Patterns of Traditional and Modern Uses of Wild Edible Native Plants of Chile: Challenges and Future Perspectives
Abstract
1. Introduction
2. Results
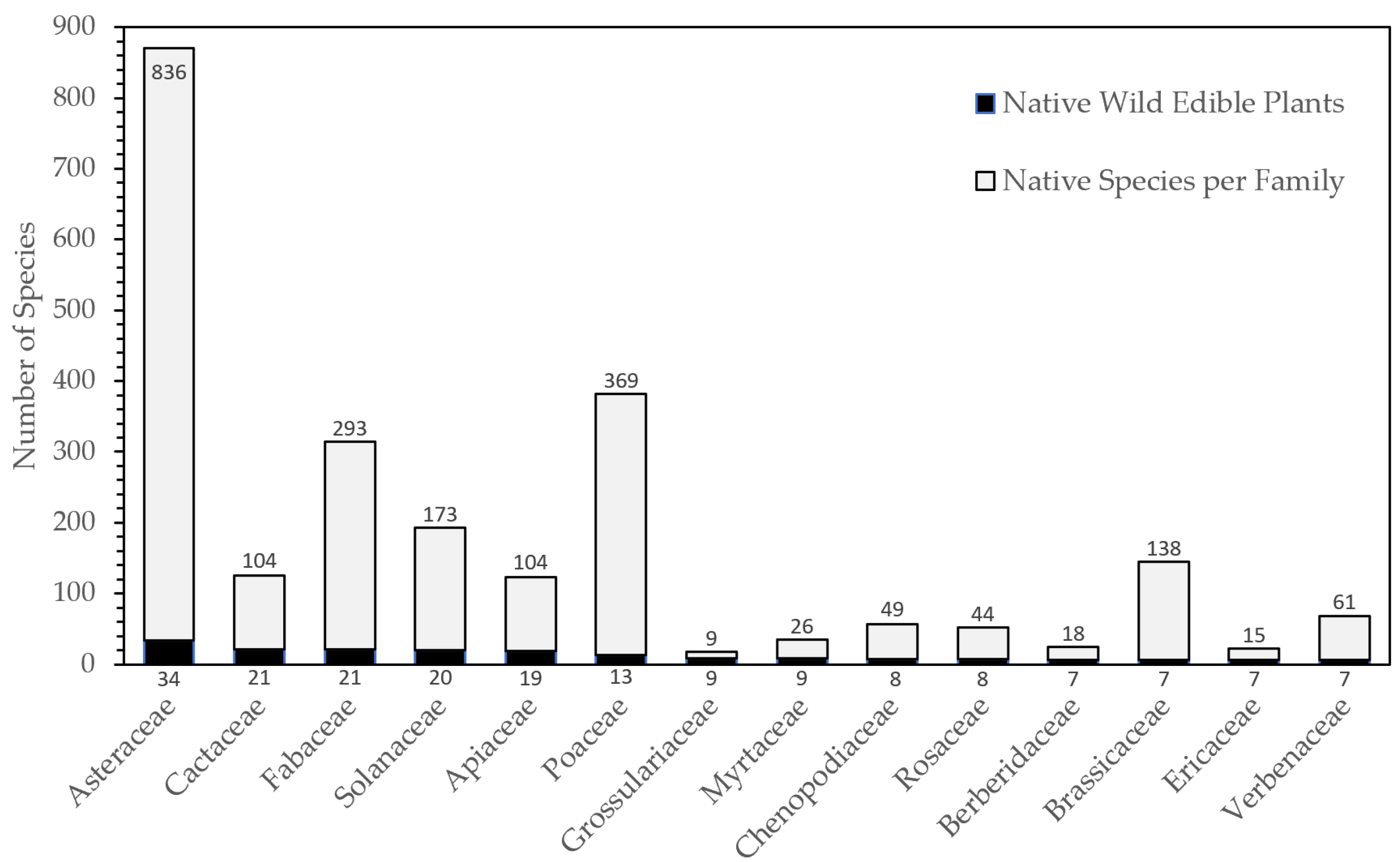
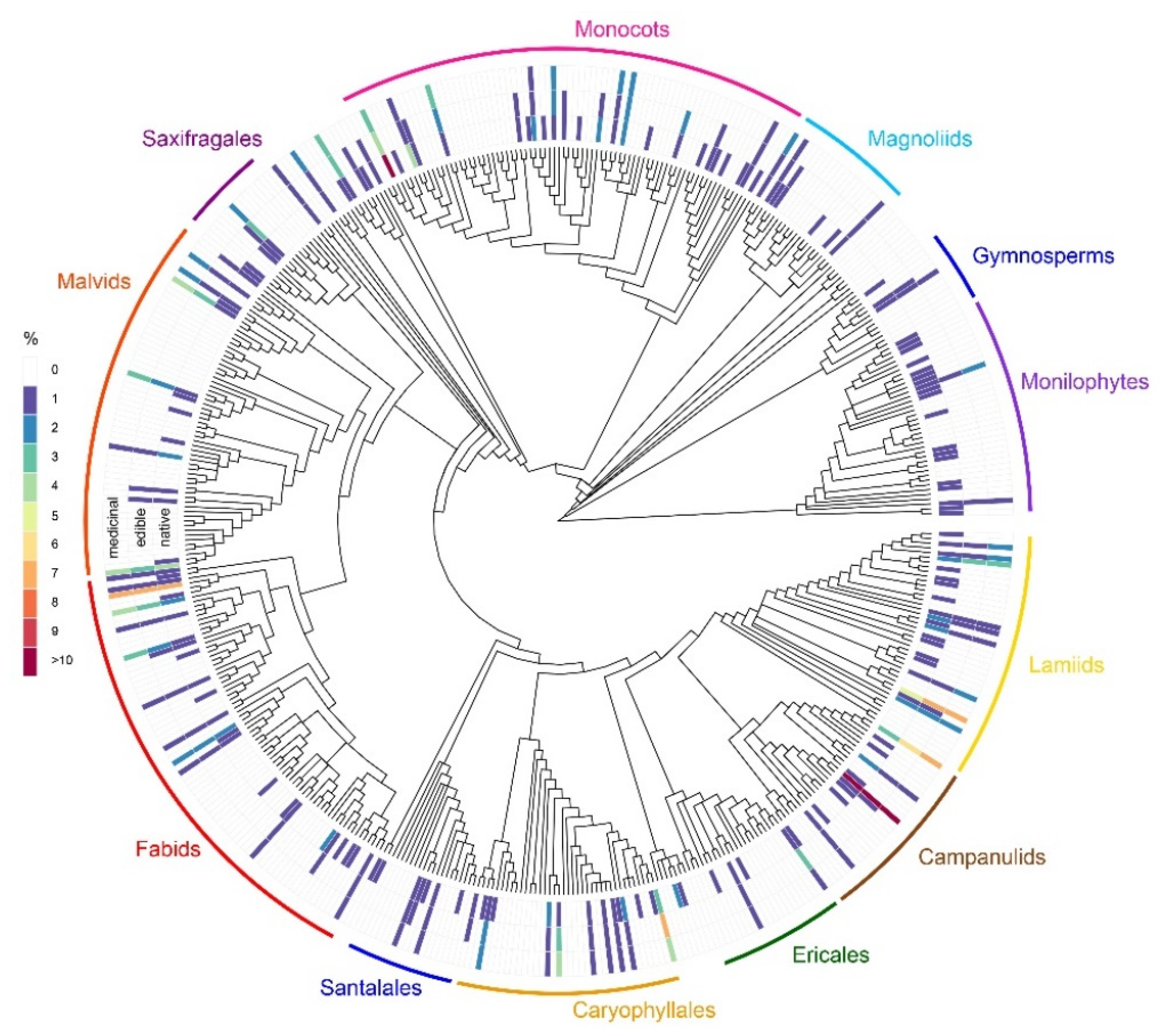
2.1. Taxonomic Diversity and Life Form
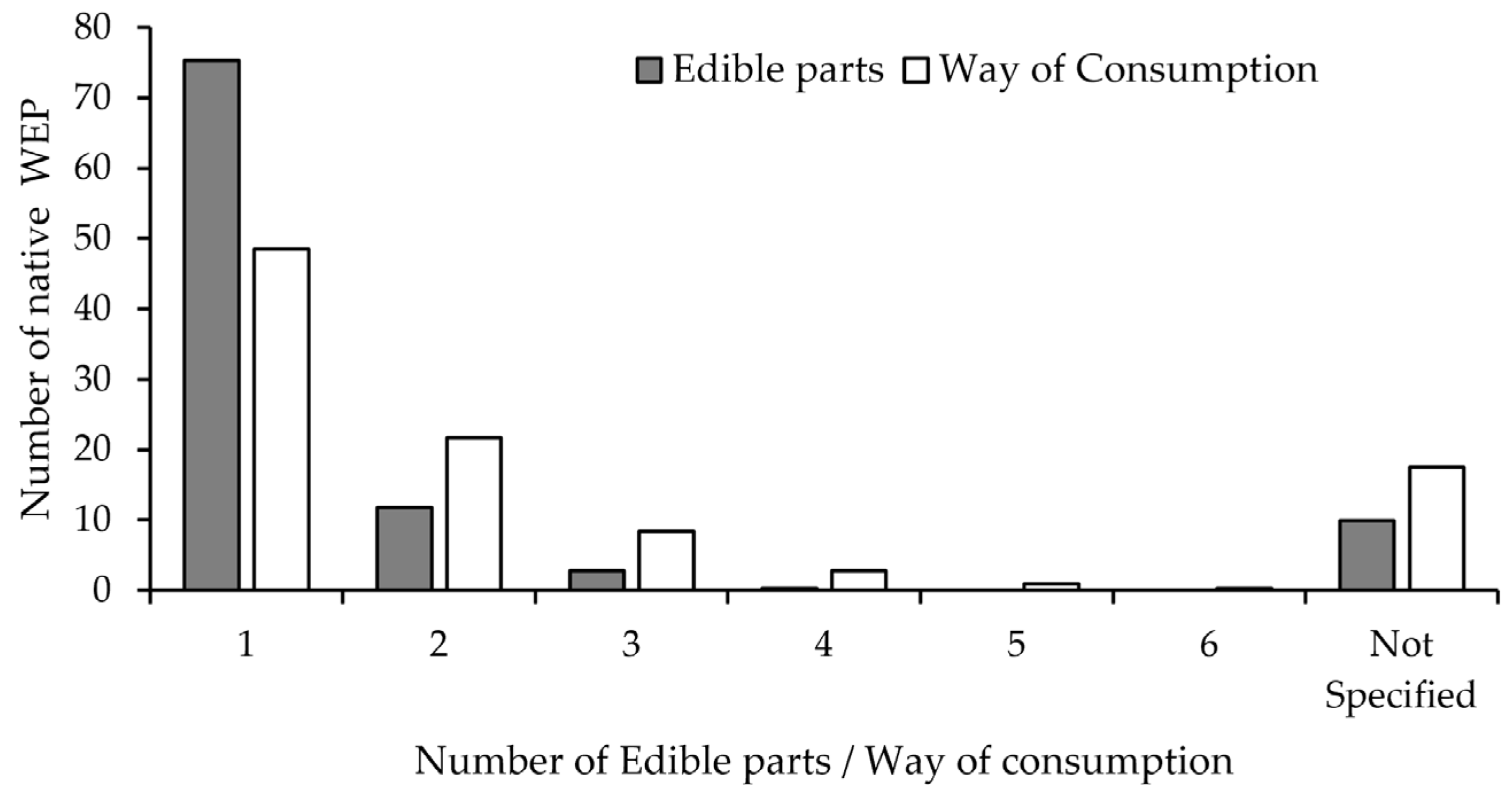
2.2. Consumption and Preparation Methods
3. Discussion
4. Material and Methods
4.1. Study Area
4.2. Literature Search
4.3. Taxonomic Diversity, Origin and Life Form
4.4. Consumption and Preparation Methods of WEP
5. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of the World’s Biodiversity for Food and Agriculture; Bélanger, J., Pilling, D., Eds.; FAO Commission on Genetic Resources for Food and Agriculture Assessments: Rome, Italy, 2019; Available online: http://www.fao.org/3/CA3129EN/CA3129EN.pdf (accessed on 12 February 2022).
- Gee, E.; Borelli, T.; de Oliveira Beltrame, D.M.; Neves Soares Oliveira, C.; Coradin, L.; Wasike, V.; Manjella, A.; Samarasinghe, G.; Güner, B.; Tan, A.; et al. The ABC of Mainstreaming Biodiversity for Food and Nutrition: Concepts, Theory and Practice. In Biodiversity, Food and Nutrition: A New Agenda for Sustainable Food Systems, 1st ed.; Issues in Agricultural Biodiversity; Hunter, D., Borelli, T., Gee, E., Eds.; Routledge: London, UK, 2020; pp. 85–186. [Google Scholar]
- CBD. Convention on Biological Diversity. Secretariat of the Convention on Biological Diversity, United Nations Environmental Programme. 1992. Available online: https://www.cbd.int/doc/legal/cbd-en.pdf (accessed on 12 February 2022).
- CBD. Global Strategy for Plant Conservation: 2011–2020; Botanic Gardens Conservation International: Richmond, UK, 2012. [Google Scholar]
- FAO. International Treaty on Plant Genetic Resources for Food and Agriculture. A Global Treaty for Food Security and Sustainable Agriculture; Food and Agriculture Organization of the United Nations: Rome, Italy, 2011. [Google Scholar]
- United Nations. Sustainable Development Goals. 17 Goals to Transform Our World. Goal Two: Zero Hunger. Available online: https://www.un.org/sustainabledevelopment/hunger/ (accessed on 13 February 2022).
- Ulian, T.; Diazgranados, M.; Pironon, S.; Padulosi, S.; Liu, U.; Davies, L.; Howes, M.-J.R.; Borrell, J.S.; Ondo, I.; Pérez-Escobar, O.A.; et al. Unlocking plant resources to support food security and promote sustainable agriculture. Plants People Planet 2020, 2, 421–445. [Google Scholar] [CrossRef]
- Borelli, T.; Hunter, D.; Powell, B.; Ulian, T.; Mattana, E.; Termote, C.; Pawera, L.; Beltrame, D.; Penafiel, D.; Tan, A.; et al. Born to Eat Wild: An Integrated Conservation Approach to Secure Wild Food Plants for Food Security and Nutrition. Plants 2020, 9, 1299. [Google Scholar] [CrossRef] [PubMed]
- Turner, N.J.; Luczaj, L.J.; Migliorini, P.; Pieroni, A.; Dreon, A.L.; Sacchetti, L.E.; Paoletti, M.N. Edible and tender wild plants, traditional ecological Knowledge and Agroecology. Crit. Rev. Plant Sci. 2011, 30, 198–225. [Google Scholar] [CrossRef]
- Carvalho, A.M.; Barata, A.M. The Consumption of Wild Edible Plants. In Wild Plants, Mushrooms and Nuts: Functional Food Properties and Applications, 1st ed.; Ferreira, I.C.F.R., Morales, P., Barros, L., Eds.; John Wiley & Sons: Chichester, UK, 2017; pp. 159–198. [Google Scholar]
- Ladio, A.; Lozada, M. Patterns of use and knowledge of wild edible plants in distinct ecological environments: A case study of a Mapuche community from NW Patagonia. Biodivers. Conserv. 2004, 13, 1153–1173. [Google Scholar] [CrossRef]
- Tardío, J.; de Santayana, M.P.; Morales, R. Ethnobotanical review of wild edible plants in Spain. Bot. J. Linn. Soc. 2006, 152, 27–71. [Google Scholar] [CrossRef]
- Afolayan, A.J.; Jimoh, F.O. Nutritional quality of some wild leafy vegetables in South Africa. Int. J. Food Sci. Nutr. 2009, 60, 2913–2926. [Google Scholar] [CrossRef]
- Bharucha, Z.; Pretty, J. The roles and values of wild foods in agricultural systems. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2913–2926. [Google Scholar] [CrossRef]
- Lulekal, E.; Asfaw, Z.; Kelbessa, E.; van Damme, P. Wild edible plants in Ethiopia: A review on their potential to combat food insecurity. Afr. Focus 2011, 24, 71–121. [Google Scholar] [CrossRef]
- Pereira, C.; Barros, L.; Carvalho, A.M.; Ferreira, I.C.F.R. Nutritional composition and bioactive properties of commonly consumed wild greens: Potential sources for new trends in modern diets. Food Res. Int. 2011, 44, 2634–2640. [Google Scholar] [CrossRef]
- Łuczaj, L.; Pieroni, A.; Tardio, J.; Pardo de Santayana, M.; Soukand, R.; Svanberg, I.; Kalle, R. Wild food plant use in 21st century Europe: The disappearance of old traditions and the search new cuisines involving wild edibles. Acta Soc. Bot. Pol. 2012, 81, 359–370. [Google Scholar] [CrossRef]
- Łuczaj, L.; Pieroni, A. Nutritional Ethnobotany in Europe: From Emergency Foods to Healthy Folk Cuisines and Contemporary Foraging Trends. In Mediterranean Wild Edible Plants. Ethnobotany and Food Composition Tables; de Cortes Sánchez-Mata, M., Tardío, J., Eds.; Springer: Heidelberg, Germany, 2016; pp. 33–56. [Google Scholar]
- Cámara, M.; Fernández-Ruiz, V.; Ruiz-Rodríguez, B.M. Wild edible plants as sources of carotenoids, fibre, phenolics and other non-nutrient bioactive compounds. In Mediterranean Wild Edible Plants. Ethnobotany and Food Composition Tables; de Cortes Sánchez-Mata, M., Tardío, J., Eds.; Springer: Heidelberg, Germany, 2016; pp. 171–208. [Google Scholar]
- Datta, S.; Sinha, B.K.; Bhattacharjee, S.; Seal, T. Nutritional composition, mineral content, antioxidant activity and quantitative estimation of water-soluble vitamins and phenolics by RP-HPLC in some lesser used wild edible plants. Heliyon 2019, 5, e01431. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Omega-3 fatty acids and antioxidants in edible wild plants. Biol. Res. 2004, 37, 263–277. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.C.; Giusti, M.M. (Eds.) Anthocyanins in Health and Disease; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Bacchetta, L.; Visioli, F.; Cappelli, G.; Caruso, E.; Martin, G.; Nemeth, E.; Bacchetta, G.; Bedini, G.; Wezel, A.; van Asseldonk, T.; et al. A manifesto for the valorisation of wild plants. J. Ethnopharmacol. 2016, 191, 180–187. [Google Scholar] [CrossRef]
- García-Herrera, P.; de Cortes Sánchez-Mata, M. The Contribution of Wild Plants to Dietary Intakes of Micronutrients (II): Mineral Elements. In Mediterranean Wild Edible Plants. Ethnobotany and Food Composition Tables; de Cortes Sánchez-Mata, M., Tardío, J., Eds.; Springer: Heidelberg, Germany, 2016; pp. 141–171. [Google Scholar]
- Heinrich, M.; Kerrouche, S.; Bharij, K.S. Recent Advances in Research on Wild Food Plants and Their Biological-Pharmacological Activity. In Mediterranean Wild Edible Plants. Ethnobotany and Food Composition Tables; de Cortes Sánchez-Mata, M., Tardío, J., Eds.; Springer: Heidelberg, Germany, 2016; pp. 253–272. [Google Scholar]
- Marrelli, M.; Statti, G.; Conforti, F. A Review of Biologically Active Natural Products from Mediterranean Wild Edible Plants: Benefits in the Treatment of Obesity and Its Related Disorders. Molecules 2020, 25, 649. [Google Scholar] [CrossRef]
- Mateos-Maces, L.; Chávez-Servia, J.L.; Vera-Guzmán, A.M.; Aquino-Bolaños, E.N.; Alba-Jiménez, J.E.; Villagómez-González, B.B. Edible Leafy Plants from Mexico as Sources of Antioxidant Compounds, and Their Nutritional, Nutraceutical and Antimicrobial Potential: A Review. Antioxidants 2020, 9, 541. [Google Scholar] [CrossRef] [PubMed]
- Falguera, V.; Aliguer, N.; Falguera, M. An integrated approach to current trends in food consumption: Moving toward functional and organic products? Food Control 2012, 26, 274–281. [Google Scholar] [CrossRef]
- Bibar, J. Crónica y Relación Copiosa y Verdadera de los Reinos de Chile; Publicado por el Fondo Histórico y Bibliográfico José Toribio Medina en 1966; Biblioteca Nacional de Chile: Santiago, Chile, 1558; Available online: http://www.memoriachilena.gob.cl/602/w3-article-8175.html (accessed on 12 February 2022).
- Rosales, D. Historia General del Reino de Chile. Flandes Indiano. Dedicada al Rey de España D. Carlos II; Publicada en 1877 por Don Benjamín Vicuña Mackenna; Imprenta de el Mercurio: Valparaíso, Chile, 1672; Available online: http://www.memoriachilena.gob.cl/602/w3-article-3356.html#documentos (accessed on 12 February 2022).
- Molina, J.I. Ensayo sobre la Historia natural de Chile; Traducción del Prof. Dr, Rodolfo Jaramillo de la Academia de las Ciencias del Instituto de Bolonia; Ediciones Maule: Santiago, Chile, 1810; Available online: http://www.memoriachilena.gob.cl/archivos2/pdfs/MC0002868.pdf (accessed on 12 February 2022).
- Gay, C. Historia Física y Política de Chile: Según Documentos Adquiridos en esta República durante Doce Años de Residencia en Ella; Flora Chilena, Botánica: Paris, France, 1852; pp. 1845–1852. Available online: http://www.memoriachilena.gob.cl/602/w3-article-94223.html (accessed on 12 February 2022).
- Gusinde, M. Plantas medicinales que los indios araucanos recomiendan. Anthropos 1936, 31, 850–873. [Google Scholar]
- Castro, V.; Villagrán, C.; Arroyo, M.T.K. Estudio Etnobotánico en la Precordillera y Altiplano de los Andes de Arica (18–19° S). In El Ambiente Natural y las Poblaciones Humanas de los Andes del Norte Grande de Chile (Arica, Lat. 18°28′ S); Veloso, A., Bustos, E., Eds.; ROSTLAC–UNESCO (Regional Office Science Tecnoligie Latinoamérica and Caribe): Montevideo, Uruguay, 1982; pp. 133–205. [Google Scholar]
- De Mösbach, E.W. Botánica Indígena de Chile; Museo Chileno de Arte Precolombino, Editorial Andrés Bello: Santiago, Chile, 1992. [Google Scholar]
- Villagrán, C.; Castro, V. Ciencia Indígena de los Andes del Norte de Chile; Editorial Universitaria: Santiago, Chile, 2004; p. 361. [Google Scholar]
- Diaz-Forestier, J.; León-Lobos, P.; Marticorena, A.; Celis-Diez, J.L.; Giovannini, P. Native useful plants of Chile: A review and use patterns. Econ. Bot. 2019, 73, 112–126. [Google Scholar] [CrossRef]
- Murillo, D.A. Memoria Sobre las Plantas Medicinales de Chile, y el Uso que de Ellas se Hace en el País; Imprenta del Ferrocarril: Santiago, Chile, 1861. [Google Scholar]
- Espinoza, E. Plantas Medicinales de Chile: Fragmento de la Cuarta Edición de la Geografía Descriptiva de la República; Imprenta y Encuadernación Barcelona: Santiago, Chile, 1897. [Google Scholar]
- Massardo, F.; Rozzi, R. Valoración de la Biodiversidad: Usos medicinales de la flora nativa chilena. Ambiente Desarro. 1996, 12, 76–81. [Google Scholar]
- Montenegro, G.; Timmermann, B.N. Chile, Nuestra Flora Útil. Especies de Uso Apicola, Alimentario en Medicina Folclórica, Artesanal y Ornamental; Abaco Press: Santiago, Chile, 2000. [Google Scholar]
- Reiche, K. Geografía Botánica de Chile. Cámara Chilena de la Construcción, Pontificia Universidad Católica de Chile, Dirección de Bibliotecas; Archivos y Museos: Santiago, Chile, 2013. [Google Scholar]
- Velázquez, P.; Montenegro, G. Chilean Endemic/Native Plant Resources as Functional and Superfoods. In Superfood and Functional Food—An Overview of Their Processing and Utilisation; Waisundara, V., Shiomi, N., Eds.; InTech Publisher: London, UK, 2017; pp. 131–154. [Google Scholar] [CrossRef]
- Ulloa-Inostroza, E.M.; Ulloa-Inostroza, E.G.; Alberdi, M.; Peña-Sanhueza, D.; González-Villagra, J.; Jaakola, L.; Reyes-Díaz, M. Native Chilean Fruits and the Effects of Their Functional Compounds on Human Health. In Superfood and Functional Food—An Overview of Their Processing and Utilisation; Waisundara, V., Shiomi, N., Eds.; InTech Publisher: London, UK, 2017; pp. 99–130. [Google Scholar]
- MMA. Procesos de Clasificación. Listado de Especies Clasificadas desde el 1° al 15° Proceso de Clasificación RCE (actualizado a julio de 2020). 2020. Available online: https://clasificacionespecies.mma.gob.cl/procesos-de-clasificacion/ (accessed on 12 February 2022).
- Cook, F.E.M. Economic Botany Data Collection Standard; Prepared for the International Working Group on Taxonomic Databases for Plant Sciences (TDWG); Royal Botanic Gardens, Kew: London, UK, 1995; ISBN 0947643710. [Google Scholar]
- Ortiz, J. Plantas Silvestres de Frutos Comestibles por el Hombre; Contribuciones Arqueológicas N°8; Museo de La Serena: La Serena, Chile, 1969. [Google Scholar]
- Muñoz, M.; Barrera, E.; Meza, I. El Uso Medicinal y Alimenticio de las Plantas Nativas y Naturalizadas en Chile; Publicaciones ocasionales del Museo Nacional de Historia Natural: Santiago, Chile, 1981; Volume 33, pp. 3–91. [Google Scholar]
- Latcham, R. La Agricultura Precolombiana en Chile y los Países Vecinos; Ediciones de la Universidad de Chile: Santiago, Chile, 1936; p. 336. [Google Scholar]
- Van den Eynden, V.; Cueva, E. Las Plantas en la Alimentación. In Enciclopedia de las Plantas Útiles del Ecuador; de la Torre, L., Navarrete, H., Muriel, P., Macía, M.J., Balslev, H., Eds.; Herbario QCA & Herbario AAU. Quito & Aarhus: Ecuador & Denmark, 2008; pp. 62–66. [Google Scholar]
- Ulian, T.; Pritchard, H.W.; Casas, A.; Mattana, E.; Liu, U.; Castillo-Lorenzo, E.; Way, M.; Dávila Aranda, P.; Lira, R. Neglected and Underutilized Species for Sustainable Food and Nutrition Security and Role of Conservation in Mexico. In Orphan Crops for Sustainable Food and Nutrition Security: Promoting Neglected and Underutilised Species (Issues in Agricultural Biodiversity); Padulosi, S., Israel, E.D., King, O., Hunter, D., Swaminathan, M.S., Eds.; Routledge: Oxon, UK, 2021. [Google Scholar]
- Ray, A.; Ray, R.; Reevidya, E.A. How Many Wild Edible Plants Do We Eat-Their Diversity, Use, and Implications for Sustainable Food System: An Exploratory Analysis in India. Front. Sustain. Food Syst. 2020, 4, 56. [Google Scholar] [CrossRef]
- Hadjichambis, A.C.; Paraskeva-Hadjichambi, D.; Della, T.; Giusti, M.E.; de Pasquale, C.; Lenzarini, C.; Censorii, E.; Gonzales-Tejero, M.R.; Sánchez-Rojas, C.P.; Ramiro-Gutiérrez, J.M.; et al. Wild and semi-domesticated food plant consumption in seven circum-Mediterranean areas. Int. J. Food Sci. Nutr. 2008, 59, 383–414. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, R.; Marticorena, C.; Alarcón, D.; Baeza, C.; Cavieres, L.; Finot, V.L.; Fuentes, N.; Kiessling, A.; Mihoc, M.; Pauchard, A.; et al. Catálogo de las plantas vasculares de Chile. Gayana Botánica 2018, 75, 1–430. [Google Scholar] [CrossRef]
- Scarpa, G.F. Wild food plants used by the indigenous peoples of the South American Gran Chaco: A general synopsis and intercultural comparison. J. Appl. Bot. Food Qual. 2009, 83, 90–101. [Google Scholar]
- Uprety, Y.; Poudel, R.C.; Shrestha, K.K.; Rajbhandary, S.; Tiwari, N.N.; Shrestha, U.B.; Asselin, H. Diversity of use and local knowledge of wild edible plant resources in Nepal. J. Ethnobiol. Ethnomedicine 2012, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Kuhnlein, H.V.; Turner, N.J. Traditional Plant Foods of Canadian Indigenous Peoples: Nutrition, Botany and Use; Gordon and Breach Publishers: Amsterdam, The Netherlands, 1991. [Google Scholar]
- Simkova, K.; Polesny, Z. Ethnobotanical review of wild edible plants used in the Czech Republic. J. Appl. Bot. Food Qual. 2015, 88, 49–67. [Google Scholar] [CrossRef]
- Nedelcheva, A. An ethnobotanical study of wild edible plants in Bulgaria. EurAsian J. BioSci. 2013, 7, 77–94. [Google Scholar] [CrossRef]
- Leonti, M.; Nebel, S.; Rivera, D.; Heinrich, M. Wild Gathered Food Plants in the European Mediterranean: A Comparative Analysis. Econ. Bot. 2006, 60, 130–142. [Google Scholar] [CrossRef]
- Zhou, L.G.; Ren, G.-X.; Chen, F. Chinese Edible Botanicals: Types, Efficacy and Safety. In Handbook of Food Science, Technology, and Engineering; Hui, Y.H., Ed.; Taylor & Francis Group: London, UK, 2005; Volume 2, pp. 98-1–98-28. [Google Scholar]
- Rapoport, E.H. Cuántas Especies Comemos y Cuántas Existen; Anales de la Academia Nacional de Agronomía y Veterinaria; Trabajo en Tomo LXI.; Universidad Nacional del Comahue, CRUB: Bariloche, Argentina, 2007; pp. 373–388. Available online: http://sedici.unlp.edu.ar/handle/10915/27759 (accessed on 12 February 2022).
- Tardío, J.; Pardo-de-Santayana, M. Ethnobotanical Analysis of Wild Fruits and Vegetables Traditionally Consumed in Spain. In Mediterranean Wild Edible Plants Ethnobotany and Food Composition Tables; Cortes Sánchez-Mata, M., Tardío, J., Eds.; Springer: Heidelberg, Germany, 2016; pp. 57–80. [Google Scholar]
- FAO. Quillaia Extracts: Type 1 and Type 2. 65th Joint FAO/WHO Expert Committee on Food Additives (JECFA) Meeting - Chemical and Technical Assessment (CTA) 2005; Food and Agriculture Organization (FAO): Rome, Italy, 2005. [Google Scholar]
- Mortensen, A.; Aguilar, F.; Crebelli, R.; di Domenico, A.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Lambré, C.; Leblanc, J.C.; et al. Re-evaluation of tara gum (E 417) as a food additive. EFSA J. 2017, 15, 4863. [Google Scholar] [CrossRef]
- Aceituno-Mata, L.; Tardío, J.; Pardo de Santayana, M. The Persistence of Flavour: Past and Present Use of Wild Food Plants in Sierra Norte de Madrid, Spain. Front. Sustain. Food Syst. 2021, 4, 610238. [Google Scholar] [CrossRef]
- Aswani, S.; Lemahieu, A.; Sauer, W.H.H. Global trends of local ecological knowledge and future implications. PloS ONE 2018, 13, e0195440. [Google Scholar] [CrossRef]
- Cruz, M.P.; Peroni, N.; Albuquerque, U.P. Knowledge, use and management of native wild edible plants from a seasonal dry forest (NE, Brazil). J. Ethnobiol. Ethnomed. 2013, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Ogle, B.M.; Tuyet, H.T.; Duyet, H.N.; Xuan Dung, N.N. Food, Feed or Medicine: The Multiple Functions of Edible Wild Plants in Vietnam. Econ. Bot. 2003, 57, 103–117. [Google Scholar] [CrossRef][Green Version]
- Shikov, A.N.; Tsitsilin, A.N.; Pozharitskaya, O.N.; Makarov, V.G.; Heinrich, M. Traditional and Current Food Use of Wild Plants Listed in the Russian Pharmacopoeia. Front. Pharmacol. 2017, 8, 841. [Google Scholar] [CrossRef] [PubMed]
- Benítez, G.; Molero-Mesa, J.; González-Tejero, M.R. Gathering an edible wild plant: Food or medicine? A case study on wild edibles and functional foods in Granada, Spain. Acta Soc. Bot. Pol. 2017, 86, 3550. [Google Scholar] [CrossRef]
- Molina, M.; Pardo-de-Santayana, M.; Tardío, J. Natural Production and Cultivation of Mediterranean Wild Edibles. In Mediterranean Wild Edible Plants Ethnobotany and Food Composition Tables; Cortes Sánchez-Mata, M., Tardío, J., Eds.; Springer: Heidelberg, Germany, 2006; pp. 81–107. [Google Scholar]
- Pinela, J.; Carvalho, A.M.; Ferreira, I.C.F.R. Wild edible plants: Nutritional and toxicological characteristics, retrieval strategies and importance for today’s society. Food Chem. Toxicol. 2017, 110, 165–188. [Google Scholar] [CrossRef]
- CORFO. Transforma Alimentos, Programa Estratégico Nacional; Gobierno de Chile: Santiago, Chile, 2018. [Google Scholar]
- CORFO. Diversificación y Sofisticación de la Industria de Alimentos en Chile. Transforma Alimento. 2015–2025; Gobierno de Chile: Santiago, Chile, 2017. [Google Scholar]
- MMA. Estrategia Nacional de Biodiversidad 2017–2030; Ministerio del Medio Ambiente, Gobierno de Chile: Santiago, Chile, 2017; Available online: https://biodiversidad.mma.gob.cl/ (accessed on 12 February 2022).
- Martínez-Crovetto, R. Estudios etonobotánicos IV. Nombres de plantas y su utilidad, según los indios onas de Tierra del Fuego. Etnobiología 1968, 3, 1–20. [Google Scholar]
- Ramirez, C.R. Ethnobotany and the Loss of Traditional Knowledge in the 21st Century. Ethnobot. Res. Appl. 2007, 5, 245–247. [Google Scholar] [CrossRef]
- Pandey, A.K.; Tripathi, Y.C. Ethnobotany and its relevance in contemporary research. J. Med. Plants Stud. 2017, 5, 123–129. [Google Scholar]
- Pardo de Santayana, M.; Macía, M.J. The benefits of traditional knowledge. Nature 2015, 518, 487–488. [Google Scholar] [CrossRef]
- Hunter, D.; Borelli, T.; Gee, E. (Eds.) Biodiversity, Food and Nutrition. A New Agenda for Sustainable Food Systems. Biodiversity International, 1st ed.; Routledge: Oxford, UK, 2020. [Google Scholar] [CrossRef]
- Mueso de Arte Precolombino, Santiago Chile. Pueblos Originarios de Chile. Available online: https://precolombino.cl/culturas-americanas/pueblos-originarios-de-chile/ (accessed on 2 February 2022).
- Gobierno de Chile. Guia de Antecedentes Territoriales y Culturales de los Pueblos Indígenas de Chile; Director General de Obras Públicas, Ministerio de Obras Públicas, Gobierno de Chile, ANDROS Impresores: Santiago, Chile, 2012. Available online: http://transparencia.dgop.cl/OtrosAntecedentes/docs/Guia_asuntos_indigenas.pdf (accessed on 2 February 2022).
- IPNI. International Plant Names Index. Facilitated by The Royal Botanic Gardens, Kew, Harvard University Herbaria & Libraries and Australian National Botanic Gardens. 2021. Available online: http://www.ipni.org/ (accessed on 13 February 2022).
- WCVP. World Checklist of Vascular Plants, Version 2.0; Royal Botanic Gardens: Richmond, UK, 2021; Available online: http://wcvp.science.kew.org/ (accessed on 13 February 2022).
- Wickham, H.; Wickham, M.H. Package ‘plyr’ [Online]. 2020. Available online: https://cran.r-project.org/web/packages/plyr/index.html (accessed on 13 February 2022).
- Bivand, R.; Keitt, T.; Rowlingson, B.; Pebesma, E.; Sumner, M.; Hijmans, R.; Rouault, E.; Bivand, M.R. Package ‘rgdal’. Bindings for the Geospatial Data Abstraction Library 2015. Available online: https://cran.r-project.org/web/packages/rgdal/index.html (accessed on 13 February 2022).
- Wickham, H.; François, R.; Henry, L.; Müller, K. dplyr: A Grammar of Data Manipulation [Online]. 2021. Available online: https://CRAN.R-project.org/package=dplyr (accessed on 13 February 2022).
- Højsgaard, S.; Halekoh, U.; Højsgaard, M.S.; Imports, M. Package ‘doBy’ [Online]. 2019. Available online: https://cran.r-project.org/web/packages/doBy/index.html (accessed on 13 February 2022).
- Gastauer, M.; Meira, J.A. Updated angiosperm family tree for analysing phylogenetic diversity and community structure. Acta Bot. Bras. 2017, 31, 191–198. [Google Scholar] [CrossRef]
- Yu, G.; Smith, D.K.; Zhu, H.; Guan, Y.; Lam, T.T.Y. ggtree: An r package for visualisation and annotation of phylogenetic trees with their covariates and other associated data. Methods Ecol. Evol. 2017, 8, 28–36. [Google Scholar] [CrossRef]
- Wickham, H.; Chang, W.; Wickham, M.H. Package ‘ggplot2’. Create Elegant Data Visualisations Using the Grammar of Graphics, 2016; Version, 2; Springer: New York, NY, USA, 2016; pp. 1–189. [Google Scholar]
- Wang, L.G.; Lam, T.T.Y.; Xu, S.; Dai, Z.; Zhou, L.; Feng, T.; Guo, P.; Dunn, C.W.; Jones, B.R.; Bradley, T. Treeio: An R package for phylogenetic tree input and output with richly annotated and associated data. Mol. Biol. Evol. 2020, 37, 599–603. [Google Scholar] [CrossRef] [PubMed]

| Life Form | Species Number | (%) |
|---|---|---|
| Tree | 36 | |
| Succulent tree | 3 | |
| Shrub/small tree | 8 | |
| Total trees | 47 | 14.2 |
| Shrub | 90 | |
| Parasitic shrub | 2 | |
| Succulent shrub | 18 | |
| Climbing shrub | 6 | |
| Total shrubs | 116 | 35.2 |
| Perennial herb | 120 | |
| Aquatic perennial herb | 2 | |
| Epiphytic/terrestrial perennial herb | 1 | |
| Parasitic perennial herb | 1 | |
| Climbing perennial herb | 5 | |
| Succulent perennial herb | 1 | |
| Total perennial herbs | 130 | 39.4 |
| Annual herb | 30 | |
| Annual/bi-annual herb | 7 | |
| Total herbs | 37 | 11.2 |
| Total | 330 |
| Plant Parts Used | Description | Number of Species | % | Number of Families | Main Families |
|---|---|---|---|---|---|
| Infrutescences | Fruits, entire immature fruits, entire mature fruits, deseeded fruits, fruit pulp, fruit juice, epicarp. | 118 | 35.8 | 33 | Cactaceae, Grossulariaceae, Myrtaceae, Berberidaceae, Ericaceae |
| Roots | Debarked ‘roots’, bulbs, corms, tubers, tubercles, nodules, aerial roots, pneumatophores, rhizomes | 71 | 21.5 | 25 | Apiaceae, Fabaceae, Dioscoreaceae, Alstromeriaceae, Solanaceae |
| Leaves | Cotyledons, young leaves, old leaves, fallen leaves, leaflets, stipules, leaf blades, leaf buds, petioles | 66 | 20.0 | 14 | Asteraceaae, Apiaceae, Chenopodiaceae, Lamiaceae, Oxalidaceae |
| Seeds | Arils, entire seeds, seed hairs, seeds without testa, kernels, seed oil, seed cake, solid albumen, liquid albumen | 30 | 9.1 | 11 | Chenopodiaceae, Fabaceae, Poaceae, Proteaceae, Celastraceae |
| Stems | Plumules, leafy stems/branches, defoliated stems/branches, stolons, tendrils | 23 | 7.0 | 18 | Asteraceae, Rubiaceae, Oxalidaceae, Montiaceae |
| Inflorescences | Bracts, spathes, spadices, flowers, flower buds, peduncles, receptacles, calyces, corollas, stamens, pollen, pistils. | 10 | 3.0 | 8 | Asteraceae, Berberidaceae, Philesiaceae |
| Exudates | Sap, latex, leaf juice, gum, resin, nectar | 9 | 2.7 | 4 | Fabaceae, Arecaceae, Asteraceae, Nothofagaceae |
| Bark | Stem bark, inner bark, root bark | 3 | 0.9 | 2 | Rosaceae, Quillajaceae |
| Seedlings | Seedlings, germinated seeds | 1 | 0.3 | 1 | Chenopodiaceae |
| Entire plant | Leaves, stems, flowers and roots | 4 | 1.2 | 3 | Asteraceae, Brassicaceae |
| Unspecified aerial parts | 18 | 5.5 | 4 | Asteraceae, Apiaceae, Aizoaceae | |
| Unspecified parts | 32 | 9.7 | 20 | Brassicaceae, Polygonaceae, Verbenaceae |
| Preparations | Description | Number of Species | % | Number of Families | Main Families |
|---|---|---|---|---|---|
| Raw | Unprocessed | 142 | 43.0 | 44 | Cactaceae, Apiaceae, Grossulariaceae, Myrtaceae, Solanaceae |
| Beverages | Alcoholic beverages, non-alcoholic beverages, juices, infusions/tisanes, coffee substitutes, tea substitutes | 89 | 27.0 | 40 | Grossulariaceae, Asteraceae, Fabaceae Anacardiaceae, Rosaceae |
| Savoury preparations | Soups and diverse cooked dishes (boiled, toasted, fried) | 91 | 27.6 | 39 | Apiaceae, Asteraceae, Fabaceae, Solanaceae, Chenopodiaceae |
| Sweet dishes | Confectionery, jams, jellies, syrups, ice creams | 44 | 13.3 | 20 | Grossulariaceae, Anacardiaceae, Berberidaceae, Ericaceae, Fabaceae |
| Seasoning | Condiments, relishes, chutneys, dressings | 30 | 9.1 | 15 | Asteraceae, Apiaceae, Verbenaceae |
| Cereal/starch-based preparations | Porridges, cakes pastry/shortening | 27 | 8.2 | 11 | Poaceae, Chenopodiaceae, Fabaceae |
| Oils | 7 | 2.1 | 4 | Asteraceae, Fabaceae, Proteaceae | |
| Other preparations | Dehydrated, lyophilised, colorants | 13 | 3.9 | 6 | Cactaceae, Elaeocarpaceae, Quillajaceae |
| Unspecified | 62 | 18.8 | 25 | Asteraceae, Solanaceae, Poaceae, Dioscoreaceae |
| Part Uses: | Infructescences (118) | Roots (71) | Leaves (66) | Seeds (30) | Stems (23) | Inflorescences (10) | Unspecified Parts (50) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Consumption: | n | % | n | % | n | % | n | % | n | % | n | % | n | % |
| Raw | 95 | 80.5 | 27 | 38.0 | 17 | 25.8 | 6 | 20.0 | 4 | 17.4 | 3 | 30.0 | 1 | 2.0 |
| Beverages | 36 | 30.5 | 3 | 4.2 | 26 | 39.4 | 8 | 26.7 | 1 | 4.3 | 1 | 10.0 | 26 | 52.0 |
| Savoury preparations | 6 | 5.1 | 39 | 54.9 | 33 | 50.0 | 11 | 36.7 | 15 | 65.2 | 3 | 30.0 | 4 | 8.0 |
| Sweet dishes | 40 | 33.9 | 1 | 1.5 | 4 | 13.3 | 2 | 20.0 | ||||||
| Seasoning | 2 | 1.7 | 2 | 2.8 | 13 | 19.7 | 1 | 3.3 | 12 | 24.0 | ||||
| Cereal/starch-based preparations | 8 | 6.8 | 7 | 9.9 | 2 | 6.7 | 3 | 6.0 | ||||||
| Oil | 6 | 20.0 | ||||||||||||
| Other preparations | 5 | 4.2 | 1 | 1.4 | 1 | 1.5 | 3 | 10.0 | 1 | 4.3 | ||||
| Unspecified | 6 | 5.1 | 27 | 38.0 | 9 | 13.6 | 3 | 10.0 | 1 | 4.3 | 4 | 40.0 | 12 | 24.0 |
| Scientific Name | Species Common Name | Found in Products as |
|---|---|---|
| Aristotelia chilensis (Molina) Stuntz | maqui | Dehydrated, lyophilised, colorants, beverages, sweet dishes |
| Ugni molinae Turcz. | murta, Chilean guava | Sweet dishes, beverages |
| Gevuina avellana Molina | Chilean hazelnut | Savoury preparations, sweet dishes, cereal/starch-based preparations, oils, beverages |
| Berberis microphylla G. Forst. | calafate | Dehydrated, lyophilised, beverages, sweet dishes |
| Araucaria araucana (Molina) K. Koch | araucaria (piñones) | Cereal/starch-based preparations, savoury preparations, beverages |
| Eulychnia acida Phil. | copao (rumpa) | Beverages, sweet dishes |
| Lapageria rosea Ruiz & Pav. | copihue | Sweet dishes, seasoning (dressing) |
| Geoffroea decorticans (Gillies ex Hook. & Arn.) Burkart | chañar | Sweet dishes, beverages |
| Aloysia deserticola (Phil.) Lu-Irving & O’Leary | rika rika | Seasoning, beverages |
| Drimys winteri J.R. Forst. & G. Forst. | canelo | Seasoning |
| Blechnum chilense (Kaulf.) Mett. | helecho costilla de vaca | Pickles |
| Amomyrtus luma (Molina) D. Legrand & Kausel | luma | Beverages |
| Ribes magellanicum Poir. | zarzaparrilla | Beverages, savoury preparations |
| Solanum tuberosum L. | papa | Savoury preparations |
| Jubaea chilensis (Molina) Baill. | Chilean palm (coquitos) | Sweet dishes |
| Peumus boldus Molina | boldo | Sweet dishes, beverages, oil |
| Haplopappus baylahuen J. Remy | baylahuen | Beverages |
| Buddleja globosa Hope | matico | Beverages |
| Gunnera tinctoria (Molina) Mirb. | nalca | Sweet dishes, seasoning (chutneys) |
| Prosopis alba Griseb. | algarrobo | Sweet dishes |
| Acacia caven (Molina) Molina | espino | Sweet dishes, beverages |
| Cryptocarya alba (Molina) Looser | peumo | Sweet dishes, beverages |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
León-Lobos, P.; Díaz-Forestier, J.; Díaz, R.; Celis-Diez, J.L.; Diazgranados, M.; Ulian, T. Patterns of Traditional and Modern Uses of Wild Edible Native Plants of Chile: Challenges and Future Perspectives. Plants 2022, 11, 744. https://doi.org/10.3390/plants11060744
León-Lobos P, Díaz-Forestier J, Díaz R, Celis-Diez JL, Diazgranados M, Ulian T. Patterns of Traditional and Modern Uses of Wild Edible Native Plants of Chile: Challenges and Future Perspectives. Plants. 2022; 11(6):744. https://doi.org/10.3390/plants11060744
Chicago/Turabian StyleLeón-Lobos, Pedro, Javiera Díaz-Forestier, Rodrigo Díaz, Juan L. Celis-Diez, Mauricio Diazgranados, and Tiziana Ulian. 2022. "Patterns of Traditional and Modern Uses of Wild Edible Native Plants of Chile: Challenges and Future Perspectives" Plants 11, no. 6: 744. https://doi.org/10.3390/plants11060744
APA StyleLeón-Lobos, P., Díaz-Forestier, J., Díaz, R., Celis-Diez, J. L., Diazgranados, M., & Ulian, T. (2022). Patterns of Traditional and Modern Uses of Wild Edible Native Plants of Chile: Challenges and Future Perspectives. Plants, 11(6), 744. https://doi.org/10.3390/plants11060744






