Phenolic Profile and In Vitro Antioxidant Activity of Different Corn and Rice Varieties
Abstract
1. Introduction
2. Results and Discussion
2.1. Total Anthocyanin Content
2.2. Soluble Polyphenols Content
2.2.1. Rice
2.2.2. Corn
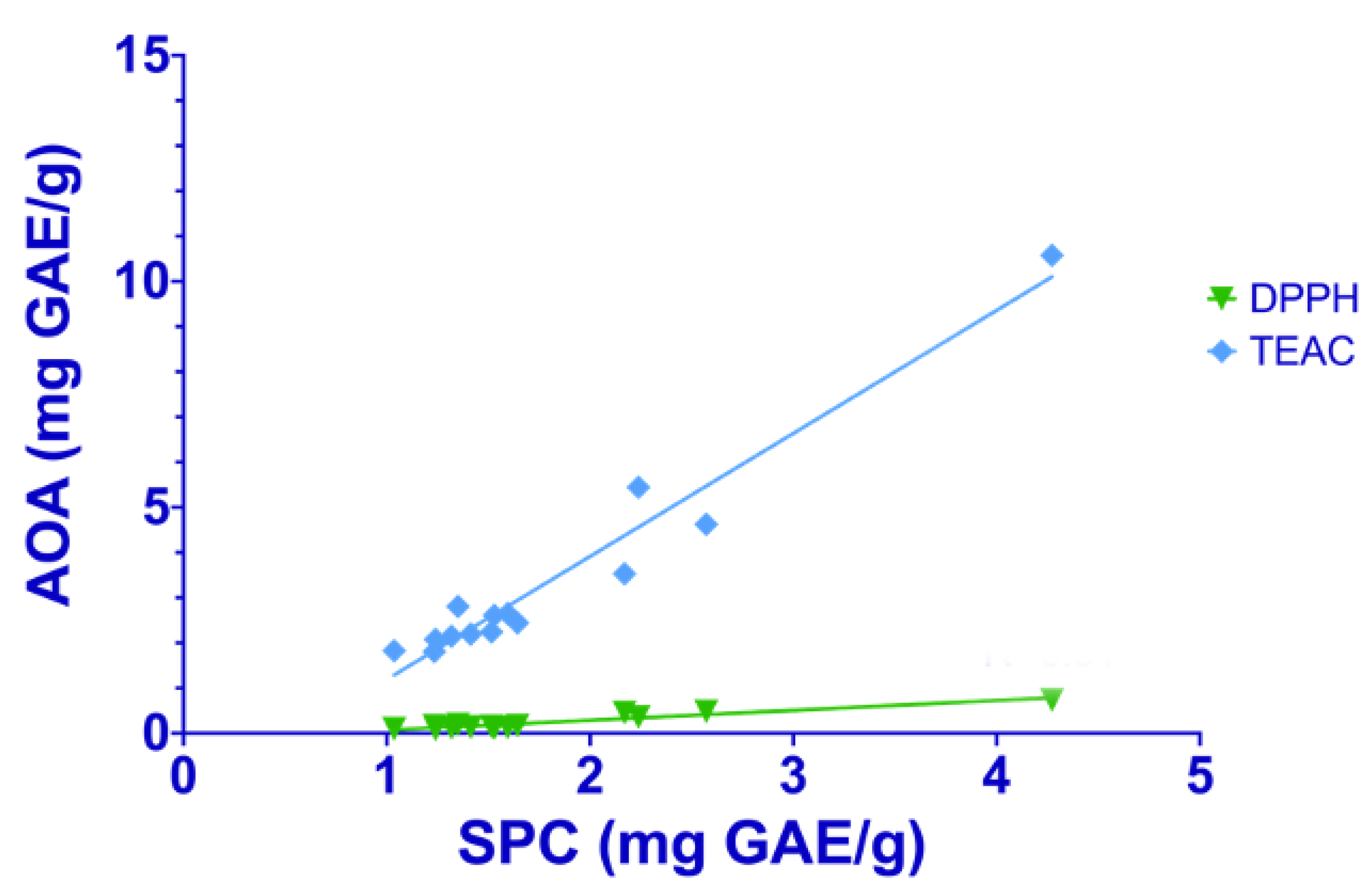
2.3. Total Antioxidant Capacity
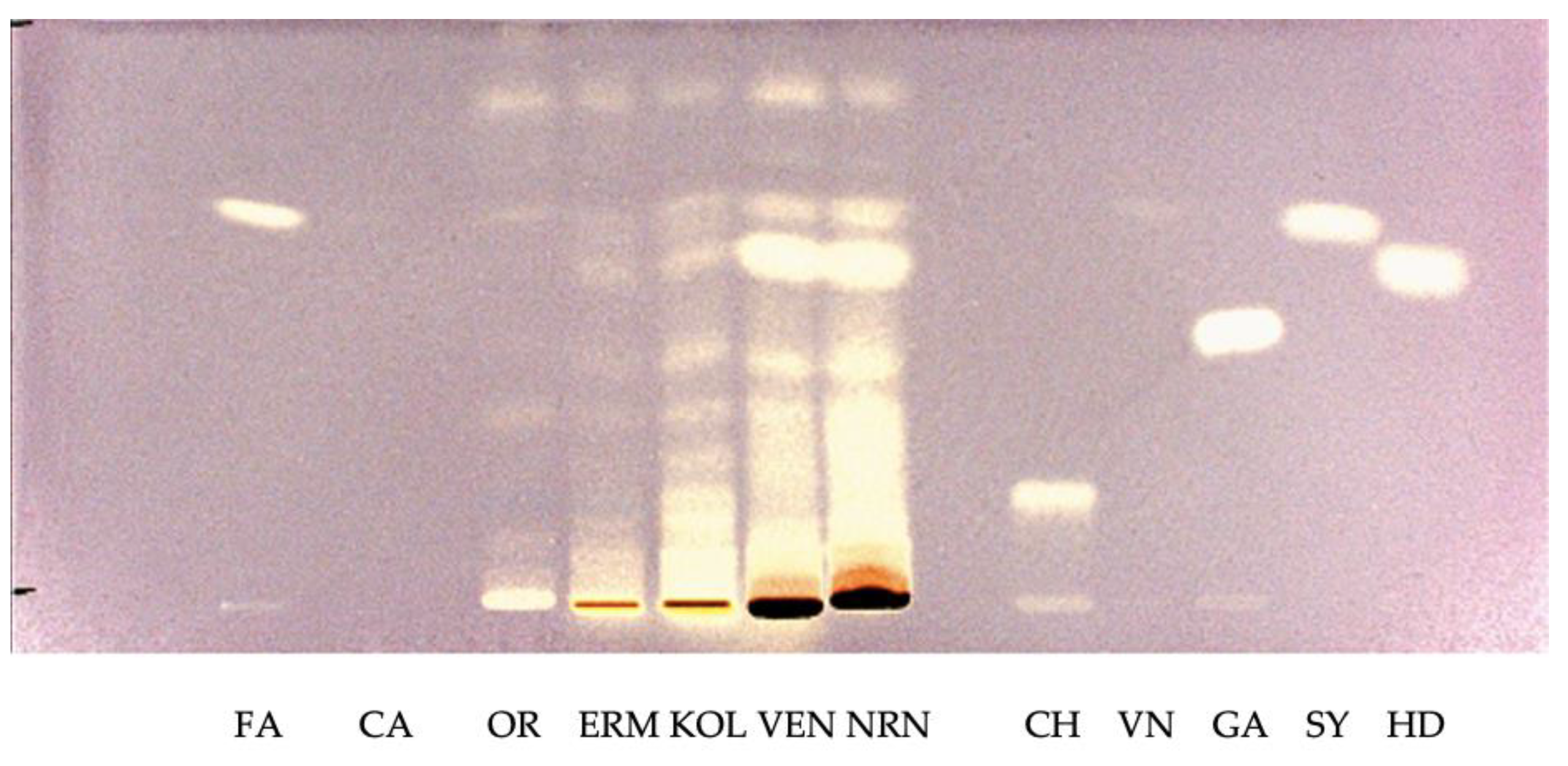
2.4. High Performance Thin Layer Chromatography
2.4.1. Validation Data
2.4.2. Rice
2.4.3. Corn
3. Materials and Methods
3.1. Samples
3.2. Determination of Total Anthocyanin Content
3.2.1. Extraction of Anthocyanins
3.2.2. pH Differential Assay
3.3. Extraction of Soluble Phenolic Compounds
3.4. Determination of Soluble Phenolic Content
Folin–Ciocalteu’s Assay
3.5. Determination of In Vitro Antioxidant Capacity
3.5.1. DPPH Assay
3.5.2. TEAC Assay
3.5.3. High Performance Thin Layer Chromatography
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Catassi, C.; Verdu, E.F.; Bai, J.C.; Lionetti, E. Coeliac Disease. Lancet 2022, 399, 2413–2426. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Arora, A.; Strand, T.A.; Leffler, D.A.; Catassi, C.; Green, P.H.; Kelly, C.P.; Ahuja, V.; Makharia, G.K. Global Prevalence of Celiac Disease: Systematic Review and Meta-Analysis. Clin. Gastroenterol. Hepatol. 2018, 16, 823–836. [Google Scholar] [CrossRef]
- Makovicky, P.; Makovicky, P.; Caja, F.; Rimarova, K.; Samasca, G.; Vannucci, L. Celiac Disease and Gluten-Free Diet: Past, Present, and Future. Gastroenterol. Hepatol. 2020, 13, 1–7. [Google Scholar]
- Vici, G.; Belli, L.; Biondi, M.; Polzonetti, V. Gluten Free Diet and Nutrient Deficiencies: A Review. Clin. Nutr. 2016, 35, 1236–1241. [Google Scholar] [CrossRef] [PubMed]
- Lebwohl, B.; Sanders, D.S.; Green, P.H.R. Coeliac Disease. Lancet 2018, 391, 70–81. [Google Scholar] [CrossRef]
- Aljada, B.; Zohni, A.; El-Matary, W. The Gluten-Free Diet for Celiac Disease and Beyond. Nutrients 2021, 13, 3993. [Google Scholar] [CrossRef]
- Dias, R.; Pereira, C.B.; Pérez-Gregorio, R.; Mateus, N.; Freitas, V. Recent Advances on Dietary Polyphenol’s Potential Roles in Celiac Disease. Trends Food Sci. Technol. 2021, 107, 213–225. [Google Scholar] [CrossRef]
- Freeman, H.J. Adverse Effects of Gluten-Free Diets. Int. J. Celiac Dis. 2018, 6, 71–73. [Google Scholar]
- Caio, G.; Volta, U.; Sapone, A.; Leffler, D.A.; de Giorgio, R.; Catassi, C.; Fasano, A. Celiac Disease: A Comprehensive Current Review. BMC Med. 2019, 17, 142. [Google Scholar] [CrossRef]
- Colombo, F.; Di Lorenzo, C.; Petroni, K.; Silano, M.; Pilu, R.; Falletta, E.; Biella, S.; Restani, P. Pigmented Corn Varieties as Functional Ingredients for Gluten-Free Products. Foods 2021, 10, 1770. [Google Scholar] [CrossRef]
- Ito, V.C.; Lacerda, L.G. Black Rice (Oryza Sativa L.): A Review of Its Historical Aspects, Chemical Composition, Nutritional and Functional Properties, and Applications and Processing Technologies. Food Chem. 2019, 301, 125304. [Google Scholar] [CrossRef]
- Piazza, S.; Colombo, F.; Bani, C.; Fumagalli, M.; Vincentini, O.; Sangiovanni, E.; Martinelli, G.; Biella, S.; Silano, M.; Restani, P.; et al. Evaluation of the Potential Anti-Inflammatory Activity of Black Rice in the Framework of Celiac Disease. Foods 2023, 12, 63. [Google Scholar] [CrossRef]
- Yang, Z.; Qin, C.; Weng, P.; Zhang, X.; Xia, Q.; Wu, Z.; Liu, L.; Xiao, J. In Vitro Evaluation of Digestive Enzyme Inhibition and Antioxidant Effects of Naked Oat Phenolic Acid Compound (OPC). Int. J. Food Sci. Technol. 2020, 55, 2531–2540. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y.; Ren, X.; Zhang, X.; Wu, Z.; Liu, L. The Positive Correlation of Antioxidant Activity and Prebiotic Effect about Oat Phenolic Compounds. Food Chem. 2023, 402, 134231. [Google Scholar] [CrossRef]
- Zhu, F. Anthocyanins in Cereals: Composition and Health Effects. Food Res. Int. 2018, 109, 232–249. [Google Scholar] [CrossRef]
- Boue, S.M.; Daigle, K.W.; Chen, M.H.; Cao, H.; Heiman, M.L. Antidiabetic Potential of Purple and Red Rice (Oryza Sativa L.) Bran Extracts. J. Agric. Food Chem. 2016, 64, 5345–5353. [Google Scholar] [CrossRef]
- Shahidi, F.; Yeo, J.D. Bioactivities of Phenolics by Focusing on Suppression of Chronic Diseases: A Review. Int. J. Mol. Sci. 2018, 19, 1573. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Colombo, F.; Biella, S.; Stockley, C.; Restani, P. Polyphenols and Human Health: The Role of Bioavailability. Nutrients 2021, 13, 273. [Google Scholar] [CrossRef]
- Llauradó Maury, G.; Méndez Rodríguez, D.; Hendrix, S.; Escalona Arranz, J.C.; Fung Boix, Y.; Pacheco, A.O.; García Díaz, J.; Morris Quevedo, H.J.; Ferrer Dubois, A.; Isaac Aleman, E.; et al. Antioxidants in Plants: A Valorization Potential Emphasizing the Need for the Conservation of Plant Biodiversity in Cuba. Antioxidants 2020, 9, 1048. [Google Scholar] [CrossRef]
- Ciulu, M.; de la Luz Cádiz-Gurrea, M.; Segura-Carretero, A. Extraction and Analysis of Phenolic Compounds in Rice: A Review. Molecules 2018, 23, 2890. [Google Scholar] [CrossRef]
- Melini, V.; Panfili, G.; Fratianni, A.; Acquistucci, R. Bioactive Compounds in Rice on Italian Market: Pigmented Varieties as a Source of Carotenoids, Total Phenolic Compounds and Anthocyanins, before and after Cooking. Food Chem. 2019, 277, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Cömert, E.D.; Gökmen, V. Antioxidants Bound to an Insoluble Food Matrix: Their Analysis, Regeneration Behavior, and Physiological Importance. Compr. Rev. Food Sci. Food Saf. 2017, 16, 382–399. [Google Scholar] [CrossRef] [PubMed]
- Das, A.K.; Singh, V. Antioxidative Free and Bound Phenolic Constituents in Botanical Fractions of Indian Specialty Maize (Zea Mays L.) Genotypes. Food Chem. 2016, 201, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Li, X.; Zhang, Y.; Yang, Y.; Sun, R.; Li, Y.; Gao, J.; Han, Y. Differential Flavonoids and Carotenoids Profiles in Grains of Six Poaceae Crops. Foods 2022, 11, 2068. [Google Scholar] [CrossRef] [PubMed]
- Mattioli, R.; Francioso, A.; Mosca, L.; Silva, P. Anthocyanins: A Comprehensive Review of Their Chemical Properties and Health Effects on Cardiovascular and Neurodegenerative Diseases. Molecules 2020, 25, 3809. [Google Scholar] [CrossRef]
- Rodríguez-Salinas, P.A.; Zavala-García, F.; Urías-Orona, V.; Muy-Rangel, D.; Heredia, J.B.; Niño-Medina, G. Chromatic, Nutritional and Nutraceutical Properties of Pigmented Native Maize (Zea Mays L.) Genotypes from the Northeast of Mexico. Arab. J. Sci. Eng. 2020, 45, 95–112. [Google Scholar] [CrossRef]
- Goufo, P.; Trindade, H. Rice Antioxidants: Phenolic Acids, Flavonoids, Anthocyanins, Proanthocyanidins, Tocopherols, Tocotrienols, c-Oryzanol, and Phytic Acid. Food Sci. Nutr. 2014, 2, 75–104. [Google Scholar] [CrossRef]
- Hosoda, K.; Sasahara, H.; Matsushita, K.; Tamura, Y.; Miyaji, M.; Matsuyama, H. Anthocyanin and Proanthocyanidin Contents, Antioxidant Activity, and in Situ Degradability of Black and Red Rice Grains. Asian-Australas. J. Anim. Sci. 2018, 31, 1213–1220. [Google Scholar] [CrossRef]
- Lopez-Martinez, L.X.; Oliart-Ros, R.M.; Valerio-Alfaro, G.; Lee, C.H.; Parkin, K.L.; Garcia, H.S. Antioxidant Activity, Phenolic Compounds and Anthocyanins Content of Eighteen Strains of Mexican Maize. LWT—Food Sci. Technol. 2009, 42, 1187–1192. [Google Scholar] [CrossRef]
- Stuper-Szablewska, K.; Perkowski, J. Phenolic Acids in Cereal Grain: Occurrence, Biosynthesis, Metabolism and Role in Living Organisms. Crit. Rev. Food Sci. Nutr. 2019, 59, 664–675. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food Sources and Bioavailability 1,2. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Wang, T.; He, F.; Chen, G. Improving Bioaccessibility and Bioavailability of Phenolic Compounds in Cereal Grains through Processing Technologies: A Concise Review. J. Funct. Foods 2014, 7, 101–111. [Google Scholar] [CrossRef]
- Yodmanee, S.; Karrila, T.T.; Pakdeechanuan, P. Physical, Chemical and Antioxidant Properties of Pigmented Rice Grown in Southern Thailand. Int. Food Res. J. 2011, 18, 901–906. [Google Scholar]
- Gulcin, İ. Antioxidants and Antioxidant Methods: An Updated Overview. Arch. Toxicol. 2020, 94, 651–715. [Google Scholar] [CrossRef]
- Salinas-Moreno, Y.; García-Salinas, C.; Ramírez-Díaz, J.L.; Alemán-de la Torre, I. Phenolic Compounds in Maize Grains and Its Nixtamalized Products. In Phenolic Compounds—Natural Sources, Importance and Applications; Soto-Hernández, M., Ed.; InTech: London, UK, 2017; pp. 215–232. [Google Scholar]
- Petroni, K.; Pilu, R.; Tonelli, C. Anthocyanins in Corn: A Wealth of Genes for Human Health. Planta 2014, 240, 901–911. [Google Scholar] [CrossRef]
- Horwitz, W. AOAC Official Method 2005.02: Total Monomeric Anthocyanin Pigment Content of Fruit Juices, Beverages, Natural Colorants, and Wines. In Official Methods of Analysis; AOAC IN TER NA TIONA: Gaithersburg, MD, USA, 2005; ISBN 0-935584-77-3. [Google Scholar]
- Singleton, V.L.; Rossi, J.A., Jr. Colorimetry of Total Phenolics with Phosphomolybdic-Phosphothungstic Acid Reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant Activity Applying an Improved ABTS Radical Cation Decolorization Assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Reich, E.; Schibli, A. High Performance Thin Layer Chromatography for the Analysis of Medicinal Plants; Schibli, A., Ed.; Thieme: New York, NY, USA, 2007. [Google Scholar]




| Cereals | Sample Code | Kernel Colour | TA (mg CY/g) Mean ± SD |
|---|---|---|---|
| Rice | ERM | Red | ND |
| KOL | ND | ||
| VEN | Black | 1.18 ± 0.07 a | |
| NRN | 3.04 ± 0.18 b | ||
| VIO | 3.98 ± 0.38 c | ||
| Corn | RRb | Red | ND |
| PG_2018 | ND | ||
| PGb_2018 | ND | ||
| PGb_2019 | ND | ||
| Pop BRP | Red/Violet | 0.45 ± 0.07 a | |
| Su/Su BRP | 1.05 ± 0.06 b | ||
| Scagliolo BRP | 0.52 ± 0.06 a |
| Sample Code | Kernel Colour | SPC (mg GAE/g) Mean ± SD |
|---|---|---|
| OR | White | 0.72 ± 0.06 a |
| ERM | Red | 2.65 ± 0.16 b |
| KOL | 4.84 ± 0.21 c | |
| VEN | Black | 4.42 ± 0.36 c |
| NRN | 7.39 ± 0.22 d | |
| VIO | 5.69 ± 0.62 e |
| Sample Code | Kernel Colour | SPC (mg GAE/g) Mean ± SD |
|---|---|---|
| OSb | White | 1.04 ± 0.13 a |
| OTb | Yellow | 1.60 ± 0.04 c |
| NSb_2018 | 1.53 ± 0.07 b,c | |
| NSb_2019 | 1.24 ± 0.05 a,b | |
| Pop | 1.32 ± 0.17 a,b,c | |
| Su/Su | 1.35 ± 0.06 a,b,c | |
| Scagliolo | 1.23 ± 0.33 a,b | |
| RRb | Red | 2.17 ± 0.02 d |
| PG_2018 | 1.41 ± 0.02 b,c | |
| PGb_2018 | 1.52 ± 0.07 b,c | |
| PGb_2019 | 1.64 ± 0.06 c | |
| Pop BRP | Red/Violet | 2.24 ± 0.13 d |
| Su/Su BRP | 4.27 ± 0.43 f | |
| Scagliolo BRP | 2.57 ± 0.30 e |
| Cereal | Sample Code | Kernel Colour | AOA | |
|---|---|---|---|---|
| (mg GAE/g) Mean ± SD | (mg TE/g) Mean ± SD | |||
| Rice | OR | White | 0.14 ± 0.02 a | 1.18 ± 0.10 a |
| ERM | Red | 0.89 ± 0.03 b | 5.28 ± 0.24 b | |
| KOL | 1.70 ± 0.09 d | 9.20 ± 0.81 c | ||
| VEN | Black | 1.39 ± 0.16 c | 8.22 ± 0.69 c | |
| NRN | 2.26 ± 0.09 e | 14.25 ± 0.61 d | ||
| VIO | 1.65 ± 0.11 d | * | ||
| Corn | OSb | White | 0.098 ± 0.013 a,b | 1.83 ± 0.08 a |
| OTb | Yellow | 0.134 ± 0.002 a,b,c | 2.65 ± 0.12 d,e | |
| NSb_2018 | 0.112 ± 0.004 a,b,c | 2.61 ± 0.18 c,d,e | ||
| NSb_2019 | 0.091 ± 0.001 a | 2.08 ± 0.06 a,b | ||
| Pop | 0.120 ± 0.014 a,b,c | 2.15 ± 0.17 a,b,c | ||
| Su/Su | 0.197 ± 0.044 d | 2.80 ± 0.06 e | ||
| Scagliolo | 0.165 ± 0.017 c,d | 1.81 ± 0.16 a | ||
| RRb | Red | 0.455 ± 0.041 f | 3.53 ± 0.12 f | |
| PG_2018 | 0.138 ± 0.002 a,b,c | 2.19 ± 0.10 a,b,c,d | ||
| PGb_2018 | 0.151 ± 0.011 b,c,d | 2.24 ± 0.15 a,b,c,d | ||
| PGb_2019 | 0.167 ± 0.010 c,d | 2.44 ± 0.11 b,c,d,e | ||
| Pop BRP | Red/Violet | 0.354 ± 0.016 e | 5.44 ± 0.52 h | |
| Su/Su BRP | 0.736 ± 0.044 g | 10.58 ± 0.28 i | ||
| Scagliolo BRP | 0.480 ± 0.039 f | 4.63 ± 0.66 g | ||
| Compound | Ratio frontis Mean ± SD | LOD | ||
|---|---|---|---|---|
| λ (nm) | ng * | mg/g ** | ||
| Chlorogenic acid | 0.24 ± 0.02 | 366 | 100 | 6.7 |
| Gallic acid | 0.45 ± 0.02 | 254, 366, vis | 200 | 13.3 |
| Dihydroxybenzoic acid | 0.54 ± 0.02 | vis | 40 | 2.7 |
| Syringic acid | 0.58 ± 0.01 | vis | 100 | 6.7 |
| Coumaric acid | 0.60 ± 0.02 | 254 | 200 | 13.3 |
| Ferulic acid | 0.60 ± 0.01 | 366 | 100 | 6.7 |
| Vanillic acid | 0.60 ± 0.01 | 254 | 100 | 6.7 |
| Cereal | Sample | Sample Code | Provenience | Pigmentation |
|---|---|---|---|---|
| Rice | Originario brown | OR | Italian market | White |
| Ermes brown | ERM | Italian market | Red | |
| Kolorado brown | KOL | Italian market | Red | |
| Venere brown | VEN | Italian market | Black | |
| Nerone brown | NRN | Italian market | Black | |
| Violet brown | VIO | Azienda Agricola Bertolone | Black | |
| Corn | Ostenga brown | OSb | Azienda Agricola Caretto | White |
| Ottofile brown 2019 | OTb | Azienda Agricola Caretto | Yellow | |
| Nostrano Isola brown 2018 | NSb_2018 | Azienda Agricola Caretto | Yellow | |
| Nostrano Isola brown 2019 | NSb_2019 | Azienda Agricola Caretto | Yellow | |
| Rosso di Rovetta brown | RRb | Associazione Rosso Mais | Red | |
| Pignoletto 2018 | PG_2018 | Azienda Agricola Caretto | Red | |
| Pignoletto brown 2018 | PGb_2018 | Azienda Agricola Caretto | Red | |
| Pignoletto brown 2019 | PGb_2019 | Azienda Agricola Caretto | Red | |
| POP | POP | Bioscience Dept.—UNIMI | Yellow | |
| su/su | su/su | Bioscience Dept.—UNIMI | Yellow | |
| Scagliolo | Sca | Bioscience Dept.—UNIMI | Yellow | |
| POP BRP | POP BRP | Bioscience Dept.—UNIMI | Red/Violet | |
| su/su BRP | su/su BRP | Bioscience Dept.—UNIMI | Red/Violet | |
| Scagliolo BRP | Sca BRP | Bioscience Dept.—UNIMI | Red/Violet |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bani, C.; Di Lorenzo, C.; Restani, P.; Mercogliano, F.; Colombo, F. Phenolic Profile and In Vitro Antioxidant Activity of Different Corn and Rice Varieties. Plants 2023, 12, 448. https://doi.org/10.3390/plants12030448
Bani C, Di Lorenzo C, Restani P, Mercogliano F, Colombo F. Phenolic Profile and In Vitro Antioxidant Activity of Different Corn and Rice Varieties. Plants. 2023; 12(3):448. https://doi.org/10.3390/plants12030448
Chicago/Turabian StyleBani, Corinne, Chiara Di Lorenzo, Patrizia Restani, Francesca Mercogliano, and Francesca Colombo. 2023. "Phenolic Profile and In Vitro Antioxidant Activity of Different Corn and Rice Varieties" Plants 12, no. 3: 448. https://doi.org/10.3390/plants12030448
APA StyleBani, C., Di Lorenzo, C., Restani, P., Mercogliano, F., & Colombo, F. (2023). Phenolic Profile and In Vitro Antioxidant Activity of Different Corn and Rice Varieties. Plants, 12(3), 448. https://doi.org/10.3390/plants12030448







