Productivity and Species Diversity of Plant Communities Are Higher inside than outside the West Ordos National Nature Reserve, Northern China
Abstract
1. Introduction
2. Results
2.1. Comparison of Biomass of Plant Communities inside and outside Nature Reserve
2.2. Comparison of Plant Community Biodiversity inside and outside Nature Reserve
2.2.1. Plant Community α-Diversity
2.2.2. Plant Community β-Diversity
2.3. Comparison of Soil Nutrient Contents inside and outside Nature Reserve
2.4. The Relationship between Plant Community Aboveground Biomass and α-Diversity
2.5. Correlation of Plant Community α-Diversity with Soil Nutrients
3. Discussion
3.1. Differences in Plant Community Characteristics and Soil Nutrients inside and outside Desert Steppe Nature Reserves
3.2. The Relationship between Plant Community Aboveground Biomass and α-Diversity
3.3. Correlation of Plant Community α-Diversity with Soil Nutrients
4. Materials and Methods
4.1. Study Site
4.2. Sample Selection and Setting
4.3. Sample Collection and Determination Method
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Perkins, D.M.; Bailey, R.A.; Dossena, M.; Gamfeldt, L.; Reiss, J.; Trimmer, M.; Woodward, G. Higher biodiversity is required to sustain multiple ecosystem processes across temperature regimes. Global Change Biol. 2015, 21, 396–406. [Google Scholar] [CrossRef]
- She, Y.; Li, X.; Li, C.; Yang, P.; Song, Z.; Zhang, J. Relationship between Species Diversity and Community Stability in Degraded Alpine Meadows during Bare Patch Succession. Plants 2023, 12, 3582. [Google Scholar] [CrossRef]
- Yan, P.; Lu, X.; Li, W.; Zhang, J.; Li, P.; Li, Y.; Wang, K.; Ding, S. Seasonal Variations in Plant Species Diversity and Phylogenetic Diversity in Abandoned Farmland of China’s Huang–Huai Plain. Diversity 2023, 15, 922. [Google Scholar] [CrossRef]
- Isbell, F.; Calcagno, V.; Hector, A.; Connolly, J. High plant diversity is needed to maintain ecosystem services. Nature 2011, 477, 199–202. [Google Scholar] [CrossRef]
- Liang, H.; Fu, T.; Gao, H.; Li, M.; Liu, J. Climatic and Non-Climatic Drivers of Plant Diversity along an Altitudinal Gradient in the Taihang Mountains of Northern China. Diversity 2023, 15, 66. [Google Scholar] [CrossRef]
- Arroyo-Rodríguez, V.; Rös, M.; Escobar, F.; Melo, F.P.L.; Santos, B.A.; Tabarelli, M.; Chazdon, R. Plant β-diversity in fragmented rain forests: Testing floristic homogenization and differentiation hypotheses. J. Ecol. 2013, 101, 1449–1458. [Google Scholar] [CrossRef]
- Wang, J.; Wang, Y.; Li, M.; He, N.; Li, J. Divergent roles of environmental and spatial factors in shaping plant β-diversity of different growth forms in drylands. Glob. Ecol. Conserv. 2021, 26, e01487. [Google Scholar] [CrossRef]
- Song, C.; Dannenberg, M.P.; Hwang, T. Optical remote sensing of terrestrial ecosystem primary productivity. Prog. Phys. Geog. 2013, 37, 834–854. [Google Scholar] [CrossRef]
- Cox, S.B.; Bloch, C.P.; Stevens, R.D.; Huenneke, L.F. Productivity and species richness in an arid ecosystem: A long-term perspective. Plant Ecol. 2006, 186, 1–12. [Google Scholar] [CrossRef]
- Paz-Kagan, T.; Shachak, M.; Zaady, E.; Karnieli, A. Evaluation of ecosystem responses to land-use change using soil quality and primary productivity in a semi-arid area, Israel. Agr. Ecosyst. Environ. 2014, 193, 9–24. [Google Scholar] [CrossRef]
- Sun, J.; Wang, N.; Niu, Z. Effect of Soil Environment on Species Diversity of Desert Plant Communities. Plants 2023, 12, 3465. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Jiang, L.; Wang, Z.; Song, M.; Wang, S. Phosphorus Enrichment Increased Community Stability by Increasing Asynchrony and Dominant Species Stability in Alpine Meadow of Qinghai-Tibet Plateau. J. Geophys. Res.-Biogeosci. 2022, 127, e2022JG006819. [Google Scholar] [CrossRef]
- Wang, X.; Dong, S.; Yang, B.; Li, Y.; Su, X. The effects of grassland degradation on plant diversity, primary productivity, and soil fertility in the alpine region of Asia’s headwaters. Environ. Monit. Assess. 2014, 186, 6903–6917. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yu, W.; Ma, L.; Ye, X.; Erdenebileg, E.; Wang, R.; Huang, Z.; Indree, T.; Liu, G. Biotic and abiotic drivers of ecosystem multifunctionality: Evidence from the semi-arid grasslands of northern China. Sci. Total Environ. 2023, 887, 164158. [Google Scholar] [CrossRef] [PubMed]
- Long, W.; Yang, X.; Li, D. Patterns of species diversity and soil nutrients along a chronosequence of vegetation recovery in Hainan Island, South China. Ecol. Res. 2012, 27, 561–568. [Google Scholar] [CrossRef]
- Stevens, M.H.H.; Carson, W.P. Phenological complementarity, species diversity, and ecosystem function. Oikos 2001, 92, 291–296. [Google Scholar] [CrossRef]
- Zuo, X.; Mao, W.; Qu, H.; Chen, M.; Zhao, S.; Liu, L.; Wang, S.; Yue, P.; Ma, X.; Zhao, X.; et al. Scale effects on spatial heterogeneity of herbaceous vegetation in desert steppe depend on plant community type. Ecol. Indic. 2021, 127, 107769. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, Z.; Han, G.; Wu, Q.; Schellenberg, M.P.; Gu, C. Grazing induced changes in plant diversity is a critical factor controlling grassland productivity in the Desert Steppe, Northern China. Agr. Ecosyst. Environ. 2018, 265, 73–83. [Google Scholar] [CrossRef]
- Xin, L.; Jin, Y.; Zhu, Y.; Luo, J.; Wang, L.; Chen, B.; Wang, W. Development effectiveness indicators of the nature reserve in China: Case study of the Anxi National Nature Reserve. J. Desert Res. 2015, 35, 1693–1699. [Google Scholar]
- Wu, R.; Zhang, S.; Yu, D.; Zhao, P.; Li, X.; Wang, L.; Yu, Q.; Ma, J.; Chen, A.; Long, Y. Effectiveness of China’s nature reserves in representing ecological diversity. Front. Ecol. Environ. 2011, 9, 383–389. [Google Scholar] [CrossRef]
- Burgess, N.D.; Loucks, C.; Stolton, S.; Dudley, N. The potential of forest reserves for augmenting the protected area network in Africa. Oryx 2007, 41, 151–159. [Google Scholar] [CrossRef]
- Victoria, C.J.; Cheng, M.; Chen, X. The development of landscape analysis of wetland function: A GIS based wetland functional assessment tool. In Proceedings of the WEFTEC 2002 Proceedings 75th Annual Conference and Exposition, Chicago, IL, USA, 28 September–2 October 2002. [Google Scholar]
- Tang, Z.; Fang, J.; Sun, J.; Gaston, K. Effectiveness of protected areas in maintaining plant production. PLoS ONE 2011, 6, e19116. [Google Scholar] [CrossRef]
- Hu, B.; Wen, Q.; Xi, F.; Li, M.; Wang, L.; Ren, Y. Effects of Enclosure on Grassland Ecological and Economic Benefits in Northern China. Chin. Geogr. Sci. 2023, 34, 67–78. [Google Scholar] [CrossRef]
- Munkhzul, O.; Oyundelger, K.; Narantuya, N.; Tuvshintogtokh, I.; Oyuntsetseg, B.; Wesche, K.; Jäschke, Y. Grazing Effects on Mongolian Steppe Vegetation-A Systematic Review of Local Literature. Front. Ecol. Evol. 2021, 9, 703220. [Google Scholar] [CrossRef]
- Liu, X.; Ma, Z.; Huang, X.; Li, L. How does grazing exclusion influence plant productivity and community structure in alpine grasslands of the Qinghai-Tibetan Plateau? Glob. Ecol. Conserv. 2020, 23, e01066. [Google Scholar] [CrossRef]
- Gagnaire-Renou, E.; Benoit, M.; Forget, P. Degradation of sandy arid shrubland environments: Observations, process modelling, and management implications. J. Arid Environ. 2001, 47, 123–144. [Google Scholar]
- Schlesinger, W.H.; Raikes, J.A. On the spatial pattern of soil nutrients in desert ecosystems. Ecology 1996, 77, 364–374. [Google Scholar] [CrossRef]
- Howard, K.S.C.; Eldridge, D.J.; Soliveres, S. Positive effects of shrubs on plant species diversity do not change along a gradient in grazing pressure in an arid shrubland. Basic Appl. Ecol. 2012, 13, 159–168. [Google Scholar] [CrossRef]
- Soliveres, S.; Eldridge, D.; Maestre, F.T.; Bowker, M.A.; Tighe, M.; Escudero, A. Microhabitat amelioration and reduced competition among understorey plants as drivers of facilitation across environmental gradients: Towards a unifying framework. Perspect. Plant Ecol. 2011, 13, 247–258. [Google Scholar] [CrossRef]
- Tilman, D.; Wedin, D.; Knops, J. Productivity and sustainability influenced by biodiversity in grassland ecosystems. Nature 1996, 379, 718–720. [Google Scholar] [CrossRef]
- Geldmann, J.; Barnes, M.; Coad, L.; Craigie, I.D.; Hockings, M.; Burgess, N.D. Effectiveness of terrestrial protected areas in reducing habitat loss and population declines. Biol. Conserv. 2013, 161, 230–238. [Google Scholar] [CrossRef]
- Coetzee, B.W.T.; Gaston, K.J.; Chown, S.L. Local scale comparisons of biodiversity as a test for global protected area ecological performance: A meta-analysis. PLoS ONE 2014, 9, e105824. [Google Scholar] [CrossRef]
- Huang, Y.; Fu, J.; Wang, W.; Li, J. Development of China’s nature reserves over the past 60 years: An overview. Land Use Policy 2019, 80, 224–232. [Google Scholar] [CrossRef]
- Shan, G.L.; Zhu, X. Influence of exclosure year on community structure and species diversity on a typical steppe. Acta Prataculturae Sin. 2008, 17, 772. [Google Scholar]
- Oba, G.; Vetaas, O.R.; Stenseth, N.C. Relationships between biomass and plant species richness in arid-zone grazing lands. J. Appl. Ecol. 2001, 38, 836–845. [Google Scholar] [CrossRef]
- Turner, M.G.; Gardner, R.H.; O’Neill, R.V. Landscape Ecology in Theory and Practice; Springer: New York, NY, USA, 2001. [Google Scholar]
- Peng, H.; Li, X.; Tong, S. Effects of shrub encroachment on biomass and biodiversity in the typical steppe of Inner Mongolia. Acta Ecol. Sin. 2013, 33, 7221–7229. [Google Scholar] [CrossRef]
- Wang, L.; Shen, Y.; Cheng, R.; Xiao, W.; Zeng, L. Nitrogen addition promotes early-stage and inhibits late-stage decomposition of fine roots in Pinus massoniana plantation. Front. Plant Sci. 2022, 13, 1048153. [Google Scholar] [CrossRef] [PubMed]
- Tilman, D.; Lehman, C.L.; Thomson, K.T. Plant diversity and ecosystem productivity: Theoretical considerations. Proc. Natl. Acad. Sci. USA 1997, 94, 1857–1861. [Google Scholar] [CrossRef]
- Ma, W.; Fang, J. The relationship between species richness and productivity in four typical grasslands of northern China. Biodivers. Sci. 2006, 14, 21. [Google Scholar] [CrossRef]
- Hector, A.; Schmid, B.; Beierkuhnlein, C.; Caldeira, M.C.; Dimitrakopoulos, P.G.; Finn, J.A.; Freitas, H.; Giller, P.S.; Good, J.; Harris, R.; et al. Plant Diversity and Productivity Experiments in European Grasslands. Science 1999, 286, 1123–1127. [Google Scholar] [CrossRef]
- Mittelbach, G.G.; Steiner, C.F.; Scheiner, S.M.; Gross, K.L.; Reynolds, H.L.; Waide, R.B.; Willig, M.R.; Dodson, S.I.; Gough, L. What is the observed relationship between species richness and productivity? Ecology 2001, 82, 2381–2396. [Google Scholar] [CrossRef]
- Zhao, L.; Gao, D.; Xiong, B.; Chen, T.; Li, Y.; Li, F. Relationship between the aboveground biomass and species diversity of sandy communities during the process of restoring succession in the Horqin Sandy Land, China. Acta Ecol. Sin. 2017, 37, 4108–4117. [Google Scholar]
- Xu, Y.; Zhang, Y.; Yang, J.; Lu, Z. Influence of tree functional diversity and stand environment on fine root biomass and necromass in four types of evergreen broad-leaved forests. Glob. Ecol. Conserv. 2020, 21, e00832. [Google Scholar] [CrossRef]
- Wei, Y.; Liu, M.; Xie, Y.; Gao, Y.; Liu, M.; Yue, P.; Liu, L. Study on soil organic carbon of plant communities in enclosed area of Xilamuren desert grassland. Res. Soil Water Conserv. 2024, 31, 35–43. [Google Scholar]
- Crepin, J.; Johnson, L.R. Soil Sampling and Methods of Analysis. J. Environ. Qual. 1993, 38, 15–24. [Google Scholar]
- Whittaker, R.H. Evolution and measurement of species diversity. Taxon 1972, 21, 213–251. [Google Scholar] [CrossRef]
- Ma, K.; Liu, C.; Liu, Y. Measurement of biotic community diversity: II β diversity. Biodivers. Sci. 1995, 3, 38–43. [Google Scholar] [CrossRef]
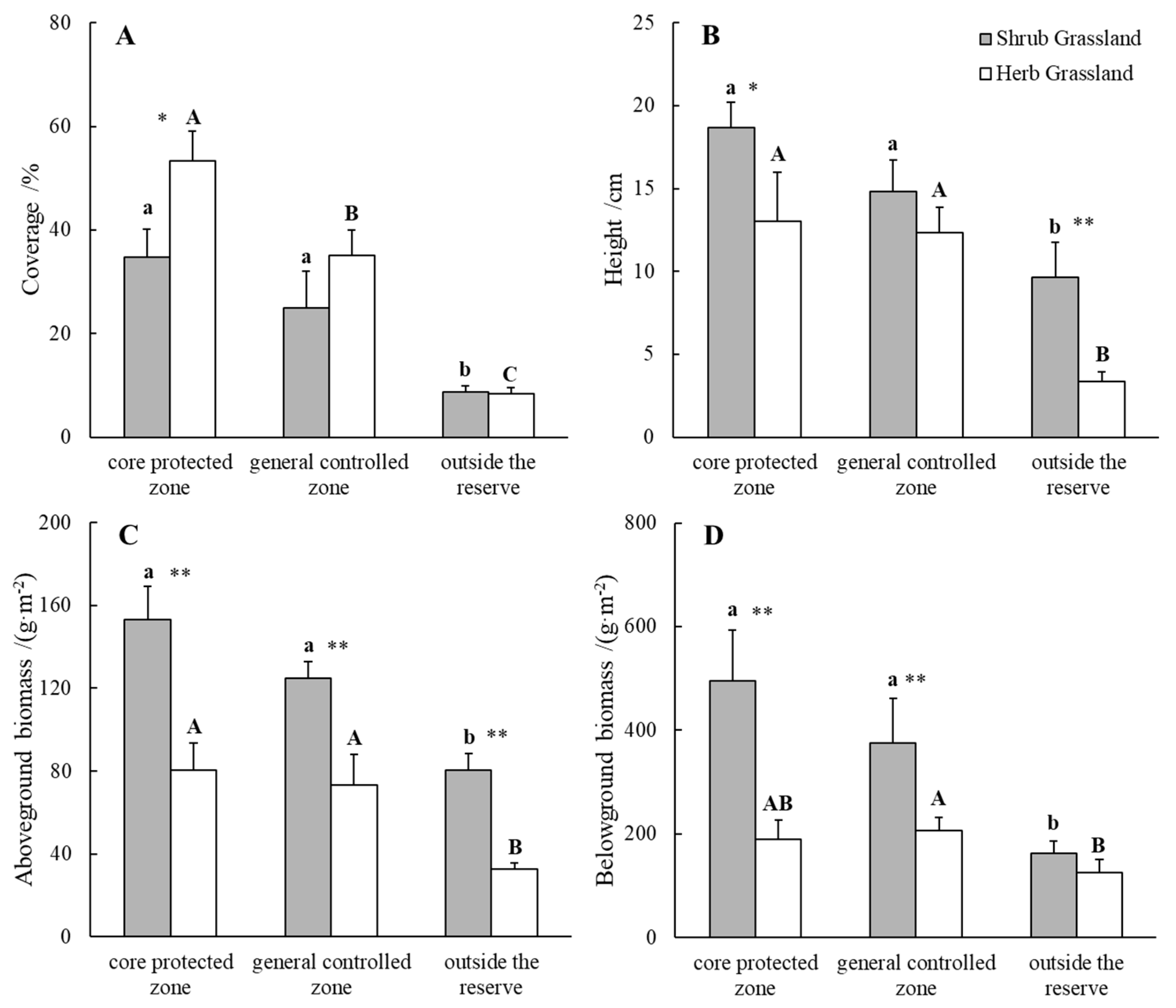
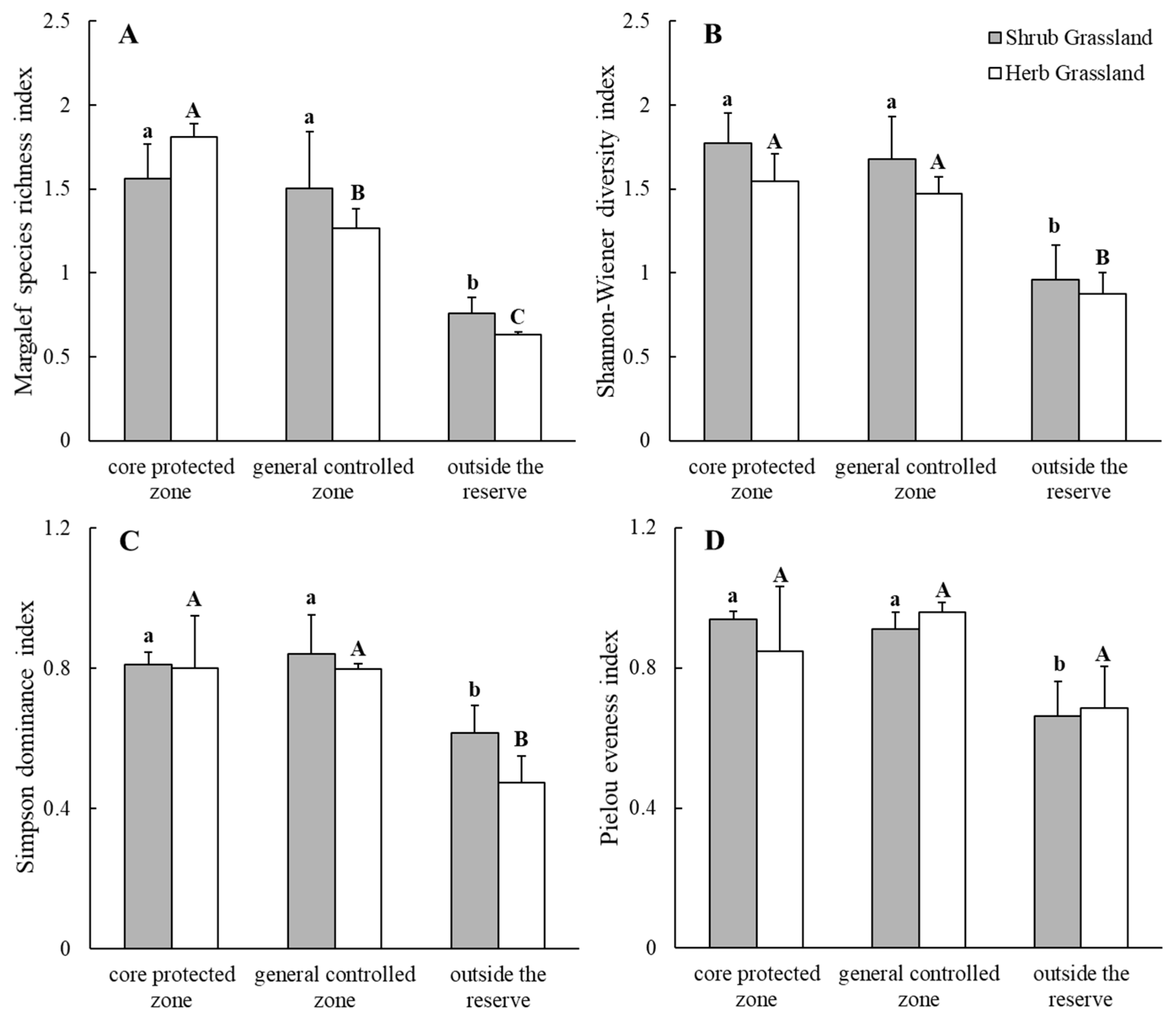
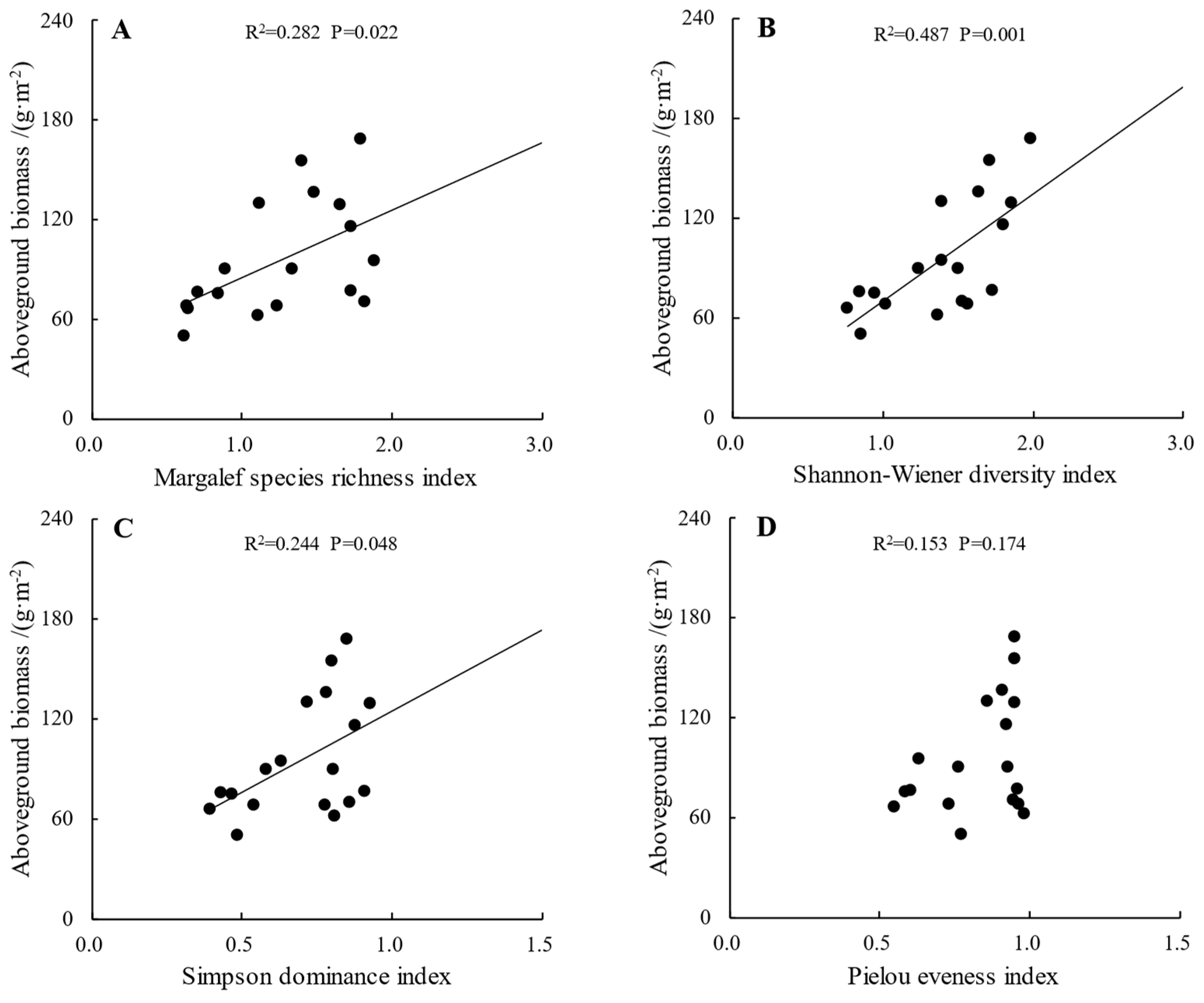
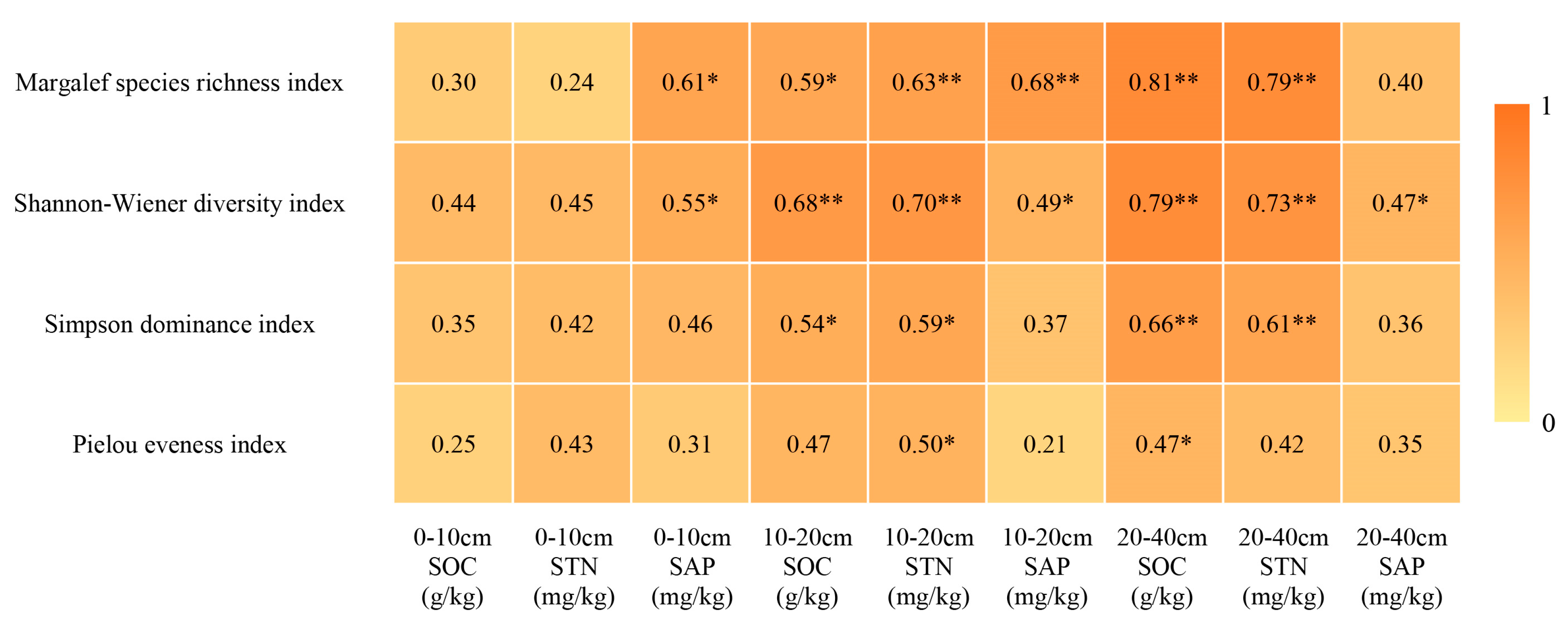
| Plots | Core Protected Zone | General Controlled Zone | Outside the Reserve | ||||
|---|---|---|---|---|---|---|---|
| Vegetation Types | Shrub Grassland | Herb Grassland | Shrub Grassland | Herb Grassland | Shrub Grassland | Herb Grassland | |
| Core protected zone | Shrub Grassland | 1 | 0.333 | 0.231 | 0.167 | 0.385 | 0.308 |
| Herb Grassland | 1 | 0.231 | 0.313 | 0.200 | 0.214 | ||
| General controlled zone | Shrub Grassland | 1 | 0.214 | 0.273 | 0.300 | ||
| Herb Grassland | 1 | 0.267 | 0.125 | ||||
| Outside the reserve | Shrub Grassland | 1 | 0.154 | ||||
| Herb Grassland | 1 | ||||||
| Vegetation Types | Soil Depth | Plots | SOC/g·kg−1 | STN/mg·kg−1 | SAP/mg·kg−1 |
|---|---|---|---|---|---|
| Shrub Grassland | 0–10 cm | Core protected zone | 4.70 ± 0.63 | 0.56 ± 0.11 | 4.79 ± 0.20 a |
| General controlled zone | 4.50 ± 0.62 | 0.48 ± 0.08 | 3.84 ± 0.89 ab | ||
| Outside the reserve | 4.05 ± 1.06 | 0.42 ± 0.07 | 2.52 ± 0.63 b | ||
| 10–20 cm | Core protected zone | 6.83 ± 1.91 a | 0.76 ± 0.21 a | 3.57 ± 0.56 a | |
| General controlled zone | 4.94 ± 0.44 ab | 0.59 ± 0.04 ab | 2.56 ± 0.60 ab | ||
| Outside the reserve | 3.04 ± 0.41 b | 0.38 ± 0.04 b | 1.71 ± 0.07 b | ||
| 20–40 cm | Core protected zone | 4.90 ± 0.51 a | 0.55 ± 0.01 a | 3.36 ± 1.19 | |
| General controlled zone | 4.32 ± 0.50 a | 0.51 ± 0.08 a | 2.09 ± 0.39 | ||
| Outside the reserve | 2.42 ± 0.22 b | 0.29 ± 0.04 b | 1.55 ± 0.19 | ||
| Herb Grassland | 0–10 cm | Core protected zone | 3.08 ± 0.45 | 0.33 ± 0.03 | 4.20 ± 0.90 |
| General controlled zone | 4.00 ± 1.40 | 0.51 ± 0.16 | 2.82 ± 1.11 | ||
| Outside the reserve | 2.84 ± 0.11 | 0.32 ± 0.06 | 3.17 ± 0.75 | ||
| 10–20 cm | Core protected zone | 3.95 ± 0.96 a | 0.52 ± 0.10 a | 3.31 ± 1.17 | |
| General controlled zone | 3.76 ± 0.52 ab | 0.47 ± 0.08 ab | 2.03 ± 0.14 | ||
| Outside the reserve | 2.20 ± 0.48 b | 0.26 ± 0.07 b | 2.05 ± 0.57 | ||
| 20–40 cm | Core protected zone | 4.24 ± 1.05 a | 0.52 ± 0.13 a | 2.03 ± 0.14 | |
| General controlled zone | 2.60 ± 0.26 b | 0.32 ± 0.06 ab | 1.83 ± 0.38 | ||
| Outside the reserve | 1.86 ± 0.26 b | 0.25 ± 0.04 b | 1.75 ± 0.21 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, P.; Lu, Q.; Li, S. Productivity and Species Diversity of Plant Communities Are Higher inside than outside the West Ordos National Nature Reserve, Northern China. Plants 2024, 13, 660. https://doi.org/10.3390/plants13050660
Guo P, Lu Q, Li S. Productivity and Species Diversity of Plant Communities Are Higher inside than outside the West Ordos National Nature Reserve, Northern China. Plants. 2024; 13(5):660. https://doi.org/10.3390/plants13050660
Chicago/Turabian StyleGuo, Pu, Qi Lu, and Shuai Li. 2024. "Productivity and Species Diversity of Plant Communities Are Higher inside than outside the West Ordos National Nature Reserve, Northern China" Plants 13, no. 5: 660. https://doi.org/10.3390/plants13050660
APA StyleGuo, P., Lu, Q., & Li, S. (2024). Productivity and Species Diversity of Plant Communities Are Higher inside than outside the West Ordos National Nature Reserve, Northern China. Plants, 13(5), 660. https://doi.org/10.3390/plants13050660





