Seed Biofortification and Phytic Acid Reduction: A Conflict of Interest for the Plant?
Abstract
:1. Introduction
2. Biosynthetic Pathways
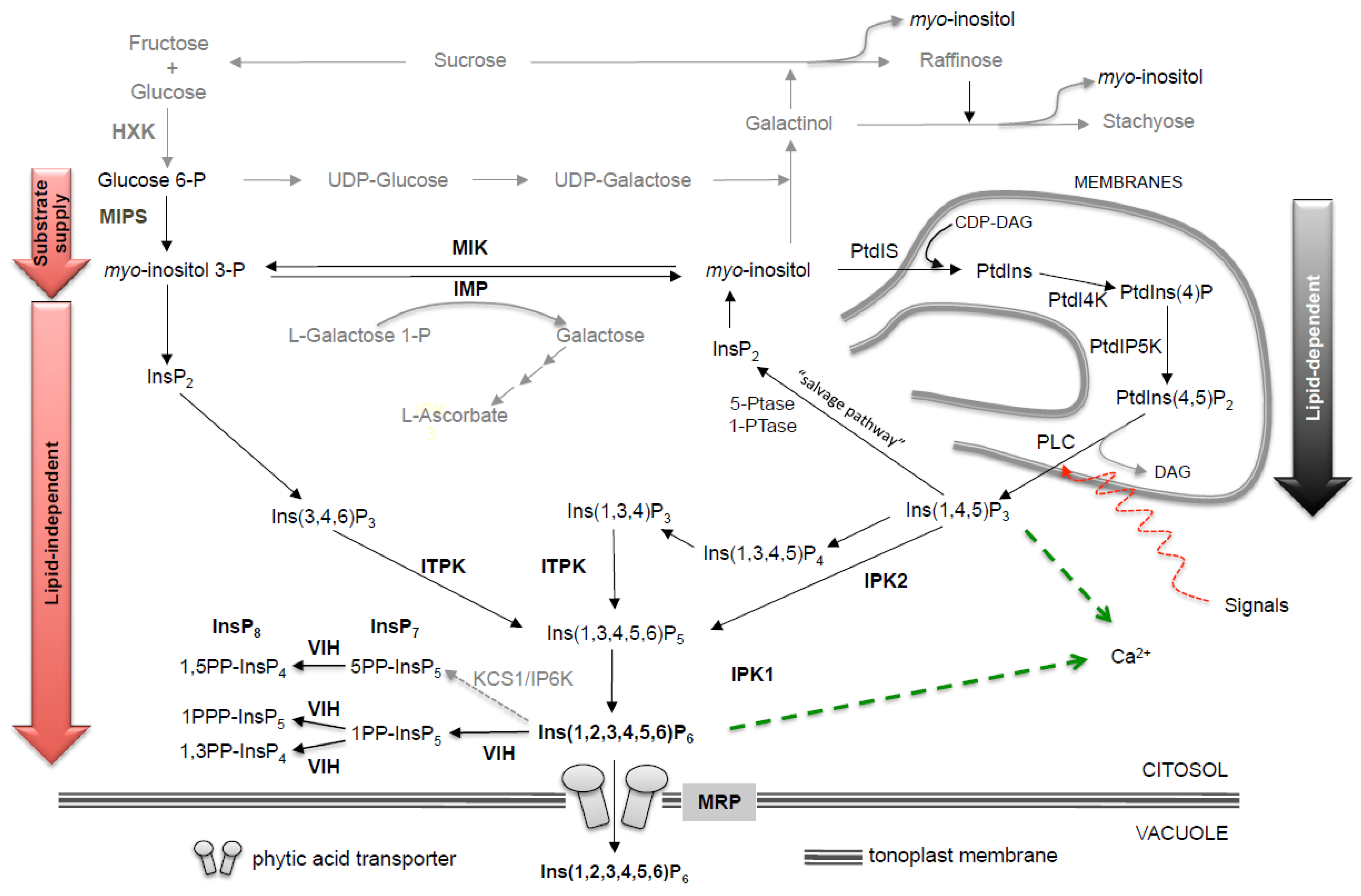
2.1. Genomic Organization and Regulation of Phytic Acid Pathway Genes
2.1.1. MIPS
2.1.2. Myo-Inositol Reversible Dephosphsorylation: IMP and MIK
2.1.3. PGK
2.1.4. IPK2
2.1.5. ITPK
2.1.6. IPK1
2.1.7. MRP
3. Low Phytic Acid (lpa) Mutants
| Class | Gene Function | Effects on the Pathway |
|---|---|---|
| Type 1 | MIPS | Decrease in phytic acid accompanied by a molar increase in free phosphate |
| MIK | ||
| IMP | ||
| Type 2 | 2PGK | Decrease in phytic acid accompanied by a low increase in free phosphate and increased content of lower inositol phosphates (InsPs) |
| IPK2 | ||
| ITPK | ||
| IPK1 | ||
| Type 3 | MRP | Decrease in phytic acid accompanied by a molar increase in free phosphate and/or decrease in phytic acid in specific seed tissues |
| Putative sulfate transporter (sultr3;3) |
Metabolic and Phenotypic Traits of Low Phytic Acid Mutants
4. Enzymes and Metabolites of the Phytic Acid Pathway Have Regulatory Roles in Cell Signaling and Plant Processes
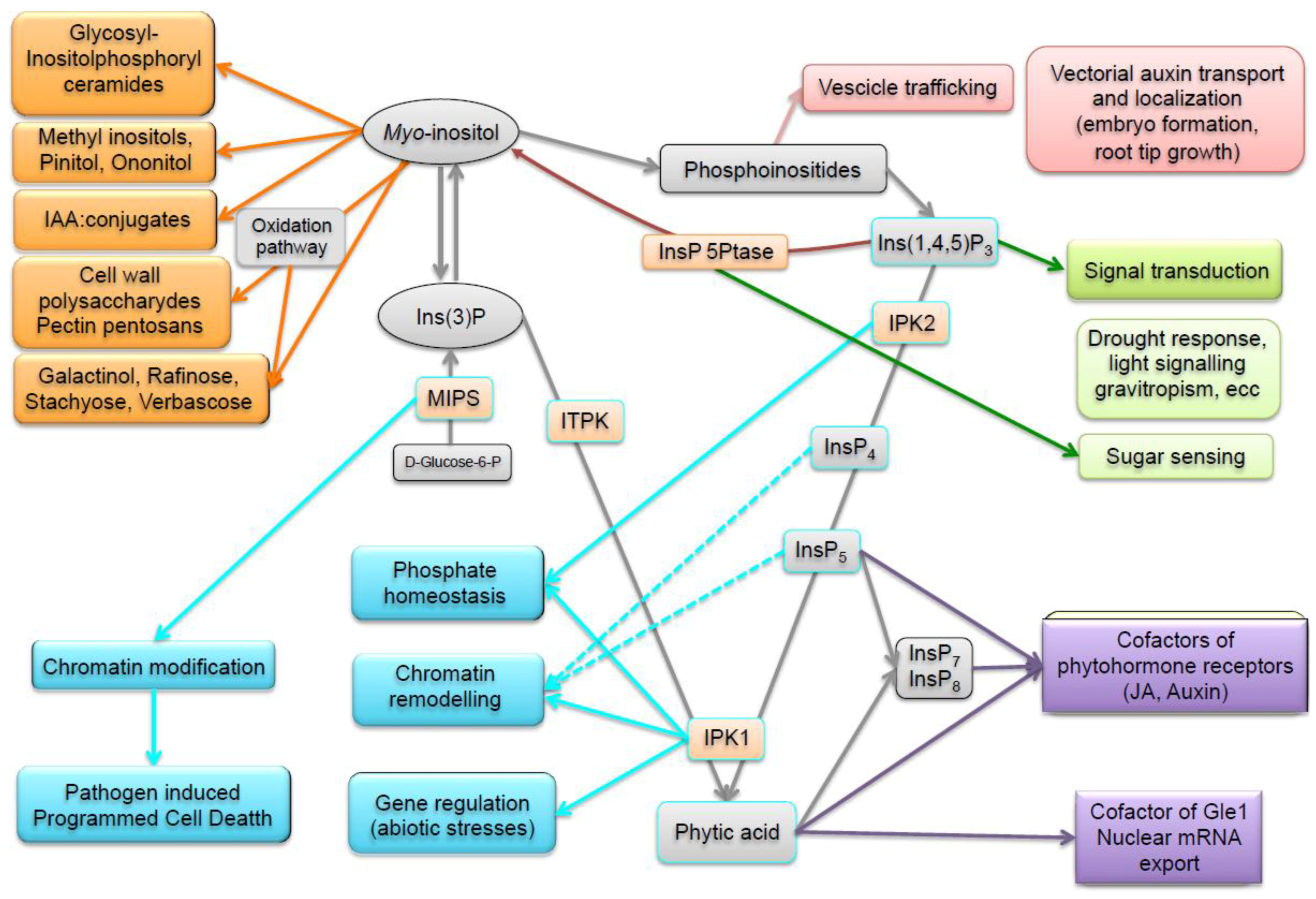
4.1. Signal Transduction
4.2. Vesicle Trafficking and Polar Auxin Transport
4.3. Biotic and Abiotic Stress Response
4.4. Nuclear Functions and Regulation of Phosphorus Homeostasis
4.5. Hormonal Signaling
5. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Shears, S. Assessing the omnipotence of inositol hexakisphosphate. Cell. Signal. 2001, 13, 151–158. [Google Scholar] [CrossRef]
- Coelho, C.; Tsai, S.; Vitorello, V. Dynamics of inositol phosphate pools (tris-, tetrakis- and pentakisphosphate) in relation to the rate of phytate synthesis during seed development in common bean (Phaseolus vulgaris). J. Plant Physiol. 2005, 162, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hatzack, F.; Johansen, K.; Rasmussen, S. Nutritionally relevant parameters in low-phytate barley (Hordeum vulgare L.) grain mutants. J. Agric. Food Chem. 2000, 48, 6074–6080. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Tanaka, K.; Kasai, Z. Phytic acid formation in dissected ripening rice grains. Agric. Biol. Chem. 1979, 43, 2211–2213. [Google Scholar] [CrossRef]
- Otegui, M.; Capp, R.; Staehelin, L. Developing seeds of Arabidopsis store different minerals in two types of vacuoles and in the endoplasmic reticulum. Plant Cell 2002, 14, 1311–1327. [Google Scholar] [CrossRef] [PubMed]
- Greenwood, J.; Gifford, D.; Bewley, J. Seed development in Ricinus-communis cv Hale (astor bean) 2. Accumulation of phytic acid in the developing endosperm and embryo in relation to the deposition of lipid, protein, and phosphorus. Can. J. Bot. 1984, 62, 255–261. [Google Scholar] [CrossRef]
- Greenwood, J.; Bewley, J.D. Seed development in Ricinus-communis cv Hale (astor bean). 3. Pattern of storage protein and phytin accumulation in the endosperm. Can. J. Bot. 1985, 63, 2121–2128. [Google Scholar] [CrossRef]
- O’Dell, B.L.; de Boland, A.R.; Koirtyohann, S.T. Distribution of phytate and nutritionally important elements among the morphological components of cereal grains. J. Agric. Food Chem. 1972, 20, 718–721. [Google Scholar] [CrossRef]
- Ariza-Nieto, M.; Blair, M.; Welch, R.; Glahn, R. Screening of iron bloavallability patterns in eight bean (Phaseolus vulgaris L.) genotypes using the Caco-2 cell in vitro model. J. Agric. Food Chem. 2007, 55, 7950–7956. [Google Scholar] [CrossRef] [PubMed]
- Raboy, V. Myo-inositol-1,2,3,4,5,6-hexakisphosphate. Phytochemistry 2003, 64, 1033–1043. [Google Scholar] [CrossRef]
- Raboy, V. Seeds for a better future: “Low phytate”, grains help to overcome malnutrition and reduce pollution. Trends Plant Sci. 2001, 6, 458–462. [Google Scholar] [CrossRef]
- Schlemmer, U.; Frølich, W.; Prieto, R.M.; Grases, F. Phytate in foods and significance for humans: Food sources, intake, processing, bioavailability, protective role and analysis. Mol. Nutr. Food Res. 2009, 53, S330–S375. [Google Scholar] [CrossRef] [PubMed]
- Leytem, A.B.; Maguire, R.O. Environmental implications of inositol phosphates in animal manures. In Inositol Phosphates: Linking Agriculture and the Environment; Turner, B.L., Richardson, A.E., Mullaney, E.J., Eds.; CAB International: Wallingford, UK, 2007; pp. 150–168. [Google Scholar]
- Raboy, V. Approaches and challenges to engineering seed phytate and total phosphorus. Plant Sci. 2009, 177, 281–296. [Google Scholar] [CrossRef]
- Gerke, J. Phytate (inositol hexakisphosphate) in soil and phosphate acquisition from inositol phosphates by higher plants. A review. Plants 2015, 4, 253–266. [Google Scholar] [CrossRef]
- Lott, J.; Ockenden, I.; Raboy, V.; Batten, G. Phytic acid and phosphorus in crop seeds and fruits: A global estimate. Seed Sci. Res. 2000, 10, 11–33. [Google Scholar]
- Rose, T.; Liu, L.; Wissuwa, M. Improving phosphorus efficiency in cereal crops: Is breeding for reduced grain phosphorus concentration part of the solution? Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Raboy, V.; Cichy, K.; Peterson, K.; Reichman, S.; Sompong, U.; Srinives, P.; Saneoka, H. Barley (Hordeum vulgare L.) low phytic acid 1-1: An endosperm-specific, filial determinant of seed total phosphorus. J. Hered. 2014, 105, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Torabinejad, J.; Donahue, J.; Gunesekera, B.; Allen-Daniels, M.; Gillaspy, G. VTC4 is a bifunctional enzyme that affects myoinositol and ascorbate biosynthesis in plants. Plant Physiol. 2009, 150, 951–961. [Google Scholar] [CrossRef] [PubMed]
- Laing, W.; Bulley, S.; Wright, M.; Cooney, J.; Jensen, D.; Barraclough, D.; MacRae, E. A highly specific l-galactose-1-phosphate phosphatase on the path to ascorbate biosynthesis. Proc. Natl. Acad. Sci. USA 2004, 101, 16976–16981. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wang, H.; Hazebroek, J.; Ertl, D.; Harp, T. The maize low-phytic acid 3 encodes a myo-inositol kinase that plays a role in phytic acid biosynthesis in developing seeds. Plant J. 2005, 42, 708–719. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Tai, T. Genetic analysis of two OsLpa1-like genes in Arabidopsis reveals that only one is required for wild-type seed phytic acid levels. Planta 2010, 232, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Andaya, C.; Newman, J.; Goyal, S.; Tai, T. Isolation and characterization of a low phytic acid rice mutant reveals a mutation in the rice orthologue of maize MIK. Theor. Appl. Genet. 2008, 117, 1291–1301. [Google Scholar] [CrossRef] [PubMed]
- Odom, A.; Stahlberg, A.; Wente, S.; York, J. A role for nuclear inositol 1,4,5-trisphosphate kinase in transcriptional control. Science 2000, 287, 2026–2029. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Andaya, C.; Goyal, S.; Tai, T. The rice OsLpa1 gene encodes a novel protein involved in phytic acid metabolism. Theor. Appl. Genet. 2008, 117, 769–779. [Google Scholar] [CrossRef] [PubMed]
- Tagashira, Y.; Shimizu, T.; Miyamoto, M.; Nishida, S.; Yoshida, K. Overexpression of a gene involved in phytic acid biosynthesis substantially increases phytic acid and total phosphorus in rice seeds. Plants 2015, 4, 196–208. [Google Scholar] [CrossRef]
- Sparvoli, F.; Cominelli, E. Phytate transport by MRPs. In Plant ABC Transporters; Gesler, M., Ed.; Springer: Berlin, Germany, 2014; Volume 22, pp. 19–38. [Google Scholar]
- Phillippy, B.; Ullah, A.; Ehrlich, K. Purification and some properties of inositol 1,3,4,5,6-pentakisphosphate 2-kinase from immature soybean seeds. J. Biol. Chem. 1994, 269, 28393–28399. [Google Scholar] [PubMed]
- Phillippy, B. Identification of inositol 1,3,4-trisphosphate 5-kinase and inositol 1,3,4,5-tetrakisphosphate 6-kinase in immature soybean seeds. Plant Physiol. 1998, 116, 291–297. [Google Scholar] [CrossRef]
- Stevenson-Paulik, J.; Odom, A.; York, J. Molecular and biochemical characterization of two plant inositol polyphosphate 6-/3-/5-kinases. J. Biol. Chem. 2002, 277, 42711–42718. [Google Scholar] [CrossRef] [PubMed]
- Sweetman, D.; Johnson, S.; Caddick, S.; Hanke, D.; Brearley, C. Characterization of an Arabidopsis inositol 1,3,4,5,6-pentakisphosphate 2-kinase (AtIPK1). Biochem. J. 2006, 394, 95–103. [Google Scholar] [CrossRef] [PubMed]
- Sweetman, D.; Stavridou, I.; Johnson, S.; Green, P.; Caddick, S.; Brearley, C. Arabidopsis thaliana inositol 1,3,4-trisphosphate 5/6-kinase 4 (AtITPK4) is an outlier to a family of ATP-grasp fold proteins from Arabidopsis. FEBS Lett. 2007, 581, 4165–4171. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Thompson, M.; Lin, G.; Butler, H.; Gao, Z.; Thornburgh, S.; Yau, K.; Smith, D.; Shukla, V. Inositol 1,3,4,5,6-pentakisphosphate 2-kinase from maize: Molecular and biochemical characterization. Plant Physiol. 2007, 144, 1278–1291. [Google Scholar] [CrossRef] [PubMed]
- Josefsen, L.; Bohn, L.; Sorensen, M.; Rasmussen, S. Characterization of a multifunctional inositol phosphate kinase from rice and barley belonging to the ATP-grasp superfamily. Gene 2007, 397, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Tai, T. Identification of genes necessary for wild-type levels of seed phytic acid in Arabidopsis thaliana using a reverse genetics approach. Mol. Genet. Genom. 2011, 286, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Tanaka, K.; Kuwano, M.; Yoshida, K. Expression pattern of inositol phosphate-related enzymes in rice (Oryza sartiva L.): Implications for the phytic acid biosynthetic pathway. Gene 2007, 405, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Fileppi, M.; Galasso, I.; Tagliabue, G.; Daminati, M.; Campion, B.; Doria, E.; Sparvoli, F. Characterisation of structural genes involved in phytic acid biosynthesis in common bean (Phaseolus vulgaris L.). Mol. Breed. 2010, 25, 453–470. [Google Scholar] [CrossRef]
- Bhati, K.; Aggarwal, S.; Sharma, S.; Mantri, S.; Singh, S.; Bhalla, S.; Kaur, J.; Tiwari, S.; Roy, J.; Tuli, R.; et al. Differential expression of structural genes for the late phase of phytic acid biosynthesis in developing seeds of wheat (Triticum aestivum L.). Plant Sci. 2014, 224, 74–85. [Google Scholar] [CrossRef] [PubMed]
- Stiles, A.; Qian, X.; Shears, S.; Grabau, E. Metabolic and signaling properties of an ITPK gene family in glycine max. FEBS Lett. 2008, 582, 1853–1858. [Google Scholar] [CrossRef] [PubMed]
- Chappell, A.; Scaboo, A.; Wu, X.; Nguyen, H.; Pantalone, V.; Bilyeu, K. Characterization of the MIPS gene family in Glycine max. Plant Breed. 2006, 125, 493–500. [Google Scholar] [CrossRef]
- Larson, S.; Raboy, V. Linkage mapping of maize and barley myo-inositol 1-phosphate synthase dna sequences: Correspondence with a low phytic acid mutation. Theor. Appl. Genet. 1999, 99, 27–36. [Google Scholar] [CrossRef]
- Yoshida, K.; Wada, T.; Koyama, H.; Mizobuchi-Fukuoka, R.; Naito, S. Temporal and spatial patterns of accumulation of the transcript of myo-inositol-1-phosphate synthase and phytin-containing particles during seed development in rice. Plant Physiol. 1999, 119, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Hitz, W.; Carlson, T.; Kerr, P.; Sebastian, S. Biochemical and molecular characterization of a mutation that confers a decreased raffinosaccharide and phytic acid phenotype on soybean seeds. Plant Physiol. 2002, 128, 650–660. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Qin, G.; Zhang, J.; Liang, Y.; Song, Y.; Zhao, M.; Tsuge, T.; Aoyama, T.; Liu, J.; Gu, H.; et al. D-myo-inositol-3-phosphate affects phosphatidylinositol-mediated endomembrane function in Arabidopsis and is essential for auxin-regulated embryogenesis. Plant Cell 2011, 23, 1352–1372. [Google Scholar] [CrossRef] [PubMed]
- Nunes, A.; Vianna, G.; Cuneo, F.; Amaya-Farfan, J.; de Capdeville, G.; Rech, E.; Aragao, F. Rnai-mediated silencing of the myo-inositol-1-phosphate synthase gene (GmMIPS1) in transgenic soybean inhibited seed development and reduced phytate content. Planta 2006, 224, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Kuwano, M.; Mimura, T.; Takaiwa, F.; Yoshida, K. Generation of stable “low phytic acid” transgenic rice through antisense repression of the 1D-myo-inositol 3-phosphate synthase gene (RINO1) using the 18-kda oleosin promoter. Plant Biotechnol. J. 2009, 7, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Zhao, H.; Ren, X.; Zhu, S.; Fu, X.; Shu, Q. Generation and characterization of two novel low phytate mutations in soybean (Glycine max L. Merr.). Theor. Appl. Genet. 2007, 115, 945–957. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.; Otto, B.; Brearley, C.; Carr, J.; Hanke, D. A role for inositol hexakisphosphate in the maintenance of basal resistance to plant pathogens. Plant J. 2008, 56, 638–652. [Google Scholar] [CrossRef] [PubMed]
- Mitsuhashi, N.; Kondo, M.; Nakaune, S.; Ohnishi, M.; Hayashi, M.; Hara-Nishimura, I.; Richardson, A.; Fukaki, H.; Nishimura, M.; Mimura, T. Localization of myo-inositol-1-phosphate synthase to the endosperm in developing seeds of Arabidopsis. J. Exp. Bot. 2008, 59, 3069–3076. [Google Scholar] [CrossRef] [PubMed]
- Chiera, J.; Grabau, E. Localization of myo-inositol phosphate synthase (GmMIPS-1) during the early stages of soybean seed development. J. Exp. Bot. 2007, 58, 2261–2268. [Google Scholar] [CrossRef] [PubMed]
- Fu, J.; Peterson, K.; Guttieri, M.; Souza, E.; Raboy, V. Barley (Hordeum vulgare L.) inositol monophosphatase: Gene structure and enzyme characteristics. Plant Mol. Biol. 2008, 67, 629–642. [Google Scholar] [CrossRef] [PubMed]
- Gillaspy, G.E.; Keddie, J.S.; Oda, K.; Gruissem, W. Plant inositol monophosphatase is a lithium-sensitive enzyme encoded by a multigene family. Plant Cell 1995, 7, 2175–2185. [Google Scholar] [CrossRef] [PubMed]
- Nourbakhsh, A.; Collakova, E.; Gillaspy, G.E. Characterization of the inositol monophosphatase gene family in Arabidopsis. Front. Plant Sci. 2014, 5, 725. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Yazawa, K.; Yoshida, S.; Tamaoki, M.; Nakajima, N.; Iwai, H.; Ishii, T.; Satoh, S. Expression and functions of myo-inositol monophosphatase family genes in seed development of Arabidopsis. J. Plant Res. 2011, 124, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Stevenson-Paulik, J.; Bastidas, R.; Chiou, S.; Frye, R.; York, J. Generation of phytate-free seeds in Arabidopsis through disruption of inositol polyphosphate kinases. Proc. Natl. Acad. Sci. USA 2005, 102, 12612–12617. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Brearley, C.A.; Lin, W.H.; Wang, Y.; Ye, R.; Mueller-Roeber, B.; Xu, Z.H.; Xue, H.W. A role of Arabidopsis inositol polyphosphate kinase, AtIPK2α, in pollen germination and root growth. Plant Physiol. 2005, 137, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.B.; Yang, G.; Arana, F.; Chen, Z.; Li, Y.; Xia, H.J. Arabidopsis inositol polyphosphate 6-/3-kinase (AtIPK2β) is involved in axillary shoot branching via auxin signaling. Plant Physiol. 2007, 144, 942–951. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Tang, R.; Zhu, J.; Liu, H.; Mueller-Roeber, B.; Xia, H.; Zhang, H. Enhancement of stress tolerance in transgenic tobacco plants constitutively expressing AtIPK2β, an inositol polyphosphate 6-/3-kinase from Arabidopsis thaliana. Plant Mol. Biol. 2008, 66, 329–343. [Google Scholar] [CrossRef] [PubMed]
- Wilson, M.; Majerus, P. Characterization of a cDNA encoding Arabidopsis thaliana inositol 1,3,4-trisphosphate 5/6-kinase. Biochem. Biophys. Res. Commun. 1997, 232, 678–681. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Niu, X.; Chai, M.; Chen, J.; Liu, Q.; Wang, X. Isolation of an Arabidopsis gene encoding Ins (1,3,4) p-3 5/6-kinase-like protein and involved in plant response to abiotic stresses. Acta Bot. Sin. 2003, 45, 211–218. [Google Scholar]
- Raboy, V.; Gerbasi, P.; Young, K.; Stoneberg, S.; Pickett, S.; Bauman, A.; Murthy, P.; Sheridan, W.; Ertl, D. Origin and seed phenotype of maize low phytic acid 1-1 and low phytic acid 2-1. Plant Physiol. 2000, 124, 355–368. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wang, H.; Wu, Y.; Hazebroek, J.; Meeley, R.; Ertl, D. The maize low-phytic acid mutant 1pa2 is caused by mutation in an inositol phosphate kinase gene. Plant Physiol. 2003, 131, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Tai, T. Identification of novel rice low phytic acid mutations via TILLING by sequencing. Mol. Breed. 2014, 34, 1717–1729. [Google Scholar] [CrossRef]
- Qin, Z.X.; Chen, Q.J.; Tong, Z.; Wang, X.C. The Arabidopsis inositol 1,3,4-trisphosphate 5/6 kinase, AtITPK-1, is involved in plant photomorphogenesis under red light conditions, possibly via interaction with COP9 signalosome. Plant Physiol. Biochem. 2005, 43, 947–954. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Zhu, D.; Tan, Y.; Dong, D.; Fu, X.; Zhu, S.; Li, B.; Shu, Q. Identification and characterization of the soybean IPK1 ortholog of a low phytic acid mutant reveals an exon-excluding splice-site mutation. Theor. Appl. Genet. 2012, 125, 1413–1423. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Wang, H.; Schellin, K.; Li, B.; Faller, M.; Stoop, J.; Meeley, R.; Ertl, D.; Ranch, J.; Glassman, K. Embryo-specific silencing of a transporter reduces phytic acid content of maize and soybean seeds. Nat. Biotech. 2007, 25, 930–937. [Google Scholar] [CrossRef] [PubMed]
- Nagy, R.; Grob, H.; Weder, B.; Green, P.; Klein, M.; Frelet-Barrand, A.; Schjoerring, J.; Brearley, C.; Martinoia, E. The Arabidopsis ATP-binding cassette protein AtMRP5/AtABCC5 is a high affinity inositol hexakisphosphate transporter involved in guard cell signaling and phytate storage. J. Biol. Chem. 2009, 284, 33614–33622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klein, M.; Perfus-Barbeoch, L.; Frelet, A.; Gaedeke, N.; Reinhardt, D.; Mueller-Roeber, B.; Martinoia, E.; Forestier, C. The plant multidrug resistance ABC transporter AtMRP5 is involved in guard cell hormonal signalling and water use. Plant J. 2003, 33, 119–129. [Google Scholar] [CrossRef] [PubMed]
- Suh, S.J.; Wang, Y.F.; Frelet, A.; Leonhardt, N.; Klein, M.; Forestier, C.; Mueller-Roeber, B.; Cho, M.H.; Martinoia, E.; Schroeder, J.I. The ATP binding cassette transporter AtMRP5 modulates anion and calcium channel activities in Arabidopsis guard cells. J. Biol. Chem. 2007, 282, 1916–1924. [Google Scholar] [CrossRef] [PubMed]
- Klein, M.; Burla, B.; Martinoia, E. The multidrug resistance-associated protein (MRP/ABCC) subfamily of ATP-binding cassette transporters in plants. FEBS Lett. 2006, 580, 1112–1122. [Google Scholar] [CrossRef] [PubMed]
- Wanke, D.; Kolukisaoglu, H. An update on the ABCC transporter family in plants: Many genes, many proteins, but how many functions? Plant Biol. 2010, 12, 15–25. [Google Scholar] [PubMed]
- Kang, J.; Park, J.; Choi, H.; Burla, B.; Kretzschmar, T.; Lee, Y.; Martinoia, E. Plant ABC transporters. Arab. Book 2011, 9, e0153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, X.; Zhao, H.; Liu, Q.; Frank, T.; Engel, K.; An, G.; Shu, Q. Mutations of the multi-drug resistance-associated protein ABC transporter gene 5 result in reduction of phytic acid in rice seeds. Theor. Appl. Genet. 2009, 119, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Gaedeke, N.; Klein, M.; Kolukisaoglu, U.; Forestier, C.; Muller, A.; Ansorge, M.; Becker, D.; Mamnun, Y.; Kuchler, K.; Schulz, B.; et al. The Arabidopsis thaliana ABC transporter AtMRP5 controls root development and stomata movement. EMBO J. 2001, 20, 1875–1887. [Google Scholar] [CrossRef] [PubMed]
- Winter, D.; Vinegar, B.; Nahal, H.; Ammar, R.; Wilson, G.; Provart, N. An “electronic fluorescent pictograph” browser for exploring and analyzing large-scale biological data sets. PLoS ONE 2007, 2. [Google Scholar] [CrossRef] [PubMed]
- Panzeri, D.; Cassani, E.; Doria, E.; Tagliabue, G.; Forti, L.; Campion, B.; Bollini, R.; Brearley, C.A.; Pilu, R.; Nielsen, E.; et al. A defective ABC transporter of the MRP family, responsible for the bean lpa1 mutation, affects the regulation of the phytic acid pathway, reduces seed myo-inositol and alters aba sensitivity. New Phytol. 2011, 191, 70–83. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillman, J.; Pantalone, V.; Bilyeu, K. The low phytic acid phenotype in soybean line CX1834 is due to mutations in two homologs of the maize low phytic acid gene. Plant Genome 2009, 2, 179–190. [Google Scholar] [CrossRef]
- Patel, R.; Nahal, H.; Breit, R.; Provart, N. Bar expressolog identification: Expression profile similarity ranking of homologous genes in plant species. Plant J. 2012, 71, 1038–1050. [Google Scholar] [CrossRef] [PubMed]
- Pilu, R.; Panzeri, D.; Gavazzi, G.; Rasmussen, S.; Consonni, G.; Nielsen, E. Phenotypic, genetic and molecular characterization of a maize low phytic acid mutant (lpa241). Theor. Appl. Genet. 2003, 107, 980–987. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; VanToai, T.; Pratt, R. Expression and nucleotide sequence of an Ins (3) P-1 synthase gene associated with low-phytate kernels in maize (Zea mays L.). J. Agric. Food Chem. 2004, 52, 4565–4570. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Xu, X.; Ren, X.; Fu, H.; Wu, D.; Shu, Q. Generation and characterization of low phytic acid germplasm in rice (Oryza sativa L.). Theor. Appl. Genet. 2007, 114, 803–814. [Google Scholar] [CrossRef] [PubMed]
- Campion, B.; Sparvoli, F.; Doria, E.; Tagliabue, G.; Galasso, I.; Fileppi, M.; Bollini, R.; Nielsen, E. Isolation and characterisation of an lpa (low phytic acid) mutant in common bean (Phaseolus vulgaris L.). Theor. Appl. Genet. 2009, 118, 1211–1221. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, J.; Premachandra, G.; Young, K.; Raboy, V. Isolation of high seed inorganic P, low-phytate soybean mutants. Crop Sci. 2000, 40, 1601–1605. [Google Scholar] [CrossRef]
- Maroof, M.; Glover, N.; Biyashev, R.; Buss, G.; Grabau, E. Genetic basis of the low-phytate trait in the soybean line CX1834. Crop Sci. 2009, 49, 69–76. [Google Scholar] [CrossRef]
- Gillman, J.; Baxter, I.; Bilyeu, K. Phosphorus partitioning of soybean lines containing different mutant alleles of two soybean seed-specific adenosine triphosphate-binding cassette phytic acid transporter paralogs. Plant Genome 2013, 6. [Google Scholar] [CrossRef]
- Meis, S.; Fehr, W.; Schnebly, S. Seed source effect on field emergence of soybean lines with reduced phytate and raffinose saccharides. Crop Sci. 2003, 43, 1336–1339. [Google Scholar] [CrossRef]
- Guttieri, M.; Bowen, D.; Dorsch, J.; Raboy, V.; Souza, E. Identification and characterization of a low phytic acid wheat. Crop Sci. 2004, 44, 418–424. [Google Scholar] [CrossRef]
- Pilu, R.; Landoni, M.; Cassani, E.; Doria, E.; Nielsen, E. The maize lpa241 mutation causes a remarkable variability of expression and some pleiotropic effects. Crop Sci. 2005, 45, 2096–2105. [Google Scholar] [CrossRef]
- Bregitzer, P.; Raboy, V. Effects of four independent low-phytate mutations in barley (Hordeum vulgare L.) on seed phosphorus characteristics and malting quality. Cereal Chem. 2006, 83, 460–464. [Google Scholar] [CrossRef]
- Campion, B.; Glahn, R.; Tava, A.; Perrone, D.; Doria, E.; Sparvoli, F.; Cecotti, R.; Dani, V.; Nielsen, E. Genetic reduction of antinutrients in common bean (Phaseolus vulgaris L.) seed, increases nutrients and in vitro iron bioavailability without depressing main agronomic traits. Field Crop. Res. 2013, 141, 27–37. [Google Scholar] [CrossRef]
- Ertl, D.; Young, K.; Raboy, V. Plant genetic approaches to phosphorus management in agricultural production. J. Environ. Qual. 1998, 27, 299–304. [Google Scholar] [CrossRef]
- Bregitzer, P.; Raboy, V. Effects of four independent low-phytate mutations on barley agronomic performance. Crop Sci. 2006, 46, 1318–1322. [Google Scholar] [CrossRef]
- Raboy, V.; Peterson, K.; Jackson, C.; Marshall, J.; Hu, G.; Saneoka, H.; Bregitzer, P. A substantial fraction of barley (Hordeum vulgare L.) low phytic acid mutations have little or no effect on yield across diverse production environments. Plants 2015, 4, 225–239. [Google Scholar] [CrossRef]
- Bregitzer, P.; Raboy, V.; Obert, D.; Windes, J.; Whitmore, J. Registration of “Herald” barley. Crop Sci. 2007, 47, 441–442. [Google Scholar] [CrossRef]
- Bregitzer, P.; Raboy, V.; Obert, D.; Windes, J.; Whitmore, J. Registration of “Clearwater” low-phytate hulless spring barley. J. Plant Regist. 2008, 2, 1–4. [Google Scholar] [CrossRef]
- Valluru, R.; van den Ende, W. Myo-inositol and beyond—Emerging networks under stress. Plant Sci. 2011, 181, 387–400. [Google Scholar] [CrossRef] [PubMed]
- Gillaspy, G. The cellular language of myo-inositol signaling. New Phytol. 2011, 192, 823–839. [Google Scholar] [CrossRef] [PubMed]
- Dieck, C.; Boss, W.; Perera, I. A role for phosphoinositides in regulating plant nuclear functions. Front. Plant Sci. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Gillaspy, G.E. The role of phosphoinositides and inositol phosphates in plant cell signaling. In Lipid-Mediated Protein Signaling; Capelluto, D., Ed.; Springer: Dordrecht, The Netherlands, 2013; Volume 991, pp. 141–157. [Google Scholar]
- Heilmann, M.; Heilmann, I. Plant phosphoinositides-complex networks controlling growth and adaption. Biochim. Biophys. Acta 2015, 1851, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Zhu, D.; Deng, B.; Fu, X.; Dong, D.; Zhu, S.; Li, B.; Shu, Q. Effects of two low phytic acid mutations on seed quality and nutritional traits in soybean (Glycine max L. Merr). J. Agric. Food Chem. 2009, 57, 3632–3638. [Google Scholar] [CrossRef] [PubMed]
- Meng, P.; Raynaud, C.; Tcherkez, G.; Blanchet, S.; Massoud, K.; Domenichini, S.; Henry, Y.; Soubigou-Taconnat, L.; Lelarge-Trouverie, C.; Saindrenan, P.; et al. Crosstalks between myo-inositol metabolism, programmed cell death and basal immunity in Arabidopsis. PLoS ONE 2009, 4. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Xiong, L. Myo-inositol-1-phosphate synthase is required for polar auxin transport and organ development. J. Biol. Chem. 2010, 285, 24238–24247. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ockenden, I.; Truax, M.; Lott, J. Phytic acid-phosphorus and other nutritionally important mineral nutrient elements in grains of wild-type and low phytic acid (lpa1-1) rice. Seed Sci. Res. 2004, 14, 109–116. [Google Scholar]
- Cerino Badone, F.; Amelotti, M.; Cassani, E.; Pilu, R. Study of low phytic acid1-7 (lpa1-7), a new ZmMRP4 mutation in maize. J. Hered. 2012, 103, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Doria, E.; Galleschi, L.; Calucci, L.; Pinzino, C.; Pilu, R.; Cassani, E.; Nielsen, E. Phytic acid prevents oxidative stress in seeds: Evidence from a maize (Zea mays L.) low phytic acid mutant. J. Exp. Bot. 2009, 60, 967–978. [Google Scholar] [CrossRef] [PubMed]
- Karner, U.; Peterbauer, T.; Raboy, V.; Jones, D.; Hedley, C.; Richter, A. Myo-inositol and sucrose concentrations affect the accumulation of raffinose family oligosaccharides in seeds. J. Exp. Bot. 2004, 55, 1981–1987. [Google Scholar] [CrossRef] [PubMed]
- Frank, T.; Meuleye, B.; Miller, A.; Shu, Q.; Engel, K. Metabolite profiling of two low phytic acid (lpa) rice mutants. J. Agric. Food Chem. 2007, 55, 11011–11019. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Yan, W.; Bock, C.; Nokhrina, K.; Keller, W.; Georges, F. Perturbing the metabolic dynamics of myo-inositol in developing brassica napus seeds through in vivo methylation impacts its utilization as phytate precursor and affects downstream metabolic pathways. BMC Plant Biol. 2013, 13, 84. [Google Scholar] [CrossRef] [PubMed]
- Keller, R.; Brearley, C.; Trethewey, R.; Muller-Rober, B. Reduced inositol content and altered morphology in transgenic potato plants inhibited for 1D-myo-inositol 3-phosphate synthase. Plant J. 1998, 16, 403–410. [Google Scholar] [CrossRef]
- Donahue, J.; Alford, S.; Torabinejad, J.; Kerwin, R.; Nourbakhsh, A.; Ray, W.; Hernick, M.; Huang, X.; Lyons, B.; Hein, P.; et al. The Arabidopsis thaliana myo-inositol 1-phosphate synthase1 gene is required for myo-inositol synthesis and suppression of cell death. Plant Cell 2010, 22, 888–903. [Google Scholar] [CrossRef] [PubMed]
- Burnette, R.; Gunesekera, B.; Gillaspy, G. An Arabidopsis inositol 5-phosphatase gain-of-function alters abscisic acid signaling. Plant Physiol. 2003, 132, 1011–1019. [Google Scholar] [CrossRef] [PubMed]
- Gunesekera, B.; Torabinejad, J.; Robinson, J.; Gillaspy, G. Inositol polyphosphate 5-phosphatases 1 and 2 are required for regulating seedling growth. Plant Physiol. 2007, 143, 1408–1417. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Lee, B.H.; Ishitani, M.; Lee, H.; Zhang, C.; Zhu, J.K. FIERY1 encoding an inositol polyphosphate 1-phosphatase is a negative regulator of abscisic acid and stress signaling in Arabidopsis. Genes Dev. 2001, 15, 1971–1984. [Google Scholar] [CrossRef] [PubMed]
- Ananieva, E.A.; Gillaspy, G.E. Switches in nutrient and inositol signaling. Plant Signal. Behav. 2009, 4, 304–306. [Google Scholar] [CrossRef] [PubMed]
- Loewus, F.A. Inositol and plant cell wall polysaccharide biogenesis. Subcell. Biochem. 2006, 39, 21–45. [Google Scholar] [PubMed]
- Lorence, A.; Chevone, B.I.; Mendes, P.; Nessler, C.L. Myo-inositol oxygenase offers a possible entry point into plant ascorbate biosynthesis. Plant Physiol. 2004, 134, 1200–1205. [Google Scholar] [CrossRef] [PubMed]
- Lemtiri-Chlieh, F.; MacRobbie, E.; Webb, A.; Manison, N.; Brownlee, C.; Skepper, J.; Chen, J.; Prestwich, G.; Brearley, C. Inositol hexakisphosphate mobilizes an endomembrane store of calcium in guard cells. Proc. Natl. Acad. Sci. USA 2003, 100, 10091–10095. [Google Scholar] [CrossRef] [PubMed]
- Sztein, A.; Cohen, J.; Cooke, T. Evolutionary patterns in the auxin metabolism of green plants. Int. J. Plant Sci. 2000, 161, 849–859. [Google Scholar] [CrossRef]
- Thole, J.; Nielsen, E. Phosphoinositides in plants: Novel functions in membrane trafficking. Curr.Opin. Plant Biol. 2008, 11, 620–631. [Google Scholar] [CrossRef] [PubMed]
- Munnik, T.; Vermeer, J. Osmotic stress-induced phosphoinositide and inositol phosphate signalling in plants. Plant Cell Environ. 2010, 33, 655–669. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.; Chang, T.; Chiang, S.; Wang, W.; Charng, Y.; Chiou, T. Arabidopsis inositol pentakisphosphate 2-kinase, AtIPK1, is required for growth and modulates phosphate homeostasis at the transcriptional level. Plant J. 2014, 80, 503–515. [Google Scholar] [CrossRef] [PubMed]
- Latrasse, D.; Jegu, T.; Meng, P.; Mazubert, C.; Hudik, E.; Delarue, M.; Charon, C.; Crespi, M.; Hirt, H.; Raynaud, C.; et al. Dual function of MIPS1 as a metabolic enzyme and transcriptional regulator. Nucl. Acids Res. 2013, 41, 2907–2917. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Lee, D.; Cho, H.; Kim, S.; Auh, J.; Pai, H. InsP6-sensitive variants of the Gle1 mRNA export factor rescue growth and fertility defects of the ipk1 low-phytic-acid mutation in Arabidopsis. Plant Cell 2015, 27, 417–431. [Google Scholar] [CrossRef] [PubMed]
- Lemtiri-Chlieh, F.; MacRobbie, E.; Brearley, C. Inositol hexakisphosphate is a physiological signal regulating the K+-inward rectifying conductance in guard cells. Proc. Natl. Acad. Sci. USA 2000, 97, 8687–8692. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Calderon-Villalobos, L.I.; Sharon, M.; Zheng, C.; Robinson, C.V.; Estelle, M.; Zheng, N. Mechanism of auxin perception by the tir1 ubiquitin ligase. Nature 2007, 446, 640–645. [Google Scholar] [CrossRef] [PubMed]
- Laha, D.; Johnen, P.; Azevedo, C.; Dynowski, M.; Weiss, M.; Capolicchio, S.; Mao, H.; Iven, T.; Steenbergen, M.; Freyer, M.; et al. VIH2 regulates the synthesis of inositol pyrophosphate InsP8 and jasmonate-dependent defenses in Arabidopsis. Plant Cell 2015, 27, 1082–1097. [Google Scholar] [CrossRef] [PubMed]
- Fassetti, F.; Leone, O.; Palopoli, L.; Rombo, S.E.; Saiardi, A. IP6K gene identification in plant genomes by tag searching. BMC Proc. 2011, 5. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.; Rangarajan, P.; Donahue, J.; Williams, S.; Land, E.; Mandal, M.; Phillippy, B.; Perera, I.; Raboy, V.; Gillaspy, G. Two inositol hexakisphosphate kinases drive inositol pyrophosphate synthesis in plants. Plant J. 2014, 80, 642–653. [Google Scholar] [CrossRef] [PubMed]
- Shears, S.B. Inositol pyrophosphates: Why so many phosphates? Adv. Biol. Regul. 2015, 57, 203–216. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.; Torabinejad, J.; Cohick, E.; Parker, K.; Drake, E.; Thompson, J.; Hortter, M.; DeWald, D. Mutations in the Arabidopsis phosphoinositide phosphatase gene SAC9 lead to overaccumulation of Ptdins(4,5)P2 and constitutive expression of the stress-response pathway. Plant Physiol. 2005, 138, 686–700. [Google Scholar] [CrossRef] [PubMed]
- Perera, I.; Hung, C.; Moore, C.; Stevenson-Paulik, J.; Boss, W. Transgenic Arabidopsis plants expressing the type 1 inositol 5-phosphatase exhibit increased drought tolerance and altered abscisic acid signaling. Plant Cell 2008, 20, 2876–2893. [Google Scholar] [CrossRef] [PubMed]
- Im, Y.; Perera, I.; Brglez, I.; Davis, A.; Stevenson-Paulik, J.; Phillippy, B.; Johannes, E.; Allen, N.; Boss, W. Increasing plasma membrane phosphatidylinositol(4,5)bisphosphate biosynthesis increases phosphoinositide metabolism in nicotiana tabacum. Plant Cell 2007, 19, 1603–1616. [Google Scholar] [CrossRef] [PubMed]
- Perera, I.; Love, J.; Heilmann, I.; Thompson, W.; Boss, W. Up-regulation of phosphoinositide metabolism in tobacco cells constitutively expressing the human type i inositol polyphosphate 5-phosphatase. Plant Physiol. 2002, 129, 1795–1806. [Google Scholar] [CrossRef] [PubMed]
- Perera, I.; Hung, C.; Brady, S.; Muday, G.; Boss, W. A universal role for inositol 1,4,5-trisphosphate-mediated signaling in plant gravitropism. Plant Physiol. 2006, 140, 746–760. [Google Scholar] [CrossRef] [PubMed]
- Ananieva, E.; Gillaspy, G.; Ely, A.; Burnette, R.; Erickson, F. Interaction of the WD40 domain of a myoinositol polyphosphate 5-phosphatase with SnRK1 links inositol, sugar, and stress signaling. Plant Physiol. 2008, 148, 1868–1882. [Google Scholar] [CrossRef] [PubMed]
- Baena-Gonzalez, E.; Sheen, J. Convergent energy and stress signaling. Trends Plant Sci. 2008, 13, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Lin, W.; Wang, Y.; Luan, S.; Xue, H. An inositol polyphosphate 5-phosphatase functions in phototropin1 signaling in arabidopis by altering cytosolic Ca2+. Plant Cell 2008, 20, 353–366. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yang, X.; Tangchaiburana, S.; Ndeh, R.; Markham, J.; Tsegaye, Y.; Dunn, T.; Wang, G.; Bellizzi, M.; Parsons, J.; et al. An inositolphosphorylceramide synthase is involved in regulation of plant programmed cell death associated with defense in Arabidopsis. Plant Cell 2008, 20, 3163–3179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Vanneste, S.; Brewer, P.; Michniewicz, M.; Grones, P.; Kleine-Vehn, J.; Lofke, C.; Teichmann, T.; Bielach, A.; Cannoot, B.; et al. Inositol trisphosphate-induced Ca2+ signaling modulates auxin transport and pin polarity. Dev. Cell 2011, 20, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Bruggeman, Q.; Prunier, F.; Mazubert, C.; de Bont, L.; Garmier, M.; Lugan, R.; Benhamed, M.; Bergounioux, C.; Raynaud, C.; Delarue, M. Involvement of Arabidopsis hexokinase1 in cell death mediated by myo-inositol accumulation. Plant Cell 2015, 27, 1801–1814. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.; Aspesi, P.; Hunter, M.; Lomax, A.; Perera, I. Phosphoinositide-signaling is one component of a robust plant defense response. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Mosblech, A.; Thurow, C.; Gatz, C.; Feussner, I.; Heilmann, I. Jasmonic acid perception by coi1 involves inositol polyphosphates in Arabidopsis thaliana. Plant J. 2011, 65, 949–957. [Google Scholar] [CrossRef] [PubMed]
- Zhai, H.; Wang, F.; Si, Z.; Huo, J.; Xing, L.; An, Y.; He, S.; Liu, Q. A myo-inositol-1-phosphate synthase gene, IbMIPS1, enhances salt and drought tolerance and stem nematode resistance in transgenic sweet potato. Plant Biotechnol. J. 2015. [Google Scholar] [CrossRef] [PubMed]
- Alcazar-Roman, A.; Wente, S. Inositol polyphosphates: A new frontier for regulating gene expression. Chromosoma 2008, 117, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Brearley, C.; Elge, S.; Kaplan, B.; Fromm, H.; Mueller-Roeber, B. Arabidopsis inositol polyphosphate 6-/3-kinase is a nuclear protein that complements a yeast mutant lacking a functional ArgR-Mcm1 transcription complex. Plant Cell 2003, 15, 449–463. [Google Scholar] [CrossRef] [PubMed]
- Monserrate, J.; York, J. Inositol phosphate synthesis and the nuclear processes they affect. Curr. Opin. Cell Biol. 2010, 22, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Xiao, H.; Ranallo, R.; Wu, W.; Wu, C. Modulation of ATP-dependent chromatin-remodeling complexes by inositol polyphosphates. Science 2003, 299, 112–114. [Google Scholar] [CrossRef] [PubMed]
- Saiardi, A.; Caffrey, J.; Snyder, S.; Shears, S. Inositol polyphosphate multikinase (ArgRIII) determines nuclear mRNA export in Saccharomyces cerevisiae. FEBS Lett. 2000, 468, 28–32. [Google Scholar] [CrossRef]
- Steger, D.; Haswell, E.; Miller, A.; Wente, S.; O’Shea, E. Regulation of chromatin remodeling by inositol polyphosphates. Science 2003, 299, 114–116. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liao, H.; Lucas, W. Molecular mechanisms underlying phosphate sensing, signaling, and adaptation in plants. J. Integr. Plant Biol. 2014, 56, 192–220. [Google Scholar] [CrossRef] [PubMed]
- Belgaroui, N.; Zaidi, I.; Farhat, A.; Chouayekh, H.; Bouain, N.; Chay, S.; Curie, C.; Mari, S.; Masmoudi, K.; Davidian, J.; et al. Over-expression of the bacterial phytase US417 in Arabidopsis reduces the concentration of phytic acid and reveals its involvement in the regulation of sulfate and phosphate homeostasis and signaling. Plant Cell Physiol. 2014, 55, 1912–1924. [Google Scholar] [CrossRef] [PubMed]
- Sheard, L.; Tan, X.; Mao, H.; Withers, J.; Ben-Nissan, G.; Hinds, T.; Kobayashi, Y.; Hsu, F.; Sharon, M.; Browse, J.; et al. Jasmonate perception by inositol-phosphate-potentiated COI1-JAZ co-receptor. Nature 2010, 468, U400–U301. [Google Scholar] [CrossRef] [PubMed]
- Santner, A.; Estelle, M. Recent advances and emerging trends in plant hormone signalling. Nature 2009, 459, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sparvoli, F.; Cominelli, E. Seed Biofortification and Phytic Acid Reduction: A Conflict of Interest for the Plant? Plants 2015, 4, 728-755. https://doi.org/10.3390/plants4040728
Sparvoli F, Cominelli E. Seed Biofortification and Phytic Acid Reduction: A Conflict of Interest for the Plant? Plants. 2015; 4(4):728-755. https://doi.org/10.3390/plants4040728
Chicago/Turabian StyleSparvoli, Francesca, and Eleonora Cominelli. 2015. "Seed Biofortification and Phytic Acid Reduction: A Conflict of Interest for the Plant?" Plants 4, no. 4: 728-755. https://doi.org/10.3390/plants4040728
APA StyleSparvoli, F., & Cominelli, E. (2015). Seed Biofortification and Phytic Acid Reduction: A Conflict of Interest for the Plant? Plants, 4(4), 728-755. https://doi.org/10.3390/plants4040728






