Fissistigma oldhamii (Hemsl.) Merr.: Ethnomedicinal, Phytochemistry, and Pharmacological Aspects
Abstract
:1. Introduction
2. Phytochemical Content of Fissistigma oldhamii
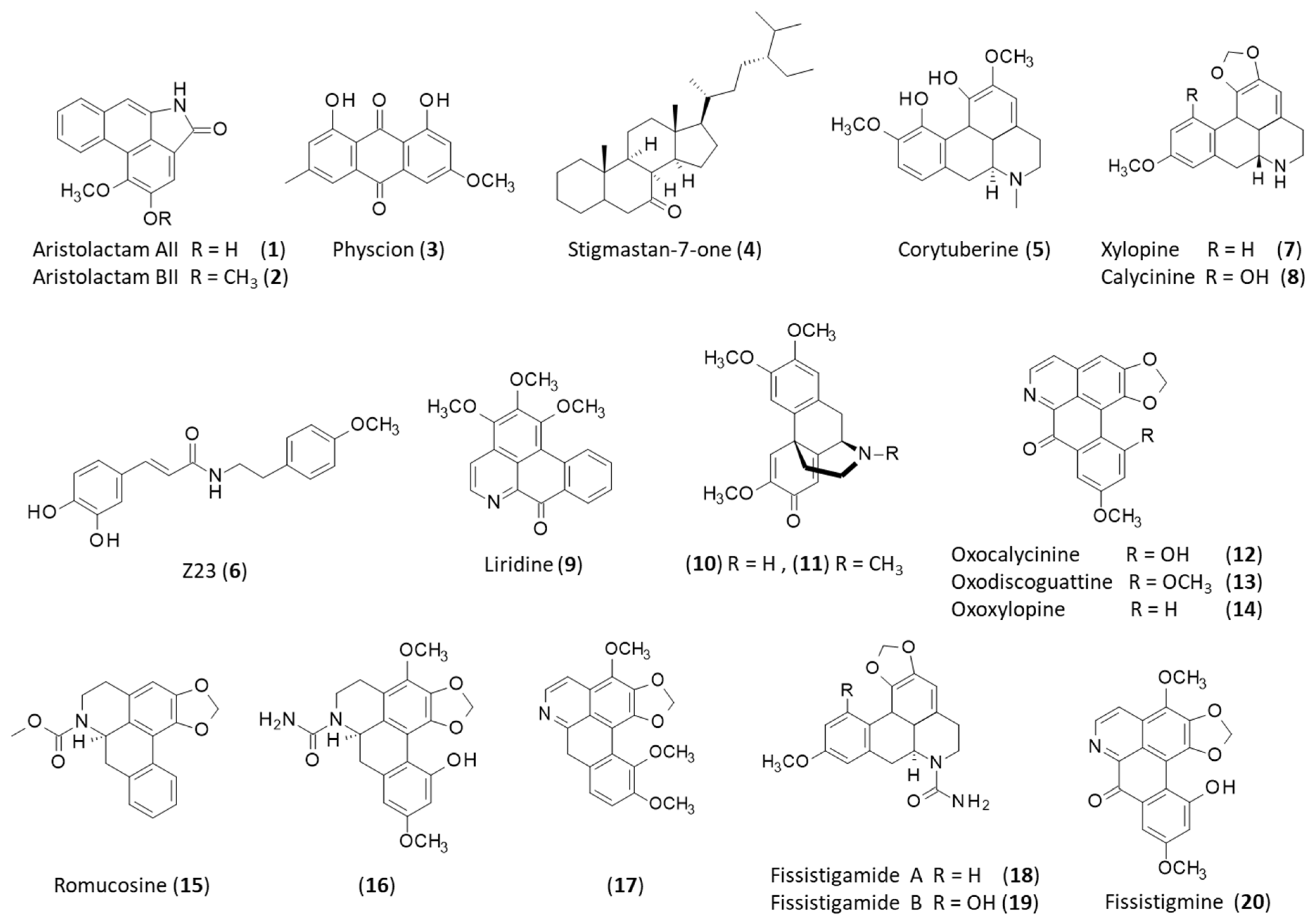
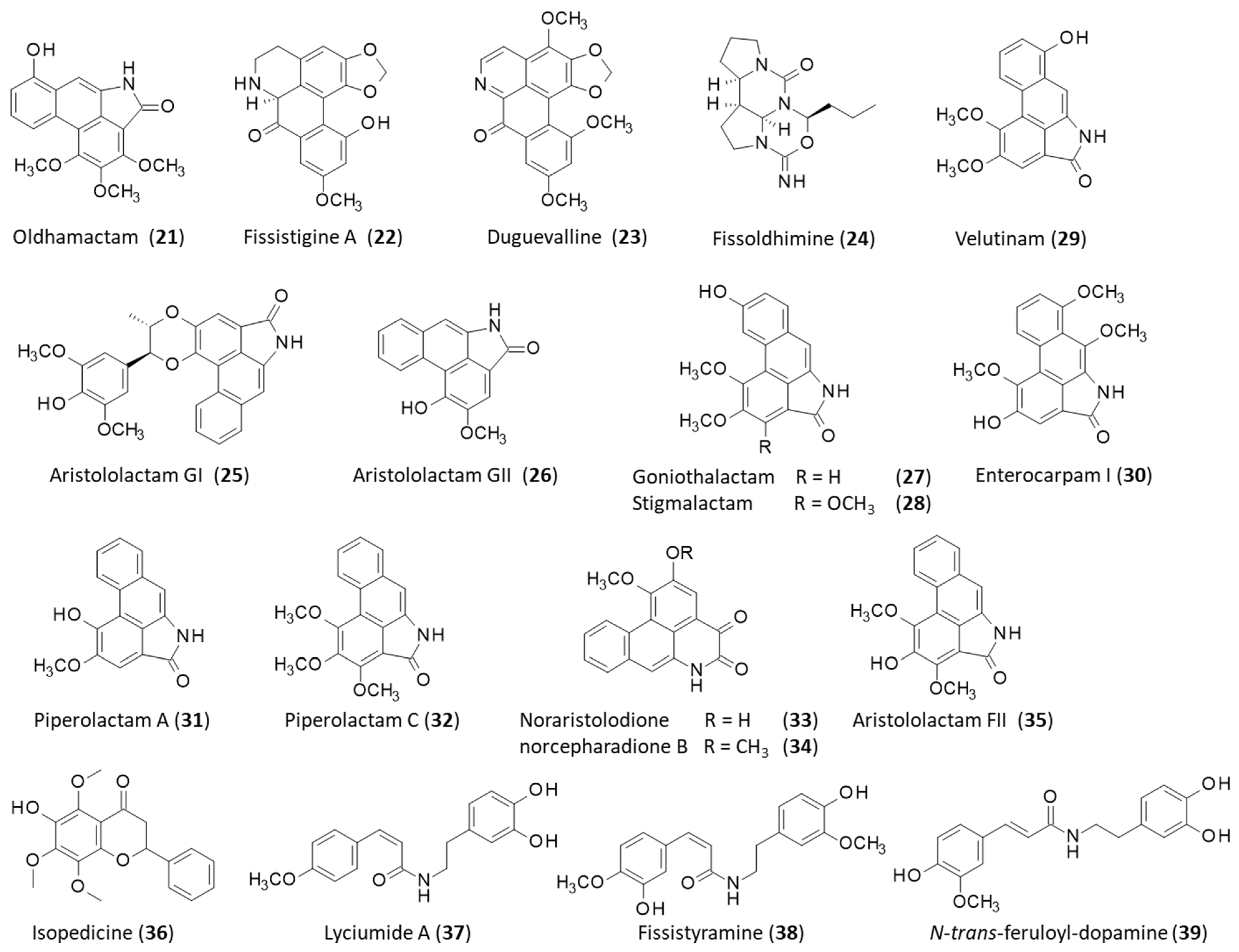
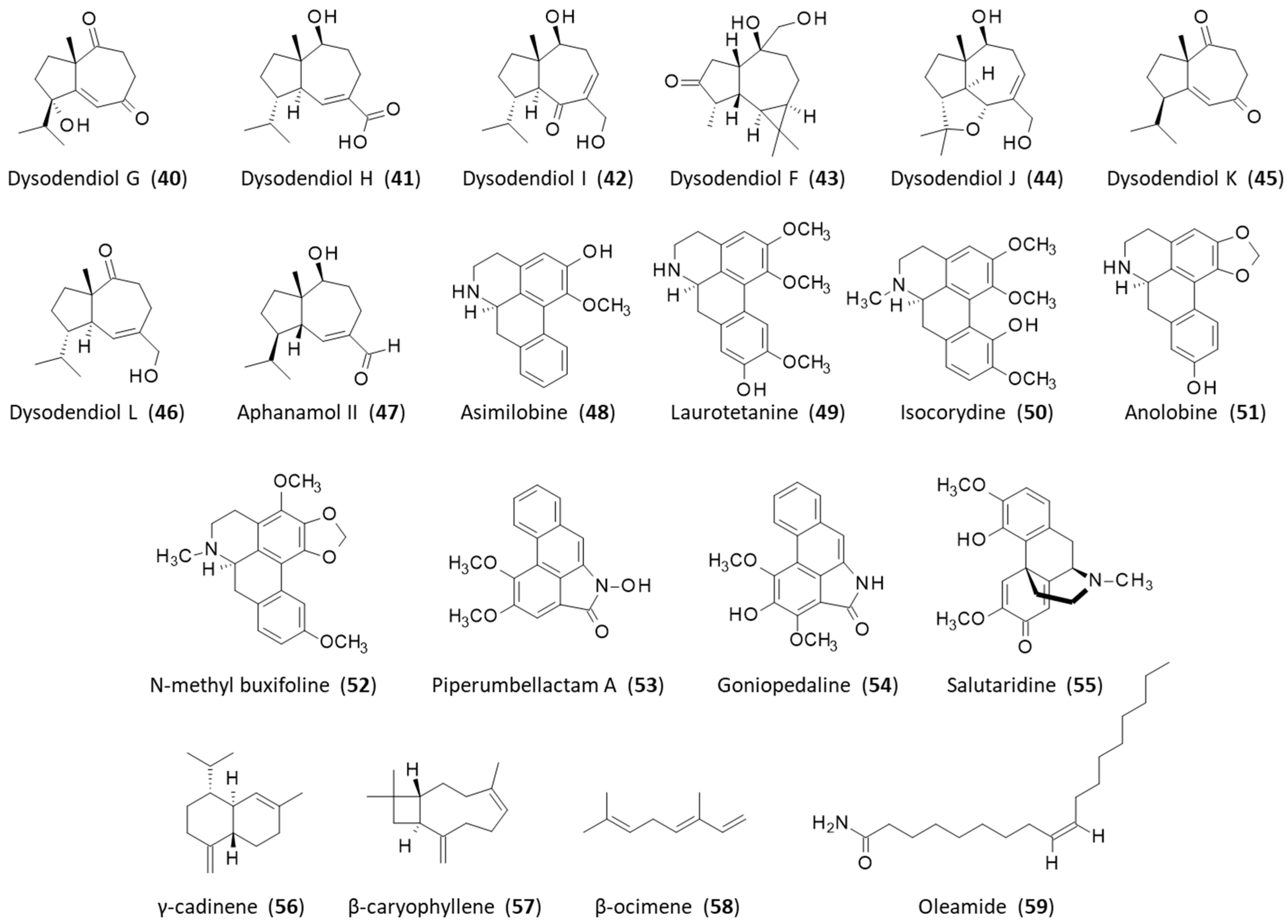
2.1. Natural Products from the Roots
2.2. Natural Products from the Stems
2.3. Natural Products from the Leaves
2.4. Natural Products from the Fruits
2.5. Natural Products from the Seeds
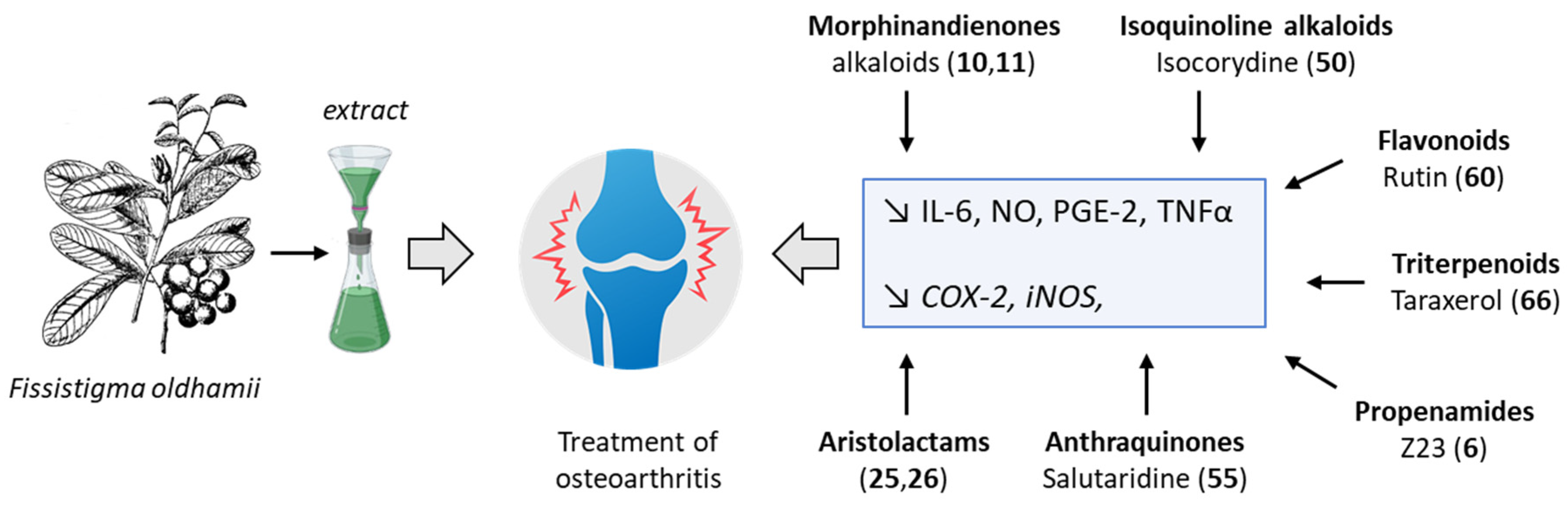
3. Discussion
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aminimoghadamfarouj, N.; Nematollahi, A.; Wiart, C. Annonaceae: Bio-resource for tomorrow’s drug discovery. J. Asian Nat. Prod. Res. 2011, 13, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Al Kazman, B.S.M.; Harnett, J.E.; Hanrahan, J.R. Traditional Uses, Phytochemistry and Pharmacological Activities of Annonacae. Molecules 2022, 27, 3462. [Google Scholar] [CrossRef] [PubMed]
- Pham, G.N.; Nguyen-Ngoc, H. Fissistigma genus—A review on phytochemistry and pharmacological activities. Nat. Prod. Res. 2021, 35, 5209–5223. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Zhu, C.C.; Luo, Y.X.; Zhang, B.; Ji, X.S.; Song, X.M.; Zhou, X.M. Sesquiterpenes from Fissistigma glaucescens inhibiting the proliferation of synoviocytes. J. Asian Nat. Prod. Res. 2022, 24, 550–555. [Google Scholar] [CrossRef]
- How to Care with Fissistigma oldhamii. Available online: https://www.picturethisai.com/care/Fissistigma_oldhamii.html (accessed on 6 September 2023).
- Xu, F.X.; Ronse de Craene, L.P. Pollen morphology and ultrastructure of selected species from Annonaceae. Plant Syst. Evol. 2013, 299, 11–24. [Google Scholar] [CrossRef]
- Butterfly Wing Scale Digital Image Gallery. Available online: https://micro.magnet.fsu.edu/optics/olympusmicd/galleries/butterfly/fourbarswallowtailr1.html (accessed on 6 September 2023).
- Hu, H.; Lee-Fong, Y.; Peng, J.; Hu, B.; Li, J.; Li, Y.; Huang, H. Comparative Research of Chemical Profiling in Different Parts of Fissistigma oldhamii by Ultra-High-Performance Liquid Chromatography Coupled with Hybrid Quadrupole-Orbitrap Mass Spectrometry. Molecules 2021, 26, 960. [Google Scholar] [CrossRef]
- Hu, H.; Yang, Y.; Aissa, A.; Tekin, V.; Li, J.; Panda, S.K.; Huang, H.; Luyten, W. Ethnobotanical study of Hakka traditional medicine in Ganzhou, China and their antibacterial, antifungal, and cytotoxic assessments. BMC Complement. Med. Ther. 2022, 22, 244. [Google Scholar] [CrossRef]
- Zheng, Z.P.; Liang, J.Y.; Hu, L.H. Studies on the active constituents of Fissistigma oldhamii. Chin. J. Nat. Med. 2005, 3, 151–154. [Google Scholar]
- Peng, X.; Zhou, Y.; Gao, Y.; Wu, T. The chemical constituents of Fissistigma oldhamii (III). Chin. Tradit. Pat. Med. 1992, 12, wpr-578757. [Google Scholar]
- Hu, X.D.; Zhong, X.G.; Zhang, X.H.; Zhang, Y.N.; Zheng, Z.P.; Zhou, Y.; Tang, W.; Yang, Y.; Yang, Y.F.; Hu, L.H.; et al. 7′-(3′,4′-dihydroxyphenyl)-N-[(4-methoxyphenyl)ethyl]propenamide (Z23), an effective compound from the Chinese herb medicine Fissistigma oldhamii (Hemsl.) Merr, suppresses T cell-mediated immunity in vitro and in vivo. Life Sci. 2007, 81, 1677–1684. [Google Scholar] [CrossRef]
- Chen, J.; Jin, C.; Xu, B.; Shu, J.; Shao, F.; Yuan, C.; Li, F.; Huang, L.; Huang, H. New compounds from the stems of Fissistigma oldhamii var. longistipitatum and their cytotoxic activities. Fitoterapia 2021, 151, 104883. [Google Scholar] [PubMed]
- Ge, Y.W.; Zhu, S.; Shang, M.Y.; Zang, X.Y.; Wang, X.; Bai, Y.J.; Li, L.; Komatsu, K.; Cai, S.Q. Aristololactams and aporphines from the stems of Fissistigma oldhamii (Annonaceae). Phytochemistry 2013, 86, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.B.; Scheng, Y.D.; Kuo, S.C.; Wu, T.S.; Iitaka, Y.; Ebizuka, Y.; Sankawa, U. Fissoldhimine, a Novel Skeleton Alkaloid from Fissistigma oldhamii. Chem. Pharm. Bull. 1994, 42, 2202–2204. [Google Scholar] [CrossRef]
- Chia, Y.C.; Chang, F.R.; Teng, C.M.; Wu, Y.C. Aristolactams and dioxoaporphines from Fissistigma balansae and Fissistigma oldhamii. J. Nat. Prod. 2000, 63, 1160–1163. [Google Scholar] [CrossRef] [PubMed]
- Hwang, T.L.; Li, G.L.; Lan, Y.H.; Chia, Y.C.; Hsieh, P.W.; Wu, Y.H.; Wu, Y.C. Potent inhibition of superoxide anion production in activated human neutrophils by isopedicin, a bioactive component of the Chinese medicinal herb Fissistigma oldhamii. Free Radic. Biol. Med. 2009, 46, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.C.; Luo, Y.X.; Wu, Y.; Yang, J.Y.; Ji, X.S.; Zhou, X.M. A New N-cis-Coumaroyltyramine Derivative from Fissistigma oldhamii. Chem. Nat. Compd. 2021, 57, 832–834. [Google Scholar] [CrossRef]
- Zhou, X.M.; Zheng, C.J.; Zhang, Y.Q.; Zhang, X.P.; Song, X.P.; Xu, W.; Chen, G.Y. Guaiane-Type Sesquiterpenoids from Fissistigma oldhamii Inhibit the Proliferation of Synoviocytes. Planta Med. 2017, 83, 217–223. [Google Scholar] [CrossRef]
- Zhong, S.H.; Fu, Y.H.; Zhou, X.M.; Song, X.P.; Chen, G.Y. Studies on alkaloids from Fissistigma oldhamii. Zhongguo Zhong Yao Za Zhi 2016, 41, 2838–2842. [Google Scholar]
- Thang, T.D.; Dung, N.X. Progress in the study of some species from Vietnam. In Aromatic Plants from Asia, Their Chemistry and Application in Food and Therapy; Jirovetz, L., Dung, N.X., Varshney, V.K., Eds.; Har Krishnan Bhalla & Sons: Dehradun, India, 2007. [Google Scholar]
- Aodah, A.H.; Devi, S.; Alkholifi, F.K.; Yusufoglu, H.S.; Foudah, A.I.; Alam, A. Effects of Taraxerol on Oxidative and Inflammatory Mediators in Isoproterenol-Induced Cardiotoxicity in an Animal Model. Molecules 2023, 28, 4089. [Google Scholar] [CrossRef]
- Rathore, J.S.; Garg, S.K.; Nagar, A.; Sharma, N.D.; Gupta, S.R. New phenolic components of Didymocarpus pedicellata. Planta Med. 1981, 43, 86–88. [Google Scholar] [CrossRef]
- Dung, V.C. The chemical composition of fruit oil fatty acids of Fissistigma oldhamii (Hemsl.) Merr. From Vietnam. Báo cáo khoa học về sinh thái và tài nguyên sinh vật (Hội nghị khoa học toàn quốc lần thứ năm) 2013, 204, 998–1000. [Google Scholar]
- Dung, V.C.; Quynh Giang, N.T.; Thang, T.D.; Hoang, V.D. Chemical constituents of the fruits from Fissistigma oldhamii (Hemsl.) Merr. Growing in Vietnam. Vietnam J. Chem. Int. Ed. 2016, 54, 467–470. [Google Scholar]
- Dung, V.C.; Thang, T.D. Triterpenoids from the fruit os Fissistigma oldhamii (Hemsl.) Merr. Tạp chí khoa học 2018, 47, 11–15. (In Vietnamese) [Google Scholar]
- Chia, Y.C.; Chang, F.R.; Wu, Y.C. Fissohamione, a novel furanone from Fissistigma oldhamii. Tetrahedron Lett. 1999, 40, 7513–7514. [Google Scholar] [CrossRef]
- Kim, S.K.; Ryu, S.Y.; No, J.; Choi, S.U.; Kim, Y.S. Cytotoxic alkaloids from Houttuynia cordata. Arch. Pharm. Res. 2001, 24, 518–521. [Google Scholar] [CrossRef]
- Choi, Y.L.; Kim, J.K.; Choi, S.U.; Min, Y.K.; Bae, M.A.; Kim, B.T.; Heo, J.N. Synthesis of aristolactam analogues and evaluation of their antitumor activity. Bioorg. Med. Chem. Lett. 2009, 19, 3036–3040. [Google Scholar] [CrossRef]
- Wang, Y.H.; Liu, Y.P.; Zhu, J.Q.; Zhou, G.H.; Zhang, F.; An, Q.; Yang, J.; Cho, K.W.; Jin, S.N.; Wen, J.F. Physcion prevents high-fat diet-induced endothelial dysfunction by inhibiting oxidative stress and endoplasmic reticulum stress pathways. Eur. J. Pharmacol. 2023, 943, 175554. [Google Scholar] [CrossRef]
- Li, J.; Zhu, Y.; Xu, M.; Li, P.; Zhou, Y.; Song, Y.; Cai, Q. Physcion prevents induction of optic nerve injury in rats via inhibition of the JAK2/STAT3 pathway. Exp. Ther. Med. 2023, 26, 381. [Google Scholar] [CrossRef]
- Dong, X.; Wang, L.; Song, G.; Cai, X.; Wang, W.; Chen, J.; Wang, G. Physcion Protects Rats Against Cerebral Ischemia-Reperfusion Injury via Inhibition of TLR4/NF-kB Signaling Pathway. Drug Des. Dev. Ther. 2021, 15, 277–287. [Google Scholar] [CrossRef]
- Trybus, W.; Król, T.; Trybus, E.; Stachurska, A. Physcion Induces Potential Anticancer Effects in Cervical Cancer Cells. Cells 2021, 10, 2029. [Google Scholar] [CrossRef]
- Zhang, L.; Dong, R.; Wang, Y.; Wang, L.; Zhou, T.; Jia, D.; Meng, Z. The anti-breast cancer property of physcion via oxidative stress-mediated mitochondrial apoptosis and immune response. Pharm. Biol. 2021, 59, 303–310. [Google Scholar] [CrossRef]
- Liu, X.; Hu, H.; Liu, J.; Chen, J.; Chu, J.; Cheng, H. Physcion, a novel anthraquinone derivative against Chlamydia psittaci infection. Vet. Microbiol. 2023, 279, 109664. [Google Scholar] [CrossRef]
- Lao, Z.; Fan, Y.; Huo, Y.; Liao, F.; Zhang, R.; Zhang, B.; Kong, Z.; Long, H.; Xie, J.; Sang, C.; et al. Physcion, a novel inhibitor of 5α-reductase that promotes hair growth in vitro and in vivo. Arch. Dermatol. Res. 2022, 314, 41–51. [Google Scholar] [CrossRef]
- Liu, W.; Han, C.; Hu, L.; Chen, K.; Shen, X.; Jiang, H. Characterization and inhibitor discovery of one novel malonyl-CoA: Acyl carrier protein transacylase (MCAT) from Helicobacter pylori. FEBS Lett. 2006, 580, 697–702. [Google Scholar] [CrossRef]
- Kumar, V.; Sharma, A.; Pratap, S.; Kumar, P. Biophysical and in silico interaction studies of aporphine alkaloids with Malonyl-CoA: ACP transacylase (FabD) from drug resistant Moraxella catarrhalis. Biochimie 2018, 149, 18–33. [Google Scholar] [CrossRef]
- Hu, X.D.; Yang, Y.; Zhong, X.G.; Zhang, X.H.; Zhang, Y.N.; Zheng, Z.P.; Zhou, Y.; Tang, W.; Yang, Y.F.; Hu, L.H.; et al. Anti-inflammatory effects of Z23 on LPS-induced inflammatory responses in RAW264.7 macrophages. J. Ethnopharmacol. 2008, 120, 447–451. [Google Scholar] [CrossRef]
- Anis, E.; Anis, I.; Ahmed, S.; Mustafa, G.; Malik, A.; Afza, N.; Hai, S.M.; Shahzad-ul-hussan, S.; Choudhary, M.I. Alpha-glucosidase inhibitory constituents from Cuscuta reflexa. Chem. Pharm. Bull. 2002, 50, 112–114. [Google Scholar] [CrossRef]
- Tanruean, K.; Poolprasert, P.; Kumla, J.; Suwannarach, N.; Lumyong, S. Bioactive compounds content and their biological properties of acetone extract of Cuscuta reflexa Roxb. grown on various host plants. Nat. Prod. Res. 2019, 33, 544–547. [Google Scholar] [CrossRef]
- Attiq, A.; Jalil, J.; Husain, K. Annonaceae: Breaking the Wall of Inflammation. Front. Pharmacol. 2017, 8, 752. [Google Scholar] [CrossRef]
- Wu, J.B.; Cheng, Y.D.; Chiu, N.Y.; Huang, S.C.; Kuo, S.C. A Novel Morphinandienone Alkaloid from Fissistigma oldhamii. Planta Med. 1993, 59, 179–180. [Google Scholar] [CrossRef]
- Silva, L.R.; Alves, A.F.; Cavalcante-Silva, L.H.A.; Braga, R.M.; de Almeida, R.N.; Barbosa-Filho, J.M.; Piuvezam, M.R. Milonine, a Morphinandienone Alkaloid, Has Anti-Inflammatory and Analgesic Effects by Inhibiting TNF-α and IL-1β Production. Inflammation 2017, 40, 2074–2085. [Google Scholar] [CrossRef]
- Santos, L.S.; Silva, V.R.; Menezes, L.R.A.; Soares, M.B.P.; Costa, E.V.; Bezerra, D.P. Xylopine Induces Oxidative Stress and Causes G2/M Phase Arrest, Triggering Caspase-Mediated Apoptosis by p53-Independent Pathway in HCT116 Cells. Oxidative Med. Cell. Longev. 2017, 2017, 7126872. [Google Scholar] [CrossRef]
- Lu, S.T.; Wu, Y.C.; Leou, S.P. Alkaloids of formosan Fissistigma and Goniothalamus species. Phytochemistry 1985, 24, 1829–1834. [Google Scholar] [CrossRef]
- Menezes, L.R.; Costa, C.O.; Rodrigues, A.C.; Santo, F.R.; Nepel, A.; Dutra, L.M.; Silva, F.M.; Soares, M.B.; Barison, A.; Costa, E.V.; et al. Cytotoxic Alkaloids from the Stem of Xylopia laevigata. Molecules 2016, 21, 890. [Google Scholar] [CrossRef]
- Zhang, Y.N.; Zhong, X.G.; Zheng, Z.P.; Hu, X.D.; Zuo, J.P.; Hu, L.H. Discovery and synthesis of new immunosuppressive alkaloids from the stem of Fissistigma oldhamii (Hemsl.) Merr. Bioorg. Med. Chem. 2007, 15, 988–996. [Google Scholar] [CrossRef]
- Fu, C.Y.; Yin, W.Q.; Zhou, Z.L. Studies on the aporphine alkaloids from Fissistigma oldhamii (Hemsl.) Merr. Zhong Yao Cai 2007, 30, 409–412. [Google Scholar]
- Kuo, R.Y.; Chang, F.R.; Chen, C.Y.; Teng, C.M.; Yen, H.F.; Wu, Y.C. Antiplatelet activity of N-methoxycarbonyl aporphines from Rollinia mucosa. Phytochemistry 2001, 57, 421–425. [Google Scholar] [CrossRef]
- Fu, C.Y.; Lu, Y.H.; Zhou, Z.L.; Yin, W.Q.; Chen, K.L. Inhibitory activity of the total alkaloids from Fissistigma oldhamii (Hemsl.) Merr. and its three aporphine alkaloids against pathogenic fungi. J. Henan Agric. Sci. 2010, 4, 70–72. [Google Scholar]
- Sala, E.; Guasch, L.; Iwaszkiewicz, J.; Mulero, M.; Salvadó, M.J.; Bladé, C.; Ceballos, M.; Valls, C.; Zoete, V.; Grosdidier, A.; et al. Identification of human IKK-2 inhibitors of natural origin (Part II): In Silico prediction of IKK-2 inhibitors in natural extracts with known anti-inflammatory activity. Eur. J. Med. Chem. 2011, 46, 6098–6103. [Google Scholar] [CrossRef]
- Zhong, R.J.; Li, H.Y.; Xie, E.L.; Wu, S.B.; Tang, J.; Zhou, G.P. HPLC simultaneous determination of fissistigine A and duguevanine in Fissistigma oldhamii (Hemsl.) Merr. Chin. J. Pharm. Anal. 2011, 1, 27–29. [Google Scholar]
- Zeng, L.; Xing, R.; Fu, C. Research Progress on Chemical Component and Pharmacological Activity of Fissistigma oldhamii (Hems.) Merr. Guangdong Chem. Ind. 2017, 3, 89–90. [Google Scholar]
- Perez, E.; Saez, J.; Blair, S.; Franck, X.; Figadere, B. Isoquinoline Alkaloids from Duguetia Vallicola Stem Bark with Antiplasmodial Activity. Lett. Org. Chem. 2004, 1, 102–104. [Google Scholar] [CrossRef]
- Chanakul, W.; Tuchinda, P.; Anantachoke, N.; Pohmakotr, M.; Piyachaturawat, P.; Jariyawat, S.; Suksen, K.; Jaipetch, T.; Nuntasaen, N.; Reutrakul, V. Cytotoxic alkaloids from stems, leaves and twigs of Dasymaschalon blumei. Fitoterapia 2011, 82, 964–968. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.T.; Wu, Y.C. A new aporphine alkaloid, fissoldine, from Fissistigma oldhamii (Hemsl.) Merr. Heterocycles 1983, 20, 813–815. [Google Scholar] [CrossRef]
- Chen, K.S.; Ko, F.N.; Teng, C.M.; Wu, Y.C. Antiplatelet and vasorelaxing actions of some aporphinoids. Planta Med. 1996, 62, 133–136. [Google Scholar] [CrossRef]
- Costa, E.V.; Pinheiro, M.L.; de Souza, A.D.; Barison, A.; Campos, F.R.; Valdez, R.H.; Ueda-Nakamura, T.; Filho, B.P.; Nakamura, C.V. Trypanocidal activity of oxoaporphine and pyrimidine-β-carboline alkaloids from the branches of Annona foetida Mart. (Annonaceae). Molecules 2011, 16, 9714–9720. [Google Scholar] [CrossRef]
- Gontijo, D.C.; Brandão, G.C.; Nascimento, M.F.A.D.; Oliveira, A.B. Antiplasmodial activity and cytotoxicity, isolation of active alkaloids, and dereplication of Xylopia sericea leaves ethanol extract by UPLC-DAD-ESI-MS/MS. J. Pharm. Pharmacol. 2019, 71, 260–269. [Google Scholar] [CrossRef]
- Rahman, M.M.; Lopa, S.S.; Sadik, G.; Harun-Or-Rashid Islam, R.; Khondkar, P.; Alam, A.H.; Rashid, M.A. Antibacterial and cytotoxic compounds from the bark of Cananga odorata. Fitoterapia 2005, 76, 758–761. [Google Scholar] [CrossRef]
- Costa, E.V.; Pinheiro, M.L.; Barison, A.; Campos, F.R.; Salvador, M.J.; Maia, B.H.; Cabral, E.C.; Eberlin, M.N. Alkaloids from the bark of Guatteria hispida and their evaluation as antioxidant and antimicrobial agents. J. Nat. Prod. 2010, 73, 1180–1183. [Google Scholar] [CrossRef]
- Zhou, Q.; Fu, Y.H.; Zhang, Y.Q.; Wu, S.Y.; Song, X.P.; Xu, W.; Chen, G.Y. A new morphinandienone alkaloid from the stems of Fissistigma tungfangense. Nat. Prod. Res. 2019, 33, 374–379. [Google Scholar] [CrossRef]
- Twin, H.; Wen, W.W.H.; Powell, D.A.; Lough, A.J.; Batey, R.A. A biogenetically inspired heterodimerization approach to the synthesis of the core structure of the alkaloid fissoldhimine. Tetrahedron Lett. 2007, 48, 1841–1844. [Google Scholar] [CrossRef]
- Luong, T.M.; Pilkington, L.I.; Barker, D. Stereoselective Total Synthesis of (+)-Aristolactam GI. J. Org. Chem. 2019, 84, 5747–5756. [Google Scholar] [CrossRef] [PubMed]
- Chia, Y.C.; Wu, J.B.; Wu, Y.C. Two novel cyclopentenones from Fissistigma oldhamii. Tetrahedron Lett. 2000, 41, 2199–2201. [Google Scholar] [CrossRef]
- Jia, X.; Liu, Y.; Li, X.; Huo, C.; Li, D.; Xu, R.; Hou, L.; Wang, X. Norcepharadione B attenuates H2O2-induced neuronal injury by upregulating cellular antioxidants and inhibiting volume-sensitive Cl- channel. Exp. Biol. Med. 2019, 244, 1463–1474. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, Y.; Chen, D.; Weng, H.; Li, H.; Lu, Y. Oral subacute nephrotoxicity of aristololactam I in rats. Toxicology 2022, 475, 153228. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Wang, C.; Bao, W.; Weng, H.; Chen, D.; Lu, Y. In vitro nephrotoxicity and quantitative UPLC-MS analysis of three aristololactams in Houttuynia cordata. J. Pharm. Biomed. Anal. 2023, 227, 115289. [Google Scholar] [CrossRef] [PubMed]
- Grollman, A.P. Aristolochic acid nephropathy: Harbinger of a global iatrogenic disease. Environ. Mol. Mutagen. 2013, 54, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Lone, I.A.; Athar, M.; Alam, M.S. Protective effect of Didymocarpus pedicellata on ferric nitrilotriacetate (Fe-NTA) induced renal oxidative stress and hyperproliferative response. Chem. Biol. Interact. 2007, 165, 33–44. [Google Scholar] [CrossRef]
- Ahmad, W.; Zaidi, S.M.A.; Ahmad, S. Quality control analysis of Didymocarpous pedicellata R. Br. Indian J. Tradit. Knowl. 2014, 13, 175–180. [Google Scholar]
- Fu, C.Y.; Liu, Y.H.; Chen, D.W.; Tang, H.; Feng, F.; Zhou, Z.L. Study on extraction and purification techniques and free radical scavenging activity of total flavonoids from Fissistigma oldhamii. Zhong Yao Cai 2011, 34, 446–449. [Google Scholar]
- Zou, C.; Zhao, Q.; Chen, C.X. The Structure of Lyciumide A. Plant Divers. 1999, 21, 1–3. [Google Scholar]
- Affes, M.; Fakhfakh, J.; Daoud, I.; Brieudes, V.; Halabalaki, M.; El Feki, A.; Allouche, N. UHPLC/HR-ESI-MS/MS Profiling of Phenolics from Tunisian Lycium arabicum Boiss. Antioxidant and Anti-lipase Activities’ Evaluation. Chem. Biodivers. 2017, 14, e1700095. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.M.; Zhang, B.; Zhang, Y.Q.; Chen, G.Y.; Xu, W.; Cai, J.; Liao, S. A new fatty acid methyl ester from Fissistigma oldhamii inhibiting proliferation of synoviocytes. Zhongguo Zhong Yao Za Zhi 2018, 43, 1754–1757. [Google Scholar] [PubMed]
- Kim, H.S.; Park, H.; Lim, J.; Lim, C.; Kim, T.; Lee, S.; Hur, J.; Sim, J.; Choi, H.J.; Suh, Y.G. Collective Syntheses of Guaiane Sesquiterpenes: Stereoselective Syntheses of (+)-Dysodensiol F, (+)-10β,14-Dihydroxy-allo-aromadendrane, and (-)-Dendroside C Aglycon. J. Org. Chem. 2020, 85, 13779–13792. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Zou, H.; Huang, G.; Chen, G.; Zhou, X.; Huang, S. Design, synthesis and anti-rheumatoid arthritis evaluation of double-ring conjugated enones. Bioorg. Chem. 2021, 109, 104701. [Google Scholar] [CrossRef]
- Wu, M.Z.; Xu, B.Q.; Zhang, X.Z.; Liu, S.; Luo, Y.P.; Zhou, X.M.; Chen, G.Y. Guaiane-Type Sesquiterpenes from the Stems of Fissistigma oldhamii. Chem. Biodivers. 2023, 20, e202300338. [Google Scholar] [CrossRef]
- Tu, Y.; Li, X.; Fu, Y.; Chen, Y.; Fang, H.; Li, Y.; Gu, Y.; Zhang, J. Isocorydine Ameliorates IL-6 Expression in Bone Marrow-Derived Macrophages and Acute Lung Injury Induced by Lipopolysaccharide. Int. J. Mol. Sci. 2023, 24, 4629. [Google Scholar] [CrossRef]
- Luo, J.; Wang, N.; Hua, L.; Deng, F.; Liu, D.; Zhou, J.; Yuan, Y.; Ouyang, F.; Chen, X.; Long, S.; et al. The Anti-Sepsis Effect of Isocorydine Screened from Guizhou Ethnic Medicine is Closely Related to Upregulation of Vitamin D Receptor Expression and Inhibition of NFκB p65 Translocation into the Nucleus. J. Inflamm. Res. 2022, 15, 5649–5664. [Google Scholar] [CrossRef]
- Hung, N.V.; Dai, D.N.; Thai, T.H.; Thang, T.D.; Ogunwande, I.A. Essential Oil from the Fruits of Fissistigma bracteolatum and Fissistigma maclurei. Chem. Sci. Int. J. 2016, 17, 1–7. [Google Scholar] [CrossRef]
- Höferl, M.; Dai, N.G.; Thang, T.D.; Jirovetz, L.; Schmidt, E. Leaf Essential Oils of Six Vietnamese Species of Fissistigma (Annonaceae). Nat. Prod. Commun. 2013, 8, 663–665. [Google Scholar] [CrossRef]
- Cheng, M.C.; Lin, L.Y.; Yu, T.H.; Peng, R.Y. Hypolipidemic and antioxidant activity of mountain celery (Cryptotaenia japonica Hassk) seed essential oils. J. Agric. Food Chem. 2008, 56, 3997–4003. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Jiménez, A.L.; González-Coloma, A.; Andrés-Yeves, M.F.; Ruiz-Sánchez, E.; Heredia, G.; Peraza-Sánchez, S.R.; Medina-Baizabal, I.L.; Reyes-Estebanez, M.; Canto-Canché, B.; Gamboa-Angulo, M. Insect deterrent and nematicidal screening of microfungi from Mexico and anti-aphid compounds from Gliomastix masseei. Rev. Argent. Microbiol. 2017, 49, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Alias, Y.; Awang, K.; Hadi, A.H.; Thoison, O.; Sévenet, T.; Païs, M. An antimitotic and cytotoxic chalcone from Fissistigma lanuginosum. J. Nat. Prod. 1995, 58, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Mizushina, Y.; Nakanishi, R.; Kuriyama, I.; Kamiya, K.; Satake, T.; Shimazaki, N.; Koiwai, O.; Uchiyama, Y.; Yonezawa, Y.; Takemura, M.; et al. Beta-sitosterol-3-O-beta-D-glucopyranoside: A eukaryotic DNA polymerase lambda inhibitor. J. Steroid Biochem. Mol. Biol. 2006, 99, 100–107. [Google Scholar] [CrossRef]
- Cascioferro, S.; Totsika, M.; Schillaci, D. Sortase A: An ideal target for anti-virulence drug development. Microb. Pathog. 2014, 77, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Alharthi, S.; Alavi, S.E.; Moyle, P.M.; Ziora, Z.M. Sortase A (SrtA) inhibitors as an alternative treatment for superbug infections. Drug Discov. Today 2021, 26, 2164–2172. [Google Scholar] [CrossRef]
- Mus, A.A.; Goh, L.P.W.; Marbawi, H.; Gansau, J.A. The Biosynthesis and Medicinal Properties of Taraxerol. Biomedicines 2022, 10, 807. [Google Scholar] [CrossRef]
- Huo, B.; Song, Y.; Tan, B.; Li, J.; Zhang, J.; Zhang, F.; Chang, L. Research on the mechanisms of taraxerol for the treatment of gastric cancer effect based on network pharmacology. Int. J. Immunopathol. Pharmacol. 2022, 36, 20587384211063962. [Google Scholar] [CrossRef]
- Liu, Z.; Yoon, C.S.; Lee, H.; Lee, H.K.; Lee, D.S. Linderone Isolated from Lindera erythrocarpa Exerts Antioxidant and Anti-Neuroinflammatory Effects via NF-κB and Nrf2 Pathways in BV2 and HT22 Cells. Int. J. Mol. Sci. 2023, 24, 7569. [Google Scholar] [CrossRef]
- Lu, Z.; Chen, H.; Lin, C.; Ou, G.; Li, J.; Xu, W. Ethnobotany of medicinal plants used by the Yao people in Gongcheng County, Guangxi, China. J. Ethnobiol. Ethnomed. 2022, 18, 49. [Google Scholar] [CrossRef]
- Xu, C.R. Studies on the chemical constituents of Fissistigma oldhamii. Zhong Yao Tong Bao 1982, 7, 30–31. [Google Scholar] [PubMed]
- Liu, H.; Chen, J.; Yuan, C.J.; He, J.; Chen, H.; Jin, C.; Guo, Q.; Huang, H. Rapid Identification of Alkaloids and Flavonoids in Fissistigma oldhamii var. longistipitatum by Ultra High-Performance Liquid Chromatography and Quadrupole Time-of-Flight Tandem Mass Spectrometry. J. Chromatogr. Sci. 2023, 61, 814–826. [Google Scholar] [PubMed]
- Jourjine, I.A.P.; Bauernschmidt, C.; Müller, C.; Bracher, F. A GC-MS Protocol for the Identification of Polycyclic Aromatic Alkaloids from Annonaceae. Molecules 2022, 27, 8217. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Shao, P.; Wang, Y.; Chen, Y.; Cui, S. Anti-inflammatory mechanism of the optimized active ingredients of Sargentodoxa cuneata and Patrinia villosa. Int. Immunopharmacol. 2023, 120, 110337. [Google Scholar] [CrossRef]
- Zou, W.; Gong, L.; Zhou, F.; Long, Y.; Li, Z.; Xiao, Z.; Ouyang, B.; Liu, M. Anti-inflammatory effect of traditional Chinese medicine preparation Penyanling on pelvic inflammatory disease. J. Ethnopharmacol. 2021, 266, 113405. [Google Scholar] [CrossRef]


| Plant Parts | Main Natural Products | References |
|---|---|---|
| Roots | Aristolactams A-B; corytuberine; physcion; stigmastanone; Z23 | [10,11,12] |
| Stems | Aristololactams AII-AIIIa, BII, FI-FII, GI-GII; calycinine; duguevalline; dysodensiols G-L; enterocarpam; fissistigamides A-B; fissistigine A; fissistigmine; fissistyramine; fissoldine; goniothalactam; isocorydine; isopedicin; lyciumide A; noraristolodione; norcepharadione B; oldhamactam; O-methylmoschatoline; oxocalycinine; oxodiscoguattine; oxoxylopine; piperolactams A-C; romucosine; stigmalactam; velutinam; xylopine | [10,13,14,15,16,17,18,19,20] |
| Leaves | γ-Cadinene; β-caryophyllene; β-ocimene | [21,22] |
| Fruits | Methyl-octadecenoate, methyl hexadecanoate, methyl oxononanoic acid; octadecenamide; pedicine; quercetin; rutin; β-sitosterol; taraxerol; taraxerenol | [23,24,25,26] |
| Seeds | Fissohamione; stigmahamones I–II | [8,16,27] |
| Patent (CIB #) | Title | Application |
|---|---|---|
| A23L 1/29 (201510503771.7) CN-24.05.2017 | Fissistigma oldhamii wind-dispelling dampness-eliminating soup base and preparation method. | Method to prepare a plant soup containing F. oldhamii, useful in promoting blood circulation, arresting pain, and relaxing tendons |
| A61K 36/896 (202111278310.6) CN-14.12.2021 | Traditional Chinese medicine patch for treating sciatica and preparation method of traditional Chinese medicine patch | A recipe to prepare a TCM patch for the treatment of sciatica, including roots of F. oldhamii and many other plants. |
| A23F 3/14 (201510482935.2) CN-24.05.2017 | Allophylus viridis Radlk wind-dispelling dampness-eliminating tea and preparation method. | Method to prepare a dampness-eliminating tea including roots of F. oldhamii and many other plants. The preparation is used to promote blood circulation to remove blood stasis and relax tendons. |
| A61K 36/899 (201610201188.5) CN-22.06.2016 | Traditional Chinese medicine composition for treating qi-blood weakness type dysmenorrhea. | Composition and preparation of a TCM tablet used to treat qi-blood weakness. It includes F. oldhamii and many other plants, used together to treat dysmenorrhea. |
| A61K 36/898 (201410322104.4) CN-17.09.2014 | Chinese herba preparation capable of treating qi and blood deficiency osteoarthritis and preparation method. | Method to prepare an herbal mixture of F. oldhamii and other plants, used to reduce phlegm and for dissipating stasis, dispelling dampness, and dredging collaterals. |
| A61K 36/87 (102016000646482) CN-23.11.2016 | Medicine for treating hepatitis containing herba senecionis scandentis. | A multiherbal TCM containing F. oldhamii, used internally and externally to combat hepatitis (prevention and treatment). |
| A61K 36/898 (201710376789.4) CN-15.09.2017 | Traditional Chinese medicine patch for treating hyperostosis. | F. oldhamii is included in a patch developed to promote nourishing yin, tonify the kidneys, expel wind, remove cold, remove dampness, and relieve pain. |
| A61K 36/899 (201310663260.2) CN-05.03.2014 | Traditional Chinese medicine for treating acute suppurative cholangitis. | Recipe for a multiherbal preparation including F. oldhamii, designed to treat suppurative cholangitis. |
| A23L 1/39 (102015000316564) CN-04.01.2017 | Schefflera arboricola wind dispelling and pain stopping oyster seafood soup materials and preparation method. | Recipe for a tasty oyster seafood soup including F. oldhamii, used for dispelling wind and stopping pain. |
| A61K 36/9066 (201410548829.5) CN-07.01.2015 | Traditional Chinese medicine preparation for treating apoplexy sequela due to vital energy deficiency and blood stasis and preparation method of traditional Chinese medicine preparation. | A TCM preparation containing F. oldhamii with various effects: benefiting vital energy, activating blood, strengthening healthy energy, and eliminating evil. For the treatment of the apoplexy sequela due to vital energy deficiency and blood stasis. |
| A61K 36/8945 (201510138462.4) CN-15.07.2015 | Qi and blood deficiency type migraine treating drug and preparation method. | A TCM preparation containing F. oldhamii for the treatment of qi- and blood-deficiency-type migraine. |
| A61K 31/575 (102016000273430) CN-31.08.2016 | Application of steroids in preparing drugs for treating rheumatoid arthritis. | Method to extract steroids from F. oldhamii and their use in treating rheumatoid arthritis. |
| A61K 36/84 (201710801775.2) CN-15.12.2017 | Decoction medicine for treating cervical and lumbar spine disease and preparation method. | Preparation method for a multiherbal TCM decoction including F. oldhamii used to treat cervical and lumbar spine diseases. |
| A61K 36/899 (201710436824.7) CN-22.09.2017 | Facial paralysis treating traditional Chinese medicine composition. | A TCM preparation containing F. oldhamii for treatment of facial paralysis with the following effects: wind evil dispelling, collateral dredging, heat clearing, blood circulation activation, phlegm dissipating, and nutrient qi regulation. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bailly, C. Fissistigma oldhamii (Hemsl.) Merr.: Ethnomedicinal, Phytochemistry, and Pharmacological Aspects. Plants 2023, 12, 4094. https://doi.org/10.3390/plants12244094
Bailly C. Fissistigma oldhamii (Hemsl.) Merr.: Ethnomedicinal, Phytochemistry, and Pharmacological Aspects. Plants. 2023; 12(24):4094. https://doi.org/10.3390/plants12244094
Chicago/Turabian StyleBailly, Christian. 2023. "Fissistigma oldhamii (Hemsl.) Merr.: Ethnomedicinal, Phytochemistry, and Pharmacological Aspects" Plants 12, no. 24: 4094. https://doi.org/10.3390/plants12244094
APA StyleBailly, C. (2023). Fissistigma oldhamii (Hemsl.) Merr.: Ethnomedicinal, Phytochemistry, and Pharmacological Aspects. Plants, 12(24), 4094. https://doi.org/10.3390/plants12244094






