Field Application of Tea Volatiles Mediating the Selectivity of Aleurocanthus spiniferus on Four Tea Cultivars
Abstract
1. Introduction
2. Results
2.1. Population Dynamics of A. spiniferus on Four Tea Cultivars
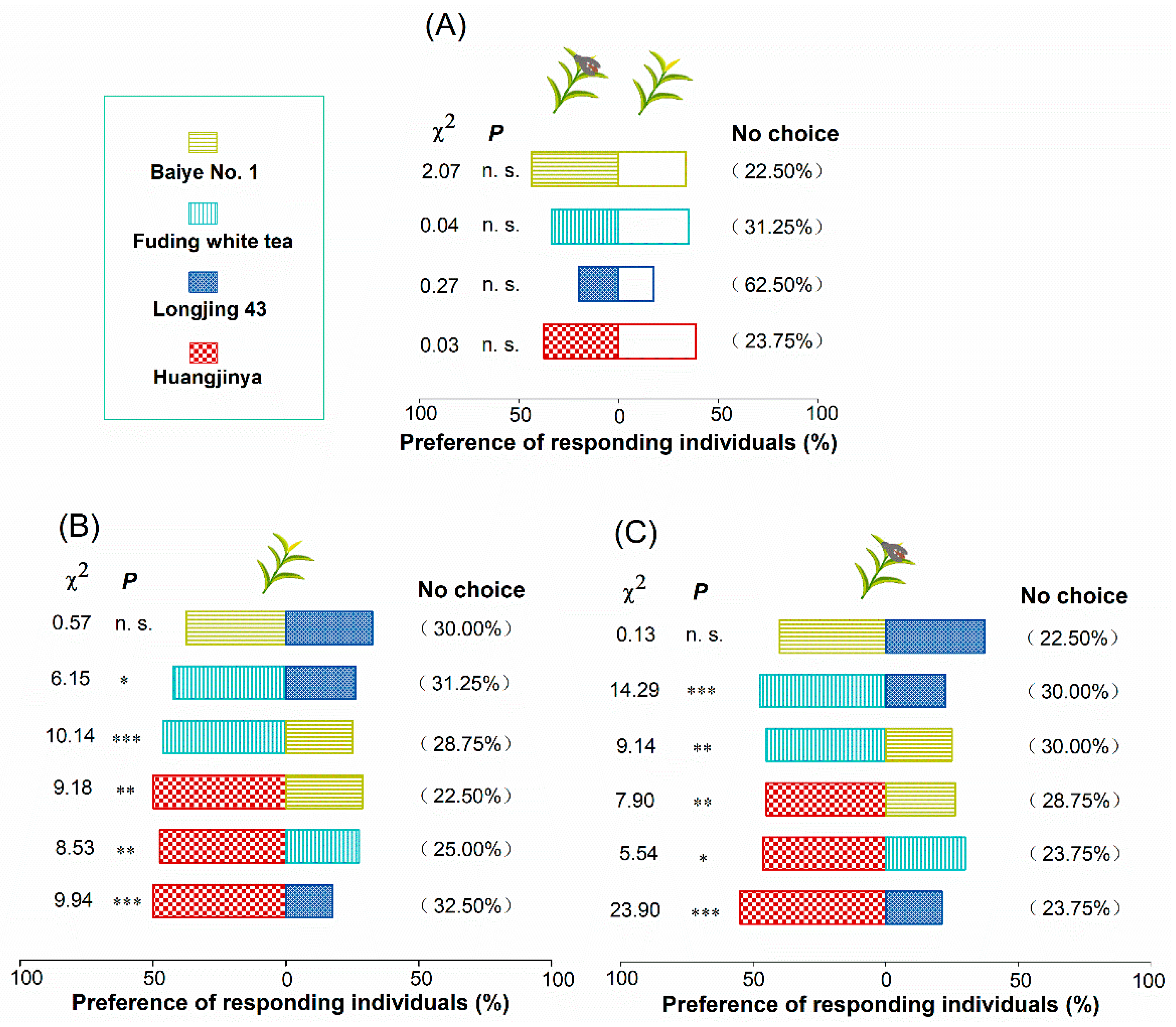
2.2. Preference of A. spiniferus Among Four Tea Cultivars
2.3. Volatile Profiles of Tested Tea Cultivars
2.4. Electroantennogram (EAG) Responses Elicited by Synthetic Compounds of Tea Volatiles
2.5. Preference of A. spiniferus for Synthetic Compounds of Tea Volatiles
2.6. Field Trials of Synthetic Compounds and Blends of Tea Volatiles
3. Discussion
4. Materials and Methods
4.1. Tea Cultivars, Insect Collection, and Chemicals
4.2. Population Dynamics of A. spiniferus on Four Tea Cultivars
4.3. Y-Tube Test for Behavioral Responses of Adult A. spiniferus to Tea Shoots and Synthetic Volatile Compounds
4.4. Volatile Collection and Analysis
4.5. Electroantennogram Experiments
4.6. Field Trial
4.7. Data Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhao, M.Y.; Zhang, N.; Gao, T.; Jin, J.Y.; Jing, T.T.; Wang, J.M.; Wu, Y.; Wan, X.C.; Schwab, W.; Song, C.K. Sesquiterpene glucosylation mediated by glucosyltransferase UGT91Q2 is involved in the modulation of cold stress tolerance in tea plants. New Phytol. 2020, 226, 362–372. [Google Scholar] [CrossRef]
- Samanta, S. Potential bioactive components and health promotional benefits of tea (Camellia sinensis). J. Am. Nutr. Assoc. 2020, 41, 65–93. [Google Scholar] [CrossRef]
- Kaczyński, P.; Iwaniuk, P.; Jankowska, M.; Orywal, K.; Socha, K.; Perkowski, M.; Farhan, J.A.; Łozowicka, B. Pesticide residues in common and herbal teas combined with risk assessment and transfer to the infusion. Chemosphere 2024, 367, 143550. [Google Scholar] [CrossRef]
- Wang, Q.; Liu, X.; Zhang, X.; Hou, Y.; Hu, X.; Liang, X.; Chen, X. Influence of tea saponin on enhancing accessibility of pyrene and cadmium phytoremediated with Lolium multiflorum in co-contaminated soils. Environ. Sci. Pollut. Res. 2016, 23, 5705–5711. [Google Scholar] [CrossRef] [PubMed]
- Cay, S. Enhancement of cadmium uptake by Amaranthus caudatus, an ornamental plant, using tea saponin. Environ. Monit. Assess. 2016, 188, 320. [Google Scholar] [CrossRef]
- Liu, X.; Cao, L.; Wang, Q.; Zhang, X.; Hu, X. Effect of tea saponin on phytoremediation of Cd and pyrene in contaminated soils by Lolium multiflorum. Environ. Sci. Pollut. Res. 2017, 24, 18946–18952. [Google Scholar] [CrossRef]
- Ye, M.; Sun, M.; Xie, S.; Liu, K.; Feng, Y.; Zhao, Y.; Wan, J.; Hu, F.; Li, H.; Zong, L.; et al. Feasibility of tea saponin-enhanced soil washing in a soybean oil-water solvent system to extract PAHs/Cd/Ni efficiently from a coking plant site. Pedosphere 2017, 27, 452–464. [Google Scholar] [CrossRef]
- Yu, X.L.; He, Y. Development of a rapid and simple method for preparing tea-leaf saponins and investigation on their surface tension differences compared with tea-seed saponins. Molecules 2018, 23, 1796. [Google Scholar] [CrossRef]
- Demirbas, A. Tea seed upgrading facilities and economic assessment of biodiesel production from tea seed oil. Energy Convers. Manag. 2010, 51, 2595–2599. [Google Scholar] [CrossRef]
- Tang, X.T.; Tao, H.H.; Du, Y.Z. Microsatellite-based analysis of the genetic structure and diversity of Aleurocanthus spiniferus (Hemiptera: Aleyrodidae) from tea plants in China. Gene 2015, 560, 107–113. [Google Scholar] [CrossRef]
- Byrne, D.N.; Bellows, T.S. Whitefly biology. Annu. Rev. Entomol. 1991, 36, 431–457. [Google Scholar] [CrossRef]
- Uesugi, R.; Sato, Y.; Han, B.Y.; Huang, Z.D.; Yara, K.; Furuhashi, K. Molecular evidence for multiple phylogenetic groups within two species of invasive spiny whiteflies and their parasitoid wasp. Bull. Entomol. Res. 2016, 106, 328–340. [Google Scholar] [CrossRef]
- Bragard, C.; Dehnen-Schmutz, K.; Di Serio, F.; Gonthier, P.; Jacques, M.A.; Miret, J.A.J.; Justesen, A.F.; Magnusson, C.S.; Milonas, P.; Navas-Cortes, J.A.; et al. Pest categorisation of Aleurocanthus spp. EFSA J. 2018, 16, 5436. [Google Scholar] [CrossRef]
- Yang, T.B.; Song, X.H.; Zhong, Y.; Wang, B.; Zhou, C. Field investigation– and dietary metabarcoding-based screening of arthropods that prey on primary tea pests. Ecol. Evol. 2022, 12, e9060. [Google Scholar] [CrossRef]
- Tian, Y.Y.; Chen, Z.J.; Huang, X.Q.; Zhang, L.; Zhang, Z. Evaluation of botanicals for management of piercing-sucking pests and the effect on beneficial arthropod populations in tea trees Camellia sinensis (L.) O. Kuntze (Theaceae). J. Insect Sci. 2020, 20, 27. [Google Scholar] [CrossRef]
- Heshmati, A.; Fereshteh, M.; Khaneghah, A.M. Simultaneous multi-determination of pesticide residues in black tea leaves and infusion: A risk assessment study. Environ. Sci. Pollut. Res. 2021, 28, 13725–13735. [Google Scholar] [CrossRef]
- Notification. Exceedance of the Maximum Residue Level (MRL) for Pesticide Dinotefuran in Black Tea From Taiwan. 2022. Available online: https://webgate.ec.europa.eu/rasff-window/screen/notification/566619 (accessed on 6 August 2025).
- Notification. Exceeding the Maximum Permissible Level of Pesticide Residues: Dinotefuran and Tolfenpyrad in the Product Entitled: “Black Tea—Black Tea. 2022. Available online: https://webgate.ec.europa.eu/rasff-window/screen/notification/569674 (accessed on 6 August 2025).
- Notification. Fenobucarb in Black Leaf Tea. 2024. Available online: https://webgate.ec.europa.eu/rasff-window/screen/notification/653652 (accessed on 6 August 2025).
- Gao, J.; Zhang, T.; Fang, Y.; Zhao, Y.; Yang, M.; Zhao, L.; Li, Y.; Huang, Y.; Zhu, G.; Guo, Y. On-site rapid detection of multiple pesticide residues in tea leaves by lateral flow immunoassay. J. Pharm. Anal. 2024, 14, 276–283. [Google Scholar] [CrossRef]
- Bruce, T.J.A.; Wadhams, L.J.; Woodcock, C.M. Insect host location: A volatile situation. Trends Plant Sci. 2005, 10, 269–274. [Google Scholar] [CrossRef]
- Matsui, K.; Engelberth, J. Green leaf volatiles—The forefront of plant responses against biotic attack. Plant Cell Physiol. 2022, 63, 1378–1390. [Google Scholar] [CrossRef]
- Ranger, C.M.; Winter, R.E.K.; Backus, E.A.; Rottinghaus, G.E.; Ellersieck, M.R.; Johnson, D.W. Discrimination by the potato leafhopper (Hemiptera: Cicadellidae) of host volatiles from resistant and susceptible alfalfa, Medicago sativa L. Environ. Entomol. 2005, 34, 271–280. [Google Scholar] [CrossRef]
- Huang, Q.T.; Han, X.Q.; Zhang, G.J.; Zhu-Salzman, K.; Cheng, W.N. Plant volatiles mediate host selection of Sitodiplosis mosellana (Diptera: Cecidomyiidae) among wheat varieties. J. Agric. Food Chem. 2022, 70, 10466–10475. [Google Scholar] [CrossRef]
- Yuan, G.G.; Zhao, L.C.; Du, Y.W.; Yu, H.; Shi, X.B.; Chen, W.C. Repellence or attraction: Secondary metabolites in pepper mediate attraction and defense against Spodoptera litura. Pest Manag. Sci. 2022, 78, 4859–4870. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Bian, L.; Sun, X.L.; Luo, Z.X.; Xin, Z.J.; Luo, F.J. Electrophysiological and behavioural responses of the tea geometrid Ectropis obliqua (Lepidoptera: Geometridae) to volatiles from a non-host plant, rosemary, Rosmarinus officinalis (Lamiaceae). Pest Manag. Sci. 2015, 71, 96–104. [Google Scholar] [CrossRef]
- Li, Y.F.; Zhong, S.T.; Qin, Y.C.; Zhang, S.Q.; Gao, Z.L.; Dang, Z.H.; Pan, W.L. Identification of plant chemicals attracting and repelling whiteflies. Arthropod-Plant Interact. 2014, 8, 183–190. [Google Scholar] [CrossRef]
- Zhang, L.; Su, Q.F.; Wang, L.S.; Lv, M.W.; Hou, Y.X.; Li, S.S. Linalool: A ubiquitous floral volatile mediating the communication between plants and insects. J. Syst. Evol. 2023, 61, 538–549. [Google Scholar] [CrossRef]
- Lin, J.; Wang, D.; Chen, X.; Köllner, T.G.; Mazarei, M.; Guo, H.; Pantalone, V.R.; Arelli, P.; Stewart, C.N., Jr.; Wang, N.N.; et al. An (E,E)-α-farnesene synthase gene of soybean has a role in defence against nematodes and is involved in synthesizing insect-induced volatiles. Plant Biotechnol. J. 2017, 15, 510–519. [Google Scholar] [CrossRef]
- Birkett, M.A.; Bruce, T.J.A.; Martin, J.L.; Smart, L.E.; Oakley, J.; Wadhams, L.J. Responses of female orange wheat blossom midge, Sitodiplosis mosellana, to wheat panicle volatiles. J. Chem. Ecol. 2004, 30, 1319–1328. [Google Scholar] [CrossRef]
- Yang, G.; Zhang, Y.N.; Gurr, G.M.; Vasseur, L.; You, M.S. Electroantennogram and behavioral responses of Cotesia plutellae to plant volatiles. Insect Sci. 2016, 23, 245–252. [Google Scholar] [CrossRef]
- Xiu, C.L.; Xu, B.; Pan, H.S.; Zhang, W.; Yang, Y.Z.; Lu, Y.H. Volatiles from Sophora japonica flowers attract Harmonia axyridis adults (Coleoptera: Coccinellidae). J. Integr. Agric. 2019, 18, 873–883. [Google Scholar] [CrossRef]
- Wang, J.; Wei, J.; Yi, T.; Li, Y.Y.; Xu, T.; Chen, L.; Xu, H.H. A green leaf volatile, (Z)-3-hexenyl-acetate, mediates differential oviposition by Spodoptera frugiperda on maize and rice. BMC Biol. 2023, 21, 140. [Google Scholar] [CrossRef]
- Baraki, Y.; Woldehawariat, Y.; Dekker, T.; Biasazin, T.D. Comparison of electrophysiological and behavioral responses of tephritid (Diptera: Tephritidae) pests to cucurbit host odors. J. Insect Physiol. 2025, 165, 104865. [Google Scholar] [CrossRef]
- Chen, K.; Huang, M.X.; Shi, Q.C.; Xie, X.; Jin, L.H.; Xu, W.M.; Li, X.Y. Screening of a potential leafhopper attractants and their applications in tea plantations. J. Environ. Sci. Health Part B 2019, 54, 858–865. [Google Scholar] [CrossRef]
- Wang, H.M.; Bai, P.H.; Zhang, J.; Zhang, X.M.; Hui, Q.; Zheng, H.X.; Zhang, X.H. Attraction of bruchid beetles Callosobruchus chinensis (L.)(Coleoptera: Bruchidae) to host plant volatiles. J. Integr. Agric. 2020, 19, 3035–3044. [Google Scholar] [CrossRef]
- Cai, X.M.; Luo, Z.X.; Meng, Z.N.; Liu, Y.; Chu, B.; Bian, L.; Li, Z.Q.; Xin, Z.J.; Chen, Z.M. Primary screening and application of repellent plant volatiles to control tea leafhopper, Empoasca onukii Matsuda. Pest Manag. Sci. 2020, 76, 1304–1312. [Google Scholar] [CrossRef]
- Sun, X.L.; Wang, G.C.; Gao, Y.; Zhang, X.Z.; Xin, Z.J.; Chen, Z.M. Volatiles emitted from tea plants infested by Ectropis obliqua larvae are attractive to conspecific moths. J. Chem. Ecol. 2014, 40, 1080–1089. [Google Scholar] [CrossRef]
- Mu, D.; Cui, L.; Ge, J.; Wang, M.X.; Liu, L.F.; Yu, X.P.; Zhang, Q.H.; Han, B.Y. Behavioral responses for evaluating the attractiveness of specific tea shoot volatiles to the tea green leafhopper, Empoaca vitis. Insect Sci. 2012, 19, 229–238. [Google Scholar] [CrossRef]
- Koczor, S.; Vuts, J.; Caulfield, J.C.; Withall, D.M.; Sarria, A.; Pickett, J.A.; Birkett, M.A.; Csonka, É.B.; Tóth, M. Sex pheromone of the alfalfa plant bug, Adelphocoris lineolatus: Pheromone composition and antagonistic effect of 1-hexanol (Hemiptera: Miridae). J. Chem. Ecol. 2021, 47, 525–533. [Google Scholar] [CrossRef]
- Machado, V.E.; Silva, F.B.D.; Goulart, T.M.; Pinto, M.C. Behavioral responses of sand fly Nyssomyia neivai (Psychodidae: Phlebotominae) to 1-hexanol and light. Acta Trop. 2022, 236, 106680. [Google Scholar] [CrossRef]
- Yu, H.L.; Zhang, Y.J.; Wu, K.M.; Gao, X.W.; Guo, Y.Y. Field-testing of synthetic herbivore-induced plant volatiles asattractants for beneficial insects. Environ. Entomol. 2008, 37, 1410–1415. [Google Scholar] [CrossRef]
- Tian, Y.; Zhao, Y.; Zhang, L.; Mu, W.; Zhang, Z.Q. Morphological, physiological, and biochemical responses of two tea cultivars to Empoasca onukii (Hemiptera: Cicadellidae) infestation. J. Econ. Entomol. 2018, 111, 899–908. [Google Scholar] [CrossRef]
- Lu, C.; Shen, N.; Jiang, W.; Xie, B.; Zhao, N.; Zhou, G.L.; Zhao, D.G.; He, Y.Q.; Chen, W.L. Different tea germplasms distinctly influence the adaptability of Toxoptera aurantii (Hemiptera: Aphididae). Insects 2023, 14, 695. [Google Scholar] [CrossRef]
- He, Y.; Lu, C.; Jiang, W.; Chen, W.L.; Fan, J.J.; Niu, S.Z.; Zhao, D.G. Adaptability of Toxoptera aurantii (Hemiptera: Aphididae) to different tea cultivars. Agriculture 2023, 13, 2039. [Google Scholar] [CrossRef]
- Liang, L.Y.; Liu, L.F.; Yu, X.P.; Han, B.Y. Evaluation of the resistance of different tea cultivars to tea aphids by EPG technique. J. Integr. Agric. 2012, 11, 2028–2034. [Google Scholar] [CrossRef]
- Khan, M.; Khan, M.; Alshareef, E.; Alaqeel, S.L.; Alkhathlan, H.Z. Chemical characterization and chemotaxonomic significance of essential oil constituents of Matricaria aurea grown in two different Agro-Climatic conditions. Plants 2023, 12, 3553. [Google Scholar] [CrossRef]
- Bruce, T.J.A.; Pickett, J.A. Perception of plant volatile blends by herbivorous insects−finding the right mix. Phytochemistry 2011, 72, 1605–1611. [Google Scholar] [CrossRef]
- Li, C.F.; Ma, J.Q.; Huang, D.J.; Ma, C.L.; Jin, J.Q.; Yao, M.Z.; Chen, L. Comprehensive dissection of metabolic changes in albino and green tea cultivars. J. Agric. Food Chem. 2018, 66, 2040–2048. [Google Scholar] [CrossRef]
- Conboy, N.J.; McDaniel, T.; Ormerod, A.; George, D.; Gatehouse, A.M.R.; Wharton, E.; Donohoe, P.; Curtis, R.; Tosh, C.R. Companion planting with French marigolds protects tomato plants from glasshouse whiteflies through the emission of airborne limonene. PLoS ONE 2019, 14, e0213071. [Google Scholar] [CrossRef]
- Wang, X.W.; Zeng, L.T.; Liao, Y.Y.; Li, J.L.; Tang, J.C.; Yang, Z.Y. Formation of α-farnesene in tea (Camellia sinensis) leaves induced by herbivore-derived wounding and its effect on neighboring tea plants. Int. J. Mol. Sci. 2019, 20, 4151. [Google Scholar] [CrossRef]
- Dong, F.; Yang, Z.; Baldermann, S.; Sato, Y.; Asai, T.; Watanabe, N. Herbivore-induced volatiles from tea (Camellia sinensis) plants and their involvement in intraplant communication and changes in endogenous nonvolatile metabolites. J. Agric. Food Chem. 2011, 59, 13131–13135. [Google Scholar] [CrossRef]
- Cai, X.M.; Sun, X.L.; Dong, W.X.; Wang, G.C.; Chen, Z.M. Herbivore species, infestation time, and herbivore density affect induced volatiles in tea plants. Chemoecology 2014, 24, 1–14. [Google Scholar] [CrossRef]
- Jing, T.T.; Zhang, N.; Gao, T.; Zhao, M.Y.; Jin, J.Y.; Chen, Y.X.; Xu, M.J.; Wan, X.C.; Schwab, W.; Song, C.K. Glucosylation of (Z)-3-hexenol informs intraspecies interactions in plants: A case study in Camellia sinensis. Plant Cell Environ. 2019, 42, 1352–1367. [Google Scholar] [CrossRef]
- Karmakar, A.; Mitra, P.; Koner, A.; Das, S.; Barik, A. Fruit volatiles of creeping cucumber (Solena amplexicaulis) attract a generalist insect herbivore. J. Chem. Ecol. 2020, 46, 275–287. [Google Scholar] [CrossRef]
- Galizia, C.G.; Rössler, W. Parallel olfactory systems in insects: Anatomy and function. Annu. Rev. Entomol. 2010, 55, 399–420. [Google Scholar] [CrossRef]
- Barbosa-Cornelio, R.; Cantor, F.; Coy-Barrera, E.; Rodríguez, D. Tools in the investigation of volatile semiochemicals on insects: From sampling to statistical analysis. Insects 2019, 10, 241. [Google Scholar] [CrossRef]
- Sánchez-Gracia, A.; Vieira, F.G.; Rozas, J. Molecular evolution of the major chemosensory gene families in insects. Heredity 2009, 103, 208–216. [Google Scholar] [CrossRef]
- Yi, X.; Zhang, Y.; Wang, P.; Qi, J.W.; Hu, M.Y.; Zhong, G.H. Ligands binding and molecular simulation: The potential investigation of a biosensor based on an insect odorant binding protein. Int. J. Biol. Sci. 2015, 11, 75–87. [Google Scholar] [CrossRef]
- Lizana, P.; Mutis, A.; Quiroz, A.; Venthur, H. Insights into chemosensory proteins from non-model insects: Advances and perspectives in the context of pest management. Front. Physiol. 2022, 13, 924750. [Google Scholar] [CrossRef]
- Gao, Y.Q.; Chen, Z.Z.; Liu, M.Y.; Song, C.Y.; Jia, Z.F.; Liu, F.H.; Qu, C.; Dewer, Y.; Zhao, H.P.; Xu, Y.Y.; et al. Characterization of antennal chemosensilla and associated chemosensory genes in the orange spiny whitefly, Aleurocanthus spiniferus (Quaintanca). Front. Physiol. 2022, 13, 847895. [Google Scholar] [CrossRef]
- Yan, H.X.; Zeng, J.W.; Zhong, G.Y. The push-pull strategy for citrus psyllid control. Pest Manag. Sci. 2015, 71, 893–896. [Google Scholar] [CrossRef]
- Renkema, J.M.; Buitenhuis, R.; Hallett, R.H. Optimizing trap design and trapping protocols for Drosophila suzukii (Diptera: Drosophilidae). J. Econ. Entomol. 2014, 107, 2107–2118. [Google Scholar] [CrossRef]
- Jakuš, R.; Modlinger, R.; Kašpar, J.; Majdák, A.; Blaženec, M.; Korolyova, N.; Jirošová, A.; Schlyter, F. Testing the efficiency of the push-and-pull strategy during severe Ips typographus outbreak and extreme drought in norway spruce stands. Forests 2022, 13, 2175. [Google Scholar] [CrossRef]
- Wang, L.Y.; Yang, C.X.; Mei, X.D.; Guo, B.B.; Yang, X.L.; Zhang, T.; Ning, J. Identification of sex pheromone in Macdunnoughia crassisigna Warren (Lepidoptera: Noctuidae) and field optimization of the sex attractant. Pest Manag. Sci. 2024, 80, 577–585. [Google Scholar] [CrossRef]
- Ryall, K.L.; Silk, P.J.; Fidgen, J.; Mayo, P.; Lavallée, R.; Guertin, C.; Scarr, T. Effects of pheromone release rate and trap placement on trapping of Agrilus planipennis (Coleoptera: Buprestidae) in Canada. Environ. Entomol. 2015, 44, 734–745. [Google Scholar] [CrossRef]
- Lopez, M.D.; Maudhuit, A.; Pascual-Villalobos, M.J.; Poncelet, D. Development of formulations to improve the controlled-release of linalool to be applied as an insecticide. J. Agric. Food Chem. 2012, 60, 1187–1192. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Chen, L.; Liu, Y.; Chen, J.L.; Francis, F. Use of slow-release plant infochemicals to control aphids: A first investigation in a Belgian wheat field. Sci. Rep. 2016, 6, 31552. [Google Scholar] [CrossRef]
- Jing, T.; Qian, X.; Du, W.; Gao, T.; Li, D.F.; Guo, D.Y.; He, F.; Yu, G.M.; Li, S.P.; Schwab, W.; et al. Herbivore-induced volatiles influence moth preference by increasing the β-ocimene emission of neighbouring tea plants. Plant Cell Environ. 2021, 44, 3667–3680. [Google Scholar] [CrossRef]
- Xin, Z.; Ge, L.; Chen, S.; Sun, X.L. Enhanced transcriptome responses in herbivore-infested tea plants by the green leaf volatile (Z)-3-hexenol. J. Plant Res. 2019, 132, 285–293. [Google Scholar] [CrossRef]
- Liu, M.Y.; Wang, F.D.; Zhao, Y.J.; Liu, Y.N.; Kang, Z.W.; Xu, Y.Y.; Chen, Z.Z. Study on overwintering stage and spatial distribution pattern of Aleurocanthus spiniferus in tea plantation. J. Environ. Entomol. 2023, 45, 73–82. [Google Scholar] [CrossRef]
- Gong, X.; Huang, J.; Xu, Y.; Li, Z.B.; Li, L.; Li, D.; Belwal, T.; Jeamdet, P.; Luo, Z.S.; Xu, Y.Q. Deterioration of plant volatile organic compounds in food: Consequence, mechanism, detection, and control. Trends Food Sci. Technol. 2023, 131, 61–76. [Google Scholar] [CrossRef]
- De Brito-Machado, D.; Ramos, Y.J.; Defaveri, A.C.A.E.; Queiroz, G.A.D.; Guimarães, E.F.; Moreira, D.D.L. Volatile chemical variation of essential oils and their correlation with insects, phenology, ontogeny and microclimate: Piper mollicomum Kunth, a case of study. Plants 2022, 11, 3535. [Google Scholar] [CrossRef]
- Vallat, A.; Gu, H.; Dorn, S. How rainfall, relative humidity and temperature influence volatile emissions from apple trees in situ. Phytochemistry 2005, 66, 1540–1550. [Google Scholar] [CrossRef]
- Cai, X.; Guo, Y.; Bian, L.; Luo, Z.X.; Li, Z.Q.; Xiu, C.L.; Fu, N.X.; Chen, Z.M. Variation in the ratio of compounds in a plant volatile blend during transmission by wind. Sci. Rep. 2022, 12, 6176. [Google Scholar] [CrossRef]
- Heil, M.; Silva Bueno, J.C. Within-plant signaling by volatiles leads to induction and priming of an indirect plant defense in nature. Proc. Natl. Acad. Sci. USA 2007, 104, 5467–5472. [Google Scholar] [CrossRef]
- Xu, X.X.; Cai, X.M.; Bian, L.; Luo, Z.X.; Li, Z.Q.; Chen, Z.M. Does background odor in tea gardens mask attractants? Screening and application of attractants for Empoasca onukii Matsuda. J. Econ. Entomol. 2017, 110, 2357–2363. [Google Scholar] [CrossRef]
- Douma, J.C.; Ganzeveld, L.N.; Unsicker, S.B.; Boeckler, G.A.; Dicke, M. What makes a volatile organic compound a reliable indicator of insect herbivory? Plant Cell Environ. 2019, 42, 3308–3325. [Google Scholar] [CrossRef]
- Cha, D.H.; Loeb, G.M.; Linn, C.E.J.; Hesler, S.P.; Landolt, P.J. A multiple-choice bioassay approach for rapid screening of key attractant volatiles. Environ. Entomol. 2018, 47, 946–950. [Google Scholar] [CrossRef]
- Qu, C.J.; Shi, J.T.; Wang, C.Z.; Guo, H.; Jiang, X.J. Identification of an adult attractant for Anomala corpulenta by the reverse chemical ecology approach. J. Pest Sci. 2024, 97, 1853–1863. [Google Scholar] [CrossRef]
- Han, B.Y.; Chen, Z.M. Behavioral and electrophysiological responses ofnatural enemies to synomones from tea shoots and kairomones from tea aphids, Toxoptera aurantii. J. Chem. Ecol. 2002, 28, 2203–2219. [Google Scholar] [CrossRef]
- Sun, X.L.; Wang, G.C.; Cai, X.M.; Jin, S.; Gao, Y.; Chen, Z.M. The tea weevil, Myllocerinus aurolineatus, is attracted to volatiles induced by conspecifics. J. Chem. Ecol. 2010, 36, 388–395. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, J.; Du, W.; Guo, D.Y.; Tang, X.Y.; Zhao, W.; Lu, M.; Yu, K.K.; Luo, Z.W.; Chen, Y.S.; et al. Distribution of and temporal variation in volatiles in tea (Camellia sinensis) flowers during the opening stages. J. Agric. Food Chem. 2023, 71, 19682–19693. [Google Scholar] [CrossRef]
- Ayelo, P.M.; Yusuf, A.A.; Pirk, C.W.; Chailleux, A.; Mohamed, S.A.; Deletre, E. Terpenes from herbivore-induced tomato plant volatiles attract Nesidiocoris tenuis (Hemiptera: Miridae), a predator of major tomato pests. Pest Manag. Sci. 2021, 77, 5255–5267. [Google Scholar] [CrossRef]
- Li, X.; Zhang, X.G.; Chun, X.; Gao, Y.L.; Dong, W.X. Behavioral responses of potato tuber moth (Phthorimaea operculella) to tobacco plant volatiles. J. Integr. Agric. 2020, 19, 325–332. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, X.; Wang, R.; Chen, S.L.; Zhang, T. Monoterpene alcohols induced by sweet potato weevil larvae deter conspecific adults from feeding and oviposition. Pest Manag. Sci. 2023, 79, 3504–3510. [Google Scholar] [CrossRef]
- Delorme, J.D.; Payne, T.L. Antennal olfactory responses of black turpentine beetle, Dendroctonus terebrans (Olivier), to bark beetle pheromones and host terpenes. J. Chem. Ecol. 1990, 16, 1321–1329. [Google Scholar] [CrossRef]









| Volatile Compounds | CAS | Retention Time (min) | Retention Index † | Retention Index ‡ | Concentration (ng plant−1 h−1) | |||
|---|---|---|---|---|---|---|---|---|
| Baiye No. 1 | Fuding White Tea | Huangjinya | Longjing 43 | |||||
| Hexanol | 111-27-3 | 3.21 | 859 | 863 | 6712 ± 562 a | 3728 ± 959.3 ab | 1668 ± 322.8 b | 5634 ± 688.4 a |
| (E)-2-Hexenal | 6728-26-3 | 7.21 | 867 | 868 | 235.8 ± 49.13 ab | 167.6 ± 21.43 ab | 90.83 ± 21.33 b | 249.5 ± 37.39 a |
| (Z)-3-Hexenol | 928-96-1 | 7.42 | 875 | 877 | 48.28 ± 4.37 a | 29.11 ± 4.10 a | ND | 32.96 ± 4.83 a |
| Benzaldehyde | 100-52-7 | 8.43 | 957 | 956 | 567.6 ± 124.8 a | 406 ± 84.73 a | 172.9 ± 88.29 a | 235.6 ± 35.35 a |
| 2-Ethyl-1-hexanol | 104-76-7 | 11.80 | 1031 | 1038 | 228.9 ± 64.03 a | 251 ± 51.94 a | 220.9 ± 61.29 a | 425.1 ± 67.08 a |
| (E)-β-Ocimene | 3779-61-1 | 12.94 | 1052 | 1058 | 1045 ± 398.3 a | 789.5 ± 158.5 a | 566 ± 154.6 a | 540 ± 169.5 a |
| Linalool | 78-70-6 | 15.62 | 1082 | 1075 | ND | 96.29 ± 23.63 | ND | ND |
| Nonanal | 124-19-6 | 15.93 | 1102 | 1095 | 60.37 ± 18.47 a | 34.35 ± 4.22 a | 14.54 ± 3.72 a | ND |
| (Z)-3-Hexenyl butanoate | 16491-36-4 | 19.11 | 1179 | 1186 | 1025 ± 248.8 a | 858.8 ± 149.7 a | 689 ± 179.8 a | 784.7 ± 237.2 a |
| 7-Methyl-heptadecane | 20959-33-5 | 26.62 | 1334 | 1330 | 681.6 ± 202 b | 636 ± 82.64 b | 1816 ± 137.9 a | 1356 ± 336.4 ab |
| (E,E)-α-Farnesene | 21499-64-9 | 30.69 | 1505 § | 1510 | ND | ND | 34.25 ± 2.60 | ND |
| Cadalene | 483-78-3 | 40.56 | 1673 | 1674 | 814.2 ± 256.8 a | 963.2 ± 174.4 a | 766.7 ± 160 a | 1714 ± 303.5 a |
| Caffeine | 58-08-2 | 46.29 | 1840 | 1835 | 227.8 ± 47.99 a | 303.4 ± 41.73 a | 252.3 ± 50.87 a | 415.1 ± 64.31 a |
| Code | Component | Ratio |
|---|---|---|
| Blend 1 | Hexanol, nonanal, (E)-2-hexenal, (Z)-3-hexenol, linalool, (E,E)-α-farnesene | 55:1:3:1:3:1 (v/v/v/v/v/v) |
| Blend 2 | Hexanol, (Z)-3-hexenol, linalool, (E,E)-α-farnesene | 55:1:3:1 (v/v/v) |
| Blend 3 | Nonanal, (E)-2-hexenal | 1:3 (v/v) |
| Blend 4 | (E,E)-α-farnesene, linalool | 1:3 (v/v) |
| Blend 5 | (Z)-3-hexenol, (E,E)-α-farnesene | 1:1 (v/v) |
| Blend 6 | (Z)-3-hexenol, linalool | 1:3 (v/v) |
| Blend 7 | Hexanol, (Z)-3-hexenol | 55:1(v/v) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, Z.; Ge, X.; Bian, Y.; Song, K.; Li, D.; Song, D.; Ding, S.; Xu, Y.; Chen, Z. Field Application of Tea Volatiles Mediating the Selectivity of Aleurocanthus spiniferus on Four Tea Cultivars. Plants 2025, 14, 2653. https://doi.org/10.3390/plants14172653
Jia Z, Ge X, Bian Y, Song K, Li D, Song D, Ding S, Xu Y, Chen Z. Field Application of Tea Volatiles Mediating the Selectivity of Aleurocanthus spiniferus on Four Tea Cultivars. Plants. 2025; 14(17):2653. https://doi.org/10.3390/plants14172653
Chicago/Turabian StyleJia, Zhifei, Xiaoyu Ge, Yanan Bian, Kai Song, Dandan Li, Dapeng Song, Shibo Ding, Yongyu Xu, and Zhenzhen Chen. 2025. "Field Application of Tea Volatiles Mediating the Selectivity of Aleurocanthus spiniferus on Four Tea Cultivars" Plants 14, no. 17: 2653. https://doi.org/10.3390/plants14172653
APA StyleJia, Z., Ge, X., Bian, Y., Song, K., Li, D., Song, D., Ding, S., Xu, Y., & Chen, Z. (2025). Field Application of Tea Volatiles Mediating the Selectivity of Aleurocanthus spiniferus on Four Tea Cultivars. Plants, 14(17), 2653. https://doi.org/10.3390/plants14172653






