The Genus Conradina (Lamiaceae): A Review
Abstract
:1. Introduction
2. Description
3. Phylogenetic Studies
4. Essential Oils and Non-Volatile Components
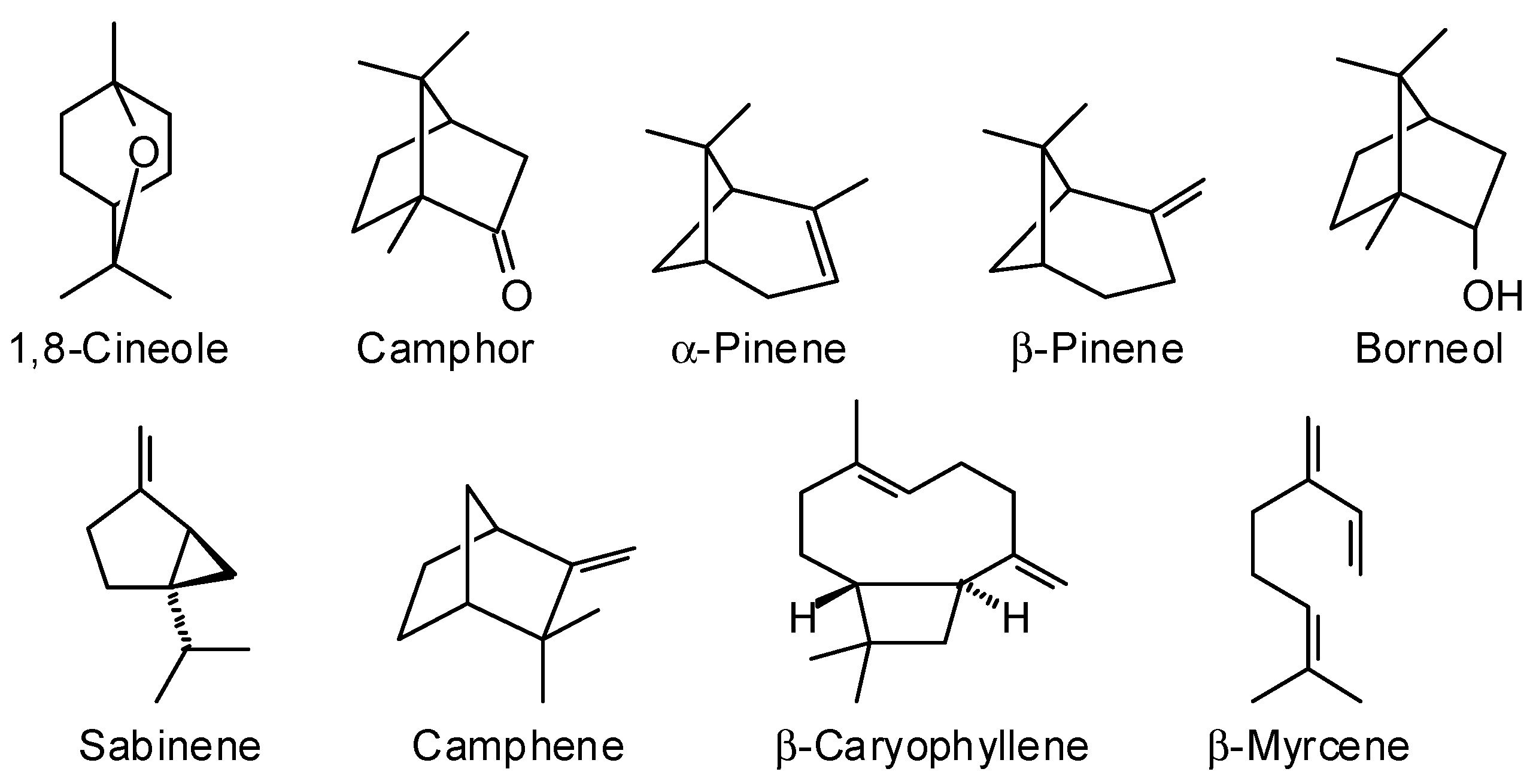
4.1. Essential Oils
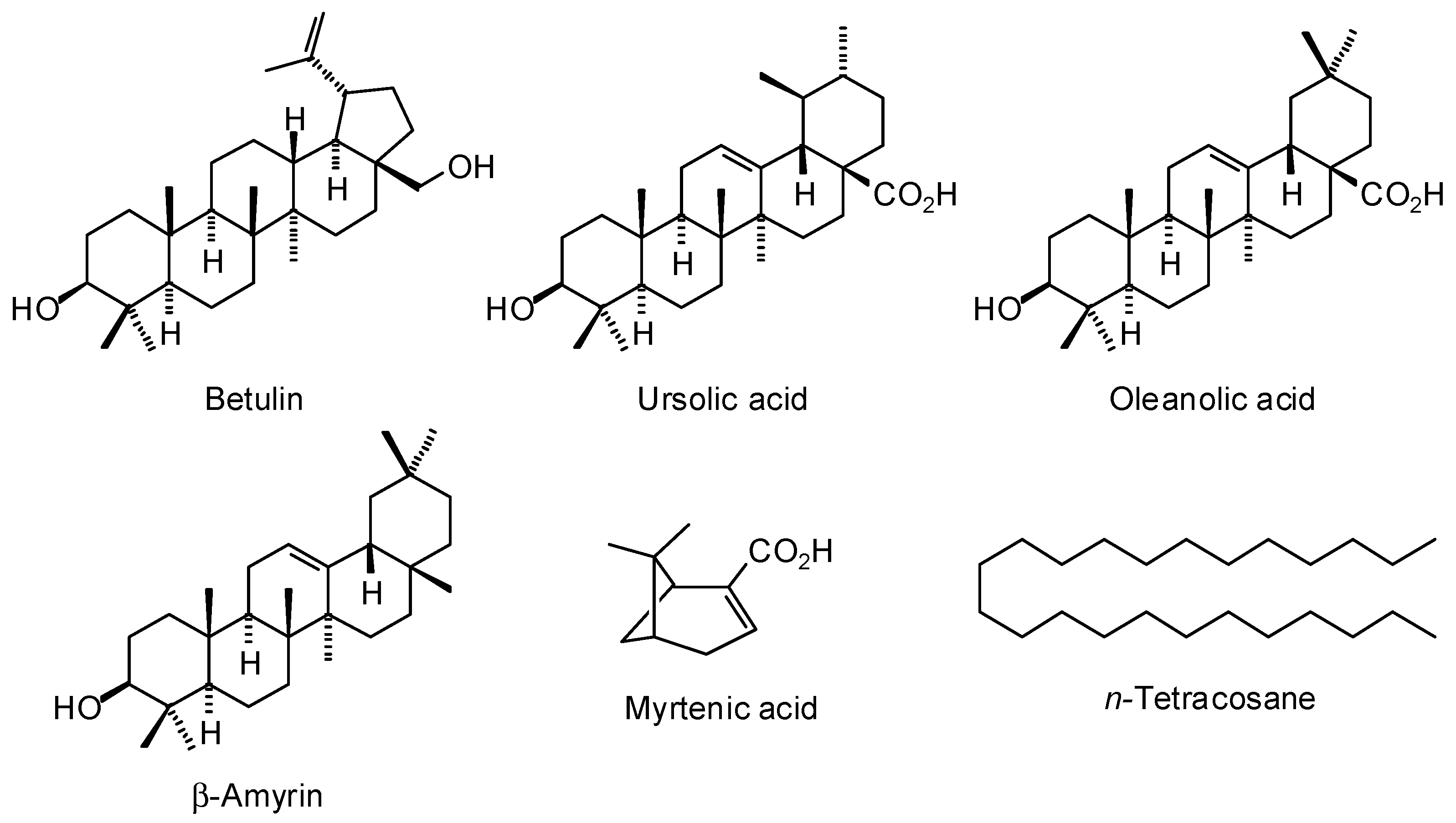
4.2. Nonvolatile Components
5. Biological Activities
5.1. Allelopathic Activity
5.2. Antibacterial and Antifungal Activity
5.3. Cytotoxic Activity
5.4. Antileishmanial Activity
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gray, T.C. A Monograph of the Genus Conradina; Vanderbilt University: Nashville, TN, USA, 1965. [Google Scholar]
- The Plant List. Available online: http://www.theplantlist.org/tpl1.1/search?q=conradina (accessed on 8 March 2018).
- The Missouri Botanical Garden. Available online: http://www.tropicos.org/NameSearch.aspx?name=Conradina&commonname= (accessed on 8 March 2018).
- Shinners, L.H. Synopsis of Conradina (Labiatae). Sida 1962, 1, 84–88. [Google Scholar]
- U.S. Fish and Wildlife Service, (USFWS). Endangered and Threatened Wildlife and plants: Endangered or threatened status for five Florida plants. USFWS Fed. Regist. Rules Regul. 1993, 58, 37432–37443. [Google Scholar]
- Kral, R.; McCartney, R.B. A new species of Conradina (Lamiaceae) from northeastern peninsular Florida. Sida 1991, 14, 391–398. [Google Scholar]
- Peterson, C.L. Analysis of the Essential Oils, Leaf Ultrastructure, and the In Vitro Growth Response of the Mint Genus Conradina; Florida Institute of Technology: Melbourne, FL, USA, 1998. [Google Scholar]
- Harrison, M. Groundcovers for the South; Pineapple Press: Sarasota, FL, USA, 2006. [Google Scholar]
- Looney, P.B.; Gibson, D.J. The relationship between the soil seed bank and above-ground vegetation of a coastal barrier island. J. Veg. Sci. 1995, 6, 825–836. [Google Scholar] [CrossRef]
- Wunderlin, R.P.; Richardson, D.; Hansen, B. Status Report on Conradina brevifolia; U.S. Fish and Wildlife Service: Jacksonville, FL, USA, 1980. [Google Scholar]
- Kral, R. A Report on Some Rare, Threatened, or Endangered Forest-Related Vascular Plants of the South; USDA Forest Service: Atlanta, GA, USA, 1983. [Google Scholar]
- Christman, S.P.; Judd, W.S. Notes on plants endemic to Florida scrub. Fla. Sci. 1990, 53, 52–73. [Google Scholar]
- Christman, S.P. Endemism and Florida’s Interior Sand Pine Scrub; Final Report No. GFC-84-101; Florida Game and Fresh Water Fish Commission, Nongame Wildlife Program: Tallahassee, FL, USA, 1988. [Google Scholar]
- U.S. Fish and Wildlife Service (USFWS). Short-Leaved Rosemary (Conradina brevifolia): Five-Year Review; USFWS: Atlanta, GA, USA, 2008.
- U.S. Fish and Wildlife Service (USFWS). Recovery Plan for Etonia rosemary (Conradina etonia); USFWS: Atlanta, GA, USA, 1994.
- U.S. Fish and Wildlife Service, (USFWS). Conradina etonia, Five-Year Review; USFWS: Atlanta, GA, USA, 2007.
- Peterson, C.L.; Weigel, R.C. In vitro propagation of Conradina etonia. Fla. Sci. 2002, 65, 201–207. [Google Scholar]
- U.S. Fish and Wildlife Service, (USFWS). Etonia rosemary (Conradina etonia); USFWS: Atlanta, GA, USA, 2005.
- U.S. Fish and Wildlife Service, (USFWS). Recovery Plan for Apalachicola rosemary (Conradina glabra); USFWS: Atlanta, GA, USA, 1994.
- U.S. Fish and Wildlife Service, (USFWS). Conradina glabra (Apalachicola rosemary): Five-Year Review; USFWS: Atlanta, GA, USA, 2009.
- Jennison, H.M. A new species of Conradina from Tennessee. J. Elisha Mitchell Sci. Soc. 1933, 48, 268–269. [Google Scholar]
- U.S. Fish and Wildlife Service, (USFWS). Recovery Plan for Nineteen Central Florida Scrub and High Pineland Plants (Revised); USFWS: Atlanta, GA, USA, 1996.
- Albrecht, M.A.; Penagos, J.C.Z. Seed germination ecology of three imperiled plants of rock outcrops in the southeastern United States. J. Torrey Bot. Soc. 2012, 139, 86–95. [Google Scholar] [CrossRef]
- U.S. Fish and Wildlife Service, (USFWS). Endangered and Threatened Wildlife and plants: Conradina verticillata (Cumberland rosemary) determined to be threatened. USFWS Fed. Regist. Rules Regul. 1991, 56, 60937–60941. [Google Scholar]
- Edwards, C.E.; Judd, S.; Ionta, G.M.; Herring, B. Using population genetic data as a tool to identify new species: Conradina cygniflora (Lamiaceae), a new, endangered species from Florida. Syst. Bot. 2009, 34, 747–759. [Google Scholar] [CrossRef]
- Small, J.K. Manual of the Southeastern Flora; The University of North Carolina Press: Chapel Hill, NC, USA, 1933. [Google Scholar]
- Small, J.K. Studies in the botany of the Southeastern United States-XIV. Bull. Torrey Bot. Club 1898, 25, 469–484. [Google Scholar] [CrossRef]
- Trusty, J.L.; Olmstead, R.G.; Bogler, D.J.; Guerra, A.S.; Ortega, J.F. Using molecular data to test a biogeographic connection of the Macaronesian genus Bystropogon (Lamiaceae) to the New World: A case of conflicting phylogenies. Syst. Bot. 2014, 29, 702–715. [Google Scholar] [CrossRef]
- Edwards, C.E.; Soltis, D.E.; Soltis, P.S. Molecular phylogeny of Conradina and other scrub mints (Lamiaceae) from the southeastern USA: Evidence for hybridization in pleistocene refugia? Syst. Bot. 2006, 31, 193–207. [Google Scholar] [CrossRef]
- Edwards, C.E.; Lefkowitz, D.; Soltis, D.; Soltis, P. Phylogeny of Conradina and related Southeastern scrub mints (Lamiaceae) based on GapC gene sequences. Int. J. Plant Sci. 2008, 169, 579–594. [Google Scholar] [CrossRef]
- Edwards, C.E.; Soltis, D.E.; Soltis, P.S. Using patterns of genetic structure based on microsatellite loci to test hypotheses of current hybridization, ancient hybridization and incomplete lineage sorting in Conradina (Lamiaceae). Mol. Ecol. 2008, 17, 5157–5174. [Google Scholar] [CrossRef] [PubMed]
- Quinn, B.P.; Bernier, U.R.; Booth, M.M. Identification of compounds from Etonia rosemary (Conradina etonia). J. Chromatogr. A 2007, 1160, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Dosoky, N.S.; Stewart, C.D.; Setzer, W.N. Identification of essential oil components from Conradina canescens. Am. J. Essent. Oils Nat. Prod. 2014, 2, 24–28. [Google Scholar]
- Williamson, G.B.; Fischer, N.H.; Richardson, D.R.; de la Peña, A. Chemical inhibition of fire-prone grasses by fire-sensitive shrub, Conradina canescens. J. Chem. Ecol. 1989, 15, 1567–1577. [Google Scholar] [CrossRef] [PubMed]
- Fischer, N.H.; Williamson, G.B.; Weidenhamer, J.D.; Richardson, D.R. In search of allelopathy in the Florida scrub: The role of terpenoids. J. Chem. Ecol. 1994, 20, 1355–1380. [Google Scholar] [CrossRef] [PubMed]
- Dosoky, N.S.; Moriarity, D.M.; Setzer, W.N. Phytochemical and biological investigations of Conradina canescens Gray. Nat. Prod. Commun. 2016, 11, 25–28. [Google Scholar] [PubMed]
- Dosoky, N.S. Isolation and Identification of Bioactive Compounds from Conradina canescens Gray; University of Alabama in Huntsville: Huntsville, AL, USA, 2015. [Google Scholar]
- Rice, E.L. Allelopathy, 2nd ed.; Academic Press: Orlando, FL, USA, 1984. [Google Scholar]
- Macias, F.A.; Molinillo, J.M.G.; Varela, R.M.; Galindo, J.C.G. Allelopathy-a natural alternative for weed control. Pest Manag. Sci. 2007, 63, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Ungar, I.A. The effect of salinity and temperature on the germination of polymorphic seeds and growth of Atriplex triangularis Willd. Am. J. Bot. 1984, 71, 481–489. [Google Scholar] [CrossRef]
- Muller, W.H.; Muller, C.H. Volatile growth inhibitors produced by Salvia species. Bull. Torrey Bot. Club 1964, 91, 327–330. [Google Scholar] [CrossRef]
- De la Peña, A.C. Terpenoids from Conradina canescens (Labiatae) with Possible Allelopathic Activity; Louisiana State University: Baton Rouge, LA, USA, 1985. [Google Scholar]
- Fischer, N.H.; Tanrisever, N.; Williamson, G.B. Allelopathy in the Florida scrub community as a model for natural herbicide actions. In Biologically Active Natural Products: Potential Use in Agriculture; Cutler, H.G., Ed.; American Chemical Society: Washington, DC, USA, 1988. [Google Scholar]
- Qiu, X.; Yu, S.; Wang, Y.; Fang, B.; Cai, C.; Liu, S. Identification and allelopathic effects of 1,8-cineole from Eucalyptus urophylla on lettuce. Allelopath. J. 2010, 26, 255–264. [Google Scholar]
- Nishida, N.; Tamotsu, S.; Nagata, N.; Saito, C.; Sakai, A. Allelopathic effects of volatile monoterpenoids produced by Salvia leucophylla: Inhibition of cell proliferation and DNA synthesis in the root apical meristem of Brassica campestris seedlings. J. Chem. Ecol. 2005, 31, 1187–1203. [Google Scholar] [CrossRef] [PubMed]
- Zunino, M.P.; Zygadlo, J.A. Effect of monoterpenes on lipid oxidation in maize. Planta 2004, 219, 303–309. [Google Scholar] [PubMed]
- Singh, H.P.; Batish, D.R.; Kohli, R.K. Allelopathic effect of two volatile monoterpenes against bill goat weed (Ageratum conyzoides L.). Crop Prot. 2002, 21, 347–350. [Google Scholar] [CrossRef]
- Abrahim, D.; Braguini, W.L.; Kelmer-Bracht, A.M.; Ishii-Iwamoto, E.L. Effects of four monoterpenes on germination, primary root growth, and mitochondrial respiration of maize. J. Chem. Ecol. 2000, 26, 611–624. [Google Scholar] [CrossRef]
- Macias, F.A.; Galindo, J.C.G.; Molinillo, J.M.G. Allelopathy: Chemistry and Mode of Action of Allelochemicals, 1st ed.; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Rizvi, S.J. Allelopathy: Basic and Applied Aspects; Springer Science & Business Media: Dordrecht, The Netherlands, 2012. [Google Scholar]
- Szakiel, A.; Grzelak, A.; Dudek, P.; Janiszowska, W. Biosynthesis of oleanolic acid and its glycosides in Calendula officinalis suspension culture. Plant Physiol. Biochem. 2003, 41, 271–275. [Google Scholar] [CrossRef]
- Weidenhamer, J.D.; Macias, F.A.; Fischer, N.H.; Williamson, G.B. Just how insoluble are monoterpenes? J. Chem. Ecol. 1993, 19, 1799–1807. [Google Scholar] [CrossRef] [PubMed]
- Wolska, K.I.; Grudniak, A.M.; Fiecek, B.; Kraczkiewicz-Dowjat, A.; Kurek, A. Antibacterial activity of oleanolic and ursolic acids and their derivatives. Cent. Eur. J. Biol. 2010, 5, 543–553. [Google Scholar] [CrossRef]

| Species | Morphological Characteristics | References |
|---|---|---|
| C. canescens | Small shrub, up to 1 m high. Leaves are 7 to 20 mm long, mostly longer than the internodes. Leaf blades are pubescent on both sides. One to three flowers per axil, lower corolla-lip 8–10-mm long; lateral lobes longer than wide. Calyx-tube hirsute or villiou-hirsute. | [5] |
| C. brevifolia a | Small shrub, up to 1 m high. Leaves are short fleshy 6.0 to 8.2 mm long, mostly shorter than the internodes, covered with short downy hairs and many tiny glands on the upper side. One to six lavender flowers per axil. | [5,11] |
| C. etonia | Straight slender shrub, about 1.5 m high. Leaves have hairy, veiny, glandular blades 1.5–3 cm long and 3–9 mm wide with tightly rolled edges. Three to seven flowers per axil. Pink to lavender in color with darker dotted lower petal. | [5,15,18] |
| C. glabra | Small shrub, about 80 cm high but some individuals reach up to 2 m. Leaves are opposite, up to 1.5 cm long, hairless on the upper surface. Two to three flowers per axil. Corolla is 1.5–2 cm long, white to pale lavender in color with a band of purple dots on the lower lip. | [5,19,20] |
| C. grandiflora | Erect shrub, 1.5–2.0 m high, with hairy branches and twigs. Leaves are hairy, glandular, up to 1.5 cm long. Year-round hairy lavender flowers with darker lavender spots, lower lip is 12–15 mm long with lateral lobes longer than wide. This species has the largest flowers of genus Conradina. | [5] |
| C. verticillata | Erect shrub, 0.5 m high with reclining branches. Leaves are about 2.5 cm long, very narrow, and arranged in tight bunches that appear as whorls around the stems. Flowers are 2.5 cm long, purple to white and borne in leaf-like clusters of bracts at the ends of the stems. | [24] |
| C. montana b | Short shrub less than 0.5 m high with diffuse branches. Leaves are narrowly linear, 5–16 mm long. Leaf blades are glabrous on the upper surface. Minute flowers with corolla 3.5–4 mm long. Calyx-tube hirsutulous. | [23] |
| C. puberula a | Short shrub of about 0.5 m high with numerous slender branches. Leaves are narrowly linear but strongly revolute and clavate, 12–20 mm long. Calyx-tube minutely canescent, 5–7 mm long. Flowers appear in racemes of 2–6 per axil, with corolla 4–5 mm long. | [26,27] |
| C. cygniflora | Virgate shrub up to 1 m high, branches are erect to spreading, internodes 5–43 mm long. Leaves persistent, appearing fascicled- verticillate; narrowly obovate, 9–33 mm long. The abaxial leaf surface is densely-covered by simple unicellular hairs. Cymes carry 1–5 subsessile flowers, densely pubescent, 1.3–12.5 mm long. Large calyx of 8.5–11 mm long; densely covered with simple hairs, upper lip upcurved, 3.6–4.4 mm long, lower lip 4.3–5.5 mm long. Corolla strongly bilabiate, 20–29 mm long, lavender, shading to white in throat, with purple spots; abaxial surface of upper lip darker lavender. | [25] |
| Species | Major Oil Components (%) | Unique Component(s) | Reference |
|---|---|---|---|
| C. brevifolia a | Camphor (9.7–17.54%) and 1,8-cineole (1.97–4.86%) | α- and β-farnesene | [7] |
| C. canescens a | Camphor (0.27–23.64%), 1,8-cineole (0.17–3.34%), cis-pinocamphone (0–8.74%) | none | [7] |
| C. canescens b | 1,8-cineole (5.2–25.2%), camphor (5.7–8.0%), α-pinene (3.2–5.6%), p-cymene (3.3–5.9%), cis-pinocamphone (1.3–5.5%), myrtenal (5.2–8.1%), myrtenol (3.4–9.2%), verbenone (4–4.5%), and myrtenyl acetate (5.0–5.4%) | - | [33] |
| C. etonia a | Camphor (30.55–35.65%), limonene (3.77–6.33%), camphene (2.92–3.75%), and β-caryophyllene (2.95–6.54%) | β-elemene, 4-carene and α-terpineol | [32] |
| C. glabra a | 1,8-cineole (2.38–7.34%) and camphor (11.78–15.88%) | Dolcymene and bornyl acetate | [7] |
| C. grandiflora a | β-pinene (4.38–5.81%) and β-cubebene (1.95–6.56%) | Calarene and β-pinone | [7] |
| C. verticillata a | 1,8-cineole (3.15–3.78%) and camphor (5.81–8.35%) | Germacrene B and 2,5,6-trimethyl-1,3,6-heptatriene | [7] |
| C. cygniflora | Data not available | N/A | N/A |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dosoky, N.S.; Setzer, W.N. The Genus Conradina (Lamiaceae): A Review. Plants 2018, 7, 19. https://doi.org/10.3390/plants7010019
Dosoky NS, Setzer WN. The Genus Conradina (Lamiaceae): A Review. Plants. 2018; 7(1):19. https://doi.org/10.3390/plants7010019
Chicago/Turabian StyleDosoky, Noura S., and William N. Setzer. 2018. "The Genus Conradina (Lamiaceae): A Review" Plants 7, no. 1: 19. https://doi.org/10.3390/plants7010019
APA StyleDosoky, N. S., & Setzer, W. N. (2018). The Genus Conradina (Lamiaceae): A Review. Plants, 7(1), 19. https://doi.org/10.3390/plants7010019







