Do Genetic Drift and Gene Flow Affect the Geographic Distribution of Female Plants in Gynodioecious Lobelia siphilitica?
Abstract
1. Introduction
2. Results
2.1. Data Characteristics
2.1.1. Demography
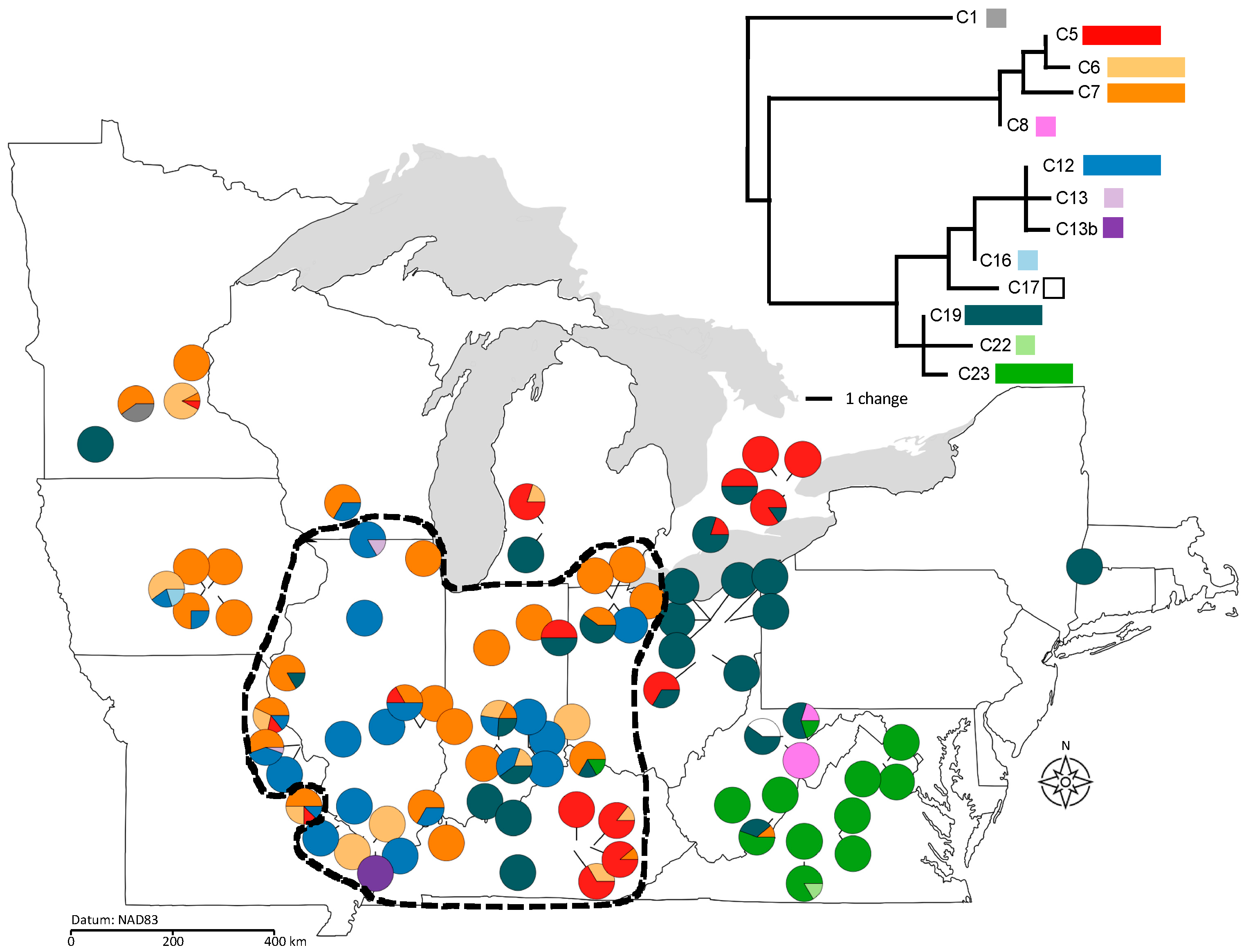
2.1.2. Plastid Sequence Data
2.1.3. Nuclear Microsatellite Diversity
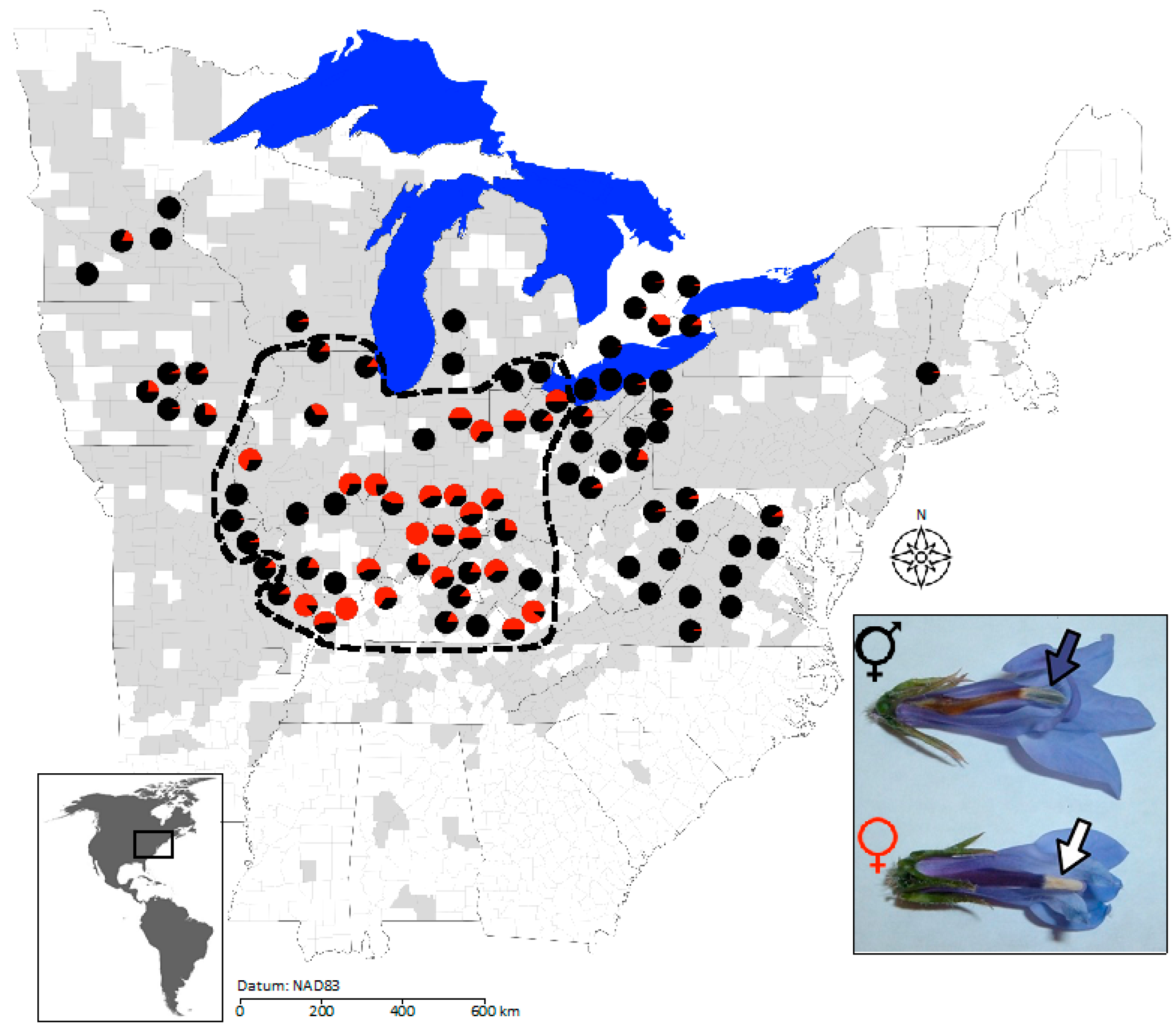
2.2. Geographic Structure in Population Sex Ratios, Population Size, and Neutral Genetic Diversity
2.3. Population Differentiation and Gene Flow
2.4. Population Genetic Structure in Relation to Variation in Sex Ratios, Population Size, and Geography
3. Discussion
4. Materials and Methods
4.1. Study System
4.2. Range-Wide Sampling of Population Size and Sex Ratio
4.3. Tissue Sampling and DNA Extraction
4.4. Plastid Sequencing and Microsatellite Genotyping
4.5. Statistical Analyses
4.5.1. Geography of Population Size and Sex Ratio in L. siphilitica
4.5.2. Relationships between Sex Ratio and Genetic Diversity
4.5.3. Estimating Population Differentiation, Gene Flow, and Genetic Drift across the Species Range
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barrett, S.C.H. Understanding plant reproductive diversity. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 99–109. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.C.H.; Dorken, M.E.; Case, A.L. A geographical context for the evolution of plant reproductive systems. In Integrating Ecological and Evolutionary Processes in a Spatial Context; Silvertown, J., Antonovics, J., Eds.; Blackwell: Oxford, UK, 2001; pp. 341–364. [Google Scholar]
- Dufaÿ, M.; Billard, E. How much better are females? The occurrence of female advantage, its proximal causes and its variation within and among gynodioecious species. Ann. Bot. 2012, 109, 505–519. [Google Scholar] [CrossRef]
- Sinclair, J.P.; Emlen, J.; Freeman, D.C. Biased sex ratios in plants: Theory and trends. Bot. Rev. 2012, 78, 63–86. [Google Scholar] [CrossRef]
- Varga, S.; Soulsbury, C.D. Environmental stressors affect sex ratios in sexually dimorphic plant sexual systems. Plant Biol. 2020, 22, 890–898. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.C.H. The evolution of plant reproductive systems: How often are transitions irreversible? Proc. R. Soc. B 2013, 280, 20130913. [Google Scholar] [CrossRef] [PubMed]
- Caruso, C.M.; Eisen, K.; Case, A.L. An angiosperm-wide analysis of the correlates of gynodioecy. Int. J. Plant Sci. 2016, 177, 115–121. [Google Scholar] [CrossRef]
- Budar, F.; Touzet, P.; De Paepe, R. The nucleo-mitochondrial conflict in cytoplasmic male sterilities revisited. Genetica 2003, 117, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Schnable, P.S.; Wise, R.P. The molecular basis of cytoplasmic male sterility and fertility restoration. Trends Plant Sci. 1998, 3, 175–180. [Google Scholar] [CrossRef]
- Delph, L.F.; Touzet, P.; Bailey, M.F. Merging theory and mechanism in studies of gynodioecy. Trends Ecol. Evol. 2007, 22, 17–24. [Google Scholar] [CrossRef]
- Dudle, D.A.; Mutikainen, P.; Delph, L.F. Genetics of sex determination in the gynodioecious species Lobelia siphilitica: Evidence from two populations. Heredity 2001, 86, 265–276. [Google Scholar] [CrossRef]
- Hanson, M.R. Plant mitochondrial mutations and male sterility. Annu. Rev. Genet. 1991, 25, 461–486. [Google Scholar] [CrossRef] [PubMed]
- Touzet, P.; Budar, F. Unveiling the molecular arms race between two conflicting genomes in cytoplasmic male sterility? Trends Plant Sci. 2004, 9, 568–570. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.; Caruso, C.M.; Case, A.L. Beyond balancing selection: Frequent mitochondrial recombination contributes to high-female frequencies in gynodioecious Lobelia siphilitica (Campanulaceae). New Phytol. 2019, 224, 1381–1393. [Google Scholar] [CrossRef] [PubMed]
- Caruso, C.M.; Case, A.L.; Bailey, M.F. The evolutionary ecology of cytonuclear interactions in angiosperms. Trends Plant Sci. 2012, 17, 638–643. [Google Scholar] [CrossRef] [PubMed]
- Dufaÿ, M.; Cuguen, J.; Arnaud, J.F.; Touzet, P. Sex ratio variation among gynodioecious populations of sea beet: Can it be explained by negative frequency-dependent selection? Evolution 2009, 63, 1483–1497. [Google Scholar] [CrossRef]
- McCauley, D.E.; Bailey, M.F. Recent advances in the study of gynodioecy: The interface of theory and empiricism. Ann. Bot. 2009, 104, 611–620. [Google Scholar] [CrossRef]
- Case, A.L.; Finseth, F.R.; Barr, C.M.; Fishman, L. Selfish evolution of cytonuclear hybrid incompatibility in Mimulus. Proc. R. Soc. B 2016, 283, 20161493. [Google Scholar] [CrossRef]
- Byers, D.L.; Warsaw, A.; Meagher, T.R. Consequences of prairie fragmentation on the progeny sex ratio of a gynodioecious species, Lobelia spicata (Campanulaceae). Heredity 2005, 95, 69–75. [Google Scholar] [CrossRef][Green Version]
- Manicacci, D.; Couvet, D.; Belhassen, E.; Gouyon, P.-H.; Atlan, A. Founder effects and sex ratio in the gynodioecious Thymus vulgaris L. Mol. Ecol. 1996, 5, 63–72. [Google Scholar] [CrossRef]
- Murayama, K.; Yahara, T.; Terachi, T. Variation of female frequency and cytoplasmic male-sterility gene frequency among natural gynodioecious populations of wild radish (Raphanus sativus L.). Mol. Ecol. 2004, 13, 2459–2464. [Google Scholar] [CrossRef]
- Nilsson, E.; Ågren, J. Population size, female fecundity, and sex ratio variation in gynodioecious Plantago maritima. J. Evol. Biol. 2006, 19, 825–833. [Google Scholar] [CrossRef] [PubMed]
- Bailey, M.F.; Delph, L.F. A field guide to models of sex-ratio evolution in gynodioecious species. Oikos 2007, 116, 1609–1617. [Google Scholar] [CrossRef]
- Bailey, M.F.; McCauley, D.E. Offspring sex ratio under inbreeding and outbreeding in a gynodioecious plant. Evolution 2005, 59, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Barr, C.M. Hybridization and regional sex ratios in Nemophila menziesii. J. Evol. Biol. 2004, 17, 786–794. [Google Scholar] [CrossRef]
- Castilla, A.R.; Alonso, C.; Herrera, C.M. Genetic structure of the shrub Daphne laureola across the Baetic Ranges, a Mediterranean glacial refugium and biodiversity hotspot. Plant Biol. 2012, 14, 515–524. [Google Scholar] [CrossRef]
- Eckert, C.G.; Samis, K.E.; Lougheed, S.C. Genetic variation across species’ geographical ranges: The central-marginal hypothesis and beyond. Mol. Ecol. 2008, 17, 1170–1188. [Google Scholar] [CrossRef]
- Sagarin, R.D.; Gaines, S.D. The ‘abundant centre’ distribution: To what extent is it a biogeographical rule? Ecol. Lett. 2002, 5, 137–147. [Google Scholar] [CrossRef]
- Vucetich, J.A.; Waite, T.A. Spatial patterns of demography and genetic processes across the species’ range: Null hypotheses for landscape conservation genetics. Conserv. Genet. 2003, 4, 639–645. [Google Scholar] [CrossRef]
- Kartesz, J.T. The Biota of North America Program (BONAP). Available online: http://bonap.net/napa (accessed on 1 June 2020).
- Caruso, C.M.; Case, A.L. Sex ratio variation in gynodioecious Lobelia siphilitica: Effects of population size and geographic location. J. Evol. Biol. 2007, 20, 1396–1405. [Google Scholar] [CrossRef]
- Omernik, J.M. Ecoregions of the conterminous United States. Ann. Assoc. Am. Geogr. 1987, 77, 118–125. [Google Scholar] [CrossRef]
- Delph, L.F.; Kelly, J.K. On the importance of balancing selection in plants. New Phytol. 2014, 201, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Arnaud, J.F.; Fenart, S.; Gode, C.; Deledicque, S.; Touzet, P.; Cuguen, J. Fine-scale geographical structure of genetic diversity in inland wild beet populations. Mol. Ecol. 2009, 18, 3201–3215. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, E.; López, S. Sex ratio and sex-specific latitudinal variation in floral characteristics of gynodioecious Kallstroemia grandiflora (Zygophyllaceae) in Mexico. Biotropica 2011, 43, 317–323. [Google Scholar] [CrossRef]
- Ruffatto, D.M.; Zaya, D.N.; Molano-Flores, B. Reproductive success of the gynodioecious Lobelia spicata Lam. (Campanulaceae): Female frequency, population demographics, and latitudinal patterns. Int. J. Plant Sci. 2015, 176, 120–130. [Google Scholar] [CrossRef]
- Cuguen, J.; Wattier, R.; Saumitou-Laprade, P.; Forcioli, D.; Mörchen, M.; Van Dijk, H.; Vernet, P. Gynodioecy and mitochondrial DNA polymorphism in natural populations of Beta vulgaris ssp maritima. Genet. Sel. Evol. 1994, 26, S87. [Google Scholar] [CrossRef]
- Soltis, D.E.; Morris, A.B.; McLachlan, J.S.; Manos, P.S.; Soltis, P.S. Comparative phylogeography of unglaciated eastern North America. Mol. Ecol. 2006, 15, 4261–4293. [Google Scholar] [CrossRef] [PubMed]
- Barnard-Kubow, K.B.; Debban, C.L.; Galloway, L.F. Multiple glacial refugia lead to genetic structuring and the potential for reproductive isolation in a herbaceous plant. Am. J. Bot. 2015, 102, 1842–1853. [Google Scholar] [CrossRef]
- Koski, M.H.; Layman, N.C.; Prior, C.J.; Busch, J.W.; Galloway, L.F. Selfing ability and drift load evolve with range expansion. Evol. Lett. 2019, 3, 500–512. [Google Scholar] [CrossRef]
- Bailey, M.F. A cost of restoration of male fertility in a gynodioecious species, Lobelia siphilitica. Evolution 2002, 56, 2178–2186. [Google Scholar] [CrossRef]
- Caruso, C.M.; Case, A.L. Testing models of sex ratio evolution in a gynodioecious plant: Female frequency covaries with the cost of male fertility restoration. Evolution 2013, 67, 561–566. [Google Scholar] [CrossRef]
- Case, A.L.; Caruso, C.M. A novel approach to estimating the cost of male fertility restoration in gynodioecious plants. New Phytol. 2010, 186, 549–557. [Google Scholar] [CrossRef] [PubMed]
- McVaugh, R. Studies in the taxonomy and distribution of the eastern North American species of Lobelia. Rhodora 1936, 38, 241–263. [Google Scholar]
- Caruso, C.M.; Maherali, H.; Jackson, R.B. Gender-specific floral and physiological traits: Implications for the maintenance of females in gynodioecious Lobelia siphilitica. Oecologia 2003, 135, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Doyle, J.J.; Doyle, J.L. Isolation of plant DNA from fresh tissue. Focus 1990, 12, 13–15. [Google Scholar]
- Codes, G. Sequencher 4.10.1—Build 5829; Gene Codes Corportation: Ann Arbor, MI, USA, 2010. [Google Scholar]
- Swofford, D.L. PAUP* Phylogenetic Analysis Using Parsimony (*and Other Methods); Sinauer Associates: Sunderland, MA, USA, 2002. [Google Scholar]
- Proell, J.M. Population Sex Ratio and Size Affect Pollination, Reproduction Success, and Seed Germination in Gynodioecious Lobelia siphilitica: Evidence Using Experimental Populations and Microsatellite Genotypes. Master’s Thesis, Kent State University, Kent, OH, USA, 2009. [Google Scholar]
- Becker, R.A.; Wilks, A.R.; Brownrigg, R.; Minka, T.; Deckmyn, A. Draw Geographical Maps; 3.4.0; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. Available online: https://www.R-project.org/ (accessed on 1 July 2020).
- Lemon, J. Plotrix: A package in the red light district of R. R News 2006, 6, 8–12. [Google Scholar]
- Paradis, E.; Schliep, K. ape 5.0: An environment for modern phylogenetics and evolutionary analyses in R. Bioinformatics 2019, 35, 526–528. [Google Scholar] [CrossRef]
- SAS. JMP Statistics and Graphics Guide; SAS Institute: Cary, NC, USA, 2009. [Google Scholar]
- Nei, M.; Tajima, F. DNA polymorphism detectable by restriction endonucleases. Genetics 1981, 97, 145–163. [Google Scholar] [CrossRef] [PubMed]
- Peakall, R.; Smouse, P.E. GENALEX 6: Genetic analysis in Excel. Population genetic software for teaching and research. Mol. Ecol. Notes 2006, 6, 288–295. [Google Scholar] [CrossRef]
- Excoffier, L.; Lischer, H.E. Arlequin suite ver 3.5: A new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef]
- Slatkin, M. A measure of population subdivision based on microsatellite allele frequencies. Genetics 1995, 139, 457–462. [Google Scholar] [CrossRef]
- Chapuis, M.P.; Estoup, A. Microsatellite null alleles and estimation of population differentiation. Mol. Biol. Evol. 2007, 24, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Hudson, R.R.; Slatkin, M.; Maddison, W.P. Estimation of levels of gene flow from DNA sequence data. Genetics 1992, 132, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Vavrek, M.J. Fossil: Palaeoecological and palaeogeographical analysis tools. Palaeontol. Electron. 2011, 14, 16. [Google Scholar]
| AMOVA Group | # of Groups | Among Groups | Among Populations within Groups | Within Populations |
|---|---|---|---|---|
| Plastid haplogroup (n = 83) | ||||
| Female frequency | 4 | 4.2 | 69.8 *** | 26.0 *** |
| Population size | 2 | 0 | 74.4 *** | 26.4 *** |
| Central vs. peripheral | 2 | 10.2 * | 64.8 *** | 24.9 *** |
| Nuclear microsatellites (n = 90) | ||||
| Female frequency | 4 | 1.0 * | 14.4 *** | 84.5 *** |
| Population size | 2 | 0 | 15.3 *** | 84.7 *** |
| Central vs. peripheral | 2 | 2.1 * | 14.0 *** | 83.9 *** |
| Evidence of Spatial Structure | Variation with Population Size | Variation with Female Frequency | |
|---|---|---|---|
| Female frequency | Very strong; 7x higher in south–central populations | None | – |
| Population size | Slightly smaller at lower latitudes | – | None |
| Plastid haplo-group diversity | Individual haplogroups structured longitudinally | None | None |
| Nuclear microsatellite diversity | Weak to none | Slightly more alleles in larger populations | Slightly fewer alleles in populations with no female plants |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Appiah-Madson, H.J.; Knox, E.B.; Caruso, C.M.; Case, A.L. Do Genetic Drift and Gene Flow Affect the Geographic Distribution of Female Plants in Gynodioecious Lobelia siphilitica? Plants 2022, 11, 825. https://doi.org/10.3390/plants11060825
Appiah-Madson HJ, Knox EB, Caruso CM, Case AL. Do Genetic Drift and Gene Flow Affect the Geographic Distribution of Female Plants in Gynodioecious Lobelia siphilitica? Plants. 2022; 11(6):825. https://doi.org/10.3390/plants11060825
Chicago/Turabian StyleAppiah-Madson, Hannah J., Eric B. Knox, Christina M. Caruso, and Andrea L. Case. 2022. "Do Genetic Drift and Gene Flow Affect the Geographic Distribution of Female Plants in Gynodioecious Lobelia siphilitica?" Plants 11, no. 6: 825. https://doi.org/10.3390/plants11060825
APA StyleAppiah-Madson, H. J., Knox, E. B., Caruso, C. M., & Case, A. L. (2022). Do Genetic Drift and Gene Flow Affect the Geographic Distribution of Female Plants in Gynodioecious Lobelia siphilitica? Plants, 11(6), 825. https://doi.org/10.3390/plants11060825






