CrossRef] [PubMed]
- Stulen, I.; Posthumus, F.S.; Amâncio, S.; Masselink-Beltman, I.; Müller, M.; De Kok, L.J. Mechanism of H2S phytotoxicity. In Sulfur Nutrition and Sulfur Assimilation in Higher Plants: Molecular, Biochemical and Physiological Aspects, 1st ed.; Brunold, C., Rennenberg, H., De Kok, L.J., Davidian, J.C., Eds.; Paul Haupt: Bern, Switzerland, 2000; pp. 381–382. [Google Scholar]
- Stuiver, C.E.E.; De Kok, L.J. Atmospheric H2S as sulphur source for sulphur deprived Brassica oleracea L. and Hordeum vulgare L. In Sulphur Metabolism in Higher Plants: Molecular, Ecophysiological and Nutritional Aspects, 1st ed.; Cram, W.J., De Kok, L.J., Stulen, I., Brunold, C., Rennenberg, H., Eds.; Backhuys Publishers: Leiden, The Netherlands, 1997; pp. 292–294. [Google Scholar]
- Ausma, T.; Parmar, S.; Hawkesford, M.J.; De Kok, L.J. Impact of atmospheric H2S, salinity and anoxia on sulfur metabolism in Zea mays. In Sulfur Metabolism in Higher Plants: Fundamental, Environmental and Agricultural Aspects, 1st ed.; De Kok, L.J., Hawkesford, M.J., Haneklaus, S.H., Schnug, E., Eds.; Springer: Dordrecht, The Netherlands, 2017; pp. 93–101. [Google Scholar]
- De Kok, L.J.; Stahl, K.; Rennenberg, H. Fluxes of atmospheric hydrogen sulfide to plant shoots. New Phytol. 1989, 112, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Zuidersma, E.I.; Ausma, T.; Stuiver, C.E.E.; Prajapati, D.H.; Hawkesford, M.J.; De Kok, L.J. Molybdate toxicity in Chinese cabbage is not the direct consequence of changes in sulphur metabolism. Plant Biol. 2020, 22, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Reich, M.; Shahbaz, M.; Prajapati, D.H.; Parmar, S.; Hawkesford, M.J.; De Kok, L.J. Interactions of sulfate with other nutrients as revealed by H2S fumigation of Chinese cabbage. Front. Plant Sci. 2016, 7, 541. [Google Scholar] [CrossRef] [PubMed]
- Smith, F.W.; Hawkesford, M.J.; Ealing, P.M.; Clarkson, D.T.; Van den Berg, P.J.; Belcher, A.R.; Warrilow, A.G.S. Regulation of expression of a cDNA from barley roots encoding a high affinity sulphate transporter. Plant J. 1999, 12, 875–884. [Google Scholar] [CrossRef] [PubMed]
- Vidmar, J.J.; Schjoerring, J.K.; Touraine, B.; Glass, A.D.M. Regulation of the hvst1 gene encoding a high-affinity sulfate transporter from Hordeum vulgare. Plant Mol. Biol. 1999, 40, 883–892. [Google Scholar] [CrossRef]
- Tausz, M.; Weidner, W.; Wonisch, A.; De Kok, L.J.; Grill, D. Uptake and distribution of 35S-sulfate in needles and roots of spruce seedlings as affected by exposure to SO2 and H2S. J. Exp. Bot. 2003, 50, 211–220. [Google Scholar] [CrossRef]
- Westerman, S.; De Kok, L.J.; Stuiver, C.E.E.; Stulen, I. Interaction between metabolism of atmospheric H2S in the shoot and sulfate uptake by the roots of curly kale (Brassica oleracea). Physiol. Plant 2000, 109, 443–449. [Google Scholar] [CrossRef]
- Buchner, P.; Stuiver, C.E.E.; Westerman, S.; Wirtz, M.; Hell, R.; Hawkesford, M.J.; De Kok, L.J. Regulation of sulfate uptake and expression of sulfate transporter genes in Brassica oleracea L. as affected by atmospheric H2S and pedospheric sulfate nutrition. Plant Physiol. 2004, 136, 3396–3408. [Google Scholar] [CrossRef] [PubMed]
- Koralewska, A.; Posthumus, F.S.; Stuiver, C.E.E.; Buchner, P.; De Kok, L.J. The characteristic high sulfate content in Brassica oleracea is controlled by the expression and activity of sulfate transporters. Plant Biol. 2007, 9, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Koralewska, A.; Stuiver, C.E.E.; Posthumus, F.S.; Kopriva, S.; Hawkesford, M.J.; De Kok, L.J. Regulation of sulfate uptake, expression of the sulfate transporters Sultr1;1 and Sultr1;2, and APS reductase in Chinese cabbage (Brassica pekinensis) as affected by atmospheric H2S nutrition and sulfate deprivation. Funct. Plant Biol. 2008, 35, 318–327. [Google Scholar] [CrossRef]
- Aghajanzadeh, T.; Hawkesford, M.J.; De Kok, L.J. Atmospheric H2S and SO2 as sulfur sources for Brassica juncea and Brassica rapa: Regulation of sulfur uptake and assimilation. Env. Exp. Bot. 2016, 124, 1–10. [Google Scholar] [CrossRef]
- Maas, F.M.; De Kok, L.J.; Hoffmann, I.; Kuiper, P.J.C. Plant responses to H2S and SO2 fumigation. I. Effects on growth, transpiration and sulfur content of spinach. Physiol. Plant 1987, 70, 713–721. [Google Scholar] [CrossRef]
- Maas, F.M.; De Kok, L.J.; Peters, J.L.; Kuiper, P.J.C. A comparative study on the effects of H2S and SO2 fumigation on the growth and accumulation of sulfate and sulfhydryl compounds in Trifolium pratense L., Glycine max Merr and Phaseolus vulgaris L. J. Exp. Bot. 1987, 38, 1459–1469. [Google Scholar] [CrossRef]
- Van Klink, R.; Van Laar-Wiersma, J.; Vorst, O.; Smit, C. Rewilding with large herbivores: Positive direct and delayed effects of carrion on plant and arthropod communities. PLoS ONE 2020, 15, e0226946. [Google Scholar] [CrossRef]
- Almario, J.; Jeena, G.; Wunder, J.; Langen, G.; Zucarro, A.; Coupland, G.; Buchner, M. Root-associated fungal microbiota of nonmycorrhizal Arabis alpina and its contribution to plant phosphorus nutrition. Proc. Natl. Acad. Sci. USA 2017, 114, 9403–9412. [Google Scholar] [CrossRef]
- De Kok, L.J.; Buwalda, F.; Bosma, W. Determination of cysteine and its accumulation in spinach leaf tissue upon exposure to excess sulfur. J. Plant Physiol. 1988, 133, 502–505. [Google Scholar] [CrossRef]
- Maas, F.M.; Hoffmann, I.; Van Harmelen, M.J.; De Kok, L.J. Refractometric determination of sulfate and anions in plants separated by high performance liquid chromatography. Plant Soil 1986, 91, 129–132. [Google Scholar] [CrossRef]
- Huang, X.Y.; Chao, D.Y.; Koprivova, A.; Danku, J.; Wirtz, M.; Muller, S.; Sandoval, F.J.; Bauwe, H.; Roje, S.; Dilkes, B. Nuclear localized MORE SULPHUR ACCUMULATION1 epigenetically regulates sulphur homeostasis in Arabidopsis thaliana. PLoS Genet. 2016, 12, e1006298. [Google Scholar] [CrossRef]
- Stuiver, C.E.E.; De Kok, L.J.; Westerman, S. Sulfur deficiency in Brassica oleracea L.: Development, biochemical characterization, and sulfur/nitrogen interactions. Russ. J. Plant Physiol. 1997, 44, 505–513. [Google Scholar]
- Durenkamp, M.; De Kok, L.J.; Kopriva, S. Adenosine 5’-phosphosulphate reductase is regulated differently in Allium cepa L. and Brassica oleracea L. upon exposure to H2S. J. Exp. Bot. 2007, 58, 1571–1579. [Google Scholar] [CrossRef] [PubMed]
- Ferdous, J.; Li, Y.; Reid, N.; Langridge, P.; Shi, B.J.; Tricker, P.J. Identification of reference genes for quantitative expression of microRNAs and mRNAs in barley under various stress conditions. PLoS ONE 2015, 10, e0118503. [Google Scholar] [CrossRef] [PubMed]
- Reid, R.; Gridley, K.; Kawamata, Y.; Zhu, Y. Arsenite elicits anomalous sulfur starvation responses in barley. Plant Physiol. 2013, 162, 401–409. [Google Scholar] [CrossRef]
- Ramakers, C.; Ruijter, J.M.; Deprez, R.H.; Moorman, A.F. Assumption-free analysis of quantitative real-time polymerase chain reaction (PCR) data. Neurosci. Lett. 2003, 339, 62–66. [Google Scholar] [CrossRef]
- Ruijter, J.M.; Ramakers, C.; Hoogaars, W.M.; Karlen, Y.; Bakker, O.; van den Hoff, M.J.; Moorman, A.F. Amplification efficiency: Linking baseline and bias in the analysis of quantitative PCR data. Nucleic Acids Res. 2009, 37, e45. [Google Scholar] [CrossRef]
- Dalla Benetta, E.; Beukeboom, L.W.; van de Zande, L. Adaptive differences in circadian clock gene expression patterns and photoperiodic diapause induction in Nasonia vitripennis. Am. Nat. 2019, 193, 881–896. [Google Scholar] [CrossRef]
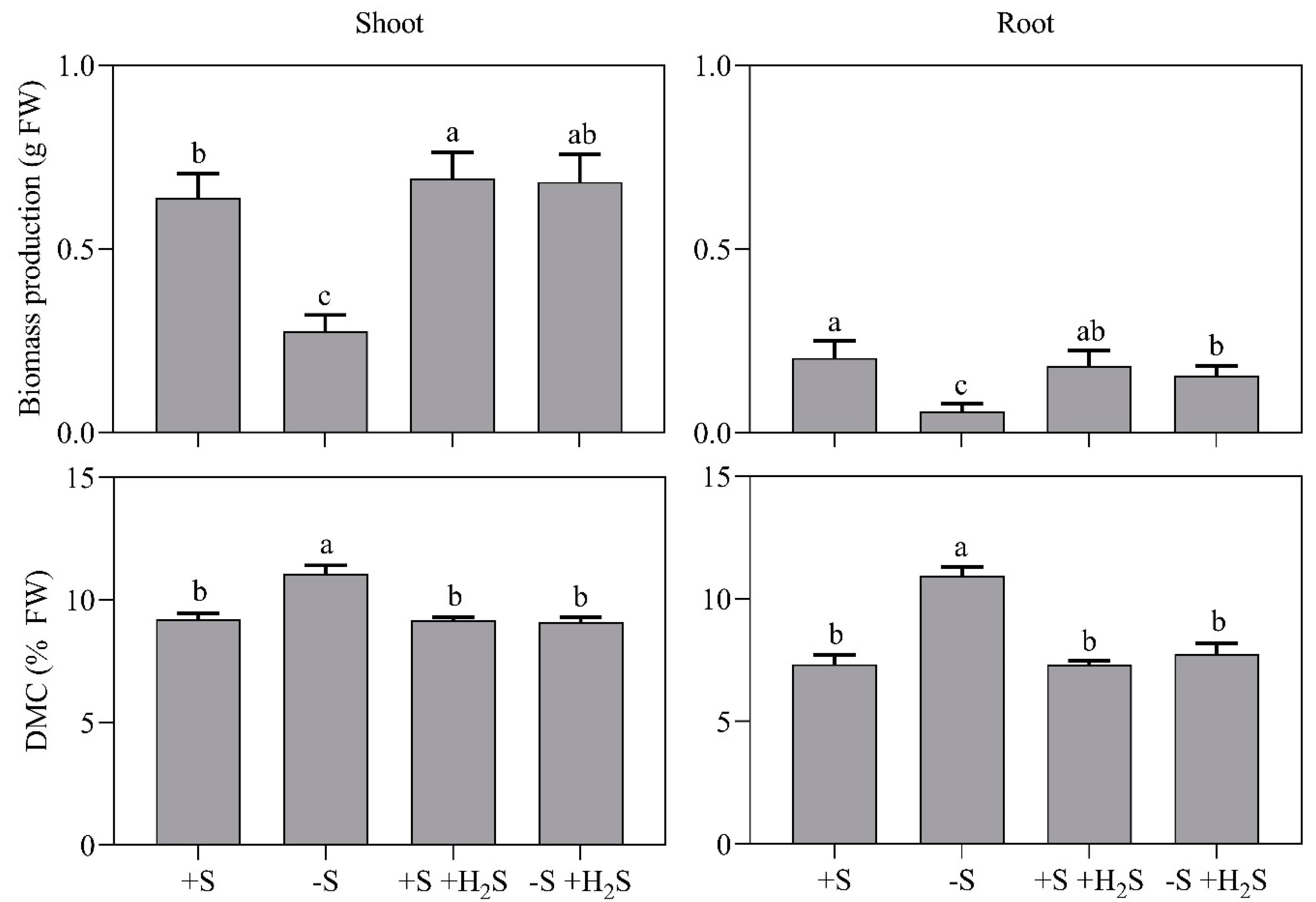
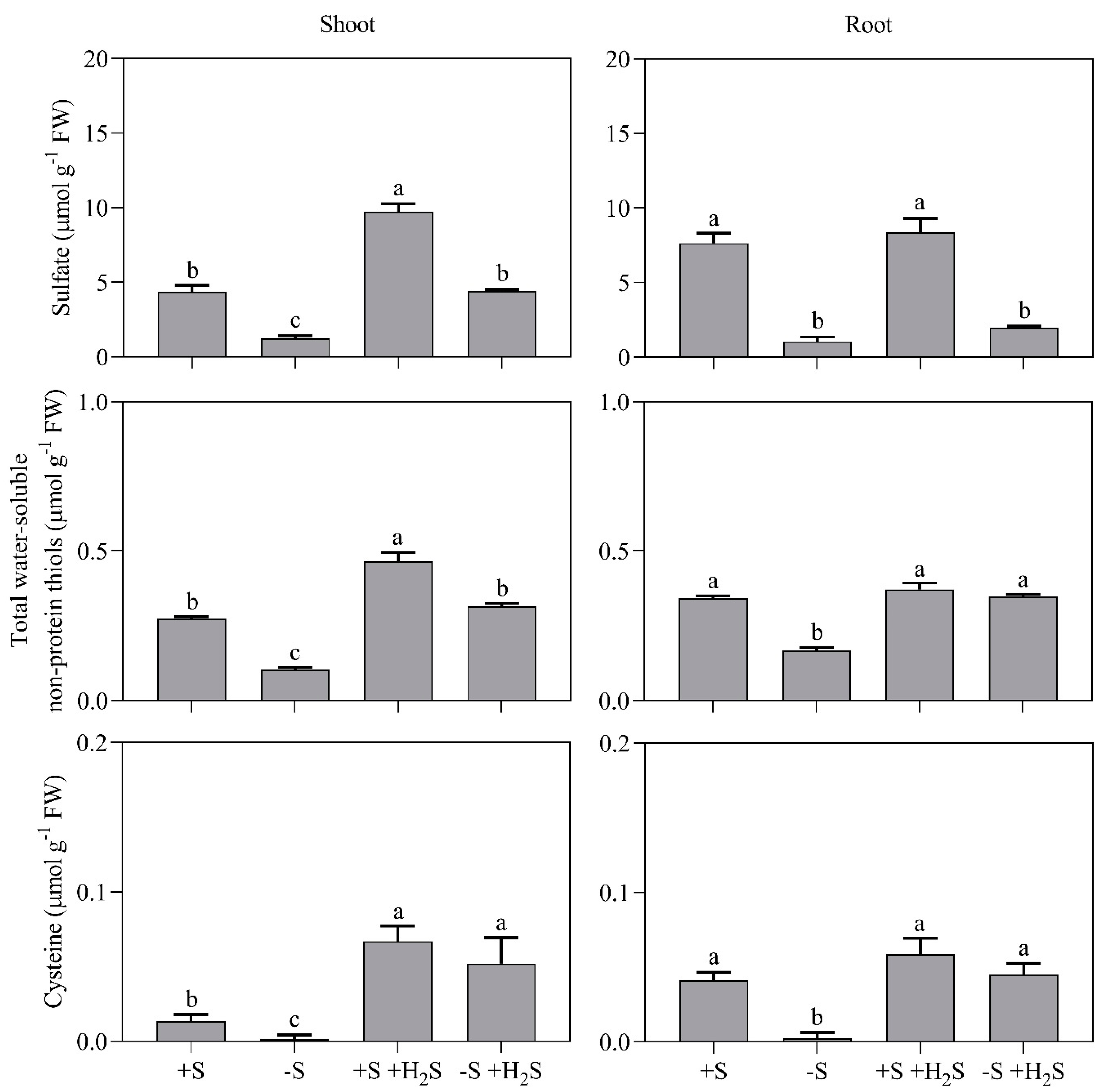
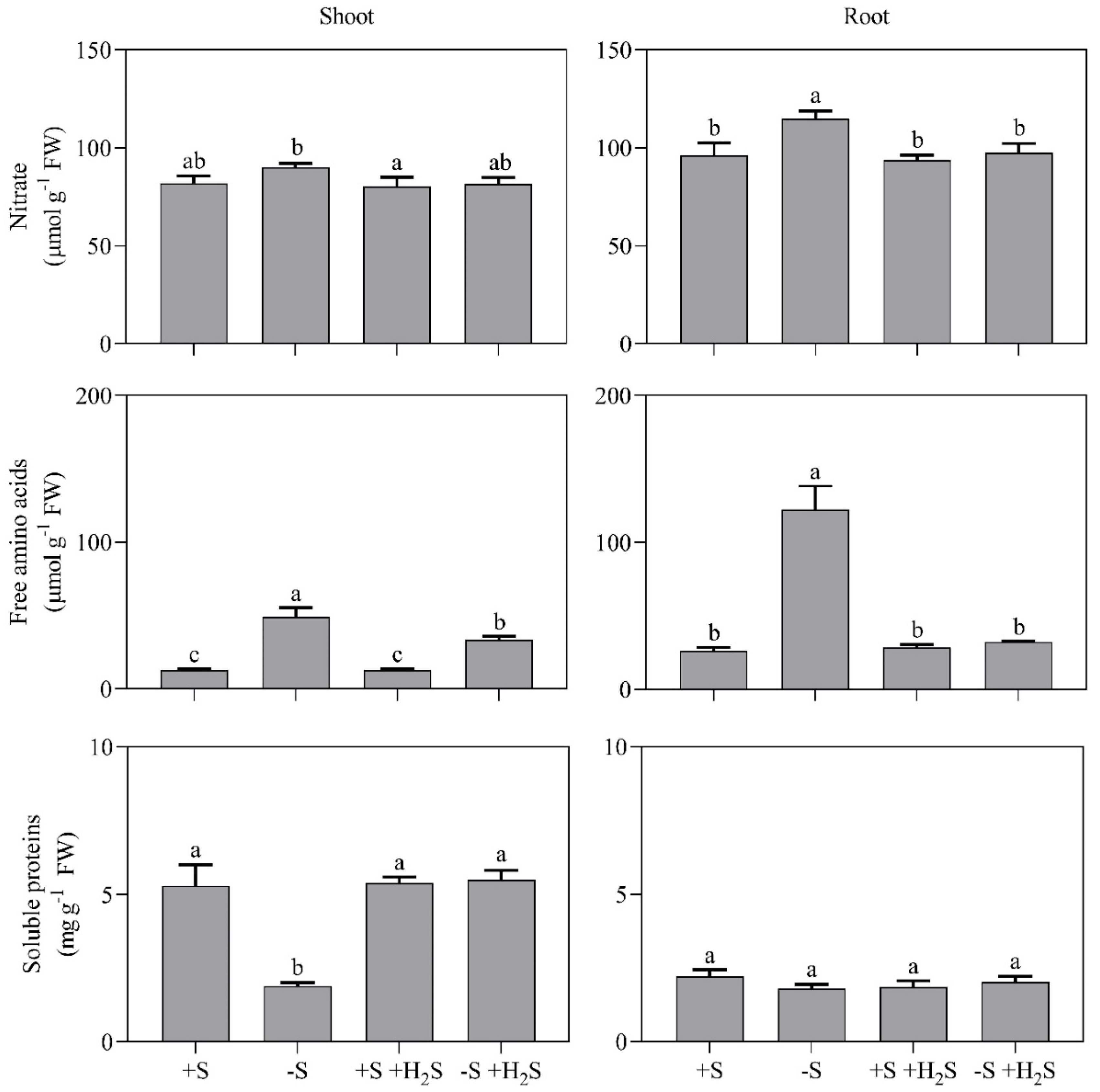
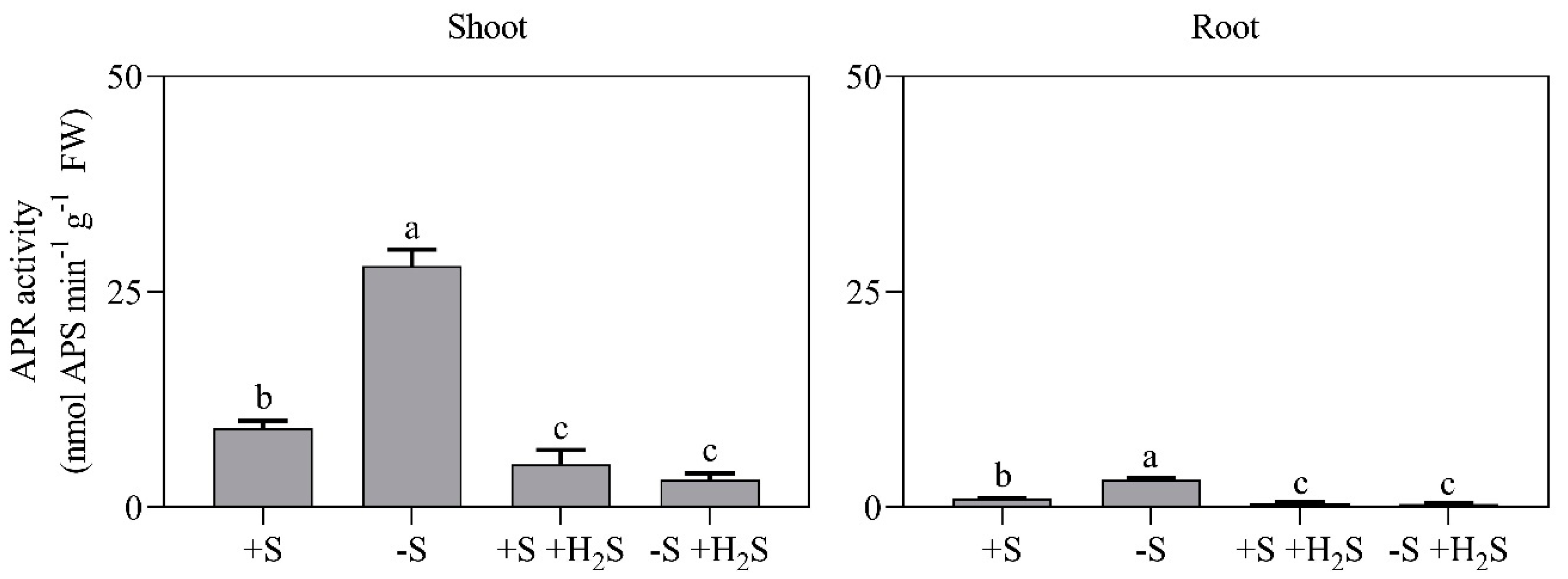
| Mineral Nutrient Content | 0 µL L−1 H2S | 0.6 µL L−1 H2S | ||
|---|---|---|---|---|
| (µmol g−1 DW) | +S | −S | +S | −S |
| Shoot | ||||
| Calcium | 103 ± 10 a | 84 ± 3 a | 100 ± 29 a | 81 ± 8.0 a |
| Copper | 0.29 ± 0.03 a | 0.25 ± 0.01 a | 0.29 ± 0.05 a | 0.25 ± 0.03 a |
| Iron | 1.92 ± 0.12 a | 1.65 ± 0.07 a | 2.06 ± 0.45 a | 1.72 ± 0.20 a |
| Magnesium | 81 ± 7 a | 76 ± 3 a | 88 ± 16 a | 71 ± 8 a |
| Manganese | 0.73 ± 0.06 ab | 0.66 ± 0.03 ab | 0.83 ± 0.14 a | 0.59 ± 0.05 b |
| Molybdenum | 0.04 ± 0.00 c | 0.25 ± 0.01 a | 0.04 ± 0.01 c | 0.19 ± 0.03 b |
| Nitrogen | 3521 ± 150 a | 3216 ± 61 b | 3412 ± 71 ab | 3418 ± 69 ab |
| Phosphorus | 192 ± 9 b | 250 ± 11 a | 204 ± 34 ab | 180 ± 22 b |
| Potassium | 1758 ± 79 a | 1637 ± 94 a | 1852 ± 205 a | 1570 ± 161 a |
| Sulfur | 99 ± 4 b | 36 ± 2 c | 163 ± 16 a | 100 ± 12 b |
| Zinc | 0.63 ± 0.07 ab | 0.77 ± 0.05 a | 0.67 ± 0.11 ab | 0.50 ± 0.03 b |
| Root | ||||
| Calcium | 26 ± 6 a | 38 ± 4 a | 27 ± 3 a | 21 ± 1 a |
| Copper | 2.05 ± 0.43 a | 1.13 ± 0.02 b | 1.83 ± 0.15 a | 1.45 ± 0.11 ab |
| Iron | 2.92 ± 1.14 a | 3.50 ± 0.46 a | 2.34 ± 0.05 a | 2.78 ± 0.12 a |
| Magnesium | 28 ± 6 a | 31 ± 3 a | 30 ± 6 a | 25 ± 1 a |
| Manganese | 2.21 ± 0.37 a | 1.17 ± 0.08 b | 2.36 ± 0.21 a | 1.34 ± 0.11 b |
| Molybdenum | 0.14 ± 0.02 b | 0.42 ± 0.07 a | 0.17 ± 0.03 b | 0.18 ± 0.01 b |
| Nitrogen | 3231 ± 133 a | 2917 ± 114 a | 3174 ± 182 a | 3152 ± 121 a |
| Phosphorus | 149 ± 21 a | 100 ± 8 b | 153 ± 12 a | 134 ± 16 ab |
| Potassium | 842 ± 103 a | 662 ± 21 b | 844 ± 42 a | 719 ± 56 ab |
| Sulfur | 82 ± 8 a | 28 ± 0 b | 94 ± 9 a | 44 ± 4 b |
| Zinc | 0.59 ± 0.12 a | 0.40 ± 0.05 b | 0.57 ± 0.04 ab | 0.47 ± 0.03 ab |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ausma, T.; De Kok, L.J. Regulation of Sulfate Uptake and Assimilation in Barley (Hordeum vulgare) as Affected by Rhizospheric and Atmospheric Sulfur Nutrition. Plants 2020, 9, 1283. https://doi.org/10.3390/plants9101283
Ausma T, De Kok LJ. Regulation of Sulfate Uptake and Assimilation in Barley (Hordeum vulgare) as Affected by Rhizospheric and Atmospheric Sulfur Nutrition. Plants. 2020; 9(10):1283. https://doi.org/10.3390/plants9101283
Chicago/Turabian StyleAusma, Ties, and Luit J. De Kok. 2020. "Regulation of Sulfate Uptake and Assimilation in Barley (Hordeum vulgare) as Affected by Rhizospheric and Atmospheric Sulfur Nutrition" Plants 9, no. 10: 1283. https://doi.org/10.3390/plants9101283
APA StyleAusma, T., & De Kok, L. J. (2020). Regulation of Sulfate Uptake and Assimilation in Barley (Hordeum vulgare) as Affected by Rhizospheric and Atmospheric Sulfur Nutrition. Plants, 9(10), 1283. https://doi.org/10.3390/plants9101283





