Interspecific Competition and Intraspecific Facilitation Shape Coastal Dune Shrub Responses to Experimental Drought
Abstract
1. Introduction
2. Results
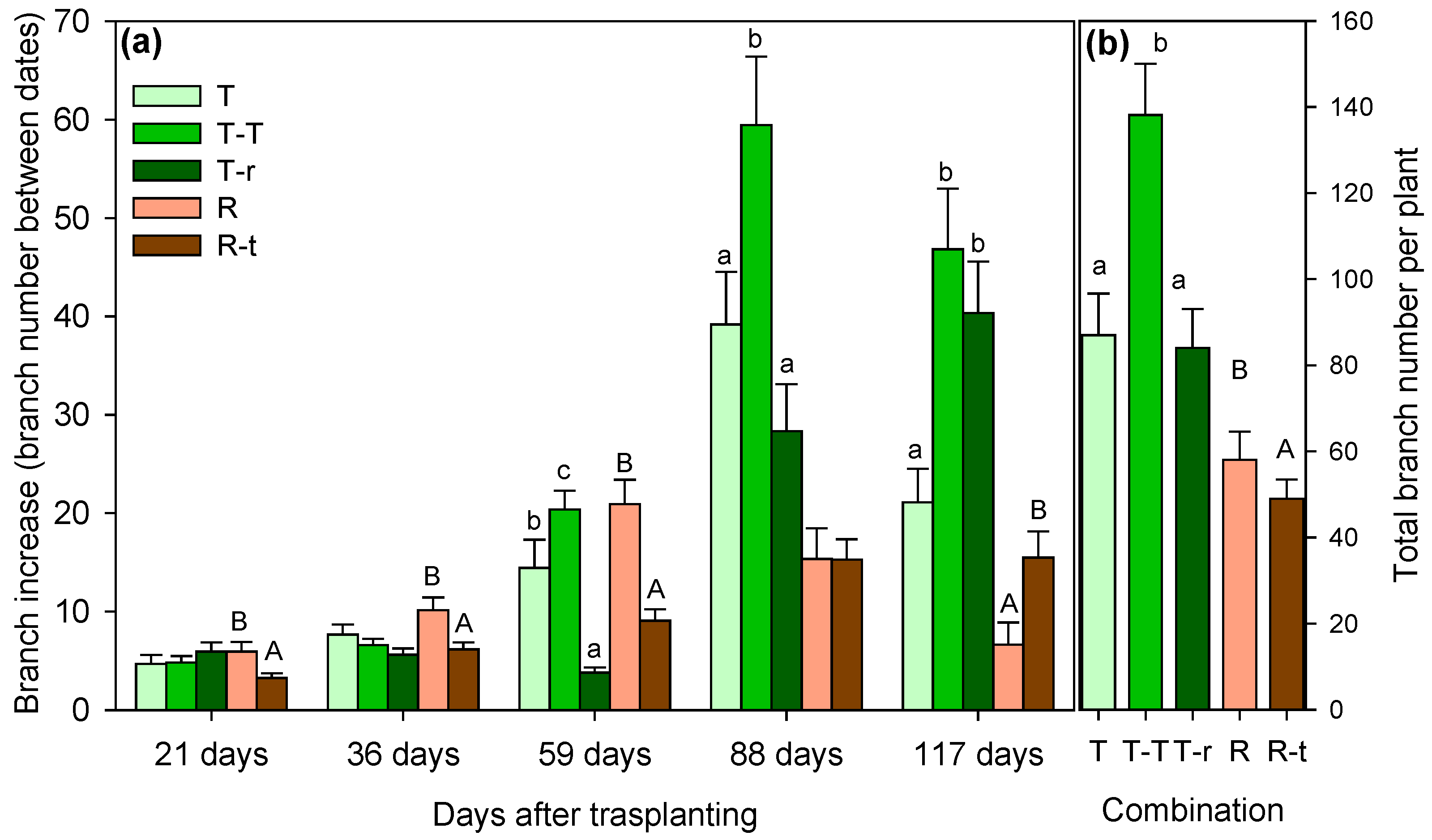
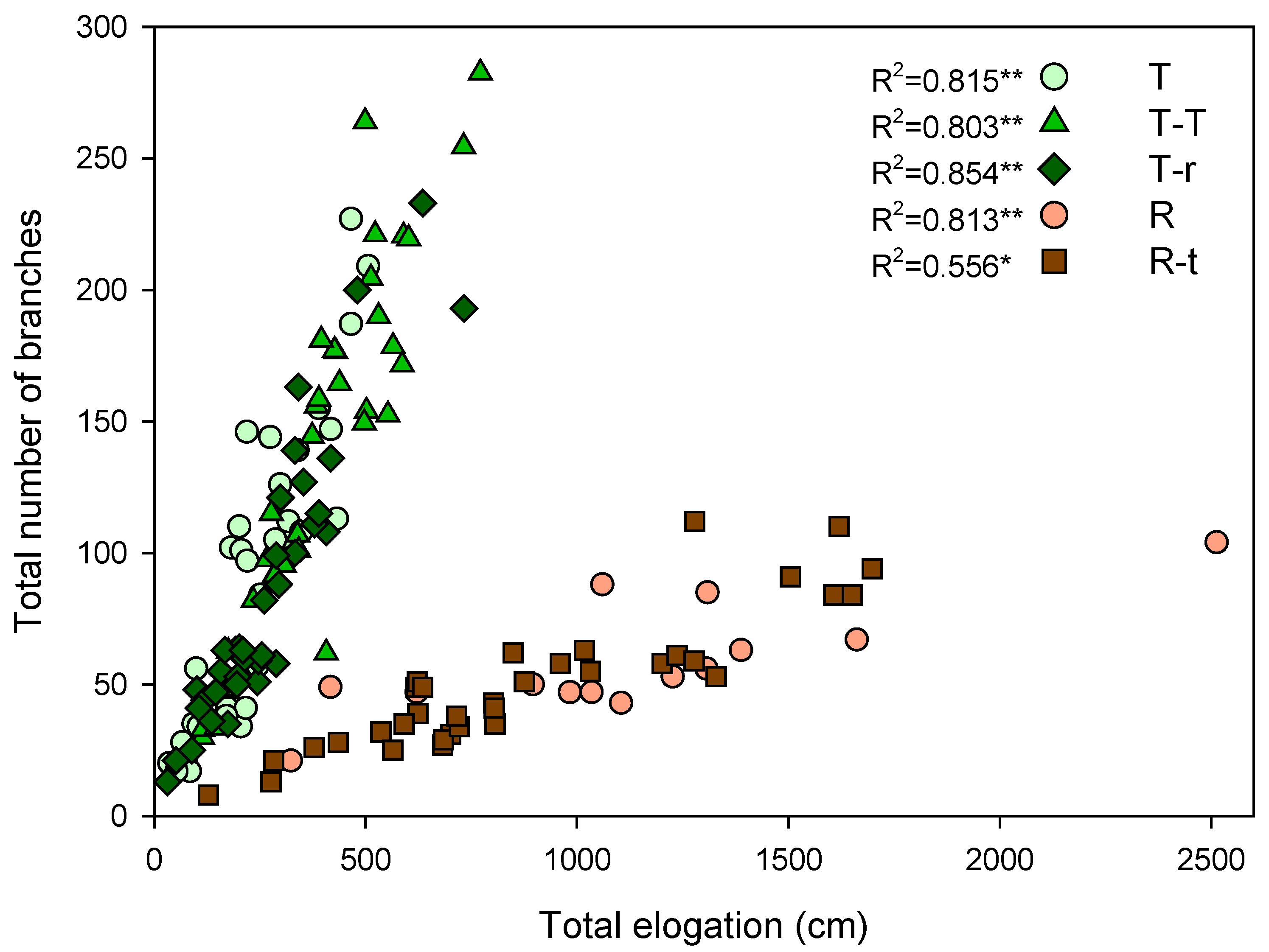
2.1. Growth Pattern Before the Water Restriction Experiment
2.2. Water Restriction Experiment
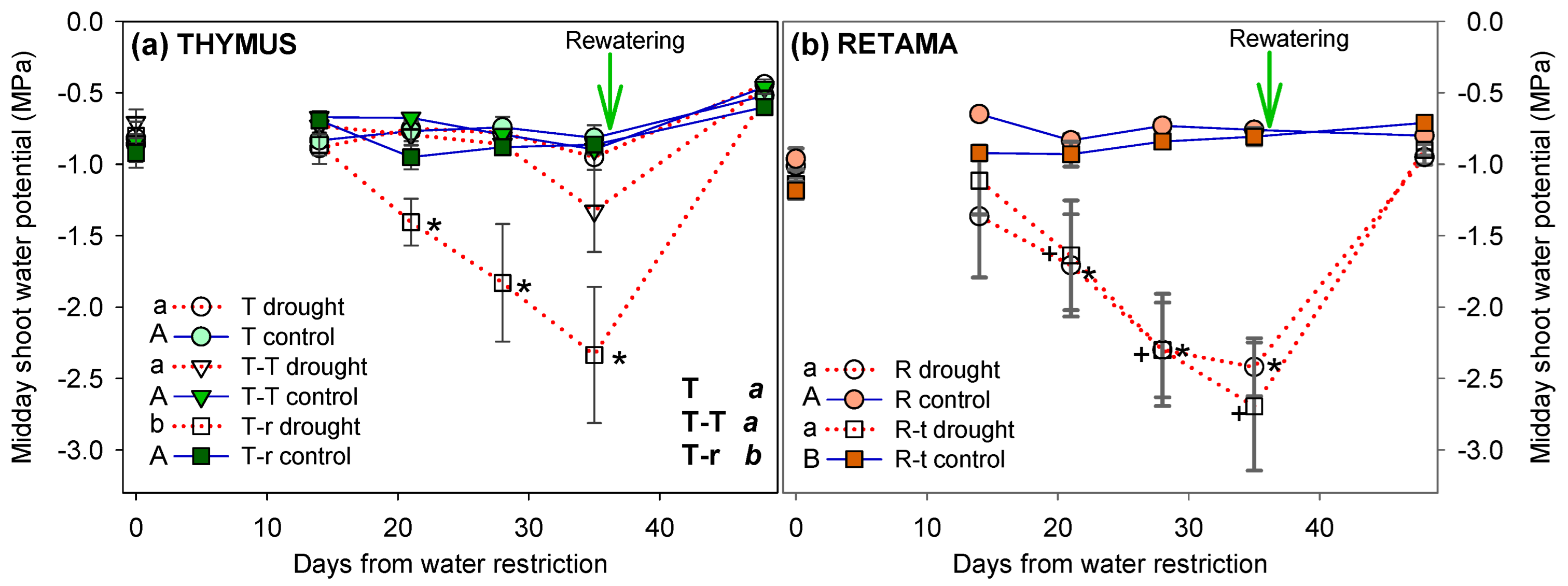
2.2.1. Shoot Water Potential
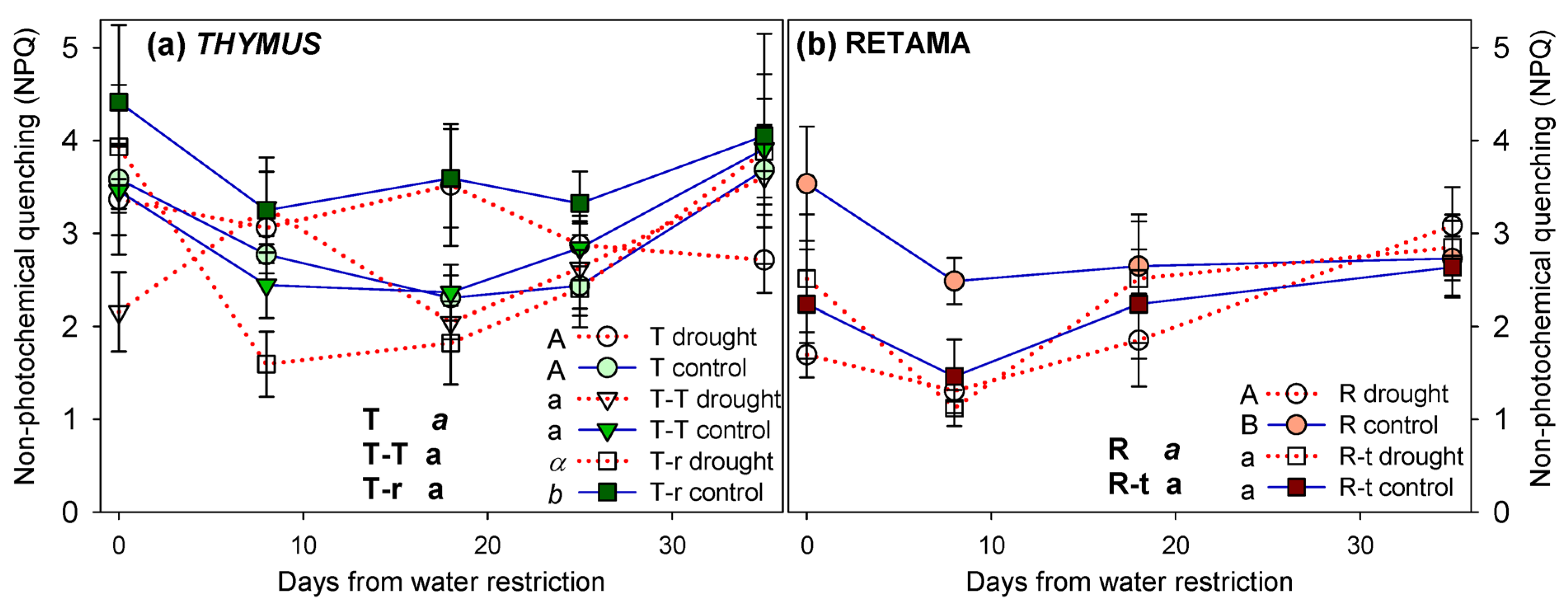
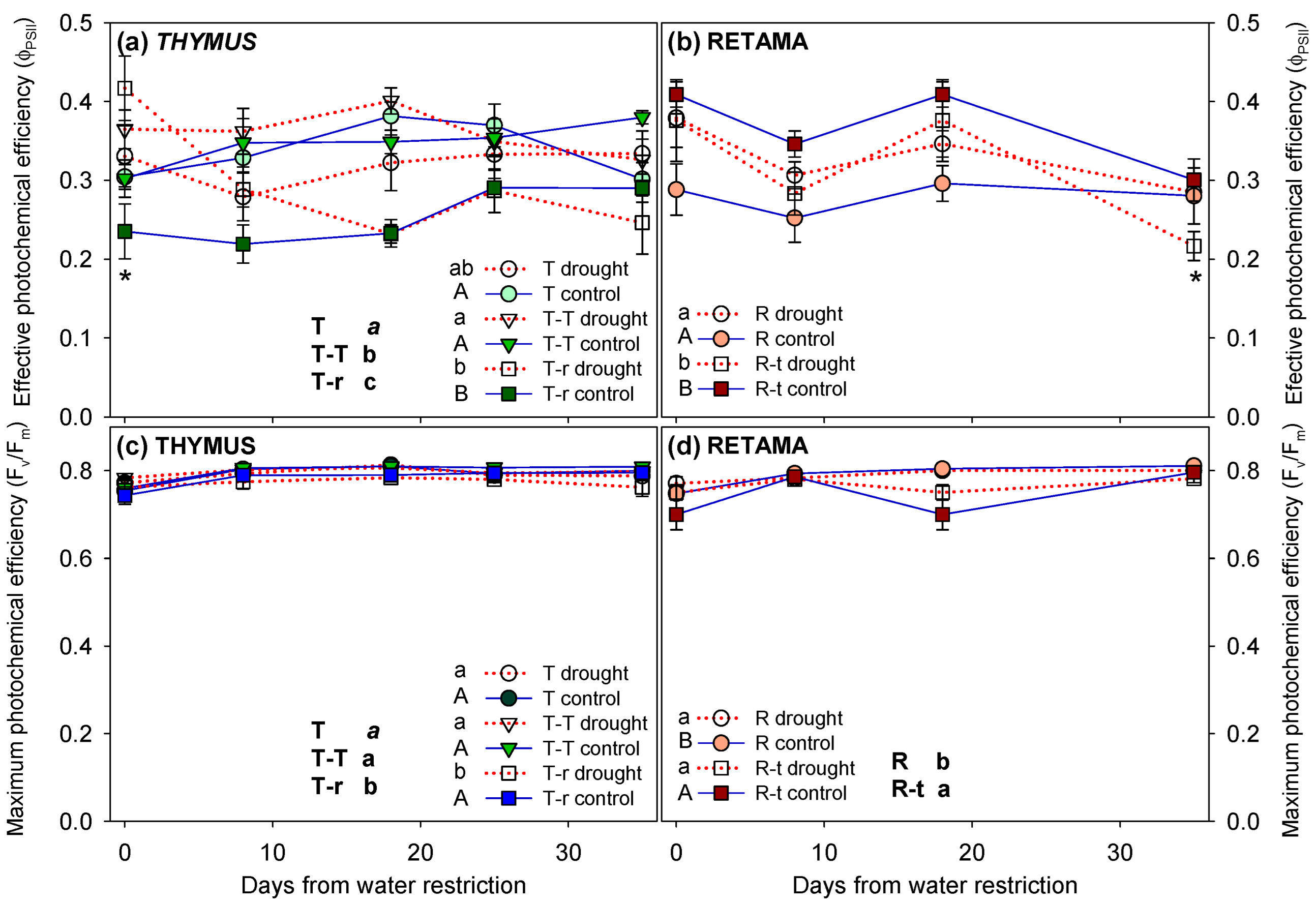
2.2.2. Photochemical Efficiency
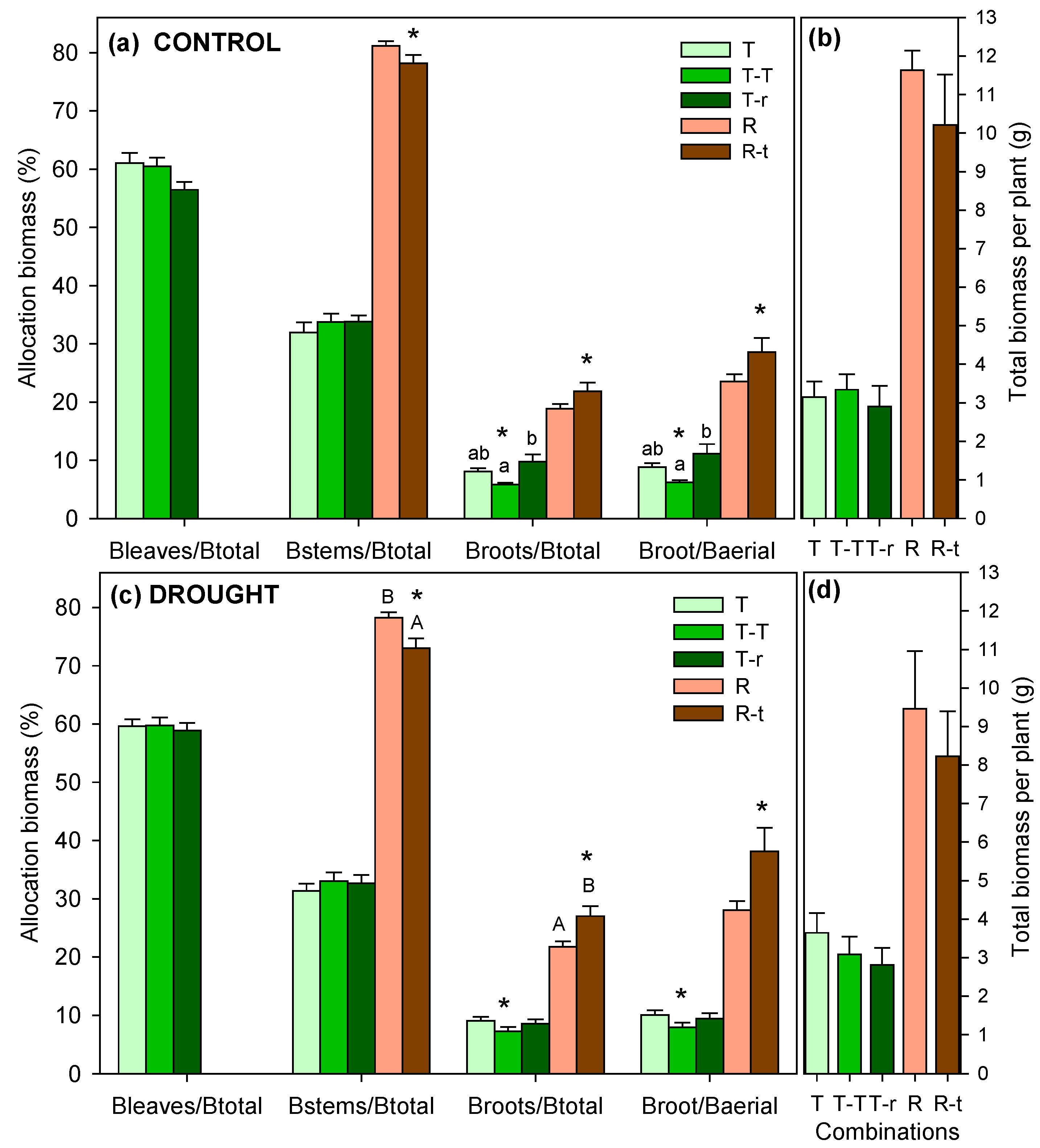
2.3. Biomass Allocation
2.4. Leaf Chemistry Analyses
3. Discussion
3.1. Morphological Responses
3.2. Physiological Responses to Drought and Plant–Plant Interactions
4. Materials and Methods
4.1. Study Species
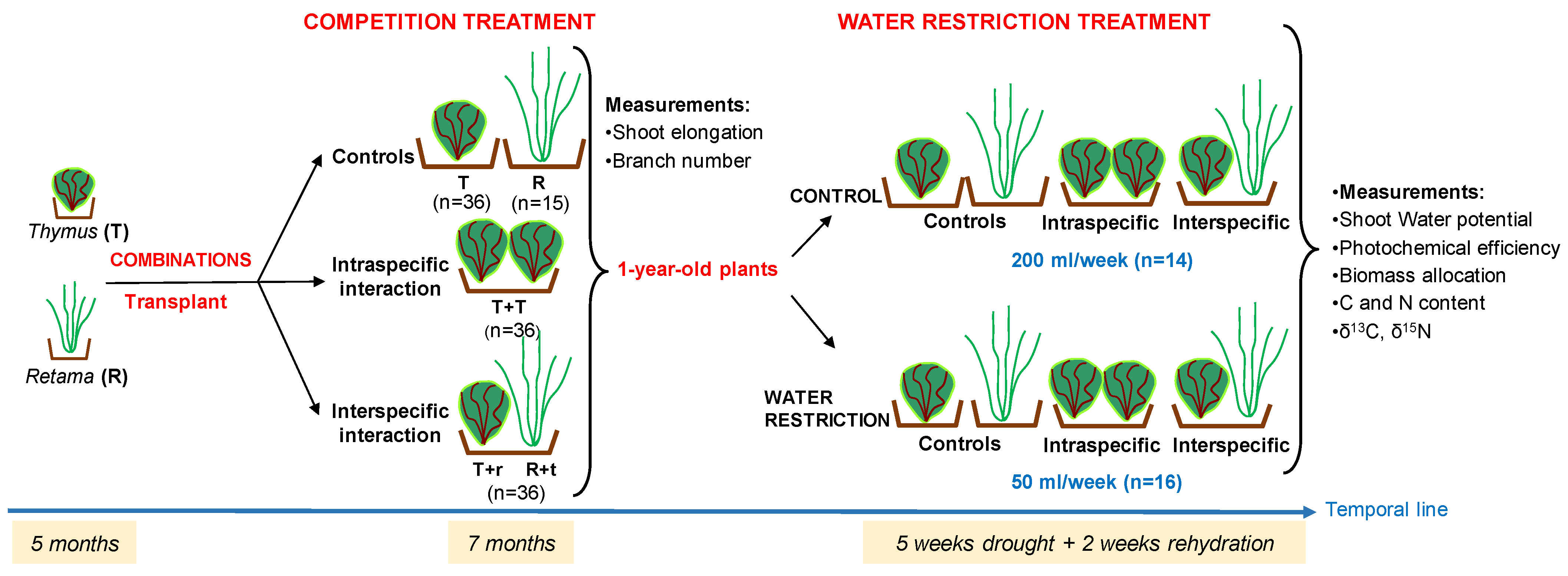
4.2. Experimental Design
4.3. Plant Transplanting
- Control: 36 pots with one Thymus plant (T) and 15 pots with one Retama plant (R);
- Intraspecific competition: 36 pots with two Thymus plants (T-T);
- Interspecific competition 36 pots with one Thymus and one Retama plant, (T-r or R-t). In this case, we distinguish between T–r and R–t, depending on which species was measured in the shared pot: T–r indicates measurements taken on Thymus growing with Retama, and R–t indicates measurements on Retama growing with Thymus.
4.4. Morphological Measurements
4.5. Water Restriction Experiment
4.5.1. Physiological Measurements
4.5.2. Biomass Measurement
4.5.3. Leaf and Cladode Isotopic Analysis and N and C Content
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| RER | Relative elongation rate |
| ΔR | Branching increment |
| Ψm | Shoot water potential |
| ΦPSII | Effective photochemical efficiency |
| Fv/Fm | Maximum photochemical efficiency |
| NPQ | Non-photochemical quenching |
| BL/BT | Leaf mass allocation |
| BS/BT | Stem mass allocation |
| BR/BT | Root mass allocation |
| BR/BA | Root-biomass-to-aboveground-biomass ratio |
| δ13C | Leaf carbon isotope signature (13C/12C) |
| δ15N | Leaf nitrogen isotope signature (15N/14N) |
| WUEi | Intrinsic water-use efficiency |
Appendix A
Appendix A.1
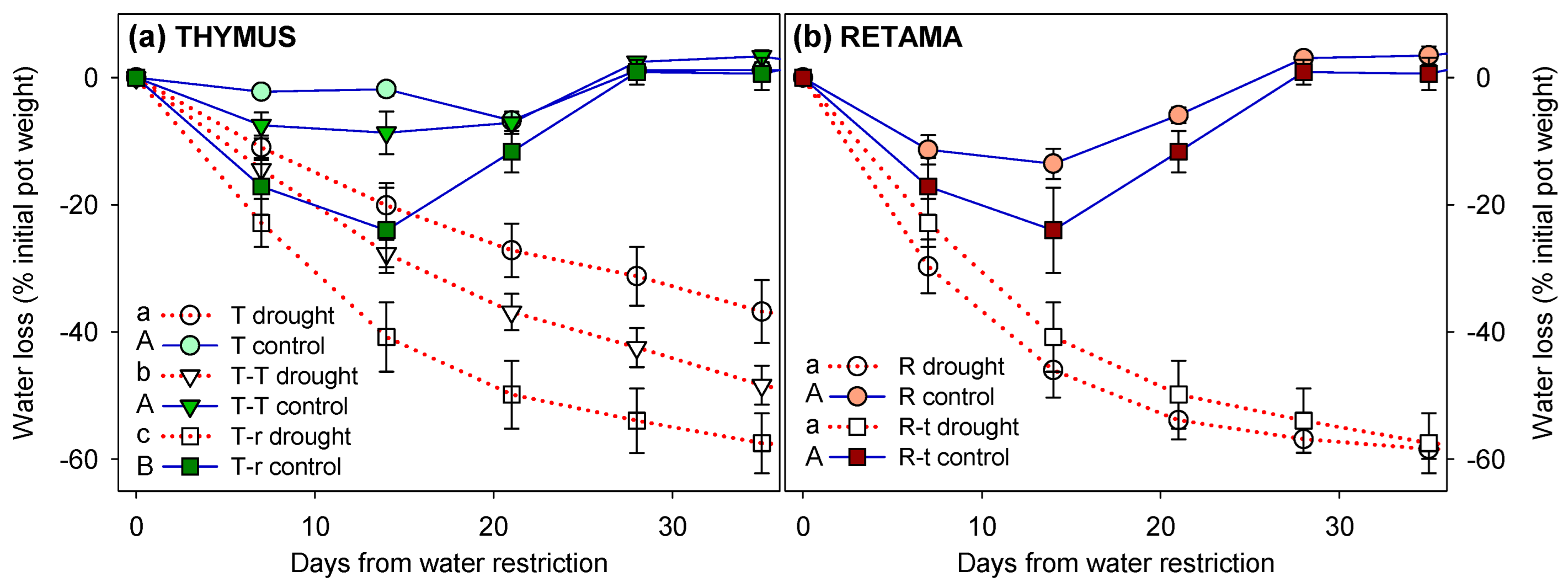
Appendix A.2
References
- Kaisermann, A.; de Vries, F.T.; Griffiths, R.I.; Bardgett, R.D. Legacy Effects of Drought on Plant–Soil Feedbacks and Plant–Plant Interactions. New Phytol. 2017, 215, 1413–1424. [Google Scholar] [CrossRef]
- van der Putten, W.H.; Bardgett, R.D.; Bever, J.D.; Bezemer, T.M.; Casper, B.B.; Fukami, T.; Kardol, P.; Klironomos, J.N.; Kulmatiski, A.; Schweitzer, J.A.; et al. Plant–Soil Feedbacks: The Past, the Present and Future Challenges. J. Ecol. 2013, 101, 265–276. [Google Scholar] [CrossRef]
- Brooker, R.W. Plant–Plant Interactions and Environmental Change. New Phytol. 2006, 171, 271–284. [Google Scholar] [CrossRef]
- Hammond, W.M.; Williams, A.P.; Abatzoglou, J.T.; Adams, H.D.; Klein, T.; López, R.; Sáenz-Romero, C.; Hartmann, H.; Breshears, D.D.; Allen, C.D. Global Field Observations of Tree Die-off Reveal Hotter-Drought Fingerprint for Earth’s Forests. Nat. Commun. 2022, 13, 1761. [Google Scholar] [CrossRef]
- Lloret, F.; Jaime, L.A.; Margalef-Marrase, J.; Pérez-Navarro, M.A.; Batllori, E. Short-Term Forest Resilience after Drought-Induced Die-off in Southwestern European Forests. Sci. Total Environ. 2022, 806, 150940. [Google Scholar] [CrossRef]
- Kardol, P.; Campany, C.E.; Souza, L.; Norby, R.J.; Weltzin, J.F.; Classen, A.T. Climate Change Effects on Plant Biomass Alter Dominance Patterns and Community Evenness in an Experimental Old-Field Ecosystem. Glob. Change Biol. 2010, 16, 2676–2687. [Google Scholar] [CrossRef]
- Ploughe, L.W.; Jacobs, E.M.; Frank, G.S.; Greenler, S.M.; Smith, M.D.; Dukes, J.S. Community Response to Extreme Drought (CRED): A Framework for Drought-Induced Shifts in Plant–Plant Interactions. New Phytol. 2019, 222, 52–69. [Google Scholar] [CrossRef]
- Evenari, M.; Noy-Meir, I.; Goodall, D.W. Hot Deserts and Arid Shrublands; Elsevier: Amsterdam, The Netherlands, 1985; ISBN 978-0-444-42296-5. [Google Scholar]
- Padilla, F.M.; Miranda, J.D.; Jorquera, M.J.; Pugnaire, F.I. Variability in Amount and Frequency of Water Supply Affects Roots but Not Growth of Arid Shrubs. Plant Ecol. 2009, 204, 261–270. [Google Scholar] [CrossRef]
- Stuart-Haëntjens, E.; De Boeck, H.J.; Lemoine, N.P.; Mänd, P.; Kröel-Dulay, G.; Schmidt, I.K.; Jentsch, A.; Stampfli, A.; Anderegg, W.R.L.; Bahn, M.; et al. Mean Annual Precipitation Predicts Primary Production Resistance and Resilience to Extreme Drought. Sci. Total Environ. 2018, 636, 360–366. [Google Scholar] [CrossRef]
- Joffre, R.; Rambal, S.; Ratte, J.P. The Dehesa System of Southern Spain and Portugal as a Natural Ecosystem Mimic. Agrofor. Syst. 1999, 45, 57–79. [Google Scholar] [CrossRef]
- Seifan, M.; Tielbörger, K.; Kadmon, R. Direct and Indirect Interactions among Plants Explain Counterintuitive Positive Drought Effects on an Eastern Mediterranean Shrub Species. Oikos 2010, 119, 1601–1609. [Google Scholar] [CrossRef]
- Van der Maarel, E. Dry Coastal Ecosystems: Polar Regions and Europe; Ecosystems of the world, 2A.; Elsevier: Amsterdam, The Netherlands, 1993; ISBN 978-0-444-87348-4. [Google Scholar]
- IPCC. IPCC Climate Change 2023 Synthesis Report. In Climate Change 2023: Synthesis Report. Contribution of Working Groups I, II and III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; IPCC: Geneva, Switzerland, 2023; pp. 35–115. [Google Scholar] [CrossRef]
- Maestre, F.; Eldridge, D.; Soliveres, S.; Kéfi, S.; Delgado-Baquerizo, M.; Bowker, M.; García-Palacios, P.; Gaitan, J.; Gallardo, A.; Lazaro, R.; et al. Structure and Functioning of Dryland Ecosystems in a Changing World. Annu. Rev. Ecol. Evol. Syst. 2016, 47, 215–237. [Google Scholar] [CrossRef] [PubMed]
- Chazarra Bernabé, A.; Lorenzo Mariño, B.; Romero Fresneda, R.; Moreno García, J.V. Evolución de los climas de Köppen en España en el periodo 1951–2020; Agencia Estatal de Meteorología: Madrid, Spain, 2022. [Google Scholar]
- Lavorel, S.; Canadell, J.; Rambal, S.; Terradas, J. Mediterranean Terrestrial Ecosystems: Research Priorities on Global Change Effects. Glob. Ecol. Biogeogr. Lett. 1998, 7, 157–166. [Google Scholar] [CrossRef]
- Domènech, R.; Vilà, M. Response of the Invader Cortaderia selloana and Two Coexisting Natives to Competition and Water Stress. Biol. Invasions 2008, 10, 903–912. [Google Scholar] [CrossRef]
- Grime, J.P.; Hillier, S.H. The Contribution of Seedling Regeneration to the Structure and Dynamics of Plant Communities, Ecosystems, and Larger Units of the Landscape. In Seeds: The Ecology of Regeneration in Plant Communities; CABI Publishing: Wallingford, UK, 2000; pp. 361–374. [Google Scholar] [CrossRef]
- Harper, J.L. Population Biology of Plants; Academic Press: Cambridge, MA, USA, 1977; ISBN 978-0-12-325850-2. [Google Scholar]
- Evans, C.E.; Etherington, J.R. The Effect of Soil Water Potential on Seedling Growth of Some British Plants. New Phytol. 1991, 118, 571–579. [Google Scholar] [CrossRef]
- Niering, W.A.; Whittaker, R.H.; Lowe, C.H. The Saguaro: A Population in Relation to Environment. Science 1963, 142, 15–23. [Google Scholar] [CrossRef]
- Mingo, A. Size-Uneven Competition and Resource Availability: A Factorial Experiment on Seedling Establishment of Three Mediterranean Species. Plant Biosyst. 2009, 143, 181–189. [Google Scholar] [CrossRef]
- Callaway, R.; Pugnaire, F. Facilitation in Plant Communities. In Functional Plant Ecology; CRC Press: Boca Raton, FL, USA, 1999; pp. 623–648. [Google Scholar]
- Pugnaire, F.I.; Luque, M.T. Changes in Plant Interactions along a Gradient of Environmental Stress. Oikos 2001, 93, 42–49. [Google Scholar] [CrossRef]
- Brouwer, R. Nutritive Influences on the Distribution of Dry Matter in the Plant. NJAS 1962, 10, 399–408. [Google Scholar] [CrossRef]
- Peperkorn, R.; Werner, C.; Beyschlag, W. Phenotypic Plasticity of an Invasive Acacia versus Two Native Mediterranean Species. Funct. Plant Biol. 2005, 32, 933–944. [Google Scholar] [CrossRef]
- Arndt, S.K.; Wanek, W. Use of Decreasing Foliar Carbon Isotope Discrimination during Water Limitation as a Carbon Tracer to Study Whole Plant Carbon Allocation. Plant Cell Environ. 2002, 25, 609–616. [Google Scholar] [CrossRef]
- Poorter, H.; Nagel, O.W. The Role of Biomass Allocation in the Growth Response of Plants to Different Levels of Light, CO2, Nutrients and Water: A Quantitative Review. Funct. Plant Biol. 2000, 27, 595–607. [Google Scholar] [CrossRef]
- Smith, T.; Huston, M. A Theory of the Spatial and Temporal Dynamics of Plant Communities. Vegetatio 1989, 83, 49–69. [Google Scholar] [CrossRef]
- Brisson, J.; Reynolds, J.F. Effects of Compensatory Growth on Population Processes: A Simulation Study. Ecology 1997, 78, 2378–2384. [Google Scholar] [CrossRef]
- Casper, B.B.; Jackson, R.B. Plant Competition Underground. Annu. Rev. Ecol. Evol. Syst. 1997, 28, 545–570. [Google Scholar] [CrossRef]
- Campbell, B.D.; Grime, J.P. A New Method of Exposing Developing Root Systems to Controlled Patchiness in Mineral Nutrient Supply. Ann. Bot. 1989, 63, 395–400. [Google Scholar] [CrossRef]
- de Kroons, H.; Hutchings, M.J. Morphological Plasticity in Clonal Plants: The Foraging Concept Reconsidered. J. Ecol. 1995, 83, 143–152. [Google Scholar] [CrossRef]
- Gersani, M.; Abramsky, Z.; Falik, O. Density-Dependent Habitat Selection in Plants. Evol. Ecol. 1998, 12, 223–234. [Google Scholar] [CrossRef]
- Mahall, B.E.; Callaway, R.M. Root Communication among Desert Shrubs. Proc. Natl. Acad. Sci. USA 1991, 88, 874–876. [Google Scholar] [CrossRef]
- Schenk, H.J. Root Competition: Beyond Resource Depletion. J. Ecol. 2006, 94, 725–739. [Google Scholar] [CrossRef]
- Gersani, M.; Brown, J.S.; O’Brien, E.E.; Maina, G.M.; Abramsky, Z. Tragedy of the Commons as a Result of Root Competition. J. Ecol. 2001, 89, 660–669. [Google Scholar] [CrossRef]
- Armas, C.; Pugnaire, F.I. Belowground Zone of Influence in a Tussock Grass Species. Acta Oecol. 2011, 37, 284–289. [Google Scholar] [CrossRef]
- Bais, H.P.; Vepachedu, R.; Gilroy, S.; Callaway, R.M.; Vivanco, J.M. Allelopathy and Exotic Plant Invasion: From Molecules and Genes to Species Interactions. Science 2003, 301, 1377–1380. [Google Scholar] [CrossRef]
- Bertin, C.; Weston, L.A.; Huang, T.; Jander, G.; Owens, T.; Meinwald, J.; Schroeder, F.C. Grass Roots Chemistry: Meta-Tyrosine, an Herbicidal Nonprotein Amino Acid. Proc. Natl. Acad. Sci. USA 2007, 104, 16964–16969. [Google Scholar] [CrossRef]
- Beyer, F.; Hertel, D.; Jung, K.; Fender, A.-C.; Leuschner, C. Competition Effects on Fine Root Survival of Fagus sylvatica and Fraxinus excelsior. For. Ecol. Manag. 2013, 302, 14–22. [Google Scholar] [CrossRef]
- Caldwell, M.M.; Manwaring, J.H.; Durham, S.L. Species Interactions at the Level of Fine Roots in the Field: Influence of Soil Nutrient Heterogeneity and Plant Size. Oecologia 1996, 106, 440–447. [Google Scholar] [CrossRef]
- Villar-Salvador, P.; Cuesta, B. Retama monosperma (L.) Boiss. y Retama sphaerocarpa (L.) Boiss. In Producción y Manejo de Semillas y Plantas Forestales; Organismo Autónomo Parques Naturales: Madrid, Spain, 2013; pp. 342–353. ISBN 978-84-8014-846-7. [Google Scholar]
- Kith y Tasara, M. El problema de las dunas del SO de España. Montes 1946, 11, 414–419. [Google Scholar]
- Möllerová, J. Notes on Invasive and Expansive Trees and Shrubs. J. For. Sci. 2005, 51, 19–23. [Google Scholar] [CrossRef]
- Hejda, M.; Sádlo, J.; Kutlvašr, J.; Petřík, P.; Vítková, M.; Vojík, M.; Pyšek, P.; Pergl, J. Impact of Invasive and Native Dominants on Species Richness and Diversity of Plant Communities. Preslia 2021, 93, 181–201. [Google Scholar] [CrossRef]
- Esquivias, M.P.; Zunzunegui, M.; Díaz Barradas, M.C.; Álvarez-Cansino, L. Competitive Effect of a Native-Invasive Species on a Threatened Shrub in a Mediterranean Dune System. Oecologia 2015, 177, 133–146. [Google Scholar] [CrossRef]
- Muñoz Vallés, S.; Gallego Fernández, J.B.; Dellafiore, C.; Cambrollé, J. Effects on Soil, Microclimate and Vegetation of the Native-Invasive Retama monosperma (L.) in Coastal Dunes. Plant Ecol. 2011, 212, 169–179. [Google Scholar] [CrossRef]
- García-de-Lomas, J.; Fernández, L.; Martín, I.; Saavedra, C.; Rodríguez-Hiraldo, C.; Gallego-Fernández, J.B. Management of Coastal Dunes Affected by Shrub Encroachment: Are Rabbits an Ally or an Enemy of Restoration? J. Coast. Conserv. 2023, 27, 8. [Google Scholar] [CrossRef]
- Esquivias, M.P.; Zunzunegui, M.; Barradas, M.C.D.; Álvarez-Cansino, L. The Role of Water Use and Uptake on Two Mediterranean Shrubs’ Interaction in a Brackish Coastal Dune Ecosystem. Ecohydrology 2014, 7, 783–793. [Google Scholar] [CrossRef]
- Zunzunegui, M.; Esquivias, M.; Oppo, F.; Gallego-Fernández, J. Interspecific Competition and Livestock Disturbance Control the Spatial Patterns of Two Coastal Dune Shrubs. Plant Soil. 2012, 354, 299–309. [Google Scholar] [CrossRef]
- Naumann, G.; Alfieri, L.; Wyser, K.; Mentaschi, L.; Betts, R.A.; Carrao, H.; Spinoni, J.; Vogt, J.; Feyen, L. Global Changes in Drought Conditions Under Different Levels of Warming. Geophys. Res. Lett. 2018, 45, 3285–3296. [Google Scholar] [CrossRef]
- Lambers, H.; Poorter, H. Inherent Variation in Growth Rate Between Higher Plants: A Search for Physiological Causes and Ecological Consequences. Adv. Ecol. Res. 1992, 23, 187–261. [Google Scholar] [CrossRef]
- Rascher, K.G.; Hellmann, C.; Máguas, C.; Werner, C. Community Scale 15N Isoscapes: Tracing the Spatial Impact of an Exotic N2-Fixing Invader. Ecol. Lett. 2012, 15, 484–491. [Google Scholar] [CrossRef]
- Fernández-Martínez, M.; Jiménez-Carrasco, C.; Barradas, M.C.D.; Gallego-Fernández, J.B.; Zunzunegui, M. Ecophysiological Keys to the Success of a Native-Expansive Mediterranean Species in Threatened Coastal Dune Habitats. Plants 2025, 14, 2342. [Google Scholar] [CrossRef]
- Fogarty, G.; Facelli, J.M. Growth and Competition of Cytisus scoparius, an Invasive Shrub, and Australian Native Shrubs. Plant Ecol. 1999, 144, 27–35. [Google Scholar] [CrossRef]
- Fajardo, A.; McIntire, E.J.B. Under Strong Niche Overlap Conspecifics Do Not Compete but Help Each Other to Survive: Facilitation at the Intraspecific Level. J. Ecol. 2011, 99, 642–650. [Google Scholar] [CrossRef]
- Martinez-Oró, D.; Parraga-Aguado, I.; Querejeta, J.I.; Conesa, H.M. Importance of Intra- and Interspecific Plant Interactions for the Phytomanagement of Semiarid Mine Tailings Using the Tree Species Pinus halepensis. Chemosphere 2017, 186, 405–413. [Google Scholar] [CrossRef]
- Noto, A.E.; Hughes, A.R. Genotypic Diversity Weakens Competition within, but not between, Plant Species. J. Ecol. 2020, 108, 2212–2220. [Google Scholar] [CrossRef]
- Armas, C.; Pugnaire, F.I. Plant Interactions Govern Population Dynamics in a Semi-arid Plant Community. J. Ecol. 2005, 93, 978–989. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Alegre, L. The Xanthophyll Cycle Is Induced by Light Irrespective of Water Status in Field-Grown Lavender (Lavandula stoechas) Plants. Physiol. Plant. 2000, 108, 147–151. [Google Scholar] [CrossRef]
- Genty, B.; Briantais, J.-M.; Silva, J.B.V.D. Effects of Drought on Primary Photosynthetic Processes of Cotton Leaves. Plant Physiol. 1987, 83, 360–364. [Google Scholar] [CrossRef]
- Peñuelas, J.; Munné-Bosch, S.; Llusià, J.; Filella, I. Leaf Reflectance and Photo- and Antioxidant Protection in Field-Grown Summer-Stressed Phillyrea angustifolia. Optical Signals of Oxidative Stress? New Phytol. 2004, 162, 115–124. [Google Scholar] [CrossRef]
- Valladares, F.; Sánchez-Gómez, D. Ecophysiological Traits Associated with Drought in Mediterranean Tree Seedlings: Individual Responses versus Interspecific Trends in Eleven Species. Plant Biol. 2006, 8, 688–697. [Google Scholar] [CrossRef]
- Callaway, R.M.; Walker, L.R. Competition and Facilitation: A Synthetic Approach to Interactions in Plant Communities. Ecology 1997, 78, 1958–1965. [Google Scholar] [CrossRef]
- Maestre, F.T.; Callaway, R.M.; Valladares, F.; Lortie, C.J. Refining the Stress-gradient Hypothesis for Competition and Facilitation in Plant Communities. J. Ecol. 2009, 97, 199–205. [Google Scholar] [CrossRef]
- Gimeno, T.E.; Escudero, A.; Valladares, F. Different Intra- and Interspecific Facilitation Mechanisms between Two Mediterranean Trees under a Climate Change Scenario. Oecologia 2015, 177, 159–169. [Google Scholar] [CrossRef]
- Gómez-Aparicio, L. The Role of Plant Interactions in the Restoration of Degraded Ecosystems: A Meta-Analysis across Life-Forms and Ecosystems. J. Ecol. 2009, 97, 1202–1214. [Google Scholar] [CrossRef]
- Holmgren, M.; Gómez-Aparicio, L.; Quero, J.L.; Valladares, F. Non-Linear Effects of Drought under Shade: Reconciling Physiological and Ecological Models in Plant Communities. Oecologia 2012, 169, 293–305. [Google Scholar] [CrossRef]
- Rajaniemi, T.K. Why Does Fertilization Reduce Plant Species Diversity? Testing Three Competition-Based Hypotheses. J. Ecol. 2002, 90, 316–324. [Google Scholar] [CrossRef]
- Sardans, J.; Peñuelas, J. Plant-Soil Interactions in Mediterranean Forest and Shrublands: Impacts of Climatic Change. Plant Soil. 2013, 365, 1–33. [Google Scholar] [CrossRef]
- Correia, O.; Martins, A.; Catarino, F. Comparative Phenology and Seasonal Foliar Nitrogen Variation in Mediterranean Species of Portugal. Ecol. Mediterr. 1992, 18, 7–18. [Google Scholar] [CrossRef]
- Werner, C.; Correia, O.; Beyschlag, W. Two Different Strategies of Mediterranean Macchia Plants to Avoid Photoinhibitory Damage by Excessive Radiation Levels during Summer Drought. Acta Oecol. 1999, 20, 15–23. [Google Scholar] [CrossRef]
- Fernández, R.J.; Reynolds, J.F. Potential Growth and Drought Tolerance of Eight Desert Grasses: Lack of a Trade-Off? Oecologia 2000, 123, 90–98. [Google Scholar] [CrossRef]
- Reich, P.B.; Tjoelker, M.G.; Walters, M.B.; Vanderklein, D.W.; Buschena, C. Close Association of RGR, Leaf and Root Morphology, Seed Mass and Shade Tolerance in Seedlings of Nine Boreal Tree Species Grown in High and Low Light. Funct. Ecol. 1998, 12, 327–338. [Google Scholar] [CrossRef]
- Hodge, A.; Stewart, J.; Robinson, D.; Griffiths, B.S.; Fitter, A.H. Spatial and Physical Heterogeneity of N Supply from Soil Does Not Influence N Capture by Two Grass Species. Funct. Ecol. 2000, 14, 645–653. [Google Scholar] [CrossRef]
- Olson, B.E.; Blicker, P.S. Response of the Invasive Centaurea maculosa and Two Native Grasses to N-Pulses. Plant Soil. 2003, 254, 457–467. [Google Scholar] [CrossRef]
- Dawson, T.E.; Mambelli, S.; Plamboeck, A.H.; Templer, P.H.; Tu, K.P. Stable Isotopes in Plant Ecology. Annu. Rev. Ecol. Syst. 2002, 33, 507–559. [Google Scholar] [CrossRef]
- Högberg, P. Tansley Review No. 95 15 N Natural Abundance in Soil-Plant Systems. New Phytol. 1997, 137, 179–203. [Google Scholar] [CrossRef]
- Evans, R.D. Physiological Mechanisms Influencing Plant Nitrogen Isotope Composition. Trends Plant Sci. 2001, 6, 121–126. [Google Scholar] [CrossRef]
- Hobbie, E.A.; Högberg, P. Nitrogen Isotopes Link Mycorrhizal Fungi and Plants to Nitrogen Dynamics. New Phytol. 2012, 196, 367–382. [Google Scholar] [CrossRef]
- Cornelissen, J.; Lavorel, S.; Garnier, E.; Diaz, S.; Buchmann, N.; Gurvich, D.; Reich, P.; ter Steege, H.; Morgan, H.D.G.; Van der Heijden, M.; et al. Handbook of Protocols for Standardised and Easy Measurement of Plant Functional Traits Worldwide. Aust. J. Bot. 2003, 51, 335–380. [Google Scholar] [CrossRef]
- Cabezudo, B.; Talavera, S.; Blanca, G.; Cueto, M.; Valdés, B.; Hernández Bermejo, J.; Herrera, C.; Rodríguez Hiraldo, C.; Navas, D. Lista roja de la Flora vascular de Andalucía; Consejería de Medio Ambiente: Sevilla, Spain, 2005; p. 126. ISBN 978-84-96329-62-1. [Google Scholar]
- Carapeto, A.; Francisco, A.; Pereira, P.; Porto, M. Lista Vermelha da Flora Vascular de Portugal Continental; Botânica em Português; Sociedade Portuguesa de Botânica, Associação Portuguesa de Ciência da Vegetação—PHYTOS e Instituto da Conservação da Natureza e das Florestas.; Imprensa Nacional-Casa da Moeda: Lisboa, Portugal, 2000; Volume 7, ISBN 978-972-27-2876. [Google Scholar]
- Comunidades Europeas. Directiva 97/62/CE del Consejo, de 27 de Octubre de 1997, por la que se Adapta al Progreso Científico y técnico la Directiva 92/43/CEE, Relativa a la Conservación de los Hábitats Naturales y de Fauna y Flora Silvestres; Comunidades Europeas: Brussels, Belgium, 1997; Volume 305. [Google Scholar]
- Talavera, S.; Castroviejo, S. Flora Iberica: Plantas vasculares de la Penínsular Ibérica e Islas Baleares: Vol. VII (I) Leguminosae (partim); Editorial CSIC—CSIC Press: Madrid, Spain, 1999; ISBN 978-84-00-07821-8. [Google Scholar]
- Gallego-Fernández, J.B.; Muñoz-Valles, S.; Dellafiore, C.M. Spatio-Temporal Patterns of Colonization and Expansion of Retama monosperma on Developing Coastal Dunes. J. Coast. Conserv. 2015, 19, 577–587. [Google Scholar] [CrossRef]
- Muñoz-Vallés, S.; Gallego-Fernández, J.B.; Cambrollé, J. The Role of the Expansion of Native-Invasive Plant Species in Coastal Dunes: The Case of Retama monosperma in SW Spain. Acta Oecol. 2014, 54, 82–89. [Google Scholar] [CrossRef]
- Pérez-Fernández, M.; Gómez Gutiérrez, J.M. Importancia e interpretación de la latencia y germinación de semillas en ambientes naturales. In Restauración de Ecosistemas Mediterráneos; Universidad de Alcalá: Madrid, Spain, 2003; pp. 87–112. [Google Scholar]
- Streeter, J.; Wong, P.P. Inhibition of Legume Nodule Formation and N2 Fixation by Nitrate. Crit. Rev. Plant Sci. 1988, 7, 1–23. [Google Scholar] [CrossRef]
- Gower, S.T.; Vogt, K.A.; Grier, C.C. Carbon Dynamics of Rocky Mountain Douglas-Fir: Influence of Water and Nutrient Availability. Ecol. Monogr. 1992, 62, 43–65. [Google Scholar] [CrossRef]
- Perkins, S.R.; Keith Owens, M. Growth and Biomass Allocation of Shrub and Grass Seedlings in Response to Predicted Changes in Precipitation Seasonality. Plant Ecol. 2003, 168, 107–120. [Google Scholar] [CrossRef]
- Farquhar, G.; O’Leary, M.H.O.; Berry, J. On the Relationship between Carbon Isotope Discrimination and the Intercellular Carbon Dioxide Concentration in Leaves. Aust. J. Plant Physiol. 1982, 9, 121–137. [Google Scholar] [CrossRef]
- Werner, C.; Máguas, C. Carbon Isotope Discrimination as a Tracer of Functional Traits in a Mediterranean Macchia Plant Community. Funct. Plant Biol. 2010, 2010, 467–477. [Google Scholar] [CrossRef]
- Farquhar, G.D.; Hubick, K.T.; Condon, A.G.; Richards, R.A. Carbon Isotope Fractionation and Plant Water-Use Efficiency. In Proceedings of the Stable Isotopes in Ecological Research; Rundel, P.W., Ehleringer, J.R., Nagy, K.A., Eds.; Springer: New York, NY, USA, 1989; pp. 21–40. [Google Scholar]
- Pérez-Harguindeguy, N.; Díaz, S.; Garnier, E.; Lavorel, S.; Poorter, H.; Jaureguiberry, P.; Bret-Harte, M.S.; Cornwell, W.K.; Craine, J.M.; Gurvich, D.E.; et al. New Handbook for Standardised Measurement of Plant Functional Traits Worldwide. Aust. J. Bot. 2013, 61, 167. [Google Scholar] [CrossRef]

| RER | Thymus | Retama | ||||
|---|---|---|---|---|---|---|
| df | F | p | df | F | p | |
| Competition | 2 | 6.8 | 0.001 | 1 | 4.678 | 0.036 |
| Time | 4 | 318.5 | 0.001 | 4 | 46.332 | 0.000 |
| Competition × Time | 8 | 10.1 | 0.001 | 4 | 0.548 | 0.463 |
| Ψm | ΦPSII | Fv/Fm | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Thymus | df | F | p | df | F | p | df | F | p |
| Competition | 2 | 15.830 | 0.001 | 2 | 26.994 | 0.001 | 2 | 15.486 | 0.001 |
| Watering | 1 | 13.621 | 0.001 | 1 | 2.091 | 0.150 | 1 | 2.199 | 0.140 |
| Time | 5 | 13.089 | 0.001 | 4 | 0.954 | 0.434 | 4 | 20.989 | 0.001 |
| C × W | 2 | 8.641 | 0.001 | 2 | 3.156 | 0.045 | 2 | 0.883 | 0.415 |
| C × T | 10 | 2.805 | 0.003 | 8 | 2.630 | 0.009 | 8 | 0.454 | 0.887 |
| W × T | 5 | 5.607 | 0.001 | 4 | 5.110 | 0.001 | 4 | 5.057 | 0.001 |
| C × W × T | 10 | 2.437 | 0.009 | 8 | 2.424 | 0.016 | 8 | 0.229 | 0.985 |
| Retama | df | F | p | df | F | p | df | F | p |
| Competition | 1 | 0.240 | 0.625 | 1 | 0.225 | 0.636 | 1 | 8.335 | 0.005 |
| Watering | 1 | 52.657 | 0.001 | 1 | 0.000 | 0.983 | 1 | 0.001 | 0.977 |
| Time | 5 | 6.152 | 0.001 | 3 | 8.028 | 0.001 | 3 | 13.240 | 0.001 |
| C × W | 1 | 0.198 | 0.657 | 1 | 15.172 | 0.001 | 1 | 0.030 | 0.864 |
| C × T | 5 | 0.108 | 0.990 | 3 | 1.964 | 0.125 | 3 | 0.859 | 0.466 |
| W × T | 5 | 7.250 | 0.001 | 3 | 1.263 | 0.292 | 3 | 3.348 | 0.022 |
| C × W × T | 5 | 0.266 | 0.931 | 3 | 0.194 | 0.900 | 3 | 0.621 | 0.603 |
| Thymus | BL/BT | BS/BT | BR/BT | BR/BA | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| df | F | p | df | F | p | df | F | p | df | F | p | |
| Competition | 2 | 1.520 | 0.225 | 2 | 0.881 | 0.418 | 2 | 7.339 | 0.001 | 2 | 6.996 | 0.002 |
| Watering | 1 | 0.135 | 0.715 | 1 | 0.467 | 0.496 | 1 | 0.418 | 0.520 | 1 | 0.287 | 0.593 |
| C × W | 2 | 0.656 | 0.522 | 2 | 0.021 | 0.979 | 2 | 1.949 | 0.149 | 2 | 1.988 | 0.143 |
| Retama | BS/BT | BR/BT | BR/BA | |||||||||
| df | F | p | df | F | p | df | F | p | ||||
| Competition | 1 | 5.618 | 0.023 | 1 | 5.618 | 0.023 | 1 | 4.719 | 0.036 | |||
| Watering | 1 | 5.456 | 0.025 | 1 | 5.456 | 0.025 | 1 | 4.115 | 0.050 | |||
| C × W | 1 | 0.449 | 0.507 | 1 | 0.449 | 0.507 | 1 | 0.551 | 0.463 | |||
| %C | %N | C/N | δ13C | δ15N | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | C | D | C | D | C | D | C | D | C | D | |
| T | Mean | 47.9 ab | 47.9 a | 1.5 | 1.4 | 32.8 | 39.7 | −28.8 | −27.0 a | 5.1 a | 4.1 a |
| SD | 1.3 | 1.6 | 0.4 | 0.6 | 9.7 | 16.1 | 2.1 | 3.6 | 1.8 | 1.6 | |
| T-T | Mean | 49.1 a | 49.0 a | 1.7 | 1.5 | 31.8 | 34.5 | −28.5 | −28.6 ab | 3.7 ab | 3.3 ab |
| SD | 1.6 | 1.1 | 0.5 | 0.4 | 9.0 | 8.2 | 2.7 | 1.1 | 0.3 | 0.6 | |
| T-r | Mean | 49.6 a | 50.0 a | 1.4 | 1.6 | 35.3 | 33.9 | −29.1 | −29.3 ab | 2.8 b | 2.6 b |
| SD | 3.6 | 2.7 | 0.3 | 0.4 | 6.3 | 9.7 | 2.1 | 1.4 | 0.5 | 1.0 | |
| R-t | Mean | 42.9 b | 42.9 b | 1.4 | 1.3 | 32.9 | 36.2 | −30.5 | −31.3 b | 2.5 b | 2.7 b |
| SD | 1.6 | 2.0 | 0.4 | 0.3 | 8.2 | 8.6 | 0.8 | 0.9 | 1.8 | 1.0 | |
| % C | % N | C/N | δ13C | δ15N | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| df | F | p | df | F | p | df | F | p | df | F | p | df | F | p | |
| Competition | 3 | 20.886 | 0.001 | 3 | 0.956 | 0.423 | 3 | 0.359 | 0.783 | 3 | 4.212 | 0.011 | 3 | 6.698 | 0.001 |
| Watering | 1 | 0.038 | 0.847 | 1 | 0.940 | 0.338 | 1 | 1.554 | 0.220 | 1 | 0.059 | 0.809 | 1 | 1.114 | 0.297 |
| C × W | 3 | 0.029 | 0.993 | 3 | 0.632 | 0.599 | 3 | 0.638 | 0.595 | 3 | 0.684 | 0.567 | 3 | 0.380 | 0.768 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zunzunegui, M.; Esquivias, M.P.; Díaz Barradas, M.C.; Gallego-Fernández, J.B.; Álvarez-Cansino, L. Interspecific Competition and Intraspecific Facilitation Shape Coastal Dune Shrub Responses to Experimental Drought. Plants 2025, 14, 2663. https://doi.org/10.3390/plants14172663
Zunzunegui M, Esquivias MP, Díaz Barradas MC, Gallego-Fernández JB, Álvarez-Cansino L. Interspecific Competition and Intraspecific Facilitation Shape Coastal Dune Shrub Responses to Experimental Drought. Plants. 2025; 14(17):2663. https://doi.org/10.3390/plants14172663
Chicago/Turabian StyleZunzunegui, María, Mari Paz Esquivias, Mari Cruz Díaz Barradas, Juan B. Gallego-Fernández, and Leonor Álvarez-Cansino. 2025. "Interspecific Competition and Intraspecific Facilitation Shape Coastal Dune Shrub Responses to Experimental Drought" Plants 14, no. 17: 2663. https://doi.org/10.3390/plants14172663
APA StyleZunzunegui, M., Esquivias, M. P., Díaz Barradas, M. C., Gallego-Fernández, J. B., & Álvarez-Cansino, L. (2025). Interspecific Competition and Intraspecific Facilitation Shape Coastal Dune Shrub Responses to Experimental Drought. Plants, 14(17), 2663. https://doi.org/10.3390/plants14172663







