Gene Functional Networks from Time Expression Profiles: A Constructive Approach Demonstrated in Chili Pepper (Capsicum annuum L.)
Abstract
1. Introduction
2. Methods
2.1. Prerequisites
2.2. Congruent Gene Correlations
2.3. Robustness of a Relation between Genes
2.4. The “Gene2Gene” Algorithm
- The genes of interest, say a set of “g” gene identifiers.
- The genotypes (or accessions) where the estimation will be performed, i.e., a set of “a” genotypes.
- A threshold for the FDR, “f”.
- A threshold for the minimum value of Pearson’s determination coefficient, “”.
- A threshold to eliminate putative regression outliers, “q”.
- The minimum number of genotypes where the gene relation must be found to be reported in the output, “x” ().
2.5. Constructing GFN: A Bottom-Up Approach
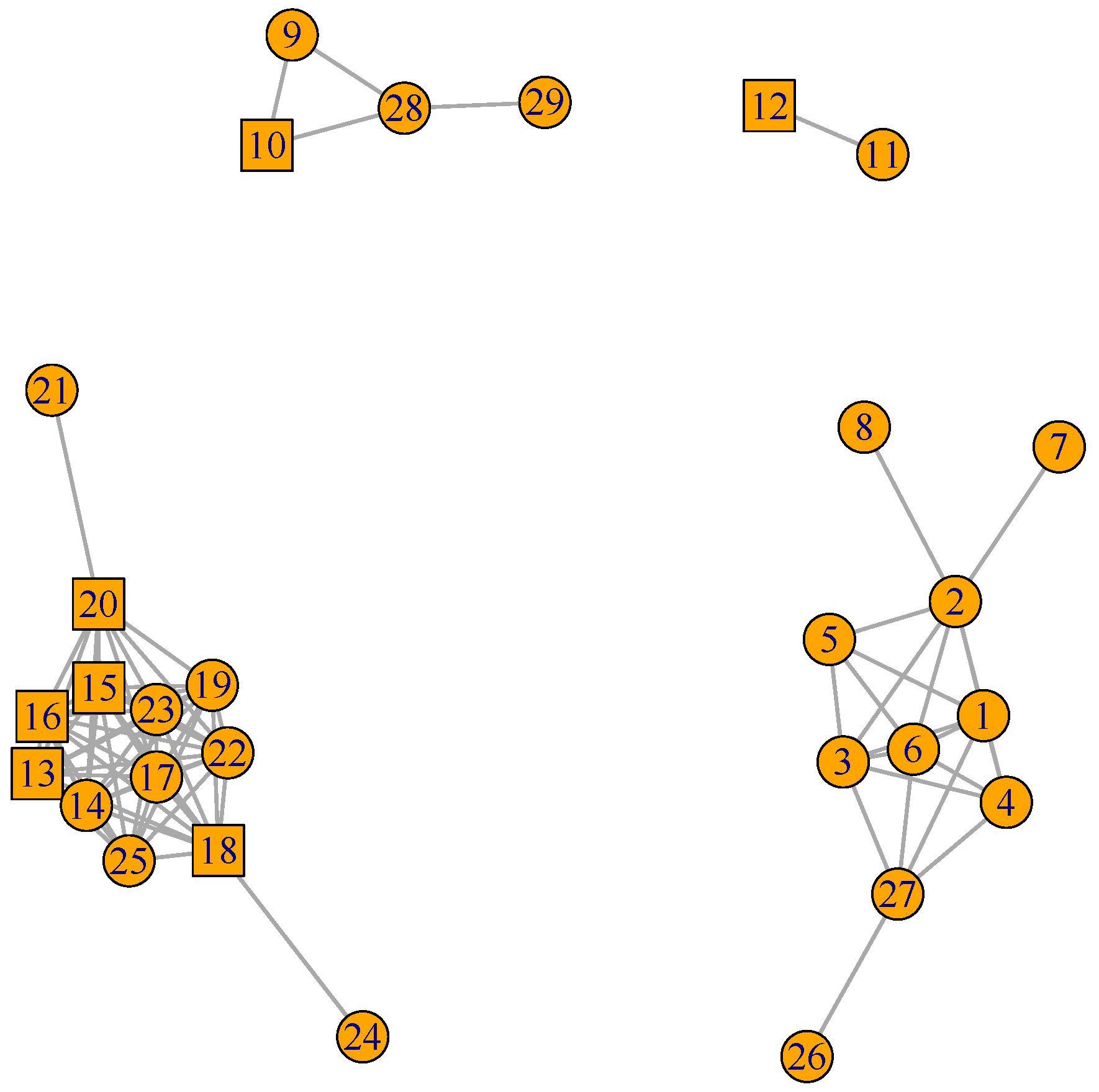
3. Results
3.1. Data Analyses
3.1.1. GFN for Three Biological Processes (BPs)
3.1.2. A Meta-Network
3.2. Finding Transcription Factors for Hub Genes
3.3. Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BP | Biological Process |
| celcy | Cell cycle (BP) |
| D | Domesticated accession (genotype) |
| DAA | Days After Anthesis |
| FDR | False Discovery Rate |
| GCN | Gene Coexpression Network |
| GFN | Gene Functional Network |
| MN | Meta Network |
| rep | Reproduction (BP) |
| SEP | Standardized Expression Profile |
| TF | Transcription Factor |
| W | Wild accession (genotype) |
| vir | Response to virus (BP) |
References
- Reverter, A.; Chan, E.K. Combining partial correlation and an information theory approach to the reversed engineering of gene co-expression networks. Bioinformatics 2008, 24, 2491–2497. [Google Scholar] [CrossRef]
- Pavlopoulos, G.A.; Secrier, M.; Moschopoulos, C.N.; Soldatos, T.G.; Kossida, S.; Aerts, J.; Schneider, R.; Bagos, P.G. Using graph theory to analyze biological networks. BioData Min. 2011, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Allocco, D.J.; Kohane, I.S.; Butte, A.J. Quantifying the relationship between co-expression, co-regulation and gene function. BMC Bioinform. 2004, 5, 18. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Li, L.; Ye, H.; Chen, H.; Shen, W.; Zhong, Y.; Tian, T.; He, H. From Saccharomyces cerevisiae to human: The important gene co-expression modules. Biomed. Rep. 2017, 7, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Kleine, L.; Leal, L.; Lopez, C. Biostatistical approaches for the reconstruction of gene co-expression networks based on transcriptomic data. Briefings Funct. Genom. 2013, 12, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Aoki, K.; Ogata, Y.; Shibata, D. Approaches for extracting practical information from gene co-expression networks in plant biology. Plant Cell Physiol. 2007, 48, 381–390. [Google Scholar] [CrossRef]
- Schaefer, R.J.; Michno, J.M.; Myers, C.L. Unraveling gene function in agricultural species using gene co-expression networks. Biochim. Biophys. Acta (BBA)-Gene Regul. Mech. 2017, 1860, 53–63. [Google Scholar] [CrossRef]
- Xu, Y. Molecular Plant Breeding; CAB International: Wallingford, UK, 2010. [Google Scholar]
- Serrano, M.a.; De Los Rios, P. Interfaces and the edge percolation map of random directed networks. Phys. Rev. E 2007, 76, 056121. [Google Scholar] [CrossRef]
- Hafri, A.; Firestone, C. The perception of relations. Trends Cogn. Sci. 2021, 25, 475–492. [Google Scholar] [CrossRef]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene ontology: Tool for the unification of biology. Nat. Genet. 2000, 25, 25–29. [Google Scholar] [CrossRef]
- Lægreid, A.; Hvidsten, T.R.; Midelfart, H.; Komorowski, J.; Sandvik, A.K. Predicting gene ontology biological process from temporal gene expression patterns. Genome Res. 2003, 13, 965–979. [Google Scholar] [CrossRef]
- Rhee, S.Y.; Wood, V.; Dolinski, K.; Draghici, S. Use and misuse of the gene ontology annotations. Nat. Rev. Genet. 2008, 9, 509. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, E.; Levanon, E.Y. Human housekeeping genes, revisited. Trends Genet. 2013, 29, 569–574. [Google Scholar] [CrossRef] [PubMed]
- Martínez, O.; Arce-Rodríguez, M.L.; Hernandez-Godínez, F.; Escoto-Sandoval, C.; Cervantes-Hernandez, F.; Hayano-Kanashiro, C.; Ordaz-Ortiz, J.J.; Reyes-Valdes, M.H.; Razo-Mendivil, F.G.; Garces-Claver, A.; et al. Transcriptome analyses throughout chili pepper fruit development reveal novel insights into the domestication process. Plants 2021, 10, 585. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Tan, H.; Sun, M.; Han, Y.; Chen, W.; Qiu, S.; Zheng, K.; Wei, G.; Ni, T. Independent component analysis based gene co-expression network inference (ICAnet) to decipher functional modules for better single-cell clustering and batch integration. Nucleic Acids Res. 2021, 49, e54. [Google Scholar] [CrossRef]
- Jimmy, J.L.; Babu, S. Variations in the structure and evolution of Rice WRKY genes in Indica and japonica genotypes and their co-expression network in mediating disease resistance. Evol. Bioinform. 2019, 15, 1176934319857720. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Escoto-Sandoval, C.; Flores-Diaz, A.; Reyes-Valdes, M.H.; Ochoa-Alejo, N.; Martinez, O. A method to analyze time expression profiles demonstrated in a database of chili pepper fruit development. Sci. Rep. 2021, 11, 13181. [Google Scholar] [CrossRef]
- Martinez, O. Time course gene expression experiments. In Transcriptome Profiling: Progress and Prospects; Ali, M.A., Lee, J., Eds.; Elsevier: Amsterdam, The Netherlands, 2022; Chapter 4; pp. 85–110. [Google Scholar]
- Margolin, A.A.; Nemenman, I.; Basso, K.; Wiggins, C.; Stolovitzky, G.; Dalla Favera, R.; Califano, A. ARACNE: An algorithm for the reconstruction of gene regulatory networks in a mammalian cellular context. BMC Bioinform. 2006, 7, S7. [Google Scholar] [CrossRef]
- Izadi, F.; Zarrini, H.N.; Kiani, G.; Jelodar, N.B. A comparative analytical assay of gene regulatory networks inferred using microarray and RNA-seq datasets. Bioinformation 2016, 12, 340. [Google Scholar] [CrossRef]
- Arce-Rodríguez, M.L.; Ochoa-Alejo, N. An R2R3-MYB transcription factor regulates capsaicinoid biosynthesis. Plant Physiol. 2017, 174, 1359–1370. [Google Scholar] [CrossRef] [PubMed]
- Martinez, O.; Escoto-Sandoval, C. Salsa: An R Package of Data Mining Facilities for Capsicum Gene Expression Profiles. Zenodo 2021. [Google Scholar] [CrossRef]
- Ma, P.; Castillo-Davis, C.I.; Zhong, W.; Liu, J.S. A data-driven clustering method for time course gene expression data. Nucleic Acids Res. 2006, 34, 1261–1269. [Google Scholar] [CrossRef] [PubMed]
- Cloonan, N.; Forrest, A.R.; Kolle, G.; Gardiner, B.; Faulkner, G.J.; Brown, M.K.; Taylor, D.F.; Steptoe, A.L.; Wani, S.; Bethel, G.; et al. Stem cell transcriptome profiling via massive-scale mRNA sequencing. Nat. Methods 2008, 5, 613–619. [Google Scholar] [CrossRef]
- Wang, Z.; Gerstein, M.; Snyder, M. RNA-Seq: A revolutionary tool for transcriptomics. Nat. Rev. Genet. 2009, 10, 57–63. [Google Scholar] [CrossRef]
- Casati, P.; Walbot, V. Gene expression profiling in response to ultraviolet radiation in maize genotypes with varying flavonoid content. Plant Physiol. 2003, 132, 1739–1754. [Google Scholar] [CrossRef]
- Gibson, G. The environmental contribution to gene expression profiles. Nat. Rev. Genet. 2008, 9, 575–581. [Google Scholar] [CrossRef]
- Grishkevich, V.; Yanai, I. The genomic determinants of genotype × environment interactions in gene expression. Trends Genet. 2013, 29, 479–487. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Mao, L.; Van Hemert, J.L.; Dash, S.; Dickerson, J.A. Arabidopsis gene co-expression network and its functional modules. BMC Bioinform. 2009, 10, 346. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. Eigengene networks for studying the relationships between co-expression modules. BMC Syst. Biol. 2007, 1, 54. [Google Scholar] [CrossRef]
- Mitra, K.; Carvunis, A.R.; Ramesh, S.K.; Ideker, T. Integrative approaches for finding modular structure in biological networks. Nat. Rev. Genet. 2013, 14, 719–732. [Google Scholar] [CrossRef] [PubMed]
- Zaret, K.S. Pioneer transcription factors initiating gene network changes. Annu. Rev. Genet. 2020, 54, 367–385. [Google Scholar] [CrossRef] [PubMed]
- Arce-Rodriguez, M.L.; Ochoa-Alejo, N. Silencing AT3 gene reduces the expression of pAmt, BCAT, Kas, and Acl genes involved in capsaicinoid biosynthesis in chili pepper fruits. Biol. Plant. 2015, 59, 477–484. [Google Scholar] [CrossRef]
- Arce-Rodríguez, M.L.; Ochoa-Alejo, N. Biochemistry and molecular biology of capsaicinoid biosynthesis: Recent advances and perspectives. Plant Cell Rep. 2019, 38, 1017–1030. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.B.; Gao, Y.; Palsson, B.O.; Lee, S.Y. DeepTFactor: A deep learning-based tool for the prediction of transcription factors. Proc. Natl. Acad. Sci. USA 2021, 118, e2021171118. [Google Scholar] [CrossRef]
- Lugar, D.J.; Mack, S.G.; Sriram, G. NetRed, an algorithm to reduce genome-scale metabolic networks and facilitate the analysis of flux predictions. Metab. Eng. 2021, 65, 207–222. [Google Scholar] [CrossRef]
- Opgen-Rhein, R.; Strimmer, K. From correlation to causation networks: A simple approximate learning algorithm and its application to high-dimensional plant gene expression data. BMC Syst. Biol. 2007, 1, 37. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. WGCNA: An R package for weighted correlation network analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef]
- Amar, D.; Safer, H.; Shamir, R. Dissection of regulatory networks that are altered in disease via differential co-expression. PLoS Comput. Biol. 2013, 9, e1002955. [Google Scholar] [CrossRef]
- Watson, M. CoXpress: Differential co-expression in gene expression data. BMC Bioinform. 2006, 7, 509. [Google Scholar] [CrossRef]
- Tesson, B.M.; Breitling, R.; Jansen, R.C. DiffCoEx: A simple and sensitive method to find differentially coexpressed gene modules. BMC Bioinform. 2010, 11, 497. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Banerjee, B.; Karasik, D.; Frenkel-Morgenstern, M. mRNA-lncRNA Co-Expression Network Analysis Reveals the Role of lncRNAs in Immune Dysfunction during Severe SARS-CoV-2 Infection. Viruses 2021, 13, 402. [Google Scholar] [CrossRef] [PubMed]
- Oliveira de Biagi, C.A., Jr.; Nociti, R.P.; Brotto, D.B.; Funicheli, B.O.; Cassia Ruy, P.d.; Bianchi Ximenez, J.P.; Alves Figueiredo, D.L.; Araujo Silva, W., Jr. CeTF: An R/Bioconductor package for transcription factor co-expression networks using regulatory impact factors (RIF) and partial correlation and information (PCIT) analysis. BMC Genom. 2021, 22, 624. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.; Mendoza, L.; Monzon-Sandoval, J.; Urrutia, A.O.; Gutierrez, H. Emergence of co-expression in gene regulatory networks. PLoS ONE 2021, 16, e0247671. [Google Scholar] [CrossRef]
- Cong, B.; Liu, J.; Tanksley, S.D. Natural alleles at a tomato fruit size quantitative trait locus differ by heterochronic regulatory mutations. Proc. Natl. Acad. Sci. USA 2002, 99, 13606–13611. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Thelen, J.J. Plastid uridine salvage activity is required for photoassimilate allocation and partitioning in arabidopsis. Plant Cell 2011, 23, 2991–3006. [Google Scholar] [CrossRef]
- Csardi, G.; Nepusz, T. The igraph software package for complex network research. InterJ. Complex Syst. 2006, 1695, 1–9. [Google Scholar]
- Escoto-Sandoval, C.; Ochoa-Alejo, N.; Martínez, O. Inheritance of gene expression throughout fruit development in chili pepper. Sci. Rep. 2021, 11, 16. [Google Scholar] [CrossRef]
- Gómez-García, M.d.R.; Ochoa-Alejo, N. Biochemistry and molecular biology of carotenoid biosynthesis in chili peppers (Capsicum spp.). Int. J. Mol. Sci. 2013, 14, 19025–19053. [Google Scholar] [CrossRef]
- Góngora-Castillo, E.; Ibarra-Laclette, E.; Trejo-Saavedra, D.L.; Rivera-Bustamante, R.F. Transcriptome analysis of symptomatic and recovered leaves of geminivirus-infected pepper (Capsicum annuum). Virol. J. 2012, 9, 295. [Google Scholar] [CrossRef]
- Hödl, M.; Basler, K. Transcription in the absence of histone h3. 2 and h3k4 methylation. Curr. Biol. 2012, 22, 2253–2257. [Google Scholar] [CrossRef] [PubMed]
- Jeevalatha, A.; Siddappa, S.; Kumar, A.; Kaundal, P.; Guleria, A.; Sharma, S.; Nagesh, M.; Singh, B.P. An insight into differentially regulated genes in resistant and susceptible genotypes of potato in response to tomato leaf curl new delhi virus-[potato] infection. Virus Res. 2017, 232, 22–33. [Google Scholar] [CrossRef] [PubMed]
- Kwak, K.J.; Kim, J.Y.; Kim, Y.O.; Kang, H. Characterization of transgenic arabidopsis plants overexpressing high mobility group b proteins under high salinity, drought or cold stress. Plant Cell Physiol. 2007, 48, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Lachmann, A.; Giorgi, F.M.; Lopez, G.; Califano, A. Aracne-ap: Gene network reverse engineering through adaptive partitioning inference of mutual information. Bioinformatics 2016, 32, 2233–2235. [Google Scholar] [CrossRef] [PubMed]
- Laurencelle, L. The exact binomial test between two independent proportions: A companion. Quant. Methods Psychol. 2021, 17, 76–79. [Google Scholar] [CrossRef]
- Lee, Y.R.J.; Liu, B. Cytoskeletal motors in arabidopsis. sixty-one kinesins and seventeen myosins. Plant Physiol. 2004, 136, 3877–3883. [Google Scholar] [CrossRef]
- Ohler, L.; Niopek-Witz, S.; Mainguet, S.E.; Möhlmann, T. Pyrimidine salvage: Physiological functions and interaction with chloroplast biogenesis. Plant Physiol. 2019, 180, 1816–1828. [Google Scholar] [CrossRef]
- Petricka, J.J.; Clay, N.K.; Nelson, T.M. Vein patterning screens and the defectively organized tributaries mutants in arabidopsis thaliana. Plant J. 2008, 56, 251–263. [Google Scholar] [CrossRef]
- Redon, C.; Pilch, D.; Rogakou, E.; Sedelnikova, O.; Newrock, K.; Bonner, W. Histone h2a variants h2ax and h2az. Curr. Opin. Genet. Dev. 2002, 12, 162–169. [Google Scholar] [CrossRef]
- Song, Q.; Chen, S.; Wu, Y.; He, Y.; Feng, J.; Yang, Z.; Lin, W.; Zheng, G.; Li, Y.; Chen, H. Comparative transcriptome analyses of gene response to different light conditions of camellia oleifera leaf using illumina and single-molecule real-time-based rna-sequencing. Forests 2020, 11, 91. [Google Scholar] [CrossRef]
- Spies, D.; Ciaudo, C. Dynamics in transcriptomics: Advancements in rna-seq time course and downstream analysis. Comput. Struct. Biotechnol. J. 2015, 13, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Spies, D.; Renz, P.F.; Beyer, T.A.; Ciaudo, C. Comparative analysis of differential gene expression tools for rna sequencing time course data. Briefings Bioinform. 2019, 20, 288–298. [Google Scholar] [CrossRef]
- Strober, B.; Elorbany, R.; Rhodes, K.; Krishnan, N.; Tayeb, K.; Battle, A.; Gilad, Y. Dynamic genetic regulation of gene expression during cellular differentiation. Science 2019, 364, 1287–1290. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Yang, Y.; Moore, M.J.; Brockington, S.F.; Walker, J.F.; Brown, J.W.; Liang, B.; Feng, T.; Edwards, C.; Mikenas, J.; et al. Evolution of portulacineae marked by gene tree conflict and gene family expansion associated with adaptation to harsh environments. Mol. Biol. Evol. 2019, 36, 112–126. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, Z.; Ma, B.; Hou, Q.; Wan, X. Phylogeny and functions of lob domain proteins in plants. Int. J. Mol. Sci. 2020, 21, 2278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liu, J.; Huber, D.J.; Qu, H.; Yun, Z.; Li, T.; Jiang, Y. Transcriptome, degradome and physiological analysis provide new insights into the mechanism of inhibition of litchi fruit senescence by melatonin. Plant Sci. 2021, 308, 110926. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Bai, X.; Jiang, C.; Li, Z. Phosphoproteomic analysis of two contrasting maize inbred lines provides insights into the mechanism of salt-stress tolerance. Int. J. Mol. Sci. 2019, 20, 1886. [Google Scholar] [CrossRef] [PubMed]
- Zoppoli, P.; Morganella, S.; Ceccarelli, M. Timedelay-aracne: Reverse engineering of gene networks from time-course data by an information theoretic approach. BMC Bioinform. 2010, 11, 154. [Google Scholar] [CrossRef]






| BP | In | Out | % Out/In | Con. | ||
|---|---|---|---|---|---|---|
| celcy | 352 | 29 | 8 | 81 | 0.9859 | 0.00729 |
| rep | 228 | 10 | 4 | 29 | 0.9934 | 0.00257 |
| vir | 33 | 4 | 12 | 6 | 0.9936 | 0.00126 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-Díaz, A.; Escoto-Sandoval, C.; Cervantes-Hernández, F.; Ordaz-Ortiz, J.J.; Hayano-Kanashiro, C.; Reyes-Valdés, H.; Garcés-Claver, A.; Ochoa-Alejo, N.; Martínez, O. Gene Functional Networks from Time Expression Profiles: A Constructive Approach Demonstrated in Chili Pepper (Capsicum annuum L.). Plants 2023, 12, 1148. https://doi.org/10.3390/plants12051148
Flores-Díaz A, Escoto-Sandoval C, Cervantes-Hernández F, Ordaz-Ortiz JJ, Hayano-Kanashiro C, Reyes-Valdés H, Garcés-Claver A, Ochoa-Alejo N, Martínez O. Gene Functional Networks from Time Expression Profiles: A Constructive Approach Demonstrated in Chili Pepper (Capsicum annuum L.). Plants. 2023; 12(5):1148. https://doi.org/10.3390/plants12051148
Chicago/Turabian StyleFlores-Díaz, Alan, Christian Escoto-Sandoval, Felipe Cervantes-Hernández, José J. Ordaz-Ortiz, Corina Hayano-Kanashiro, Humberto Reyes-Valdés, Ana Garcés-Claver, Neftalí Ochoa-Alejo, and Octavio Martínez. 2023. "Gene Functional Networks from Time Expression Profiles: A Constructive Approach Demonstrated in Chili Pepper (Capsicum annuum L.)" Plants 12, no. 5: 1148. https://doi.org/10.3390/plants12051148
APA StyleFlores-Díaz, A., Escoto-Sandoval, C., Cervantes-Hernández, F., Ordaz-Ortiz, J. J., Hayano-Kanashiro, C., Reyes-Valdés, H., Garcés-Claver, A., Ochoa-Alejo, N., & Martínez, O. (2023). Gene Functional Networks from Time Expression Profiles: A Constructive Approach Demonstrated in Chili Pepper (Capsicum annuum L.). Plants, 12(5), 1148. https://doi.org/10.3390/plants12051148








