Antihyperglycemic Effects of Salvia polystachya Cav. and Its Terpenoids: α-Glucosidase and SGLT1 Inhibitors
Abstract
:1. Introduction
2. Results
2.1. In Vivo Assays
2.1.1. Acute Oral Toxicity of the Ethanolic Extract of the Stems from S. polystachya
2.1.2. Acute Effect of Ethanolic Extract of Stems from S. polystachya and its Products over Glycemia in Normal and Diabetic Mice
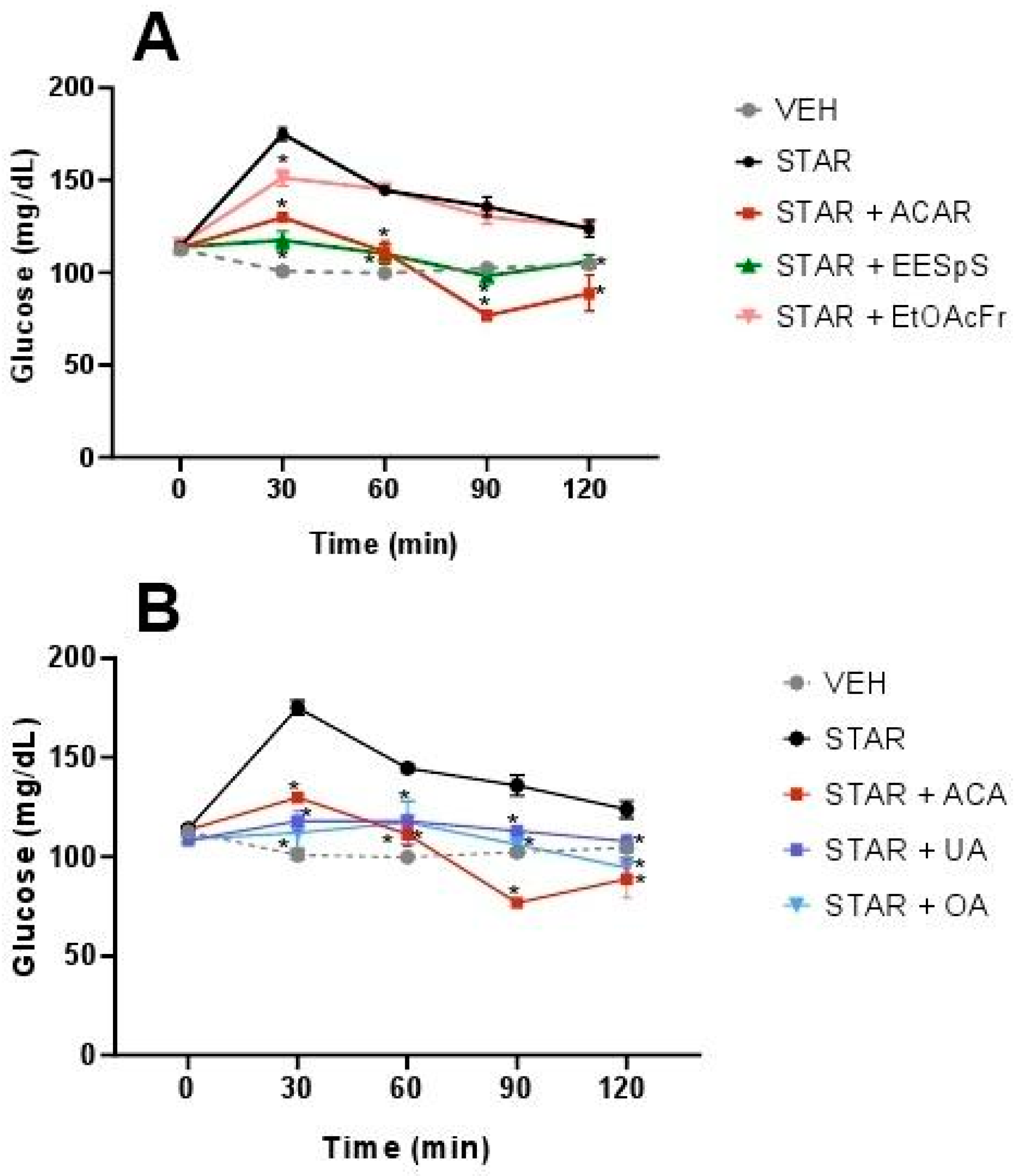
2.1.3. Oral Sucrose and Starch Tolerance Tests of the Products Obtained from Stems from S. polystachya
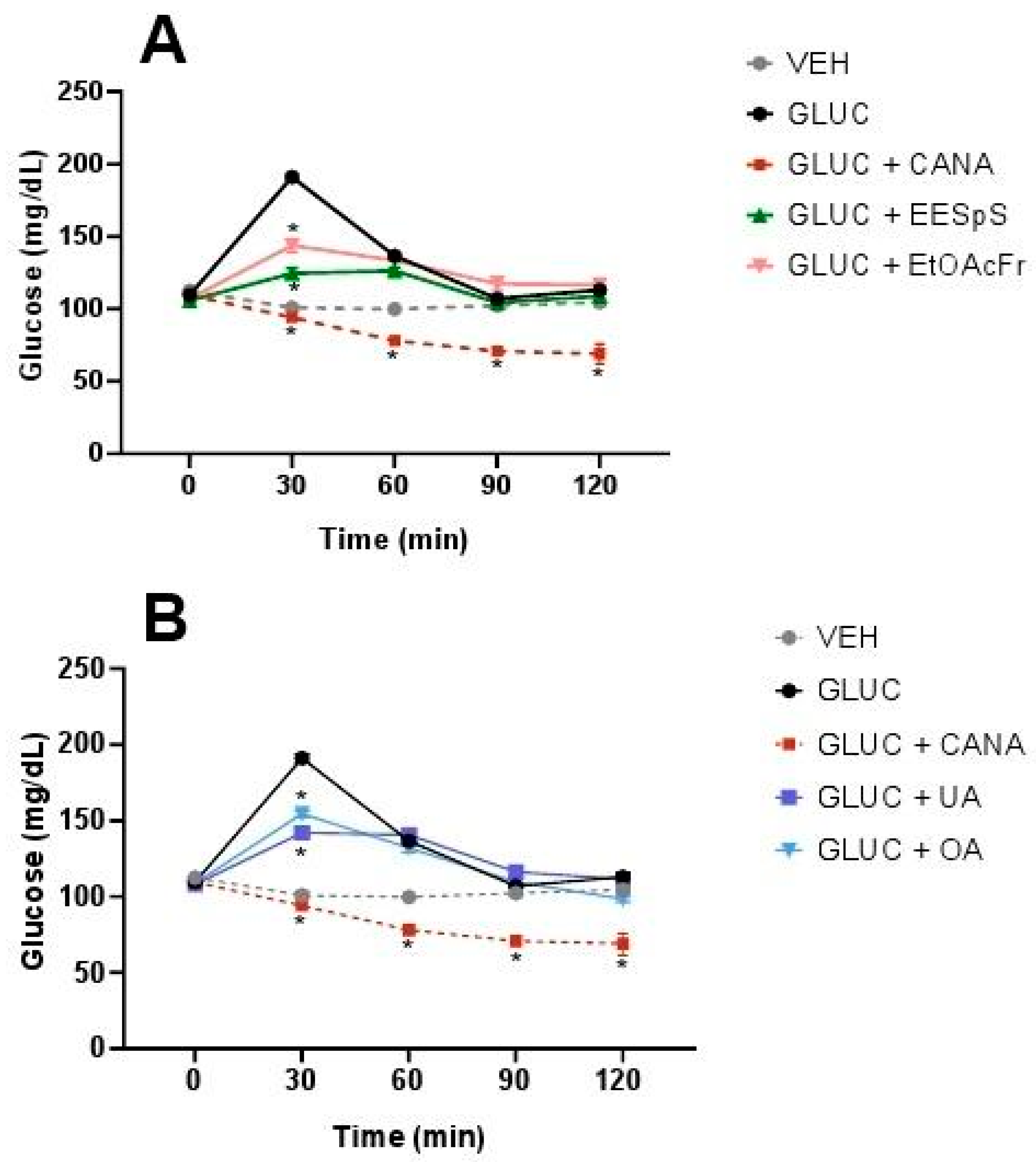
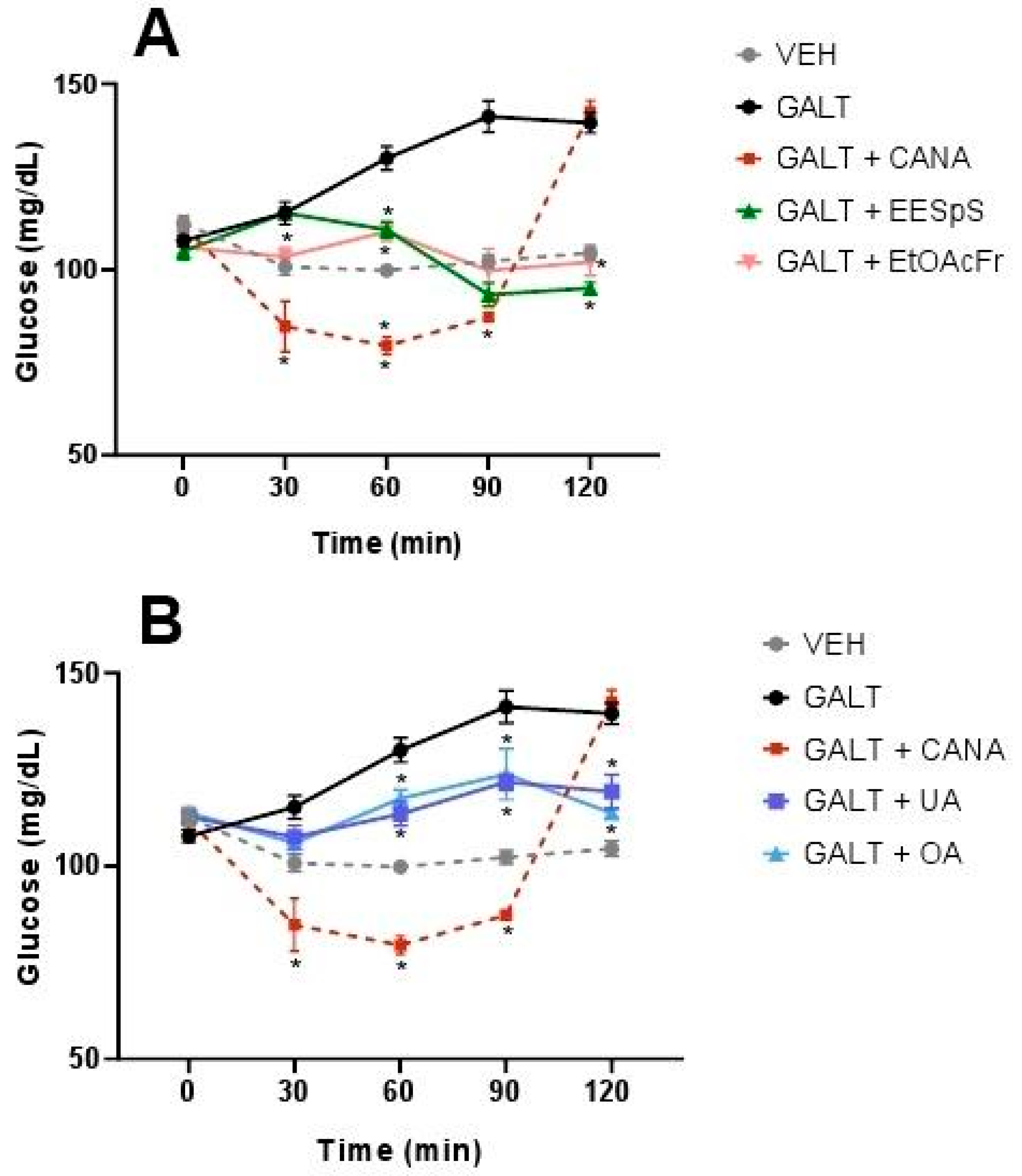
2.1.4. Oral Glucose and Galactose Tolerance Tests of the Products Obtained from Stems from S. polystachya
2.2. Ex Vivo Assays
2.2.1. Inhibition of Intestinal Sucrose Hydrolysis of the Products Obtained from the Stems from S. polystachya
2.2.2. Inhibition of Intestinal Glucose Absorption of the Products Obtained from the Stems from S. polystachya
2.3. In Silico Assays
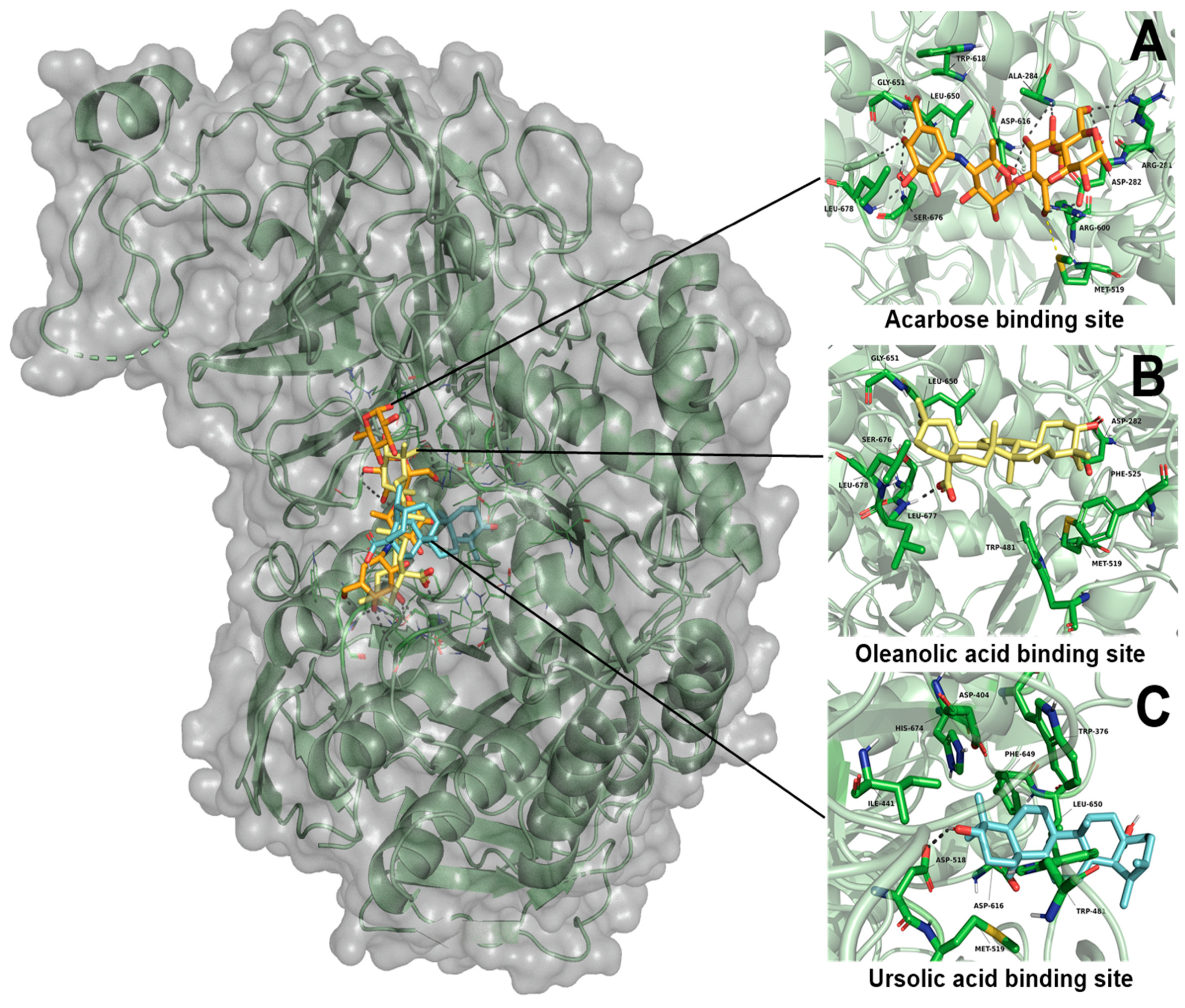
2.3.1. Molecular Docking Studies of Ursolic Acid (UA), Oleanolic Acid (OA) and Acarbose on α-Glucosidase Enzyme
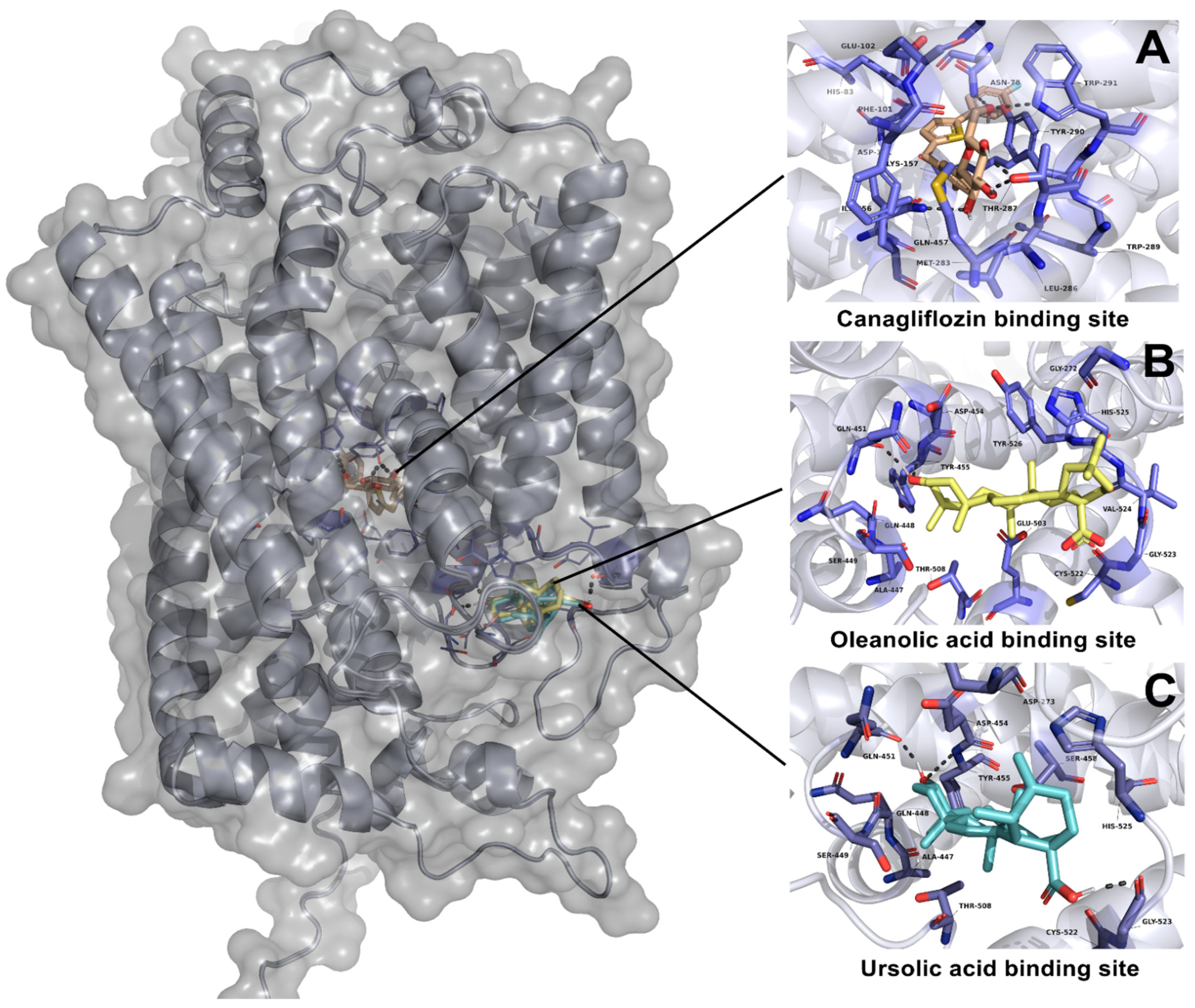
2.3.2. Molecular Docking Studies of Ursolic Acid (UA), Oleanolic Acid (OA) and Canagliflozin on SGLT1 Cotransporter
3. Discussion
4. Materials and Methods
4.1. General Information
4.2. Plant Material
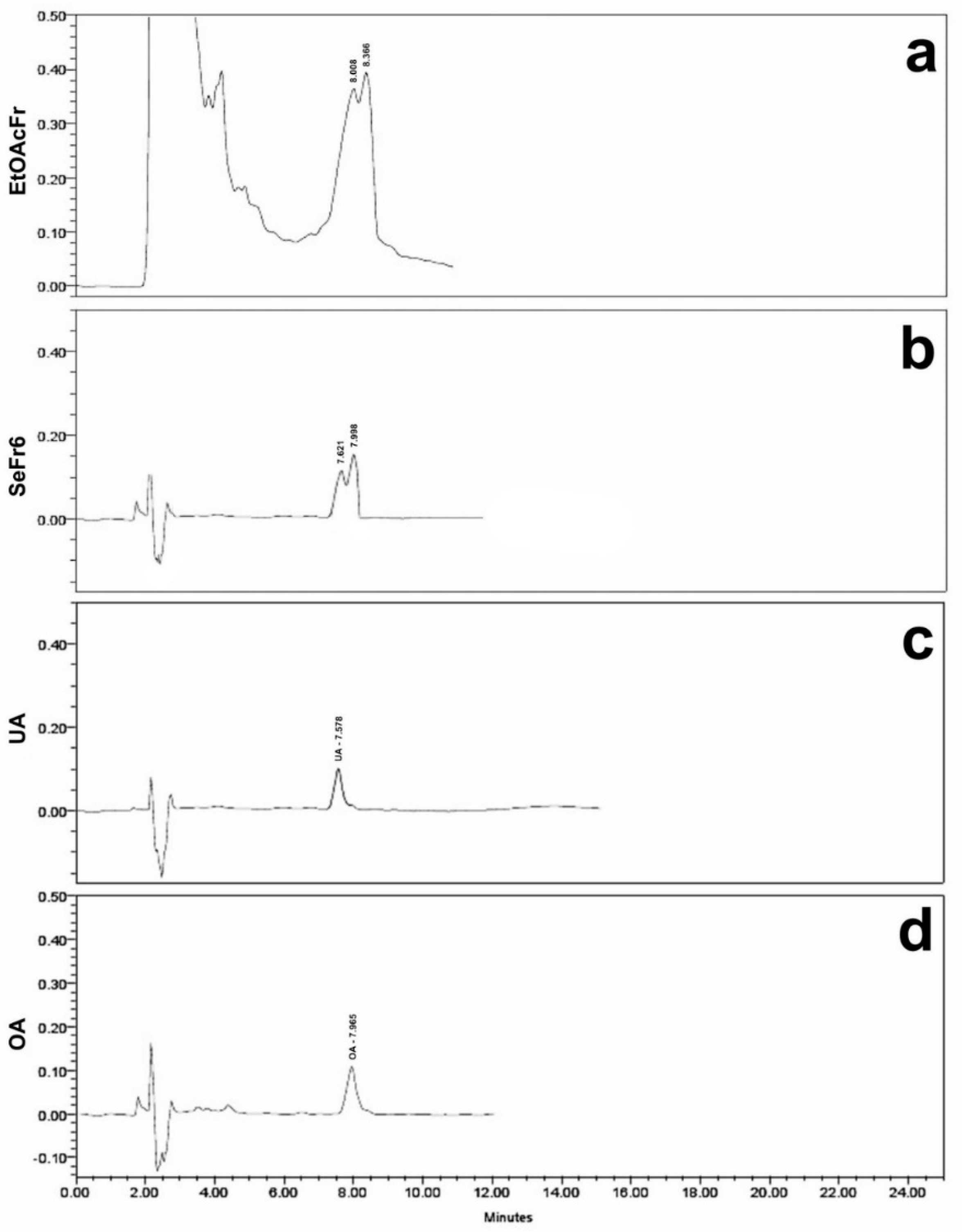
4.3. Preparation of Ethanolic Extracts, Fractionation, and Characterization of Ursolic and Oleanolic Acid
4.4. Experimental Animals
4.5. In Vivo Assays
4.5.1. Acute Toxicity Study
4.5.2. Induction of Experimental Type 2 Diabetes
4.5.3. Acute Effect of Ethanolic Extract from Stems of S. polystachya and Its Products over Glycemia
4.5.4. Oral Sucrose and Starch Tolerance Tests of the Products Obtained from Stems from S. polystachya
4.5.5. Oral Glucose and Galactose Tolerance Tests of the Products Obtained from the Stems from S. polystachya
4.6. Ex Vivo Assays
Determination of the Inhibition of Intestinal Sucrose Hydrolysis and Glucose Absorption of the Products Obtained from the Stems from S. polystachya Cav. and Its Products
4.7. In Silico Assays
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Diabetes. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 23 November 2021).
- American Diabetes Association. Diagnosis and Classification of Diabetes Mellitus. Available online: http://archives.diabetes.org/es/informacion-basica-de-la-diabetes/?loc=globalnav (accessed on 23 November 2021).
- Guthrie, R.A.; Guthrie, D.A. Pathophysiology of Diabetes Mellitus. Crit. Care Nurs. Q. 2004, 27, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Maritin, A.; Sanders, R.; Watkins, J. Diabetes, oxidative stress, and antioxidants: A review. J. Biochem. Mol. Toxicol. 2003, 17, 24–38. [Google Scholar] [CrossRef] [PubMed]
- Wada, J.; Makino, H. Inflammation and the pathogenesis of diabetic nephropathy. Clin. Sci. 2013, 124, 139–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halimi, S. DPP-4 inhibitors and GLP-1 analogues: For whom? Which place for incretins in the management of type 2 diabetic patients? Diabetes Metab. 2008, 34, S91–S95. [Google Scholar] [CrossRef]
- Lambeir, A.; Scharpé, S.; Meester, I. DPP4 inhibitors for diabetes-What next? Biochem. Pharmacol. 2008, 76, 1637–1643. [Google Scholar] [CrossRef] [PubMed]
- Tasnuva, S.; Qamar, U.; Ghafoor, K.; Sahena, F.; Jahurul, M.; Rukshana, A.; Juliana, M.; Al-Juhaimi, F.; Jalifah, L.; Jalal, K.; et al. α-glucosidase inhibitors isolated from Mimosa pudica L. Nat. Prod. Res. 2017, 33, 1495–1499. [Google Scholar] [CrossRef] [PubMed]
- Furman, B. Acarbose. In Elsevier Strathclyde Institute of Pharmacy and biomedical Sciences, 1st ed.; Elsevier: Glasgow, UK, 2017; pp. 1–3. [Google Scholar]
- Aftab, S.; Surhesh, R.; Sherali, N.; Daniyal, M.; Tsouklidis, N. Sodium-glucose cotransporter-2 (SGLT-2) inhibitors: Benefits in diabetic with cardiovascular disease. Cureus 2020, 12, e10783. [Google Scholar] [CrossRef] [PubMed]
- Alam, U.; Asghar, O.; Azmi, S.; Malik, R. General aspects of diabetes mellitus. Handb. Clin. Neurol. 2014, 126, 211–222. [Google Scholar]
- Valdés, M.; Calzada, F.; Mendieta-Wejebe, J. Structure-ativity relationship of acyclic terpenes in blood glucose levels: Potential α-glucosidase and sodium glucose cotransporter (SGLT-1) inhibitors. Molecules 2019, 24, 4020. [Google Scholar] [CrossRef] [Green Version]
- Valdés, M.; Calzada, F.; Mendieta-Wejebe, J.; Merlín-Lucas, V.; Velázquez, C.; Barbosa, E. Antihyperglycemic effects of Annona diversifolia Safford and its acyclic terpenoids: α-glucosidase and selective SGLT1 inhibitors. Molecules 2020, 25, 3361. [Google Scholar] [CrossRef]
- De Melo, E.B.; da Silveira, A.G.; Carvalho, I. α- and β-glucosidase inhibitors: Chemical structure and biological activity. Tetrahedron 2006, 62, 10277–10302. [Google Scholar] [CrossRef]
- Castrejón, V.; Carbó, R.; Martínez, M. Mecanismos moleculares que intervienen en el transporte de la glucosa. Rev. Ed. Bioq. 2007, 368, 85–97. [Google Scholar]
- Asante, D.; Effah-Yeboah, E.; Barnes, P.; Abban, H.A.; Ameyaw, E.O.; Boampong, J.N.; Ofori, E.G.; Dadzie, J. Antidiabetic effect of young and old ethanolic leaf extracts of Vernonia amygdalina: A comparative study. J. Diabetes Res. 2016, 2016, 8252741. [Google Scholar] [CrossRef] [Green Version]
- Salinas-Arellano, E.; Pérez-Vásquez, A.; Rivero-Cruz, I.; Torres-Colin, R.; González-Andrade, M.; Rangel-Grimaldo, M.; Mata, R. Flavonoids and terpenoids with PTP-1B inhibitory properties from the infusion of Salvia amarissima Ortega. Molecules 2020, 25, 3530. [Google Scholar] [CrossRef] [PubMed]
- IDF. Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; Available online: https://www.diabetesatlas.org/ (accessed on 29 November 2021).
- Brindis, F.; Rodríguez, R.; Bye, R.; González-Andrade, M.; Mata, R. (Z)-3-butylidenephthalide from Ligusticum porteri, an α-glucosidase inhibitor. J. Nat. Prod. 2011, 74, 314–320. [Google Scholar] [CrossRef]
- Solares-Pascasio, J.I.; Ceballos, G.; Calzada, F.; Barbosa, E.; Velazquez, C. Antihyperglycemic and lipid profile effects of Salvia amarissima Ortega on Streptozocin-Induced Type 2 diabetic mice. Molecule 2021, 26, 947. [Google Scholar] [CrossRef]
- Zarei, A.; Vaezi, G.; Malekirad, A.A.; Abdollahi, M. Hypoglycemic and hypolipidemic activities of Salvia hydrangea in streptozotocin-induced diabetes in rats. Iran. J. Basic Med. Sci. 2015, 18, 417–422. [Google Scholar]
- Sairafianpour, M.; Bahreininejad, B.; Witt, M.; Ziegler, H.L.; Jaroszewski, J.W.; Stærk, D. Terpenoids of Salvia hydrangea: Two new, rearranged 20-norabietanes and the effect of oleanolic acid on erythrocyte membranes. Planta Med. 2003, 69, 846–850. [Google Scholar]
- Flores-Bocanegra, L.; González-Andrade, M.; Bye, R.; Linares, E.; Mata, R. α-glucosidase inhibitors from Salvia circinata. J. Nat. Prod. 2017, 80, 1584–1593. [Google Scholar] [CrossRef]
- Bahadori, M.B.; Salehi, P.; Sonboli, A. Comparative study of the essential oil composition of Salvia urmiensis and its enzyme inhibitory activities linked to diabetes mellitus and Alzheimer’s disease. Int. J. Food Prop. 2017, 20, 2974–2981. [Google Scholar] [CrossRef] [Green Version]
- Bautista, E.; Fragoso-Serrano, M.; Ortiz-Pastrana, N.; Toscano, R.; Ortega, A. Structural elucidation and evaluation of multidrug-resistance modulatory capability of amarissinins A–C, diterpenes derived from Salvia amarissima. Fitoterapia 2016, 114, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bautista, E.; Fragoso-Serrano, M.; Toscano, R.; García-Peña, M.; Ortega, A. Teotihuacanin, a diterpene with an unusual spiro-10/6 system from Salvia amarissima with potent modulatory activity of multidrug resistance in cancer cells. Org. Lett. 2015, 17, 3280–3282. [Google Scholar] [CrossRef] [PubMed]
- Esquivel, B.; Esquivel, O.; Cárdenas, J.; Adela Sánchez, A.; Ramamoorthy, T.P.; Alfredo Toscano, R.; Rodríguez-Hahn, L. Clerodane and seco-clerodane diterpenoids from Salvia reptans. Phytochemistry 1991, 30, 2335–2338. [Google Scholar] [CrossRef]
- Aydoǧmuş, Z.; Yeşilyurt, V.; Topcu, G. Constituents of Salvia microphylla. Nat. Prod. Res. 2006, 20, 775–781. [Google Scholar] [CrossRef]
- Maldonado, E.; Cárdenas, J.; Bojórquez, H.; Escamilla, E.M.; Ortega, A. Amarisolide, a neo-clerodane diterpene glycoside from Salvia amarissima. Phytochemistry 1996, 42, 1105–1108. [Google Scholar] [CrossRef]
- Jaeger, R.; Cuny, E. Terpenoids with special pharmacological significance: A review. Nat. Prod. Commun. 2016, 11, 1373–1390. [Google Scholar] [CrossRef] [Green Version]
- Rzedowski, J.; de Rzedowski, G. Tres especies mexicanas de Lobelia (Campanulaceae, Lobelioideae), una redescubierta y dos nuevas. Acta Bot. Mex. 2001, 55, 29–38. [Google Scholar] [CrossRef] [Green Version]
- Ríos, J.V.; García, F.E. Catálogo de malezas de México. Consejo Nacional Consultivo Fitosanitario; Universidad Nacional Autónoma de México: Mexico City, Mexico, 1998. [Google Scholar]
- Ortega, A.; Bautista, E.; Maldonado, E. Polystachyne F, a 5,10-seco-neoclerodane from Salvia polystachya. Chem. Pharm. Bull. 2006, 54, 1338–1339. [Google Scholar] [CrossRef] [Green Version]
- Bushway, A.; Belyea, R.; Bushway, J. Chia seed as a source of oil, polysaccharide. Food Sci. 1981, 46, 1349–1350. [Google Scholar] [CrossRef]
- Calzada, F.; Yepez-Mulia, L.; Tapia-Contreras, A.; Bautista, E.; Maldonado, E.; Ortega, A. Evaluation of the antiprotozoal activity of neo-clerodane type diterpenes from Salvia polystachya against Entamoeba histolytica and Giardia lamblia. Phyther. Res. 2010, 24, 662–665. [Google Scholar]
- Bautista, E.; Ortiz-Pastrana, N.; Pastor-Palacios, G.; Montoya-Contreras, A.; Toscano, R.A.; Morales-Jiménez, J.; Salazar-Olivo, L.A.; Ortega, A. Neo-clerodane diterpenoids from Salvia polystachya stimulate the expression of extracellular matrix components in human dermal fibroblasts. J. Nat. Prod. 2017, 80, 3003–3009. [Google Scholar] [CrossRef]
- OCDE. Guideline for Testing if Chemicals. Acute Oral Toxicity-Acute Toxic Class Method. 2001; pp. 1–14. Available online: https://ntp.niehs.nih.gov/Iccvam/suppdocs/feddocs/oecd/oecd_gl423.pdf (accessed on 26 November 2021).
- Skudelski, T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res. 2001, 50, 537–546. [Google Scholar]
- Diab, H.; Fares, M.; Abdei-Valugerdi, M.; Kumagai-Baresch, M.; Holgersson, J.; Hassan, M. Immunotoxicological effects of streptozotocin and alloxan in vitro and in vivo studies. Imminol. Lett. 2015, 163, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Chatuphonprasert, W.; Lao-Ong, T.; Jarukamjorn, K. Improvement of superoxide dismutase and catalase in streptozotocin-nicotinamide-induced type 2-diabetes in mice by berberine and glibenclamide. Pharm. Biol. 2014, 52, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Ovalle-Magallanes, B.; Medina-Campos, O.N.; Pedraza-Chaverri, J.; Mata, R. Hypoglycemic and antihyperglycemic effects of phytopreparations and limonoids from Swietenia humilis. Phytochemistry 2015, 110, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Blain, R.B. Hormesis and plant biology. Environ. Pollut. 2009, 157, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.M.; Yee, S.T.; Choi, J.; Choi, M.S.; Do, G.M.; Jeon, S.M.; Yeo, J.; Kim, M.J.; Seo, K.-I.; Lee, M.-K. Ursolic acid enhances the cellular immune system and pancreatic β-cell function in streptozotocin-induced diabetic mice fed a high-fat diet. Int. Immunopharmacol. 2009, 9, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.J.G.; Frederico, M.J.S.; Cazarolli, L.H.; Mendes, C.P.; Bretanha, L.C.; Schmidt, É.C.; Bouzon, Z.L.; de Medeiros, V.A.P.; da Fonte, C.R.; Pizzolatti, M.G.; et al. The mechanism of action of ursolic acid as insulin secretagogue and insulinomimetic is mediated by cross-talk between calcium and kinases to regulate glucose balance. Biochim. et Biophys. Acta (BBA) Gen. Subj. 2015, 1850, 51–61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jhong, C.H.; Riyaphan, J.; Lin, S.H.; Chia, Y.C.; Weng, C.F. Screening alpha-glucosidase and alpha-amylase inhibitors from natural compounds by molecular docking in silico. BioFactors 2015, 41, 242–251. [Google Scholar] [CrossRef]
- Kuroda, S.; Kobashi, Y.; Oi, T.; Amada, H.; Okumura-Kitajima, L.; Io, F.; Yamamto, K.; Kakinuma, H. Discovery of a potent, low-absorbable sodium-dependent glucose cotransporter 1 (SGLT1) inhibitor (TP0438836) for the treatment of type 2 diabetes. Bioorganic Med. Chem. Lett. 2018, 28, 3534–3539. [Google Scholar] [CrossRef] [PubMed]
- Röder, P.V.; Geillinger, K.E.; Zietek, T.S.; Thorens, B.; Koepsell, H.; Daniel, H. The role of SGLT1 and GLUT2 in intestinal glucose transport and sensing. PLoS ONE 2014, 9, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, T.; Inoue, R.; Matsumoto, M.; Yajima, T.; Ushida, K.; Iwanaga, T. Comparative expression of hexose transporters (SGLT1, GLUT1, GLUT2, and GLUT5) throughout the mouse gastrointestinal tract. Histochem. Cell Biol. 2011, 135, 183–194. [Google Scholar] [CrossRef] [PubMed]
- Gorboulev, V.; Schurmann, A.; Vallon, V.; Kipp, H.; Jaschke, A.; Klessen, A.; Scherneck, S.; Rieg, T.; Cunard, M.; Srinivasan, A.; et al. Na(+)-D-glucose cotransporter SGLT1 is pivotal for intestinal glucose absorption and glucose-dependent incretin secretion. Diabetes 2012, 61, 187–196. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dirir, A.M.; Daou, M.; Yousef, A.F.; Yousef, L.F. A review of alpha-glucosidase inhibitors from plants as potential candidates for the treatment of type-2 diabetes. Phytochem. Rev. 2021, 1–31. [Google Scholar] [CrossRef]
- Ding, H.; Hu, X.; Xu, X.; Zhang, G.; Gong, D. Inhibitory mechanism of two allosteric inhibitors, oleanolic acid and ursolic acid on α-glucosidase. Int. J. Biol. Macromol. 2018, 107, 1844–1855. [Google Scholar] [CrossRef]
- Jäger, S.; Trojan, H.; Kopp, T.; Laszczyk, M.N.; Scheffler, A. Pentacyclic triterpene distribution in various plants—Rich sources for a new group of multi-potent plant extracts. Molecules 2009, 14, 2016–2031. [Google Scholar] [CrossRef] [Green Version]
- Poongunran, J.; Perera, H.K.I.; Jayasinghe, L.; Fernando, I.T.; Sivakanesan, R.; Araya, H.; Fujimoto, Y. Bioassay-guided fractionation and identification of α-amylase inhibitors from Syzygium cumini leaves. Pharm. Biol. 2017, 55, 206–211. [Google Scholar] [CrossRef] [Green Version]
- Castellano, J.M.; Guinda, A.; Delgado, T.; Rada, M.; Cayuela, J.A. Biochemical basis of the antidiabetic activity of oleanolic acid and related pentacyclic triterpenes. Diabetes 2013, 62, 1791–1799. [Google Scholar] [CrossRef] [Green Version]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Detection of inflammatory biomarkers in Saliva and urine: Potential in diagnosis, prevention, and treatment for chronic diseases. Exp. Biol. Med. 2016, 241, 783–799. [Google Scholar] [CrossRef] [Green Version]
- Šulniūtė, V.; Pukalskas, A.; Venskutonis, P.R. Phytochemical composition of fractions isolated from ten Salvia species by supercritical carbon dioxide and pressurized liquid extraction methods. Food Chem. 2017, 224, 37–47. [Google Scholar] [CrossRef]
- Topçu, G. Bioactive triterpenoids from Salvia species. J. Nat. Prod. 2006, 69, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Abd, A.R.; Rathi, D.G. Bioactive components of Salvia and their potential antidiabetic properties: A review. Molecules 2021, 26, 3042. [Google Scholar]
- Mahdizadeh, R.; Moein, S.; Soltani, N.; Malekzadeh, K.; Mahmoodreza, M. Study the molecular mechanism of Salvia species in prevention of diabetes. Int. J. Pharm. Sci. Res. 2018, 9, 4512–4521. [Google Scholar]
- Khan, T.; Zahid, M.; Asim, M.; Iqbal, Z.; Choudhary, M.I.; Ahmad, V.U. Pharmacological activities of crude acetone extract and purified constituents of Salvia moorcraftiana Wall. Phytomedicine 2002, 9, 749–752. [Google Scholar] [CrossRef] [PubMed]
- Nickavar, B.; Abolhasani, L. Bioactivity-guided separation of an α-amylase inhibitor flavonoid from Salvia virgata. Iran. J. Pharm. Res. 2013, 12, 57–61. [Google Scholar]
- Guzmán-Ávila, R.; Flores-Morales, V.; Paoli, P.; Camici, G.; Ramírez-Espinosa, J.J.; Cerón-Romero, L.; Navarrete-Vázquez, G.; Hidalgo-Figueroa, S.; Yolanda Rios, M.; Villalobos-Molina, R.; et al. Ursolic acid derivatives as potential antidiabetic agents: In vitro, in vivo, and in silico studies. Drug. Dev. Res. 2018, 79, 70–80. [Google Scholar] [CrossRef]
- Norma Oficial Mexicana. NOM-062-ZOO-1999: Especificaciones Técnicas Para la Producción, Cuidado y Uso de los Animals de Laboratorio. 1999. Available online: https://www.fmvz.unam.mx/fmvz/principal/archivos/062ZOO.PDF (accessed on 25 November 2021).
- Porwal, M.; Khan, N.A.; Maheshwari, K.K. Evaluation of acute and subacute oral toxicity induced by ethanolic extract of Marsdenia tenacissima leaves in experimental rats. Sci. Pha. 2017, 85, 29. [Google Scholar] [CrossRef] [Green Version]
- Hanwell, M.; Curtis, D.; Lonie, D.; Vandermeersch, T.; Zurek, E.; Hutchison, G. Avogadro: An advanced semantic chemical editor, visualization, and analysis platform. J. Cheminform. 2012, 4, 17. [Google Scholar] [CrossRef] [Green Version]
- Morris, G.; Lindstrom, W.; Sanner, M.; Belew, R.; Goodshell, D.; Olson, A. Autodock4 and AutodockTools4: Automated docking with selective receptor flexibility. J. Comput. Chem. 2009, 30, 2785–2791. [Google Scholar] [CrossRef] [Green Version]

| Group | Dose (mg/kg) | Pancreas (g) | Liver (g) | Kidneys (g) | Stomach (g) |
|---|---|---|---|---|---|
| Normal | - | 0.8 ± 0.0 | 4.9 ± 0.1 | 1.2 ± 0.05 | 1.4 ± 0.1 |
| EESpS | 50 | 0.7 ± 0.0 | 4.5 ± 0.2 | 1.1 ± 0.1 | 2.1 ± 0.1 |
| 300 | 0.6 ± 0.1 | 5.0 ± 0.3 | 1.2 ± 0.1 | 2.0 ± 0.5 | |
| 2000 | 0.8 ± 0.2 | 4.8 ± 0.2 | 1.2 ± 0.0 | 1.9 ± 0.5 |
| Group | Dose (mg/kg−1) | 0 min | 30 min | 60 min | 90 min | 120 min |
|---|---|---|---|---|---|---|
| NM control | - | 175.3 ± 1.4 | 174 ± 1.8 | 170.5 ± 2.6 | 172.8 ± 2 | 178.5 ± 1.7 |
| DM control | - | 302.5 ± 5.7 | 306 ± 3.7 | 309.4 ± 2.8 | 311.5 ± 1.5 | 294.8 ± 0.6 |
| DM + EESpS | 50 | 295.5 ± 5 | 310.3 ± 12 | 290.3 ± 14 | 300 ± 20 | 291.8 ± 1 |
| DM + EESpS | 100 | 301.8 ± 1 | 242.3 ± 0.6 * | 258.3 ± 1.2 ** | 241.3 ± 0.8 ■ | 247.3 ± 0.3 ■■ |
| DM + EESpS | 200 | 303.3 ± 10 | 320.4 ± 7.3 | 307.8 ± 1.3 | 287 ± 9.5 ■ | 313.3 ± 3.5 |
| DM + AqRFr | 50 | 309 ± 2.6 | 268.4 ± 2.7 | 231.6 ± 3.4 | 218.8 ± 1 | 213.3 ± 7.9 |
| DM + EtOAcFr | 50 | 300 ± 4.8 | 249 ± 5.9 * | 203.3 ± 1.8 ** | 213 ± 4.4 ■ | 187.5 ± 0.6 ■■ |
| DM + SeFr6 | 50 | 293.5 ± 2.5 | 338.8 ± 2.2 | 386 ± 3.9 | 330.3 ± 4.2 | 259.3 ± 5.1 ■■ |
| DM + Oleanolic acid | 50 | 283.2 ± 5.2 | 302 ± 11.4 | 302 ± 7.2 | 239.4 ± 3.1 ■ | 252.8 ± 2.9 ■■ |
| DM + Ursolic acid | 50 | 268.7 ± 4 | 220.8 ± 5.9 * | 239 ± 6.6 ** | 259.2 ± 4 ■ | 236 ± 5.5 ■■ |
| DM + Acarbose | 50 | 308.7 ± 1.6 | 236.8 ± 2.3 * | 237.8 ± 3.4 ** | 249 ± 1.5 ■ | 225 ± 2.5 ■■ |
| DM + Glibenclamide | 50 | 307.6 ± 1.8 | 243.5 ± 6.2 * | 247.8 ± 5.4 ** | 268 ± 1.9 ■ | 292 ± 2.3 ■■ |
| Treatment | Glucose (mg/dL−1) | Glucose (mg/dL−1) | % of Inhibition | IC50 |
|---|---|---|---|---|
| 0 h | 2 h | |||
| Sucrose (15%) | 0 ± 0 | 90.6 ± 5.8 | - | - |
| EESpS [200 µgmL−1] | 0 ± 0 | 71.6 ± 1.6 * | 20.8 ± 1.8 | 734.3 µgmL−1 |
| EESpS [400 µgmL−1] | 0 ± 0 | 55 ± 2 * | 39.2 ± 2.2 | |
| EESpS [800 µgmL−1] | 0 ± 0 | 44 ± 1.8 * | 51.4 ± 2 | |
| EtOAcFr [200 µgmL−1] | 0 ± 0 | 65.6 ± 1.2 * | 27.5 ± 1.3 | 573.5 µgmL−1 |
| EtOAcFr [400 µgmL−1] | 0 ± 0 | 51 ± 1.4 * | 43.7 ± 1.6 | |
| EtOAcFr [800 µg/mL−1] | 0 ± 0 | 35 ± 1.7 * | 61.3 ± 1.8 | |
| UA [200 µM] | 0 ± 0 | 59.3 ± 1.8 * | 34.5± 2 | 739.9 µM |
| UA [400 µM] | 0 ± 0 | 48.3 ± 1.2 * | 46.6 ± 1.3 | |
| UA [800 µM] | 0 ± 0 | 45.3 ± 1.9 * | 49.9± 2.1 | |
| OA [200 µM] | 0 ± 0 | 87 ± 3.9 | 3.9 ± 1.6 | 726.3 µM |
| OA [400 µM] | 0 ± 0 | 77.3 ± 1.2 * | 14.6 ± 1.3 | |
| OA [800 µM] | 0 ± 0 | 50 ± 3.6 * | 44.8 ± 4 | |
| Acarbose [200 µM] | 0 ± 0 | 38.2 ± 1.22 * | 57.8 ± 1.3 | |
| Acarbose [400 µM] | 0 ± 0 | 13.1 ± 0.75 * | 85.5 ± 0.8 | 173 µM |
| Acarbose [800 µM] | 0 ± 0 | 7 ± 0.4 * | 96.7 ± 0.18 | |
| Treatment | Glucose (mg/dL−1) | Glucose (mg/dL−1) | % Of Inhibition | IC50 |
|---|---|---|---|---|
| 0 h | 1 h | |||
| Glucose (5%) | 0 ± 0 | 217.3 ± 6.1 | - | - |
| EESpS [200 µg/mL−1] | 0 ± 0 | 216 ± 8.1 | 0.5 ± 3.7 | 1536.3 µg/mL |
| EESpS [400 µg/mL−1] | 0 ± 0 | 192.5 ± 3.7 * | 11.4 ± 1.4 | |
| EESpS [800 µg/mL−1] | 0 ± 0 | 167.7 ± 9 * | 22.8 ± 4.1 | |
| EtOAcFr [200 µg/mL−1] | 0 ± 0 | 175.3 ± 19 * | 19.3 ± 8.7 | 697.3 µg/mL |
| EtOAcFr [400 µg/mL−1] | 0 ± 0 | 162.7 ± 1.8 * | 25.1 ± 0.8 | |
| EtOAcFr [800 µg/mL−1] | 0 ± 0 | 176.5 ± 7.1 * | 18.7 ± 7.7 | |
| UA [200 µM] | 0 ± 0 | 270 ± 31.3 | 0 | 966.6 µM |
| UA [400 µM] | 0 ± 0 | 188.3 ± 4 * | 13.3 ± 1.8 | |
| UA [800 µM] | 0 ± 0 | 132.3 ± 5.3 * | 39.1 ± 2.4 | |
| OA [200 µM] | 0 ± 0 | 189.3 ± 4 * | 12.8 ± 1.8 | 849.3 µM |
| OA [400 µM] | 0 ± 0 | 132.3 ± 2.2 * | 39.1 ± 1 | |
| OA [800 µM] | 0 ± 0 | 121 ± 6.5 * | 44.3 ± 3 | |
| Canagliflozin [200 µM] | 0 ± 0 | 230 ± 13.3 | 0 | |
| Canagliflozin [400 µM] | 0 ± 0 | 113.5 ± 7.4 * | 49 ± 3.1 | 834.5 µM |
| Canagliflozin [800 µM] | 0 ± 0 | 124 ± 5.7 * | 40.5 ± 1.3 | |
| Compound | α-Glucosidase | SGLT1 | ||||||
|---|---|---|---|---|---|---|---|---|
| ΔG (kcal-mol−1) | H-BR | NPI | RMSD | ΔG (kcal-mol−1) | H-BR | NPI | RMSD | |
| Oleanolic acid | −6.41 | Leu677 | Asp282, Met519, Phe525, Leu650, Gly651, Ser676, Leu678 | - | −10.55 | Gln451 | Gly272, Ala447, Gln448, Ser449, Asp454, Tyr455, Glu503, Thr508, Cys522, Gly523, Val524, His525, Tyr526 | - |
| Ursolic acid | −5.48 | Asp518 | Trp376, Trp481, Met519, Asp616, Phe649, Leu650, His674 | - | −9.65 | Gln451, Gly523 | Asp273, Ala447, Gln448, Ser449, Asp454, Tyr455, Ser458, Thr508, Cys522 | - |
| Acarbose | −8.33 | Arg281, Asp282, Ala284, Arg600, Asp616, Gly651, Ser676, Leu678 | Met519, Trp618, Leu650 | 1.87 | - | - | - | - |
| Canagliflozin | - | - | - | - | −11.04 | Met283, Thr287, Tyr290, Trp291, Gln 457 | Asn78, His83, Phe101 | 1.44 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortega, R.; Valdés, M.; Alarcón-Aguilar, F.J.; Fortis-Barrera, Á.; Barbosa, E.; Velazquez, C.; Calzada, F. Antihyperglycemic Effects of Salvia polystachya Cav. and Its Terpenoids: α-Glucosidase and SGLT1 Inhibitors. Plants 2022, 11, 575. https://doi.org/10.3390/plants11050575
Ortega R, Valdés M, Alarcón-Aguilar FJ, Fortis-Barrera Á, Barbosa E, Velazquez C, Calzada F. Antihyperglycemic Effects of Salvia polystachya Cav. and Its Terpenoids: α-Glucosidase and SGLT1 Inhibitors. Plants. 2022; 11(5):575. https://doi.org/10.3390/plants11050575
Chicago/Turabian StyleOrtega, Rocio, Miguel Valdés, Francisco J. Alarcón-Aguilar, Ángeles Fortis-Barrera, Elizabeth Barbosa, Claudia Velazquez, and Fernando Calzada. 2022. "Antihyperglycemic Effects of Salvia polystachya Cav. and Its Terpenoids: α-Glucosidase and SGLT1 Inhibitors" Plants 11, no. 5: 575. https://doi.org/10.3390/plants11050575









