Anthocyanin Synthesis Capability of Maize Cultivars Is Associated with Their Photosynthetic Carbon Partitioning for Growth Adaptability Under Low Phosphorus
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Treatment
2.2. Experimental Design
2.3. Plant Growth Parameters
2.4. Leaf Photosynthesis Rate
2.5. Carbohydrates and Anthocyanins
2.6. Statistical Analysis
3. Results
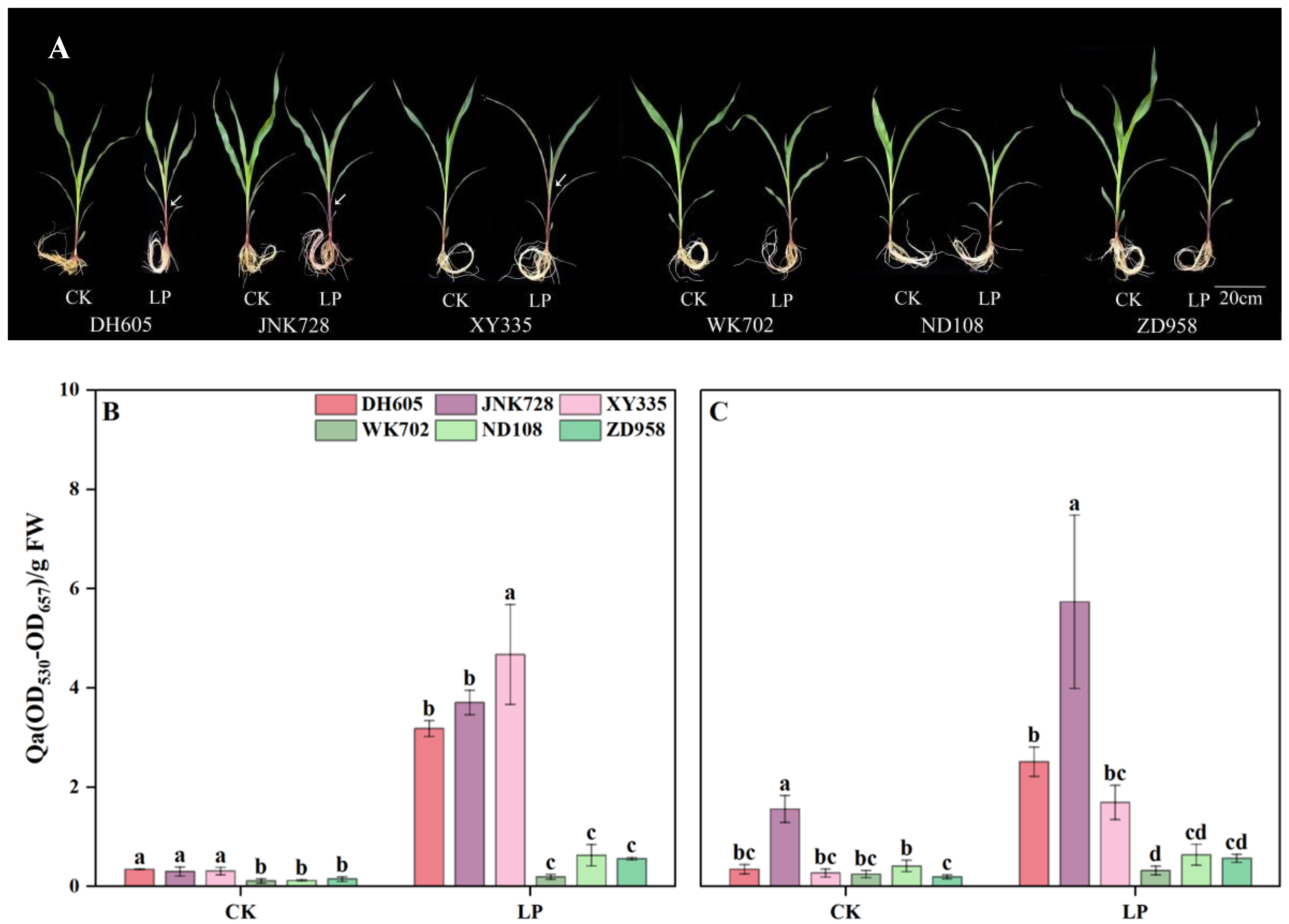
3.1. The Difference in Anthocyanin Accumulation Within Cultivars
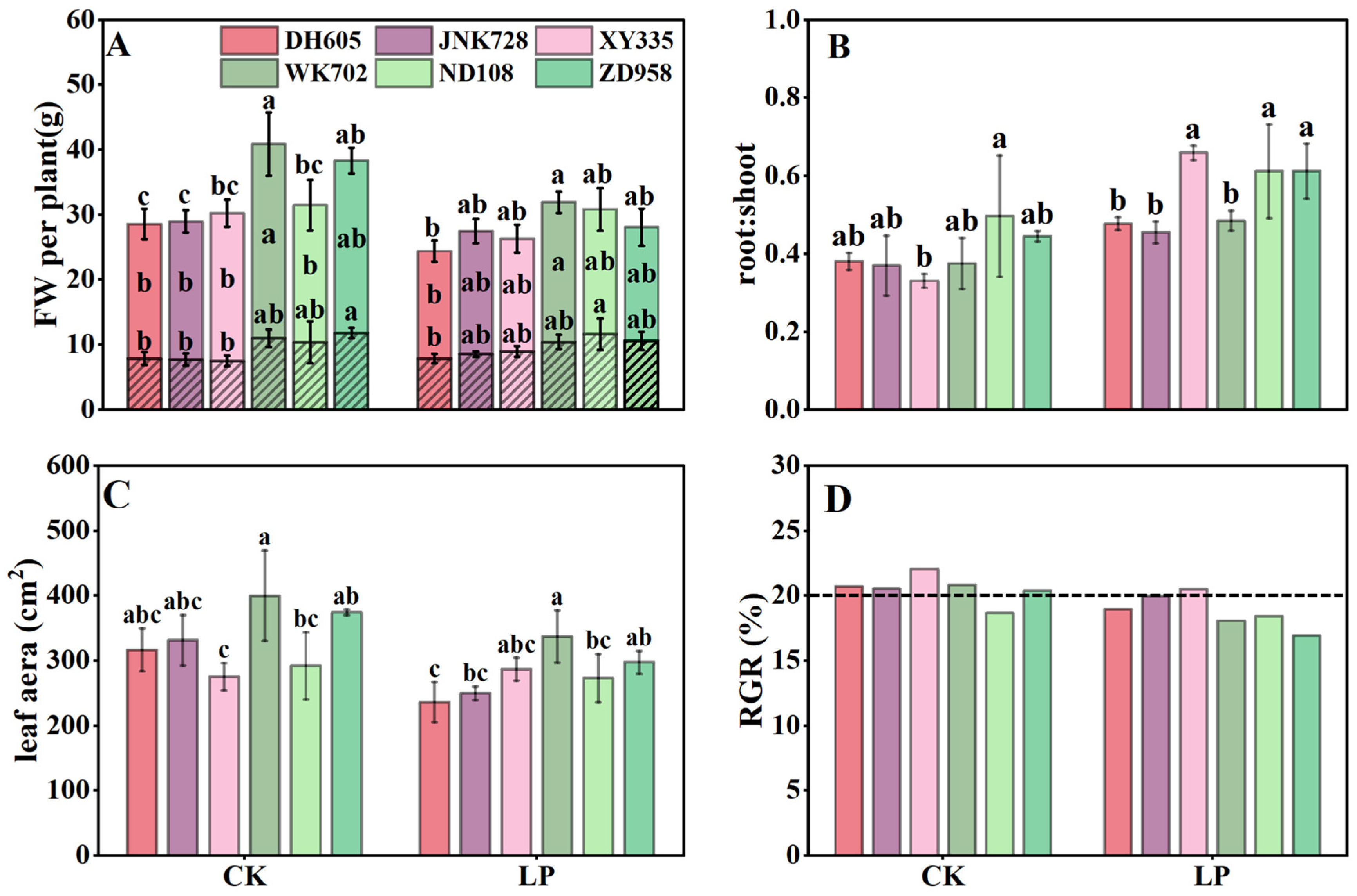
3.2. The Response of Plant Growth to P Deficiency
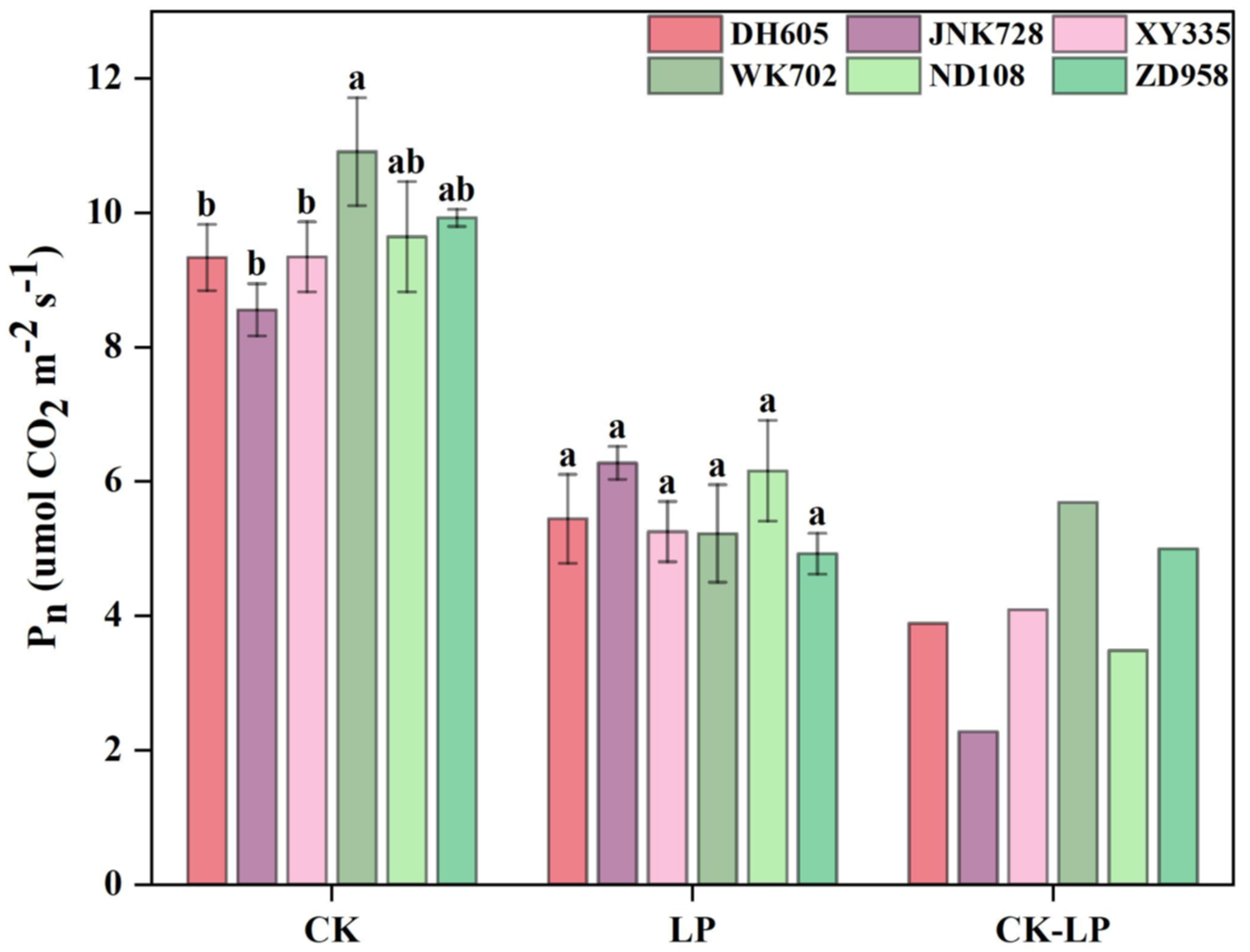
3.3. Leaf Photosynthesis Affected by LP
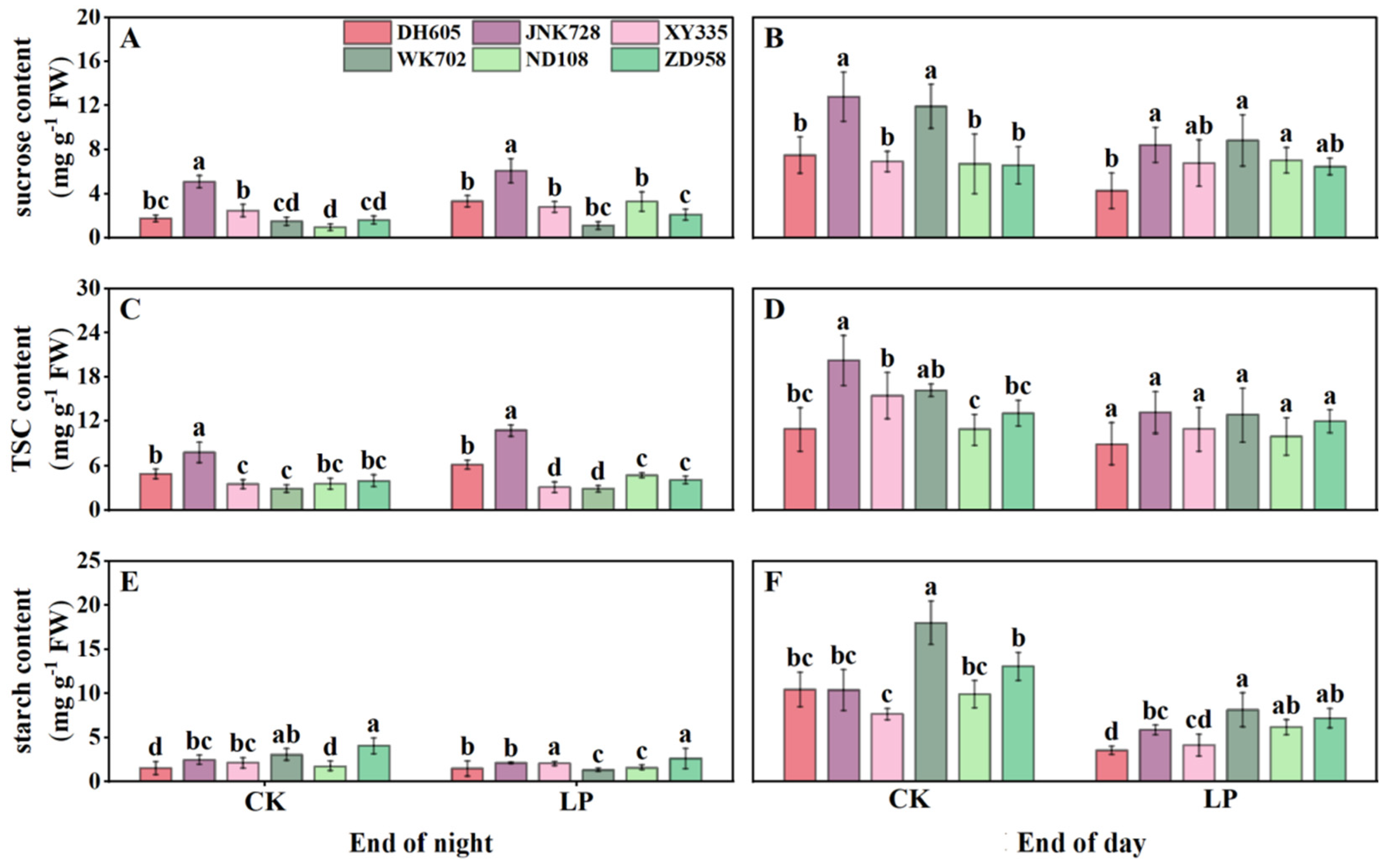
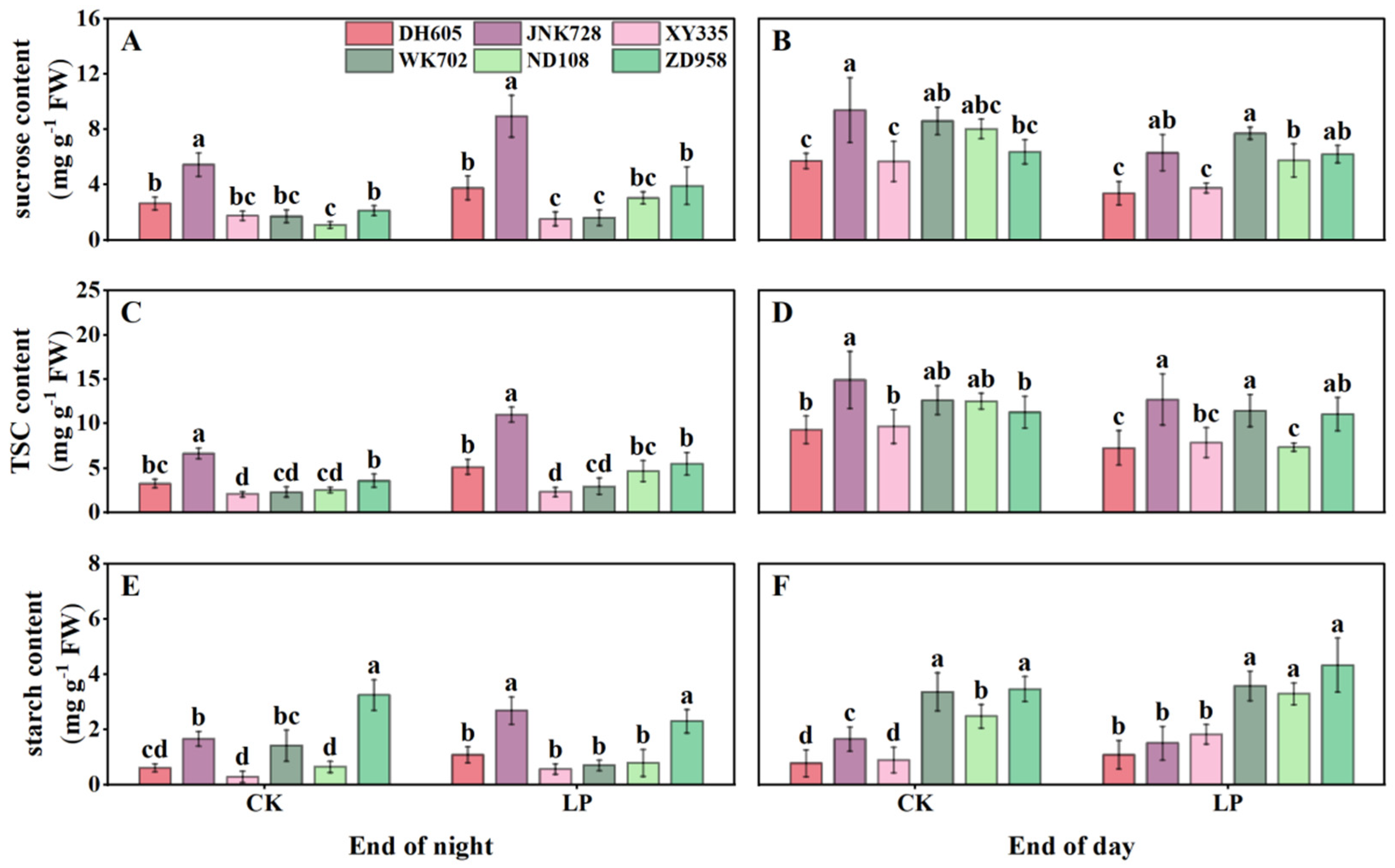
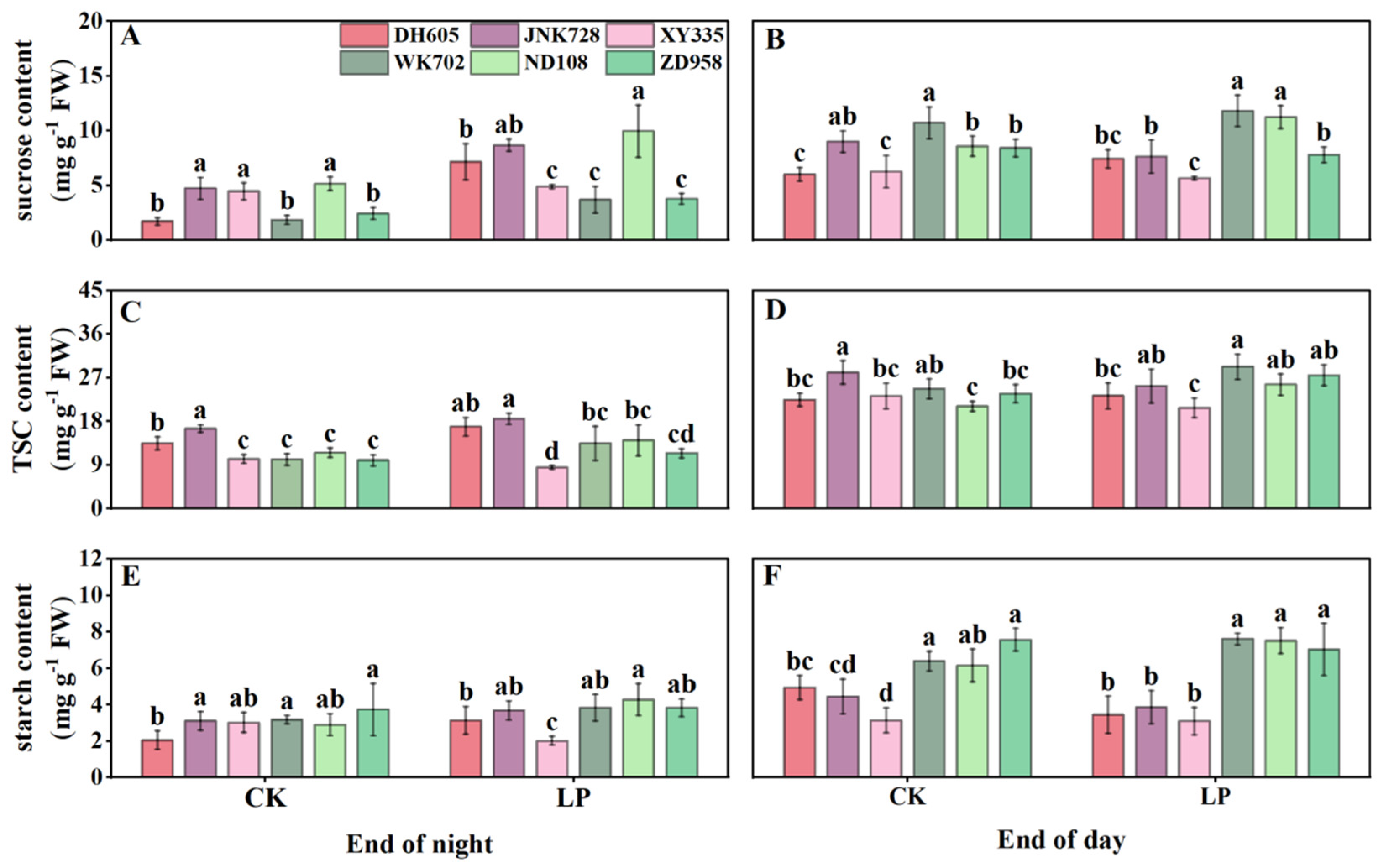
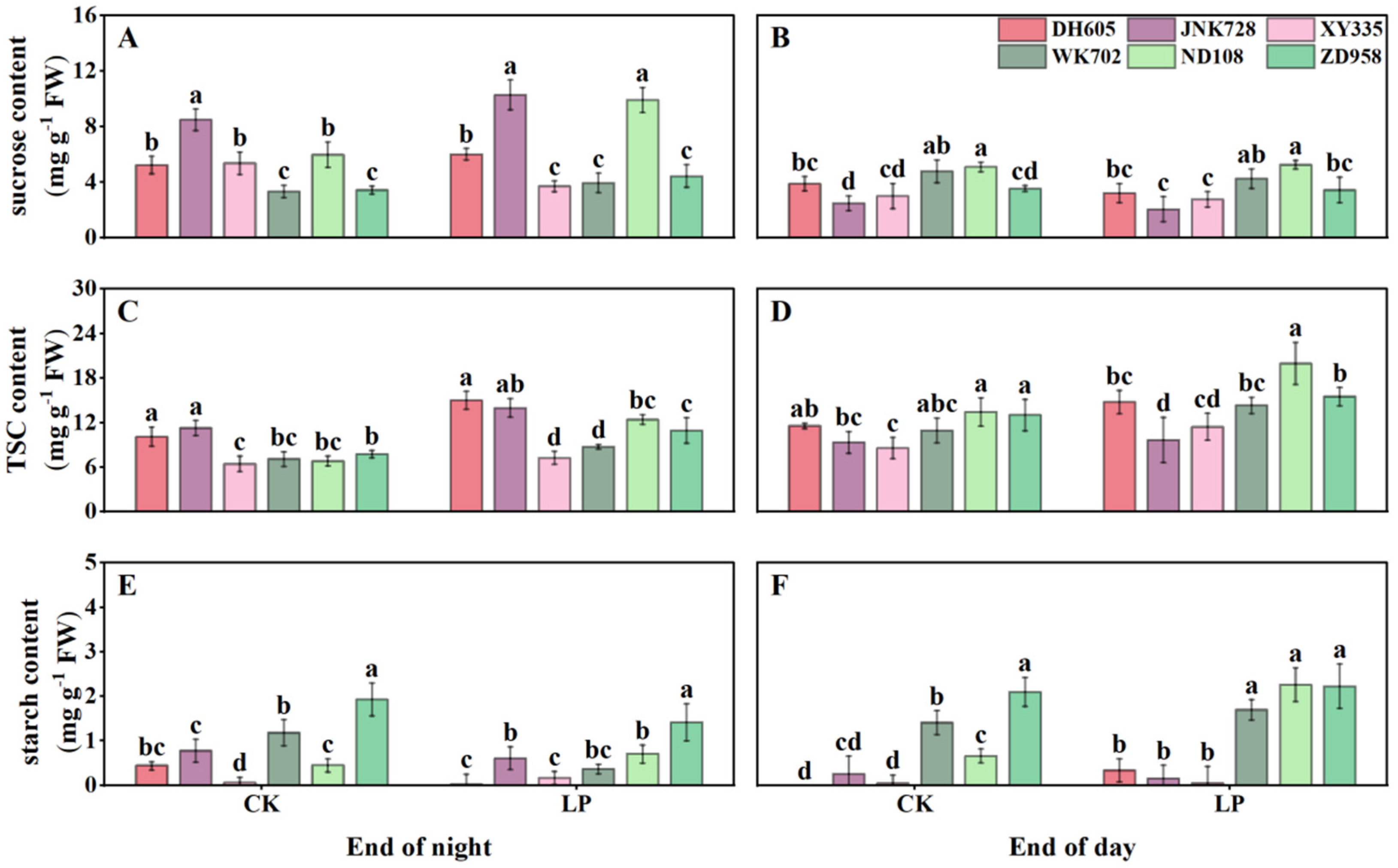
3.4. Carbohydrate Accumulation in the Fourth Leaf, Sheaths, Unexpanded New Leaves, and Roots
3.5. Correlations Among Anthocyanins, Plant Growth, Photosynthesis, and Tissue-Specific Carbohydrates
4. Discussion
4.1. The Association Between ACNs Accumulation and Carbon Partitioning for Different Cultivar Groups
4.2. Capability of ACNs Accumulation Is Associated with Photosynthesis and Growth for Different Cultivar Groups Under LP
4.3. The Stability of ACNs Synthesis Among Cultivars May Provide a Diagnostic Indicator for Growth or Resistance at an Early Stage
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACNs | Anthocyanins |
| CK | Control |
| FW | Fresh weight |
| LA | Leaf area |
| LP | Low phosphorus |
| RGR | Relative growth ratio |
| TSC | Total soluble sugars |
| DH605 | Denghai605 |
| JNK728 | Jingnongke728 |
| XY335 | Xianyu335 |
| ND108 | Nongda108 |
| WK702 | Weike702 |
| ZD958 | Zhengdan958 |
References
- Zhang, Y.; Butelli, E.; Martin, C. Engineering anthocyanin biosynthesis in plants. Curr. Opin. Plant Biol. 2014, 19, 81–90. [Google Scholar] [CrossRef]
- Riaz, B.; Chen, H.; Wang, J.; Du, L.; Wang, K.; Ye, X. Overexpression of maize ZmC1 and ZmR transcription factors in wheat regulates anthocyanin biosynthesis in a tissue-specific manner. Int. J. Mol. Sci. 2019, 20, 5806. [Google Scholar] [CrossRef]
- Li, P.; Li, Y.J.; Zhang, F.J.; Zhang, G.Z.; Jiang, X.Y.; Yu, H.M.; Hou, B.K. The Arabidopsis UDP-glycosyltransferases UGT79B2 and UGT79B3, contribute to cold, salt and drought stress tolerance via modulating anthocyanin accumulation. Plant J. 2017, 89, 85–103. [Google Scholar] [CrossRef] [PubMed]
- Naing, A.H.; Kim, C.K. Roles of R2R3-MYB transcription factors in transcriptional regulation of anthocyanin biosynthesis in horticultural plants. Plant Mol. Biol. 2018, 98, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, Z.; Wu, Y.; Zheng, L.; Zhang, G. Regulatory mechanisms of anthocyanin biosynthesis in apple and pear. Int. J. Mol. Sci. 2021, 22, 8441. [Google Scholar] [CrossRef] [PubMed]
- Yoon, H.S.; Yang, H.C.; Kang, H.M.; Jang, D.C. Mechanical stress-induced anthocyanin regulatory genes involved in anthocyanin accumulation in tomato plants. Hortic. Environ. Biotechnol. 2024, 65, 283–291. [Google Scholar] [CrossRef]
- Ma, Y.; Ma, X.; Gao, X.; Wu, W.; Zhou, B. Light induced regulation pathway of anthocyanin biosynthesis in plants. Int. J. Mol. Sci. 2021, 22, 11116. [Google Scholar] [CrossRef]
- Magaña Cerino, J.M.; Peniche Pavía, H.A.; Tiessen, A.; Gurrola Díaz, C.M. Pigmented maize (Zea mays L.) contains anthocyanins with potential therapeutic action against oxidative stress-a review. Pol. J. Food Nutr. Sci. 2020, 70, 85–99. [Google Scholar] [CrossRef]
- Paulsmeyer, M.N.; Vermillion, K.E.; Juvik, J.A. Assessing the diversity of anthocyanin composition in various tissues of purple corn (Zea mays L.). Phytochemistry 2022, 201, 113263. [Google Scholar] [CrossRef]
- Wu, Y.; Han, T.; Lyu, L.; Li, W.; Wu, W. Research progress in understanding the biosynthesis and regulation of plant anthocyanins. Sci. Hortic. 2023, 321, 112374. [Google Scholar] [CrossRef]
- Sun, Y.; Mu, C.; Chen, Y.; Kong, X.; Xu, Y.; Zheng, H.; Zhang, H.; Wang, Q.; Xue, Y.; Li, Z.; et al. Comparative transcript profiling of maize inbreds in response to long-term phosphorus deficiency stress. Plant Physiol. Biochem. 2016, 109, 467–481. [Google Scholar] [CrossRef]
- Wang, B.; Zhao, X.; Zhao, Y.; Shanklin, J.; Zhao, Q.; Liu, C.J. Arabidopsis SnRK1 negatively regulates phenylpropanoid metabolism via Kelch domain-containing F-box proteins. New Phytol. 2021, 229, 3345–3359. [Google Scholar] [CrossRef]
- Peniche-Pavía, H.A.; Guzmán, T.J.; Magaña-Cerino, J.M.; Gurrola-Díaz, C.M.; Tiessen, A. Maize flavonoid biosynthesis, regulation, and human health relevance: A review. Molecules 2022, 27, 5166. [Google Scholar] [CrossRef] [PubMed]
- Teng, S.; Keurentjes, J.; Bentsink, L.; Koornneef, M.; Smeekens, S. Sucrose-specific induction of anthocyanin biosynthesis in Arabidopsis requires the MYB75/PAP1 gene. Plant Physiol. 2005, 139, 1840–1852. [Google Scholar] [CrossRef] [PubMed]
- Solfanelli, C.; Poggi, A.; Loreti, E.; Alpi, A.; Perata, P. Sucrose-specific induction of the anthocyanin biosynthetic pathway in Arabidopsis. Plant Physiolgy 2006, 140, 637–646. [Google Scholar] [CrossRef]
- Das, P.K.; Shin, D.H.; Choi, S.B.; Park, Y.I. Sugar-hormone cross-talk in anthocyanin biosynthesis. Mol. Cells 2012, 34, 501–507. [Google Scholar] [CrossRef]
- Qin, Y.; Xiao, Z.; Zhao, H.; Wang, J.; Wang, Y.; Qiu, F. Starch phosphorylase 2 is essential for cellular carbohydrate partitioning in maize. J. Integr. Plant Biol. 2022, 64, 1755–1769. [Google Scholar] [CrossRef] [PubMed]
- Cheaib, A.; Mahmoud, L.M.; Vincent, C.; Killiny, N.; Dutt, M. Influence of anthocyanin expression on the performance of photosynthesis in sweet orange, Citrus sinensis (L.) Osbeck. Plants 2023, 12, 3965. [Google Scholar] [CrossRef]
- Kumari, V.V.; Banerjee, P.; Verma, V.C.; Sukumaran, S.; Chandran, M.A.S.; Gopinath, K.A.; Venkatesh, G.; Yadav, S.K.; Singh, V.K.; Awasthi, N.K. Plant nutrition: An effective way to alleviate abiotic stress in agricultural crops. Int. J. Mol. Sci. 2022, 23, 8519. [Google Scholar] [CrossRef]
- Wen, Z.; Li, H.; Shen, J.; Rengel, Z. Maize responds to low shoot P concentration by altering root morphology rather than increasing root exudation. Plant Soil 2017, 416, 377–389. [Google Scholar] [CrossRef]
- Xiao, Z.D.; Chen, Z.Y.; Lin, Y.H.; Liang, X.G.; Wang, X.; Huang, S.B.; Munz, S.; Graeff-Hönninger, S.; Shen, S.; Zhou, S.L. Phosphorus deficiency promotes root: Shoot ratio and carbon accumulation via modulating sucrose utilization in maize. J. Plant Physiol. 2024, 303, 154349. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.G.; Wang, K.; Xu, C.; Zou, C.; Xie, C.X.; Xu, Y.B.; Li, W.X. Strand-specific RNA-Seq transcriptome analysis of genotypes with and without low-phosphorus tolerance provides novel insights into phosphorus-use efficiency in maize. BMC Plant Biol. 2016, 16, 222. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, I.; Kolla, M.; Volker, R.; Günter, N. Impact of nutrient seed priming on germination, seedling development, nutritional status and grain yield of maize. J. Plant Nutr. 2015, 38, 1803–1821. [Google Scholar] [CrossRef]
- Liang, X.G.; Shen, S.; Gao, Z.; Zhang, L.; Zhao, X.; Zhou, S.L. Variation of carbon partitioning in newly expanded maize leaves and plant adaptive growth under extended darkness. J. Integr. Agric. 2021, 20, 2360–2371. [Google Scholar] [CrossRef]
- Dubious, M. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–366. [Google Scholar] [CrossRef]
- Wang, Y.; Tao, H.; Tian, B.; Sheng, D.; Xu, C.; Zhou, H.; Huang, S.; Wang, P. Flowering dynamics, pollen, and pistil contribution to grain yield in response to high temperature during maize flowering. Environ. Exp. Bot. 2019, 158, 80–88. [Google Scholar] [CrossRef]
- He, Y.; Zhang, X.; Li, L.; Sun, Z.; Li, J.; Chen, X.; Hong, G. SPX4 interacts with both PHR1 and PAP1 to regulate critical steps in phosphorus-status-dependent anthocyanin biosynthesis. New Phytol. 2021, 230, 205–217. [Google Scholar] [CrossRef]
- Malhotra, H.; Vandana; Sharma, S.; Pandey, R. Phosphorus nutrition: Plant growth in response to deficiency and excess. In Plant Nutrients and Abiotic Stress Tolerance; Springer: Singapore, 2018; pp. 171–190. [Google Scholar]
- Liang, X.G.; Gao, Z.; Shen, S.; Paul, M.J.; Zhang, L.; Zhao, X.; Lin, S.; Wu, G.; Chen, X.M.; Zhou, S.L. Differential ear growth of two maize varieties to shading in the field environment: Effects on whole plant carbon allocation and sugar starvation response. J. Plant Physiol. 2020, 251, 153194. [Google Scholar] [CrossRef]
- Liang, X.G.; Gao, Z.; Zhang, L.; Shen, S.; Zhao, X.; Liu, Y.P.; Zhou, L.L.; Paul, M.J.; Zhou, S.L. Seasonal and diurnal patterns of non-structural carbohydrates in source and sink tissues in field maize. BMC Plant Biol. 2019, 19, 508. [Google Scholar] [CrossRef]
- Zhang, Z.; Liang, Z.; Yin, L.; Li, Q.X.; Wu, Z. Distribution of four bioactive flavonoids in maize tissues of five varieties and correlation with expression of the biosynthetic genes. J. Agric. Food Chem. 2018, 66, 10431–10437. [Google Scholar] [CrossRef]
- Zhao, T.; Li, Q.; Yan, T.; Yu, B.; Wang, Q.; Wang, D. Sugar and anthocyanins: A scientific exploration of sweet signals and natural pigments. Plant Sci. 2025, 353, 112409. [Google Scholar] [CrossRef]
- Sadowska-Bartosz, I.; Bartosz, G. Antioxidant activity of anthocyanins and anthocyanidins: A critical review. Int. J. Mol. Sci. 2024, 25, 12001. [Google Scholar] [CrossRef]
- Pei, L.; Liu, J.; Zhou, Y.; Jiang, Y.; Li, H. Transcriptomic and metabolomic profiling reveals the protective role of anthocyanins in alleviating low phosphate stress in maize. Physiol. Mol. Biol. Plants 2021, 27, 889–905. [Google Scholar] [CrossRef]
- MacNeill, G.J.; Mehrpouyan, S.; Minow, M.A.; Patterson, J.A.; Tetlow, I.J.; Emes, M.J. Starch as a source, starch as a sink: The bifunctional role of starch in carbon allocation. J. Exp. Bot. 2017, 68, 4433–4453. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Dubos, C.; Lepiniec, L. Transcriptional control of flavonoid biosynthesis by MYB–bHLH–WDR complexes. Trends Plant Sci. 2015, 20, 176–185. [Google Scholar] [CrossRef] [PubMed]
- Cone, K.C.; Cocciolone, S.M.; Burr, F.A.; Burr, B. Maize anthocyanin regulatory gene pl is a duplicate of c1 that functions in the plant. Plant Cell 1993, 5, 1795–1805. [Google Scholar] [PubMed]
- Hollick, J.B.; Chandler, V.L. Genetic factors required to maintain repression of a paramutagenic maize pl1 allele. Genetics 2001, 157, 369–378. [Google Scholar] [CrossRef]
- Ma, R.; Liu, B.; Geng, X.; Ding, X.; Yan, N.; Sun, X.; Wang, W.L.; Sun, X.Z. Biological function and stress response mechanism of MYB transcription factor family genes. J. Plant Growth Regul. 2023, 42, 83–95. [Google Scholar] [CrossRef]
- Li, Z.G.; Wang, Y.R.; Ma, Y.W.; Xiang, Y.; Wu, S.G.; Qu, H.T.; Li, F.L.; Zhang, S.Q.; Li, G.F. Genetic analysis of color traits in sheath, silk, anther and cob of isolated population based on maize DH lines. Crops 2023, 39, 75–79, (In Chinese with English abstract). [Google Scholar]
- Liang, X.G.; Gao, Z.; Shen, S.; Zhao, X.; Zhang, L.; Zhou, S.L. Cob color, an indicator of grain dehydration and agronomic traits in maize hybrids. Crop J. 2022, 10, 582–586. [Google Scholar] [CrossRef]
- Joya-Dávila, J.G.; Gutiérrez-Miceli, F.A.; Serrano-Gómez, L.A.; Salazar-Navarro, A.; González-Mendoza, D.; Tzintzun-Camacho, O.; Santos-Espinoza, A.M.; Sánchez-Grajalez, G.; Gómez-Padilla, E.; Llaven-Martínez, J. Variability in anthocyanin expression in native maize: Purple totomoxtle as a phenotypic trait of agroecological value. Plants 2025, 14, 2511. [Google Scholar] [CrossRef]
- Chen, C.; Liu, X.Q.; Li, S.Z.; Lui, C.X.; Zhang, Y.L.; Luo, L.L.; Miao, L.Q.; Yang, W.Z.; Xiao, Z.J.; Zhou, Y.; et al. Co-expression of transcription factors ZmC1 and ZmR2 establishes an efficient and accurate haploid embryo identification system in maize. Plant J. 2022, 111, 1296–1307. [Google Scholar] [CrossRef] [PubMed]
- Zeng, M.; Wu, Y.; Lin, S.; Zhang, F.; Jiang, H.; Ma, L.; Liu, D. Disruption of ABI4 enhances anthocyanin accumulation in Arabidopsis seedlings through HY5-mediated light signaling. Plants 2025, 14, 1905. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, K.; Fu, C.; Wu, C.; Zuo, D.; Cheng, H.; Lv, L.; Zhao, H.; Wang, J.; Wu, C.; et al. Two B-Box proteins, GhBBX21 and GhBBX24, antagonistically modulate anthocyanin biosynthesis in R1 cotton. Plants 2025, 14, 2367. [Google Scholar] [CrossRef]
- Katerji, N.; Mastrorilli, M.; Lahmer, F.Z.; Oweis, T. Emergence rate as a potential indicator of crop salt-tolerance. Eur. J. Agron. 2012, 38, 1–9. [Google Scholar] [CrossRef]
- Qin, L.; Sun, L.; Wei, L.; Yuan, J.; Kong, F.; Zhang, Y.; Miao, X.; Xia, G.; Liu, S. Maize SRO1e represses anthocyanin synthesis through regulating the MBW complex in response to abiotic stress. Plant J. 2021, 105, 1010–1025. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, W.; Xiao, Z.-D.; Liang, X.-W.; Shen, S.; Liang, X.-G.; Zhou, S.-L. Anthocyanin Synthesis Capability of Maize Cultivars Is Associated with Their Photosynthetic Carbon Partitioning for Growth Adaptability Under Low Phosphorus. Plants 2025, 14, 2690. https://doi.org/10.3390/plants14172690
Tang W, Xiao Z-D, Liang X-W, Shen S, Liang X-G, Zhou S-L. Anthocyanin Synthesis Capability of Maize Cultivars Is Associated with Their Photosynthetic Carbon Partitioning for Growth Adaptability Under Low Phosphorus. Plants. 2025; 14(17):2690. https://doi.org/10.3390/plants14172690
Chicago/Turabian StyleTang, Wang, Zu-Dong Xiao, Xing-Wei Liang, Si Shen, Xiao-Gui Liang, and Shun-Li Zhou. 2025. "Anthocyanin Synthesis Capability of Maize Cultivars Is Associated with Their Photosynthetic Carbon Partitioning for Growth Adaptability Under Low Phosphorus" Plants 14, no. 17: 2690. https://doi.org/10.3390/plants14172690
APA StyleTang, W., Xiao, Z.-D., Liang, X.-W., Shen, S., Liang, X.-G., & Zhou, S.-L. (2025). Anthocyanin Synthesis Capability of Maize Cultivars Is Associated with Their Photosynthetic Carbon Partitioning for Growth Adaptability Under Low Phosphorus. Plants, 14(17), 2690. https://doi.org/10.3390/plants14172690






