Warming Scenarios and Phytophthora cinnamomi Infection in Chestnut (Castanea sativa Mill.)
Abstract
1. Introduction
2. Results
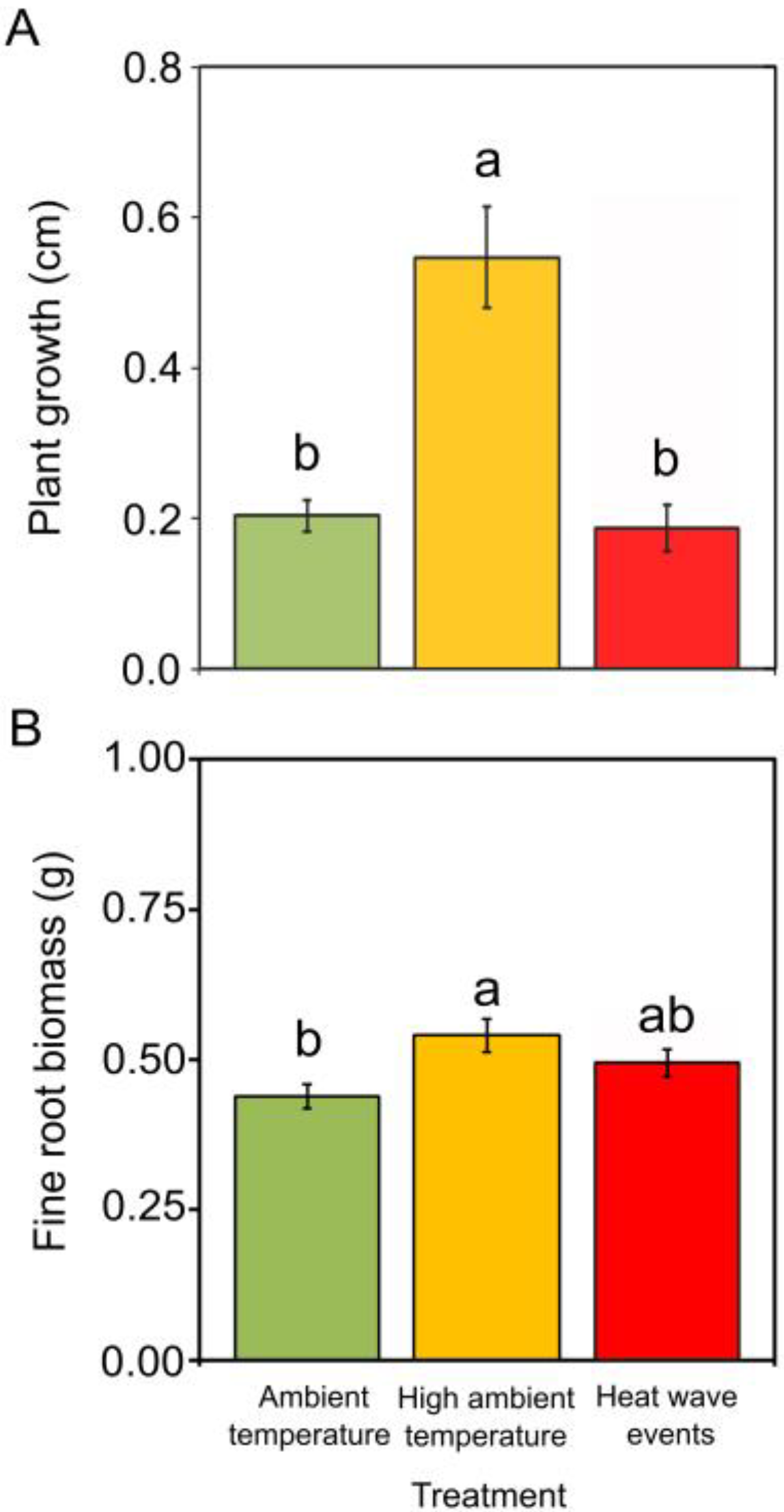
2.1. Warming Scenarios in Chestnut
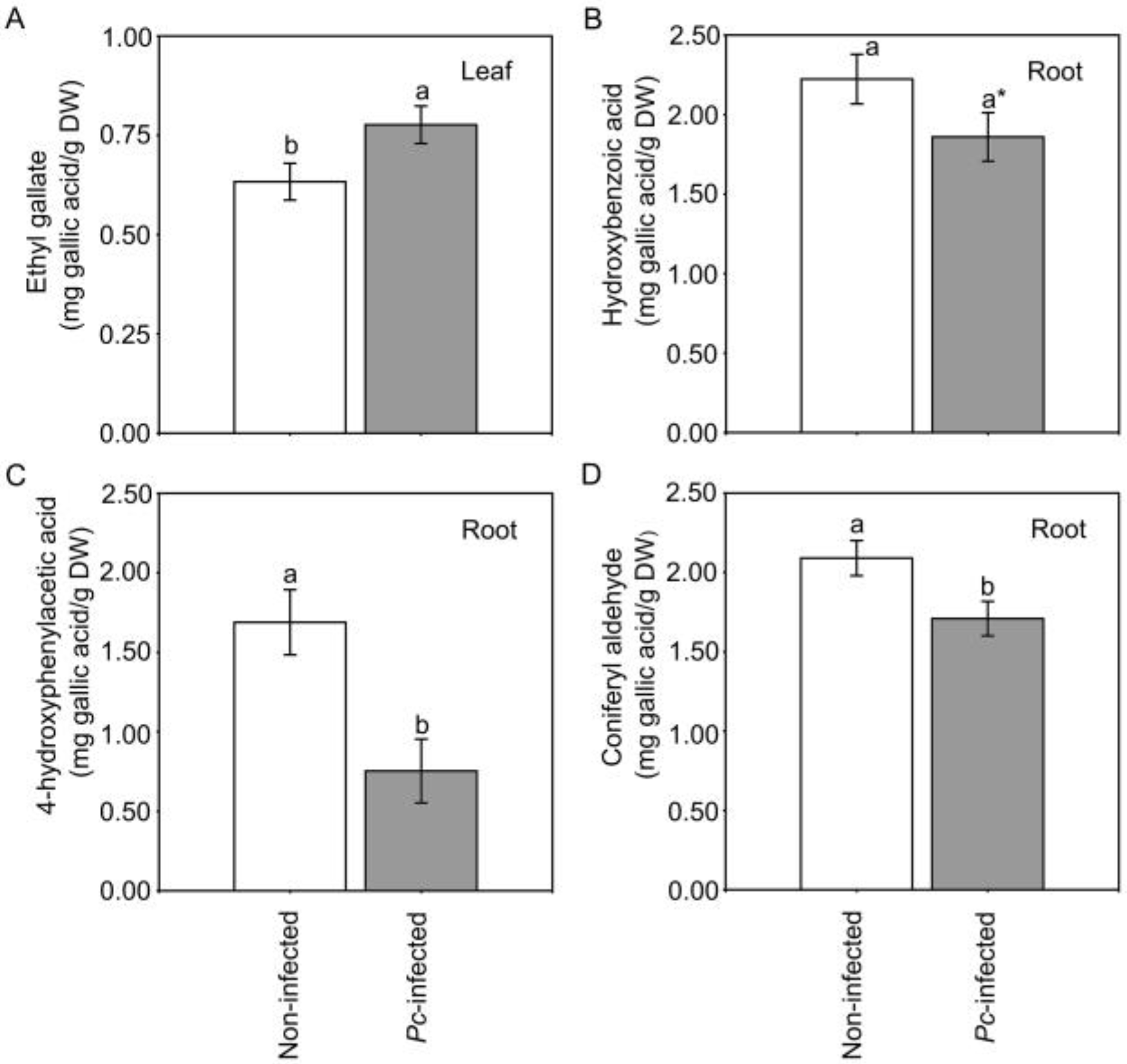
2.2. Warming Scenarios and Phytophthora cinnamomi Infection in Chestnut
3. Discussion
3.1. Effects of Temperature Increase in Chestnut
3.2. Phenolic Profile of Chestnut Seedlings after Infection by Pc
3.3. Enhanced Chestnut Resistance to Phytophthora cinnamomi after High Ambient Temperature
4. Materials and Methods
4.1. Plant Material
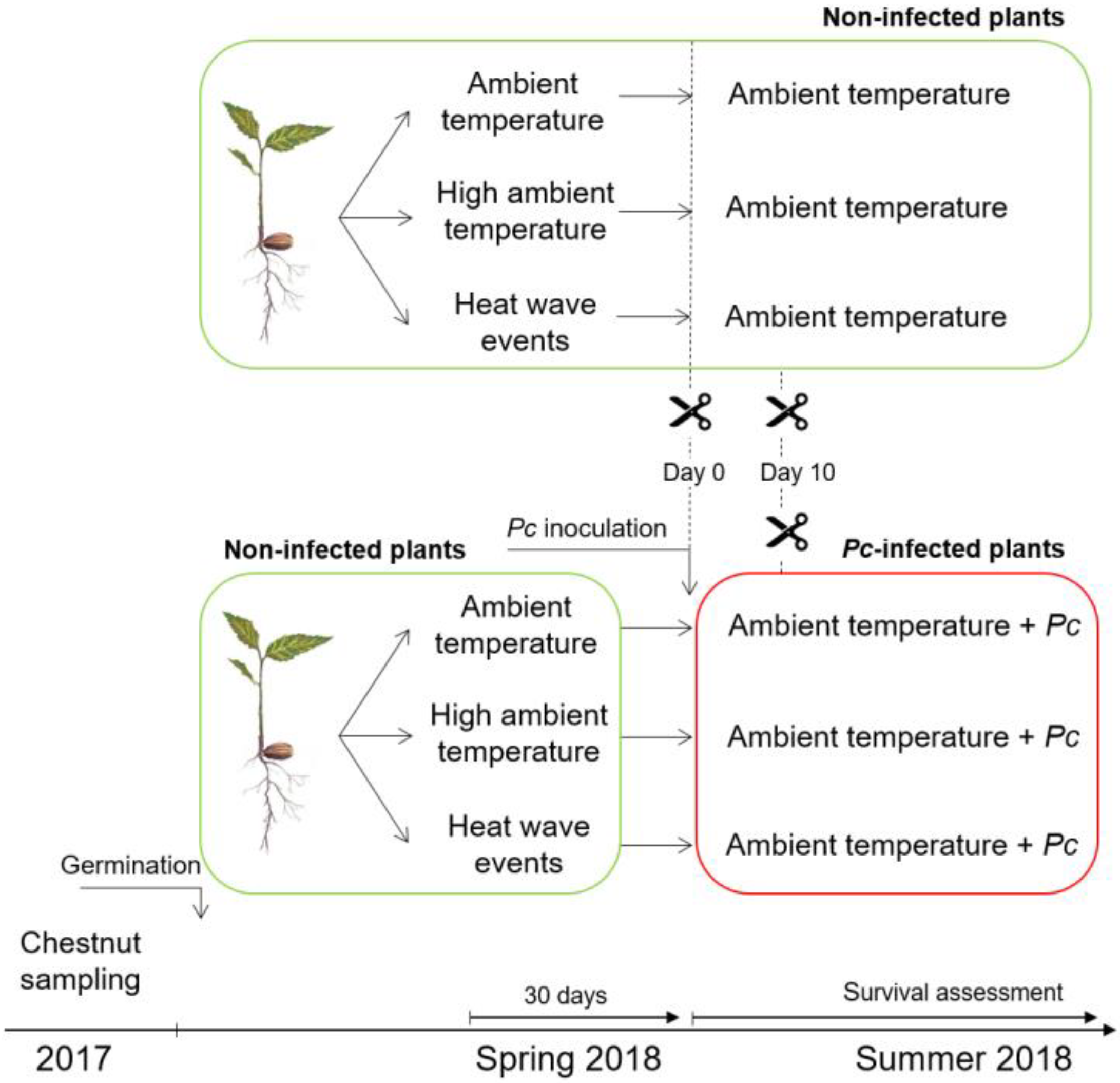
4.2. Treatments and Experimental Design
4.3. Inoculation of Phytophthora cinnamomi and Mortality Assessment
4.4. Plant Measurements and Sampling
4.5. Non-Targeted Phenolic Compound Profiling
4.6. Statistical Analysis
5. Conclusions
- Chestnut seedlings exposed to high ambient temperature (35 °C) showed the highest vigour in plant growth, fine root biomass and dynamic response of phenolic compounds to biotic stress. Plant mortality induced by Pc was 20% lower in chestnuts previously exposed to high ambient temperature (for 30 days) than in chestnuts previously exposed to ambient temperature. This result is encouraging for the future persistence of chestnut in the Mediterranean area, where temperatures are increasing and the presence of Pc is becoming more frequent.
- Two 45 °C heat waves for three days did not alter plant growth, fine root biomass or chestnut’s susceptibility to Pc. This suggests the good adaptation of chestnut to heat waves in the absence of water limitation.
- Pc was able to alter the physiology of C. sativa plants in response to temperature by homogenising the values of gas exchange parameters in leaves.
- In response to heat, changes in the phenolic compound profiles of chestnut plants exposed to high ambient temperature and heat waves were similar. However, during recovery, most phenolic compounds of plants exposed to high ambient temperature remained low, but, in plants subjected to heat waves, they increased. Changes in compounds were greater in leaves than in roots.
- Three phenolic compounds (ethyl gallate in leaves, 4-hydroxyphenylacetic acid in roots and coniferyl aldehyde in roots) showed significant variations in chestnut in response to Pc infection. Five additional phenolic compounds showed different changes in their content in response to Pc and the scenario that plants were exposed to before inoculation.
- Variation was observed in plasticity at the family level of several phenolic compounds in response to altered warming scenarios. This variation would be an opportunity for C. sativa to respond and probably adapt to global warming.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ben Rejeb, I.; Pastor, V.; Mauch-Mani, B. Plant responses to simultaneous biotic and abiotic stress: Molecular mechanisms. Plants 2014, 3, 458–475. [Google Scholar] [CrossRef] [PubMed]
- Rivero, R.M.; Mittler, R.; Blumwald, E.; Zandalinas, S.I. Developing climate-resilient crops: Improving plant tolerance to stress combination. Plant J. 2022, 109, 373–389. [Google Scholar] [CrossRef] [PubMed]
- Elvira-Recuenco, M.; Cacciola, S.O.; Sanz-Ros, A.V.; Garbelotto, M.; Aguayo, J.; Solla, A.; Mullett, M.; Drenkhan, T.; Oskay, F.; Kaya, A.G.A.; et al. Potential interactions between invasive Fusarium circinatum and other pine pathogens in Europe. Forests 2020, 11, 7. [Google Scholar] [CrossRef]
- Lombardero, M.J.; Solla, A.; Ayres, M.P. Pine Defenses against the pitch canker disease are modulated by a native insect newly associated with the invasive fungus. For. Ecol. Manag. 2019, 437, 253–262. [Google Scholar] [CrossRef]
- Desaint, H.; Aoun, N.; Deslandes, L.; Vailleau, F.; Roux, F.; Berthomé, R. Fight hard or die trying: When plants face pathogens under heat stress. New Phytol. 2021, 229, 712–734. [Google Scholar] [CrossRef]
- Milanović, S.; Mladenović, K.; Stojnić, B.; Solla, A.; Milenković, I.; Uremović, V.; Tack, A.J.M. Relationships between the pathogen Erysiphe alphitoides, the phytophagous mite Schizotetranychus garmani (Acari: Tetranychidae) and the predatory mite Euseius finlandicus (Acari: Phytoseiidae) in oak. Insects 2021, 12, 981. [Google Scholar] [CrossRef]
- Zolfaghari, R.; Dalvand, F.; Fayyaz, P.; Solla, A. Maternal drought stress on Persian oak (Quercus brantii Lindl.) affects susceptibility to single and combined drought and biotic stress in offspring. Environ. Exp. Bot. 2022, 194, 104716. [Google Scholar] [CrossRef]
- Atkinson, N.J.; Urwin, P.E. The interaction of plant biotic and abiotic stresses: From genes to the field. J. Exp. Bot. 2012, 63, 3523–3544. [Google Scholar] [CrossRef]
- Gomes Marques, I.; Solla, A.; David, T.S.; Rodríguez-González, P.M.; Garbelotto, M. Response of two riparian woody plants to Phytophthora species and drought. For. Ecol. Manag. 2022, 518, 120281. [Google Scholar] [CrossRef]
- Camisón, A.; Martín, M.Á.; Sánchez-Bel, P.; Flors, V.; Alcaide, F.; Morcuende, D.; Pinto, G.; Solla, A. Hormone and secondary metabolite profiling in chestnut during susceptible and resistant interactions with Phytophthora cinnamomi. J. Plant Physiol. 2019, 241, 153030. [Google Scholar] [CrossRef]
- Jan, R.; Asaf, S.; Numan, M.; Kim, K.M. Plant secondary metabolite biosynthesis and transcriptional regulation in response to biotic and abiotic stress conditions. Agronomy 2021, 11, 968. [Google Scholar] [CrossRef]
- Berini, J.L.; Brockman, S.A.; Hegeman, A.D.; Reich, P.B.; Muthukrishnan, R.; Montgomery, R.A.; Forester, J.D. Combinations of abiotic factors differentially alter production of plant secondary metabolites in five woody plant species in the Boreal-Temperate transition zone. Front. Plant Sci. 2018, 9, 1287. [Google Scholar] [CrossRef]
- Conrad, A.O.; McPherson, B.A.; Wood, D.L.; Madden, L.V.; Bonello, P. Constitutive phenolic biomarkers identify naïve Quercus agrifolia resistant to Phytophthora ramorum, the causal agent of sudden oak death. Tree Physiol. 2017, 37, 1686–1696. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, P.; Colavolpe, M.B.; Serrazina, S.; Costa, R.L. European and American chestnuts: An overview of the main threats and control efforts. Front. Plant Sci. 2022, 13, 951844. [Google Scholar] [CrossRef] [PubMed]
- Zaynab, M.; Fatima, M.; Abbas, S.; Sharif, Y.; Umair, M.; Zafar, M.H.; Bahadar, K. Role of secondary metabolites in plant defense against pathogens. Microb. Pathog. 2018, 124, 198–202. [Google Scholar] [CrossRef]
- IPCC Mediterranean region. In climate change 2022: Impacts, adaptation and vulnerability. In Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Pörtner, H.-O., Roberts, D.C., Tignor, M., Poloczanska, E.S., Mintenbeck, K., Alegría, A., Craig, M., Langsdorf, S., Löschke, S., Möller, V., et al., Eds.; Cambridge University Press: Cambridge, UK, 2022; pp. 2233–2272. [Google Scholar]
- Clark, P.W.; Freeman, A.J.; D’Amato, A.W.; Schaberg, P.G.; Hawley, G.J.; Evans, K.S.; Woodall, C.W. restoring a keystone tree species for the future: American chestnut assisted migration plantings in an adaptive silviculture experiment. For. Ecol. Manag. 2022, 523, 120505. [Google Scholar] [CrossRef]
- Dorado, F.J.; Solla, A.; Alcaide, F.; Martín, M.Á. Assessing heat stress tolerance in Castanea sativa. Forestry 2022, 5, 667–677. [Google Scholar] [CrossRef]
- Gustafson, E.J.; Miranda, B.R.; Dreaden, T.J.; Pinchot, C.C.; Jacobs, D.F. Beyond blight: Phytophthora root rot under climate change limits populations of reintroduced American chestnut. Ecosphere 2022, 13, e3917. [Google Scholar] [CrossRef]
- Castellana, S.; Martin, M.Á.; Solla, A.; Alcaide, F.; Villani, F.; Cherubini, M.; Neale, D.; Mattioni, C. Signatures of local adaptation to climate in natural populations of sweet chestnut (Castanea sativa Mill.) from southern Europe. Ann. For. Sci. 2021, 78, 27. [Google Scholar] [CrossRef]
- Freitas, T.R.; Santos, J.A.; Silva, A.P.; Martins, J.; Fraga, H. Climate change projections for bioclimatic distribution of Castanea sativa in Portugal. Agronomy 2022, 12, 1137. [Google Scholar] [CrossRef]
- Míguez-Soto, B.; Fernández-Cruz, J.; Fernández-López, J. Mediterranean and northern Iberian gene pools of wild Castanea sativa Mill. are two differentiated ecotypes originated under natural divergent selection. PLoS ONE 2019, 14, e0211315. [Google Scholar] [CrossRef]
- Míguez-Soto, B.; Fernández-López, J. Variation in adaptive traits among and within Spanish and European populations of Castanea sativa: Selection of trees for timber production. New For. 2015, 46, 23–50. [Google Scholar] [CrossRef]
- Alcaide, F.; Solla, A.; Mattioni, C.; Castellana, S.; Martín, M.A. Adaptive diversity and drought tolerance in Castanea sativa assessed through EST-SSR genic markers. Forestry 2019, 92, 287–296. [Google Scholar] [CrossRef]
- Camisón, A.; Martín, A.M.; Dorado, F.J.; Moreno, G.; Solla, A. Changes in carbohydrates induced by drought and waterlogging in Castanea sativa. Trees 2020, 34, 579–591. [Google Scholar] [CrossRef]
- Camisón, Á.; Martín, M.Á.; Flors, V.; Sánchez-Bel, P.; Pinto, G.; Vivas, M.; Rolo, V.; Solla, A. Exploring the use of scions and rootstocks from xeric areas to improve drought tolerance in Castanea sativa Miller. Environ. Exp. Bot. 2021, 187, 104467. [Google Scholar] [CrossRef]
- Ciordia, M.; Feito, I.; Pereira-lorenzo, S.; Fernández, A.; Majada, J. Adaptive diversity in Castanea sativa Mill. half-sib progenies in response to drought stress. Environ. Exp. Bot. 2012, 78, 56–63. [Google Scholar] [CrossRef]
- Dorado, F.J.; Pinto, G.C.; Monteiro, P.; Chaves, N.; Alías, J.C.; Rodrigo, S.; Camisón, Á.; Solla, A. Heat stress and recovery effects on the physiology and biochemistry of Castanea sativa Mill. Front. For. Glob. Chang. 2023, 5, 1072661. [Google Scholar] [CrossRef]
- Maurel, M.; Robin, C.; Simonneau, T.; Loustau, D.; Dreyer, E.; Desprez-Loustau, M.-L. Stomatal conductance and root-to-shoot signalling in chestnut saplings exposed to Phytophthora cinnamomi or partial soil drying. Funct. Plant Biol. 2004, 31, 41–51. [Google Scholar] [CrossRef]
- Hardham, A.R.; Blackman, L.M. Phytophthora cinnamomi . Mol. Plant Pathol. 2018, 19, 260–285. [Google Scholar] [CrossRef] [PubMed]
- Serrazina, S.; Santos, C.; Machado, H.; Pesquita, C.; Vicentini, R.; Pais, M.S.; Sebastiana, M.; Costa, R. Castanea root transcriptome in response to Phytophthora cinnamomi challenge. Tree Genet. Genomes 2015, 11, 6. [Google Scholar] [CrossRef]
- Vettraino, A.M.; Morel, O.; Perlerou, C.; Robin, C.; Diamandis, S.; Vannini, A. Occurrence and distribution of Phytophthora species in European chestnut stands, and their association with Ink disease and crown decline. Eur. J. Plant Pathol. 2005, 111, 169–180. [Google Scholar] [CrossRef]
- Jung, T.; Pérez-Sierra, A.; Durán, A.; Jung, M.H.; Balci, Y.; Scanu, B. Canker and decline diseases caused by soil- and airborne Phytophthora species in forests and woodlands. Persoonia 2018, 40, 182–220. [Google Scholar] [CrossRef] [PubMed]
- Gomes-Laranjo, J.; Araújo-Alves, J.; Ferreira-Cardoso, J.; Pimentel-Pereira, M.; Abreu, C.G.; Torres-Pereira, J. Effect of chestnut Ink disease on photosynthetic performance. J. Phytopathol. 2004, 152, 138–144. [Google Scholar] [CrossRef]
- Alcaide, F.; Solla, A.; Cherubini, M.; Mattioni, C.; Cuenca, B.; Camisón, Á.; Martín, M.Á. Adaptive evolution of chestnut forests to the impact of Ink disease in Spain. J. Syst. Evol. 2020, 58, 504–516. [Google Scholar] [CrossRef]
- Camisón, Á.; Martín, M.Á.; Oliva, J.; Elfstrand, M.; Solla, A. Increased tolerance to Phytophthora cinnamomi in offspring of ink-diseased chestnut (Castanea sativa Miller) trees. Ann. For. Sci. 2019, 76, 119. [Google Scholar] [CrossRef]
- Fernandes, P.; Machado, H.; Silva, M.C.; Costa, R.L. A histopathological study reveals new insights into responses of chestnut (Castanea spp.) to root infection by Phytophthora cinnamomi. Phytopathology 2021, 111, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Oliveras, J.; Ramis, X.; Caballol, M.; Serrad, F. Distribution of Phytophthora species within recreational chestnut, beech and cork oak forests. For. Ecol. Manag. 2023, 529, 120674. [Google Scholar]
- Santos, C.; Zhebentyayeva, T.; Serrazina, S.; Nelson, C.D.; Costa, R.; Ky, A. Development and characterization of EST-SSR markers for mapping reaction to Phytophthora cinnamomi in Castanea spp. Sci. Hortic. 2015, 194, 181–187. [Google Scholar] [CrossRef]
- Serrazina, S.; Machado, H.; Costa, R.L.; Duque, P.; Malhó, R. Expression of Castanea crenata allene oxide synthase in Arabidopsis improves the defense to Phytophthora cinnamomi. Front. Plant Sci. 2021, 12, 628697. [Google Scholar] [CrossRef] [PubMed]
- Saiz-Fernández, I.; Milenković, I.; Berka, M.; Černý, M.; Tomšovský, M.; Brzobohatý, B.; Kerchev, P. Integrated proteomic and metabolomic profiling of Phytophthora cinnamomi attack on sweet chestnut (Castanea sativa) reveals distinct molecular reprogramming proximal to the infection site and away from it. Int. J. Mol. Sci. 2020, 21, 8525. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, C.; Fang, Z.; Wu, Q.; Xu, Y.; Gong, B.; Jiang, X.; Lai, J.; Fan, J. A review of the stress resistance, molecular breeding, health benefits, potential food products, and ecological value of Castanea mollissima. Plants 2022, 11, 2111. [Google Scholar] [CrossRef] [PubMed]
- Martins, L.M.; Oliveira, M.T.; Abreu, C.G. Soils and climatic characteristic of chestnut stands that differ on the presence of the Ink disease. Acta Hortic. 1999, 494, 447–450. [Google Scholar] [CrossRef]
- Burgess, T.I.; Scott, J.K.; Mcdougall, K.L.; Stukely, M.J.C.; Crane, C.; Dunstan, W.A.; Brigg, F.; Andjic, V.; White, D.; Rudman, T.; et al. Current and projected global distribution of Phytophthora cinnamomi, one of the world’s worst plant pathogens. Glob. Chang. Biol. 2017, 23, 1661–1674. [Google Scholar] [CrossRef]
- Davison, E.M. Are jarrah (Eucalyptus marginata) trees killed by Phytophthora cinnamomi or waterlogging? Aust. For. 1997, 60, 116–124. [Google Scholar] [CrossRef]
- Corcobado, T.; Cech, T.L.; Brandstetter, M.; Daxer, A.; Hüttler, C.; Kudláček, T.; Jung, M.H.; Jung, T. Decline of European beech in Austria: Involvement of Phytophthora spp. and contributing biotic and abiotic factors. Forests 2020, 11, 895. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Férriz, M.; García-Garrido, S.; Aguín, O.; Elvira-Recuenco, M.; Hernandez-Escribano, L.; Martin-Benito, D.; Raposo, R. Synergistic abiotic and biotic stressors explain widespread decline of Pinus pinaster in a mixed forest. Sci. Total Environ. 2019, 685, 963–975. [Google Scholar] [CrossRef]
- Umami, M.; Parker, L.M.; Arndt, S.K. The impacts of drought stress and Phytophthora cinnamomi infection on short-term water relations in two year-old Eucalyptus obliqua. Forests 2021, 12, 109. [Google Scholar] [CrossRef]
- Gómez, F.J.R.; Pérez-de-Luque, A.; Sánchez-Cuesta, R.; Quero, J.L.; Cerrillo, R.M.N. Differences in the response to acute drought and Phytophthora cinnamomi Rands infection in Quercus ilex L. seedlings. Forests 2018, 9, 634. [Google Scholar] [CrossRef]
- San-Eufrasio, B.; Castillejo, M.Á.; Labella-Ortega, M.; Ruiz-Gómez, F.J.; Navarro-Cerrillo, R.M.; Tienda-Parrilla, M.; Jorrín-Novo, J.V.; Rey, M.D. Effect and response of Quercus ilex subsp. ballota [Desf.] Samp. seedlings from three contrasting Andalusian populations to individual and combined Phytophthora cinnamomi and drought stresses. Front. Plant Sci. 2021, 12, 722802. [Google Scholar] [CrossRef]
- Way, D.A.; Oren, R. Differential responses to changes in growth temperature between trees from different functional groups and biomes: A Review and synthesis of data. Tree Physiol. 2010, 30, 669–688. [Google Scholar] [CrossRef] [PubMed]
- Luo, H.; Xu, H.; Chu, C.; He, F.; Fang, S. High temperature can change root system architecture and intensify root interactions of plant seedlings. Front. Plant Sci. 2020, 11, 160. [Google Scholar] [CrossRef] [PubMed]
- Saxe, H.; Cannell, M.G.R.; Johnsen, Ø.; Ryan, M.G.; Vourlitis, G. Tree and forest functioning in response to global warming. New Phytol. 2001, 149, 369–399. [Google Scholar]
- Berry, J.; Bjorkman, O. Photosynthetic response and adaptation to temperature in higher plants. Annu. Rev. Plant Physiol. 1980, 31, 491–543. [Google Scholar] [CrossRef]
- Tarvainen, L.; Wittemann, M.; Mujawamariya, M.; Manishimwe, A.; Zibera, E.; Ntirugulirwa, B.; Ract, C.; Manzi, O.J.L.; Andersson, M.X.; Spetea, C.; et al. Handling the heat–photosynthetic thermal stress in tropical trees. New Phytol. 2022, 233, 236–250. [Google Scholar] [CrossRef] [PubMed]
- Virlouvet, L.; Fromm, M. Physiological and transcriptional memory in guard cells during repetitive dehydration stress. New Phytol. 2015, 205, 596–607. [Google Scholar] [CrossRef]
- Amarowicz, R.; Weidner, S.; Wójtowicz, I.; Karamac, M.; Kosińska, A.; Rybarczyk, A. Influence of low-temperature stress on changes in the composition of grapevine leaf phenolic compounds and their antioxidant properties. Funct. Plant Sci. Biotechnol. 2010, 4, 90–96. [Google Scholar]
- Weidner, S.; Brosowska-Arendt, W.; Szczechura, W.; Karamać, M.; Kosińska, A.; Amarowicz, R. Effect of osmotic stress and post-stress recovery on the content of phenolics and properties of antioxidants in germinating seeds of grapevine Vitis california. Acta Soc. Bot. Pol. 2011, 80, 11–19. [Google Scholar] [CrossRef]
- Król, A.; Amarowicz, R.; Weidner, S. Changes in the composition of phenolic compounds and antioxidant properties of grapevine roots and leaves (Vitis vinifera L.) under continuous of long-term drought stress. Acta Physiol. Plant. 2014, 36, 1491–1499. [Google Scholar] [CrossRef]
- Chaves, I.; Passarinho, J.A.P.; Capitão, C.; Chaves, M.M.; Fevereiro, P.; Ricardo, C.P.P. Temperature stress effects in Quercus suber leaf metabolism. J. Plant Physiol. 2011, 168, 1729–1734. [Google Scholar] [CrossRef]
- Weidner, S.; Kordala, E.; Brosowska-Arendt, W.; Karamać, M.; Kosińska, A.; Amarowicz, R. Phenolic compounds and properties of antioxidants in grapevine roots (Vitis vinifera L.) under low-temperature stress followed by recovery. Acta Soc. Bot. Pol. 2009, 78, 279–286. [Google Scholar] [CrossRef]
- Lukić, N.; Kukavica, B.; Davidović-Plavšić, B.; Hasanagić, D.; Walter, J. Plant stress memory is linked to high levels of anti-oxidative enzymes over several weeks. Environ. Exp. Bot. 2020, 178, 104166. [Google Scholar] [CrossRef]
- Correia, B.; Hancock, R.D.; Amaral, J.; Gomez-Cadenas, A.; Valledor, L.; Pinto, G. Combined drought and heat activates protective responses in Eucalyptus globulus that are not activated when subjected to drought or heat stress alone. Front. Plant Sci. 2018, 9, 819. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.A.; Solla, A.; Domingues, M.R.; Coimbra, M.A.; Gil, L. Exogenous phenol increase resistance of Ulmus minor to Dutch Elm disease through formation of suberin-like compounds on xylem tissues. Environ. Exp. Bot. 2008, 64, 97–104. [Google Scholar] [CrossRef]
- Kumar, S.; Abedin, M.M.; Singh, A.K.; Das, S. Role of Phenolic Compounds in plant-defensive mechanisms. In Plant Phenolics in Sustainable Agriculture; Lone, R., Shuab, R., Kamili, A.N., Eds.; Springer: Singapore, 2020; pp. 517–532. [Google Scholar]
- Goupil, P.; Benouaret, R.; Richard, C. Ethyl gallate displays elicitor activities in Tobacco plants. J. Agric. Food Chem. 2017, 65, 9006–9012. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.A.; Solla, A.; Witzell, J.; Gil, L.; García-Vallejo, M.C. Antifungal effect and reduction of Ulmus minor symptoms to Ophiostoma novo-ulmi by carvacrol and salicylic acid. Eur. J. Plant Pathol. 2010, 127, 21–32. [Google Scholar] [CrossRef]
- Widhalm, J.R.; Dudareva, N. A Familiar ring to it: Biosynthesis of plant benzoic acids. Mol. Plant 2015, 8, 83–97. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Zhang, X.; Fernando, W.G.D. Directing trophic divergence in plant-pathogen interactions: Antagonistic phytohormones with no doubt? Front. Plant Sci. 2020, 11, 600063. [Google Scholar] [CrossRef]
- Redondo, M.Á.; Pérez-Sierra, A.; Abad-Campos, P.; Torres, L.; Solla, A.; Reig-Armiñana, J.; García-Breijo, F. Histology of Quercus ilex roots during infection by Phytophthora cinnamomi. Trees 2015, 29, 1943–1957. [Google Scholar] [CrossRef]
- Van den Berg, N.; Swart, V.; Backer, R.; Fick, A.; Wienk, R.; Engelbrecht, J.; Prabhu, S.A. Advances in understanding defense mechanisms in Persea americana against Phytophthora cinnamomi. Front. Plant Sci. 2021, 12, 636339. [Google Scholar] [CrossRef] [PubMed]
- Humphreys, J.M.; Hemm, M.R.; Chapple, C. New routes for lignin biosynthesis defined by biochemical characterization of recombinant ferulate 5-hydroxylase, a multifunctional cytochrome P450-dependent monooxygenase. Proc. Natl. Acad. Sci. USA 1999, 96, 10045–10050. [Google Scholar] [CrossRef] [PubMed]
- Boudet, A.M. Lignins and lignification: Selected issues. Plant Physiol. Biochem. 2000, 38, 81–96. [Google Scholar] [CrossRef]
- Dodge, A.G.; Wackett, L.P. Metabolism of bismuth subsalicylate and intracellular accumulation of bismuth by Fusarium sp. strain BI. Appl. Environ. Microbiol. 2005, 71, 876–882. [Google Scholar] [CrossRef] [PubMed]
- Lanoue, A.; Burlat, V.; Henkes, G.J.; Koch, I.; Schurr, U.; Röse, U.S.R. De novo biosynthesis of defense root exudates in response to Fusarium attack in barley. New Phytol. 2010, 185, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Aguayo, J.; Elegbede, F.; Husson, C.; Saintonge, F.-X.; Marçais, B. Modeling climate impact on an emerging disease, the Phytophthora alni-induced alder decline. Glob. Chang. Biol. 2014, 20, 3209–3221. [Google Scholar] [CrossRef] [PubMed]
- Martín-García, J.; Solla, A.; Corcobado, T.; Siasou, E.; Woodward, S. Influence of temperature on germination of Quercus ilex in Phytophthora cinnamomi, P. gonapodyides, P. quercina and P. psychrophila infested soils. For. Pathol. 2015, 45, 215–223. [Google Scholar] [CrossRef]
- Serrano, M.S.; Romero, M.Á.; Homet, P.; Gómez-Aparicio, L. Climate change impact on the population dynamics of exotic pathogens: The case of the worldwide pathogen Phytophthora cinnamomi. Agric. For. Meteorol. 2022, 322, 109002. [Google Scholar] [CrossRef]
- Garbelotto, M.; Schmidt, D.; Popenuck, T. Pathogenicity and infectivity of Phytophthora ramorum vary depending on host species, infected plant part, inoculum potential, pathogen genotype, and temperature. Plant Pathol. 2021, 70, 287–304. [Google Scholar] [CrossRef]
- Gea-Izquierdo, G.; Natalini, F.; Cardillo, E. Holm oak death is accelerated but not sudden and expresses drought legacies. Sci. Total Environ. 2021, 754, 141793. [Google Scholar] [CrossRef] [PubMed]
- Encinas-Valero, M.; Esteban, R.; Hereş, A.M.; Vivas, M.; Fakhet, D.; Aranjuelo, I.; Solla, A.; Moreno, G.; Curiel Yuste, J. Holm oak decline is determined by shifts in fine root phenotypic plasticity in response to belowground stress. New Phytol. 2022, 235, 2237–2251. [Google Scholar] [CrossRef]
- Martínez-Arias, C.; Witzell, J.; Solla, A.; Martin, J.A.; Rodríguez-Calcerrada, J. Beneficial and pathogenic plant-microbe interactions during flooding stress. Plant Cell Environ. 2022, 45, 2875–2897. [Google Scholar] [CrossRef] [PubMed]
- Homet, P.; González, M.; Matías, L.; Godoy, O.; Pérez-Ramos, I.M.; García, L.V.; Gómez-Aparicio, L. Exploring interactive effects of climate change and exotic pathogens on Quercus suber performance: Damage caused by Phytophthora cinnamomi varies across contr asting scenarios of soil moisture. Agric. For. Meteorol. 2019, 276–277, 107605. [Google Scholar] [CrossRef]
- Dinis, L.T.; Peixoto, F.; Zhang, C.; Martins, L.; Costa, R.; Gomes-Laranjo, J. Physiological and biochemical changes in resistant and sensitive chestnut (Castanea) plantlets after inoculation with Phytophthora cinnamomi. Physiol. Mol. Plant Pathol. 2011, 75, 146–156. [Google Scholar] [CrossRef]
- Maurel, M.; Robin, C.; Capdevielle, X.; Loustau, D.; Desprez-Loustau, M.L. Effects of variable root damage caused by Phytophthora cinnamomi on water relations of chestnut saplings. Ann. For. Sci. 2001, 58, 639–651. [Google Scholar] [CrossRef]
- Bourgine, B.; Guihur, A. Heat shock signaling in land plants: From plasma membrane sensing to the transcription of small heat shock proteins. Front. Plant Sci. 2021, 12, 710801. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xu, F.; Shao, Y.; He, J. Regulatory mechanisms of heat stress response and thermomorphogenesis in plants. Plants 2022, 11, 3410. [Google Scholar] [CrossRef]
- Lindquist, S.; Craig, E.A. The Heat-Shock Proteins. Annu. Rev. Genet. 1988, 22, 631–677. [Google Scholar] [CrossRef]
- Pereira-Lorenzo, S.; Ramos-Cabrer, A.M.; Barreneche, T.; Mattioni, C.; Villani, F.; Díaz-Hernández, B.; Martín, L.M.; Robles-Loma, A.; Cáceres, Y.; Martín, A. Instant domestication process of European chestnut cultivars. Ann. Appl. Biol. 2019, 174, 74–85. [Google Scholar] [CrossRef]
- IPCC Summary for policymakers. IPCC Summary for policymakers. In climate change 2013: The physical science basis. In Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Stocker, T.F., Qin, D., Plattner, G.-K., Tignor, M., Allen, S.K., Boschung, J., Nauels, A., Xia, Y., Bex, V., Midgley, P.M., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2013; p. 20. [Google Scholar]
- Molina, M.O.; Sánchez, E.; Gutiérrez, C. Future heat waves over the Mediterranean from an Euro-CORDEX regional climate model ensemble. Sci. Rep. 2020, 10, 8801. [Google Scholar] [CrossRef] [PubMed]
- Camisón, A.; Martín, M.A.; Sánchez-Bel, P.; Flors, V.; Cubera, E.; Solla, A. Effect of grafting on phenology, susceptibility to Phytophthora cinnamomi and hormone profile of chestnut. Sci. Hortic. 2023, 311, 111789. [Google Scholar] [CrossRef]
- Jung, T.; Blaschke, H.; Neumann, P. Isolation, identification and pathogenicity of Phytophthora species from declining oak stands. Eur. J. For. Pathol. 1996, 26, 253–272. [Google Scholar] [CrossRef]
- Solla, A.; Aguín, O.; Cubera, E.; Sampedro, L.; Mansilla, J.P.; Zas, R. Survival time analysis of Pinus pinaster inoculated with Armillaria ostoyae: Genetic variation and relevance of seed and root traits. Eur. J. Plant Pathol. 2011, 130, 477–488. [Google Scholar] [CrossRef]
- StatSoft Inc. STATISTICA. In Data Analysis Software System; StatSoft, Inc.: Tulsa, OK, USA, 2011. [Google Scholar]





| Class | Subclass | Leaf | Root |
|---|---|---|---|
| Phenols | Hydroxybenzoic acids | Ethyl gallate | Hydroxybenzoic acid |
| Ellagic acid acetyl-xyloside | Gallic acid | ||
| Ellagic acid | Ellagic acid | ||
| Hydroxycinnamic acid | 3-feruloylquinic acid | ||
| Hydroxyphenylacetic acid | 4-hydroxyphenylacetic acid | ||
| Lignans | Lariciresinol | ||
| Flavonoids | Flavanols | Procyanidin | |
| Catechin | |||
| Flavonols | Miquelianin (quercetin 3-O-glucuronide) | Quercetin 3-O-galactoside | |
| Quercetin 3-O-rutinoside | Kaempferol-3-O-(6″ acetyl) glucoside 7-O rhamnoside | ||
| Quercetin 3-O-rhamnoside | |||
| Quercetin 3-O-galactoside | |||
| Other polyphenols | Hydroxytyrosol | Tyrosol | |
| Hydroxytyrosol acetate | Coniferyl aldehyde (4-hydroxy-3-methoxycinnamaldehyde) |
| Leaf | Root | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Effects | Df | Ethyl Gallate | Ellagic Acid Acetyl-xyloside | Ellagic Acid | Lariciresinol | Procyanidin | Catechin | Quercetin 3-O-glucuronide | Hydroxytyrosol Acetate | Ellagic Acid | Hydroxybenzoic Acid | Coniferyl Aldehyde |
| Scenario [S] | 2 | 0.012 | 0.004 | 0.006 | 0.019 | 0.001 | 0.002 | 0.009 | 0.039 | 0.015 | <0.001 | <0.001 |
| Mother tree [M] | 1 | * | ns | ns | ns | ns | ns | 0.034 | ns | ns | ns | 0.007 |
| Time [T] | 1 | 0.012 | 0.022 | ns | ns | 0.035 | ns | ns | 0.014 | ns | ns | ns |
| S × M | 2 | * | ns | ns | ns | ns | ns | 0.007 | ns | ns | ns | ns |
| S × T | 2 | 0.029 | 0.037 | * | ns | 0.006 | ns | <0.001 | 0.015 | 0.002 | 0.002 | 0.022 |
| M × T | 1 | * | ns | ns | ns | 0.046 | ns | ns | ns | * | ns | ns |
| S × M × T | 2 | ns | ns | ns | ns | ns | ns | ns | ns | 0.005 | 0.005 | 0.022 |
| Seed weight (g) | 1 | 0.028 | ns | ns | ns | 0.014 | ns | ns | ns | ns | ns | ns |
| Time to emerge (d) | 1 | ns | ns | ns | ns | ns | ns | 0.017 | 0.017 | ns | ns | ns |
| Plant height (cm) | 1 | 0.021 | ns | ns | ns | ns | ns | ns | ns | ns | ns | ns |
| Non-Infected Plants | |||||
|---|---|---|---|---|---|
| Day 0 | Day 10 | ||||
| Organ | Compound | High Ambient Temperature | Heat Wave Events | High Ambient Temperature | Heat Wave Events |
| Leaf | Ethyl gallate (mg gallic acid/g DW) | ns | ns | ns | ↑↑ |
| Ellagic acid acetyl-xyloside (mg gallic acid/g DW) | ↓↓ | ↓↓ | ↓↓ | ns | |
| Lariciresinol (mg gallic acid/g DW) | ↓ | ↓ | ↓↓ | ns | |
| Procyanidin (mg procyanidin/g DW) | ns | ns | ↓↓ | ↑↑ | |
| Catechin (mg catechin/g DW) | ↓↓ | ns | ↓↓ | ns | |
| Miquelianin (quercetin 3-O-glucuronide) (mg quercetin/g DW) | ns | ns | ns | ↑↑ | |
| Hydroxytyrosol acetate (mg gallic acid/g DW) | ↓↓ | ↓↓ | ns | ↑ | |
| Ellagic acid (mg ellagic acid/g DW) | ns | ns | ns | ↑↑ | |
| Root | Ellagic acid (mg ellagic acid/g DW) | ns | ns | ↑↑ | ns |
| Hydroxybenzoic acid (mg gallic acid/g DW) | ns | ns | ↓↓ | ↓↓ | |
| Coniferyl aldehyde (mg gallic acid/g DW) | ns | ns | ↓↓ | ns | |
| Leaf | Root | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Effect | Df | Ethyl Gallate | Ellagic Acid | 3-feruloylquinic Acid | Miquelianin | Hydroxytyrosol Acetate | Hydroxybenzoic Acid | 4-hydroxyphenylacetic Acid | Coniferyl Aldehyde |
| Phytophthora [Pc] | 1 | 0.032 | ns | ns | ns | ns | * | 0.006 | 0.014 |
| Scenario [S] | 2 | 0.018 | ns | 0.038 | 0.015 | <0.001 | <0.001 | 0.009 | <0.001 |
| Mother tree [M] | 1 | ns | ns | ns | * | ns | ns | ns | 0.031 |
| Pc × S | 2 | 0.016 | 0.014 | 0.044 | 0.005 | <0.001 | 0.003 | * | <0.001 |
| Pc × M | 1 | ns | ns | ns | 0.037 | 0.015 | ns | ns | ns |
| S × M | 2 | ns | ns | ns | ns | <0.001 | ns | ns | ns |
| Pc × S × M | 2 | ns | ns | ns | ns | ns | 0.031 | ns | * |
| Seed weight (g) | 1 | ns | ns | ns | ns | ns | ns | ns | ns |
| Time to emerge (d) | 1 | ns | ns | * | ns | 0.002 | ns | ns | ns |
| Plant height (cm) | 1 | ns | ns | ns | ns | ns | ns | ns | * |
| Pc-Infected Plants (Day 10) | |||
|---|---|---|---|
| Organ | Compound | High Ambient Temperature + Pc | Heat Wave Events+ Pc |
| Leaf | Ethyl gallate (mg gallic acid/g DW) | ns | ns |
| Ellagic acid (mg ellagic acid/g DW) | ns | ns | |
| 3-Feruloylquinic acid (mg gallic acid/g DW) | ↑↑ | ↑ | |
| Miquelianin (quercetin 3-O-glucuronide) (mg quercetin/g DW) | ↑↑ | ns | |
| Hydroxytyrosol acetate (mg gallic acid/g DW) | ↓↓ | ↓↓ | |
| Root | Hydroxybenzoic acid (mg gallic acid/g DW) | ns | ↑ |
| 4-Hydroxyphenylacetic acid (mg gallic acid/g DW) | ns | ns | |
| Coniferyl aldehyde (mg gallic acid/g DW) | ↓↓ | ns | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dorado, F.J.; Alías, J.C.; Chaves, N.; Solla, A. Warming Scenarios and Phytophthora cinnamomi Infection in Chestnut (Castanea sativa Mill.). Plants 2023, 12, 556. https://doi.org/10.3390/plants12030556
Dorado FJ, Alías JC, Chaves N, Solla A. Warming Scenarios and Phytophthora cinnamomi Infection in Chestnut (Castanea sativa Mill.). Plants. 2023; 12(3):556. https://doi.org/10.3390/plants12030556
Chicago/Turabian StyleDorado, F. Javier, Juan Carlos Alías, Natividad Chaves, and Alejandro Solla. 2023. "Warming Scenarios and Phytophthora cinnamomi Infection in Chestnut (Castanea sativa Mill.)" Plants 12, no. 3: 556. https://doi.org/10.3390/plants12030556
APA StyleDorado, F. J., Alías, J. C., Chaves, N., & Solla, A. (2023). Warming Scenarios and Phytophthora cinnamomi Infection in Chestnut (Castanea sativa Mill.). Plants, 12(3), 556. https://doi.org/10.3390/plants12030556







