Orchid Micropropagation Using Conventional Semi-Solid and Temporary Immersion Systems: A Review
Abstract
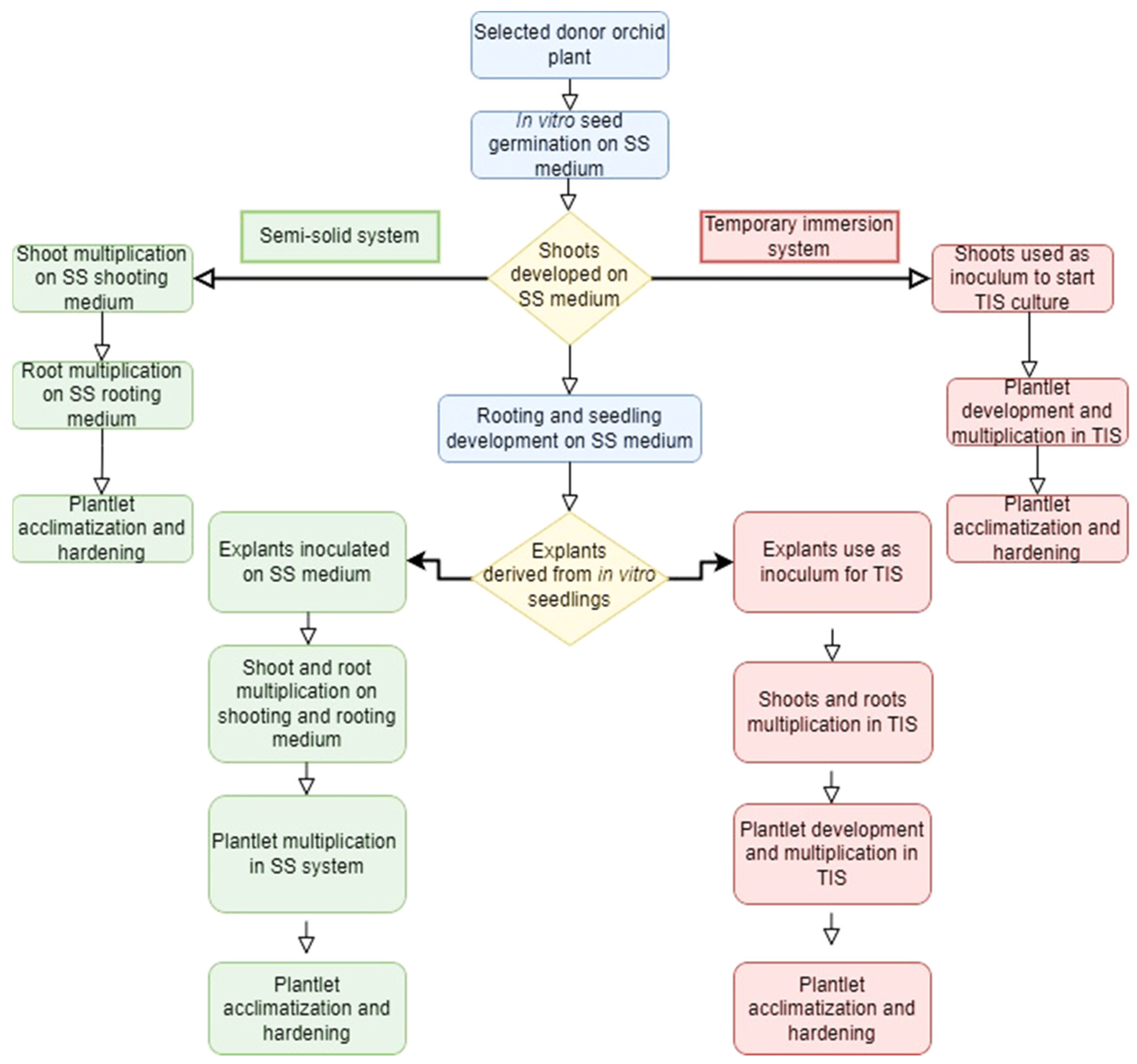
1. Introduction
2. Orchid Micropropagation Using Semi-Solid Media
2.1. Micropropagation Employing Different Explants
2.2. Limitations of SS and Liquid Culture
| Name of the Species | Different Explants Used | Culture Conditions and Growth Hormone Combinations and Concentrations Generating Optimum Culture Response | References |
|---|---|---|---|
| Anacamptis pyramidalis (L.) Rich. and Gymnadenia conopsea (L.) R. Br. | Seeds | Malmgren (MM) medium in complete darkness accelerated in vitro seed germination in both the orchids. MM + CW produced the highest germination (69–88%) in A. pyramidalis, but the maximum morphometric values of height (1.10 mm), width (1.00 mm), and bud height (0.57 mm) were obtained on MM + PE (peptone). MM + 0.3 mg L−1 2iP gave the best plant development response in G. conopsea (plant height—5.33 mm; root number—1.13; root length—12.26 mm). | [54] |
| Aerides multiflora Roxb. | Nodal and leaf segments from in vitro developed seedlings | Shoot bud number from nodal explant was maximum (8.83 ± 0.45/segment) in MS + 1.0 mg L−1 NAA + 2.0 mg L−1 BAP. Longest shoot bud was obtained in MS + 1.0 mg L−1 NAA + 1.0 mg L−1 BAP. Root proliferation and development were superior in MS + 1.0 mg L−1 IBA. PLBs formation from the leaf explant was best in MS + 1.0 mg L−1 IAA + 2.0 mg L−1 BAP. The longest length (4.17 ± 0.13 cm) of individual shoot buds/PLBs after 30 days of culture was recorded in PM + 2% (w/v) sucrose + 0.5 mg L−1 NAA + 1.0 mg L−1 BAP. | [103] |
| Brassavola nodosa (L.) Lindl. | In vitro shoot tips (0.3 to 0.5 cm) | Out of the 6 hormonal treatments used for shoot multiplication, the third treatment (T3: 2.0 mg L−1 BA and 30.0 mg adenine sulphate) generated the highest shoot number per explant, while the survival rate witnessed for 6 treatments was almost similar. After transferring to the rooting medium, the plantlets showed maximum root formation on 0.5 mg L−1 NAA supplemented medium. | [104] |
| Catasetum integerrimum Hook | Seeds | MS + 2.5 mg L−1 BAP + 5.0 mg L−1 IAA produced the highest shoots per explant (5.73 ± 0.45) and leaves per shoot (5.84 ± 0.48). MS + 2.5 mg L−1 IAA generated the best rooting response (11.20 ± 0.28 roots; 13.20 ± 0.28 cm root length). When plant parts (leaves, roots, and pseudobulbs) from in vitro seedlings were employed as explants, best leaf (5.50 ± 0.18) and root formation (4.37 ± 0.37) were achieved with pseudobulb explants. | [105] |
| Cattleya gaskelliana and C. warscewiczii | Seeds | Seeds after sterilization (chlorine 0.5%, chlorine 1%, distilled water, and sucrose) were checked for viability with 2 concentrations of tetrazolium (0.25% and 0.5%) and duration exposure of 24 h and 48 h. Seed viability was 90.6% for C. gaskelliana in 0.5% tetrazolium, while it was 90% for C. warscewiczii in 0.25% tetrazolium with exposure of 48 h in both treatments. The best seedling growth for C. gaskelliana and C. warscewiczii was witnessed in MS + CW and MS + P (pineapple juice), respectively, after 18 weeks of culture. | [106] |
| Cattleya warneri T. Moore | Seeds | Seeds germinated successfully on ½ MS + microalgal biomass or its aqueous extract (0.25, 0.5, 1.0, and 2.0 g L−1). Seed germination enhanced with the development of chlorophyllous protocorms at 4 weeks after the supplementation of biomass or extract (0.25 g L−1). The seedling development was high (greater than 95%) in all treatments with biomass and microalgal extracts (0.25 or 0.5 g L−1) after 24 weeks of culture. MS + 2.0 g L−1 AC produced elongated shoots and roots. | [107] |
| Coelogyne ovalis Lindl. | Nodal bud | PLB formation was maximum (80%) in KC + 10 µM meta-Topolin + 0.5 µM NAA. Medium augmented with 10 µM IAA was the most suitable for rooting. | [24] |
| Crepidium acuminatum (D.Don) Szlach. | Floral buds | M + 1 mg L−1 IAA + 1 mg L−1 KN + 2% Sucrose + 2 g L−1 AC produced the highest shoot bud regeneration with 8 to 10 pseudobulbous shoots per floral bud. | [108] |
| Cypripedium subtropicum | Seeds | Seed germination accelerated on medium with 2iP or BA though the higher concentration of BA (4 and 8 μM) reduced seed germination. Medium with 2ip produced the highest surviving rate for protocorms compared with those with KN or BA. The highest seedlings developed after 4 months on Norstog medium fortified with 1 mg L−1 malic acid, 20 g L−1 sucrose, and 20 g L−1 potato homogenate and solidified with 7 g L−1 agar. | [38] |
| Cymbidium eburneum Lindley. | Leaf segments of in vitro grown plants | M + 0.5 mg L−1 BAP + 2.0 mg L−1 NAA produced a plant regeneration rate (83.3%) in 5.25 weeks generating 15.7 plantlets/explant after 30 WOC. M + 2 mg L−1 BAP + 2 mg L−1 NAA promoted PLB-mediated regeneration in 66.6% of the explants within 6.32 weeks. | [109] |
| Dendrobium anosmum Lindl. | Seeds | High protocorm formation (100%) was observed in all the concentrations of BAP or KN, alone or in combination with NAA, after 10 WOC. MS + 1.0 mg L−1 KN + 0.5 mg L−1 NAA + 30 g sucrose + 8.0 g L−1 agar was suitable for shoot length growth. The best rooting response (100%) was recorded in MS + 1 mg L−1 KN + 0.2, 0.3, or 0.5 mg L−1 NAA. | [110] |
| Dendrobium chryseum Rolfe. | Seed derived protocorms | ½ MS + 2.0 mg L−1 KN + 10% CW produced highest shoot multiplication (18.75 ± 0.48 shoots/culture). MS + 1.0 mg L−1 GA3 + 10% CW yielded the longest shoots (2.0 ± 0.20 cm) and greatest shoot number (4.5 ± 0.65) per culture. Root growth and multiplication were best noticed on ½ MS +1.5 mg L−1 IAA. | [111] |
| Dendrobium crepidatum Lindley & Paxton | Seeds | The highest protocorm formation (41 ± 0.76% for the late capsule; 36.33 ± 0.96% for the early capsule) was witnessed in ½ MS medium. Maximum plant growth and development were demonstrated when the germinated seeds were transferred to ½ MS + 2 µg mL−1 BAP + 1 µg mL−1 NAA. | [112] |
| Dendrobium densiflorum Lindl. | Seeds | ½ MS +10% CW produced the highest seed germination. Root production from protocorms was maximum in MS +15% CW, while the greatest number of roots was noticed in MS +1.5 mg L−1 IBA. | [113] |
| Dendrobium heterocarpum Wall. ex Lindl. | Immature embryos | 95% germination of embryos was observed on MS + sucrose (3%, w/v) + 3 mM L−1 kinetin. Shoot, root, pseudobulb length, and leaf and root number were found maximum on MS + 3 mM L−1 KN + 12 mM L−1 NAA + sucrose (3%). | [49] |
| Dendrobium ovatum (Willd.) Kraenzl | Seeds | Seeds cultured on ½ MS +1 mg L−1 zeatin + 2% sucrose produced protocorms and PLBs after successful germination. ½ MS + 2 mg L−1 BAP was employed to grow the protocorms into plantlets. ½ MS +1 mg L−1 2,4-D + 0.5 mg L−1 6-BAP + 0.5 mg L−1 zeatin generated callus. Plantlets developed proper roots and shoots when transferred to ½ MS +1 mg L−1 zeatin + 2% sucrose. | [114] |
| Dendrobium ovatum (Willd.) Kraenzl | PLBs from in vitro germinated seeds | MS + 1.0 mg L−1 TDZ + 0.5 mg L−1 NAA produced maximum induction of embryogenic callus (EC) (58.6%) and somatic embryos (SEs) (39.8/explant). The explants in the upright orientation gave a greater percentage of EC and higher SEs/explants (EC—58.6% and SEs—39.8/explants) compared to explants with inverted orientation, irrespective of growth hormone combinations. | [115] |
| Dendrobium palpebrae Rchb. f | In vitro derived pseudobulbs | Through organogenesis, multiple shoot buds were developed from both the upper and lower parts of the pseudobulb. MS + 1.0 mg L−1 NAA + 2.0 mg L−1 BAP yielded the maximum shoot buds (8.21 ± 0.44) per segment in the lower part and the highest shoot buds (6.43 ± 0.40) per segment in the upper part of the pseudobulb. The longest root (4.82 ± 0.22 cm) and the greatest root number (2.75 ± 0.17) per shoot bud were recorded on MS + 0.5 mg L−1 NAA. | [116] |
| Dendrobium Yuki White | Apical shoot segment | MS + 0.5 mg L−1 BA + 0.1 mg L−1 NAA + 40 mg L−1 adenine sulphate produced maximum shoots (12) and root number (17) per explant within 8 weeks of culture. The in vitro generated plants were acclimatized with 97% survival rate in charcoal blocks for 6 weeks, followed by plant transfer in potting mixture with coconut fiber and charcoal (1:1). | [117] |
| Doritis pulcherrima Lindl. | In vitro derived protocorms | New Dogashima Media (NDM) was better than VW and MS media giving improved protocorm survival rate (46.70 ± 0.51%), number (11.00 ± 2.94 PLBs per protocorm), and size (6.35 ± 4.31 mm). NDM + 0.1 mg L−1 NAA + 0.1 mg L−1 BA produced maximum shoot, leaf, and root number and length. | [118] |
| Dryadella zebrina (Porsch) Luer | Seeds | The cultures with different BAP treatments showed a mean survival rate greater than 97%. However, BAP concentration higher than 9 μM significantly reduced plant survival. MS fortified with 6 μM BAP generated the highest shoot formation, while MS with 12 or 15 μM BAP yielded less shoots indicating its deleterious effect on shoot development at an elevated level. | [119] |
| Encyclia cordigera (Kunth) Dressler | Seeds | ½ MS + AC (0.15%) produced the best germination response (100%), while ¼ MS + AC (0.15%) generated seedlings with the longest height (1.53 cm). MS + AC (0.15%) gave the maximum root number (2) and root length (2 cm). | [50] |
| Epidendrum denticulatum Barb. Rod | Seeds | In vitro seed-derived plants were subjected to different LED types with blue/red (B/R) combinations for 90 days. White (W) light influenced the production of higher fresh and dry mass, while blue (B) light gave higher anthocyanins value under in vitro conditions. The total chlorophyll values were higher under B/R Light, and B and B/R wavelengths brought higher Fv/Fm values. | [120] |
| Epidendrum fulgens Brongn. | Different explants (protocorm bases, leaf, and root tips) derived from in vitro seed-derived plantlets. | The PLB induction was higher in MS +10 μM TDZ. PLB number increased with TDZ concentration higher than 15 μM, but induction frequency reduced at TDZ concentration greater than 10 μM. The most responsive explants for the highest PLB induction (90%) were protocorm bases with plants regenerated only in a single subculture to hormone-free medium in a shorter time (12 weeks) compared to other leaves (24 weeks) and root tips (60 weeks) explants. | [33] |
| Eulophia dabia (D. Don) | Axenic rhizome segments | ½ MS showed 5% seed germination efficiency. MS medium with casein hydrolysate and AC produced the best rhizome growth from rhizome-like bodies. Maximum shoot induction (96.1%) response was witnessed on MS + 4.4 μM BAP +AC using axenic rhizome with maximum shoots (4.3) and shoot length (13.4 cm). | [121] |
| Gastrochilus matsuran (Makino) | Seeds | ½ MS (without vitamins) + 1 µM NAA + 1.5 µM GA3 + 0.2% peptone + AC (0.05%) + 1% banana pulp + 3% sucrose + 0.8% agar gave the highest seed germination (93.3%). MS + 2 µM TDZ produced the best secondary protocorm formation. MS + 2 µM IBA or 1 µM NAA showed maximum conversion of protocorms into seedlings. | [70] |
| Laelia anceps ssp. anceps | Seeds | MS +2 mg L−1 BAP + 2 mg L−1 IAA + 2 mg L −1 NAA gave the highest seed germination rate (82.20%), with seedlings exhibiting the highest leaf number (1.64) and length (1.11 cm) per explant. MS +1 mg L−1 IAA + 150 mL L−1 CW produced the best rooting percentage (78.20%). | [122] |
| Ludisia discolor | Nodal | Explants treated with 0.40% HgCl2 produced the best survival (63.1%) and growth (22.5%) rate of the culture. ½ MS + 1.0 mg L−1 NAA + 0.1 mg L−1 TDZ + AC (0.2%) + 8% banana cultivar homogenate + 3% sucrose + 3.5 g L−1 generated maximum survival (42%) and plant growth rate (19.6%). | [34] |
| Malaxis acuminata D. Don | Transverse thin cell layer segments (1–4 mm) excised from the pseudobulb | Maximum shoot proliferation (21 micro-shoots/explant) was found in MS + 1.5 mg L−1 meta-Topolin (mT) + 5 mg L−1 chitosan. MS +1.5 mg L−1 IBA + 5 mg L−1 phloroglucinol (PG) produced the best rooting response (root number—7.22 ± 0.45; root length—3.62 ± 0.28 cm). | [9] |
| Mokara Sayan X Ascocenda Wangsa gold | Leaf sections | MS + 3 mg L−1 TDZ induced maximum PLBs (34 PLBs cm−2 leaf section), induction frequency (82.8%), and highest growth rate (93.7 mg day−1). The protocorms were best encapsulated at 3% sodium alginate and 75 mM calcium chloride. Furthermore, 71.2% germination frequency displayed by synthetic seeds stored at 25 °C even after 180 days, while those stored at 4 °C degenerated completely. | [123] |
| Orchis militaris L. | Seeds | mM (Malmgren modified terrestrial orchid medium) + CW (5%) + birch sap (5%) + AC (0.1%) produced the highest seed germination (82.6%). The seedling formation was through protocorm development without callus formation in all 3 modified culture media (Harvais, KC and Malmgren). Modified Harvais 2 medium was suitable for protocorm proliferation in darkness, while KC incorporated with AC was appropriate for further culture development leading to seedling formation. | [48] |
| Orchis simia Lam | Seeds | mM (modified Malmgren medium) + pineapple juice (PJ) + casein hydrolysate (CH) gave the highest seed germination (94.51 ± 0.96%). mM + CW + AV yielded the quickest seed germination (6.8 ± 0.20 days), while mM + PJ with either AV (Aminoven) or CH made larger and higher-weight protocorms. Medium with PJ + AV generated the longest plantlet (4.2 ± 0.04 cm), shoot lengths (1.96 ± 0.042 cm), and heaviest weight (0.58 ± 0.002 g), while the maximum root formation was witnessed in medium with CW and AV (5.2 ± 0.20). | [124] |
| Paphiopedilum SCBG Huihuang 90 (P. SCBG Prince × P. SCBG Miracle) | Seeds | Seeds germinated on Hyponex No. 26 medium + 0.5 g L−1 AC + 1.0 mg L−1 NAA. The protocorms produced meristem mass after transferring them to ½ MS + 0.05 mg L−1 2,4-D. Higher level of IAA and jasmonic acid (JA) promoted PLBs differentiation, while lowering GA3 concentration was essential for shoot apical meristem (SAM) development. | [37] |
| Paphiopedilum insigne | Seeds | The production of protocorm was high in ¼ MS +1 mg·dm−3 BAP + 2 mg·dm−3 TDZ (73%) and 5 mg·dm−3 KN + 1 mg·dm−3 BAP (67%). Both combinations revealed 99% leaf formation from protocorms. The fresh weight of regenerants was high (9.07 mg) in 5 mg·dm−3 KN +1 mg·dm−3 BAP. | [36] |
| Paphiopedilum niveum Rchb.f. | Seeds | The highest percentage (68.33%) of somatic embryo formation was noticed in modified VC+ 0.1 mg L−1 NAA, with the production of the maximum number of somatic embryos (5.19 ± 0.67 per explant). High fresh weight accumulated (183.33 mg) in modified VW without NAA and KN. | [69] |
| Phalaenopsis amabilis (L.) Blume | Shoot tips | ½ MS + 3% (w/v) sucrose + 0.1 g L−1 myoinositol, 2 mg L−1 thidiazuron + 1 mg L−1 BAP was employed to initiate culture with shoot-tip explants. Shoot multiplication was observed better under the blue + green light irradiation, but biomass accumulation was higher with white LEDs. The best shoot branching and multiplication were noticed with higher KN content, total cytokinins, and GA3 under blue + green lights. | [82] |
| Phalaenopsis amboinensis J. J. Sm | Seeds | Best seed germination (90.7%) and protocorm development (51.4%) were witnessed on the VW medium. Leaf, root, and plantlet development was superior in medium augmented with 15% CW + 10 g L−1 banana homogenate (BH). | [125] |
| Phalaenopsis pulcherrima (Lindl.) J. J. Sm | Leaf segments | VW + CW (2%) +100 g L−1 potato + sucrose (2%) +AC (0.2%) + 50 g L−1 banana extract + 3 mg L−1 thidiazuron, and ½ MS + 0.5 mg L−1 niacin + 0.1 mg L−1 thiamine–HCl + 0.5 mg L−1 pyridoxine–HCl + 100 mg L−1 myo-inositol + 2 mg L−1 glycine + banana extract (2%) + 3 mg L−1 thidiazuron was used for culture initiation using leaf explants. The maximum PLBs and highest PLB induction were observed under R (red): B (blue) LEDs on both MS and VW media. Shoot elongation, shoot number, and chlorophyll a and b content were promoted in response to R: B LEDs. | [126] |
| Rhynchostylis retusa (L.) Blume | Root tips | Root explants with intact tips and root caps with distal ends displayed good growth irrespective of the chemical regime. M + 3 mg L−1 KN + 1 mg L−1 NAA + 2% sucrose showed maximum culture regeneration (31%) in the proximal region of the root segment giving 28 plantlets in 15 weeks. | [89] |
| Rhynchostylis retusa (L.) Blume | Immature capsules | ½ MS and ¼ MS demonstrated the earliest seed germination and protocorm development. MS + 10% CW provided high shoot multiplication (12.8) and the longest shoot length (5.3 cm) in MS + 10% CW. The greatest root number (7.3) and root length (5.0 cm) were noticed in MS + fungal elicitor CVS4 extracted from the stem of Vanda cristata. | [127] |
| Spathoglottis plicata Blume | Leaves | The highest somatic embryogenesis (93.7%) was observed in MS +1.0 mg L−1 2,4-D. Somatic embryo proliferation and shoot bud development were high in MS + 2.0 mg L−1 BAP. ½ MS + 1.0 mg L−1 IBA generated maximum rooting (93.6%) in ½ MS + 1.0 mg L−1 IBA. Synthetic seeds were best formed with somatic embryos encapsulated in 3% sodium alginate + 100 mM CaCl2. | [128] |
| Spathoglottis plicata Blume | Seeds | High seed germination (93%) was observed on MS + 1.0 mg L−1 BAP, while lower germination was witnessed on MS with either KN, IAA, or NAA. The shoots obtained from liquid culture showed a better rooting response (94%), producing higher root numbers and lengths (13.0 ± 0.22 roots per shoot, 4.0 ± 0.25 cm length) compared to root development (6.5 ± 0.29 roots and 3.3 ± 0.19 cm length) observed on SS medium. | [6] |
| Stanhopea tigrina Bateman ex Lind. | Seeds | High seed germination (98%) was witnessed after 120 days of culture on MS + 1% AC. MS + 10 g L−1 apple extract or 10 g L−1 banana extract or 30 mL L−1 CW or 5.0 mg L−1 BAP were effective for shoot induction (1.25 ± 0.35). Maximum root formation (9.00 ± 0.68 roots) was achieved in MS + 5.0 mg L−1 IAA + 100 mL L−1 CW. | [129] |
| Tolumnia Snow Fairy | Leaf segments from in vitro plants | Maximum PLB formation was found in MS + 4.0 mg L−1 BAP + 0.5 mg L−1 NAA with an average of 24.0 PLBs. However, there was no PLB formation from the outer leaves; only the inner expanding leaves showed protocorm induction (25.5 PLBs per explant) on MS + 4.0 mg L−1 BAP + 0.5 mg L−1 NAA. The shoot generation rate from PLBs was 33.3% for the whole PLB, while upper PLB halves produced 40%. | [75] |
| Vanda bicolor Griff | Seeds | MS + 3 µM NAA + 3 µM BA showed 88.2% seed germination. The protocorms developed into plantlets with healthy leaves (6.2) and roots (3.3) on MS + 3 µM NAA + 6 µM BA + AC (0.6%). | [19] |
| Vanda brunnea Rchb.f. | Shoot tips | A high plant regeneration rate (92–100%) was witnessed with Orchimax and MS medium supplemented with 0.5 mg L−1 BA. Orchimax showed the highest plant regeneration rate (100%), irrespective of the presence of BA in the medium. The number of plants obtained in Orchimax (6.2 per explant) was two times more than the plants produced (3.1 per explant) in MS. | [130] |
| Vanda cristata Wall. ex Lindl. | Whole leaf | The regeneration responses from the leaf explants were maximum (100%) on both M and KC medium fortified with NAA (10.6 µM) and BAP (8.8 µM). The highest number of plantlets (6) were obtained after explant differentiation via callus, PLBs, and shoot bud formation on the same hormonal combination. | [131] |
| Vanilla planifolia Jacks. ex Andrews | Seeds | Seeds germinated successfully on ½ MS + 2 mg L−1 glycine + 0.5 mg L−1 niacin + 0.5 mg L−1 pyridoxine HCl + 0.1 mg L−1 thiamine + 1 g L−1 tryptone + 20 g L−1 sucrose + 7 g L−1 agar. The seed germination time increased from 75 to 90 min when the mature seeds were treated with 4% sodium chlorite solution before inoculation, and germination percentage was recorded highest with immature seeds collected 45 days after pollination. The seedlings developed after the protocorms were grown on ½ MS + 20 g L−1 sucrose + 1 g L−1 AC + 20 g L−1 potato homogenate + 7 g L−1 agar. | [23] |
| Vanilla planifolia Jacks. ex Andrews | Leaf segments | The leaf explants produced non-embryogenic calli on MS + 3.0 mg L−1 2,4-D. The non-embryonic callus acquired embryogenic potential when transferred on MS + 1.0 mg L−1 BAP + 1.0 mg L−1 KN + 2.5 mg L−1 SN. Leaf explants, however, induced direct somatic embryogenesis (92.0%) on MS + 1.0 mg L−1 BAP + 1.0 mgL −1 KN + 2.5 mg L−1 SN generating maximum somatic embryos (30.0 per explant). The embryos encapsulated and stored at −4 °C for 1 year demonstrated the highest germination (95.3 ± 0.49) and shoot multiplication (17.2 shoots per SE) on MS + 0.5 mg L−1 BAP + 0.25 mg L−1 KN + 2.5 mg L−1 SN. | [25] |
| Vanda pumila Hook.f. | Protocorms | Highest shoots (9.50 ± 0.29) per culture formed on ½ MS + 1.0 mg L−1 KN + 10% CW. The shoot length was greatest (0.78 ± 0.07 cm) per culture on MS + 2.0 mg L−1 BAP + 10% CW. The ½ MS + 0.5 mg L−1 IAA produced high root formation (5 ± 0.00) per culture with good root length (0.93 ± 0.07 cm). | [132] |
| Vanda tessellate (Roxb.) Hook. ex G. Don | Seeds | MS gave the maximum seed germination (94%). MS + 2.0 mg L−1 BAP + 0.5 mg L−1 IAA produced the highest (89.4%) calli induction. Highest somatic embryo production (96%) from PLBs was observed in MS + 1.0 mg L−1 BAP + 0.5 mg L−1 IAA. Synthetic seed formation was best in MS + 2% sodium alginate + 100 mM CaCl2. Maximum germination (91%) of the cold stored encapsulated seeds was witnessed in MS + 50 mg L−1 ascorbic acid + 25 mg L−1 each of citric acid, adenine sulphate, and L-arginine + 0.5 mg L−1 each of BAP, KN, and IAA. | [133] |
3. Orchid Micropropagation Using Temporary Immersion System (TIS)
3.1. Important Factors Influencing Temporary Immersion System (TIS)
3.2. Benefits and Drawbacks of TIS
3.3. Application of TIS for In Vitro Orchid Propagation
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Arditti, J. Fundamentals of Orchid Biology; John Wiley & Sons: New York, NY, USA, 1992. [Google Scholar]
- Nirmala, C.; Nongdam, P.; Tewari, R. Biotechnological and Molecular Approaches for Improvement of Orchids. Plant Cell Biotechnol. Mol. Biol. 2006, 7, 1–10. [Google Scholar]
- Vendrame, W.A.; Khoddamzadeh, A.A. Orchid biotechnology. In Horticultural Reviews, 1st ed.; Janick, J., Ed.; Wiley Blackwell: Hoboken, NJ, USA, 2017; pp. 173–228. [Google Scholar]
- Nongdam, P.; Nirmala, C. Genetic variability in four species of Cymbidium based on isozyme markers. Physiol. Mol. Biol. Plants 2007, 13, 65–68. [Google Scholar]
- Vendrame, W.A. Cryopreservation. In Orchid Propagation: From Laboratories to Greenhouses—Methods and Protocols; Springer Protocols Handbooks; Lee, Y.I., Yeung, E.T., Eds.; Humana Press: New York, NY, USA, 2018; pp. 283–302. [Google Scholar]
- Shekhawat, M.S.; Latha, R.; Priyadharshini, S.; Manokari, M. Improved micropropagation, morphometric traits and photosynthetic pigments content using liquid culture system in Spathoglottis plicata Blume. Vegetos 2022, 35, 9–18. [Google Scholar] [CrossRef]
- Hossain, M.M. Therapeutic orchids: Traditional uses and recent advances—An overview. Fitoterapia 2011, 82, 102–140. [Google Scholar] [CrossRef]
- Castillo-Perez, L.J.; Martinez-Soto, D.; Maldonado-Miranda, J.J.; Alonso-Castro, A.J.; Carranza-Alvarez, C. The endemic orchids of Mexico: A review. Biologia 2019, 74, 1–13. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Lalthafamkimi, L.; Sharma, T.; Kumar, D.; Kumar, A.; Kumar, D.; Kumar, S. Metabolic and tissue-specific expression profiling in micropropagated plants of Malaxis acuminata: An endangered medicinal orchid. Plant Cell Tissue Organ Cult. 2022, 151, 535–549. [Google Scholar] [CrossRef]
- Arora, M.; Arora, K.; Kaur, R. Pharmacognostic, physicochemical, phytochemical, nutraceutical evaluation and in vitro antioxidant potency of Habenaria intermedia (D. Don) Szlach-A rare orchid. S. Afr. J. Bot. 2023, 152, 278–287. [Google Scholar] [CrossRef]
- Tiwari, P.; Bose, S.K.; Gautam, A.; Chen, J.T. Emerging trends and insights into the cultivation strategies, ethnomedicinal uses, and socio-economic attributes of orchids. J. Hortic. Sci. Biotechnol. 2023, 1–26. [Google Scholar] [CrossRef]
- Tikendra, L.; Amom, T.; Nongdam, P. Molecular genetic homogeneity assessment of micropropagated Dendrobium moschatum Sw.—A rare medicinal orchid, using RAPD and ISSR markers. Plant Gene 2019, 19, 100196. [Google Scholar] [CrossRef]
- Fay, M.F. Orchid conservation: How can we meet the challenges in the twenty-first century? Bot. Stud. 2018, 59, 16. [Google Scholar] [CrossRef]
- Wraith, J.; Norman, P.; Pickering, C. Orchid conservation and research: An analysis of gaps and priorities for globally Red Listed species. Ambio 2020, 49, 1601–1611. [Google Scholar] [CrossRef]
- Wraith, J.; Pickering, C. Quantifying anthropogenic threats to orchids using the IUCN Red List. Ambio 2018, 47, 307–317. [Google Scholar] [CrossRef] [PubMed]
- IUCN (International Union for Conservation of Nature). The IUCN Red List of Threatened Species, Orchidaceae. Available online: https://www.iucnredlist.org/ (accessed on 1 December 2022).
- Nongdam, P.; Nirmala, C.; Tewari, R. In vitro multiplication of Cymbidium orchids via embryo culture. Plant Cell Biotechnol. Mol. Bio. 2006, 7, 145–150. [Google Scholar]
- Bhattacharjee, B.; Islam, S.S. Effects of plant growth regulators on multiple shoot induction in Vanda tessellata (Roxb.) Hook. Ex G. Don an endangered medicinal orchid. Int. J. Sci. Nature 2014, 5, 707–712. [Google Scholar]
- Deb, C.R.; Pongener, A.; Kikon, Z.P. Asymbiotic immature seed culture of Vanda bicolor Griff., plant regeneration and formulation of an efficient new hardening technique for direct field transfer of regenerates. S. Afr. J. Bot. 2022, 150, 744–751. [Google Scholar] [CrossRef]
- Vendrame, W.A.; Maguire, I.; Carvalho, V.S. In vitro propagation and plantlet regeneration from Doritaenopsis purple gem ‘Ching Hua’flower explants. Hort. Sci. 2007, 42, 1256–1258. [Google Scholar]
- Chugh, S.; Guha, S.; Rao, I.U. Micropropagation of orchids: A review on the potential of different explants. Sci. Hortic. 2009, 122, 507–520. [Google Scholar] [CrossRef]
- Nongdam, P.N.; Nirmala, C. In vitro rapid propagation of Cymbidium aloifolium (L.) Sw.: A medicinally important orchid via seed culture. J. Biol. Sci. 2011, 11, 254–260. [Google Scholar] [CrossRef]
- Yeh, C.H.; Chen, K.Y.; Lee, Y.I. Asymbiotic germination of Vanilla planifolia in relation to the timing of seed collection and seed pretreatments. Bot. Stud. 2021, 62, 6. [Google Scholar] [CrossRef]
- Singh, N.; Kumaria, S. A combinational phytomolecular-mediated assessment in micropropagated plantlets of Coelogyne ovalis Lindl.: A horticultural and medicinal Orchid. Proc. Natl. Acad. Sci. India Sec. B Biol. Sci. 2020, 90, 455–466. [Google Scholar] [CrossRef]
- Manokari, M.; Priyadharshini, S.; Cokulraj, M.; Dey, A.; Faisal, M.; Alatar, A.A.; Alok, A.; Shekhawat, M.S. Exogenous implications of silver nitrate on direct and indirect somatic embryogenesis and germination of cold stored synseeds of Vanilla planifolia Jacks. ex Andrews. S. Afr. J. Bot. 2022, 150, 129–138. [Google Scholar] [CrossRef]
- Reuther, G. Principles and application of the micropropagation of ornamental plants. In In Vitro Techniques: Propagation and Long-Term Storage; Schäfer-Menuhr, A., Ed.; Martinus Nijhoff: Dordrecht, The Netherlands, 1985; pp. 1–14. [Google Scholar]
- Berthouly, M.; Etienne, H. Temporary immersion system: A new concept for use liquid medium in mass propagation. In Liquid Culture Systems for in Vitro Plant Propagation; Hvoslef-Eide, A.K., Walter, P., Eds.; Springer: Heidelberg, Germany, 2005; pp. 165–195. [Google Scholar]
- Shin, K.S.; Park, S.Y.; Paek, K.Y. Physiological and biochemical changes during acclimatization in a Doritaenopsis hybrid cultivated in different microenvironments in vitro. Environ. Exp. Bot. 2014, 100, 26–33. [Google Scholar] [CrossRef]
- Ramírez-Mosqueda, M.A.; Iglesias-Andreu, L.G. Evaluation of different temporary immersion systems (BIT®, BIG, and RITA®) in the micropropagation of Vanilla planifolia Jacks. In Vitro Cell. Dev. Biol. Plant 2016, 52, 154–160. [Google Scholar] [CrossRef]
- Liu, T.H.; Kuo, S.S.; Wu, R.Y. Mass micropropagation of orchid protocorm-like bodies using air-driven periodic immersion bioreactor. Acta Hortic. 2002, 578, 187–191. [Google Scholar] [CrossRef]
- Gao, R.; Wu, S.Q.; Piao, X.C.; Park, S.Y.; Lian, M.L. Micropropagation of Cymbidium sinense using continuous and temporary airlift bioreactor systems. Acta Physiol. Plant 2014, 36, 117–124. [Google Scholar] [CrossRef]
- Zhang, B.; Song, L.; Bekele, L.D.; Shi, J.; Jia, Q.; Zhang, B.; Jin, L.; Duns, G.J.; Chen, J. Optimizing factors affecting development and propagation of Bletilla striata in a temporary immersion bioreactor system. Sci. Hortic. 2018, 232, 121–126. [Google Scholar] [CrossRef]
- Fritsche, Y.; Pinheiro, M.V.M.; Guerra, M.P. Light quality and natural ventilation have different effects on protocorm development and plantlet growth stages of the in vitro propagation of Epidendrum fulgens (Orchidaceae). S. Afr. J. Bot. 2022, 146, 864–874. [Google Scholar] [CrossRef]
- Poobathy, R.; Zakaria, R.; Murugaiyah, V.; Subramaniam, S. Surface sterilization and micropropagation of Ludisia discolor. Biocatal. Agric. Biotechnol. 2019, 22, 101380. [Google Scholar] [CrossRef]
- Tikendra, L.; Koijam, A.S.; Nongdam, P. Molecular markers based genetic fidelity assessment of micropropagated Dendrobium chrysotoxum Lindl. Meta Gene 2019, 20, 100562. [Google Scholar] [CrossRef]
- Poniewozik, M.; Parzymies, M.; Szot, P. The influence of disinfection methods and liquid phase media on Paphiopedilum insigne seeds germination and media supplements on morphological features of protocorms in tissue culture. Acta Sci. Pol. Hortorum Cultus 2020, 19, 7–14. [Google Scholar] [CrossRef]
- Guo, B.; Zeng, S.; Yin, Y.; Li, L.; Ma, G.; Wu, K.; Fang, L. Characterization of phytohormone and transcriptome profiles during protocorm-like bodies development of Paphiopedilum. BMC Genom. 2021, 22, 806. [Google Scholar] [CrossRef] [PubMed]
- Perner, H.; Zhou, R.; Perner, W.; Jiang, H.; Lee, Y.I. Cypripedium subtropicum embryo development and cytokinin requirements for asymbiotic germination. Bot. Stud. 2022, 63, 28. [Google Scholar] [CrossRef] [PubMed]
- Lal, N.; Singh, M. Prospect of plant tissue culture in orchid propagation: A review. Indian J. Biol. 2020, 7, 103–110. [Google Scholar]
- Sonia, P.; Apana, N.; Tikendra, L.; Dey, A.; Jamir, I.; Nongdam, P. Molecular clonal fidelity assessment of micropropagated orchids using dna markers. In Biotechnology and Crop Improvement, 1st ed.; Kumar, N., Ed.; CRC Press: Boca Raton, FL, USA, 2022; pp. 143–179. [Google Scholar]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Planatarum 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Mitra, G.C.; Prasad, R.N.; Roychowdhury, A. Inorganic salts and differentiation of protocorms in seed callus of orchid and correlative changes in its free amino acid content. Indian J. Exp. Biol. 1976, 14, 350–351. [Google Scholar]
- Knudson, L. A nutrient for the germination of orchid seeds. Amer. Orchid Soc. Bull. 1946, 15, 214–217. [Google Scholar]
- Vacin, E.F.; Went, F.W. Some pH changes in nutrient solutions. Bot. Gaz. 1949, 110, 605–613. [Google Scholar] [CrossRef]
- Churchill, M.E.; Ball, E.A.; Arditti, J. Tissue culture of orchids—II. Methods for root tips. Am. Orchid Soc. Bull. 1972, 41, 726–730. [Google Scholar]
- Sidek, N.; Mohd Anuar, N.S.; Naher, L.; Azmi, K.; Rahman, M.A. The effect of different nutrient media on in vitro shoot and root proliferation of Vanilla planifolia Jacks. ex Andrews. Afr. J. Biotechnol. 2018, 17, 1241–1246. [Google Scholar]
- Nongdam, P.; Chongtham, N. In vitro seed germination and mass propagation of Cymbidium dayanum Reichb: An important ornamental orchid of North-East Indian. Trends Hort. Res. 2012, 2, 28–37. [Google Scholar] [CrossRef][Green Version]
- Nabieva, A.Y. Asymbiotic seed germination and in vitro seedling development of Orchis militaris, an endangered orchid in Siberia. J. Genet. Eng. Biotechnol. 2021, 19, 1–11. [Google Scholar] [CrossRef]
- Longchar, T.B.; Deb, C.R. Optimization of in vitro propagation protocol of Dendrobium heterocarpum Wall. ex. Lindl. and clonal genetic fidelity assessment of the regenerates: An orchid of horticultural and medicinal importance. S. Afr. J. Bot. 2022, 149, 67–78. [Google Scholar] [CrossRef]
- Baltazar-Bernal, O.; De la Cruz-Martínez, V.M.; Ramírez-Mosqueda, M.A.; Zavala-Ruiz, J. In vitro seed germination and acclimatization of Encyclia cordigera (Kunth) Dressler. S. Afr. J. Bot. 2022, 151, 578–582. [Google Scholar] [CrossRef]
- Herawati, R.; Ganefianti, D.W.; Romeida, A. Addition of coconut water and banana extract on MS media to stimulate PLB (protocorm like bodies) regeneration of Dendrobium gatton sunray. In Proceedings of the International Seminar on Promoting Local Resources for Sustainable Agriculture and Development (ISPLRSAD 2020); Atlantis Press: Amsterdam, The Netherlands, 2021; pp. 251–258. [Google Scholar]
- Zhang, Y.; Lee, Y.I.; Deng, L.; Zhao, S. Asymbiotic germination of immature seeds and the seedling development of Cypripedium macranthos Sw., an endangered lady’s slipper orchid. Sci. Hortic. 2013, 164, 130–136. [Google Scholar] [CrossRef]
- Pakum, W.; Watthana, S.; orn Srimuang, K.; Kongbangkerd, A. Influence of medium component on in vitro propagation of Thai s endangered orchid: Bulbophyllum nipondhii Seidenf. Plant Tiss. Cult. Biotechnol. 2016, 26, 37–46. [Google Scholar] [CrossRef]
- Ostojić, J.; Ljubojević, M.; Narandžić, T.; Pušić, M. In vitro culture conditions for asymbiotic germination and seedling development of Anacamptis pyramidalis (L.) Rich. and Gymnadenia conopsea (L.). R. Br. S. Afr. J. Bot. 2022, 150, 829–839. [Google Scholar] [CrossRef]
- Agbadje, E.T.A.E.; Agbidinoukoun, A.; Zandjanakou-Tachin, M.; Cacai, G.T.H.; Ahanhanzo, C. Mass production of bananas and plantains (Musa spp.) plantlets through in vitro tissue culture partway: A review. Eur. J. Biol. Biotech. 2021, 2, 229. [Google Scholar] [CrossRef]
- Tikendra, L.; Apana, N.; Potshangbam, A.M.; Amom, T.; Choudhary, R.; Sanayaima, R.; Dey, A.; Nongdam, P. Dendrobium sp.: In vitro propagation of genetically stable plants and ethnomedicinal uses. In Orchids Phytochemistry, Biology and Horticulture. Reference Series in Phytochemistry; Mérillon, J.M., Kodja, H., Eds.; Springer: Cham, Switzerland, 2022. [Google Scholar] [CrossRef]
- Chen, J.T.; Chang, W.C. Effects of auxins and cytokinins on direct somatic embryogenesis on leaf explants of Oncidium ‘Gower Ramsey’. Plant Growth Regul. 2001, 34, 229–232. [Google Scholar] [CrossRef]
- Tikendra, L.; Potshangbam, A.M.; Dey, A.; Devi, T.R.; Sahoo, M.R.; Nongdam, P. RAPD, ISSR, and SCoT markers based genetic stability assessment of micropropagated Dendrobium fimbriatum Lindl. var. oculatum Hk. f.- an important endangered orchid. Physiol. Mol. Biol. Plants 2021, 27, 341–357. [Google Scholar] [CrossRef]
- Teixeira da Silva, J.A.; Tsavkelova, E.A.; Ng, T.B.; Parthibhan, S.; Dobránszki, J.; Cardoso, J.C.; Rao, M.V.; Zeng, S. Asymbiotic in vitro seed propagation of Dendrobium. Plant Cell Rep. 2015, 34, 1685–1706. [Google Scholar] [CrossRef]
- Kaur, S.; Bhutani, K.K. In vitro propagation of Vanda testacea (Lindl.) Reichb. f. a rare orchid of high medicinal value. Plant Tiss. Cult. Biotechnol. 2009, 19, 1–7. [Google Scholar]
- Wu, K.; Zeng, S.; da Silva, J.A.T.; Chen, Z.; Zhang, J.; Yang, Y.; Duan, J. Efficient regeneration of Renanthera Tom Thumb ‘Qilin’ from leaf explants. Sci. Hortic. 2012, 135, 194–201. [Google Scholar] [CrossRef]
- Do, V.N.T.; Hsu, S.-T.; Lee, Y.-I. Clonal propagation in vitro of Paphiopedilum hybrids from adult plants. Hort Sci. 2019, 54, 1565–1569. [Google Scholar] [CrossRef]
- Arditti, J.; Ghani, A.K.A. Tansley Review No. 110.: Numerical and physical properties of orchid seeds and their biological implications. New Phytol. 2000, 145, 367–421. [Google Scholar] [CrossRef] [PubMed]
- Knudson, L. Nonsymbiotic germination of orchid seeds. Bot. Gaz. 1922, 73, 1–25. [Google Scholar] [CrossRef]
- Bustam, B.M.; Dixon, K.W.; Bunn, E. In vitro propagation of temperate Australian terrestrial orchids: Revisiting asymbiotic compared with symbiotic germination. Bot. J. Linn. Soc. 2014, 176, 556–566. [Google Scholar] [CrossRef]
- Nongdam, P.; Tikendra, L. Establishment of an efficient in vitro regeneration protocol for rapid and mass propagation of Dendrobium chrysotoxum Lindl. using seed culture. Sci. World J. 2014, 740150. [Google Scholar]
- Rodrigues, L.A.; Paiva Neto, V.B.D.; Boaretto, A.G.; Oliveira, J.F.D.; Torrezan, M.D.A.; Lima, S.F.D.; Otoni, W.C. In vitro propagation of Cyrtopodium saintlegerianum rchb. f.(orchidaceae), a native orchid of the Brazilian savannah. Crop Breed. Appl. Biotechnol. 2015, 15, 10–17. [Google Scholar] [CrossRef]
- Tikendra, L.; Amom, T.; Nongdam, P. Effect of phytohormones on rapid in vitro propagation of Dendrobium thyrsiflorum Rchb. f.: An endangered medicinal orchid. Pharmacogn. Mag. 2018, 14, 495–500. [Google Scholar]
- Soonthornkalump, S.; Nakkanong, K.; Meesawat, U. In vitro cloning via direct somatic embryogenesis and genetic stability assessment of Paphiopedilum niveum (Rchb. f.) Stein: The endangered Venus’s slipper orchid. In Vitro. Cell. Dev. Biol. Plant 2019, 55, 265–276. [Google Scholar] [CrossRef]
- Kang, H.; Kang, K.W.; Kim, D.H.; Sivanesan, I. In vitro propagation of Gastrochilus matsuran (Makino) Schltr., an endangered epiphytic orchid. Plants 2020, 9, 524. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Gayatri, M.C.; Sarangi, S.K. In vitro seed germination and plant regeneration of an epiphytic orchid Aerides ringens (Lindl.) Fischer. Indian J. Biotechnol. 2015, 14, 574–580. [Google Scholar]
- Wu, K.; Zeng, S.; Lin, D.; Teixeira da Silva, J.A.; Bu, Z.; Zhang, J.; Duan, J. In vitro propagation and reintroduction of the endangered Renanthera imschootiana Rolfe. PLoS ONE 2014, 9, e110033. [Google Scholar] [CrossRef]
- Wimber, D.E. Additional observations on clonal multiplication of Cymbidiums through culture of shoot meristems. Cymbidium Soc. News 1965, 20, 7–10. [Google Scholar]
- Hardjo, P.H.; Savitri, W.D. Somatic embryo from basal leaf segments of Vanda tricolor Lindl. var. pallida. KnE Life Sci. 2017, 3, 173–179. [Google Scholar] [CrossRef]
- Chookoh, N.; Chiu, Y.T.; Chang, C. Micropropagation of Tolumnia orchids through induction of protocorm-like bodies from leaf segments. Hortscience 2019, 54, 1230–1236. [Google Scholar] [CrossRef]
- Pathak, P.; Verma, S.; Prakash, A.; Mahant, K.C. Regeneration competence of an ornamentally important epiphytic orchid, Rhynchostylis gigantea (Lindl.) Ridl. through leaf segments: A study in vitro. J. Orchid Soc. India 2017, 31, 97–101. [Google Scholar]
- Decruse, S.W.; Gangaprasad, A. Restoration of Smithsonia maculata (Dalz.) Saldanha, an endemic and vulnerable orchid of Western Ghats through in vitro propagation. J. Orchid Soc. India 2018, 32, 25–32. [Google Scholar]
- Morel, G.M. Producing virus free cymbidiums. Amer. Orchid Soc. Bull. 1960, 29, 495–497. [Google Scholar]
- Sopalun, K.; Thammasiri, K.; Ishikawa, K. Micropropagation of the Thai orchid Grammatophyllum speciosum blume. Plant Cell Tissue Organ Cult. 2010, 101, 143–150. [Google Scholar] [CrossRef]
- Mamun, A.A.; Islam, M.M.; Ahmed, M.; Ghosh, G.C. In vitro mass propagation from shoot tip of Dendrobium Red Bull-an endangered epiphytic orchid. Plant Tissue Cult. Biotechnol. 2018, 28, 161–169. [Google Scholar] [CrossRef]
- Ma, N.L.; Khoo, S.C.; Lee, J.X.; Soon, C.F.; Shukor, N.A.A.B. Efficient micro-propagation of Dendrobium aurantiacum from shoot explant. Plant Sci. Today 2020, 7, 476–482. [Google Scholar] [CrossRef]
- Farrokhzad, Y.; Babaei, A.; Yadollahi, A.; Kashkooli, A.B.; Mokhtassi-Bidgoli, A.; Hessami, S. Informative title: Development of lighting intensity approach for shoot proliferation in Phalaenopsis amabilis through combination with silver nanoparticles. Sci. Hortic. 2022, 292, 110582. [Google Scholar] [CrossRef]
- Priyakumari, I.; Sabrina George, T.; Rajmohan, K. Influence of plant growth regulators on in vitro clonal propagation of Dendrobium Sonia ‘Earsakul’. J. Bio. Innov. 2013, 2, 51–58. [Google Scholar]
- Preetha, L.K.; Shylaraj, K.S.; Rohini, P.C. An improved method for rapid propagation of Phalaenopsis hybrids via culture of longitudinally bisected shoot tips. J. Trop. Agric. 2017, 55, 45–51. [Google Scholar]
- Winarto, B.; Samijan. Axillary shoots derived from shoot tips in in vitro mass propagation of Anoectochilus formosanus Hayata. J. Agric. Sci. 2018, 2, 121–130. [Google Scholar]
- Pant, B.; Thapa, D. In vitro mass propagation of an epiphytic orchid, Dendrobium primulinum Lindl. through shoot tip culture. Afr. J. Biotechnol. 2012, 11, 9970–9974. [Google Scholar]
- Kerbauy, G.B. Regeneration of protocorm-like bodies through in vitro culture of root tips of Catasetum (Orchidaceae). Z. Pflanzenphysiol. 1984, 113, 287–291. [Google Scholar] [CrossRef]
- Sharma, V. In vitro conversion of root meristem into protocorm like bodies in Rhynchostylis retusa-an endangered fox tail orchid. Eco. Env. Cons. 2019, 25, 1812–1816. [Google Scholar]
- Verma, S.; Pathak, P. Regenerative competence in root explants of Cymbidium aloifolium, a medicinally important orchid: A study in vitro. Vegetos 2018, 31, 100–103. [Google Scholar] [CrossRef]
- Picolotto, D.R.N.; Paiva, V.B.D.; Barros, F.D.; Padilha, D.R.C.; Cruz, A.C.F.D.; Otoni, W.C. Micropropagation of Cyrtopodium paludicolum (Orchidaceae) from root tip explants. Crop Breed. Appl. Biotechnol. 2017, 17, 191–197. [Google Scholar] [CrossRef][Green Version]
- Maene, L.; Debergh, P. Liquid medium additions to established tissue cultures to improve elongation and rooting in vivo. Plant Cell Tissue Organ Cult. 1985, 5, 23–33. [Google Scholar] [CrossRef]
- Paek, K.Y.; Chakrabarty, D.; Hahn, E.J. Application of bioreactor systems for large scale production of horticultural and medicinal plants. Plant Cell Tissue Organ Cult. 2005, 81, 287–300. [Google Scholar] [CrossRef]
- Vasudevan, R.; Van Staden, J. Cytokinin and explant types influence in vitro plant regeneration of Leopard Orchid (Ansellia africana Lindl.). Plant Cell Tissue Organ Cult. 2011, 107, 123–129. [Google Scholar] [CrossRef]
- Chu, I. Economic analysis of automated micropropagation. In Automation and environmental control in plant tissue culture; Aitken-Christie, A., Kozai, T., Smith., M.A.L., Eds.; Springer: Dordrecht, The Netherlands, 1995; pp. 19–27. [Google Scholar]
- Etienne, H.; Berthouly, M. Temporary immersion systems in plant micropropagation. Plant Cell Tissue Organ Cult. 2002, 69, 215–231. [Google Scholar] [CrossRef]
- Ahmadian, M.; Babaei, A.; Shokri, S.; Hessami, S. Micropropagation of carnation (Dianthus caryophyllus L.) in liquid medium by temporary immersion bioreactor in comparison with solid culture. J. Genet. Eng. Biotechnol. 2017, 15, 309–315. [Google Scholar] [CrossRef]
- Cardoso, J.C.; Sheng Gerald, L.T.; Teixeira da Silva, J.A. Micropropagation in the twenty-first century. Methods Mol. Biol. 2018, 1815, 17–46. [Google Scholar]
- Robert, M.L.; Herrera-Herrera, J.L.; Herrera-Herrera, G.; Herrera-Alamillo, M.Á.; Fuentes-Carrillo, P. A new temporary immersion bioreactor system for micropropagation. Methods Mol. Biol. 2006, 318, 121–129. [Google Scholar]
- Mirzabe, A.H.; Hajiahmad, A.; Fadavi, A.; Rafiee, S. Temporary immersion systems (TISs): A comprehensive review. J. Biotechnol. 2022, 357, 56–83. [Google Scholar] [CrossRef]
- Debnath, S. Bioreactors and molecular analysis in berry crop micropropagation—A review. Can. J. Plant Sci. 2011, 91, 147–157. [Google Scholar] [CrossRef]
- Yaseen, M.; Ahmad, T.; Sablok, G.; Standardi, A.; Hafiz, I.A. Role of carbon sources for in vitro plant growth and development. Mol. Biol. Rep. 2013, 40, 2837–2849. [Google Scholar] [CrossRef] [PubMed]
- San José, M.C.; Blázquez, N.; Cernadas, M.J.; Janeiro, L.V.; Cuenca, B.; Sánchez, C.; Vidal, N. Temporary immersion systems to improve alder micropropagation. Plant Cell Tissue Organ Cult. 2020, 143, 265–275. [Google Scholar] [CrossRef]
- Bhowmik, T.K.; Rahman, M.M. In vitro study of medicinally important orchid Aerides multiflora Roxb. from nodal and leaf explants. J. Pharmacogn. Phytochem. 2020, 9, 179–184. [Google Scholar]
- Xu, J.; Beleski, D.G.; Vendrame, W.A. Effects of culture methods and plant growth regulators on in vitro propagation of Brassavola nodosa (L.) Lindl. hybrid. In Vitro Cell. Dev. Biol. Plant 2022, 58, 931–941. [Google Scholar] [CrossRef]
- Castillo-Pérez, L.J.; Alonso-Castro, A.J.; Fortanelli-Martínez, J.; Carranza-Álvarez, C. Micropropagation of Catasetum integerrimum Hook (Orchidaceae) through seed germination and direct shoot regeneration from pseudobulbs and roots. In Vitro Cell. Dev. Biol. Plant 2022, 58, 279–289. [Google Scholar] [CrossRef]
- Mercado, S.A.S.; Jaimes, Y.M.O. Implementation of organic components to the culture medium to improve the in vitro propagation of Cattleya warscewiczii and Cattleya gaskelliana. S. Afr. J. Bot. 2022, 148, 352–359. [Google Scholar] [CrossRef]
- Navarro, Q.R.; de Oliveira Corrêa, D.; Behling, A.; Noseda, M.D.; Ribas, L.L.F. Effect of microalga Desmodesmus subspicatus, polyamines and plant growth regulators on the in vitro propagation of Cattleya warneri. Plant Cell Tissue Organ Cult. 2023. [Google Scholar] [CrossRef]
- Vasundhara; Promila, P.; Prakash, A. In vitro shoot induction and regeneration potential of floral buds in Crepidium acuminatum (D. Don) Szlach., a medicinal ayurvedic plant from Northwestern Himalayas. J. Orchid Soc. India 2019, 33, 43–48. [Google Scholar]
- Sembi, J.K.; Pathak, P.; Verma, J. Regeneration competence of leaf explants in Cymbidium eburneum lindl. (orchidaceae). J. Orchid Soc. India 2020, 34, 17–21. [Google Scholar]
- Nguyen, H.T.; Dinh, S.T.; Ninh, T.T.; Nong, H.T.; Dang, T.T.; Khuat, Q.V.; Dang, A.T.; Ly, M.T.; Kirakosyan, R.N.; Kalashnikova, E.A. In Vitro Propagation of the Dendrobium anosmum Lindl. collected in Vietnam. Agronomy 2022, 12, 324. [Google Scholar] [CrossRef]
- Maharjan, S.; Thakuri, L.S.; Thapa, B.B.; Pradhan, S.; Pant, K.K.; Joshi, G.P.; Pant, B. In vitro propagation of the endangered orchid Dendrobium chryseum Rolfefrom protocorms culture. Nepal J. Sci. Tech. 2020, 19, 39–47. [Google Scholar] [CrossRef]
- Gurung, P.R.; Shrestha, A.; Adhikari, S.; Limbu, S.; Pant, B. Asymbiotic seed germination and seedling development of a medicinally important epiphytic orchid, Dendrobium crepidatum Lindl. & Paxton. J. Plant Res. 2020, 18, 226–234. [Google Scholar]
- Pant, B.; Chand, K.; Paudel, M.R.; Joshi, P.R.; Thapa, B.B.; Park, S.Y.; Shakya, S.; Thakuri, L.S.; Rajbahak, S.; Sah, A.K.; et al. Micropropagation, antioxidant and anticancer activity of pineapple orchid: Dendrobium densiflorum Lindl. J. Plant Biochem. Biotechnol. 2022, 31, 399–409. [Google Scholar] [CrossRef]
- Pujari, I.; Thomas, A.; Rai, P.S.; Satyamoorthy, K.; Babu, V.S. In vitro bioproduction and enhancement of moscatilin from a threatened tropical epiphytic orchid, Dendrobium ovatum (Willd.) Kraenzl. 3 Biotech 2021, 11, 507. [Google Scholar] [CrossRef] [PubMed]
- Pyati, A.N. In vitro Propagation of orchid (Dendrobium ovatum (L.) Kraenzl.) through somatic embryogenesis. Plant Tissue Cult. Biotechnol. 2022, 32, 53–66. [Google Scholar] [CrossRef]
- Bhowmik, T.K.; Rahman, M.M. Micropropagation of commercially important orchid Dendrobium palpebrae Lindl. through in vitro developed pseudobulb culture. J. Adv. Biotechnol. Exp. Ther. 2020, 3, 225–232. [Google Scholar] [CrossRef]
- Subrahmanyeswari, T.; Verma, S.K.; Gantait, S. One-step in vitro protocol for clonal propagation of Dendrobium Yuki White, a high value ornamental orchid hybrid. S. Afr. J. Bot. 2022, 146, 883–888. [Google Scholar] [CrossRef]
- Thipwong, J.; Kongton, K.; Samala, S. Micropropagation and somaclonal variation of Doritis pulcherrima (Lindl.). Plant Biotechnol. Rep. 2022, 16, 401–408. [Google Scholar] [CrossRef]
- Dos Santos Anjos, J.; Alves Stefanello, C.; do Nascimento Vieira, L.; Giacomolli Polesi, L.; Guerra, M.P.; Pacheco de Freitas Fraga, H. The cytokinin 6-benzylaminopurine improves the formation and development of Dryadella zebrina (Orchidaceae) in vitro shoots. Brazilian J. Bot. 2021, 44, 811–819. [Google Scholar] [CrossRef]
- Cabral, N.N.; Pescador, R.; Pinheiro, M.V.M.; Ornellas, T.S.; Rizzolo, R.G.; Bordallo, S.U.; Guterres, S.M.; Gris, T.; Schvambach, M.I.; de Souza, P.F. Different spectral qualities do not influence the in vitro and ex vitro survival of Epidendrum denticulatum Barb. Rod.: A Brazilian orchid. Vegetos 2022. [Google Scholar] [CrossRef]
- Panwar, G.S.; Joshi, B.; Joshi, R. Axenic rhizome culture and genetic fidelity assessment of Eulophia dabia (D. Don) Hochr: An endangered terrestrial orchid species. In Vitro Cell. Dev. Biol. Plant 2022, 58, 567–576. [Google Scholar] [CrossRef]
- Vera-Aguilar, A.; Ramírez-Mosqueda, M.A.; Lee-Espinosa, H.E.; Llarena-Hernández, R.C.; Rodríguez-Deméneghi, M.V.; Murguía-González, J. Efficient protocol for in vitro propagation of Laelia anceps ssp. anceps white variant from asymbiotic seed germination. S. Afr. J. Bot. 2022, 149, 376–380. [Google Scholar] [CrossRef]
- Gantait, S.; Subrahmanyeswari, T.; Sinniah, U.R. Leaf-based induction of protocorm-like bodies, their encapsulation, storage and post-storage germination with genetic fidelity in Mokara Sayan× Ascocenda Wangsa gold. S. Afr. J. Bot. 2022, 150, 893–902. [Google Scholar] [CrossRef]
- Fatahi, M.; Vafaee, Y.; Nazari, F.; Tahir, N.A.R. In vitro asymbiotic seed germination, protocorm formation, and plantlet development of Orchis simia Lam.: A threatened terrestrial orchid species. S. Afr. J. Bot. 2022, 151, 156–165. [Google Scholar] [CrossRef]
- Utami, E.S.W.; Hariyanto, S. In vitro seed germination and seedling development of a rare indonesian native orchid Phalaenopsis amboinensis JJ Sm. Scientifica 2019, 2019, 8105138. [Google Scholar] [CrossRef] [PubMed]
- Chashmi, K.A.; Omran, V.O.G.; Ebrahimi, R.; Moradi, H.; Abdosi, V. Light quality affects protocorm-like body (plb) formation, growth and development of in vitro plantlets of Phalaenopsis pulcherrima. Biol. Bull. 2022, 49, 476–484. [Google Scholar]
- Oliya, B.K.; Chand, K.; Thakuri, L.S.; Baniya, M.K.; Sah, A.K.; Pant, B. Assessment of genetic stability of micropropagated plants of Rhynchostylis retusa (L.) using RAPD markers. Sci. Hortic. 2021, 281, 110008. [Google Scholar] [CrossRef]
- Manokari, M.; Priyadharshini, S.; Shekhawat, M.S. Direct somatic embryogenesis using leaf explants and short term storage of synseeds in Spathoglottis plicata Blume. Plant Cell Tissue Organ Cult. 2021, 145, 321–331. [Google Scholar] [CrossRef]
- Castillo-Pérez, L.J.; Martínez-Soto, D.; Fortanelli-Martínez, J.; Carranza-Álvarez, C. Asymbiotic seed germination, in vitro seedling development, and symbiotic acclimatization of the Mexican threatened orchid Stanhopea tigrina. Plant Cell Tissue Organ Cult. 2021, 146, 249–257. [Google Scholar] [CrossRef]
- Nowakowska, K.; Marciniak, P.; Pacholczak, A. A protocol for efficient micropropagation of rare orchid Vanda brunnea Rchb. f. S. Afr. J. Bot. 2022, 150, 233–239. [Google Scholar] [CrossRef]
- Pathak, P.; Kumari, A.; Thakur, B. Regeneration competence of an endangered orchid, Vanda cristata Wall. ex Lindl. using leaf explants: A study in vitro. S. Afr. J. Bot. 2022, 151, 1018–1024. [Google Scholar] [CrossRef]
- Maharjan, S.; Pradhan, S.; Thapa, B.B.; Pant, B. In vitro propagation of endangered orchid, Vanda pumila Hook. f. through protocorms culture. Am. J. Plant Sci. 2019, 10, 1220–1232. [Google Scholar] [CrossRef]
- Manokari, M.; Latha, R.; Priyadharshini, S.; Jogam, P.; Shekhawat, M.S. Short-term cold storage of encapsulated somatic embryos and retrieval of plantlets in grey orchid (Vanda tessellata (Roxb.) Hook. ex G. Don). Plant Cell Tissue Organ Cult. 2021, 144, 171–183. [Google Scholar] [CrossRef]
- Albarran, J.; Bertrand, B.; Lartaud, M.; Etienne, H. Cycle characteristics in a temporary immersion bioreactor affect regeneration, morphology, water and mineral status of coffee (Coffea arabica) somatic embryos. Plant Cell Tissue Organ Cult. 2005, 81, 27–36. [Google Scholar] [CrossRef]
- Curtis, W.; Tuerk, A. Oxygen transport in plant tissue culture systems. In Plant Tissue Culture Engineering; Gupta, S.D., Ibaraki, Y., Eds.; Springer: Cham, The Netherlands, 2006; pp. 173–186. [Google Scholar]
- Yang, L.; Zambrano, Y.; Hu, C.-J.; Carmona, E.; Bernal, A.; Perez, A.; Zayas, C.M.; Li, Y.R.; Guerra, A.; Santana, I.; et al. Sugarcane metabolites produced in CO2-rich temporary immersion bioreactors (TIBs) induce tomato (Solanum lycopersicum) resistance against bacterial wilt (Ralstonia solanacearum). In Vitro Cell. Dev. Biol. Plant 2010, 46, 558–568. [Google Scholar] [CrossRef]
- Simonton, W.; Robacker, C.; Krueger, S. A programmable micropropagation apparatus using cycled liquid medium. Plant Cell Tissue Organ Cult. 1991, 27, 211–218. [Google Scholar] [CrossRef]
- Lorenzo, J.C.; González, B.L.; Escalona, M.; Teisson, C.; Borroto, C. Sugarcane shoot formation in an improved temporary immersion system. Plant Cell Tissue Organ Cult. 1998, 54, 197–200. [Google Scholar] [CrossRef]
- Escalona, M.; Lorenzo, J.C.; González, B.; Daquinta, M.; González, J.L.; Desjardins, Y.; Borroto, C.G. Pineapple (Ananas comosus L. Merr) micropropagation in temporary immersion systems. Plant Cell Rep. 1999, 18, 743–748. [Google Scholar] [CrossRef]
- Roels, S.; Escalona, M.; Cejas, I.; Noceda, C.; Rodriguez, R.; Canal, M.J.; Sandoval, J.; Debergh, P. Optimization of plantain (Musa AAB) micropropagation by temporary immersion system. Plant Cell Tissue Organ Cult. 2005, 82, 57–66. [Google Scholar] [CrossRef]
- Uma, S.; Karthic, R.; Kalpana, S.; Backiyarani, S.; Saraswathi, M.S. A novel temporary immersion bioreactor system for large scale multiplication of banana (Rasthali AAB—Silk). Sci. Rep. 2021, 11, 20371. [Google Scholar] [CrossRef]
- Monette, P.L. Influence of size of culture vessel on in vitro proliferation of grape in a liquid medium. Plant Cell Tissue Organ Cult. 1983, 2, 327–332. [Google Scholar] [CrossRef]
- Krueger, S.; Robacker, C.; Simonton, W. Culture of Amel-anchier × grandiflora in a programmable micropropagation apparatus. Plant Cell Tissue Organ Cult. 1991, 27, 219–226. [Google Scholar] [CrossRef]
- Aragón, C.E.; Sánchez, C.; Gonzalez-Olmedo, J.; Escalona, M.; Carvalho, L.; Amâncio, S. Comparison of plantain plantlets propagated in temporary immersion bioreactors and gelled medium during in vitro growth and acclimatization. Biol. Plant. 2014, 58, 29–38. [Google Scholar] [CrossRef]
- Florez, S.L.; Curtis, M.S.; Shaw, S.E.; Hamaker, N.K.; Larsen, J.S.; Curtis, W.R. A temporary immersion plant propagation bioreactor with decoupled gas and liquid flows for enhanced control of gas phase. Biotechnol. Prog. 2016, 32, 337–345. [Google Scholar] [CrossRef]
- Etienne, H.; Lartaud, M.; Michaux-Ferrière, N.; Carron, M.P.; Berthouly, M.; Teisson, C. Improvement of somatic embryogenesis in Hevea Brasiliensis (Müll. Arg.) using the temporary immersion technique. In Vitro Cell. Dev. Biol. Plant 1997, 33, 81–87. [Google Scholar] [CrossRef]
- Etienne, H.; Solano, W.; Pereira, A.; Bertrand, B.; Berthouly, M. Coffee in vitro plantlet acclimatization protocol. Plantations Rech. Dév. 1997, 4, 304–311. [Google Scholar]
- Berthouly, M.; Dufour, M.; Alvard, D.; Carasco, C.; Alemano, L.; Teisson, C. Coffee micropropagation in a liquid medium using the temporary immersion technique. In Proceedings of the 16th International Scientific Colloquium on Coffee, Kyoto, Japan, 9–14 April 1995; ASIC: Lausanne, Switzerland, 1995; pp. 514–519. [Google Scholar]
- Bello-Bello, J.J.; Schettino-Salomón, S.; Ortega-Espinoza, J.; Spinoso-Castillo, J.L. A temporary immersion system for mass micropropagation of pitahaya (Hylocereus undatus). 3 Biotech 2021, 11, 437. [Google Scholar] [CrossRef]
- Pérez-Alonso, N.; Jiménez, E.; Feria, M.D.; Capote, A.; Barbón, R.; Quiala, E.; Chávez, M. Potato microtubers using a temporary immersion system: Inoculum density, immersion time and field studies. Biotecnol. Veg. 2007, 7, 149–154. [Google Scholar]
- García-Ramírez, Y.; González-González, M.; García, S.; Freire-Seijo, M.; Pérez, M.; Trujillo, Á.; Barbon, R. Efecto de la densidad de inóculo sobre la morfología y fisiología de los brotes de Bambusa vulgaris Schrad. ex Wendl cultivados en sistema de immersión temporal. Biotecnol. Veg. 2016, 16, 231–237. [Google Scholar]
- Posada-Pérez, L.; Montesinos, Y.P.; Guerra, D.G.; Daniels, D.; Gómez-Kosky, R. Complete germination of papaya (Carica papaya L. cv. Maradol Roja) somatic embryos using temporary immersion system type RITA® and phloroglucinol in semi-solid culture medium. In Vitro Cell. Dev. Biol. Plant 2017, 53, 505–513. [Google Scholar] [CrossRef]
- Ducos, J.P.; Labbe, G.; Lambot, C.; Pétiard, V. Pilot scale process for the production of pre-germinated somatic embryos of selected robusta (Coffea canephora) clones. In Vitro Cell. Dev. Biol. Plant 2007, 43, 652–659. [Google Scholar] [CrossRef]
- Ptak, A.; Simlat, M.; Kwiecień, M.; Laurain-Mattar, D. Leucojum aestivum plants propagated in in vitro bioreactor culture and on solid media containing cytokinins. Eng. Life Sci. 2013, 13, 261–270. [Google Scholar] [CrossRef]
- Lambardi, M.; Roncasaglia, R.; Bujazha, D.; Baileiro, F.; Correira da Silva, D.; Ozudogru, E.A. Improvement of shoot proliferation by liquid culture in temporary immersion. In Proceedings of the 6th International Symposium on Production and Establishment of Micropropagated Plants, San Remo, Italy, 19–24 April 2015; ISHS. 2015. [Google Scholar]
- De Carlo, A.; Tarraf, W.; Lambardi, M.; Benelli, C. Temporary immersion system for production of biomass and bioactive compounds from medicinal plants. Agronomy 2021, 11, 2414. [Google Scholar] [CrossRef]
- Pavlov, A.; Bley, T. Betalains biosynthesis by Beta vulgaris L. hairy root culture in a temporary immersion cultivation system. Process Biochem. 2006, 41, 848–852. [Google Scholar] [CrossRef]
- Kokotkiewicz, A.; Bucinski, A.; Luczkiewicz, M. Xanthone, benzophenone and bioflavonoid accumulation in Cyclopia genistoides (L.) Vent. (honeybush) shoot cultures grown on membrane rafts and in a temporary immersion system. Plant Cell Tissue Organ Cult. 2015, 120, 373–378. [Google Scholar] [CrossRef]
- Kunakhonnuruk, B.; Kongbangkerd, A.; Inthima, P. Improving large-scale biomass and plumbagin production of Drosera communis A. St.-Hil. by temporary immersion system. Ind. Crops Prod. 2019, 137, 197–202. [Google Scholar] [CrossRef]
- Michoux, F.; Ahmad, N.; McCarthy, J.; Nixon, P.J. Contained and high-level production of recombinant protein in plant chloroplasts using a temporary immersion bioreactor. Plant Biotechnol. J. 2010, 9, 575–584. [Google Scholar] [CrossRef]
- Cuello, J.L.; Yue, L.C. Ebb-and-flow bioreactor regime and electrical elicitation: Novel strategies for hairy root biochemical production. Electron. J. Integr. Biosci. 2008, 3, 45–56. [Google Scholar]
- Steingroewer, J.; Bley, T.; Georgiev, V.; Ivanov, I.; Lenk, F.; Marchev, A.; Pavlov, A. Bioprocessing of differentiated plant in vitro systems. Eng. Life Sci. 2013, 13, 26–38. [Google Scholar] [CrossRef]
- Georgiev, V.; Schumann, A.; Pavlov, A.; Bley, T. Temporary immersion systems in plant biotechnology. Eng. Life Sci. 2014, 14, 607–621. [Google Scholar] [CrossRef]
- Tisserat, B.; Vandercook, C.E. Development of an automated plant culture system. Plant Cell Tissue Organ Cult. 1985, 5, 107–117. [Google Scholar] [CrossRef]
- Young, P.S.; Murthy, H.N.; Yoeup, P.K. Mass multiplication of protocorm-like bodies using bioreactor system and subsequent plant regeneration in Phalaenopsis. Plant Cell Tissue Organ Cult. 2000, 63, 67–72. [Google Scholar] [CrossRef]
- Rodrigues, P.H.V.; Teixeira, F.M.; Lima, A.M.L.P.; Ambrosano, G.M.B. Propagation of Heliconia plantlets in temporarily immersion bioreactor. Bragantia 2006, 65, 29–35. [Google Scholar] [CrossRef]
- Wu, R.Z.; Chakrabarty, D.; Hahn, E.J.; Paek, K.Y. Micropropagation of an endangered jewel orchid (Anoectochilus formosanus) using bioreactor system. Hortic. Environ. Biotechnol. 2007, 48, 376–380. [Google Scholar]
- Min, J.Y.; Jung, H.Y.; Kang, S.M.; Kim, Y.D.; Kang, Y.M.; Park, D.J.; Prasad, D.T.; Choi, M.S. Production of tropane alkaloids by small-scale bubble column bioreactor cultures of Scopolia parviflora adventitious roots. Bioresource Technol. 2007, 98, 1748–1753. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.A.; Wu, C.H.; Murthy, H.N.; Hahn, E.J.; Paek, K.Y. Application of an airlift bioreactor system for the production of adventitious root biomass and caffeic acid derivatives of Echinacea purpurea. Biotechnol. Bioprocess Eng. 2009, 14, 91–98. [Google Scholar] [CrossRef]
- Kim, E.K.; Hahn, E.J.; Murthy, H.N.; Paek, K.Y. Enhanced shoot and bulblet proliferation of garlic (Allium sativum L.) in bioreactor systems. J. Hortic. Sci. Biotechnol. 2004, 79, 818–822. [Google Scholar] [CrossRef]
- Piao, X.C.; Chakrabarty, D.; Hahn, E.J.; Paek, K.Y. A simple method for mass production of potato microtubers using a bioreactor system. Curr. Sci. 2003, 84, 1129–1132. [Google Scholar]
- Ekmekçigil, M.; Bayraktar, M.; Akkuş, Ö.; Gürel, A. High-frequency protocorm-like bodies and shoot regeneration through a combination of thin cell layer and RITA® temporary immersion bioreactor in Cattleya forbesii Lindl. Plant Cell Tissue Organ Cult. 2019, 136, 451–464. [Google Scholar] [CrossRef]
- Fritsche, Y.; Deola, F.; da Silva, D.A.; Holderbaum, D.F.; Guerra, M.P. Cattleya tigrina (Orchidaceae) in vitro regeneration: Main factors for optimal protocorm-like body induction and multiplication, plantlet regeneration, and cytogenetic stability. S. Afr. J. Bot. 2022, 149, 96–108. [Google Scholar] [CrossRef]
- Hempfling, T.; Preil, W. Application of a temporary immersion system in mass propagation of Phalaenopsis. In Liquid Culture Systems for in vitro Plant Propagation; Hvoslef-Eide, A.K., Preil, W., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 231–242. [Google Scholar]
- Pisowotzki, C.; Surminski, K.; Lieberei, R. Micropropagation of Phalaenopsis hybrids in temporary immersion system effects of exudated phenolic substances on plant development. Propag. Ornam. Plants 2008, 8, 221–223. [Google Scholar]
- Ramos-Castellá, A.; Iglesias-Andreu, L.G.; Bello-Bello, J.; Lee-Espinosa, H. Improved propagation of vanilla (Vanilla planifolia Jacks. ex Andrews) using a temporary immersion system. In Vitro Cell. Dev. Biol. Plant 2014, 50, 576–581. [Google Scholar] [CrossRef]
- Ramírez-Mosqueda, M.A.; Bello-Bello, J.J. SETIS™ bioreactor increases in vitro multiplication and shoot length in vanilla (Vanilla planifolia Jacks. Ex Andrews). Acta. Physiol. Plant. 2021, 43, 52. [Google Scholar] [CrossRef]
- Leyva-Ovalle, O.R.; Bello-Bello, J.J.; Murguía-González, J.; Núñez-Pastrana, R.; Ramírez-Mosqueda, M.A. Micropropagation of Guarianthe skinneri (Bateman) Dressler et WE Higging in temporary immersion systems. 3 Biotech 2020, 10, 26. [Google Scholar] [CrossRef] [PubMed]
- Kunakhonnuruk, B.; Inthima, P.; Kongbangkerd, A. In vitro propagation of rheophytic orchid, Epipactis flava Seidenf.—A comparison of semi-solid, continuous immersion and temporary immersion systems. Biology 2019, 8, 72. [Google Scholar] [CrossRef] [PubMed]
- Yoon, Y.J.; Murthy, H.N.; Hahn, E.J.; Paek, K.Y. Biomass production of Anoectochilus formosanus hayata in a bioreactor system. J. Plant Biol. 2007, 50, 573–576. [Google Scholar] [CrossRef]
- Zhang, B.; Niu, Z.; Li, C.; Hou, Z.; Xue, Q.; Liu, W.; Ding, X. Improving large-scale biomass and total alkaloid production of Dendrobium nobile Lindl. using a temporary immersion bioreactor system and MeJA elicitation. Plant Methods 2022, 18, 10. [Google Scholar] [CrossRef]
- Moreira, A.L.; Silva, A.B.; Santos, A.; Reis, C.O.; Landgraf, P.R.C. Cattleya walkeriana growth in different micropropagation systems. Revista Ciência Rural 2013, 43, 1804–1810. [Google Scholar] [CrossRef]
- Cui, H.Y.; Murthy, H.N.; Moh, S.H.; Cui, Y.Y.; Lee, E.J.; Paek, K.Y. Production of biomass and bioactive compounds in protocorm cultures of Dendrobium candidum Wall ex Lindl. using balloon type bubble bioreactors. Ind. Crops Prod. 2014, 53, 28–33. [Google Scholar] [CrossRef]
- Zhang, B.; Niu, Z.; Zhou, A.; Zhang, D.; Xue, Q.; Liu, W.; Chen, J.; Shen, J.; Ding, X. Micropropagation of Dendrobium nobile Lindl. plantlets by temporary immersion bioreactor. J. Biobased Mater. Bioenergy 2019, 13, 395–400. [Google Scholar] [CrossRef]
- Minh, T.V. Micropropagation of Mokara orchid by temporary immersion system technique. Int. J. Res. Sci. Innov. Appl. Sci. 2022, 7, 54–58. [Google Scholar] [CrossRef]
- Masnoddin, M.; Repin, R.; Aziz, Z.A. PLB regeneration of Paphiopedilum rothschildianum using callus and liquid culture system. J. Trop. Bio. Conserv. 2018, 15, 1–14. [Google Scholar] [CrossRef]
- Masnoddin, M.; Repin, R.; Aziz, Z.A. Micropropagation of an endangered borneo orchid, Paphiopedilum rothschildianum callus using temporary immersion bioreactor system. Thai J. Agric. Sci. 2016, 34, 161–171. [Google Scholar]
- Shen, R.S.; Hsu, S.T. Virus elimination through meristem culture and rapid clonal propagation using a temporary immersion system. In Orchid Propagation: From Laboratories to Greenhouses—Methods and Protocols; Springer Protocols Handbooks; Lee, Y.I., Yeung, E.T., Eds.; Humana Press: New York, NY, USA, 2018; pp. 267–282. [Google Scholar]
- Spinoso-Castillo, J.L.; Chavez-Santoscoy, R.A.; Bogdanchikova, N.; Pérez-Sato, J.A.; Morales-Ramos, V.; Bello-Bello, J.J. Antimicrobial and hormetic effects of silver nanoparticles on in vitro regeneration of vanilla (Vanilla planifolia Jacks. ex Andrews) using a temporary immersion system. Plant Cell Tissue Organ Cult. 2017, 129, 195–207. [Google Scholar] [CrossRef]
- Ramírez-Mosqueda, M.A.; Iglesias-Andreu, L.G.; Favián-Vega, E.; Teixeira da Silva, J.A.; Leyva-Ovalle, O.R.; Murguía-González, J. Morphogenetic stability of variegated Vanilla planifolia Jacks. plants micropropagated in a temporary immersion system (TIB®). Rend. Lincei Sci. Fis. Nat. 2019, 30, 603–609. [Google Scholar] [CrossRef]
- Esyanti, R.R.; Adhitama, N.; Manurung, R. Efficiency evaluation of Vanda tricolor growth in temporary immerse system bioreactor and thin layer culture system. J. Adv. Agric. Technol. 2016, 3, 63–66. [Google Scholar] [CrossRef]


| Orchid Species | Explant Type | Culture Systems | Type of Medium and Hormone Combinations Used | Medium Volume | Immersion Time and Frequency | Experimental Results | References |
|---|---|---|---|---|---|---|---|
| Cattleya tigrina A. Rich. ex Beer | Homogeneous shoots (≥5 mm) with leaves and adventitious roots | CIS (continuous immersion system) and TIS | Liquid MS fortified with 30 g L−1 sucrose and Morel vitamins (2 g L−1 Phytagel added for CIS) | 30 mL for CIS; 200 mL for TIS | - | The inoculated shoots gave rise to PLB directly and continued to proliferate without growth hormones. PLB multiplication significantly enhanced in TIS with 2-fold higher production (77.3 g) of PLBs) than those formed on the gelled medium of CIS (35.4 g PLBs). | [173] |
| Cattleya walkeriana Gardner | In vitro seedlings (1 cm long) | SSS (semi-solid system), liquid, CIS), and TIS | Liquid MS augmented with 1.34 μM NAA, 30 g L−1 of sucrose (6 g L−1 of agar added for solid medium). | 60 mL for solid and liquid medium; 600 mL for CIS and TIS. | An immersion period of 3 min every 90 min. | The longest aerial part (2.06 cm) and biggest fresh mass (0.032 g) of the propagated plant were observed in the TIS. Furthermore, the largest fresh mass was noticed in the CIS and TIS bioreactors due to the continual contact of the explants with the medium. The TIS performed best as compared to other culture systems. | [182] |
| Cymbidium sinense Willd | Rhizome segment | CIS and TIS | Medium containing g L−1 Hyponex I, 0.5 g L−1 Hyponex II, 1 g L−1 peptone, 2 mg L−1 BA, 0.2 m L−1 NAA, 0.2 mg L−1 AC and 30 g L−1 sucrose for CIS; medium with 2 g L−1 Hyponex I, 0.5 g L−1 Hyponex II, 1 g L−1 peptone, 4 mg L−1 BA, 0.2 mg L−1 NAA and 30 g L−1 sucrose for TIS. | 2000 mL for CIS and TIS. | 1 h immersion with a drying period of 1 h. | Shoot induction from rhizome failed in CIS, unlike TIS, which produced the best shooting response and plantlet generation in medium appended with 4 mg L−1 BAP and 0.2 mg L−1 NAA. The root formation rate was prominent (94.7%) in the medium enriched with 1.0 mg L−1 NAA. | [31] |
| Dendrobium candidum Wall ex Lindl. | PLBs from the nodal stem segment | Raft type (protocorms cultured on the net), CIS and TIS (ebb and flood) | ½ MS incorporated with 0.5 mg L−1 NAA, 2.5% (w/v) sucrose, 150 mg L−1 NaH2PO4 and 1% (v/v) banana homogenate. | 2000 mL | The fresh and dry biomass accumulation was highest (323.33 g L−1 and 16.13 g L−1) in CIS while it was least (270.60 g L−1 and 14.67 g L−1) with ebb and flood method demonstrating better protocorm growth in immersion cultures. Accumulation of bioactive compounds was maximum (polysaccharides—404.48 mg g−1 DW, coumarins—18.36 mg g−1 DW, polyphenolics—13.33 mg g−1 DW, and flavonoids—3.97 mg g−1 DW) in immersion cultures. An inoculum density of 50 g L−1 was appropriate for biomass and bioactive compound accumulation in cultures. | [183] | |
| Dendrobium nobile Lindl. | Protocorms from in vitro germinate seeds | SSS and TIS | The liquid ½ MS medium containing 0.5 mg L−1 NAA, 2% sucrose and 10% CW (0.7% agar added for SS culture) | 1000 mL | 5 min immersion for every 2, 4, 6, and 8 h duration. | The total fresh weight was the highest (302.85 g) with 6 h immersion frequency, while dry matter content (11.56%) was the maximum with 8 h immersion frequency. The longest shoot (72.83 mm), highest internode number (4.52), and largest stem diameter (4.05 mm) were achieved in 6 h of immersion frequency in the TIB. After acclimatization in the greenhouse, the shoot growth and plant survival rate were better with plants obtained from TIS than with the plants generated through SSS. | [184] |
| Epidendrum fulgens Brongn. | In vitro generated plantlets | Natural ventilation system (NV) and TIB system | Liquid MS appended with 3% sucrose for TIS (gelled media with 2 g L−1 phytagel poured into polypropylene containers containing PTFE filters allowing NV at a rate of 54 dm3 day−1 for NV culture system). | 400 mL of gelled media for NV system; 400 mL liquid media for TIB system | 3 min immersion every 3 h duration | The two culture systems (NV and TIBS) significantly affected plant growth and quality. The number of leaves, shoots, roots, and fresh weight was greater for plants developed in TIBS than those generated in the NV system. There was a 2-fold increase in root number for plants grown in TIBS compared to those in NV system, even though there was a significant impact on stomata number and photosynthetic pigment contents | [33] |
| Mokara Leuen Berger Gold | Callus tissue | SSS and TIS | MS incorporated with BA (0.5 mg L−1), B1 (5 mg L−1), adenin sulfate (10 mg L−1), peptone (1 g L−1), CW (10%) and sucrose (20 g L−1) for SSS; MS supplemented with CW (10%), sucrose (20 g L−1) and other growth regulators at different concentrations for TIS. | 10 m for SS and 250 mL for TIS | 1 min immersion time for 1 h interval | The callus proliferation was more prominent in TIS than SSS on MS supplemented with CW (30%), sucrose (30 g L−1), and 2.4 D (1 mg L−1). The shoots were rooted best on MS augmented with NAA (1 mg L−1), B1 (5 mg L−1), CW (10%), and sucrose (20 g L−1). | [185] |
| Paphiopedilum rothschildianum Rchb.f. | Callus induced from seeds and protocorm explants | SSS and TIS | ½ MS enriched with 0–22.6 µM 2,4-D and 4.54 µM TDZ for SS culture; ½ MS appended with 2.27 µM TDZ and 12.0 µM BAP for TIS. | 150 mL | Immersion time of 5 min after every 125 min | Callus proliferation in TIS produced a 3-fold increase in fresh weight compared to that cultured on SSS. Protocorm development from callus explant increased 3-fold in TIS with a regeneration capacity of 168 PLBs per gram calli. PLB regeneration capacity was enhanced further with increased sucrose concentration (15 µ to 58 µM) with the generation of 190 PLBs per gram calli. | [186] |
| Paphiopedilum rothschildianum Rchb.f. | Callus derived PLBs | SSS and TIS | MS supplemented with 4.54 μM TDZ singly or in association with 13.6 μM 2,4-D. | 150 mL | 5 min immersion after every 125 min. | Higher sucrose concentration promoted better PLB formation (4.0) on TIS, contrasted with greater PLBs formation in lower sucrose concentration in SSS. A 2-fold increase in PLB formation was observed on TIS compared to SSS, producing 168 PLBs per gram calli. Furthermore, the regeneration capacity in TIS enhanced to 190 PLBs per gram calli with higher sucrose concentration (58 mM). | [187] |
| Phalaenopsis | Shoot apical meristem | TIS | The liquid medium with 1.5 g L−1 Hyponex I, 0.1 mg L−1 NAA, 4 mg L−1 BAP, 200 mL L−1 CW and 20 g L−1 sucrose. | 250 mL | 10 min immersion every 4 h each time | The production of axillary shoots correlated with days in the culture. After 5 months of culture, a single virus-free short shoot segment generated around 200 plantlets on average. Shoots were separated into single and transferred to root initiation medium to get complete plantlets in 3–4 months. | [188] |
| Vanilla planifolia Jacks | In vitro shoots | Automated TIS (RITA) | MS supplemented with 2 mg L−1 BA with different concentrations of Argovit (0, 25, 50, 100 and 200 mg L−1). | 200 mL | 2 min per immersion with a time interval of every 4, 8, and 12 h. | Maximum shoots (14.89) per explant were recorded at Argovit doses of 50 mg L−1, while the least shoots (4.55) were observed at Argovit doses of 200 mg L−1. The highest shoot length (14.89 cm) was noticed at 50 mg L−1 of Argovit, and the shortest (0.82 ± 0.6 cm) was witnessed in Argovit doses of 200 mg L−1. Fresh weight was maximum (438.00 ± 18.42 mg) in shoots under 50 mg L−1 dose of Argovit and the lowest (143.80 ± 12.34 mg) was noted in 200 mg L−1 of Argovit. | [189] |
| Vanilla planifolia Jacks | Shoot nodal segments | TIS | MS basal medium supplemented with 30 g L−1 sucrose and 2.15 mg L−1 BA. | 25 mL of medium per explant. | 2 min immersion every 8 h. | The longest (2.79 cm) and maximum shoot number (9.15) per explant were recorded after the first subculture, and the least (4.57) shoots were achieved in the fourth subculture. 100% of the roots were rooted with 80% survival, but all showed variegation. 100% genetic uniformity was observed from molecular analysis with ISSR markers and morphological stability was evident from the heritability of leaf pigmentation. | [190] |
| Vanda tricolor Lindl. | In vitro grown shoots | Thin layer and TIS | MS basal medium fortified with 150 mL L−1 CW, and 30 g L−1 sucrose. | 250 mL for TIS and 5 mL for thin layer system | Immersion time of 5 min and 10 min every 12 h interval for TIS; incubated in continuous 120 rpm in a gyratory shaker for thin layer system. | The sugar utilization in the thin layer system was more (25.28%) than in the TIS bioreactor (6.31%), resulting in a higher growth rate and biomass accumulation. The largest biomass production was noticed in the thin layer system, with a growth rate of 0.056 cm per day. However, the ability to sustain shoot viability and survivability was higher in TIS compared to the thin layer system. | [191] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nongdam, P.; Beleski, D.G.; Tikendra, L.; Dey, A.; Varte, V.; EL Merzougui, S.; Pereira, V.M.; Barros, P.R.; Vendrame, W.A. Orchid Micropropagation Using Conventional Semi-Solid and Temporary Immersion Systems: A Review. Plants 2023, 12, 1136. https://doi.org/10.3390/plants12051136
Nongdam P, Beleski DG, Tikendra L, Dey A, Varte V, EL Merzougui S, Pereira VM, Barros PR, Vendrame WA. Orchid Micropropagation Using Conventional Semi-Solid and Temporary Immersion Systems: A Review. Plants. 2023; 12(5):1136. https://doi.org/10.3390/plants12051136
Chicago/Turabian StyleNongdam, Potshangbam, David G. Beleski, Leimapokpam Tikendra, Abhijit Dey, Vanlalrinchhani Varte, Soumaya EL Merzougui, Vania M. Pereira, Patricia R. Barros, and Wagner A. Vendrame. 2023. "Orchid Micropropagation Using Conventional Semi-Solid and Temporary Immersion Systems: A Review" Plants 12, no. 5: 1136. https://doi.org/10.3390/plants12051136
APA StyleNongdam, P., Beleski, D. G., Tikendra, L., Dey, A., Varte, V., EL Merzougui, S., Pereira, V. M., Barros, P. R., & Vendrame, W. A. (2023). Orchid Micropropagation Using Conventional Semi-Solid and Temporary Immersion Systems: A Review. Plants, 12(5), 1136. https://doi.org/10.3390/plants12051136







