Mitochondrial Genome of Fagus sylvatica L. as a Source for Taxonomic Marker Development in the Fagales
Abstract
1. Introduction
2. Results
2.1. Assembly and Annotation of the DNA Sequence of the Complete Mitochondrial Genome of F. sylvatica L.
2.2. Identification of Potentially Taxon-Specific SNPs in Mitochondrial Genes
2.3. Development of Selected CAPS Markers and Further Validation of Their Taxon Specificity
3. Discussion
4. Material and Methods
4.1. Plant Material
4.2. DNA Sequencing of F. sylvatica L.
4.3. RNA Sequencing of Two F. sylvatica L. Individuals
4.4. Assembly and Scaffolding of the Illumina MiSeq Reads from DNA Sequencing
4.5. Mapping of Nanopore MinION Reads to the DNA Sequence of the Mitochondrial Genome of F. sylvatica
4.6. Assembly of the RNA-Seq Data
4.7. Annotation of the DNA Sequence of the Mitochondrial Genome of F. sylvatica
4.8. Multiple Alignments of Mitochondrial Gene Sequences and Selection of SNPs Specific for Pre-Defined Taxa
4.9. DNA Preparation, PCR Amplification, and Agarose Gel Electrophoresis for CAPS Markers
4.10. Validation of the CAPS Markers
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Renner, S.S.; Grimm, G.W.; Kapli, P.; Denk, T. Species relationships and divergence times in beeches: New insights from the inclusion of 53 young and old fossils in a birth-death clock model. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371, 20150135. [Google Scholar] [CrossRef] [PubMed]
- The Plant List. Available online: http://www.theplantlist.org/ (accessed on 1 July 2020).
- Mishra, B.; Gupta, D.K.; Pfenninger, M.; Hickler, T.; Langer, E.; Nam, B.; Paule, J.; Sharma, R.; Ulaszewski, B.; Warmbier, J.; et al. A reference genome of the European beech (Fagus sylvatica L.). Gigascience 2018, 7, giy063. [Google Scholar] [CrossRef] [PubMed]
- Mader, M.; Liesebach, H.; Liesebach, M.; Kersten, B. The complete chloroplast genome sequence of Fagus sylvatica L. (Fagaceae). Mitochondrial DNA Part B 2019, 4, 1818–1819. [Google Scholar] [CrossRef]
- Yang, Y.; Zhu, J.; Feng, L.; Zhou, T.; Bai, G.; Yang, J.; Zhao, G. Plastid Genome Comparative and Phylogenetic Analyses of the Key Genera in Fagaceae: Highlighting the Effect of Codon Composition Bias in Phylogenetic Inference. Front. Plant Sci. 2018, 9, 82. [Google Scholar] [CrossRef]
- Worth, J.R.P.; Liu, L.; Wei, F.J.; Tomaru, N. The complete chloroplast genome of Fagus crenata (subgenus Fagus) and comparison with F. engleriana (subgenus Engleriana). PeerJ 2019, 7, e7026. [Google Scholar] [CrossRef]
- Park, J.S.; Jin, D.P.; Park, J.W.; Choi, B.H. Complete chloroplast genome of Fagus multinervis, a beech species endemic to Ulleung Island in South Korea. Mitochondrial DNA Part B 2019, 4, 1698–1699. [Google Scholar] [CrossRef]
- Organelle Genome Resources at NCBI. Available online: http://www.ncbi.nlm.nih.gov/genome/organelle/ (accessed on 1 July 2020).
- Bi, Q.X.; Li, D.X.; Zhao, Y.; Wang, M.K.; Li, Y.C.; Liu, X.J.; Wang, L.B.; Yu, H.Y. Complete mitochondrial genome of Quercus. variabilis. (Fagales, Fagaceae). Mitochondrial DNA Part B 2019, 4, 3927–3928. [Google Scholar] [CrossRef]
- Salojarvi, J.; Smolander, O.P.; Nieminen, K.; Rajaraman, S.; Safronov, O.; Safdari, P.; Lamminmaki, A.; Immanen, J.; Lan, T.Y.; Tanskanen, J.; et al. Genome sequencing and population genomic analyses provide insights into the adaptive landscape of silver birch. Nat. Genet. 2017, 49, 904–912. [Google Scholar] [CrossRef]
- Gualberto, J.M.; Newton, K.J. Plant Mitochondrial Genomes: Dynamics and Mechanisms of Mutation. Annu. Rev. Plant Biol. 2017, 68, 225–252. [Google Scholar] [CrossRef]
- Morley, S.A.; Nielsen, B.L. Plant mitochondrial DNA. Front. Biosci. Landmrk 2017, 22, 1023–1032. [Google Scholar]
- Ward, B.L.; Anderson, R.S.; Bendich, A.J. The Mitochondrial Genome Is Large and Variable in a Family of Plants (Cucurbitaceae). Cell 1981, 25, 793–803. [Google Scholar] [CrossRef]
- Bendich, A.J. Structural analysis of mitochondrial DNA molecules from fungi and plants using moving pictures and pulsed-field gel electrophoresis. J. Mol. Biol. 1996, 255, 564–588. [Google Scholar] [CrossRef] [PubMed]
- Oldenburg, D.J.; Bendich, A.J. DNA maintenance in plastids and mitochondria of plants. Front. Plant Sci. 2015, 6, 883. [Google Scholar] [CrossRef]
- Bendich, A.J. The size and form of chromosomes are constant in the nucleus, but highly variable in bacteria, mitochondria and chloroplasts. BioEssays 2007, 29, 474–483. [Google Scholar] [CrossRef]
- Palmer, J.D.; Herbon, L.A. Plant Mitochondrial-DNA Evolves Rapidly in Structure, but Slowly in Sequence. J. Mol. Evol. 1988, 28, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.O.; Rice, D.W.; Young, G.J.; Alverson, A.J.; Palmer, J.D. The “fossilized” mitochondrial genome of Liriodendron tulipifera: Ancestral gene content and order, ancestral editing sites, and extraordinarily low mutation rate. BMC Biol. 2013, 11, 29. [Google Scholar] [CrossRef] [PubMed]
- Wynn, E.L.; Christensen, A.C. Repeats of Unusual Size in Plant Mitochondrial Genomes: Identification, Incidence and Evolution. G3-Genes Genom. Genet. 2019, 9, 549–559. [Google Scholar] [CrossRef]
- Drouin, G.; Daoud, H.; Xia, J. Relative rates of synonymous substitutions in the mitochondrial, chloroplast and nuclear genomes of seed plants. Mol. Phylogenet. Evol. 2008, 49, 827–831. [Google Scholar] [CrossRef]
- Wolfe, K.H.; Li, W.H.; Sharp, P.M. Rates of Nucleotide Substitution Vary Greatly among Plant Mitochondrial, Chloroplast, and Nuclear Dnas. Proc. Natl. Acad. Sci. USA 1987, 84, 9054–9058. [Google Scholar] [CrossRef]
- Schuster, W.; Brennicke, A. The Plant Mitochondrial Genome-Physical Structure, Information-Content, Rna Editing, and Gene Migration to the Nucleus. Annu. Rev. Plant. Phys. 1994, 45, 61–78. [Google Scholar] [CrossRef]
- Bullerwell, C.E.; Gray, M.W. Evolution of the mitochondrial genome: Protist connections to animals, fungi and plants. Curr. Opin. Microbiol. 2004, 7, 528–534. [Google Scholar] [CrossRef] [PubMed]
- Bock, R. The give-and-take of DNA: Horizontal gene transfer in plants. Trends. Plant Sci. 2010, 15, 11–22. [Google Scholar] [CrossRef] [PubMed]
- Kleine, T.; Maier, U.G.; Leister, D. DNA transfer from organelles to the nucleus: The idiosyncratic genetics of endosymbiosis. Annu. Rev. Plant Biol. 2009, 60, 115–138. [Google Scholar] [CrossRef] [PubMed]
- Kozik, A.; Rowan, B.A.; Lavelle, D.; Berke, L.; Schranz, M.E.; Michelmore, R.W.; Christensen, A.C. The alternative reality of plant mitochondrial DNA: One ring does not rule them all. PLoS Genet. 2019, 15, e1008373. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Chen, L.; Liu, Y.; Wang, Y.; Zhang, S.; Yang, L.; Lang, X.; Zhang, S. The draft mitochondrial genome of Magnolia biondii and mitochondrial phylogenomics of angiosperms. PLoS ONE 2020, 15, e0231020. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.L.; Palmer, J.D. Evolution of mitochondrial gene content: Gene loss and transfer to the nucleus. Mol. Phylogenet. Evol. 2003, 29, 380–395. [Google Scholar] [CrossRef]
- Tsujimura, M.; Kaneko, T.; Sakamoto, T.; Kimura, S.; Shigyo, M.; Yamagishi, H.; Terachi, T. Multichromosomal structure of the onion mitochondrial genome and a transcript analysis. Mitochondrion 2019, 46, 179–186. [Google Scholar] [CrossRef]
- Backert, S.; Nielsen, B.L.; Borner, T. The mystery of the rings: Structure and replication of mitochondrial genomes from higher plants. Trends. Plant Sci. 1997, 2, 477–483. [Google Scholar] [CrossRef]
- Backert, S.; Borner, T. Phage T4-like intermediates of DNA replication and recombination in the mitochondria of the higher plant Chenopodium album (L.). Curr. Genet. 2000, 37, 304–314. [Google Scholar] [CrossRef]
- Sloan, D.B.; Alverson, A.J.; Chuckalovcak, J.P.; Wu, M.; McCauley, D.E.; Palmer, J.D.; Taylor, D.R. Rapid Evolution of Enormous, Multichromosomal Genomes in Flowering Plant Mitochondria with Exceptionally High Mutation Rates. PLoS Biol. 2012, 10, e1001241. [Google Scholar] [CrossRef]
- Jacobs, M.A.; Payne, S.R.; Bendich, A.J. Moving pictures and pulsed-field gel electrophoresis show only linear mitochondrial DNA molecules from yeasts with linear-mapping and circular-mapping mitochondrial genomes. Curr. Genet. 1996, 30, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Hammani, K.; Giege, P. RNA metabolism in plant mitochondria. Trends. Plant Sci. 2014, 19, 380–389. [Google Scholar] [CrossRef] [PubMed]
- Barcode of Life. Available online: http://www.barcodinglife.org/ (accessed on 1 July 2020).
- Hollingsworth, P.M.; Forrest, L.L.; Spouge, J.L.; Hajibabaei, M.; Ratnasingham, S.; van der Bank, M.; Chase, M.W.; Cowan, R.S.; Erickson, D.L.; Fazekas, A.J.; et al. A DNA barcode for land plants. Proc. Natl. Acad. Sci. USA 2009, 106, 12794–12797. [Google Scholar]
- Wang, Z.; Du, S.; Dayanandan, S.; Wang, D.; Zeng, Y.; Zhang, J. Phylogeny reconstruction and hybrid analysis of Populus. (Salicaceae) based on nucleotide sequences of multiple single-copy nuclear genes and plastid fragments. PLoS ONE 2014, 9, e103645. [Google Scholar] [CrossRef]
- Mader, M.; Pakull, B.; Blanc-Jolivet, C.; Paulini-Drewes, M.; Bouda, Z.H.; Degen, B.; Small, I.; Kersten, B. Complete Chloroplast Genome Sequences of Four Meliaceae Species and Comparative Analyses. Int. J. Mol. Sci. 2018, 19, 701. [Google Scholar] [CrossRef]
- Fladung, M.; Schroeder, H.; Wehenkel, C.; Kersten, B. Differentiation of six Eucalyptus trees grown in Mexico by ITS and six chloroplast barcoding markers. Silvae Genet. 2015, 64, 121–130. [Google Scholar] [CrossRef][Green Version]
- Birky, C.W. Uniparental inheritance of mitochondrial and chloroplast genes-Mechanisms and evolution. Proc. Natl. Acad. Sci. USA 1995, 92, 11331–11338. [Google Scholar] [CrossRef]
- Ahmad, N.; Nielsen, B.L. Plant Organelle DNA Maintenance. Plants 2020, 9, 683. [Google Scholar] [CrossRef]
- Morley, S.A.; Ahmad, N.; Nielsen, B.L. Plant Organelle Genome Replication. Plants 2019, 8, 358. [Google Scholar] [CrossRef]
- Oldenburg, D.J.; Kumar, R.A.; Bendich, A.J. The amount and integrity of mtDNA in maize decline with development. Planta 2013, 237, 603–617. [Google Scholar] [CrossRef]
- Kersten, B.; Rampant, P.F.; Mader, M.; Le Paslier, M.C.; Bounon, R.; Berard, A.; Vettori, C.; Schroeder, H.; Leple, J.C.; Fladung, M. Genome Sequences of Populus. tremula. Chloroplast and Mitochondrion: Implications for Holistic Poplar Breeding. PLoS ONE 2016, 11, e0147209. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Yang, Y.; Henry, R.J.; Rossetto, M.; Wang, Y.; Chen, S. Plant DNA barcoding: From gene to genome. Biol. Rev. Camb. Philos. Soc. 2015, 90, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Nevill, P.G.; Zhong, X.; Tonti-Filippini, J.; Byrne, M.; Hislop, M.; Thiele, K.; van Leeuwen, S.; Boykin, L.M.; Small, I. Large scale genome skimming from herbarium material for accurate plant identification and phylogenomics. Plant Methods 2020, 16, 1. [Google Scholar] [CrossRef] [PubMed]
- Saarela, J.M.; Burke, S.V.; Wysocki, W.P.; Barrett, M.D.; Clark, L.G.; Craine, J.M.; Peterson, P.M.; Soreng, R.J.; Vorontsova, M.S.; Duvall, M.R. A 250 plastome phylogeny of the grass family (Poaceae): Topological support under different data partitions. PeerJ 2018, 6, e4299. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, J.N.; Chaluvadi, S.R.; Rangan, L.; Bennetzen, J.L. Whole Plastome Sequences from Five Ginger Species Facilitate Marker Development and Define Limits to Barcode Methodology. PLoS ONE 2014, 9, e108581. [Google Scholar] [CrossRef]
- Kersten, B.; Voss, M.; Fladung, M. Development of mitochondrial SNP markers to differentiate Populus species. Trees–Struct. Funct. 2015, 29, 575–582. [Google Scholar] [CrossRef]
- Koch, G.; Haag, V.; Heinz, I. Pflichtbewusstsein: EUTR und Holzwerkstoffe—Prüfungen am Thünen-Kompetenzzentrum Holzherkünfte. MDF. Mag. Co. 2015, 2015, 64–67. [Google Scholar]
- EU. European Timber Regulation Verordnung (EU) Nr. 995/2010 des Europäischen Parlaments und des Rates vom 20; Oktober 2010 über die Verpflichtungen von Marktteilnehmern, die Holz und Holzerzeugnisse in Verkehr bringen; EU: Brussels, Belgium, 2010. [Google Scholar]
- The IUCN Red List of Threatened Species. Available online: http://www.iucnredlist.org/ (accessed on 3 July 2020).
- Knorn, J.; Kuemmerle, T.; Radeloff, V.C.; Keeton, W.S.; Gancz, V.; Biris, I.A.; Svoboda, M.; Griffiths, P.; Hagatis, A.; Hostert, P. Continued loss of temperate old-growth forests in the Romanian Carpathians despite an increasing protected area network. Environ. Conserv. 2013, 40, 182–193. [Google Scholar] [CrossRef]
- Liesebach, H.; Eusemann, P.; Liesebach, M. Verwandtschaftsbeziehungen innerhalb von Prüfgliedern in Herkunftsversuchen-Beispiel Buche (Fagus sylvatica L.) [Sibship structure in samples from a provenance trial. A case study in beech (Fagus sylvatica L.)]. Forstarchiv 2015, 86, 174–182. [Google Scholar]
- Greiner, S.; Lehwark, P.; Bock, R. OrganellarGenomeDRAW (OGDRAW) version 1.3.1: Expanded toolkit for the graphical visualization of organellar genomes. Nucleic Acids Res. 2019, 47, W59–W64. [Google Scholar] [CrossRef]
- OrganellarGenomeDRAW. Available online: http://ogdraw.mpimp-golm.mpg.de/ (accessed on 3 July 2020).
- Lloyd Evans, D.; Hlongwane, T.T.; Joshi, S.V.; Riano Pachon, D.M. The sugarcane mitochondrial genome: Assembly, phylogenetics and transcriptomics. PeerJ 2019, 7, e7558. [Google Scholar] [CrossRef] [PubMed]
- Zascavage, R.R.; Hall, C.L.; Thorson, K.; Mahmoud, M.; Sedlazeck, F.J.; Planz, J.V. Approaches to Whole Mitochondrial Genome Sequencing on the Oxford Nanopore MinION. Curr. Protoc. Hum. Genet. 2019, 104, e94. [Google Scholar] [CrossRef] [PubMed]
- Frommer, B.; Holtgräwe, D.; Hausmann, L.; Viehöver, P.; Huettel, B.; Töpfer, R.; Weisshaar, B. Genome sequences of both organelles of the grapevine rootstock cultivar ‘Börner’. Microbiol. Resour. Announc. 2020, 9, e01471-19. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.L.; Daley, D.O.; Whelan, J.; Palmer, J.D. Genes for two mitochondrial ribosomal proteins in flowering plants are derived from their chloroplast or cytosolic counterparts. Plant Cell 2002, 14, 931–943. [Google Scholar] [CrossRef]
- Rivarola, M.; Foster, J.T.; Chan, A.P.; Williams, A.L.; Rice, D.W.; Liu, X.; Melake-Berhan, A.; Huot Creasy, H.; Puiu, D.; Rosovitz, M.J.; et al. Castor bean organelle genome sequencing and worldwide genetic diversity analysis. PLoS ONE 2011, 6, e21743. [Google Scholar] [CrossRef]
- Shearman, J.R.; Sangsrakru, D.; Ruang-Areerate, P.; Sonthirod, C.; Uthaipaisanwong, P.; Yoocha, T.; Poopear, S.; Theerawattanasuk, K.; Tragoonrung, S.; Tangphatsornruang, S. Assembly and analysis of a male sterile rubber tree mitochondrial genome reveals DNA rearrangement events and a novel transcript. BMC Plant Biol. 2014, 14, 45. [Google Scholar] [CrossRef]
- Choi, C.; Liu, Z.; Adams, K.L. Evolutionary transfers of mitochondrial genes to the nucleus in the Populus lineage and coexpression of nuclear and mitochondrial Sdh4 genes. New Phytol. 2006, 172, 429–439. [Google Scholar] [CrossRef]
- Choi, I.S.; Schwarz, E.N.; Ruhlman, T.A.; Khiyami, M.A.; Sabir, J.S.M.; Hajarah, N.H.; Sabir, M.J.; Rabah, S.O.; Jansen, R.K. Fluctuations in Fabaceae mitochondrial genome size and content are both ancient and recent. BMC Plant Biol. 2019, 19, 448. [Google Scholar] [CrossRef]
- Alverson, A.J.; Wei, X.X.; Rice, D.W.; Stern, D.B.; Barry, K.; Palmer, J.D. Insights into the evolution of mitochondrial genome size from complete sequences of Citrullus lanatus and Cucurbita pepo (Cucurbitaceae). Mol. Biol. Evol. 2010, 27, 1436–1448. [Google Scholar] [CrossRef]
- Adams, K.L.; Rosenblueth, M.; Qiu, Y.L.; Palmer, J.D. Multiple losses and transfers to the nucleus of two mitochondrial succinate dehydrogenase genes during angiosperm evolution. Genetics 2001, 158, 1289–1300. [Google Scholar]
- Small, I.D.; Schallenberg-Rudinger, M.; Takenaka, M.; Mireau, H.; Ostersetzer-Biran, O. Plant organellar RNA editing: What 30 years of research has revealed. Plant J. 2020, 101, 1040–1056. [Google Scholar] [CrossRef] [PubMed]
- Edera, A.A.; Gandini, C.L.; Sanchez-Puerta, M.V. Towards a comprehensive picture of C-to-U RNA editing sites in angiosperm mitochondria. Plant Mol. Biol. 2018, 97, 215–231. [Google Scholar] [CrossRef]
- Quinones, V.; Zanlungo, S.; Holuigue, L.; Litvak, S.; Jordana, X. The Cox1 Initiation Codon Is Created by Rna Editing in Potato Mitochondria. Plant Physiol. 1995, 108, 1327–1328. [Google Scholar] [CrossRef] [PubMed]
- Brenner, W.G.; Mader, M.; Muller, N.A.; Hoenicka, H.; Schroeder, H.; Zorn, I.; Fladung, M.; Kersten, B. High Level of Conservation of Mitochondrial RNA Editing Sites Among Four Populus Species. G3-Genes Genom. Genet. 2019, 9, 709–717. [Google Scholar] [CrossRef]
- Hollingsworth, P.M.; Graham, S.W.; Little, D.P. Choosing and Using a Plant DNA Barcode. PLoS ONE 2011, 6, e19254. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.H.; Wu, K.C.; Chuang, L.Y.; Chang, H.W. Decision Tree Algorithm-Generated Single-Nucleotide Polymorphism Barcodes of rbcL Genes for 38 Brassicaceae Species Tagging. Evol. Bioinform. 2018, 14, 1176934318760856. [Google Scholar] [CrossRef] [PubMed]
- Arulandhu, A.J.; Staats, M.; Hagelaar, R.; Voorhuijzen, M.M.; Prins, T.W.; Scholtens, I.; Costessi, A.; Duijsings, D.; Rechenmann, F.; Gaspar, F.B.; et al. Development and validation of a multi-locus DNA metabarcoding method to identify endangered species in complex samples. Gigascience 2017, 6, 1–18. [Google Scholar] [CrossRef]
- Lee, O.R.; Kim, M.K.; Yang, D.C. Authentication of medicinal plants by SNP-based multiplex PCR. Methods Mol. Biol. 2012, 862, 135–147. [Google Scholar]
- Brunner, I.; Brodbeck, S.; Buchler, U.; Sperisen, C. Molecular identification of fine roots of trees from the Alps: Reliable and fast DNA extraction and PCR-RFLP analyses of plastid DNA. Mol. Ecol. 2001, 10, 2079–2087. [Google Scholar] [CrossRef]
- Ju, L.P.; Shih, H.C.; Chiang, Y.C. Microsatellite primers for the endangered beech tree, Fagus hayatae (Fagaceae). Am. J. Bot. 2012, 99, e453–e456. [Google Scholar] [CrossRef]
- Reutimann, O.; Gugerli, F.; Rellstab, C. A species-discriminatory single-nucleotide polymorphism set reveals maintenance of species integrity in hybridizing European white oaks (Quercus spp.) despite high levels of admixture. Ann. Bot. 2020, 125, 663–676. [Google Scholar] [CrossRef]
- Schroeder, H.; Cronn, R.; Yanbaev, Y.; Jennings, T.; Mader, M.; Degen, B.; Kersten, B. Development of Molecular Markers for Determining Continental Origin of Wood from White Oaks (Quercus L. sect. Quercus). PLoS ONE 2016, 11, e0158221. [Google Scholar] [CrossRef] [PubMed]
- Pencakowski, B.M.; Tokarski, M.; Jonkisz, A.; Czosnykowska-Lukacka, M.; Lenard, E.; Malodobra-Mazur, M. DNA profiling of oaks (Quercus spp.). Arch. Med. Sadowej. Kryminol. 2018, 68, 1–9. [Google Scholar] [PubMed]
- Coutinho, J.P.; Carvalho, A.; Martin, A.; Lima-Brito, J. Molecular characterization of Fagaceae species using inter-primer binding site (iPBS) markers. Mol. Biol. Rep. 2018, 45, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.; Helmstetter, A.J.; Devey, D.; Wilkinson, T.; Papadopulos, A.S.T. Field-based species identification of closely-related plants using real-time nanopore sequencing. Sci. Rep. 2017, 7, 8345. [Google Scholar] [CrossRef]
- Dumolin, S.; Demesure, B.; Petit, R.J. Inheritance of chloroplast and mitochondrial genomes in pedunculate oak investigated with an efficient PCR method. Theor. Appl. Genet. 1995, 91, 1253–1256. [Google Scholar] [CrossRef]
- Schalamun, M.; Nagar, R.; Kainer, D.; Beavan, E.; Eccles, D.; Rathjen, J.P.; Lanfear, R.; Schwessinger, B. Harnessing the MinION: An example of how to establish long-read sequencing in a laboratory using challenging plant tissue from Eucalyptus pauciflora. Mol. Ecol. Resour. 2019, 19, 77–89. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Grabherr, M.G.; Haas, B.J.; Yassour, M.; Levin, J.Z.; Thompson, D.A.; Amit, I.; Adiconis, X.; Fan, L.; Raychowdhury, R.; Zeng, Q.D.; et al. Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat. Biotechnol. 2011, 29, 644–652. [Google Scholar] [CrossRef]
- Tillich, M.; Lehwark, P.; Pellizzer, T.; Ulbricht-Jones, E.S.; Fischer, A.; Bock, R.; Greiner, S. GeSeq-versatile and accurate annotation of organelle genomes. Nucleic Acids Res. 2017, 45, W6–W11. [Google Scholar] [CrossRef]
- Sequin-A DNA Sequence Submission Tool. 2019. Available online: https://www.ncbi.nlm.nih.gov/Sequin/ (accessed on 3 July 2020).
- Sloan, D.B.; Wu, Z.Q.; Sharbrough, J. Correction of Persistent Errors in Arabidopsis. Reference Mitochondrial Genomes. Plant Cell 2018, 30, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Kim, J.I.; Yi, G. geneCo: A visualized comparative genomic method to analyze multiple genome structures. Bioinformatics 2019, 35, 5303–5305. [Google Scholar] [CrossRef] [PubMed]
- geneCo. The Visualization Software for Genome Data. Available online: https://bigdata.dongguk.edu/geneCo/ (accessed on 3 September 2020).
- Stajich, J.E.; Block, D.; Boulez, K.; Brenner, S.E.; Chervitz, S.A.; Dagdigian, C.; Fuellen, G.; Gilbert, J.G.R.; Korf, I.; Lapp, H.; et al. The bioperl toolkit: Perl modules for the life sciences. Genome Res. 2002, 12, 1611–1618. [Google Scholar] [CrossRef] [PubMed]
- Loytynoja, A. Phylogeny-aware alignment with PRANK. Methods Mol. Biol. 2014, 1079, 155–170. [Google Scholar] [PubMed]
- GeSeq-Annotation of Organellar Genomes. Available online: https://chlorobox.mpimp-golm.mpg.de/geseq.html (accessed on 3 September 2020).
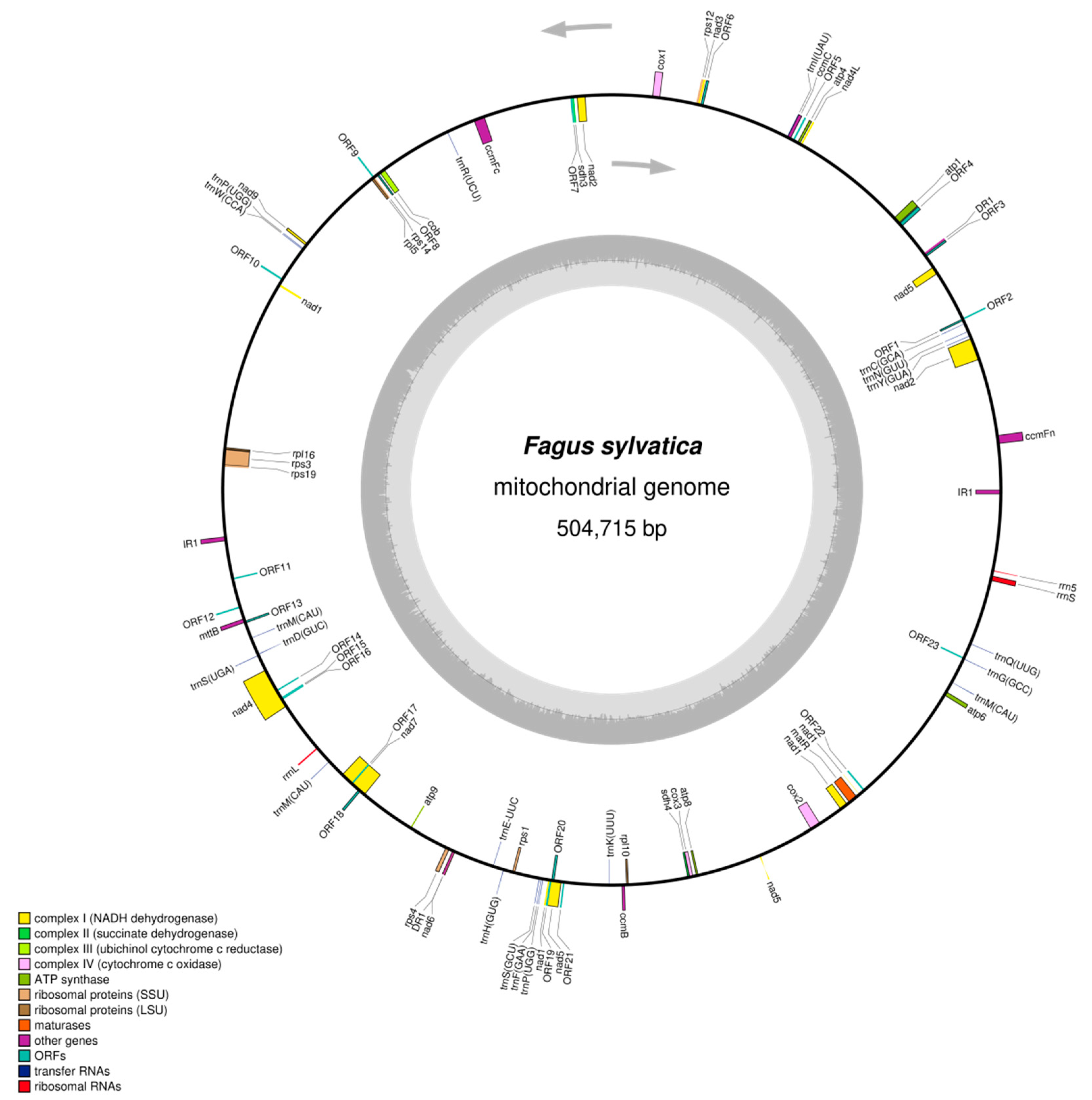

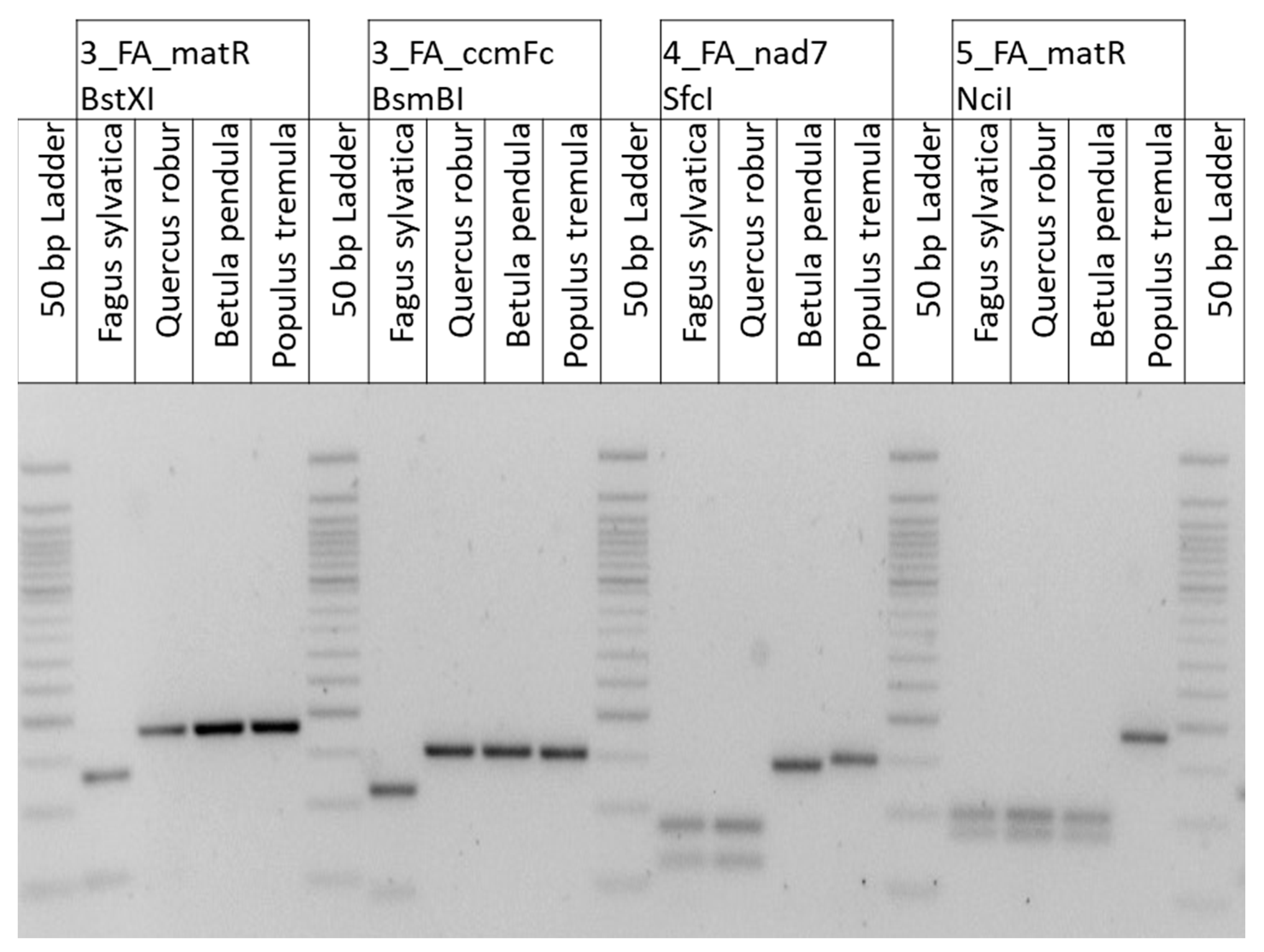
| Marker Name | Taxon Specificity * | SNP Position (in bp) | Target Allele/ Alternative Alleles | PCR Amplification in Deciduous Trees (D)/ Conifers (C) | Restriction Enzyme | Fragment Sizes (Target/Others) |
|---|---|---|---|---|---|---|
| 3_Fagus_matR | Fagus | 1530 | G/ C,T,A | D | BstXI | 53,129/ 182 |
| 3_Fagus_ccmFc | Fagus | 1862 | T/ C | D and C | BsmBI | 108,45/ 153 |
| 4_Fagaceae_nad7 | Fagaceae | 1459 | A/ G | D | SfcI | 5786/ 143 |
| 5_Fagales_matR | Fagales | 261 | G/ A | D and C | NciI | 8897/ >=185 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mader, M.; Schroeder, H.; Schott, T.; Schöning-Stierand, K.; Leite Montalvão, A.P.; Liesebach, H.; Liesebach, M.; Fussi, B.; Kersten, B. Mitochondrial Genome of Fagus sylvatica L. as a Source for Taxonomic Marker Development in the Fagales. Plants 2020, 9, 1274. https://doi.org/10.3390/plants9101274
Mader M, Schroeder H, Schott T, Schöning-Stierand K, Leite Montalvão AP, Liesebach H, Liesebach M, Fussi B, Kersten B. Mitochondrial Genome of Fagus sylvatica L. as a Source for Taxonomic Marker Development in the Fagales. Plants. 2020; 9(10):1274. https://doi.org/10.3390/plants9101274
Chicago/Turabian StyleMader, Malte, Hilke Schroeder, Thomas Schott, Katrin Schöning-Stierand, Ana Paula Leite Montalvão, Heike Liesebach, Mirko Liesebach, Barbara Fussi, and Birgit Kersten. 2020. "Mitochondrial Genome of Fagus sylvatica L. as a Source for Taxonomic Marker Development in the Fagales" Plants 9, no. 10: 1274. https://doi.org/10.3390/plants9101274
APA StyleMader, M., Schroeder, H., Schott, T., Schöning-Stierand, K., Leite Montalvão, A. P., Liesebach, H., Liesebach, M., Fussi, B., & Kersten, B. (2020). Mitochondrial Genome of Fagus sylvatica L. as a Source for Taxonomic Marker Development in the Fagales. Plants, 9(10), 1274. https://doi.org/10.3390/plants9101274






