Maize Production under Drought Stress: Nutrient Supply, Yield Prediction
Abstract
:1. Introduction
2. Materials and Methods
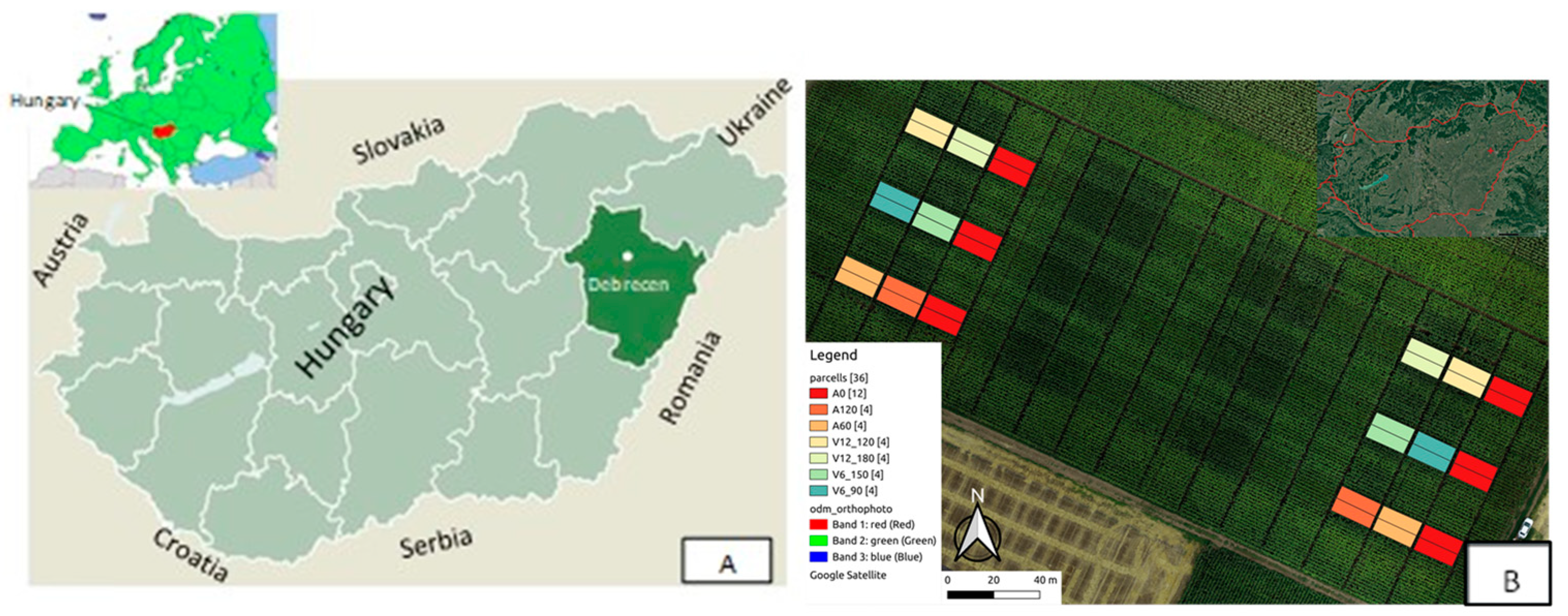
2.1. Experiment Site
2.2. Soil Data
2.3. Experiment Design and Treatments
| Name | Treatment |
| A0 | Unfertilised control; |
| A60 | 60 kg N ha−1 before sowing; |
| A120 | 120 kg N ha−1 before sowing; |
| V690 | 60 kg N ha−1 before sowing + 30 kg N ha−1 during the V6 phenophase; |
| V6150 | 120 kg N ha−1 before sowing + 30 kg N ha−1 during the V6 phenophase; |
| V12120 | 60 kg N ha−1 before sowing + 30 kg N ha−1 during the V6 phenophase + 30 kg N ha−1 during the V12 phenophase; |
| V12180 | 120 kg N ha−1 before sowing + 30 kg N ha−1 during the V6 phenophase + 30 kg N ha−1 during the V12 phenophase. |
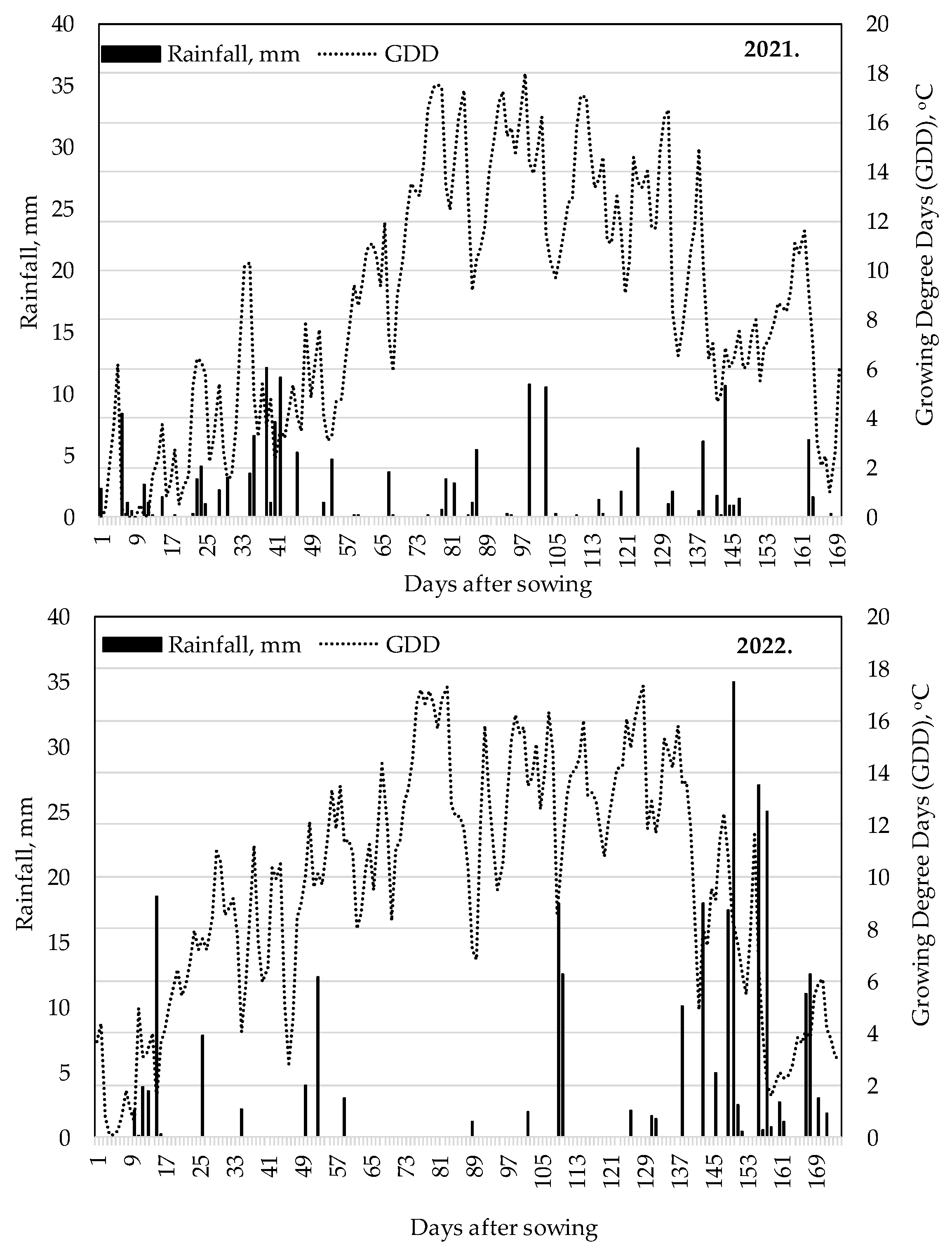
2.4. Climatic Data at the Experiment Site
2.5. Measuring Instruments and Test Methods
2.6. Statistical Analysis
3. Results
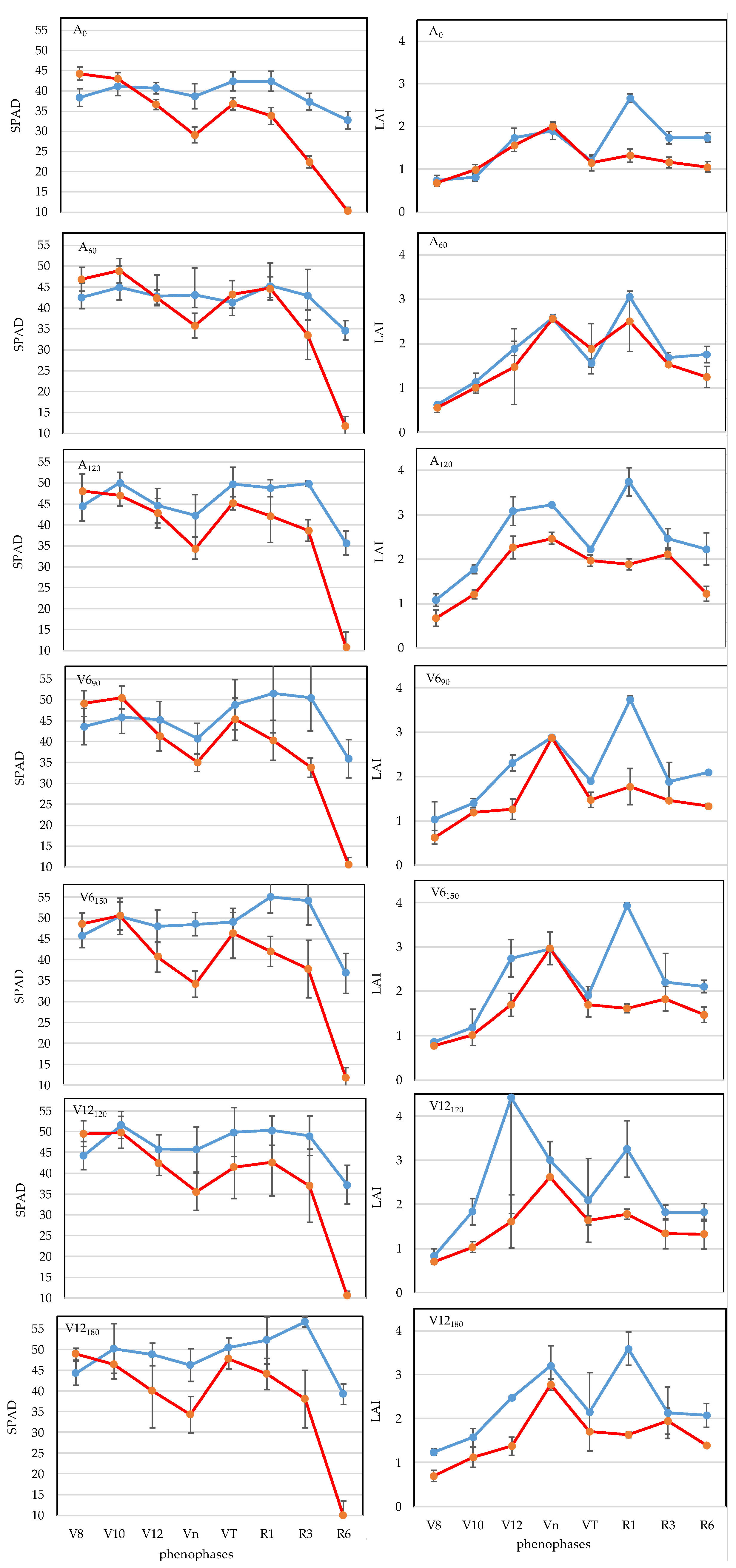
3.1. Chlorophyll Concentration (SPAD)
3.2. Leaf Area Index (LAI)
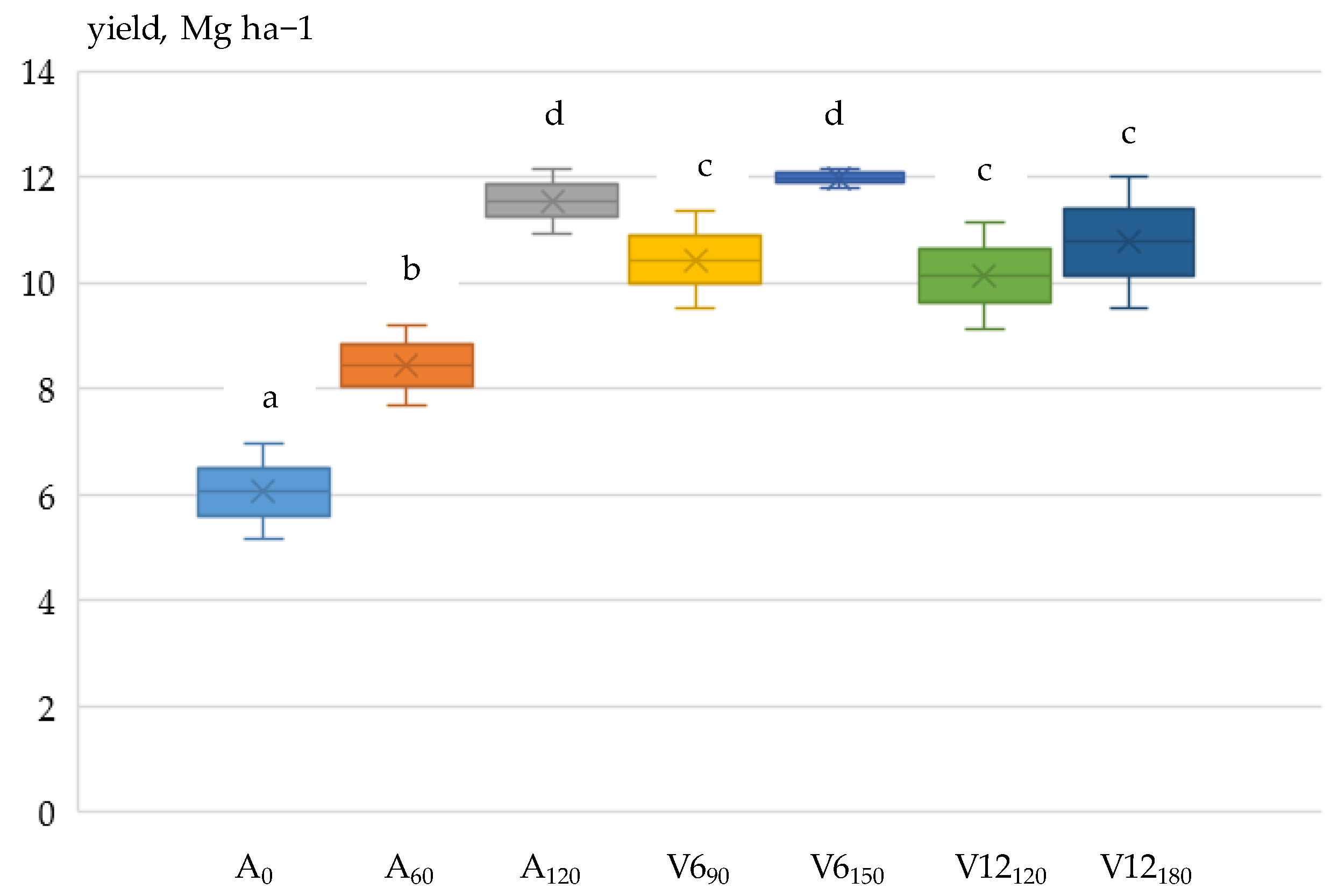
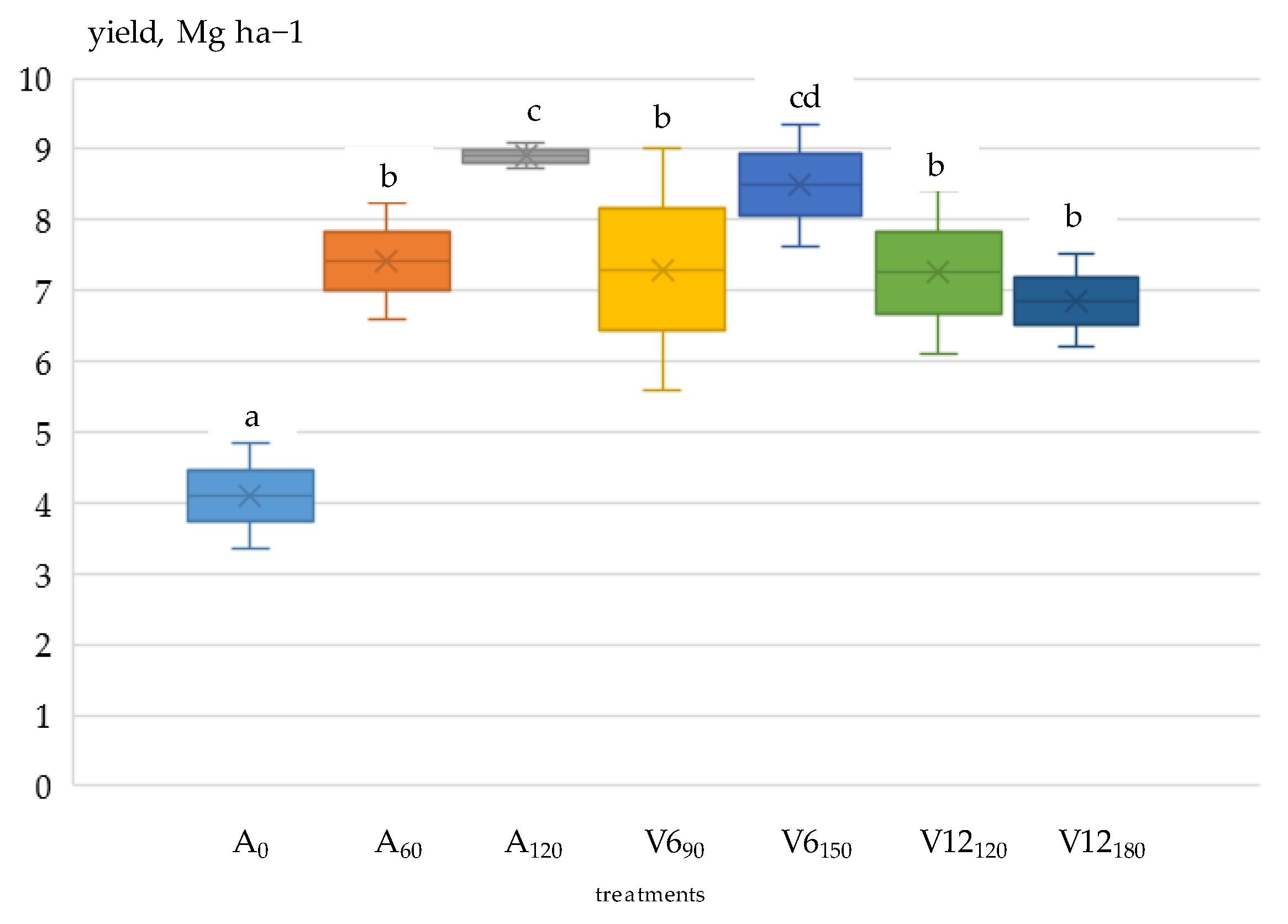
3.3. Grain Yield
3.4. Correlation Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Erenstein, O.; Jaleta, M.; Sonder, K.; Mottaleb, K.; Prasanna, B.M. Global maize production, consumption and trade: Trends and R&D implications. Food Secur. 2022, 14, 1295–1319. [Google Scholar] [CrossRef]
- FAO (Food and Agriculture Organization of the United Nation). The Future of Food and Agriculture—Drivers and Triggers for Transformation; The Future of Food and Agriculture: Rome, Italy, 2022. [Google Scholar] [CrossRef]
- KSH (Hungarian Central Statistical Office). 2022. Available online: https://www.ksh.hu/stadat?lang=hu&theme=mez (accessed on 1 January 2023).
- OMSZ (The Hungarian Meteorological Service). 2020. Available online: https://www.met.hu/eghajlat/magyarorszag_eghajlata/eghajlati_adatsorok/Debrecen/adatok/napi_adatok/index.php (accessed on 17 April 2022).
- Bihari, Z.; Babolcsai, G.; Bartholy, J.; Ferenczi, Z.; Gerhátné Kerényi, J.; Haszpra, L.; Homokiné Ujváry, K.; Kovács, T.; Lakatos, M.; Németh, Á.; et al. Éghajlat. In Magyarország Nemzeti Atlasza 2. Kötet; Kocsis, K., Horváth, G., Keresztesi, Z., Nemerkényi, Z., Eds.; Természeti környezet; MTA CSFK Földrajztudományi Intézet: Budapest, Hungary, 2018; pp. 58–69. [Google Scholar]
- Seleiman, M.F.; Al-Suhaibani, N.; Ali, N.; Akmal, M.; Alotaibi, M.; Refay, Y.; Dindaroglu, T.; Abdul-Wajid, H.H.; Battaglia, M.L. Drought Stress Impacts on Plants and Different Approaches to Alleviate Its Adverse Effects. Plants 2021, 10, 259. [Google Scholar] [CrossRef] [PubMed]
- Sheoran, S.; Kaur, Y.; Kumar, S.; Shukla, S.; Rakshit, S.; Kumar, R. Recent Advances for Drought Stress Tolerance in Maize (Zea mays L.): Present Status and Future Prospects. Front. Plant Sci. 2022, 13, 872566. [Google Scholar] [CrossRef]
- Zhang, F.; Zhou, G. Estimation of vegetation water content using hyperspectral vegetation indices: A comparison of crop water indicators in response to water stress treatments for summer maize. BMC Ecol. 2019, 19, 18. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lin, D.; Wang, J.; Yang, L.; Wang, Y. Multivariate Analysis Models Based on Full Spectra Range and Effective Wavelengths Using Different Transformation Techniques for Rapid Estimation of Leaf Nitrogen Concentration in Winter Wheat. Front. Plant Sci. 2020, 11, 755. [Google Scholar] [CrossRef]
- Ahmad, S.; Wang, G.Y.; Muhammad, I.; Chi, Y.X.; Zeeshan, M.; Nasar, J.; Zhou, X.B. Interactive Effects of Melatonin and Nitrogen Improve Drought Tolerance of Maize Seedlings by Regulating Growth and Physiochemical Attributes. Antioxidants 2022, 11, 359. [Google Scholar] [CrossRef]
- Barnabás, B.; Jäger, K.; Fehér, A. The effect of drought and heat stress on reproductive processes in cereals. Plant Cell Environ. 2008, 31, 11–38. [Google Scholar] [CrossRef] [PubMed]
- Gong, F.; Yang, L.; Tai, F.; Hu, X.; Wang, W. “Omics” of maize stress response for sustainable food production: Opportunities and challenges. Omics J. Integr. Biol. 2014, 18, 714–732. [Google Scholar] [CrossRef]
- Bhat, M.A.; Kumar, V.; Bhat, M.A.; Wani, I.A.; Dar, F.L.; Farooq, I.; Bhatti, F.; Koser, R.; Rahman, S.; Jan, A.T. Mechanistic Insights of the Interaction of Plant Growth-Promoting Rhizobacteria (PGPR) With Plant Roots Toward Enhancing Plant Productivity by Alleviating Salinity Stress. Front. Microbiol. 2020, 11, 1952. [Google Scholar] [CrossRef] [PubMed]
- Grant, R.F.; Jackson, B.S.; Kiniry, J.R.; Arkin, G.F. Water deficit timing effects on yield components in maize. Agron. J. 1989, 81, 61–65. [Google Scholar] [CrossRef]
- Ahmad, N.; Malagoli, M.; Wirtz, M.; Hell, R. Drought stress in maize causes differential acclimation responses of glutathione and sulfur metabolism in leaves and roots. BMC Plant Biol. 2016, 16, 247. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Jin, Z.N.; Zhuang, Q.L.; Ciais, P.; Bernacchi, C.; Wang, X.H.; Makowski, D.; Lobell, D. The important but weakening maize yield benefit of grain filling prolongation in the US Midwest. Glob. Chang. Boil. 2018, 24, 4718–4730. [Google Scholar] [CrossRef]
- Mimić, G.; Brdar, S.; Brkić, M.; Panić, M.; Marko, O.; Crnojević, V. Engineering Meteorological Features to Select Stress Tolerant Hybrids in Maize. Sci. Rep. 2020, 10, 3421. [Google Scholar] [CrossRef]
- Salgado-Aguilar, M.; Molnar, T.; Pons-Hernández, J.L.; Covarrubias-Prieto, J.; Ramírez-Pimentel, J.G.; Raya-Pérez, J.C.; Iturriaga, G. Physiological and biochemical analyses of novel drought-tolerant maize lines reveal osmoprotectant accumulation at silking stage. Chil. J. Agric. Res. 2020, 80, 241–252. [Google Scholar] [CrossRef]
- Krell, N.T.; Morgan, B.E.; Gower, D.; Caylor, K.K. Consequences of dryland maize planting decisions under increased seasonal rainfall variability. Water Resour. Res. 2021, 57, e2020WR029362. [Google Scholar] [CrossRef]
- Sah, R.P.; Chakraborty, M.; Prasad, K.; Pandit, M.; Tudu, V.K.; Chakravarty, M.K.; Narayan, S.C.; Rana, M.; Moharana, D. Impact of water deficit stress in maize: Phenology and yield components. Sci. Rep. 2020, 10, 1–5. [Google Scholar] [CrossRef]
- Ványiné Széles, A.; Megyes, A.; Nagy, J. Irrigation and nitrogen effects on the leaf chlorophyll content and grain yield of maize in different crop years. Agric. Water Manag. 2012, 107, 133–144. [Google Scholar] [CrossRef]
- Kalaji, H.M.; Dabrowski, P.; Cetner, M.D.; Samborska, I.A.; Lukasik, I.; Brestic, M.; Zivcak, M.; Tomasz, H.; Mojski, J.; Kociel, H.; et al. A comparison between different chlorophyll content meters under nutrient deficiency conditions. J. Plant Nutr. 2017, 40, 1024–1034. [Google Scholar] [CrossRef]
- Zhao, B.; Ata-Ul-Karim, S.T.; Duan, A.; Liu, Z.; Wang, X.; Xiao, J.; Liu, Z.; Qin, A.; Ning, D.; Zhang, W.; et al. Determination of critical nitrogen concentration and dilution curve based on leaf area index for summer maize. Field Crops Res. 2018, 228, 195–203. [Google Scholar] [CrossRef]
- Liang, L.; Geng, D.; Yan, J.; Qiu, S.; Di, L.; Wang, S.; Xu, L.; Wang, L.; Kang, J.; Li, L. Estimating Crop LAI Using Spectral Feature Extraction and the Hybrid Inversion Method. Remote Sens. 2020, 12, 3534. [Google Scholar] [CrossRef]
- Davies, B.; Coulter, J.A.; Pagliari, P.H. Timing and rate of nitrogen fertilization influence maize yield and nitrogen use efficiency. PLoS ONE 2020, 15, e0233674. [Google Scholar] [CrossRef]
- Gheith, E.M.S.; El-Badry, O.Z.; Lamlom, S.F.; Ali, H.M.; Siddiqui, M.H.; Ghareeb, R.Y.; El-Sheikh, M.H.; Jebril, J.; Abdelsalam, N.R.; Kandil, E.E. Maize (Zea mays L.) Productivity and Nitrogen Use Efficiency in Response to Nitrogen Application Levels and Time. Front. Plant Sci. 2022, 13, 941343. [Google Scholar] [CrossRef]
- Xu, K.; Chai, Q.; Hu, F.; Fan, Z.; Yin, W. N-fertilizer postponing application improves dry matter translocation and increases system productivity of wheat/maize intercropping. Sci. Rep. 2021, 11, 22825. [Google Scholar] [CrossRef] [PubMed]
- Hammad, H.M.; Ahmad, A.; Wajid, A.; Akhter, J. Maize response to time and rate of nitrogen application. Pak. J. Bot. 2011, 43, 1935–1942. [Google Scholar]
- Walling, E.; Vaneeckhaute, C. Greenhouse gas emissions from inorganic and organic fertilizer production and use: A review of emission factors and their variability. J. Environ. Manag. 2020, 276, 111211. [Google Scholar] [CrossRef] [PubMed]
- Kizilgeci, F.; Yildirim, M.; Islam, M.S.; Ratnasekera, D.; Iqbal, M.A.; Sabagh, A.E. Normalized Difference Vegetation Index and Chlorophyll Content for Precision Nitrogen Management in Durum Wheat Cultivars under Semi-Arid Conditions. Sustainability 2021, 13, 3725. [Google Scholar] [CrossRef]
- Nosheen, S.; Ajmal, I.; Song, Y. Microbes as biofertilizers, a potential approach for sustainable crop production. Sustainability 2021, 13, 1868. [Google Scholar] [CrossRef]
- Saberioon, M.M.; Amin, M.S.M.; Anuar, A.R.; Gholizadeh, A.; Wayayok, A. Assessment of rice leaf chlorophyll content using visible bands at different growth stages at both the leaf and canopy scale. Int. J. Appl. Earth Obs. Geoinf. 2014, 32, 35–45. [Google Scholar] [CrossRef]
- Zhao, G.; Miao, Y.; Wang, H.; Su, M.; Fan, M.; Zhang, F.; Jiang, R.; Zhang, Z.; Liu, C.; Liu, P.; et al. A preliminary precision rice management system for increasing both grain yield and nitrogen use efficiency. Field Crops Res. 2013, 154, 10–23. [Google Scholar] [CrossRef]
- Sun, X.; Yang, Z.; Su, P.; Wei, K.; Wang, Z.; Yang, C.; Wang, C.; Qin, M.; Xiao, L.; Yang, W.; et al. Non-destructive monitoring of maize LAI by fusing UAV spectral and textural features. Front. Plant Sci. 2023, 14, 1158837. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Gómez-Dans, J.L.; Huang, H.; Ma, H.; Wu, Q.; Lewis, P.E.; Liang, S.; Chen, Z.; Xue, J.H.; Wu, Y.; et al. Assimilation of remote sensing into crop growth models: Current status and perspectives. Agric. Meteorol. 2019, 276, 107609. [Google Scholar] [CrossRef]
- Mateo-Sanchis, A.; Piles, M.; Amorós-López, J.; Muñoz-Marí, J.; Adsuara, J.E.; Moreno-Martínez, Á.; Camps-Valls, G. Learning main drivers of crop progress and failure in Europe with interpretable machine learning. Int. J. Appl. Earth Obs. Geoinf. 2021, 104, 102574. [Google Scholar] [CrossRef]
- McMaster, G.S.; Wilhelm, W.W. Growing degree-days: One equation, two interpretations. Agric. Meteorol. 1997, 87, 291–300. [Google Scholar] [CrossRef]
- Davidson, H.R.; Campbell, C.A. The effect of temperature, moisture and nitrogen on the rate of development of spring wheat as measured by degree days. Can. J. Plant Sci. 1983, 63, 833–846. [Google Scholar] [CrossRef]
- Gallagher, J.N. Field studies of cereal leaf growth: I. Initation and expansion in relation to temperature and ontogeny. J. Exp. Bot. 1979, 30, 625–636. Available online: https://www.jstor.org/stable/23689719 (accessed on 17 April 2022). [CrossRef]
- Menyhért, Z. A Kukoricatermesztés Kézikönyve; Mezőgazdasági Kiadó: Budapest, Hungary, 1985. [Google Scholar]
- Dwyer, L.M.; Anderson, A.M.; Ma, B.L.; Stewart, D.W.; Tollenaar, M.; Gregorich, E. Quantifying the nonlinearity in chlorophyll meter response to corn leaf nitrogen concentration. Can. J. Plant Sci. 1995, 75, 179–182. [Google Scholar] [CrossRef]
- Earl, H.J.; Tollenaar, M. Maize leaf absorptance of photosynthetically active radiation and its estimation using a chlorophyll meter. Crop Sci. 1997, 37, 436–440. [Google Scholar] [CrossRef]
- Spitkó, T.; Nagy, Z.; Zsubori, Z.T.; Szőke, C.; Berzy, T.; Pintér, J.; Marton, C.L. Connection between normalized difference vegetation index and yield in maize. Plant Soil Environ. 2016, 62, 293–298. [Google Scholar] [CrossRef]
- Konica-Minolta. Chlorophyll Meter SPAD-502 Instruction Manual; Konica Minolta Sensing, Inc.: Plainfield, IL, USA, 1989. [Google Scholar]
- Jordan, D.R.; Hunt, C.H.; Cruickshank, A.W.; Borrell, A.K.; Henzell, R.G. The relationship between the stay-green trait and grain yield in elite sorghum hybrids grown in a range of environments. Crop Sci. 2012, 52, 1153–1161. [Google Scholar] [CrossRef]
- Gabriel, J.L.; Quemada, M.; Alonso-Ayuso, M.; Lizaso, J.I.; Martín-Lammerding, D. Predicting N status in maize with clip sensors: Choosing sensor, leaf sampling point, and timing. Sensors 2019, 19, 3881. [Google Scholar] [CrossRef]
- Costa, C.; Dwyer, L.M.; Dutilleul, P.; Stewart, D.W.; Ma, B.L.; Smith, D.L. Inter-relationships of applied nitrogen, SPAD, and yield of leafy and non-leafy maize genotypes. J. Plant Nutr. 2001, 24, 1173–1194. [Google Scholar] [CrossRef]
- Potter, E.; Wood, J.; Nicholl, C. SunScan Canopy Analysis System User Manual SS1-UM-1.05; Delta-T Devices: Cambridge, UK, 1996. [Google Scholar]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2023; Available online: https://www.R-project.org/ (accessed on 21 March 2023).
- Andrade, M.A.F.; Ramos-Cairo, P.A.; Santos, J.L. Water relations and photosynthesis of young coffee plants under two water regimes and different N and K doses. Agrociencia 2015, 49, 153–161. [Google Scholar]
- Yordanov, I.; Velikova, V.; Tsonev, T. Plant responses to drought, acclimation, and stress tolerance. Photosynthetica 2000, 38, 171–186. [Google Scholar] [CrossRef]
- Sun, J.; Gao, J.; Wang, Z.; Hu, S.; Zhang, F.; Bao, H.; Fan, Y. Maize Canopy Photosynthetic Efficiency, Plant Growth, and Yield Responses to Tillage Depth. Agronomy 2019, 9, 3. [Google Scholar] [CrossRef]
- Çakir, R. Effect of water stress at different development stages on vegetative and reproductive growth of corn. Field Crops Res. 2004, 89, 1–16. [Google Scholar] [CrossRef]
- Li, Y.; Tao, H.; Zhang, B.; Huang, S.; Wang, P. Timing of Water Deficit Limits Maize Kernel Setting in Association With Changes in the Source-Flow-Sink Relationship. Front. Plant Sci. 2018, 9, 1326. [Google Scholar] [CrossRef]
- Guna, A.; Zhang, J.; Tong, S.; Bao, Y.; Han, A.; Li, K. Effect of Climate Change on Maize Yield in the Growing Season: A Case Study of the Songliao Plain Maize Belt. Water 2019, 11, 2108. [Google Scholar] [CrossRef]
- Almeida, G.D.; Nair, S.; Borém, A.; Cairns, J.; Trachsel, S.; Ribaut, J.M.; Bänziger, M.; Prasanna, B.M.; Crossa, J.; Babu, R. Molecular mapping across three populations reveals a QTL hotspot region on chromosome 3 for secondary traits associated with drought tolerance in tropical maize. Mol. Breed. 2014, 34, 701–715. [Google Scholar] [CrossRef]
- Neiff, N.; Trachsel, S.; Valentinuz, O.R.; Balbi, C.N.; Andrade, F.H. High temperatures around flowering in maize: Effects on photosynthesis and grain yield in three genotypes. Crop Sci. 2016, 56, 2702–2712. [Google Scholar] [CrossRef]
- Șimon, A.; Moraru, P.I.; Ceclan, A.; Russu, F.; Chețan, F.; Bărdaș, M.; Popa, A.; Rusu, T.; Pop, A.I.; Bogdan, I. The Impact of Climatic Factors on the Development Stages of Maize Crop in the Transylvanian Plain. Agronomy 2023, 13, 1612. [Google Scholar] [CrossRef]
- Di Paolo, E.; Rinaldi, M. Yield response of corn to irrigation and nitrogen fertilization in a Mediterranean environment. Field Crops Res. 2008, 105, 202–210. [Google Scholar] [CrossRef]
- Li, S.X.; Wang, Z.H.; Li, S.Q.; Gao, Y.J. Effect of nitrogen fertilization under plastic and non-plastic mulched conditions on water use by maize plants in dryland areas of China. Agric. Water Manag. 2015, 162, 15–32. [Google Scholar] [CrossRef]
- Rutkowska, A. Productivity, nitrogen use efficiency and water use efficiency of maize for grain in long term field experiments. Pol. J. Agron. 2019, 37, 11–15. [Google Scholar] [CrossRef]
- Plett, D.C.; Ranathunge, K.; Melino, V.J.; Kuya, N.; Uga, Y.; Kronzucker, H.J. The intersection of nitrogen nutrition and water use in plants: New paths toward improved crop productivity. J. Exp. Bot. 2020, 71, 4452–4468. [Google Scholar] [CrossRef]
- Schepers, J.S.; Francis, D.D.; Vigil, M.; Below, F.E. Comparison of corn leaf nitrogen concentration and chlorophyll meter readings. Commun. Soil Sci. Plant Anal. 1992, 23, 2173–2187. [Google Scholar] [CrossRef]
- Nagy, J. Impact of Fertilization and Irrigation on the Correlation between the Soil Plant Analysis Development Value and Yield of Maize. Commun. Soil Sci. Plant Anal. 2010, 41, 1293–1305. [Google Scholar] [CrossRef]
- Rorie, R.L.; Purcell, L.C.; Mozaffari, M.; Karcher, D.E.; King, C.A.; Marsh, M.C.; Longer, D.E. Association of “greenness” in corn with yield and leaf nitrogen concentration. Agron. J. 2011, 103, 529–535. [Google Scholar] [CrossRef]
- Ványiné Széles, A.; Nagy, J. Effect of nutrition and water supply on the yield and grain protein content of maize hybrids. Aust. J. Crop Sci. 2012, 6, 290–381. [Google Scholar]
- Bai, H.; Purcell, L.C. Evaluation of Soybean Greenness from Ground and Aerial Platforms and the Association with Leaf Nitrogen Concentration in Response to Drought. Crop Sci. 2019, 59, 2763–2773. [Google Scholar] [CrossRef]
- Kaler, A.S.; Abdel-Haleem, H.; Fritschi, F.B.; Gillman, J.D.; Ray, J.D.; Smith, J.R.; Purcell, L.C. Genome-wide association mapping of dark green color index using a diverse panel of soybean accessions. Sci. Rep. 2020, 10, 5166. [Google Scholar] [CrossRef]
- Tajul, M.I.; Alam, M.M.; Hossain, S.M.; Naher, K.; Rafii, M.Y.; Latif, M.A. Influence of plant population and nitrogen-fertilizer at various levels on growth and growth efficiency of maize. Sci. World J. 2013, 2013, 193018. [Google Scholar] [CrossRef] [PubMed]
- Campos-Taberner, M.; García-Haro, F.J.; Camps-Valls, G.; Grau-Muedra, G.; Nutini, F.; Crema, A.; Boschetti, M. Multitemporal and multiresolution leaf area index retrieval for operational local rice crop monitoring. Remote Sens. Environ. 2016, 187, 102–118. [Google Scholar] [CrossRef]
- Berdjour, A.; Dugje, I.Y.; Rahman, N.A.; Odoom, D.A.; Kamara, A.Y.; Ajala, S. Direct Estimation of Maize Leaf Area Index as Influenced by Organic and Inorganic Fertilizer Rates in Guinea Savanna. J. Agric. Sci. 2020, 12, 66–76. [Google Scholar] [CrossRef]
- Ren, Y.; Li, Q.; Du, X.; Zhang, Y.; Wang, H.; Shi, G.; Wei, M. Analysis of Corn Yield Prediction Potential at Various Growth Phases Using a Process-Based Model and Deep Learning. Plants 2023, 12, 446. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Wiatrak, P. Corn production and plant characteristics response to N fertilization management in dry-land conventional tillage system. Int. J. Plant Prod. 2012, 5, 405–416. [Google Scholar] [CrossRef]
- Kogo, B.K.; Kumar, L.; Koech, R.; Langat, P. Modelling Impacts of Climate Change on Maize (Zea mays L.) Growth and Productivity: A Review of Models, Outputs and Limitations. J. Geosci. Environ. Prot. 2019, 7, 76–95. [Google Scholar] [CrossRef]
- Mbaye, M.L.; Sylla, M.B.; Tall, M. Impacts of 1.5 and 2.0 °C Global Warming on Water Balance Components over Senegal in West Africa. Atmosphere 2019, 10, 712. [Google Scholar] [CrossRef]
- Adamo, N.; Al-Ansari, N.; Sissakian, V.; Fahmi, K.J.; Abed, S.A. Climate Change: Droughts and Increasing Desertification in the Middle East, with Special Reference to Iraq. Engineering 2022, 14, 235–273. [Google Scholar] [CrossRef]
- Bista, D.R.; Heckathorn, S.A.; Jayawardena, D.M.; Mishra, S.; Boldt, J.K. Effects of Drought on Nutrient Uptake and the Levels of Nutrient-Uptake Proteins in Roots of Drought-Sensitive and -Tolerant Grasses. Plants 2018, 7, 28. [Google Scholar] [CrossRef]
- Dubey, A.; Kumar, A.; Malla, M.A.; Chowdhary, K.; Singh, G.; Ravikanth, G.; Harish Sharma, S.; Saati-Santamaria, Z.; Menéndez, E.; Dames, J.F. Approaches for the amelioration of adverse effects of drought stress on crop plants. Front. Biosci. 2021, 26, 928–947. [Google Scholar] [CrossRef]
- Gitelson, A.A.; Andrés, V.; Arkebauer, T.J.; Rundquist, D.C.; Galina, K.; Bryan, L. Remote estimation of leaf area index and green leaf biomass in maize canopies. Geophys. Res. Lett. 2003, 30, 335–343. [Google Scholar] [CrossRef]
- Zhang, Y.Q.; Wang, J.; Gong, S.; Xu, D.; Mo, Y. Straw mulching enhanced the photosynthetic capacity of field maize by increasing the leaf N use efficiency. Agric. Water Manag. 2019, 218, 60–67. [Google Scholar] [CrossRef]
- Cheng, Q.; Xu, H.; Fei, S.; Li, Z.; Chen, Z. Estimation of Maize LAI Using Ensemble Learning and UAV Multispectral Imagery under Different Water and Fertilizer Treatments. Agriculture 2022, 12, 1267. [Google Scholar] [CrossRef]
- Anjum, S.A.; Xie, X.-y.; Wang, L.-c.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar]
- Karimpour, M. Effect of Drought Stress on RWC and Chlorophyll Content on Wheat (Triticum durum L.) Genotypes. World Essays J. 2019, 7, 52–56. [Google Scholar]
- Monteoliva, M.I.; Guzzo, M.C.; Posada, G.A. Breeding for Drought Tolerance by Monitoring Chlorophyll Content. Gene Technol. 2021, 10, 165. [Google Scholar]
- Piekielek, W.P.; Fox, R.H.; Toth, J.D.; Macneal, K.E. Use of chlorophyll meter at the early dent stage of corn to evaulate nitrogen sufficiency. Agron. J. 1995, 87, 403–408. [Google Scholar] [CrossRef]
- He, M.; Dijkstra, F.A. Drought effect on plant nitrogen and phosphorus: A meta-analysis. New Phytol. 2014, 204, 924–931. [Google Scholar] [CrossRef]
- Guo, J.; Fan, J.; Xiang, Y.; Zhang, F.; Yan, S.; Zhang, X.; Zheng, J.; Hou, X.; Tang, Z.; Li, Z. Maize leaf functional responses to blending urea and slow-release nitrogen fertilizer under various drip irrigation regimes. Agric. Water Manag. 2022, 262, 107396. [Google Scholar] [CrossRef]
- Menyhért, Z.; Ángyán, J.; Radics, L. A levélfelület-index (LAI), a fényviszonyok és a termés kapcsolata eltérő vetésidejű és tenyészterületű kukorica állományokban. Növénytermelés 1980, 29, 357–369. [Google Scholar]
- Berzsenyi, Z. A növényszám hatása a kukorica növekedésének és növekedési jellemzőinek dinamikájára I. Növénytermelés 1989, 38, 395–407. [Google Scholar]
- Tong, S.Y.; Song, F.B.; Xu, H.W. Differences of Morphological Senescence of Leaves in Various Maize Varieties during Mature Period of Seed. Acta Agric. Boreali Sin. 2009, 24, 11–15. [Google Scholar] [CrossRef]
- Liu, G.Z.; Hou, P.; Xie, R.Z.; Ming, B.; Wang, K.R.; Xu, W.J.; Liu, W.M.; Yang, Y.S.; Li, S.K. Canopy characteristics of high-yield maize with yield potential of 22.5 Mg ha−1. Field Crops Res. 2017, 213, 221–230. [Google Scholar] [CrossRef]
- Ocwa, A.; Harsanyi, E.; Széles, A.; Holb, I.J.; Szabó, S.; Rátonyi, T.; Mohammed, S. A bibliographic review of climate change and fertilization as the main drivers of maize yield: Implications for food security. Agric. Food Secur. 2023, 12, 14. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, Y.; Fu, W.; Guo, W.; Ren, N.; Zhao, Y.; Ye, Y. Efficient Physiological and Nutrient Use Efficiency Responses of Maize Leaves to Drought Stress under Different Field Nitrogen Conditions. Agronomy 2020, 10, 523. [Google Scholar] [CrossRef]
- Martínez, D.E.; Guiamet, J.J. Distortion of the SPAD 502 chlorophyll meter readings by changes in irradiance and leaf water status. Agronomie 2004, 24, 41–46. [Google Scholar] [CrossRef]
- Padilla, F.M.; de Souza, R.; Peña-Fleitas, M.T.; Gallardo, M.; Giménez, C.; Thompson, R.B. Different Responses of Various Chlorophyll Meters to Increasing Nitrogen Supply in Sweet Pepper. Front. Plant Sci. 2018, 9, 1752. [Google Scholar] [CrossRef]
- Illés, A.; Bojtor, C.; Széles, A.; Mousavi, S.M.N.; Tóth, B.; Nagy, J. Analyzing the Effect of Intensive and Low-Input Agrotechnical Support for the Physiological, Phenometric, and Yield Parameters of Different Maize Hybrids Using Multivariate Statistical Methods. Int. J. Agron. 2021, 2021, 6682573. [Google Scholar] [CrossRef]
- Kira, O.; Nguy-Robertson, A.L.; Arkebauer, T.J.; Linker, R.; Gitelson, A.A. Informative spectral bands for remote green LAI estimation in C3 and C4 crops. Agric. Meteorol. 2016, 218–219, 243–249. [Google Scholar] [CrossRef]
- Liang, H.Y.; Shen, P.F.; Kong, X.Z.; Liao, Y.C.; Liu, Y.; Wen, X.X. Optimal nitrogen practice in winter wheat-summer maize rotation affecting the fates of 15 N-labeled fertilizer. Agronomy 2020, 10, 521. [Google Scholar] [CrossRef]
- Oguntunde, P.G.; Fasinmirin, J.T.; Abiolu, O.A. Performance of the SunScan canopy analysis system in estimating leaf area index of maize. Agric. Eng. Int. CIGR J. 2012, 14, 1–7. [Google Scholar]
- Wilhelm, W.; Ruwe, K.; Schlemmer, M.R. Comparison of three leaf area index meters in a corn canopy. Crop Sci. 2000, 40, 1179–1183. [Google Scholar] [CrossRef]
- Li, J.; Feng, Y.; Mou, J.; Xu, G.; Luo, Q.; Luo, K.; Huang, S.; Shi, X.; Guan, Z.; Ye, Y.; et al. Construction and Application Effect of the Leaf Value Model Based on SPAD Value in Rice. Sci. Agric. Sin. 2017, 50, 4714–4724. [Google Scholar] [CrossRef]
- Zhang, S.; Zhao, G.; Lang, K.; Su, B.; Chen, X.; Xi, X.; Zhang, H. Integrated Satellite, Unmanned Aerial Vehicle (UAV) and Ground Inversion of the SPAD of Winter Wheat in the Reviving Stage. Sensors 2019, 19, 1485. [Google Scholar] [CrossRef]
- Croft, H.; Chen, J.M.; Luo, X.Z.; Bartlett, P.; Chen, B.; Staebler, R.M. Leaf chlorophyll content as a proxy for leaf photosynthetic capacity. Glob. Chang. Biol. 2017, 23, 3513–3524. [Google Scholar] [CrossRef]
- Xie, C.; Yang, C. A review on plant high-throughput phenotyping traits using UAV-based sensors. Comput. Electron. Agric. 2020, 178, 105731. [Google Scholar] [CrossRef]
- Tsouros, D.C.; Bibi, S.; Sarigiannidis, P.G. A Review on UAV-Based Applications for Precision Agriculture. Information 2019, 10, 349. [Google Scholar] [CrossRef]
- Hunt, E.R., Jr.; Hively, W.D.; Fujikawa, S.; Linden, D.; Daughtry, C.S.; McCarty, G. Acquisition of NIR-Green-Blue Digital Photographs from Unmanned Aircraft for Crop Monitoring. Remote Sens. 2010, 2, 290–305. [Google Scholar] [CrossRef]
- Bouguettaya, A.; Zarzour, H.; Kechida, A.; Taberkit, A.M. Deep learning techniques to classify agricultural crops through UAV imagery: A review. Neural Comput. Appl. 2022, 34, 9511–9536. [Google Scholar] [CrossRef]



| Maize Phenophase | R2 (SPAD) | R2 (LAI) | R2 (SPAD + LAI) | RMSE (SPAD + LAI) |
|---|---|---|---|---|
| V8 | n.s. | 0.77 | 0.267 | 2.469 |
| V10 | 0.352 | 0.396 | 0.475 | 2.089 |
| V12 | 0.500 | 0.226 | 0.515 | 2.008 |
| Vn | 0.545 | 0.611 | 0.734 | 1.486 |
| VT | 0.624 | 0.627 | 0.762 | 1.408 |
| R1 | 0.655 | 0.575 | 0.704 | 1.569 |
| R3 | 0.716 | 0.521 | 0.733 | 1.489 |
| R6 | 0.221 | 0.526 | 0.562 | 1.908 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Széles, A.; Horváth, É.; Simon, K.; Zagyi, P.; Huzsvai, L. Maize Production under Drought Stress: Nutrient Supply, Yield Prediction. Plants 2023, 12, 3301. https://doi.org/10.3390/plants12183301
Széles A, Horváth É, Simon K, Zagyi P, Huzsvai L. Maize Production under Drought Stress: Nutrient Supply, Yield Prediction. Plants. 2023; 12(18):3301. https://doi.org/10.3390/plants12183301
Chicago/Turabian StyleSzéles, Adrienn, Éva Horváth, Károly Simon, Péter Zagyi, and László Huzsvai. 2023. "Maize Production under Drought Stress: Nutrient Supply, Yield Prediction" Plants 12, no. 18: 3301. https://doi.org/10.3390/plants12183301
APA StyleSzéles, A., Horváth, É., Simon, K., Zagyi, P., & Huzsvai, L. (2023). Maize Production under Drought Stress: Nutrient Supply, Yield Prediction. Plants, 12(18), 3301. https://doi.org/10.3390/plants12183301










