Abstract
Non-coding RNAs play vital roles in the diverse biological processes of plants, and they are becoming key topics in horticulture research. In particular, miRNAs and long non-coding RNAs (lncRNAs) are receiving increased attention in fruit crops. Recent studies in horticulture research provide both genetic and molecular evidence that miRNAs and lncRNAs regulate biological function and stress responses during fruit development. Here, we summarize multiple regulatory modules of miRNAs and lncRNAs and their biological roles in fruit sets and stress responses, which would guide the development of molecular breeding techniques on horticultural crops.
Keywords:
miRNA; long non-coding RNA; stress response; fruit; agricultural traits; CRISPR; molecular breeding 1. Introduction
Fruits are differentiated from a mature ovary of the pistil after fertilization or floral organs and comprise a variety of pericarp and seed tissue types, which can be classified as dry or fleshy according to different pericarp textures [1]. Dry fruits or their seeds are usually derived from ovary tissues, such as Arabidopsis thaliana, which produces dry dehiscent fruits (silique) [2]. Fleshy fruits are derived mostly from hypanthium tissues that are hypothesized to consist of the fused bases of the sepals, petals and stamens [1], such as tomato (Solanum lycopersicum) and strawberry (Fragaria × ananassa). Fleshy fruits play important nutritious and health roles in the human diet, and several characteristics of the fleshy fruit, including color, flavor, aroma, texture and nutrition, have been studied for their dramatic changes during growth [3,4].
Likewise, although there is a remarkable convergence between fleshy fruit species, regardless of the ontogeny of the fruit, the processes involved in fleshy fruit development can be very different. For example, tomato fruit ripens in climacteric patterns, while strawberry fruit softening during ripening is accompanied by non-climacteric behavior [5]. Fruit development is under regulation at a complex molecular level and it explains the dynamics that alter the size, color, firmness, taste and flavor, which are characteristics intimately related to fruit quality. In addition, disease-resistant and stress-tolerant varieties also contribute an important part to fruit quality, fruit production and economic value. Fruit analysis provides insights into the potential for fruit crop improvement strategies and consequently applies to agricultural production [6,7].
More recently, a great number of functional genomics has shown that non-coding RNAs, especially miRNA and lncRNA, are involved in a diversity of developmental reproductive stages, from carpel formation and ovary development to the softening of the ripe/ripened fruit [8,9,10]. Moreover, numerous genetic studies have also shown that miRNA and lncRNA regulation results in fruit development alteration, including organ pattern, fruit shape and size, as well as their developmental progress, such as miR159 involved in fruit set [11], miR160 associated with fruit shape [12], miR164 affecting locule number [13,14] and miR156 regulating fruit softening [15]. This layer of transcriptional control has been associated with ovule, seed and fruit development and fruit ripening, as well as stress responses [16,17], which are crucial developmental processes in breeding programs because of their relevance for crop production. In addition, the final ripe fruit is the result of a process under multiple complicated levels of regulation that acts to coordinate the main steps of fruit development/patterning and fruit ripening, including mechanisms orchestrated by miRNA and lncRNA.
Although miRNA and lncRNA are particularly well studied and well known in fleshy fruit, the functions of miRNA involved in development processes are different in fruit species. In contrast, lncRNAs are usually less evolutionarily conserved, but the broad functions of lncRNAs are still possible under certain interactions. In this review, we discuss miRNA/lncRNA-controlled mechanisms described in the current literature that act to coordinate the main steps of fruit development/patterning, fruit ripening and fruit stress responses. Moreover, we discuss the main aspects of agronomic traits in fleshy fruits, such as yield (fruit size, fruit set), quality (parthenocarpic fruit) and stress tolerance, and explore the outlook for uses of miRNA/lncRNA-associated traits in fruit biotechnology and fruit breeding. In such a way, we present the latest advances in miRNA and lncRNA validation and the functional analysis as strong evidence for the regulatory roles in fruit biology (Table 1).
2. Functions of miRNAs and lncRNAs in Fruit Development
Fruit size and/or fruit number are crucial for improving yield and have a positive impact on consumer preference. It has been reported that the genes controlling tissue identity are involved in modulating fruit morphology, such as MADS-box genes, which are known to regulate floral organ identity, thereby regulating fruit development in Arabidopsis [18]. The regulatory module of miR172-AP2 has been highlighted in regulating fruit development in diverse plants. In Arabidopsis, miR172 promotes the silique fruit expansion process by the negative regulation of the activity of the APETALA2-like (AP2) gene [19], which would otherwise hinder the action of AGAMOUS (AG) and FRUITFUL (FUL) [20], two MADS-box transcription factors that are essential for ovary and silique growth [21]. miR172 has an adverse influence on fruit size in apples (Malus × domestica) through the negative regulation of AP2 that is required for hypanthium development into a pome fruit [22], resulting in small fruit size and an abnormal floral organ [23]. Another study in tomato, an ovary-derived fleshy fruit, revealed that the SlMIR172c and SlMIR172d loss-of-function mutant lines (slmir172c-dCR) resulted in abnormal flower organ and number identity [24]. These findings suggested a different role of miRNAs in dry and fleshy fruit. miRNAs regulate endogenous genes to impact development responses and even drive crop domestication; these results are consistent considering that silique is a true fruit deriving from ovary tissues, while the pome is a false fruit developing mainly from extra-carpellary tissues, such as sepals.
MIR172 encodes highly similar miR172s, but exhibits differences in their distribution among fruit growth. These different biological functions and miRNA patterns in Arabidopsis, apple and tomato indicate the parallel evolution of the miRNA machinery in different fruit types. There are seven genes (SlMIR172a–g) that code for four unique species of miR172 (sly-miR172) in tomato [25], fifteen genes (mdm-MIR172a–o) in apple [26] and five genes (ath-miR172a–e) in Arabidopsis [27]. A recent study shows that the whole genome duplication (WGD) event of Populus trichocarpa stimulates the emergence of new miRNAs [28]. The number variety of miRNAs in different species may have resulted from the whole genome duplication event, thus contributing to the functional specialization of miRNAs and the functional importance of MIR genes. Many miRNAs are species-specific rather than conserved, which supports fruit type-specific divergence in miRNA evolution. Divergence in miRNAs or targets may have played important roles in horticultural crop domestication; for example, a loss-of-function mutation in MIR172p improved fruit size during apple domestication [22].
In addition, it has been observed that sly-MIR156a-c expressed in placenta, ovules and pre- and post-anthesis flowers in tomato [29], when overexpressing miR156a–c, resulted in the enhancement of vegetative development, a delay in flowering time, and a smaller number of fruits that presented ectopic leaf-like structures [15]. Moreover, the overexpression of tomato miR156 altered the expression of miR164, which is related to organ identity as well as carotenoid biosynthesis [15], suggesting that the miRNA–miRNA crosstalk and other molecular networks are also involved in fruit development. It has also been reported that miR156–miR172 pairs perform a negative correlation in flowering induction in A. thaliana, Nicotiana tabacum, D. glomerate and Oryza sativa [30,31,32,33], suggesting that miRNA–miRNA crosstalk plays an important role in the development of plant sexual organs.
Furthermore, other miRNA regulation modules have also been identified in regulating fruit size and number (Table 1, Figure 1A,B). The sly-miR171a gene regulates hormone crosstalk between auxin and gibberellin in fruit size/weight by targeting two members of the GRAS family (SlGRAS24 and SlGRAS40) known as hormone regulators [34]. In this way, SlGRAS24 silencing results in GA3 and IAA accumulation, which leads to cell division and cell growth, and then floral initiation and seed number alteration [35,36]. Furthermore, sly-miR396a-3p/5p and sly-miR396b are mainly expressed in fruit, highlighting their potential role in fruit development. Knocking down miR396 by short tandem target mimic (STTM) showed an increase in fruit weight (66%), sepal size (153%), cell number (99%) and size (65%) [37], suggesting that the attenuation of miR396 results in the enhancement of some key performance indicators for fruit production. It has also been observed that knocking out miR164a by CRISPR/Cas9 to release the expression of NAM2/3 leads to decreased tomato fruit size [38]. The knocking down of miR1917 targeting an ethylene response gene CTR4 in tomato leads to bigger fruit [39]. It is reported that sly-miR159 is essential for fruit growth in Arabidopsis, and the mir159ab double mutant leads to small siliques [40], while its silencing results in larger fruits in tomato [41], suggesting fruits developed from the ovary may have evolved a different role of miRNAs in dry and fleshy fruit.
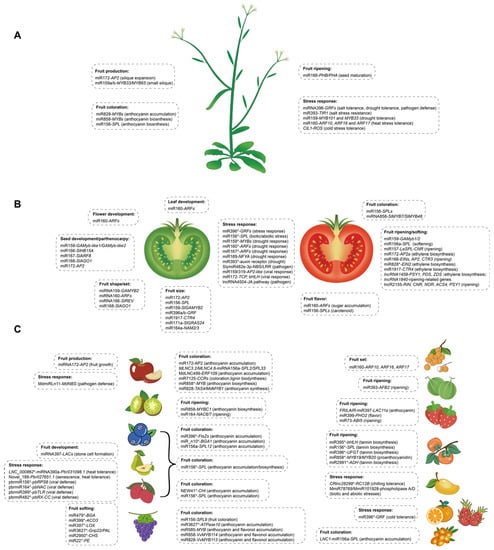
Figure 1.
Diverse function of miRNA and lncRNA members in fruit crops: (A) Arabidopsis, (B) tomato and (C) fruit crops (fruit species on left: apple, kiwifruit, blueberry, pear, litchi, grape; fruit species on right: longan, melon, strawberry, persimmon, mango, orange, pitaya, and sea buckthorn). The asterisk (*) represents bioinformatics analysis data.
Fruit set or fruit shape is a crucial indicator of fruit development affecting the economic values of fruit and consumers’ preferences. During the initial fruit growth phase, rapid growth in fruit length and width, mainly due to rapid cell division and expansion driven by phytohormones such as auxin and gibberellins (GAs), is observed in the tissues surrounding fertilized ovules [42]. It has long been known that auxin is involved in plant growth as well as development and fruit set [43,44,45]. Early studies revealed that the knockdown of sly-miR160a by STTM technology resulted in the alteration of floral organ abscission and auxin-mediated ovary patterning as well as fruit shape through the post-transcriptional regulation of the auxin response factors ARF10A, ARF10B and ARF17 [12]. The overexpression of ARF10A resulted in greener fruits before the BR stage, enhanced photosynthesis rate and sugar accumulation [12,46]. These results suggest the important roles of sly-miR160 auxin-mediated fruit shape and sugar accumulation in tomato [47].
Moreover, many transcript factors (TFs) are miRNA targets that regulate key genes involved in the floral induction and flower formation processes such as transition phases from juvenile to adult, the initiation of floral competence and flower development [48]. A large number of species-specific miRNAs have also been identified in tomato fruit development [49]. For example, silencing miR159 induced more locules inside the tomato fruit [11]. The miR159–SlGAMYB1/2 module is involved in tomato ovary development and fruit set by modulating auxin and gibberellin responses during ovule and ovary development [11]. Additionally, the overexpression of an miR166-resistant mutant of REV (35S::REVRis) results in ectopic fruits on receptacles and fused fruits [50], indicating that the functioning of REV mainly depends on the regulation of miR166 at the posttranscriptional level. In addition, is reported that the overexpression of PbrmiR397a reduced the lignin content and stone cell number in pear fruit (Pyrus bretschneideri) by inhibiting laccase (LAC) genes that encode key lignin biosynthesis enzymes [51]. The genome analysis of a single nucleotide polymorphism (SNP) in the promoter of PbrmiR397a between sixty pear varieties was found to be associated with low levels of fruit lignin [51]. Moreover, miR160 regulates longan somatic embryo development by targeting ARF10, ARF16 and ARF 7 [52]. Altogether, these results unveil the function of the miRNA-targeted key genes in regulating an agronomically important trait.
In addition, transcriptome-wide analyses have revealed the important regulatory roles of lncRNAs in a set of fruit developmental processes, such as lncRNAs function in regulating flower and fruit development in strawberry [53], peach (Prunus persica) [54] and apple [55]. Although many lncRNAs have been identified in diverse fruit species during fruit developmental stages, their biological functions will be fully elucidated in the future (Table 1, Figure 1A–C).
Parthenocarpic or seedless fruits are an important agricultural trait and a desirable trait for consumers. Methods of achieving seedless/parthenocarpic fruits have received increasing attention from researchers and breeders. Fruit and seed development held a distinct place in plant propagation and harvesting for defining crop yields; therefore, the roles played by the miRNAs in the development of fruits and seeds are of great interest. In tomato, the interaction of AGO1s–miR168 not only determines fruit initiation and growth, but also exhibits parthenocarpy in miR168-resistant 4m-SlAGO1A [56]. The overexpression of miR159 induced parthenocarpy as well as the downregulation of miR167, releasing the expression of SlARF8 [11]. It was also observed that the SlHB15A transcript is recessive-dosage-sensitivity-controlled by miR166, and Slhb15a knockdown alleles by miR166 lead to a parthenocarpic fruit set [57]. The MIR172 gene loss-of-function mutant line (slmir172c-dCR) produces an abnormal ovary expanded to form small parthenocarpic fruit-like organs [24].
It is reported that the molecular mechanisms of parthenocarpic fruit formation are mainly related to plant hormones, for example, cytokinin-treated pear (Pyrus spp.) and kiwifruit (Actinidia chinensis) [58,59], gibberellic acid-treated and auxin-induced tomato [60,61], brassinosteroids-induced apple [62], cytokinin-regulated and auxin-regulated grape (Vitis vinifera) [63] and gibberellic acid-maintained citrus (Citrus clementina) [64]. It is well established that parthenocarpy/seedless work in various fruits contributes to improving the taste quality of the fruit.
3. Functions of miRNAs and lncRNAs in Fruit Ripening
Fruit color variation is the most important agricultural trait of fruit ripening and chiefly affects the postharvest texture and consumers’ preferences. miRNAs have been extensively investigated in fruit development, and they also play an important role in fruit ripening. Interestingly, some miRNA regulations work in the same biological processes during fruit ripening. It has been found that the miR156-SPLs [65,66,67] and miR828/858-MYBs [68,69,70,71,72] modules are the conserved pathways to regulate fruit coloration in various fruit crops. For example, the miR156a-SPL12 module manipulates the accumulation of chlorophylls and anthocyanins during fruit ripening in blueberry, in which VcSPL12 interacts with VcMYBPA1 [66]. In pear, miR156-targeted SPLs interfere with the MYB-bHLH-WD40 complex in anthocyanin biosynthesis [73]. The transient overexpression of VvmiR156b/c/d in tomato promotes fruit coloring by repressing VvSPL9 transcription [74], suggesting that VvmiR156b/c/d-mediated VvSPL9 is involved in the formation of grape color. Similarly, in litchi (Litchi chinensis Sonn.), miR156a-targeted LcSPL1, interacting with LcMYB1, functions as a major cue in anthocyanin biosynthesis [67]. Moreover, the overexpression of miR156 promotes the accumulation of anthocyanins by targeting SPL9 in Arabidopsis, which negatively inhibits anthocyanin biosynthetic genes through the destabilization of an MYB-bHLH-WD40 transcriptional activation complex [65]. Another study reveals that long non-coding RNA MLNC3.2 and MLNC4.6 function as eTMs for miR156a and suppress the miR156a-mediated cleavage of SPL2-like and SPL33 during light-induced anthocyanin accumulation in apple fruit [75]. Similarly, the lncRNA LNC1-induces the downregulation of SPL9 through endogenous target mimics of miR156a, which leads to increased anthocyanin content in sea buckthorn (Hippophae rhamnoides Linn.) [76]. Moreover, ncRNAs associated with the anthocyanin biosynthesis pathway have also been reported in various regulatory modules, such as NEW41-CHI in litchi [67], miR396-FtsZs and miR_n10-BAG1 associated with blueberry [77] and miR172-AP2, miR7125-MYB16/MYB1 and MdLNC499-ERF109 involved in apple [78,79,80], all of which have been identified in anthocyanin accumulation (Table 1).
In addition, miR828 triggers the biogenesis of phasiRNAs that, in trans or in cis, regulate multiple MYBs that are involved in anthocyanin accumulation [8,68,69]. These MYBs belong to the R2R3 class, which is integrated with multiple biological processes, particularly in plant anthocyanin biosynthesis [81]. In Arabidopsis, the overexpression of miR828 reduces anthocyanin accumulation by repressing genes encoding MYB transcription factors [68]. In tomato, miR858 plays a negative role in anthocyanin biosynthesis, and the blockage of miR858 leads to increased anthocyanin accumulation by modulating the expression of SlMYB7 and SlMYB48 [70]. Another report demonstrates that miR858a represses the translation of MYBL2 in Arabidopsis seedlings as a positive regulator of anthocyanin biosynthesis [72]. In grape, miR828/miR858 targets VvMYB114, which is reported as being essential for anthocyanin and flavonol accumulation [69]. The transient overexpression of miR858 reduces anthocyanin accumulation in kiwifruit (Actinidia arguta) by repressing the target gene MYBC1 [71]. Among them, miR828 and miR858 could directly or indirectly control anthocyanin biosynthesis in apple [26]; for example, a recent study found that the overexpression of mdm-miR828 inhibited anthocyanin synthesis through the cleavage of MdTAS4 in the late fruit coloration stage, and MdMYB1 was induced in a feedback regulatory mechanism through binding to the promoter of mdm-MIR828b to promote its expression [82].
Fruit ripening is a complex biological process and is associated with many aspects of fruit flavor and textural alterations. In persimmon (Diospyros kaki Thunb.), miR395p-3p and miR858b regulate bHLH and MYB, respectively, which synergistically regulate the structural genes responsible for tannin biosynthesis [83]. In addition, many miRNAs’ target genes have been identified through high-throughput sequencing associated with regulating persimmon fruit ripening, such as miR156-SPL, miR396-UFGT, miR858-MYB19/20 and miR2991-ADH [83]. Another study on strawberry shows that the overexpression of miR399 can improve fruit quality by targeting PHO2 [84]. A novel miRNA, Fan-miR73, negatively regulates its target gene, ABI5, to control strawberry fruit ripening [85]. Knocking down pre-slymiR157 or mature slymiR157 delays tomato fruit ripening by targeting LeSPL-CNR, in turn regulating the expression of LeMADS-RIN, LeHB1, SlAP2a and SlTAGL1 [86,87]. Additionally, miR164-NAC6/7 and miR393-AFB2 are associated with fruit ripening in kiwifruit and melon (Cucumis melo) [88,89] (Table 1). These sophisticated regulatory networks might provide the accurate regulation of fruit ripening in different plants.
LncRNAs also play important roles in the fleshy fruit-ripening process. The genome-wide discovery and characterization of novel species-specific lncRNAs in fruits were conducted in various fleshy fruit species, including tomato [10,90], strawberry [53], apple [91], grape [92], kiwifruit [93], peach, mume (Prunus mume) [94], sea buckthorn [95] and melon [96]. These results present the global function of lncRNAs in different fruit species, which provides new insights into the regulation of fruit quality.
In strawberry, color change in wild varieties of Fragaria pentaphylla (F. pentaphylla) may be largely regulated by lncRNAs [97]. In tomatoes, silencing two lncRNAs, lncRNA1459 and lncRNA1840, delayed the fruit-ripening processes, which indicated the positive regulatory roles of the two members in the fruit-ripening process [10]. Furthermore, knocking down lncRNA1459 by CRISPR/Cas9 genome editing technology affected lycopene, carotenoid and ethylene biosynthesis [17]. Moreover, in tomato, 187 lncRNAs were found to be direct targets of the MADS-box transcription factor (TF) RIPENING INHIBITOR (RIN), which is a critical TF of fruit ripening [98,99]. In the fleshy fruit species, lncRNAs were also reported to be the key regulators with miRNAs under sophisticated control to perform their proper function. Some research has shown that long non-coding RNA (lncRNA) could regulate miRNAs as endogenous target mimics (eTMs) and participate in anthocyanin accumulation, such as MLNC3.2 and MLNC4.6 in apple [75] and LNC1 in sea buckthorn [76]. In strawberry, the lncRNA FRILAIR serves as a miRNA sponge by functioning as a noncanonical target mimic of strawberry miR397, which can guide the mRNA cleavage of the fruit-ripening accelerating gene LAC11a, thereby regulating the fruit-ripening process [100]. Knocking out lncRNA2155 by CRISPR/Cas9 technology delayed tomato fruit ripening with downregulated ripening-related genes, including RIN, CNR, NOR, ACS4 and PSY1 [98].
Fruit softening and fruit texture are also crucial for optimizing fruit quality. In addition, several ncRNAs are involved in fruit softening. Knocking down pre-miR156a–c or their mature SlymiR156a sequences through the VIGS system accelerates tomato fruit softening after the red ripe stage [87]. Additionally, miR479-BGA, miR2950-CHS, miR22-PE, miR3627-PAL and miR399-ACO3 are associated with fruit softening in grapes [101] (Table 1). Furthermore, the overexpression of miR399a can promote the accumulation of fructose and glucose in wild strawberry fruit [84]. In apple, the overexpression of miR7125 reduces lignin biosynthesis by targeting MdCCR during light induction [79]. Taken together, it will be important to extensively explore the underlying mechanisms in fruit ripening.
Plant hormones in fruit ripening are necessary, and the molecular mechanism and the signaling cascades of plant hormones during fruit ripening have been extensively studied in horticultural plants [102]. Non-coding RNAs are also involved in phytohormone regulation networks, such as ethylene (ETH), which is the major phytohormone in climacteric fruit ripening [103,104]. Tomato miR172 targets SlAP2 cleavage to accelerate fruit ripening and enhance ethylene biosynthesis [105]. Furthermore, slymiR1917 was reported as a negative regulator of two ET-related CTR4 splicing variants, but it is also regulated to ACS2 and ACS4, which are key genes for the establishment of the type of ET synthesis pathway [106]. In particular, Ethylene Insensitive 2 (EIN2) is targeted by miR828 [107], therefore for the onset of ethylene-dependent ripening events, a strong reduction of the expression of both miR394 and miR828 is required in tomato [108]. Moreover, some other miRNAs were found as regulators of some ET-related genes, such as the overexpression of the miR166-resistant version of SlREV downregulating EIN3, ERFs, AP2 and CTR3 in tomato [52]. The interplay may provide a mechanism to enable flexible fruit ripening. Several different types of non-coding RNAs are involved in regulating the expression of ripening genes, but further clarification of their diverse mechanisms of action is required. Further investigation might help to understand whether this behavior is relevant for development and if there are some other offset mechanisms in terms of time to ripen.
4. Functions of miRNAs and lncRNAs in Fruit Responses to Biotic and Abiotic Stress
Stress tolerance is an important breeding objective and selection criteria in breeding that is critical for fruit quality, such as disease-resistant varieties, cold-resistant varieties and drought-resistant varieties. Besides the role of miRNAs and lncRNAs in growth, development and ripening, they also act as important signaling components in stress responses. They are key modulators of the transcriptional and post-transcriptional expression of genes during defense responses, and they are shown to be required for adaptation to the changes in ambient environments. Stress-induced changes occur in multiple species and correlate with a conserved mechanism involving non-coding RNA regulations.
Salinity stress usually causes physiological disorders in fruit crops. During salinity conditions, numerous gene transcripts are variably regulated by miRNAs. The auxin signaling plays an important role in the biotic stress response, and the miR393-mediated regulation of the auxin receptor TIR1 is involved in the response to salt stress resistance and ABA-signaling pathways [109,110]. Furthermore, the miR396-GRF module was shown in pitaya (Hylocereus polyrhizus) and Arabidopsis [111,112]. Interestingly, a wide range of miRNAs was induced in date palms (Phoenix dactylifera L.) and mandarin (Citrus reticulata Blanco) under salt stress conditions [113,114], which provides insight into plants’ adaptation to salinity.
High or low temperature stress at the fruit development stage is an important factor that determines fruit quality and fruit storage time, and hot or cold temperatures influence plant growth and yields. Several miRNAs induced by high-stress conditions have been identified through the bioinformatic prediction or RLM-5′ RACE-based validation in tomato, suggesting that a miRNA-mediated regulatory network is involved in high temperature [115]. In Arabidopsis, miR160 repressed ARF10, ARF16 and ARF17 to release the expression level of heat shock protein genes to allow the plants to survive heat stress [116], while miR160-ARF18 mediated salt tolerance in peanut [117]. In pear, a novel miRNA, Novel_188, is validated to target Pbr027651.1 to mediate fruit senescence under high- or low-temperature conditions [118]. Ptr-miR396b was determined to target 1-aminocyclopropane-1carboxylic acid oxidase (ACO) in response to cold stress in orange (Poncirus trifoliata) [119]. In mango, bioinformatic analysis reveals that MmiR78769 and MmiR101928 target phospholipase A and phospholipase D, respectively, both of which are associated with plant temperature stress-responsive process [120]. Moreover, degradome-wide analyses have revealed that miR393-TIR1/AFB displays a cold stress-specific response and miR156-SPL-mediated heat stress response in banana [121].
In particular, the lncRNAs’ temperature stress responses were found to be very specific. High temperature-induced LNC_000862 is likely to delay pear fruit senescence by competing with miR390a to derepress the expression of Pbr031098.1 [122]. LncRNAs involved in the response of chilling injury in tomato fruit have been systematically identified, providing a new perspective on lncRNA roles in chilling tolerance in fruits [123]. In mango, the cold-responsive lncRNA CRlnc26299 can interact with RC12B, which is the low-temperature and salt-responsive protein [120]. A novel lncRNA, COLD INDUCED lncRNA 1 (CIL1), is a positive regulator in plant response to cold stress by regulating the expression of endogenous reactive oxygen species (ROS) in Arabidopsis [124].
Drought stress adversely affects fruit crops’ productivity and quality. Drought stress response modulation via the miRNA pathway has also been found in several plant species. In tomato, miR159, miR169, miR160, miR167 and miR393 are associated with dehydration stress tolerance, by controlling hormonal signal transduction, stomatal closure and auxin-responsive genes [125,126]. The overexpression of miR396 showed lower densities of stomata and induced drought tolerance in Arabidopsis by suppressing the expression of GRF [127], which was consistent with the finding that the miR396-GRF module is involved in stress tolerance in tomato and pitaya [112,125]. Moreover, ABA-induced miR159 inhibits the transcripts of MYB101 and MYB33 during seedling stress responses in Arabidopsis [128]. A novel lncRNA, named DROUGHT INDUCED lncRNA (DRIR), has a positive role in the response of Arabidopsis to drought and salt stress [129].
Pathogen defense is associated with fruit quality and postharvest quality. Plants are constantly exposed to a range of microbial pathogens with different lifestyles and modes of attack, including fungal, bacterial and viral pathogens, whereas RNA-based mechanisms largely regulate plant–virus interactions. Many key miRNA regulators of the stress response in fruits during pathogen infection were identified, such as miRNAs engineer Botrytis cinerea (B. cinerea) in kiwifruit [130] and specific miRNAs’ response to stress in Amur grape (vitis amurensis Rupr.) [131]. In particular, Md-miRLn11 targeted an apple nucleotide-binding site (NBS)–leucine-rich repeat (LRR) class protein coding gene (Md-NBS) to trigger host immune responses during pathogen infection [132]. SlymiR482e-3p knocking-out lines showed enhanced resistance to tomato wilt disease and regulated ethylene signaling by suppressing the expression of ethylene response factors (SlERFs) [133]. The can-miRn37a further confirmed anthracnose resistance in chili (Capsicum annuum L.) by repressing ERFs and preventing fungal colonization [134]. In tomato, sly-MIR156d/e were found induced under biotic and abiotic stress [29,135]. In addition, miR159/319 and miR172 accumulation positively correlated with immune responses during Tomato leaf curl virus (ToLCV) infection, indicating that miR159/319 and miR172 might be associated with the response to viral infection in tomato [136]. In pear, pbr-miR156, pbr-miR164, pbr-miR399 and pbr-miR482 are induced during Apple stem pitting virus (ASPV) infection and then trigger its target genes to participate in viral defense pathways [137]. Overexpressed miR396 not only plays roles in drought response in A. thaliana [127] and cold tolerance in orange [119], but also has resulted in plant tolerance under the attack of necrotrophic fungal pathogens [138]. Previous reports have shown that lncRNA not only plays essential roles in diverse biological processes, but also in various stress responses. LncRNA4504 positively regulated methyl jasmonate (MeJA)-induced tomato fruit resistance to B. cinerea by promoting the accumulation of total phenols and flavonoids and upregulating the expression of JA signal pathway genes [139].
The most effective postharvest technology to maintain fruit quality is to delay the fruit senescence process, such as cold storage after the fruit is harvested. Thus, in incorporating the dynamic environments, important alterations in non-coding RNA transcriptomes are observed in many plant species, which has led to the general view that plants utilize ncRNAs as part of their arsenal to cope with the wide array of microbial pathogens they encounter (Table 1). Further investigation might help to find clues to a better understanding of the consequences of ncRNA attenuation under biotic and abiotic stress and its putative success under field conditions.

Table 1.
miRNAs and lncRNAs with functional verification in fruit crops.
Table 1.
miRNAs and lncRNAs with functional verification in fruit crops.
| Fruit Biology | Classification | Species | Non-Coding RNA | Targets/Downstream | Functionally in Fruit Quality | Research Methods | References |
|---|---|---|---|---|---|---|---|
| Fruit development | Fruit size and number | arabidopsis | miR172C | APETALA2-like | silique fruit expansion | stable (MIR172C::GUS, MIR172CAuxRE::GUS) | [21] |
| miR159a/b | MYB33/MYB65 | altered growth habit, curled leaves, small siliques, and small seeds | T-DNA mutants (mir159ab double mutant) | [40] | |||
| apple | miR172p | AP2 | reduced fruit size, altered floral organ development | stable (MIR172p OE in tomato) | [22,23] | ||
| tomato | miR156 | SPL | fruit growth, ovary and fruit development | stable (AtMIR156b OE) | [15] | ||
| miR159 | SlGAMYB2 (GA biosynthesis gene) | larger fruits | STTM-miR159 | [41] | |||
| miR172d | AP2 | floral organ identity and number | CRISPR/Cas9 (slmir172c-dCR) | [24] | |||
| miR396a/b | GRF | a larger plant, with bigger flowers, leaves, and fruits | STTM-miR396 | [35,36,37] | |||
| miR1917 | CTR4 (altered ethylene response) | fruit size, bigger fruit | STTM-miR1917 | [39] | |||
| miR171a | SlGRAS24 and SlGRAS40 (altered gibberellin and auxin) | cell number and size, smaller tomato fruit | GRAS24 OE | [34] | |||
| miR164a | NAM2/3 | decreased fruit size | CRISPR/Cas9 (slmir164a, slmir164b, slmir164d, slmir164CR) | [38] | |||
| Fruit development | fruit set | tomato | miR159 | SlGAMYB2 (GA biosynthesis gene) | fruit morphology, precocious fruit initiationflattened, fruit with more locules inside | SlMIR159 OE | [11] |
| miR160 | ARF10, ARF16 and ARF17 | sugar accumulation, leaf and flower development, somatic embryo development, pear-shaped fruit | STTM-miR160 | [12,46,47] | |||
| miR166 | SlREV | fruit formation | Overexpression of a microRNA166-resistant version of SlREV (35S::REVRis) | [50] | |||
| miR168 | SlAGO1s | fruit initiation and growth | miR168 loss-of-function (four-point-mutated miR168-resistant 4m-SlAGO1A and 4m-SlAGO1B) | [56] | |||
| pear | PbrmiR397a | LACs | stone cell formation, reduced lignin content and stone cell number | transient (PbrmiR397a OE, pear), stable (PbrmiR397a OE, tobacco) | [51] | ||
| longan | miR160 | ARF10, -16, and -17 | somatic embryo development | target mimics down-regulate miR160 | [52] | ||
| seed development/parthenocarpy | tomato | miR159 | GAMyb-like1 and GAMyb-like2 | parthenocarpy | SlMIR159 OE | [11] | |
| miR166 | SlHB15A | parthenocarpic fruit set | used TILLING to screen for SlHB15A miR166-resistant alleles | [57] | |||
| miR167 | SlARF8 | parthenocarpy | downregulation of miR167 | [11] | |||
| miR168 | SlAGO1s | parthenocarpy | miR168-resistant 4m-SlAGO1A | [56] | |||
| miR172 | AP2 | small parthenocarpic fruit-like organ | CRISPR/Cas9 (slmir172c-dCR) | [24] | |||
| Fruit ripening | fruit color | litchi | miR156a * | LcSPL1/2 | anthocyanin biosynthesis | High-Throughput Sequencing and Degradome Analysis | [67] |
| NEW41 * | CHI | anthocyanin accumulation | |||||
| pear | miR156 * | SPL | Red Peel Coloration, anthocyanin biosynthesis | Degradome Library | [73] | ||
| blueberry | miR156a | VcSPL12 | anthocyanin accumulation | VcMIR156a OE in tomato | [66] | ||
| miR396 * | FtsZs | coloration | Small RNA and Degradome Sequencing | [77] | |||
| miR_n10 * | BAG1 | coloration | |||||
| apple | miR172 | AP2-MYB10 | flavonoidse, reduction in red coloration | miR172 OE | [80] | ||
| MLNC3.2 and MLNC4.6 (lncRNA) | miR156a-SPL2-like and SPL33 | anthocyanin biosynthesis | transient (35S::MLNC3.2, 35S::MLNC4.6, OE-miR156a) | [75] | |||
| miR7125 (light-induced) | MYB16/MYB1-CCRs | promoted anthocyanin synthesis, reduced lignin biosynthesis | transient (miR7125 OE) | [79] | |||
| MdLNC499 (lncRNA) | MdERF109 | fruit coloration | transient (TRV-MdLNC499, TRV-MdERF109, apple fruit), stable (MdLNC499 OE, MdLNC499 RNAi, MdERF109 OE, MdERF109 RNAi, apple calli) | [78] | |||
| mdm-miR828 | TAS4-MdMYB1 | inhibited anthocyanin synthesis | transient (mdm-miR828 OE, apple, stable (mdm-miR828 OE, Arabidopsis) | [82] | |||
| miR858 * | MYB | anthocyanin biosynthesis | small RNA-seq | [26] | |||
| sea buckthorn | LNC1 (lncRNA)-miR156a | SPL9 | anthocyanin accumulation | transient (TRV-LNC1) | [76] | ||
| Fruit ripening | fruit color | grape | miR858 | VvMYB114 | anthocyanin and flavonol accumulation | Degradom, transient/stable (VvMYB114 OE, tobacco) | [69] |
| miR156 | SPL9 | promoted fruit coloration | miR156b/c/d OE in tomato | [74] | |||
| miR3627 * | calcium-transporting ATPase10 | anthocyanin accumulation | sequencing small RNAs, bioinformatics analysis | [101] | |||
| miR828 | VvMYB113/VvMYB114 | anthocyanin and flavonol accumulation | vvi-miR828 OE, Arabidopsis | [69] | |||
| arabidopsis | miR828 | MYB75, MYB90, and MYB113 | anthocyanin accumulation | AtmiR828 OE | [68] | ||
| miR858a | MYB2 | anthocyanin accumulation, anthocyanin biosynthesis | STTM-miR858 | [72] | |||
| miR156 | SPL9 and SPL15 | anthocyanin biosynthesis | MIR156b OE | [65] | |||
| tomato | miR858 | SlMYB7 and SlMYB48 | anthocyanin accumulation | STTM-miR858 | [70] | ||
| kiwifruit | miR858 | AaMYBC1 | anthocyanin biosynthesis | transient (miR858 OE) | [71] | ||
| fruit ripening, fruit softening and fruit quality | persimmon | miR395 * | bHLH | tannin biosynthesis | high-throughput sequencing | [83] | |
| miR156 * | SPL | tannin biosynthesis | |||||
| miR396 * | Flavonoid 3-O-glucosyltransferase (UFGT) | tannin biosynthesis | |||||
| miR858 * | MYB19/20 | reduced the content of proanthocyanidin (PA) | |||||
| miR2991 * | ADH | tannin biosynthesis | |||||
| Fruit ripening | fruit ripening, fruit softening and fruit quality | strawberry | FRILAIR (lncRNA)-miR397 | LAC11a | delayed fruit ripening | transient (miR397 OE, Cas13b-miR397, ocotoploid strawberry) | [100] |
| fan-miR73 | ABI5 | fruit ripening | 5′ -RACE analysis | [85] | |||
| miR399 | PHO2 | flavor, sugar content | miR399a OE (woodland strawberry) | [84] | |||
| tomato | miR157 | SPL-CNR | delayed fruit ripening | miR157 OE | [86,87] | ||
| miR156 | SPL | accelerates tomato fruit softening | VIGS-miR156a | [87] | |||
| miR172 | AP2a | accelerates fruit ripening with enhanced ethylene biosynthesis | miR172 OE | [105] | |||
| miR166 | SlREV | fruit ripening | 35S::REVRis (EIN3, ERFs, AP2, and CTR3 downregulated) | [50] | |||
| miR828 * | EIN2 | ethylene-dependent ripening | high throughput sequencing | [108] | |||
| miR1917 | CTR4 | enhances ethylene response and accelerates fruit ripening | miR1917 OE | [107] | |||
| lncRNA2155 (lncRNA) | RIN, CNR, NOR, ACS4, PSY1 | delayed fruit ripening | CRISPR/Cas9 (lncRNA2155 KO) | [98] | |||
| lncRNA1459 (lncRNA) | PSY1, PDS, ZDS | ripening, ethylene biosynthesis | CRISPR/Cas9 (lncRNA1459 KO) | [10,17] | |||
| lncRNA1840 (lncRNA) | ripening-related genes | ripening, ethylene biosynthesis | TRV-lncRNA1840 | [10] | |||
| kiwifruit | miR164 | NAC6/7 | fruit ripening | miR164 OE (kiwifruit callus) | [88] | ||
| apple | miR7125 | MYB16/MYB1-CCRs | reduced lignin biosynthesis | transient (miR7125 OE, apple fruit) | [79] | ||
| melon | cme-miR393 | CmAFB2 | delayed fruit ripening | cme-miR393-OE | [89] | ||
| Fruit ripening | fruit ripening, fruit softening and fruit quality | grapes | miR479 * | BGA | fruit softing | deep sequencing, bioinformatics analysis | [101] |
| miR399 * | ACO3 | ||||||
| miR397 * | LOX | ||||||
| miR3627 * | Grip22/PAL | ||||||
| miR2950 * | CHS | ||||||
| miR22 * | PE | ||||||
| biotic and abiotic stress in fruit | cold response | arabidopsis | CIL1 (lncRNA) | ROS | enhances cold stress tolerance | T-DNA insertion mutants | [124] |
| orange | miR396b | GRF | cold tolerance | ptr-miR396b OE (transgenic lemon (Citrus limon)) | [119] | ||
| banana | miR393 * | TIR1/AFB | cold stress-specific response | bioinformatics analysis | [121] | ||
| mango | CRlnc26299 * (lncRNA) | RC12B | chilling tolerance | Computational Identification | [120] | ||
| salt tolerance | arabidopsis | miR396 | GRF | salt tolerance | target mimicry (eTM) transgene specific to miR396 | [111] | |
| miR393a/b | TIR1 | salt stress resistance and ABA signaling pathways | mir393ab double mutant | [109,110] | |||
| pitaya | miR396 * | GRF | stress response | bioinformatics analysis | [112] | ||
| heat tolerance | tomato | miR396 * | GRF | drought and heat stress | bioinformatics analysis | [125] | |
| arabidopsis | miR160 | ARF10, ARF16, and ARF17 | heat stress tolerance | eTM-miR160 | [116] | ||
| banana | miR156 * | SPL | heat stress response | bioinformatics analysis | [121] | ||
| mango | MmiR78769 and MmiR101928 (lncRNA) | phospholipase A and phospholipase D | biotic and abiotic stresses | Computational Identification | [120] | ||
| biotic and abiotic stress in fruit | heat tolerance | pear | Novel_188 | Pbr027651.1 | mediate fruit senescence | transient (Novel_188 OE) | [118] |
| LNC_000862 * (lncRNA) | miR390a-Pbr031098.1 | heat tolerance | bioinformatics analysis | [122] | |||
| drought response | arabidopsis | miR396a/b | GRF | drought tolerance | 35S::MIR396a and 35S::MIR396b | [127] | |
| miR159 | MYB101 and MYB33 | drought tolerance | miR159 OE | [128] | |||
| DRIR (lncRNA) | genes involved in ABA signaling | Enhances Drought and Salt Stress Tolerance | DRIR OE | [129] | |||
| tomato | miR169 | NFYA | drought and heat stress | STTM-miR169 | [126] | ||
| miR159 * | MYB | bioinformatics analysis | [125] | ||||
| miR160 * | ARF | ||||||
| miR167 * | ARF | ||||||
| miR393 * | auxin receptor homologous genes | ||||||
| pathogen defense | arabidopsis | miR396 | GRF | pathogen defense | miR396 target mimics lines | [138] | |
| apple | Md-miRLn11 | Md-NBS | pathogen defense | bioinformatics analysis | [132] | ||
| tomato | SlymiR482e-3p | NBS-LRR | enhanced resistance to tomato wilt disease | slymiR482e-3p KO lines | [133] | ||
| miR156 * | SPL | response to ToLCV infections | bioinformatics analysis | [29,135] | |||
| miR159/319 | AP2-like | viral response (tomato leaf curl new delhi virus (tolcndv)) | MicroRNA profiling | [136] | |||
| miR172 | TCP, bHLH | [136] | |||||
| LncRNA4504 (lncRNA) | JA signal pathway genes | pathogen defense (Botrytis cinerea) | TRV-lncRNA4504 | [139] | |||
| pear | pbr-miR156 * | pbRPS6 | viral defense | bioinformatics analysis | [137] | ||
| pbr-miR164 * | pbNAC | ||||||
| pbr-miR399 * | pbTLR | ||||||
| pbr-miR482 * | pbRX-CC |
The asterisk (*) represents bioinformatics analysis data.
5. Conclusions and Perspective
It can be inferred that miRNA and lncRNA are important regulators in fruit crops. In both dry and fleshy fruit systems, specific miRNAs and lncRNAs are identified, and their roles are essential in the organization of a functional final organ structure, fruit ripening and stress tolerance (Table 1 and Figure 1). Functional studies have revealed that some miRNA regulatory modules are universally important to fruit crops; for instance, miR828/miR858-MYB regulates anthocyanin accumulation, miR396-GRF regulates plant growth and stress response, miR156-SPL regulates fruit metabolism, and miR172-AP2 regulates fruit size and anthocyanin biosynthesis (Figure 2). Thus, miRNA-mediated genetic engineering methods could represent an effective approach for the development of superior characters. This is probably an economic way to develop important agricultural traits or innovations by fine-tuning miRNAs instead of suppressing a large number of genes, and it will also allow breeders to obtain a commercially valuable crop species in a short time.

Figure 2.
Models of miRNA regulatory modules in fruit crops. (A) The miR828/miR858-MYB module regulates anthocyanin accumulation in Arabidopsis, tomato, apple, grape, kiwifruit and persimmon, respectively. (B) The miR396-GRF module regulates plant growth and stress response in Arabidopsis, tomato, orange and pitaya, respectively. (C) The miR156-SPL module regulates fruit metabolism in Arabidopsis, apple, pear and persimmon, respectively. (D) The miR172-AP2 module regulates fruit size and anthocyanin biosynthesis in Arabidopsis, tomato and apple.
The involvement of miRNAs in several agriculturally important traits that have been improved to date using CRISPR/Cas9 include crop productivity, quality, and biotic and abiotic stress resistance [140]. For example, generating mutations in MIR396e and MIR396f increases the grain size and modulates the shoot architecture in rice using a multiplex CRISPR/Cas9-based genome engineering tool [141]. For fruit crops, few studies are available on the CRISPR/Cas9 editing of miRNA family and lncRNA mutants, and much more work is needed to decipher miRNA-mediated regulatory networks. In addition, many genome editing tools are continually developed, thus finding valuable editing switch sites that are important for creating new agronomic traits instead of one or several gene edits. This is a broadly adopted regulatory strategy during plant evolution and it is practical for crop improvement. A growing body of evidence shows the prime editing system applied in plants [142,143,144], suggesting that the prime editing tool would be a promising technology to introduce the desired modification and breed elite crop varieties in fruit crops. We believe that with further understanding of miRNA- and lncRNA-based cellular regulatory networks, additional technologies will emerge for the improvement of fruit agricultural traits.
In particular, a better characterization of the miRNAs and lncRNAs, which can coordinate the main steps of fruit development and ripening in different plant species, may result in the development of novel strategies for fruit crop management. One of the most important challenges in the future will be to functionally analyze more miRNA modules and lncRNA networks in fruit crops, as well as the further achievement of efficient transgene-free genome editing via the CRISPR system in fruit crops. We outline future perspectives in developing miRNA/lncRNA-based breeding strategies for fruit crop improvement and applying genome editing tools for modulating agriculturally important traits.
Author Contributions
Y.T. wrote the manuscript. Y.T., R.H. and D.W. conducted the literature review. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Science Foundation of China (32160070) and the Natural Science Foundation of Shandong Province (ZR2022QC054).
Data Availability Statement
The datasets generated during the current study are available at https://www.mdpi.com/ethics (accessed on 24 December 2022).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Coombe, B.G. The Development of Fleshy Fruits. Annu. Rev. Plant Physiol. 1976, 27, 207–228. [Google Scholar] [CrossRef]
- Seymour, G.B.; Østergaard, L.; Chapman, N.H.; Knapp, S.; Martin, C. Fruit Development and Ripening. Annu. Rev. Plant Biol. 2013, 64, 219–241. [Google Scholar] [CrossRef]
- Seymour, G.B.; Chapman, N.H.; Chew, B.L.; Rose, J.K.C. Regulation of Ripening and Opportunities for Control in Tomato and Other Fruits. Plant Biotechnol. J. 2013, 11, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, A.; Alquézar, B.; Peña, L. Fruit Aromas in Mature Fleshy Fruits as Signals of Readiness for Predation and Seed Dispersal. New Phytol. 2013, 197, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Kou, X.; Feng, Y.; Yuan, S.; Zhao, X.; Wu, C.; Wang, C.; Xue, Z. Different Regulatory Mechanisms of Plant Hormones in the Ripening of Climacteric and Non-Climacteric Fruits: A Review. Plant Mol. Biol. 2021, 107, 477–497. [Google Scholar] [CrossRef]
- Lin, T.; Zhu, G.; Zhang, J.; Xu, X.; Yu, Q.; Zheng, Z.; Zhang, Z.; Lun, Y.; Li, S.; Wang, X.; et al. Genomic Analyses Provide Insights into the History of Tomato Breeding. Nat. Genet. 2014, 46, 1220–1226. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Q.; Shi, J.; Scheben, A.; Zhan, J.; Wang, X.; Liu, G.; Yan, G.; King, G.J.; Edwards, D.; Wang, H. Genetic and Signalling Pathways of Dry Fruit Size: Targets for Genome Editing-based Crop Improvement. Plant Biotechnol. J. 2020, 18, 1124–1140. [Google Scholar] [CrossRef]
- Wang, W.-Q.; Allan, A.C.; Yin, X.-R. Small RNAs With a Big Impact on Horticultural Traits. Crit. Rev. Plant Sci. 2020, 39, 30–43. [Google Scholar] [CrossRef]
- Chen, C.; Zeng, Z.; Liu, Z.; Xia, R. Small RNAs, Emerging Regulators Critical for the Development of Horticultural Traits. Hortic. Res. 2018, 5, 63. [Google Scholar] [CrossRef]
- Zhu, B.; Yang, Y.; Li, R.; Fu, D.; Wen, L.; Luo, Y.; Zhu, H. RNA Sequencing and Functional Analysis Implicate the Regulatory Role of Long Non-Coding RNAs in Tomato Fruit Ripening. J. Exp. Bot. 2015, 66, 4483–4495. [Google Scholar] [CrossRef]
- da Silva, E.M.; Silva, G.F.F.E.; Bidoia, D.B.; da Silva Azevedo, M.; de Jesus, F.A.; Pino, L.E.; Peres, L.E.P.; Carrera, E.; López-Díaz, I.; Nogueira, F.T.S. MicroRNA159-targeted SlGAMYB transcription factors are required for fruit set in tomato. Plant J. 2017, 92, 95–109. [Google Scholar] [CrossRef] [PubMed]
- Damodharan, S.; Corem, S.; Gupta, S.K.; Arazi, T. Tuning of SlARF10A Dosage by Sly-MiR160a Is Critical for Auxin-Mediated Compound Leaf and Flower Development. Plant J. 2018, 96, 855–868. [Google Scholar] [CrossRef]
- Hendelman, A.; Stav, R.; Zemach, H.; Arazi, T. The Tomato NAC Transcription Factor SlNAM2 Is Involved in Flower-Boundary Morphogenesis. J. Exp. Bot. 2013, 64, 5497–5507. [Google Scholar] [CrossRef]
- Berger, Y.; Harpaz-Saad, S.; Brand, A.; Melnik, H.; Sirding, N.; Alvarez, J.P.; Zinder, M.; Samach, A.; Eshed, Y.; Ori, N. The NAC-Domain Transcription Factor GOBLET Specifies Leaflet Boundaries in Compound Tomato Leaves. Development 2009, 136, 823–832. [Google Scholar] [CrossRef]
- Silva, G.F.F.E.; Silva, E.M.; da Silva Azevedo, M.; Guivin, M.A.C.; Ramiro, D.A.; Figueiredo, C.R.; Carrer, H.; Peres, L.E.P.; Nogueira, F.T.S. MicroRNA156-targeted SPL/SBP box transcription factors regulate tomato ovary and fruit development. Plant J. 2014, 78, 604–618. [Google Scholar] [CrossRef] [PubMed]
- Asefpour Vakilian, K. Machine Learning Improves Our Knowledge about miRNA Functions towards Plant Abiotic Stresses. Sci. Rep. 2020, 10, 3041. [Google Scholar] [CrossRef]
- Li, R.; Fu, D.; Zhu, B.; Luo, Y.; Zhu, H. CRISPR/Cas9-Mediated Mutagenesis of LncRNA1459 Alters Tomato Fruit Ripening. Plant J. 2018, 94, 513–524. [Google Scholar] [CrossRef] [PubMed]
- Dinneny, J.R.; Weigel, D.; Yanofsky, M.F. A Genetic Framework for Fruit Patterning in Arabidopsisthaliana. Development 2005, 132, 4687–4696. [Google Scholar] [CrossRef]
- Sang, Q.; Vayssières, A.; Ó’Maoiléidigh, D.S.; Yang, X.; Vincent, C.; Bertran Garcia de Olalla, E.; Cerise, M.; Franzen, R.; Coupland, G. MicroRNA172 Controls Inflorescence Meristem Size through Regulation of APETALA2 in Arabidopsis. New Phytol. 2022, 235, 356–371. [Google Scholar] [CrossRef]
- Yant, L.; Mathieu, J.; Dinh, T.T.; Ott, F.; Lanz, C.; Wollmann, H.; Chen, X.; Schmid, M. Orchestration of the Floral Transition and Floral Development in Arabidopsis by the Bifunctional Transcription Factor APETALA2. Plant Cell 2010, 22, 2156–2170. [Google Scholar] [CrossRef]
- José Ripoll, J.; Bailey, L.J.; Mai, Q.-A.; Wu, S.L.; Hon, C.T.; Chapman, E.J.; Ditta, G.S.; Estelle, M.; Yanofsky, M.F. MicroRNA Regulation of Fruit Growth. Nat. Plants 2015, 1, 15036. [Google Scholar] [CrossRef]
- Yao, J.-L.; Xu, J.; Cornille, A.; Tomes, S.; Karunairetnam, S.; Luo, Z.; Bassett, H.; Whitworth, C.; Rees-George, J.; Ranatunga, C.; et al. A MicroRNA Allele That Emerged Prior to Apple Domestication May Underlie Fruit Size Evolution. Plant J. 2015, 84, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.-L.; Tomes, S.; Xu, J.; Gleave, A.P. How MicroRNA172 Affects Fruit Growth in Different Species Is Dependent on Fruit Type. Plant Signal. Behav. 2016, 11, e1156833. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Gupta, S.K.; Arazi, T.; Spitzer-Rimon, B. MIR172d Is Required for Floral Organ Identity and Number in Tomato. Int. J. Mol. Sci. 2021, 22, 4659. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, T.C.D.S.; Alves, T.C.; Caneschi, C.M.; Santana, D.D.R.G.; Fernandes-Brum, C.N.; Reis, G.L.D.; Daude, M.M.; Ribeiro, T.H.C.; Gómez, M.M.D.; Lima, A.A.; et al. New Insights into Tomato MicroRNAs. Sci. Rep. 2018, 8, 16069. [Google Scholar] [CrossRef]
- Xia, R.; Zhu, H.; An, Y.; Beers, E.P.; Liu, Z. Apple miRNAs and TasiRNAs with Novel Regulatory Networks. Genome Biol. 2012, 13, R47. [Google Scholar] [CrossRef]
- Ó’Maoiléidigh, D.S.; van Driel, A.D.; Singh, A.; Sang, Q.; Le Bec, N.; Vincent, C.; de Olalla, E.B.G.; Vayssières, A.; Romera Branchat, M.; Severing, E.; et al. Systematic Analyses of the MIR172 Family Members of Arabidopsis Define Their Distinct Roles in Regulation of APETALA2 during Floral Transition. PLoS Biol. 2021, 19, e3001043. [Google Scholar] [CrossRef]
- Xie, J.; Yang, X.; Song, Y.; Du, Q.; Li, Y.; Chen, J.; Zhang, D. Adaptive Evolution and Functional Innovation of Populus-Specific Recently Evolved MicroRNAs. New Phytol. 2017, 213, 206–219. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Goswami, K.; Tiwari, M.; Mukherjee, S.K.; Sanan-Mishra, N. Identification and Comparative Analysis of MicroRNAs from Tomato Varieties Showing Contrasting Response to ToLCV Infections. Physiol. Mol. Biol. Plants 2018, 24, 185–202. [Google Scholar] [CrossRef]
- Guo, C.; Xu, Y.; Shi, M.; Lai, Y.; Wu, X.; Wang, H.; Zhu, Z.; Poethig, R.S.; Wu, G. Repression of MiR156 by MiR159 Regulates the Timing of the Juvenile-to-Adult Transition in Arabidopsis. Plant Cell 2017, 29, 1293–1304. [Google Scholar] [CrossRef]
- Zhang, T.; Wang, J.; Zhou, C. The Role of MiR156 in Developmental Transitions in Nicotiana tabacum. Sci. China Life Sci. 2015, 58, 253–260. [Google Scholar] [CrossRef]
- Feng, G.; Xu, L.; Wang, J.; Nie, G.; Bushman, B.S.; Xie, W.; Yan, H.; Yang, Z.; Guan, H.; Huang, L.; et al. Integration of Small RNAs and Transcriptome Sequencing Uncovers a Complex Regulatory Network during Vernalization and Heading Stages of Orchardgrass (Dactylis Glomerata L.). BMC Genom. 2018, 19, 727. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Sun, S.; Jin, J.; Fu, D.; Yang, X.; Weng, X.; Xu, C.; Li, X.; Xiao, J.; Zhang, Q. Coordinated Regulation of Vegetative and Reproductive Branching in Rice. Proc. Natl. Acad. Sci. USA 2015, 112, 15504–15509. [Google Scholar] [CrossRef]
- Huang, W.; Xian, Z.; Kang, X.; Tang, N.; Li, Z. Genome-Wide Identification, Phylogeny and Expression Analysis of GRAS Gene Family in Tomato. BMC Plant Biol. 2015, 15, 209. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Peng, S.; Xian, Z.; Lin, D.; Hu, G.; Yang, L.; Ren, M.; Li, Z. Overexpression of a Tomato MiR171 Target Gene SlGRAS24 Impacts Multiple Agronomical Traits via Regulating Gibberellin and Auxin Homeostasis. Plant Biotechnol. J. 2017, 15, 472–488. [Google Scholar] [CrossRef]
- Li, M.; Wang, X.; Li, C.; Li, H.; Zhang, J.; Ye, Z. Silencing GRAS2 Reduces Fruit Weight in Tomato. J. Integr. Plant Biol. 2018, 60, 498–513. [Google Scholar] [CrossRef] [PubMed]
- Peng, T.; Qiao, M.; Liu, H.; Teotia, S.; Zhang, Z.; Zhao, Y.; Wang, B.; Zhao, D.; Shi, L.; Zhang, C.; et al. A Resource for Inactivation of MicroRNAs Using Short Tandem Target Mimic Technology in Model and Crop Plants. Mol. Plant 2018, 11, 1400–1417. [Google Scholar] [CrossRef]
- Gupta, S.K.; Vishwakarma, A.; Kenea, H.D.; Galsurker, O.; Cohen, H.; Aharoni, A.; Arazi, T. CRISPR/Cas9 Mutants of Tomato MICRORNA164 Genes Uncover Their Functional Specialization in Development. Plant Physiol. 2021, 187, 1636–1652. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Wang, Y.; Liu, H.; Zhang, W.; Chai, M.; Tang, G.; Zhang, Z. MicroRNA1917-CTR1-LIKE PROTEIN KINASE 4 Impacts Fruit Development via Tuning Ethylene Synthesis and Response. Plant Sci. 2020, 291, 110334. [Google Scholar] [CrossRef]
- Allen, R.S.; Li, J.; Stahle, M.I.; Dubroué, A.; Gubler, F.; Millar, A.A. Genetic Analysis Reveals Functional Redundancy and the Major Target Genes of the Arabidopsis MiR159 Family. Proc. Natl. Acad. Sci. USA 2007, 104, 16371–16376. [Google Scholar] [CrossRef]
- Zhao, P.; Wang, F.; Deng, Y.; Zhong, F.; Tian, P.; Lin, D.; Deng, J.; Zhang, Y.; Huang, T. Sly-miR159 Regulates Fruit Morphology by Modulating GA Biosynthesis in Tomato. Plant Biotechnol. J. 2022, 20, 833–845. [Google Scholar] [CrossRef] [PubMed]
- Vivian-Smith, A.; Koltunow, A.M. Genetic Analysis of Growth-Regulator-Induced Parthenocarpy in Arabidopsis. Plant Physiol. 1999, 121, 437–452. [Google Scholar] [CrossRef] [PubMed]
- Fenn, M.A.; Giovannoni, J.J. Phytohormones in Fruit Development and Maturation. Plant J. 2021, 105, 446–458. [Google Scholar] [CrossRef]
- de Jong, M.; Mariani, C.; Vriezen, W.H. The Role of Auxin and Gibberellin in Tomato Fruit Set. J. Exp. Bot. 2009, 60, 1523–1532. [Google Scholar] [CrossRef]
- Pattison, R.J.; Csukasi, F.; Catalá, C. Mechanisms Regulating Auxin Action during Fruit Development. Physiol. Plant. 2014, 151, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Mei, L.; Wu, M.; Wei, W.; Shan, W.; Gong, Z.; Zhang, Q.; Yang, F.; Yan, F.; Zhang, Q.; et al. SlARF10, an Auxin Response Factor, Is Involved in Chlorophyll and Sugar Accumulation during Tomato Fruit Development. J. Exp. Bot. 2018, 69, 5507–5518. [Google Scholar] [CrossRef] [PubMed]
- Damodharan, S.; Zhao, D.; Arazi, T. A Common miRNA160-Based Mechanism Regulates Ovary Patterning, Floral Organ Abscission and Lamina Outgrowth in Tomato. Plant J. 2016, 86, 458–471. [Google Scholar] [CrossRef]
- Hong, Y.; Jackson, S. Floral Induction and Flower Formation—The Role and Potential Applications of miRNAs. Plant Biotechnol. J. 2015, 13, 282–292. [Google Scholar] [CrossRef]
- Itaya, A.; Bundschuh, R.; Archual, A.; Joung, J.; Fei, Z.; Dai, X.; Zhao, P.; Tang, Y.; Nelson, R.; Ding, B. Small RNAs in Tomato Fruit and Leaf Development. Biochim. Biophys. Acta BBA-Gene Regul. Mech. 2008, 1779, 99–107. [Google Scholar] [CrossRef]
- Hu, G.; Fan, J.; Xian, Z.; Huang, W.; Lin, D.; Li, Z. Overexpression of SlREV Alters the Development of the Flower Pedicel Abscission Zone and Fruit Formation in Tomato. Plant Sci. 2014, 229, 86–95. [Google Scholar] [CrossRef]
- Xue, C.; Yao, J.-L.; Qin, M.-F.; Zhang, M.-Y.; Allan, A.C.; Wang, D.-F.; Wu, J. PbrmiR397a Regulates Lignification during Stone Cell Development in Pear Fruit. Plant Biotechnol. J. 2019, 17, 103–117. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Lai, Z.; Tian, Q.; Lin, L.; Lai, R.; Yang, M.; Zhang, D.; Chen, Y.; Zhang, Z. Endogenous Target Mimics Down-Regulate MiR160 Mediation of ARF10, -16, and -17 Cleavage during Somatic Embryogenesis in Dimocarpus Longan Lour. Front. Plant Sci. 2015, 6, 956. [Google Scholar] [CrossRef]
- Kang, C.; Liu, Z. Global Identification and Analysis of Long Non-Coding RNAs in Diploid Strawberry Fragaria vesca during Flower and Fruit Development. BMC Genom. 2015, 16, 815. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Ren, F.; Wang, X.; Qiu, K.; Sheng, Y.; Xie, Q.; Shi, P.; Zhang, J.; Pan, H. Genome-Wide Identification and Characterization of Long Noncoding RNAs during Peach (Prunus Persica) Fruit Development and Ripening. Sci. Rep. 2022, 12, 11044. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Guo, M.; Huang, K.; Qi, Q.; Li, W.; Yan, J.; He, J.; Guan, Q.; Ma, F.; Xu, J. Genome-Wide Identification and Characterization of Long Noncoding RNAs Involved in Apple Fruit Development and Ripening. Sci. Hortic. 2022, 295, 110898. [Google Scholar] [CrossRef]
- Xian, Z.; Huang, W.; Yang, Y.; Tang, N.; Zhang, C.; Ren, M.; Li, Z. MiR168 Influences Phase Transition, Leaf Epinasty, and Fruit Development via SlAGO1s in Tomato. J. Exp. Bot. 2014, 65, 6655–6666. [Google Scholar] [CrossRef]
- Clepet, C.; Devani, R.S.; Boumlik, R.; Hao, Y.; Morin, H.; Marcel, F.; Verdenaud, M.; Mania, B.; Brisou, G.; Citerne, S.; et al. The MiR166–SlHB15A Regulatory Module Controls Ovule Development and Parthenocarpic Fruit Set under Adverse Temperatures in Tomato. Mol. Plant 2021, 14, 1185–1198. [Google Scholar] [CrossRef]
- Murakami, S. Study of Parthenocarpy in Three Kiwifruit Commercial Cultivars. Int. J. Fruit Sci. 2020, 20, S802–S811. [Google Scholar] [CrossRef]
- Cong, L.; Wu, T.; Liu, H.; Wang, H.; Zhang, H.; Zhao, G.; Wen, Y.; Shi, Q.; Xu, L.; Wang, Z. CPPU May Induce Gibberellin-Independent Parthenocarpy Associated with PbRR9 in ‘Dangshansu’ Pear. Hortic. Res. 2020, 7, 68. [Google Scholar] [CrossRef]
- Shinozaki, Y.; Ezura, H.; Ariizumi, T. The Role of Ethylene in the Regulation of Ovary Senescence and Fruit Set in Tomato (Solanum Lycopersicum). Plant Signal. Behav. 2018, 13, e1146844. [Google Scholar] [CrossRef]
- Serrani, J.C.; Carrera, E.; Ruiz-Rivero, O.; Gallego-Giraldo, L.; Peres, L.E.P.; García-Martínez, J.L. Inhibition of Auxin Transport from the Ovary or from the Apical Shoot Induces Parthenocarpic Fruit-Set in Tomato Mediated by Gibberellins. Plant Physiol. 2010, 153, 851–862. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, L.Y.; Kotb, H.R.M. Effect of Brassinosteroids and Gibberellic Acid on Parthenocarpic Fruit Formation and Fruit Quality of Sugar Apple Annona squamosa L. Middle East J. Agric. Res. 2018, 7, 1341–1351. [Google Scholar]
- Lu, L.; Liang, J.; Zhu, X.; Xiao, K.; Li, T.; Hu, J. Auxin- and Cytokinin-Induced Berries Set in Grapevine Partly Rely on Enhanced Gibberellin Biosynthesis. Tree Genet. Genomes 2016, 12, 41. [Google Scholar] [CrossRef]
- Mesejo, C.; Yuste, R.; Reig, C.; Martínez-Fuentes, A.; Iglesias, D.J.; Muñoz-Fambuena, N.; Bermejo, A.; Germanà, M.A.; Primo-Millo, E.; Agustí, M. Gibberellin Reactivates and Maintains Ovary-Wall Cell Division Causing Fruit Set in Parthenocarpic Citrus Species. Plant Sci. 2016, 247, 13–24. [Google Scholar] [CrossRef]
- Gou, J.-Y.; Felippes, F.F.; Liu, C.-J.; Weigel, D.; Wang, J.-W. Negative Regulation of Anthocyanin Biosynthesis in Arabidopsis by a MiR156-Targeted SPL Transcription Factor. Plant Cell 2011, 23, 1512–1522. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hou, Y.; Xie, X.; Li, H.; Li, X.; Zhu, Y.; Zhai, L.; Zhang, C.; Bian, S. A Blueberry MIR156a–SPL12 Module Coordinates the Accumulation of Chlorophylls and Anthocyanins during Fruit Ripening. J. Exp. Bot. 2020, 71, 5976–5989. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Lai, B.; Hu, B.; Qin, Y.; Hu, G.; Zhao, J. Identification of MicroRNAs and Their Target Genes Related to the Accumulation of Anthocyanins in Litchi Chinensis by High-Throughput Sequencing and Degradome Analysis. Front. Plant Sci. 2017, 7, 2059. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Cai, J.; Yang, Y.; Liu, Z. Overexpression of MicroRNA828 Reduces Anthocyanin Accumulation in Arabidopsis. Plant Cell Tissue Organ Cult. PCTOC 2013, 15, 159–167. [Google Scholar] [CrossRef]
- Tirumalai, V.; Swetha, C.; Nair, A.; Pandit, A.; Shivaprasad, P.V. miR828 and miR858 Regulate VvMYB114 to Promote Anthocyanin and Flavonol Accumulation in Grapes. J. Exp. Bot. 2019, 70, 4775–4792. [Google Scholar] [CrossRef]
- Jia, X.; Shen, J.; Liu, H.; Li, F.; Ding, N.; Gao, C.; Pattanaik, S.; Patra, B.; Li, R.; Yuan, L. Small Tandem Target Mimic-Mediated Blockage of MicroRNA858 Induces Anthocyanin Accumulation in Tomato. Planta 2015, 242, 283–293. [Google Scholar] [CrossRef]
- Li, Y.; Cui, W.; Qi, X.; Lin, M.; Qiao, C.; Zhong, Y.; Hu, C.; Fang, J. MicroRNA858 Negatively Regulates Anthocyanin Biosynthesis by Repressing AaMYBC1 Expression in Kiwifruit (Actinidia Arguta). Plant Sci. 2020, 296, 110476. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, Y.; Song, Z.; Zhang, H. Repression of MYBL2 by Both MicroRNA858a and HY5 Leads to the Activation of Anthocyanin Biosynthetic Pathway in Arabidopsis. Mol. Plant 2016, 9, 1395–1405. [Google Scholar] [CrossRef]
- Qian, M.; Ni, J.; Niu, Q.; Bai, S.; Bao, L.; Li, J.; Sun, Y.; Zhang, D.; Teng, Y. Response of MiR156-SPL Module during the Red Peel Coloration of Bagging-Treated Chinese Sand Pear (Pyrus Pyrifolia Nakai). Front. Physiol. 2017, 8, 550. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Wang, X.; Xuan, X.; Sheng, Z.; Jia, H.; Emal, N.; Liu, Z.; Zheng, T.; Wang, C.; Fang, J. Characterization and Action Mechanism Analysis of VvmiR156b/c/d-VvSPL9 Module Responding to Multiple-Hormone Signals in the Modulation of Grape Berry Color Formation. Foods 2021, 10, 896. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Ma, H.; Zhang, J.; Wu, T.; Song, T.; Tian, J.; Yao, Y. Systematic Identification of Long Noncoding RNAs Expressed during Light-Induced Anthocyanin Accumulation in Apple Fruit. Plant J. 2019, 100, 572–590. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Chen, D.; Zhang, T.; Duan, A.; Zhang, J.; He, C. Transcriptomic and Functional Analyses Unveil the Role of Long Non-Coding RNAs in Anthocyanin Biosynthesis during Sea Buckthorn Fruit Ripening. DNA Res. 2018, 25, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Zhai, L.; Li, X.; Xue, Y.; Wang, J.; Yang, P.; Cao, C.; Li, H.; Cui, Y.; Bian, S. Comparative Analysis of Fruit Ripening-Related miRNAs and Their Targets in Blueberry Using Small RNA and Degradome Sequencing. Int. J. Mol. Sci. 2017, 18, 2767. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Yang, T.; Li, Y.; Zhang, J.; Wu, T.; Song, T.; Yao, Y.; Tian, J. The Long Noncoding RNA MdLNC499 Bridges MdWRKY1 and MdERF109 Function to Regulate Early-Stage Light-Induced Anthocyanin Accumulation in Apple Fruit. Plant Cell 2021, 33, 3309–3330. [Google Scholar] [CrossRef]
- Hu, Y.; Cheng, H.; Zhang, Y.; Zhang, J.; Niu, S.; Wang, X.; Li, W.; Zhang, J.; Yao, Y. The MdMYB16/MdMYB1-MiR7125-MdCCR Module Regulates the Homeostasis between Anthocyanin and Lignin Biosynthesis during Light Induction in Apple. New Phytol. 2021, 231, 1105–1122. [Google Scholar] [CrossRef]
- Ding, T.; Tomes, S.; Gleave, A.P.; Zhang, H.; Dare, A.P.; Plunkett, B.; Espley, R.V.; Luo, Z.; Zhang, R.; Allan, A.C.; et al. MicroRNA172 Targets APETALA2 to Regulate Flavonoid Biosynthesis in Apple (Malus domestica). Hortic. Res. 2022, 9, uhab007. [Google Scholar] [CrossRef]
- Ramsay, N.A.; Glover, B.J. MYB–BHLH–WD40 Protein Complex and the Evolution of Cellular Diversity. Trends Plant Sci. 2005, 10, 63–70. [Google Scholar] [CrossRef]
- Zhang, B.; Yang, H.-J.; Yang, Y.-Z.; Zhu, Z.-Z.; Li, Y.-N.; Qu, D.; Zhao, Z.-Y. Mdm-MiR828 Participates in the Feedback Loop to Regulate Anthocyanin Accumulation in Apple Peel. Front. Plant Sci. 2020, 11, 608109. [Google Scholar] [CrossRef]
- Luo, Y.; Zhang, X.; Luo, Z.; Zhang, Q.; Liu, J. Identification and Characterization of MicroRNAs from Chinese Pollination Constant Non-Astringent Persimmon Using High-Throughput Sequencing. BMC Plant Biol. 2015, 15, 11. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, J.; Cui, W.; Guan, C.; Mao, W.; Zhang, Z. Improvement in Fruit Quality by Overexpressing MiR399a in Woodland Strawberry. J. Agric. Food Chem. 2017, 65, 7361–7370. [Google Scholar] [CrossRef]
- Li, D.; Mou, W.; Luo, Z.; Li, L.; Limwachiranon, J.; Mao, L.; Ying, T. Developmental and Stress Regulation on Expression of a Novel miRNA, Fan-MiR73 and Its Target ABI5 in Strawberry. Sci. Rep. 2016, 6, 28385. [Google Scholar] [CrossRef]
- Karlova, R.; van Haarst, J.C.; Maliepaard, C.; van de Geest, H.; Bovy, A.G.; Lammers, M.; Angenent, G.C.; de Maagd, R.A. Identification of MicroRNA Targets in Tomato Fruit Development Using High-Throughput Sequencing and Degradome Analysis. J. Exp. Bot. 2013, 64, 1863–1878. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Kong, J.; Lai, T.; Manning, K.; Wu, C.; Wang, Y.; Qin, C.; Li, B.; Yu, Z.; Zhang, X.; et al. Tuning LeSPL-CNR Expression by SlymiR157 Affects Tomato Fruit Ripening. Sci. Rep. 2015, 5, 7852. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, J.; Wu, Y.; Li, D.; Allan, A.C.; Yin, X. Genome-Wide Analysis of Coding and Non-Coding RNA Reveals a Conserved MiR164-NAC Regulatory Pathway for Fruit Ripening. New Phytol. 2020, 225, 1618–1634. [Google Scholar] [CrossRef] [PubMed]
- Bai, S.; Tian, Y.; Tan, C.; Bai, S.; Hao, J.; Hasi, A. Genome-Wide Identification of MicroRNAs Involved in the Regulation of Fruit Ripening and Climacteric Stages in Melon (Cucumis melo). Hortic. Res. 2020, 7, 106. [Google Scholar] [CrossRef]
- Wang, M.; Zhao, W.; Gao, L.; Zhao, L. Genome-Wide Profiling of Long Non-Coding RNAs from Tomato and a Comparison with MRNAs Associated with the Regulation of Fruit Ripening. BMC Plant Biol. 2018, 18, 75. [Google Scholar] [CrossRef]
- An, N.; Fan, S.; Wang, Y.; Zhang, L.; Gao, C.; Zhang, D.; Han, M. Genome-Wide Identification, Characterization and Expression Analysis of Long Non-Coding RNAs in Different Tissues of Apple. Gene 2018, 666, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, G.; Sharma, S.; Upadhyay, S.K.; Singh, K. Long Non-Coding RNAs Coordinate Developmental Transitions and Other Key Biological Processes in Grapevine. Sci. Rep. 2019, 9, 3552. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, C.; Feng, X.; Lai, R.; Gao, M.; Chen, W.; Wu, R. Integrated Analysis of LncRNA and MRNA Transcriptomes Reveals the Potential Regulatory Role of LncRNA in Kiwifruit Ripening and Softening. Sci. Rep. 2021, 11, 1671. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Shi, T.; Iqbal, S.; Zhang, Y.; Liu, L.; Gao, Z. Genome-Wide Discovery and Characterization of Flower Development Related Long Non-Coding RNAs in Prunus mume. BMC Plant Biol. 2019, 19, 64. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Duan, A.; Zhang, J.; He, C. Genome-Wide Analysis of Long Non-Coding RNAs at the Mature Stage of Sea Buckthorn (Hippophae Rhamnoides Linn) Fruit. Gene 2017, 596, 130–136. [Google Scholar] [CrossRef]
- Tian, Y.; Bai, S.; Dang, Z.; Hao, J.; Zhang, J.; Hasi, A. Genome-Wide Identification and Characterization of Long Non-Coding RNAs Involved in Fruit Ripening and the Climacteric in Cucumis melo. BMC Plant Biol. 2019, 19, 369. [Google Scholar] [CrossRef]
- Bai, L.; Chen, Q.; Jiang, L.; Lin, Y.; Ye, Y.; Liu, P.; Wang, X.; Tang, H. Comparative Transcriptome Analysis Uncovers the Regulatory Functions of Long Noncoding RNAs in Fruit Development and Color Changes of Fragaria pentaphylla. Hortic. Res. 2019, 6, 42. [Google Scholar] [CrossRef]
- Yu, T.; Tzeng, D.T.W.; Li, R.; Chen, J.; Zhong, S.; Fu, D.; Zhu, B.; Luo, Y.; Zhu, H. Genome-Wide Identification of Long Non-Coding RNA Targets of the Tomato MADS Box Transcription Factor RIN and Function Analysis. Ann. Bot. 2019, 123, 469–482. [Google Scholar] [CrossRef]
- Vrebalov, J.; Ruezinsky, D.; Padmanabhan, V.; White, R.; Medrano, D.; Drake, R.; Schuch, W.; Giovannoni, J. A MADS-Box Gene Necessary for Fruit Ripening at the Tomato Ripening-Inhibitor (Rin) Locus. Science 2002, 296, 343–346. [Google Scholar] [CrossRef]
- Tang, Y.; Qu, Z.; Lei, J.; He, R.; Adelson, D.L.; Zhu, Y.; Yang, Z.; Wang, D. The Long Noncoding RNA FRILAIR Regulates Strawberry Fruit Ripening by Functioning as a Noncanonical Target Mimic. PLoS Genet. 2021, 17, e1009461. [Google Scholar] [CrossRef]
- Xue, M.; Yi, H.; Wang, H. Identification of miRNAs Involved in SO2 Preservation in Vitis vinifera L. by Deep Sequencing. Environ. Exp. Bot. 2018, 153, 218–228. [Google Scholar] [CrossRef]
- Kumar, R.; Khurana, A.; Sharma, A.K. Role of Plant Hormones and Their Interplay in Development and Ripening of Fleshy Fruits. J. Exp. Bot. 2013, 65, 4561–4575. [Google Scholar] [CrossRef]
- Alexander, L. Ethylene Biosynthesis and Action in Tomato: A Model for Climacteric Fruit Ripening. J. Exp. Bot. 2002, 53, 2039–2055. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Chen, K.; Grierson, D. A Critical Evaluation of the Role of Ethylene and MADS Transcription Factors in the Network Controlling Fleshy Fruit Ripening. New Phytol. 2019, 221, 1724–1741. [Google Scholar] [CrossRef] [PubMed]
- Chung, M.-Y.; Nath, U.K.; Vrebalov, J.; Gapper, N.; Lee, J.M.; Lee, D.-J.; Kim, C.K.; Giovannoni, J. Ectopic Expression of miRNA172 in Tomato (Solanum Lycopersicum) Reveals Novel Function in Fruit Development through Regulation of an AP2 Transcription Factor. BMC Plant Biol. 2020, 20, 283. [Google Scholar] [CrossRef]
- Liu, M.; Pirrello, J.; Chervin, C.; Roustan, J.-P.; Bouzayen, M. Ethylene Control of Fruit Ripening: Revisiting the Complex Network of Transcriptional Regulation. Plant Physiol. 2015, 169, 2380–2390. [Google Scholar] [CrossRef]
- Moxon, S.; Jing, R.; Szittya, G.; Schwach, F.; Rusholme Pilcher, R.L.; Moulton, V.; Dalmay, T. Deep Sequencing of Tomato Short RNAs Identifies MicroRNAs Targeting Genes Involved in Fruit Ripening. Genome Res. 2008, 18, 1602–1609. [Google Scholar] [CrossRef]
- Zuo, J.; Zhu, B.; Fu, D.; Zhu, Y.; Ma, Y.; Chi, L.; Ju, Z.; Wang, Y.; Zhai, B.; Luo, Y. Sculpting the Maturation, Softening and Ethylene Pathway: The Influences of MicroRNAs on Tomato Fruits. BMC Genom. 2012, 13, 7. [Google Scholar] [CrossRef]
- Chen, Z.; Hu, L.; Han, N.; Hu, J.; Yang, Y.; Xiang, T.; Zhang, X.; Wang, L. Overexpression of a MiR393-Resistant Form of Transport Inhibitor Response Protein 1 (MTIR1) Enhances Salt Tolerance by Increased Osmoregulation and Na+ Exclusion in Arabidopsis Thaliana. Plant Cell Physiol. 2015, 56, 73–83. [Google Scholar] [CrossRef]
- Denver, J.B.; Ullah, H. MiR393s Regulate Salt Stress Response Pathway in Arabidopsis thaliana through Scaffold Protein RACK1A Mediated ABA Signaling Pathways. Plant Signal. Behav. 2019, 14, 1600394. [Google Scholar] [CrossRef]
- Pegler, J.L.; Nguyen, D.Q.; Oultram, J.M.J.; Grof, C.P.L.; Eamens, A.L. Molecular Manipulation of the MiR396/GRF Expression Module Alters the Salt Stress Response of Arabidopsis thaliana. Agronomy 2021, 11, 1751. [Google Scholar] [CrossRef]
- Cai, X.; Zhang, L.; Xiao, L.; Wen, Z.; Hou, Q.; Yang, K. Genome-Wide Identification of GRF Gene Family and Their Contribution to Abiotic Stress Response in Pitaya (Hylocereus polyrhizus). Int. J. Biol. Macromol. 2022, 223, 618–635. [Google Scholar] [CrossRef] [PubMed]
- Yaish, M.W.; Sunkar, R.; Zheng, Y.; Ji, B.; Al-Yahyai, R.; Farooq, S.A. A Genome-Wide Identification of the miRNAome in Response to Salinity Stress in Date Palm (Phoenix dactylifera L.). Front. Plant Sci. 2015, 6, 946. [Google Scholar] [CrossRef] [PubMed]
- Xie, R.; Zhang, J.; Ma, Y.; Pan, X.; Dong, C.; Pang, S.; He, S.; Deng, L.; Yi, S.; Zheng, Y.; et al. Combined Analysis of MRNA and miRNA Identifies Dehydration and Salinity Responsive Key Molecular Players in Citrus Roots. Sci. Rep. 2017, 7, 42094. [Google Scholar] [CrossRef]
- Pan, C.; Ye, L.; Zheng, Y.; Wang, Y.; Yang, D.; Liu, X.; Chen, L.; Zhang, Y.; Fei, Z.; Lu, G. Identification and Expression Profiling of MicroRNAs Involved in the Stigma Exsertion under High-Temperature Stress in Tomato. BMC Genom. 2017, 18, 843. [Google Scholar] [CrossRef]
- Lin, J.-S.; Kuo, C.-C.; Yang, I.-C.; Tsai, W.-A.; Shen, Y.-H.; Lin, C.-C.; Liang, Y.-C.; Li, Y.-C.; Kuo, Y.-W.; King, Y.-C.; et al. MicroRNA160 Modulates Plant Development and Heat Shock Protein Gene Expression to Mediate Heat Tolerance in Arabidopsis. Front. Plant Sci. 2018, 9, 68. [Google Scholar] [CrossRef]
- Tang, Y.; Du, G.; Xiang, J.; Hu, C.; Li, X.; Wang, W.; Zhu, H.; Qiao, L.; Zhao, C.; Wang, J.; et al. Genome-Wide Identification of Auxin Response Factor (ARF) Gene Family and the MiR160-ARF18-Mediated Response to Salt Stress in Peanut (Arachis hypogaea L.). Genomics 2022, 114, 171–184. [Google Scholar] [CrossRef]
- Gu, C.; Xu, H.-Y.; Zhou, Y.-H.; Yao, J.-L.; Xie, Z.-H.; Chen, Y.-Y.; Zhang, S.-L. Multiomics Analyses Unveil the Involvement of MicroRNAs in Pear Fruit Senescence under High- or Low-Temperature Conditions. Hortic. Res. 2020, 7, 196. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, W.; Wang, M.; Zhang, H.-Y.; Liu, J.-H. The MiR396b of Poncirus Trifoliata Functions in Cold Tolerance by Regulating ACC Oxidase Gene Expression and Modulating Ethylene–Polyamine Homeostasis. Plant Cell Physiol. 2016, 57, 1865–1878. [Google Scholar] [CrossRef]
- Moh, N.M.M.; Zhang, P.; Chen, Y.; Chen, M. Computational Identification of miRNAs and Temperature-Responsive LncRNAs From Mango (Mangifera indica L.). Front. Genet. 2021, 12, 607248. [Google Scholar] [CrossRef]
- Zhu, H.; Zhang, Y.; Tang, R.; Qu, H.; Duan, X.; Jiang, Y. Banana SRNAome and Degradome Identify MicroRNAs Functioning in Differential Responses to Temperature Stress. BMC Genom. 2019, 20, 33. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Wang, X.; Qi, K.; Bao, J.; Zhang, S.; Gu, C. Involvement of Long Non-Coding RNAs in Pear Fruit Senescence under High- and Low-Temperature Conditions. Hortic. Plant J. 2022, in press. [Google Scholar] [CrossRef]
- Wang, Y.; Gao, L.; Zhu, B.; Zhu, H.; Luo, Y.; Wang, Q.; Zuo, J. Integrative Analysis of Long Non-Coding RNA Acting as CeRNAs Involved in Chilling Injury in Tomato Fruit. Gene 2018, 667, 25–33. [Google Scholar] [CrossRef]
- Liu, G.; Liu, F.; Wang, Y.; Liu, X. A Novel Long Non-Coding RNA CIL1 Enhances Cold Stress Tolerance in Arabidopsis. Plant Sci. 2022, 323, 111370. [Google Scholar] [CrossRef]
- Zhou, R.; Yu, X.; Ottosen, C.-O.; Zhang, T.; Wu, Z.; Zhao, T. Unique miRNAs and Their Targets in Tomato Leaf Responding to Combined Drought and Heat Stress. BMC Plant Biol. 2020, 20, 107. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.; Balyan, S.; Jha, S.; Mathur, S. Novel Insights into Expansion and Functional Diversification of MIR169 Family in Tomato. Planta 2020, 251, 55. [Google Scholar] [CrossRef]
- Liu, D.; Song, Y.; Chen, Z.; Yu, D. Ectopic Expression of MiR396 Suppresses GRF Target Gene Expression and Alters Leaf Growth in Arabidopsis. Physiol. Plant. 2009, 136, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Reyes, J.L.; Chua, N.-H. ABA Induction of MiR159 Controls Transcript Levels of Two MYB Factors during Arabidopsis Seed Germination. Plant J. 2007, 49, 592–606. [Google Scholar] [CrossRef]
- Qin, T.; Zhao, H.; Cui, P.; Albesher, N.; Xiong, L. A Nucleus-Localized Long Non-Coding RNA Enhances Drought and Salt Stress Tolerance. Plant Physiol. 2017, 175, 1321–1336. [Google Scholar] [CrossRef]
- Li, Z.; Yang, S.; Tang, J.; Song, H. miRNA Expression Profiling Reveals the Regulators of Kiwifruit Response to Botrytis cinerea. Physiol. Mol. Plant Pathol. 2022, 120, 101851. [Google Scholar] [CrossRef]
- Wang, C.; Han, J.; Liu, C.; Kibet, K.N.; Kayesh, E.; Shangguan, L.; Li, X.; Fang, J. Identification of MicroRNAs from Amur Grape (Vitis amurensis Rupr.) by Deep Sequencing and Analysis of MicroRNA Variations with Bioinformatics. BMC Genom. 2012, 13, 122. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.; Lu, Y.; Bai, S.; Zhang, W.; Duan, X.; Meng, D.; Wang, Z.; Wang, A.; Zhou, Z.; Li, T. Cloning and Characterization of miRNAs and Their Targets, Including a Novel miRNA-Targeted NBS–LRR Protein Class Gene in Apple (Golden Delicious). Mol. Plant 2014, 7, 218–230. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Li, S.; Zhang, S.; Feng, T.; Zhang, Z.; Luo, S.; Mao, H.; Borkovich, K.A.; Ouyang, S. SlymiR482e-3p Mediates Tomato Wilt Disease by Modulating Ethylene Response Pathway. Plant Biotechnol. J. 2021, 19, 17–19. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Mohanty, J.N.; Chand, S.K.; Joshi, R.K. Can-MiRn37a Mediated Suppression of Ethylene Response Factors Enhances the Resistance of Chilli against Anthracnose Pathogen Colletotrichum truncatum L. Plant Sci. 2018, 267, 135–147. [Google Scholar] [CrossRef]
- Liu, M.; Yu, H.; Zhao, G.; Huang, Q.; Lu, Y.; Ouyang, B. Identification of Drought-Responsive MicroRNAs in Tomato Using High-Throughput Sequencing. Funct. Integr. Genom. 2018, 18, 67–78. [Google Scholar] [CrossRef]
- Naqvi, A.R.; Haq, Q.M.; Mukherjee, S.K. MicroRNA Profiling of Tomato Leaf Curl New Delhi Virus (Tolcndv) Infected Tomato Leaves Indicates That Deregulation of Mir159/319 and Mir172 Might Be Linked with Leaf Curl Disease. Virol. J. 2010, 7, 281. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, Y.; Wang, S.; Hao, L.; Wang, S.; Xu, C.; Jiang, F.; Li, T. Characterization of Genome-Wide MicroRNAs and Their Roles in Development and Biotic Stress in Pear. Planta 2019, 249, 693–707. [Google Scholar] [CrossRef]
- Soto-Suárez, M.; Baldrich, P.; Weigel, D.; Rubio-Somoza, I.; San Segundo, B. The Arabidopsis MiR396 Mediates Pathogen-Associated Molecular Pattern-Triggered Immune Responses against Fungal Pathogens. Sci. Rep. 2017, 7, 44898. [Google Scholar] [CrossRef] [PubMed]
- Fu, X.; Li, J.; Min, D.; Zhao, X.; Liu, J.; Li, F.; Li, X.; Zhang, X. LncRNA4504 Involved in Methyl Jasmonate-Induced Resistance to Botrytis Cinerea in Postharvest Tomato Fruit. Sci. Hortic. 2022, 305, 111381. [Google Scholar] [CrossRef]
- Tang, J.; Chu, C. MicroRNAs in Crop Improvement: Fine-Tuners for Complex Traits. Nat. Plants 2017, 3, 17077. [Google Scholar] [CrossRef]
- Miao, C.; Wang, D.; He, R.; Liu, S.; Zhu, J. Mutations in MIR396e and MIR396f Increase Grain Size and Modulate Shoot Architecture in Rice. Plant Biotechnol. J. 2020, 18, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Zong, Y.; Xue, C.; Wang, S.; Jin, S.; Zhu, Z.; Wang, Y.; Anzalone, A.V.; Raguram, A.; Doman, J.L.; et al. Prime Genome Editing in Rice and Wheat. Nat. Biotechnol. 2020, 38, 582–585. [Google Scholar] [CrossRef] [PubMed]
- Molla, K.A.; Sretenovic, S.; Bansal, K.C.; Qi, Y. Precise Plant Genome Editing Using Base Editors and Prime Editors. Nat. Plants 2021, 7, 1166–1187. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Li, J.; Liu, X.; Shan, T.; Qin, R.; Wei, P. Development of Plant Prime-Editing Systems for Precise Genome Editing. Plant Commun. 2020, 1, 100043. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).