Influence of Sucrose and Activated Charcoal on Phytochemistry and Vegetative Growth in Zephyranthes irwiniana (Ravenna) Nic. García (Amaryllidaceae)
Abstract
1. Introduction
2. Results
2.1. Vegetative Growth in a Greenhouse
2.2. Vegetative Growth In Vitro and Influence of Sucrose and Activated Charcoal
2.3. Ethanolic Extract
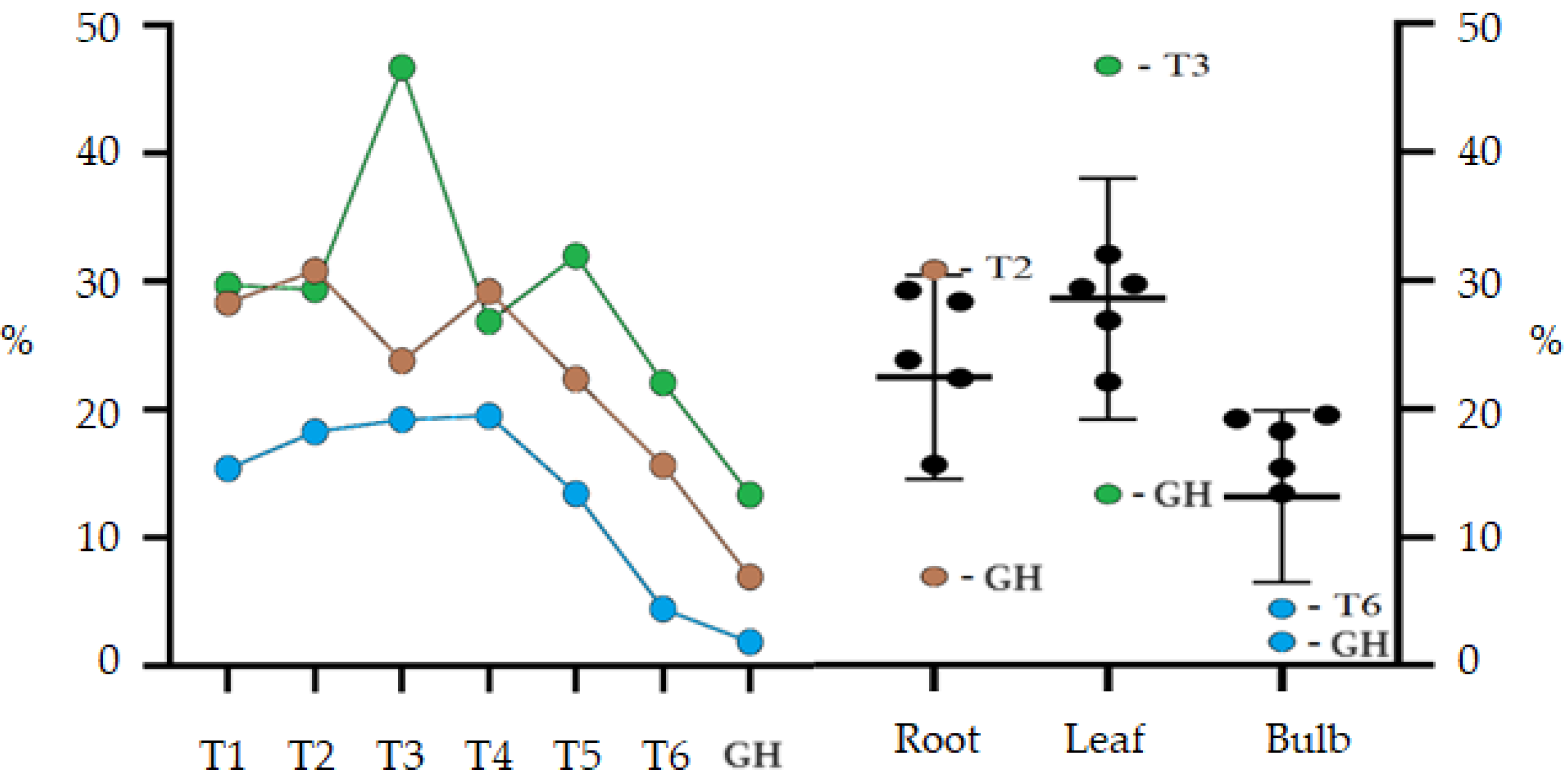
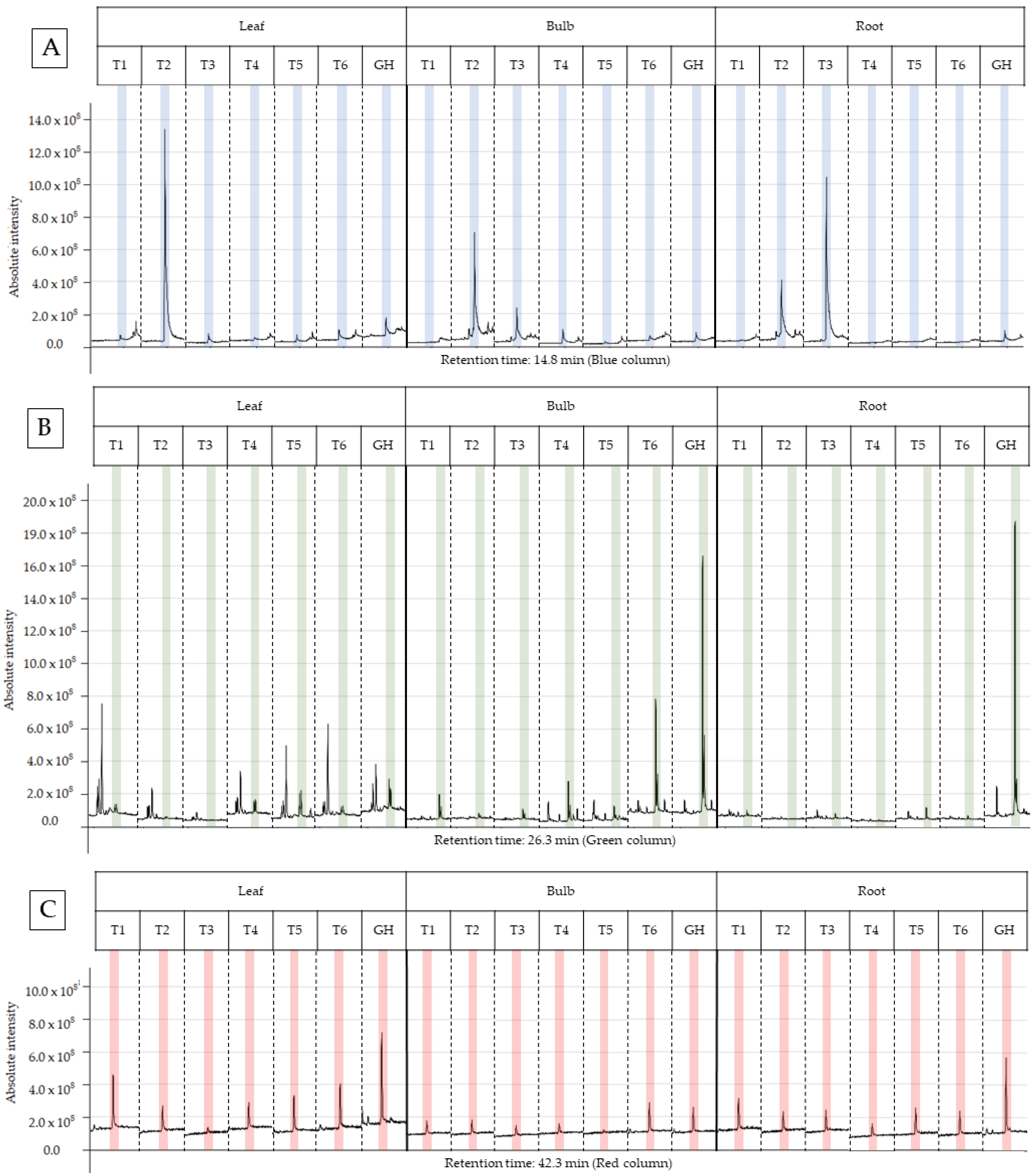
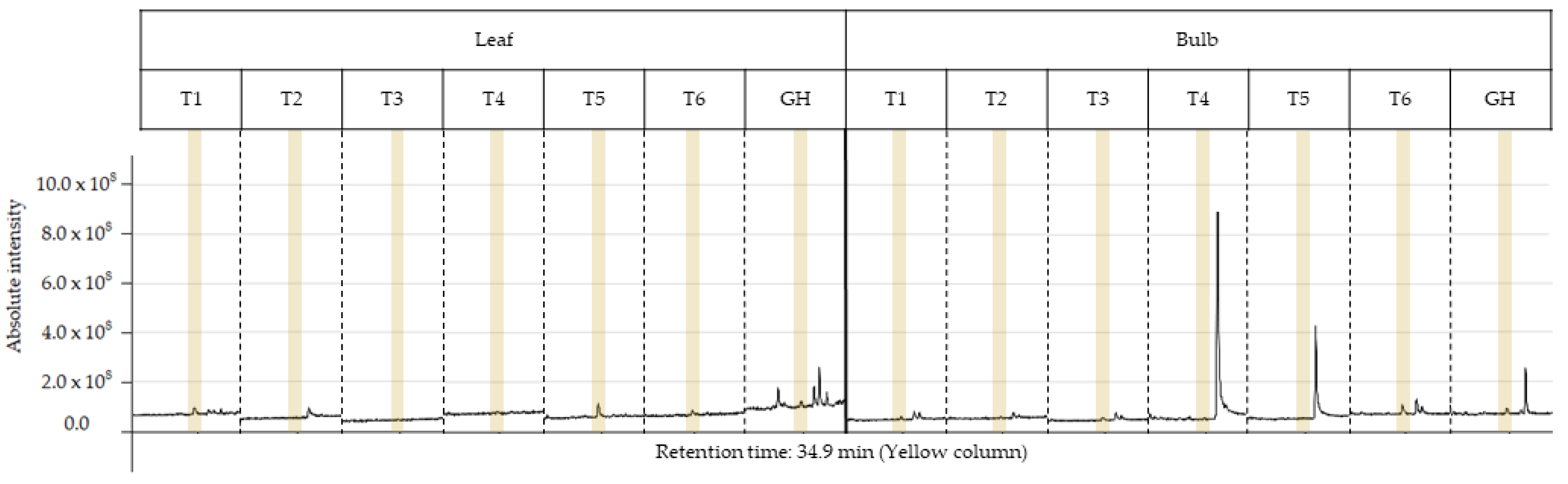
2.4. Phytochemical Profile GC–MS
2.4.1. Alpha-Terpinyl-Acetate
2.4.2. Ethyl Linoleate
2.4.3. Clionasterol
2.4.4. Lycorine
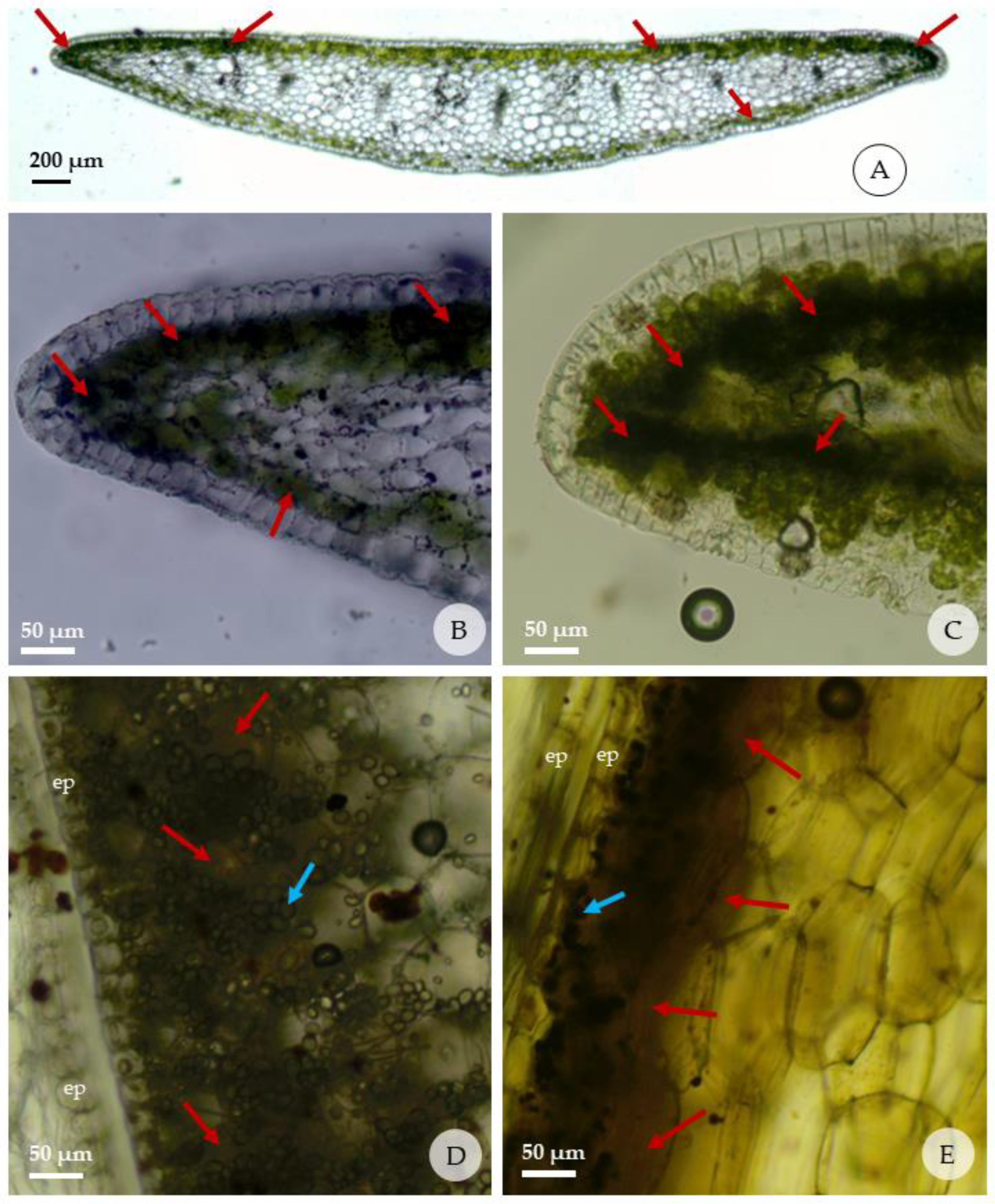
2.5. Histolocalization of Alkaloids in Leaves and Bulblets
3. Discussion
4. Materials and Methods
4.1. Plant Material
4.2. Germination and Cultivation in a Greenhouse
4.3. In Vitro Germination
4.4. Micropropagation of Bulblets
4.5. Seedling Growth Analysis
4.6. Zephyranthes irwiniana Extract
4.7. GC–MS Analysis
4.8. Compounds Identification
4.9. Histolocalization of Alkaloids in the Leaf and Bulb of Zephyranthes irwiniana
4.10. Statistical and Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, C.W.; Okubo, H.; Uemoto, S. Comparison of bulblet formation from twin scales and single scales in Hippeastrum hybridum cultured in vitro. Sci. Hortic. 1990, 42, 151–160. [Google Scholar] [CrossRef]
- Rahimi Khonakdari, M.; Rezadoost, H.; Heydari, R.; Mirjalili, M.H. Effect of photoperiod and plant growth regulators on in vitro mass bulblet proliferation of Narcissus tazzeta L. (Amaryllidaceae), a potential source of galantamine. Plant Cell Tissue Organ Cult. 2020, 142, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Us-Camas, R.; Rivera-Solís, G.; Duarte-Aké, F.; De-la-Peña, C. In vitro culture: An epigenetic challenge for plants. Plant Cell Tissue Organ Cult. 2014, 118, 187–201. [Google Scholar] [CrossRef]
- Ng, Y.P.; Or, T.C.T.; Ip, N.Y. Plant alkaloids as drug leads for Alzheimer’s disease. Neurochem. Int. 2015, 89, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Zaragoza-Puchol, D.; Ortiz, J.E.; Orden, A.A.; Sanchez, M.; Palermo, J.; Tapia, A.; Bastida, J.; Feresin, G.E. Alkaloids analysis of Habranthus cardenasianus (Amaryllidaceae), anti-cholinesterase activity and biomass production by propagation strategies. Molecules 2021, 26, 192–209. [Google Scholar] [CrossRef] [PubMed]
- IUCN. The IUCN Red List of Threatened Species. Version 2022-1. 2022. Available online: https://www.iucnredlist.org (accessed on 7 September 2022).
- BRASIL. Ministério do Meio Ambiente. Gabinete do Ministro. Portaria MMA nº 148, de 7 de junho de 2022. Available online: https://www.in.gov.br/en/web/dou/-/portaria-mma-n-148-de-7-de-junho-de-2022-406272733 (accessed on 7 September 2022).
- Paiva, J.R.; Souza, A.S.Q.; Pereira, R.C.A.; Ribeiro, P.R.V.; Zocolo, G.J.; Brito, E.S.; Pessoa, O.D.L.; Canuto, K.M. Development and validation of a UPLC-ESI-MS method for quantitation of the anti-Alzheimer drug galantamine and other amaryllidaceae alkaloids in plants. J. Braz. Chem. Soc. 2020, 31, 265–272. [Google Scholar] [CrossRef]
- da Costa, G.G.P.; Silva, C.A.G.; Gomes, J.V.D.; Torres, A.G.; Santos, I.R.I.; Almeida, F.T.C.; Fagg, C.W.; Simeoni, L.A.; Silveira, D.; Gomes-Copeland, K.K.P. Influence of in vitro micropropagation on lycorine biosynthesis and anticholinesterase activity in Hippeastrum goianum. Rev. Bras. Farmacogn. 2019, 29, 262–265. [Google Scholar] [CrossRef]
- Ortiz, J.E.; Berkov, S.; Pigni, N.B.; Theoduloz, C.; Roitman, G.; Tapia, A.; Bastida, J.; Feresin, G.E. Wild Argentinian Amaryllidaceae, a new renewable source of the acetylcholinesterase inhibitor galanthamine and other alkaloids. Molecules 2012, 17, 13473–13482. [Google Scholar] [CrossRef]
- Juan-Vicedo, J.; Pavlov, A.; Ríos, S.; Casas, J.L. In vitro culture and micropropagation of the Baetic-Moroccan endemic plant Lapiedra martinezii Lag. (Amaryllidaceae). In Vitro Cell. Dev. Biol. Plant 2019, 55, 725–732. [Google Scholar] [CrossRef]
- Koirala, M.; Karimzadegan, V.; Liyanage, N.S.; Mérindol, N.; Desgagné-Penix, I. Biotechnological Approaches to Optimize the Production of Amaryllidaceae Alkaloids. Biomolecules 2022, 12, 893. [Google Scholar] [CrossRef]
- Ptak, A.; Morańska, E.; Skrzypek, E.; Warchol, M.; Spina, R.; Laurian-Mattar, D.; Simlat, M. Carbohydrates stimulated Amaryllidaceae alkaloids biosynthesis in Leucojum aestivum L. plants cultured in RITA® bioreactor. PeerJ 2020, 8, e8688. [Google Scholar] [CrossRef] [PubMed]
- Thomas, T.D. The role of activated charcoal in plant tissue culture. Biotechnol. Adv. 2008, 26, 618–631. [Google Scholar] [CrossRef] [PubMed]
- Gomez, A.A.; Mercado, M.I.; Belizán, M.M.E.; Ponessa, G.; Vattuone, M.A.; Sampietro, D.A. In situ histochemical localization of alkaloids in leaves and pods of Prosopis ruscifolia. Flora 2019, 256, 1–6. [Google Scholar] [CrossRef]
- Chowdhury, S.; Kumar, S. Alpha-terpinyl acetate: A natural monoterpenoid from Elettaria cardamomum as multi-target directed ligand in Alzheimer’s disease. J. Funct. Foods 2020, 68, 103892. [Google Scholar] [CrossRef]
- Kissling, J.; Ioset, J.; Marston, A.; Hostettmann, K. Bio-guided isolation of cholinesterase inhibitors from the bulbs of Crinum × powellii (Amaryllidaceae). Phytother. Res. 2005, 19, 984–987. [Google Scholar] [CrossRef]
- Aly, S.H.; Eldahshan, O.A.; Al-Rashood, S.T.; Binjubair, F.A.; Hassab, M.A.; Eldehna, W.M.; Dall’Acqua, S.; Zengin, G. Chemical constituents, antioxidant, and enzyme inhibitory activities supported by in-silico study of n-hexane extract and essential oil of guava leaves. Molecules 2022, 27, 8979. [Google Scholar] [CrossRef]
- Mcnulty, J.; Nair, J.J.; Little, J.R.L.; Brennan, J.D.; Bastida, J. Structure–activity studies on acetylcholinesterase inhibition in the lycorine series of Amaryllidaceae alkaloids. Bioorg. Med. Chem. Lett. 2010, 20, 5290–5294. [Google Scholar] [CrossRef]
- Herranz, R.; Copete, M.A.; Herranz, J.M.; Copete, E.; Ferrandis, P. Optimization of plant production by seed treatment in two wild subspecies of Narcissus pseudonarcissus Rich in Alkaloids. Molecules 2020, 25, 4439–4457. [Google Scholar] [CrossRef]
- Rodrigues, A.M.; Carvalho, T.C.; Ayub, R.A. Germinação in vitro de sementes de Hippeastrum hybridum. Braz. J. Appl. Technol. Agric. Sci. 2020, 13, 1–6. [Google Scholar]
- Herranz, J.M.; Copete, M.A.; Ferrandis, P. Environmental regulation of embryo growth, dormancy breaking and germination in Narcissus alcaracensis (Amaryllidaceae), a threatened endemic Iberian daffodil. Am. Midl. Nat. 2013, 169, 147–167. [Google Scholar] [CrossRef]
- Herranz, J.M.; Copete, E.; Ferrandis, P. Non-deep complex morphophysiological dormancy in Narcissus longispathus (Amaryllidaceae): Implications for evolution of dormancy levels within section Pseudonarcissi. Seed Sci. Res. 2013, 23, 141–155. [Google Scholar] [CrossRef]
- Staikidou, I.; Watson, S.; Harvey, B.M.R.; Selby, C. Narcissus bulblet formation in vitro: Effects of carbohydrate type and osmolarity of the culture medium. Plant Cell Tissue Organ Cult. 2005, 80, 313–320. [Google Scholar] [CrossRef]
- Berkov, S.; Georgieva, L.; Kondakova, V.; Atanassov, A.; Viladomat, F.; Bastida, J.; Codina, C. Plant sources of galanthamine: Phytochemical and biotechnological aspects. Biotechnol. Biotechnol. Equip. 2009, 23, 1170–1176. [Google Scholar] [CrossRef]
- Berkov, S.; Pavlov, A.; Georgiev, V.; Bastida, J.; Burrus, M.; Ilieva, M.; Codina, C. Alkaloid synthesis and accumulation in Leucojum aestivum in vitro cultures. Nat. Prod. Commun. 2009, 4, 359–364. [Google Scholar] [CrossRef]
- Sellés, M.; Bergoñón, S.; Viladomat, F.; Bastida, J.; Codina, C. Effect of sucrose on growth and galanthamine production in shoot-clump cultures of Narcissus confusus in liquid-shake medium. Plant Cell Tissue Organ Cult. 1997, 49, 129–136. [Google Scholar] [CrossRef]
- El Tahchy, A.; Bordage, S.; Ptak, A.; Dupire, F.; Barre, E.; Guillou, C.; Henry, M.; Chapleur, Y.; Laurian-Mattar, D. Effects of sucrose and plant growth regulators on acetylcholinesterase inhibitory activity of alkaloids accumulated in shoot cultures of Amaryllidaceae. Plant Cell Tissue Organ Cult. 2011, 106, 381–390. [Google Scholar] [CrossRef]
- Pan, M.J.; Staden, J.V. The use of charcoal in in vitro culture—A review. Plant Growth Regul. 1998, 26, 155–163. [Google Scholar] [CrossRef]
- Baker, F.S.; Miller, C.E.; Repik, A.J.; Tolles, D. Activated carbon. In Kirk-Othmer Encyclopedia Chemical Technology, 1st ed.; Wiley Blackwell: Hoboken, NJ, USA, 2000. [Google Scholar]
- Tallini, L.R.; Bastida, J.; Cortes, N.; Osorio, E.H.; Theoduloz, C.; Schmeda-Hirschmann, G. Cholinesterase inhibition activity, alkaloid profiling and molecular docking of Chilean Rhodophiala (Amaryllidaceae). Molecules 2018, 23, 1532. [Google Scholar] [CrossRef]
- Moreno, R.; Tallini, L.R.; Salazar, C.; Osorio, E.H.; Montero, E.; Bastida, J.; Oleas, N.H.; León, K.A. Chemical profiling and cholinesterase inhibitory activity of five Phaedranassa Herb. (Amaryllidaceae) species from Ecuador. Molecules 2020, 25, 2092. [Google Scholar] [CrossRef]
- Ferdausi, A.; Chang, X.; Hall, A.; Jones, M. Galanthamine production in tissue culture and metabolomic study on Amaryllidaceae alkaloids in Narcissus pseudonarcissus cv. Carlton. Ind. Crops Prod. 2020, 144, 112058. [Google Scholar] [CrossRef]
- Vaičiulyte, V.; Ložienė, K.; Švedienė, J.; Raudonienė, V.; Paškevičius, A. α-Terpinyl acetate: Occurrence in essential oils bearing Thymus pulegioides, phytotoxicity, and antimicrobial effects. Molecules 2021, 26, 1065. [Google Scholar] [CrossRef]
- Kumar, S.; Kashyap, P.; Chowdhury, S.; Kumar, S.; Panwar, A.; Kumar, A. Identification of phytochemicals as potential therapeutic agents that binds to Nsp15 protein target of coronavirus (SARS-CoV-2) that are capable of inhibiting virus replication. Phytomedicine 2021, 85, 153317. [Google Scholar] [CrossRef]
- Michet, A.; Chalchat, J.; Figuérédo, G.; Thébaud, G.; Billy, F.; Pétel, G. Chemotypes in the volatiles of wild thyme (Thymus pulegioides L.). J. Essent. Oil Res. 2008, 20, 101–103. [Google Scholar] [CrossRef]
- Alam, A.; Majumdar, R.S.; Alam, P. Development of HPTLC method for determination of α-terpinyl acetate, and evaluation of antioxidant properties of essential oils in Elettaria cardamomum. Trop. J. Pharm. Res. 2019, 18, 2139–2145. [Google Scholar] [CrossRef]
- Huang, X.; Zhao, Y.; Hou, Z. Purification of ethyl linoleate from foxtail millet (Setaria italica) bran oil via urea complexation and molecular distillation. Foods 2021, 10, 1925. [Google Scholar] [CrossRef] [PubMed]
- Koo, H.J.; Park, H.J.; Byeon, H.E.; Kwak, J.H.; Um, S.H.; Kwon, S.T.; Rhee, D.K.; Pyo, S. Chinese yam extracts containing β-sitosterol and ethyl linoleate protect against atherosclerosis in apolipoprotein E-deficient mice and inhibit muscular expression of VCAM-1 in vitro. J. Food Sci. 2014, 79, H719–H729. [Google Scholar] [CrossRef]
- Park, S.Y.; Seetharaman, R.; Ko, M.J.; Kim, D.Y.; Kim, T.H.; Yoon, M.K.; Kwak, J.H.; Lee, S.J.; Bae, Y.S.; Choi, Y.W. Ethyl linoleate from garlic attenuates lipopolysaccharide-induced pro-inflammatory cytokine production by inducing heme oxygenase-1 in RAW264. 7 cells. Int. Immunopharmacol. 2014, 19, 253–261. [Google Scholar] [CrossRef]
- Kwon, Y.; Haam, C.E.; Byeon, S.; Choi, S.J.; Shin, D.; Choi, S.; Lee, Y. Vasodilatory effect of Phellinus linteus extract in rat mesenteric arteries. Molecules 2020, 25, 3160. [Google Scholar] [CrossRef] [PubMed]
- Shah, M.; Murad, W.; Rehman, N.U.; Mubin, S.; Al-Sababi, J.N.; Ahmad, M.; Zahoor, M.; Ullah, O.; Waqas, M.; Ullah, S.; et al. GC-MS analysis and biomedical therapy of oil from n-hexane fraction of Scutellaria edelbergii Rech. f.: In vitro, in vivo, and in silico approach. Molecules 2021, 26, 7676. [Google Scholar] [CrossRef]
- Sundarraj, S.; Thangam, R.; Sreevani, V.; Kaveri, K.; Gunasekaran, P.; Achiraman, S.; Kannan, S. Gamma-sitosterol from Acacia nilotica L. induces G2/M cell cycle arrest and apoptosis through c-Myc suppression in MCF-7 and A549 cells. J. Ethnopharmacol. 2012, 141, 803–809. [Google Scholar] [CrossRef]
- Balamurugan, R.; Stalin, A.; Ignacimuthus, S. Molecular docking of γ-sitosterol with some targets related to diabetes. Eur. J. Med. Chem. 2012, 47, 38–43. [Google Scholar] [CrossRef] [PubMed]
- Cerqueira, F.; Watanadilok, R.; Sonchaeng, P.; Kijjoa, A.; Pinto, M.; Ufford, H.Q.; Kroes, B.; Beukelman, C.; Nascimento, M.S.J. Clionasterol: A potent inhibitor of complement component C1. Planta Med. 2003, 69, 174–176. [Google Scholar] [CrossRef]
- Shahid, A.; Khan, K.R.; Rao, H.; Aati, H.Y.; Sherif, A.E.; Khan, D.A.; Basit, A.; Umair, M.; Mueed, A.; Esatbeyoglu, T.; et al. Phytochemical profiling of the ethanolic extract of Zaleya pentandra L. Jaffery and its biological activities by in-vitro assays and in-silico molecular docking. Appl. Sci. 2023, 13, 584. [Google Scholar] [CrossRef]
- Khoba, K.; Kumar, S.; Chatterjee, S.; Purty, R.S. Isolation, characterization, and in silico interaction studies of bioactive compounds from Caesalpinia bonducella with target proteins involved in alzheimer’s disease. Appl. Biochem. Biotechnol. 2023, 195, 2216–2234. [Google Scholar] [CrossRef] [PubMed]
- Godara, P.; Dulara, B.K.; Barwer, N.; Chaudhary, N.S. Comparative GC-MS analysis of bioactive phytochemicals from different plant parts and callus of Leptadenia reticulata Wight and Arn. Pharmacogn. J. 2019, 11, 129–140. [Google Scholar] [CrossRef]
- Shehu, A.; Issa, S.B.; Lawal, A.Z.; Egharevba, G.O.; Bale, M.I.; Ojo, J.B.; Salami, L.B.; Taiwo, L.O.; Yusuf, S.A.; Adeyemo, J.; et al. Chemical constituents, antibacterial, antifungal and antioxidant activities from the leaf extracts of Drynaria laurentii. J. Phytomed. Ther. 2022, 20, 690–701. [Google Scholar]
- Marrelli, M.; Russo, C.; Statti, G.; Argentieri, M.P.; Meleleo, D.; Mallamaci, R.; Avato, P.; Conforti, F. Phytochemical and biological characterization of dry outer scales extract from Tropea red onion (Allium cepa L. var. Tropea)—A promising inhibitor of pancreatic lipase. Phytomed. Plus 2022, 2, 100235. [Google Scholar] [CrossRef]
- Roy, M.; Liang, L.; Xiao, X.; Feng, P.; Ye, M.; Liu, J. Lycorine: A prospective natural lead for anticancer drug discovery. Biomed. Pharmacother. 2018, 107, 615–624. [Google Scholar] [CrossRef]
- Tasker, S.Z.; Cowfer, A.E.; Hergenrother, P.J. Preparation of structurally diverse compounds from the natural product lycorine. Org. Lett. 2018, 20, 5894–5898. [Google Scholar] [CrossRef]
- Bores, G.M.; Huger, F.P.; Petko, W.; Mutlib, A.E.; Camacho, F.; Rush, D.K.; Selk, D.E.; Wolf, V.; Kosley, R.W.; Davis, L.; et al. Pharmacological evaluation of novel Alzheimer’s disease therapeutics: Acetylcholinesterase inhibitors related to galanthamine. J. Pharmacol. Exp. Ther. 1996, 277, 728–738. [Google Scholar]
- Elgorashi, E.E.; Malan, S.F.; Stafford, G.I.; Staden, J. Quantitative structure–activity relationship studies on acetylcholinesterase enzyme inhibitory effects of Amaryllidaceae alkaloids. S. Afr. J. Bot. 2006, 72, 224–231. [Google Scholar] [CrossRef]
- Cortes, N.; Posada-Duque, R.A.; Alvarez, R.; Alzate, F.; Berkov, S.; Cardona-Gómez, G.P.; Osorio, E. Neuroprotective activity and acetylcholinesterase inhibition of five Amaryllidaceae species: A comparative study. Life Sci. 2015, 122, 42–50. [Google Scholar] [CrossRef]
- Evidente, A.; Kornienko, A. Anticancer evaluation of structurally diverse Amaryllidaceae alkaloids and their synthetic derivatives. Phytochem. Rev. 2009, 8, 449–459. [Google Scholar] [CrossRef]
- BESSA, C.D.P.B.; Andrade, J.P.; Oliveira, R.S.; Domingos, E.; Santos, H.; Romão, W.; Bastida, J.; Borges, W.S. Identification of alkaloids from Hippeastrum aulicum (Ker Gawl.) Herb. (Amaryllidaceae) using CGC-MS and ambient ionization mass spectrometry (PS-MS and LS-MS). J. Braz. Chem. Soc. 2017, 28, 819–830. [Google Scholar] [CrossRef]
- Berkov, S.; Georgieva, L.; Sidjimova, B.; Nikolova, M.; Stanilova, M.; Bastida, J. In vitro propagation and biosynthesis of Sceletium-type alkaloids in Narcissus pallidulus and Narcissus cv. Hawera. S. Afr. J. Bot. 2021, 136, 190–194. [Google Scholar] [CrossRef]
- Andrade, J.P.; Pigni, N.B.; Torras-Claveria, L.; Berkov, S.; Codina, C.; Viladomat, F.; Bastida, J. Bioactive alkaloid extracts from Narcissus broussonetii: Mass spectral studies. J. Pharm. Biomed. Anal. 2012, 70, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.P.; Guo, Y.; Font-Bardia, M.; Calvet, T.; Dutilh, J.; Viladomat, F.; Codina, C.; Nair, J.J.; Zuanazzi, J.A.S.; Bastida, J. Crinine-type alkaloids from Hippeastrum aulicum and H. calyptratum. Phytochemistry 2014, 103, 188–195. [Google Scholar] [CrossRef]
- Reyes-Chilpa, R.; Berkov, S.; Hernández-Ortega, S.; Jankowski, C.K.; Arseneau, S.; Clotet-Codina, I.; Esté, J.A.; Codina, C.; Viladomat, F.; Bastida, J. Acetylcholinesterase-inhibiting alkaloids from Zephyranthes concolor. Molecules 2011, 16, 9520–9533. [Google Scholar] [CrossRef]
- Katoch, D.; Kumar, S.; Kumar, N.; Singh, B. Simultaneous quantification of Amaryllidaceae alkaloids from Zephyranthes grandiflora by UPLC–DAD/ESI-MS/MS. J. Pharm. Biomed. Anal. 2012, 71, 187–192. [Google Scholar] [CrossRef]
- Kulhánková, A.; Cahlíková, L.; Novák, Z.; Macáková, K.; Kuneš, J.; Opletal, L. Alkaloids from Zephyranthes robusta Baker and their acetylcholinesterase and butyrylcholinesterase inhibitory activity. Chem. Biodivers. 2013, 10, 1120–1127. [Google Scholar] [CrossRef]
- Zhan, G.; Gao, B.; Zhou, J.; Liu, T.; Zheng, G.; Jin, Z.; Yao, G. Structurally diverse alkaloids with nine frameworks from Zephyranthes candida and their acetylcholinesterase inhibitory and anti-inflammatory activities. Phytochemistry 2023, 207, 113564. [Google Scholar] [CrossRef] [PubMed]
- Zhan, G.; Liu, J.; Zhou, J.; Sun, B.; Aisa, H.A.; Yao, G. Amaryllidaceae alkaloids with new framework types from Zephyranthes candida as potent acetylcholinesterase inhibitors. Eur. J. Med. Chem. 2017, 127, 771–780. [Google Scholar] [CrossRef] [PubMed]
- Shitara, N.; Hirasawa, Y.; Hasumi, S.; Sasaki, T.; Matsumoto, M.; Wong, C.P.; Kaneda, T.; Asakawa, Y.; Morita, H. Four new Amaryllidaceae alkaloids from Zephyranthes candida. J. Nat. Med. 2014, 68, 610–614. [Google Scholar] [CrossRef]
- Luo, Z.; Wang, F.; Zhang, J.; Li, X.; Zhang, M.; Hao, X.; Xue, Y.; Li, Y.; Horgen, F.D.; Yao, G.; et al. Cytotoxic alkaloids from the whole plants of Zephyranthes candida. J. Nat. Prod. 2012, 75, 2113–2120. [Google Scholar] [CrossRef]
- Kohelová, E.; Maříková, J.; Korábečný, J.; Hulcová, D.; Kučera, T.; Jun, D.; Chlebek, J.; Jenčo, J.; Šafratová, M.; Hrabinová, M.; et al. Alkaloids of Zephyranthes citrina (Amaryllidaceae) and their implication to Alzheimer’s disease: Isolation, structural elucidation and biological activity. Bioorg. Chem. 2021, 107, 104567. [Google Scholar] [CrossRef] [PubMed]
- Centeno-Betanzos, L.Y.; López-Caamal, A.; Rendon, N.C.; Santiago, M.L.; Osorio, E.; Bastida, J.; Cano-Santana, Z.; Reyes-Chilpa, R.; Tovar-Sánchez, E. Microsatellites, morphological, and alkaloids characterization of Zephyranthes fosteri and Z. alba (Amaryllidaceae): Allopatric populations. Biochem. Syst. Ecol. 2022, 101, 104398. [Google Scholar] [CrossRef]
- Silva, S.M.; Araújo, S.F.; Santos, A.S. Detecção de alcaloides em Crinum americanum (Amaryllidaceae). In Proceedings of the 64º Congresso Nacional de Botânica, Belo Horizonte, Brazil, 19 September 2013. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Svendsen, A.B.; Verpoorte, R. Chromatography of Alkaloids. Part A: Thin-Layer Chromatography, 1st ed.; Elsevier Scientific Publishing Company: Amsterdam, The Netherlands, 1983. [Google Scholar]
- Furr, M.; Mahlberg, P.G. Histochemical analyses of laticifers and glandular trichomes in Cannabis sativa. J. Nat. Prod. 1981, 44, 153–159. [Google Scholar] [CrossRef]
- Pavlov, A.; Berkov, S.; Courot, E.; Gocheva, T.; Tuneva, D.; Pandova, B.; Georgiev, M.; Georgiev, V.; Yanev, S.; Burrus, M.; et al. Galanthamine production by Leucojum aestivum in vitro systems. Process Biochem. 2007, 42, 734–739. [Google Scholar] [CrossRef]

| (gL−1) | Treatment | Leaf nº | Root nº | Leaf le. (cm) | Root le. (cm) | Fresh biom. (g) | Dry biom. (g) | |
|---|---|---|---|---|---|---|---|---|
| Sucrose | 30.0 | T1-T2 | 3.78 a | 5.33 a | 20.25 a | 16.83 a | 0.44 a | 0.05 a |
| 45.0 | T3-T4 | 3.86 a | 7.97 b | 20.33 a | 18.19 a | 0.66 b | 0.06 a | |
| 60.0 | T5-T6 | 4.02 a | 10.89 c | 19.53 a | 17.64 a | 0.83 c | 0.10 b | |
| AC | 0.0 | T1, T3, T5 | 3.63 a | 8.89 a | 18.31 a | 14.95 a | 0.61 a | 0.06 a |
| 1.0 | T2, T4, T6 | 4.15 b | 7.24 b | 21.76 b | 20.16 b | 0.68 a | 0.07 a |
| AC (gL−1) | Sucrose (gL−1) | Treatment | Leaf nº | Root nº | Leaf le. (cm) | Root le. (cm) | Fresh biom. (g) | Dry biom. (g) |
|---|---|---|---|---|---|---|---|---|
| 0.0 | 30.0 | T1 | 3.67 a | 6.11 a | 18.89 a | 13.71 a | 0.47 a | 0.04 a |
| 45.0 | T3 | 3.61 a | 8.33 b | 18.91 a | 16.55 b | 0.64 ab | 0.06 a | |
| 60.0 | T5 | 3.66 a | 12.22 c | 17.13 b | 14.59 ab | 0.71 b | 0.09 b | |
| 1.0 | 30.0 | T2 | 3.89 a | 4.56 a | 21.60 a | 19.96 a | 0.40 a | 0.05 a |
| 45.0 | T4 | 4.11 a | 7.61 b | 21.74 a | 19.83 a | 0.67 b | 0.06 a | |
| 60.0 | T6 | 4.44 a | 9.56 b | 21.93 a | 20.69 a | 0.96 c | 0.11 b |
| AC (gL−1) | Sucrose (gL−1) | Treatment | Leaf nº | Root nº | Leaf le. (cm) | Root le. (cm) | Fresh biom. (g) | Dry biom. (g) |
|---|---|---|---|---|---|---|---|---|
| 0.0 | 30.0 | T1 | 3.67 a | 6.11 a | 18.90 a | 13.71 a | 0.47 a | 0.04 a |
| 1.0 | T2 | 3.89 a | 4.56 a | 21.60 b | 19.96 b | 0.40 a | 0.05 a | |
| 0.0 | 45.0 | T3 | 3.61 a | 8.33 a | 18.91 a | 16.55 a | 0.64 a | 0.06 a |
| 1.0 | T4 | 4.11 b | 7.61 a | 21.75 b | 19.83 b | 0.67 a | 0.06 a | |
| 0.0 | 60.0 | T5 | 3.61 a | 12.22 a | 17.13 a | 14.59 a | 0.71 a | 0.09 a |
| 1.0 | T6 | 4.44 b | 9.56 b | 21.93 b | 20.69 b | 0.96 b | 0.11 a |
| Treat. | %TIC | RT (min) | RI (exp) | RI (Lit) | Compound | Mass Fragmentation (Relative Intensity) |
|---|---|---|---|---|---|---|
| T3 (root) | 40.8 | 14.9 | 1351 | 1350 ± 3 | α-Terpenyl acetate | 121 (100), 93 (62), 43 (47), 136 (39), 67 (28), 68 (22), 107 (20), 91 (17), 79 (16), 77 (15) |
| T1 (leaf) | 2.9 | 15.7 | 1390 | 1396 ± 2 | Ethyl decanoate | 88 (100), 101 (51), 70 (29), 73 (39), 157 (25), 41 (25), 61 (24), 155 (22), 60 (21), 43 (20) |
| T2 (leaf) | 1.7 | 17.3 | 1499 | 1499 ± 8 | Eremophylene | 55 (100), 107 (97), 189 (96), 81 (88), 79 (82), 105 (73), 108 (57), 121 (55), 133 (54), 161 (41) |
| T1 (bulb) | 3.5 | 18.3 | 1590 | 1595 ± 2 | Ethyl decanoate | 88 (100), 101 (38), 73 (26), 70 (23), 41 (21), 43 (20), 55 (18), 69 (14), 57 (13), 160 (12) |
| T1 (leaf) | 4.7 | 21.4 | 1832 | 1837 ± 5 | Neophytadiene | 68 (100), 69 (70), 82 (63), 95 (56), 57 (53), 67 (49), 71 (44), 81 (40), 55 (38), 41 (36) |
| T6 (bulb) | 3.7 | 22.7 | 1922 | 1926 ± 2 | Methyl palmitate | 74 (100), 87 (47), 43 (15), 75 (14), 143 (11), 227 (9), 83 (6), 59 (5), 129 (5), 171 (5) |
| T2 (bulb) | 1.4 | 23.2 | 1956 | 1968 ± 7 | Palmitic acid | 60 (100), 43 (92), 41 (81), 55 (65), 129 (64), 69 (61), 83 (47), 213 (33), 185 (28), 256 (27) |
| T6 (bulb) | 12.3 | 23.7 | 1990 | 1993 ± 3 | Ethyl palmitate | 88 (100), 70 (27), 43 (23), 41 (19), 55 (18), 57 (16), 69 (15), 89 (15), 157 (15), 241 (11) |
| GH (root) | 4.0 | 25.2 | 2090 | 2092 ± 4 | Methyl linoleate | 81 (100), 67 (70), 68 (47), 41 (41), 82 (33), 96 (31), 69 (28), 79 (27), 64 (24), 109 (21) |
| T1 (leaf) | 6.4 | 25.3 | 2095 | 2098 ± 3 | Methyl linolenate | 79 (100), 67 (83), 95 (56), 108 (48), 80 (48), 93 (47), 55 (43), 69 (28), 77 (28), 107 (26) |
| GH (root) | 38.4 | 26.3 | 2156 | 2162 ± 6 | Ethyl linoleate | 67 (100), 81 (94), 95 (57), 82 (48), 55 (48), 79 (43), 41 (42), 68 (42), 69 (35), 109 (32) |
| T5 (leaf) | 7.0 | 26.4 | 2163 | 2171 ± 13 | Ethyl linolenate | 79 (100), 95 (82), 81 (62), 108 (40), 41 (40), 44 (32), 55 (29), 93 (25), 149 (17), 119 (16) |
| T5 (bulb) | 28.6 | 29.3 | 2353 | 2374 ± 25 | Octadecanamide | 59 (100), 72 (79), 41 (31), 55 (30), 69 (24), 43 (22), 83 (15), 67 (14), 81 (13), 126 (12) |
| T5 (leaf) | 2.6 | 34.9 | 2734 | 2747 − N/A | Lycorine | 226 (100), 250 (62), 227 (52), 287 (23), 268 (19), 286 (16), 228 (10), 240 (6), 269 (6), 270 (5) |
| GH (leaf) | 11.6 | 42.3 | 3310 | 3321 ± 31 | Clionasterol | 43 (100), 107 (88), 145 (82), 414 (77), 55 (77), 81 (75), 161 (72), 95 (72), 213 (68), 93 (66) |
| Compound | Tests | Leaves | Bulbs |
|---|---|---|---|
| Alkaloid | Dragendorff | + | + |
| Wagner | ++ | ++ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dewes Neto, B.; Gomes-Copeland, K.K.P.; Silveira, D.; Gomes, S.M.; Craesmeyer, J.M.M.; de Castro Nizio, D.A.; Fagg, C.W. Influence of Sucrose and Activated Charcoal on Phytochemistry and Vegetative Growth in Zephyranthes irwiniana (Ravenna) Nic. García (Amaryllidaceae). Plants 2024, 13, 569. https://doi.org/10.3390/plants13050569
Dewes Neto B, Gomes-Copeland KKP, Silveira D, Gomes SM, Craesmeyer JMM, de Castro Nizio DA, Fagg CW. Influence of Sucrose and Activated Charcoal on Phytochemistry and Vegetative Growth in Zephyranthes irwiniana (Ravenna) Nic. García (Amaryllidaceae). Plants. 2024; 13(5):569. https://doi.org/10.3390/plants13050569
Chicago/Turabian StyleDewes Neto, Bertholdo, Kicia Karinne Pereira Gomes-Copeland, Dâmaris Silveira, Sueli Maria Gomes, Julia Marina Muller Craesmeyer, Daniela Aparecida de Castro Nizio, and Christopher William Fagg. 2024. "Influence of Sucrose and Activated Charcoal on Phytochemistry and Vegetative Growth in Zephyranthes irwiniana (Ravenna) Nic. García (Amaryllidaceae)" Plants 13, no. 5: 569. https://doi.org/10.3390/plants13050569
APA StyleDewes Neto, B., Gomes-Copeland, K. K. P., Silveira, D., Gomes, S. M., Craesmeyer, J. M. M., de Castro Nizio, D. A., & Fagg, C. W. (2024). Influence of Sucrose and Activated Charcoal on Phytochemistry and Vegetative Growth in Zephyranthes irwiniana (Ravenna) Nic. García (Amaryllidaceae). Plants, 13(5), 569. https://doi.org/10.3390/plants13050569







