LC-MS Analysis and Antifungal Activity of Turnera subulata Sm.
Abstract
1. Introduction
2. Results
2.1. Identification of Chemical Composition
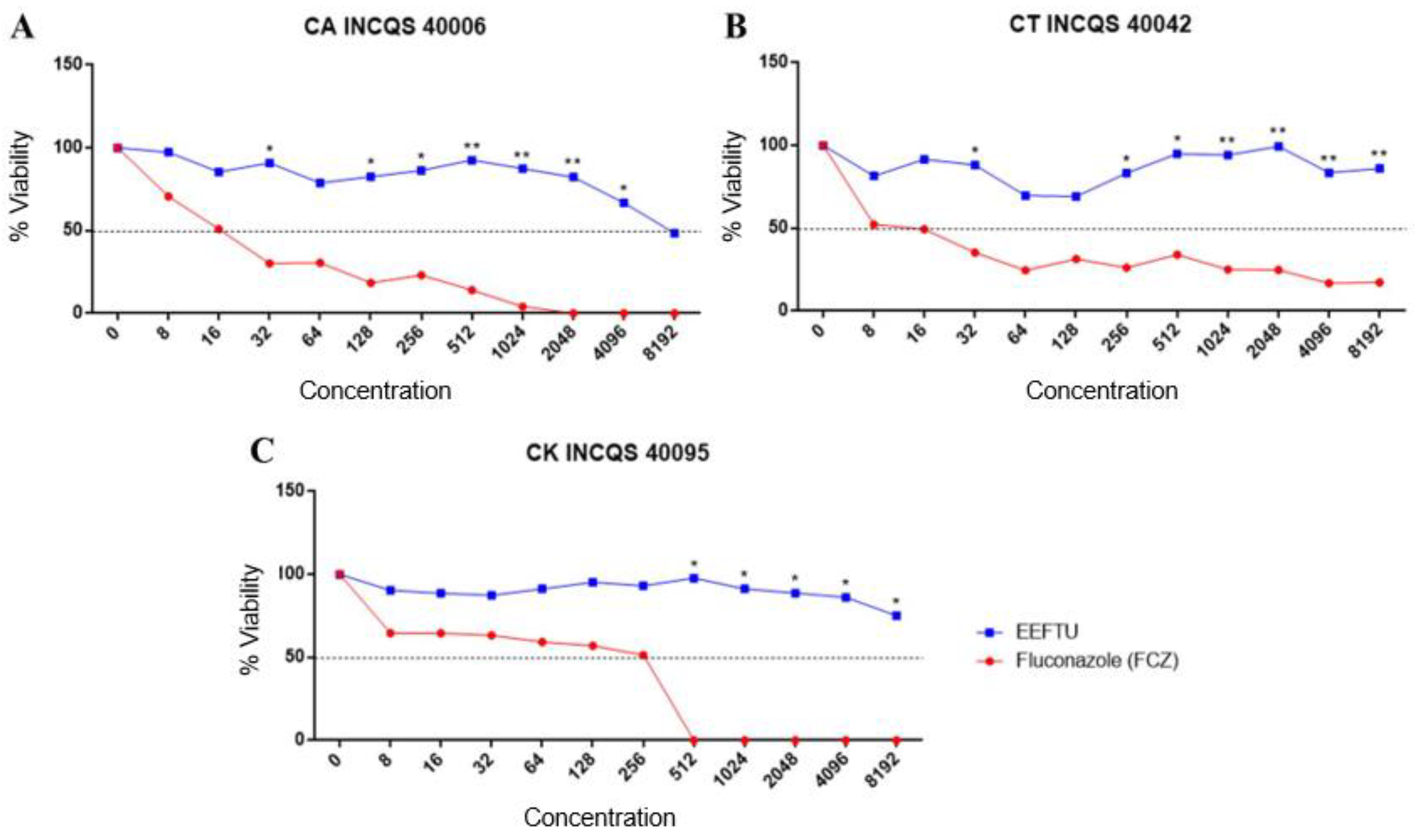
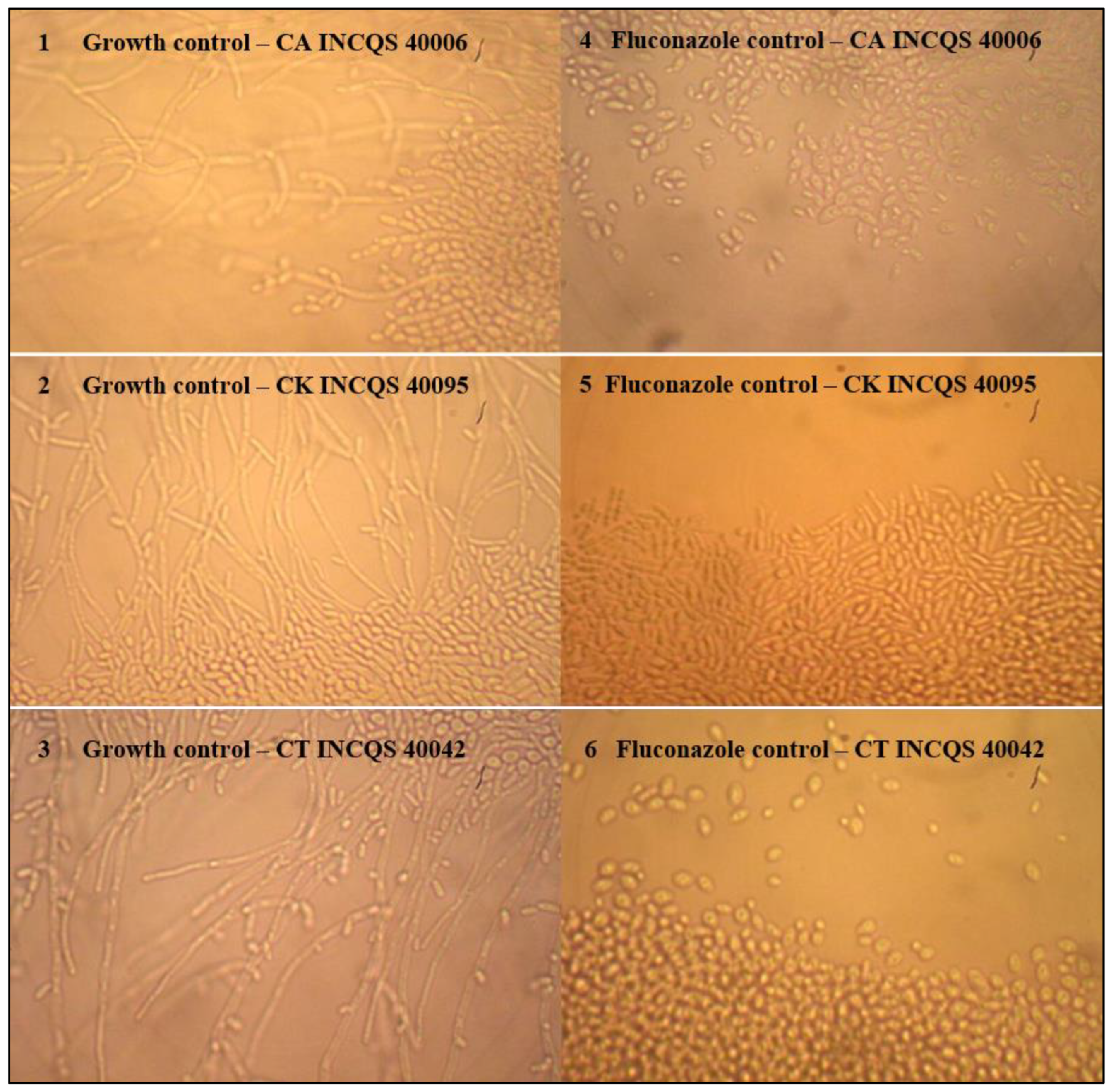
2.2. Antifungal Tests
3. Discussion
4. Materials and Methods
4.1. Plant Collection
4.2. Preparation of Ethanol Extract
4.3. LC-MS Conditions
4.4. Antifungal Assays
4.4.1. Microorganisms
4.4.2. Growth Media
4.4.3. Inoculum Preparation
4.4.4. Drugs and Reagents
4.4.5. Intrinsic Activity of the Antifungal Effect of EELTS and Fluconazole
4.4.6. Determination of the Minimum Fungicide Concentration
4.4.7. Effect of EELTS on the Fungal Dimorphism of Candida spp.
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Campos, T.; Simioni, P.U. Avaliação do Comportamento de Leveduras do Gênero Candida a Fármacos Antifúngicos. Rev. Cienc. Inovação-FAM 2020, 4, 1–17. [Google Scholar]
- Janbon, G.; Quintin, J.; Lanternier, F.; d’Enfert, C. Studying Fungal Pathogens of Humans and Fungal Infections: Fungal Diversity and Diversity of Approaches. Microbes Infect. 2019, 21, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Kadosh, D. Regulatory Mechanisms Controlling Morphology and Pathogenesis in Candida albicans. Curr. Opin. Microbiol. 2019, 52, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Kornitzer, D. Regulation of Candida Albicans Hyphal Morphogenesis by Endogenous Signals. J. Fungi 2019, 5, 21. [Google Scholar] [CrossRef]
- Sharma, J.; Rosiana, S.; Razzaq, I.; Shapiro, R.S. Linking Cellular Morphogenesis with Antifungal Treatment and Susceptibility in Candida Pathogens. J. Fungi 2019, 5, 17. [Google Scholar] [CrossRef]
- Albuquerque, H.; Dias, J.; Dantas, R.; Cabral, I.; Medeiros, A.; Santos, V. Potencial Atividade Antiulcerogênica da Espécie Spondias mombin. J. Biol. Pharm. Agric. Manag. 2015, 10. [Google Scholar]
- Almeida-Bezerra, J.W.; Costa, A.R.; de Freitas, M.A.; Rodrigues, F.C.; de Souza, M.A.; da Silva, A.R.P.; dos Santos, A.T.L.; Linhares, K.V.; Coutinho, H.D.M.; Silva, J.R.L.; et al. Chemical Composition, Antimicrobial, Modulator and Antioxidant Activity of Essential Oil of Dysphania ambrosioides (L.) Mosyakin & Clemants. Comp. Immunol. Microbiol. Infect. Dis. 2019, 65, 58–64. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.R.; Bezerra, J.W.A.; da Cruz, R.P.; de Freitas, M.A.; da Silva, V.B.; Neto, J.C.; Dos Santos, A.T.L.; Braga, M.F.B.M.; da Silva, L.A.; Rocha, M.I.; et al. In Vitro Antibiotic and Modulatory Activity of Mesosphaerum suaveolens (L.) Kuntze against Candida Strains. Antibiotics 2020, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Arbo, M.M. Turneraceae. In Flowering Plants· Eudicots; Springer: Berlin/Heidelberg, Germany, 2007; pp. 458–466. [Google Scholar]
- Arbo, M.M.; Espert, S.M. Morphology, Phylogeny and Biogeography of Turnera L. (Turneraceae). Taxon 2009, 58, 457–467. [Google Scholar] [CrossRef]
- Thulin, M.; Razafimandimbison, S.G.; Chafe, P.; Heidari, N.; Kool, A.; Shore, J.S. Phylogeny of the Turneraceae Clade (Passifloraceae s.l.): Trans-Atlantic Disjunctions and Two New Genera in Africa. Taxon 2012, 61, 308–323. [Google Scholar] [CrossRef]
- Arbo, M.M. Estudios Sistemáticos en Turnera (Turneraceae); IV. Series Leiocarpae, Sessilifoliae y Conciliatae. Bonplandia 2008, 17, 107–334. [Google Scholar] [CrossRef]
- Rocha, L.N.G.; de Melo, J.I.M.; Camacho, R.G.V. Flora of Rio Grande do Norte, Brazil: Turneraceae Kunth ex DC. Rodriguésia 2012, 63, 1085–1099. [Google Scholar] [CrossRef]
- Coutinho, H.D.M.; Costa, J.G.M.; Lima, E.O.; Falcão-Silva, V.S.; Siqueira, J.P. Herbal Therapy Associated with Antibiotic Therapy: Potentiation of the Antibiotic Activity against Methicillin-Resistant Staphylococcus aureus by Turnera ulmifolia L. BMC Complement. Altern. Med. 2009, 9, 1–4. [Google Scholar] [CrossRef]
- Coutinho, H.D.M.; Costa, J.G.M.; Lima, E.O.; Falcão-Silva, V.S.; Siqueira-Júnior, J.P. Increasing of the Aminoglicosyde Antibiotic Activity against a Multidrug-Resistant E. Coli by Turnera ulmifolia L. and Chlorpromazine. Biol. Res. Nurs. 2010, 11, 332–335. [Google Scholar] [CrossRef] [PubMed]
- Santos, K.K.A.; Matias, E.F.F.; Souza, C.E.S.; Tintino, S.R.; Braga, M.F.B.M.; Guedes, G.M.M.; Nogueira, L.F.B.; Morais, E.C.; Costa, J.G.M.; Menezes, I.R.A.; et al. Anti-Candida Activity of Mentha arvensis and Turnera ulmifolia. J. Med. Food 2012, 15, 322–324. [Google Scholar] [CrossRef]
- Oliveira, A.F.; Costa Junior, L.M.; Lima, A.S.; Silva, C.R.; Ribeiro, M.N.S.; Mesquista, J.W.C.; Rocha, C.Q.; Tangerina, M.M.P.; Vilegas, W. Anthelmintic Activity of Plant Extracts from Brazilian Savanna. Vet. Parasitol. 2017, 236, 121–127. [Google Scholar] [CrossRef]
- Antônio, M.A.; Souza Brito, A.R.M. Oral Anti-Inflammatory and Anti-Ulcerogenic Activities of a Hydroalcoholic Extract and Partitioned Fractions of Turnera ulmifolia (Turneraceae). J. Ethnopharmacol. 1998, 61, 215–228. [Google Scholar] [CrossRef]
- Gracioso, J.S.; Vilegas, W.; Hiruma-Lima, C.A.; Brito, A.R.M.S. Effects of Tea from Turnera ulmifolia L. on Mouse Gastric Mucosa Support the Turneraceae as a New Source of Antiulcerogenic Drugs. Biol. Pharm. Bull. 2002, 25, 487–491. [Google Scholar] [CrossRef]
- Nascimento, M.A.; Silva, A.K.; França, L.C.B.; Quignard, E.L.J.; López, J.A.; Almeida, M.G. Turnera ulmifolia L. (Turneraceae): Preliminary Study of Its Antioxidant Activity. Bioresour. Technol. 2006, 97, 1387–1391. [Google Scholar] [CrossRef]
- Henciya, S.; Seturaman, P.; James, A.R.; Tsai, Y.H.; Nikam, R.; Wu, Y.C.; Dahms, H.U.; Chang, F.R. Biopharmaceutical Potentials of Prosopis Spp. (Mimosaceae, Leguminosa). J. Food Drug Anal. 2017, 25, 187–196. [Google Scholar] [CrossRef]
- Abu-Reidah, I.M.; Contreras, M.M.; Arráez-Román, D.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Reversed-Phase Ultra-High-Performance Liquid Chromatography Coupled to Electrospray Ionization-Quadrupole-Time-of-Flight Mass Spectrometry as a Powerful Tool for Metabolic Profiling of Vegetables: Lactuca sativa as an Example of Its Application. J. Chromatogr. A 2013, 1313, 212–227. [Google Scholar] [CrossRef]
- Abdelaziz, S.; Al Yousef, H.M.; Al-Qahtani, A.S.; Hassan, W.H.B.; Fantoukh, O.I.; El-Sayed, M.A. Phytochemical Profile, Antioxidant and Cytotoxic Potential of Parkinsonia aculeata L. Growing in Saudi Arabia. Saudi Pharm. J. 2020, 28, 1129–1137. [Google Scholar] [CrossRef]
- Llorent-Martínez, E.J.; Spínola, V.; Castilho, P.C. Phenolic Profiles of Lauraceae Plant Species Endemic to Laurisilva Forest: A Chemotaxonomic Survey. Ind. Crops Prod. 2017, 107, 1–12. [Google Scholar] [CrossRef]
- Jin, J.; Lao, J.; Zhou, R.; He, W.; Qin, Y.; Zhong, C.; Xie, J.; Liu, H.; Wan, D.; Zhang, S. Simultaneous Identification and Dynamic Analysis of Saccharides during Steam Processing of Rhizomes of Polygonatum cyrtonema by HPLC–QTOF–MS/MS. Molecules 2018, 23, 2855. [Google Scholar] [CrossRef]
- Leyva-Jimenez, F.J.; Lozano-Sanchez, J.; Borras-Linares, I.; de la Luz Cadiz-Gurrea, M.; Mahmoodi-Khaledi, E. Potential Antimicrobial Activity of Honey Phenolic Compounds against Gram Positive and Gram Negative Bacteria. LWT 2019, 101, 236–245. [Google Scholar] [CrossRef]
- Brito, A.; Ramirez, J.E.; Areche, C.; Sepúlveda, B.; Simirgiotis, M.J. HPLC-UV-MS Profiles of Phenolic Compounds and Antioxidant Activity of Fruits from Three Citrus Species Consumed in Northern Chile. Molecules 2014, 19, 17400–17421. [Google Scholar] [CrossRef] [PubMed]
- Galvão, W.R.A.; Braz Filho, R.; Canuto, K.M.; Ribeiro, P.R.V.; Campos, A.R.; Moreira, A.; Silva, S.O.; Mesquita Filho, F.A.; Santos, S.; Junior, J.M.A.M. Gastroprotective and Anti-Inflammatory Activities Integrated to Chemical Composition of Myracrodruon urundeuva Allemão-A Conservationist Proposal for the Species. J. Ethnopharmacol. 2018, 222, 177–189. [Google Scholar] [CrossRef] [PubMed]
- Morcol, T.B.; Wysocki, K.; Sankaran, R.P.; Matthews, P.D.; Kennelly, E.J. UPLC-QTof-MSE Metabolomics Reveals Changes in Leaf Primary and Secondary Metabolism of Hop (Humulus lupulus L.) Plants under Drought Stress. J. Agric. Food Chem. 2020, 68, 14698–14708. [Google Scholar] [CrossRef] [PubMed]
- Costa, C.; Pires, C.; Cabrito, T.R.; Renaudin, A.; Ohno, M.; Chibana, H.; Sá-Correia, I.; Teixeira, M.C. Candida Glabrata Drug: H+ Antiporter CgQdr2 Confers Imidazole Drug Resistance, Being Activated by Transcription Factor CgPdr1. Antimicrob. Agents Chemother. 2013, 57, 3159–3167. [Google Scholar] [CrossRef] [PubMed]
- Alcazar-Fuoli, L.; Mellado, E. Current Status of Antifungal Resistance and Its Impact on Clinical Practice. Br. J. Haematol. 2014, 166, 471–484. [Google Scholar] [CrossRef]
- Braga, A.L.; da Cruz, R.P.; Carneiro, J.N.P.; dos Santos, A.T.L.; Sales, D.L.; Bezerra, C.F.; Fonseca, V.J.A.; Rocha, J.E.; de Freitas, T.S.; Campina, F.F.; et al. Piper regnellii (Miq.) C. DC.: Chemical Composition, Antimicrobial Effects, and Modulation of Antimicrobial Resistance. S. Afr. J. Bot. 2021, 142, 495–501. [Google Scholar] [CrossRef]
- Fernandes de Morais, L.V. Atividade Antimicrobiana e Antioxidante de Licania rigida e Turnera ulmifolia. Master’s Thesis, Centro de Ciências da Saúde, Universidade Federal do Rio Grande do Norte, Natal, Brazil, 2015; p. 64. [Google Scholar]
- Peralta, M.A.; Da Silva, M.A.; Ortega, M.G.; Cabrera, J.L.; Paraje, M.G. Antifungal Activity of a Prenylated Flavonoid from Dalea elegans against Candida albicans Biofilms. Phytomedicine 2015, 22, 975–980. [Google Scholar] [CrossRef]
- Jin, Y.S. Recent Advances in Natural Antifungal Flavonoids and Their Derivatives. Bioorg. Med. Chem. Lett. 2019, 29, 126589. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Kim, Y.G.; Khadke, S.K.; Lee, J. Antibiofilm and Antifungal Activities of Medium-Chain Fatty Acids against Candida albicans via Mimicking of the Quorum-Sensing Molecule Farnesol. Microb. Biotechnol. 2021, 14, 1353–1366. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, T.M.M.; Santos, A.T.L.; de Freitas, M.A.; Almeida-Bezerra, J.W.; Fonseca, V.J.A.; Coutinho, H.D.M.; Morais-Braga, M.F.B.; de Miranda, G.E.C.; Andrade-Pinheiro, J.C. Antifungal Activity of Gracilaria cervicornis (Turner) J. Agardh against Candida Spp. S. Afr. J. Bot. 2022, 150, 146–152. [Google Scholar] [CrossRef]
- Arendrup, M.C.; Patterson, T.F. Multidrug-Resistant Candida: Epidemiology, Molecular Mechanisms, and Treatment. J. Infect. Dis. 2017, 216, S445–S451. [Google Scholar] [CrossRef]
- Brasileiro, B.G.; Pizziolo, V.R.; Raslan, D.S.; Jamal, C.M.; Silveira, D. Antimicrobial and Cytotoxic Activities Screening of Some Brazilian Medicinal Plants Used in Governador Valadares District. Rev. Bras. Ciencias Farm. J. Pharm. Sci. 2006, 42, 195–202. [Google Scholar] [CrossRef]
- Souza, A.B.; Souza, L.M.S.; Carvalho, J.C.T.; Maistro, E.L. No Clastogenic Activity of Caesalpinia ferrea Mart. (Leguminosae) Extract on Bone Marrow Cells of Wistar Rats. Genet. Mol. Biol. 2006, 29, 380–383. [Google Scholar] [CrossRef]
- Javadpour, M.M.; Juban, M.M.; Lo, W.C.J.; Bishop, S.M.; Alberty, J.B.; Cowell, S.M.; Becker, C.L.; McLaughlin, M.L. De Novo Antimicrobial Peptides with Low Mammalian Cell Toxicity. J. Med. Chem. 1996, 39, 3107–3113. [Google Scholar] [CrossRef]
- Morais-Braga, M.F.B.; Sales, D.L.; Carneiro, J.N.P.; Machado, A.J.T.; Santos, A.T.L.; Freitas, M.A.; Martins, G.M.d.A.B.; Leite, N.F.; de Matos, Y.M.L.S.; Tintino, S.R.; et al. Psidium guajava L. and Psidium brownianum Mart Ex DC.: Chemical Composition and Anti-Candida Effect in Association with Fluconazole. Microb. Pathog. 2016, 95, 200–207. [Google Scholar] [CrossRef]
- Ernst, E.J.; Klepser, M.E.; Ernst, M.E.; Messer, S.A.; Pfaller, M.A. In Vitro Pharmacodynamic Characteristics of Flucytosine Determined by Time-Kill Methods. Diagn. Microbiol. Infect. Dis. 2000, 36, 101–105. [Google Scholar] [CrossRef]
- Sidrim, J.J.C.; Rocha, M.F.G. Micologia Médica à Luz de Autores Contemporâneos. Rio de Janeiro, RJ 2010; Guanabara Koogan: Rio De Janeiro, Brazil, 2010. [Google Scholar]
- Mendes, J.M. Investigação da Atividade Antifúngica do Óleo Essencial de Eugenia caryophyllata Thunb. Sobre Cepas de Candida tropicalis. Master’s Thesis, Universidade Federal da Paraíba, João Pessoa, Brazil, 2011; pp. 10–14. [Google Scholar]


| Peak N° | Rt Min | [M-H]- Observed | [M-H]- Calculated | Product Ions (MS/MS) | Empirical Formula | Ppm Error | Putative Identification | Ref. |
|---|---|---|---|---|---|---|---|---|
| 1 | 1.14 | 191.0550 | 191.0556 | - | C7H11O6 | −3.1 | Quinic acid * | [22] |
| 2 | 1.33 | 133.0133 | 130.0137 | - | C4H5O5 | −3.0 | Malic acid * | [22] |
| 3 | 2.05 | 130.0863 | 130.0868 | - | C6H12NO2 | −3.8 | Leucine * | [22] |
| 4 | 3.03 | 409.1877 | 409.1862 | 179, 119 | C21H29O8 | 3.7 | Unknown | - |
| 5 | 3.36 | 657.1106 | 657.1092 | 577, 341 | C30H25O17 | 2.1 | Unknown | - |
| 6 | 3.65 | 593.1507 | 593.1506 | 473, 429, 369, 357, 327 | C27H29O15 | 0.2 | Rhamnosyl isoorientin | [23] |
| 7 | 3.85 | 577.1560 | 577.1557 | 413, 293 | C27H29O14 | 0.5 | Rhamnosyl vitexin | [24] |
| 8 | 4.17 | 553.3001 | 553.3013 | 507, 375 | C29H45O10 | −2.2 | Unknown | - |
| 9 | 4.44 | 305.1603 | 305.1600 | 179, 161 | C14H25O7 | 1.0 | Hexose derivative | [25] |
| 10 | 4.78 | 187.0966 | 187.0970 | 125 | C9H15O4 | −2.1 | Azelaic acid | [26] |
| 11 | 5.35 | 577.1564 | 577.1557 | 269 | C27H29O14 | 1.2 | Apigenin 7-O-neohesperidoside | [27] |
| 12 | 5.75 | 577.1558 | 577.1557 | 269, 431 | C27H29O14 | 0.2 | Apigenin 7-O-rutinoside | [27] |
| 13 | 6.28 | 329.2322 | 329.2328 | 229, 211, 171 | C18H33O5 | −1.8 | Trihydroxyoctadecaenoic acid | [28] |
| 14 | 6.63 | 723.1807 | 723.1773 | 269 | C32H35O19 | 4.7 | Apigenin derivative | [27] |
| 15 | 6.82 | 725.1939 | 725.1929 | 453, 271 | C32H37O19 | 1.4 | Unknown | |
| 16 | 7.76 | 293.1766 | 293.1753 | 236, 221 | C17H25O4 | 4.4 | Unknown terpene | - |
| 17 | 8.94 | 675.3601 | 675.3592 | 415, 397, 277, 235 | C33H55O14 | 1.3 | Digalactosylmono acylglycerol | [29] |
| 18 | 10.38 | 555.2793 | 555.2805 | 255 | C28H43O11 | −2.2 | Unknown fatty acid | - |
| Tested Products | Strains | ||
|---|---|---|---|
| CA INCQS 40006 | CT INCQS 40042 | CK INCQS 40095 | |
| Fluconazole (FCZ) | 16.70 μg/mL | 9.30 μg/mL | 133.32 μg/mL |
| EELTS | 7544.60 μg/mL | - | 16,087.37 μg/mL |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andrade-Pinheiro, J.C.; Sobral de Souza, C.E.; Ribeiro, D.A.; Silva, A.d.A.; da Silva, V.B.; dos Santos, A.T.L.; Juno Alencar Fonseca, V.; de Macêdo, D.G.; da Cruz, R.P.; Almeida-Bezerra, J.W.; et al. LC-MS Analysis and Antifungal Activity of Turnera subulata Sm. Plants 2023, 12, 415. https://doi.org/10.3390/plants12020415
Andrade-Pinheiro JC, Sobral de Souza CE, Ribeiro DA, Silva AdA, da Silva VB, dos Santos ATL, Juno Alencar Fonseca V, de Macêdo DG, da Cruz RP, Almeida-Bezerra JW, et al. LC-MS Analysis and Antifungal Activity of Turnera subulata Sm. Plants. 2023; 12(2):415. https://doi.org/10.3390/plants12020415
Chicago/Turabian StyleAndrade-Pinheiro, Jacqueline Cosmo, Celestina Elba Sobral de Souza, Daiany Alves Ribeiro, Andressa de Alencar Silva, Viviane Bezerra da Silva, Antonia Thassya Lucas dos Santos, Victor Juno Alencar Fonseca, Delmacia Gonçalves de Macêdo, Rafael Pereira da Cruz, José Weverton Almeida-Bezerra, and et al. 2023. "LC-MS Analysis and Antifungal Activity of Turnera subulata Sm." Plants 12, no. 2: 415. https://doi.org/10.3390/plants12020415
APA StyleAndrade-Pinheiro, J. C., Sobral de Souza, C. E., Ribeiro, D. A., Silva, A. d. A., da Silva, V. B., dos Santos, A. T. L., Juno Alencar Fonseca, V., de Macêdo, D. G., da Cruz, R. P., Almeida-Bezerra, J. W., Machado, A. J. T., de Freitas, T. S., de Brito, E. S., Ribeiro, P. R. V., da Costa, J. G. M., Coutinho, H. D. M., Kowalska, G., Rowiński, R., Kowalski, R., & Morais-Braga, M. F. B. (2023). LC-MS Analysis and Antifungal Activity of Turnera subulata Sm. Plants, 12(2), 415. https://doi.org/10.3390/plants12020415









