Phytochemical Screening of Rosmarinus officinalis L. as a Potential Anticholinesterase and Antioxidant–Medicinal Plant for Cognitive Decline Disorders
Abstract
:1. Introduction
2. Results
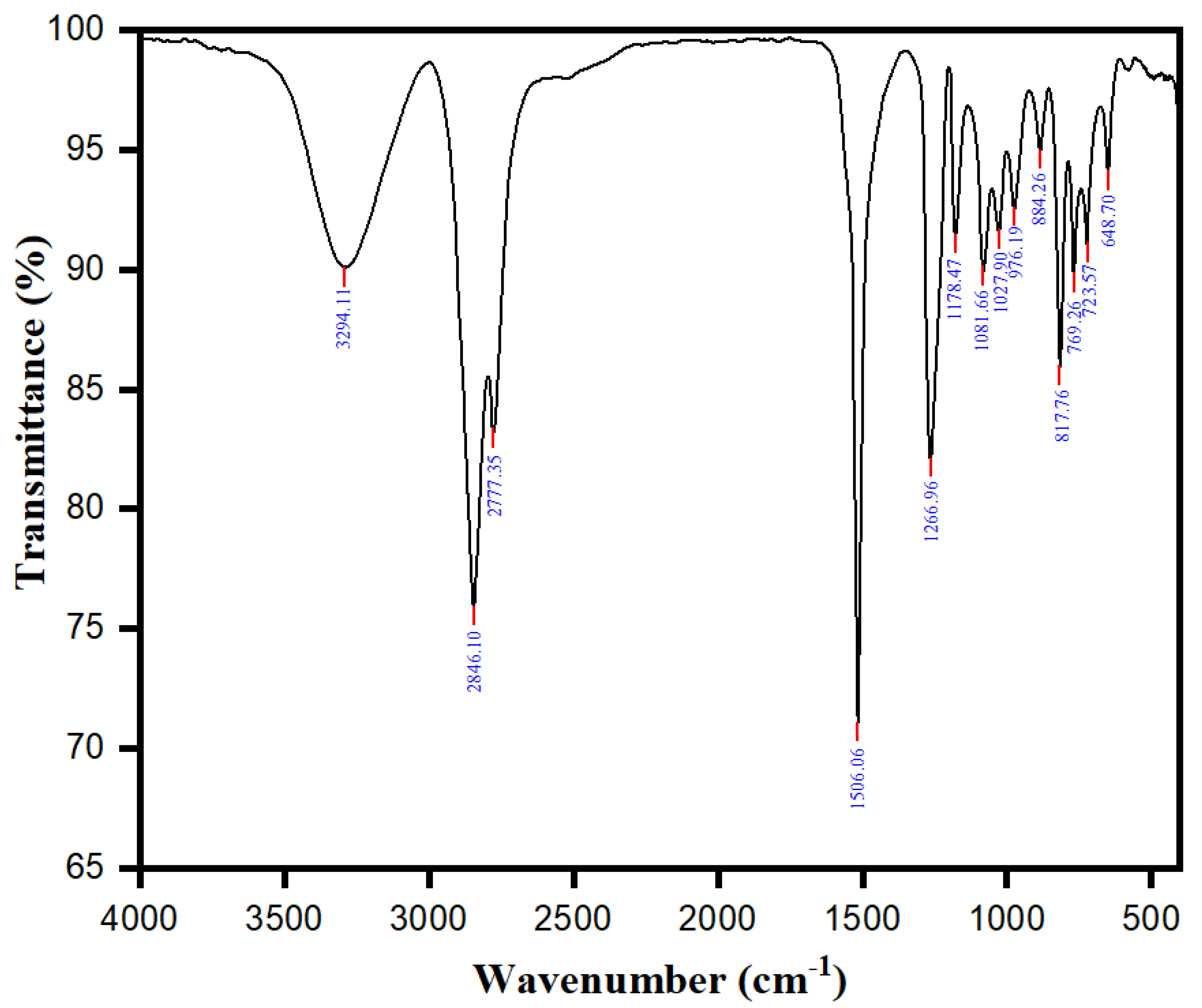
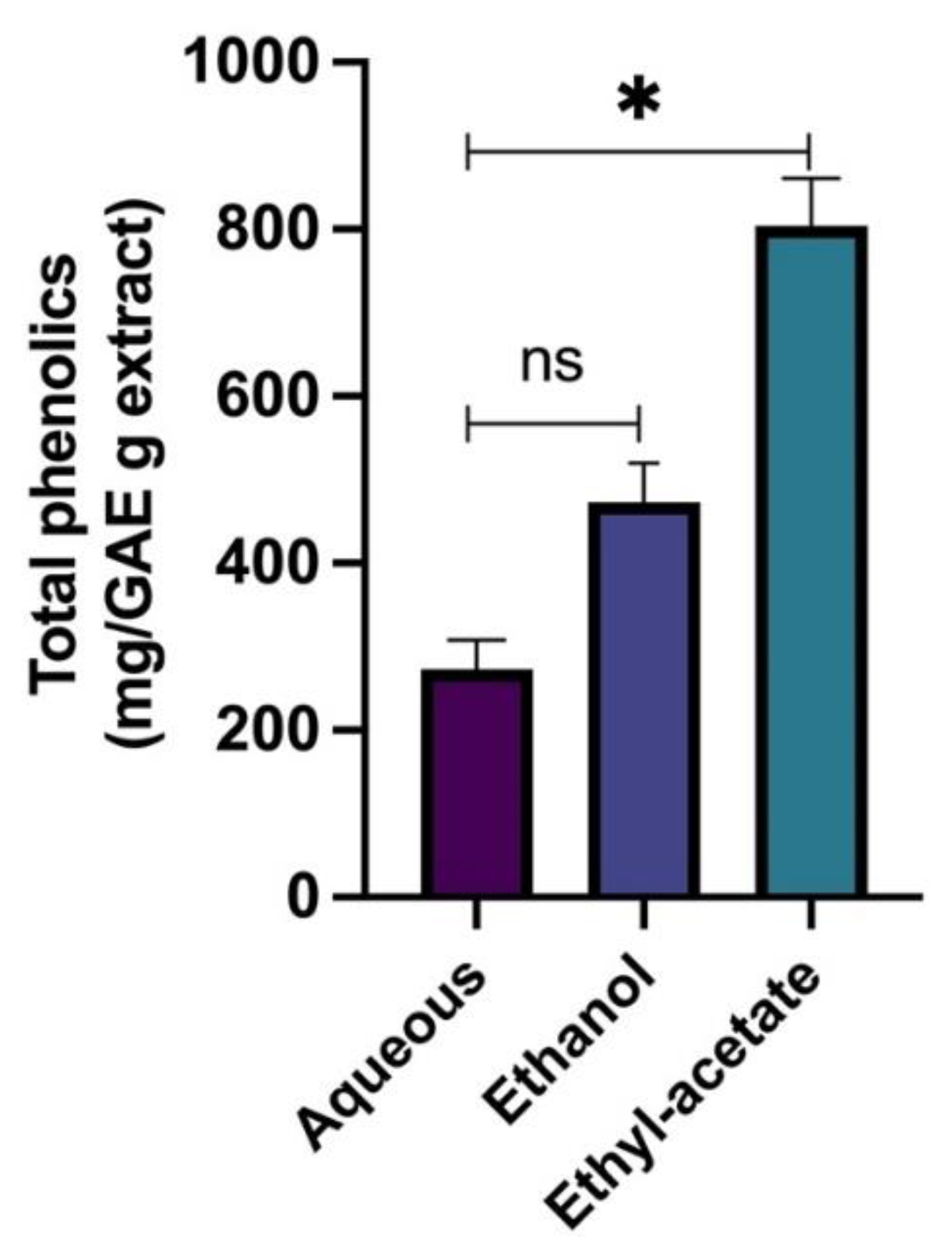
2.1. Functional Group Determination and Total Phenolic Content of the R. officinalis Extracts
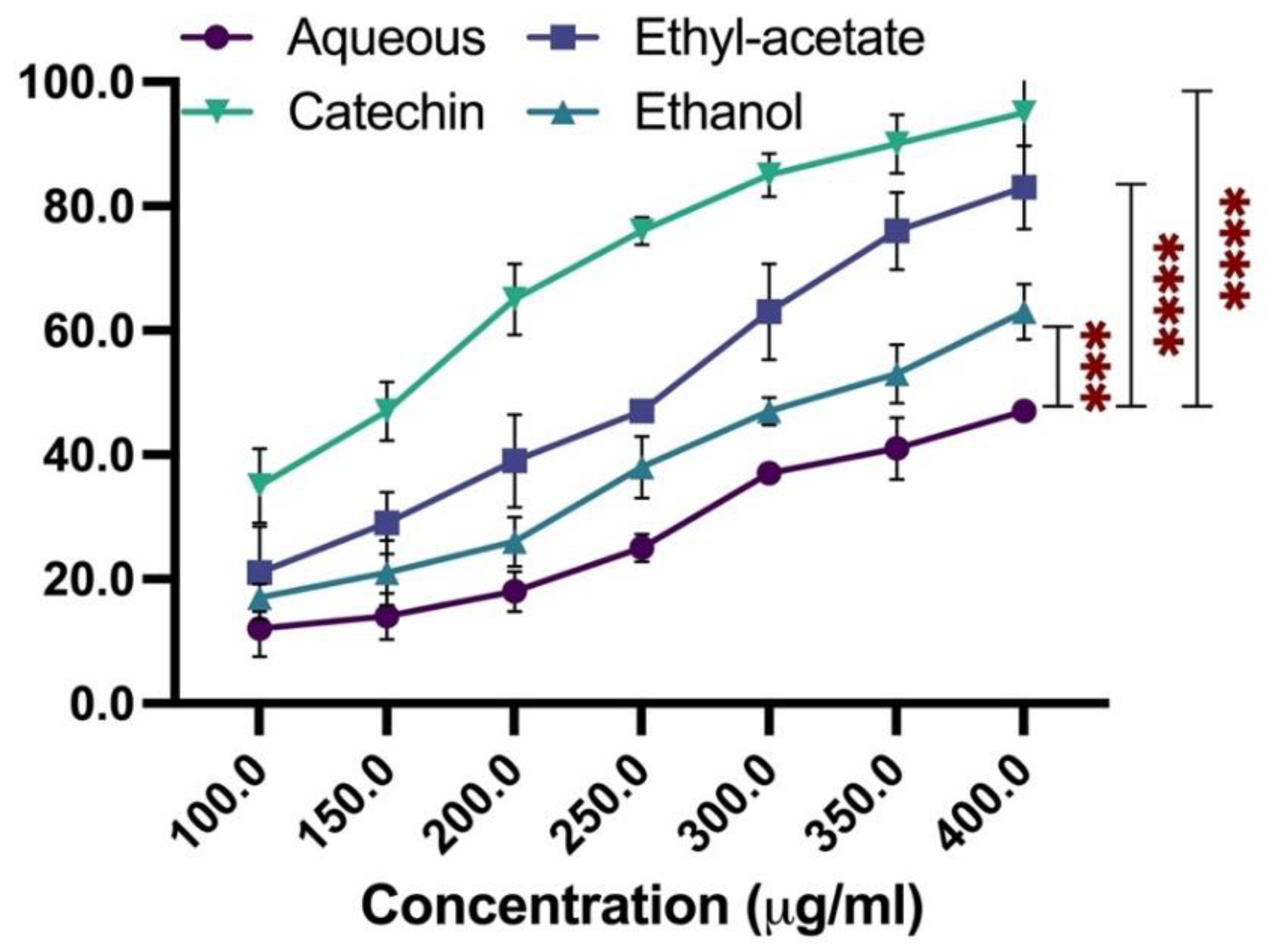
2.2. Free Radical Scavenging Potential of the Rosemarinus Officinalis Extracts
2.3. Inhibition of Acetylcholinesterase by the Extracts
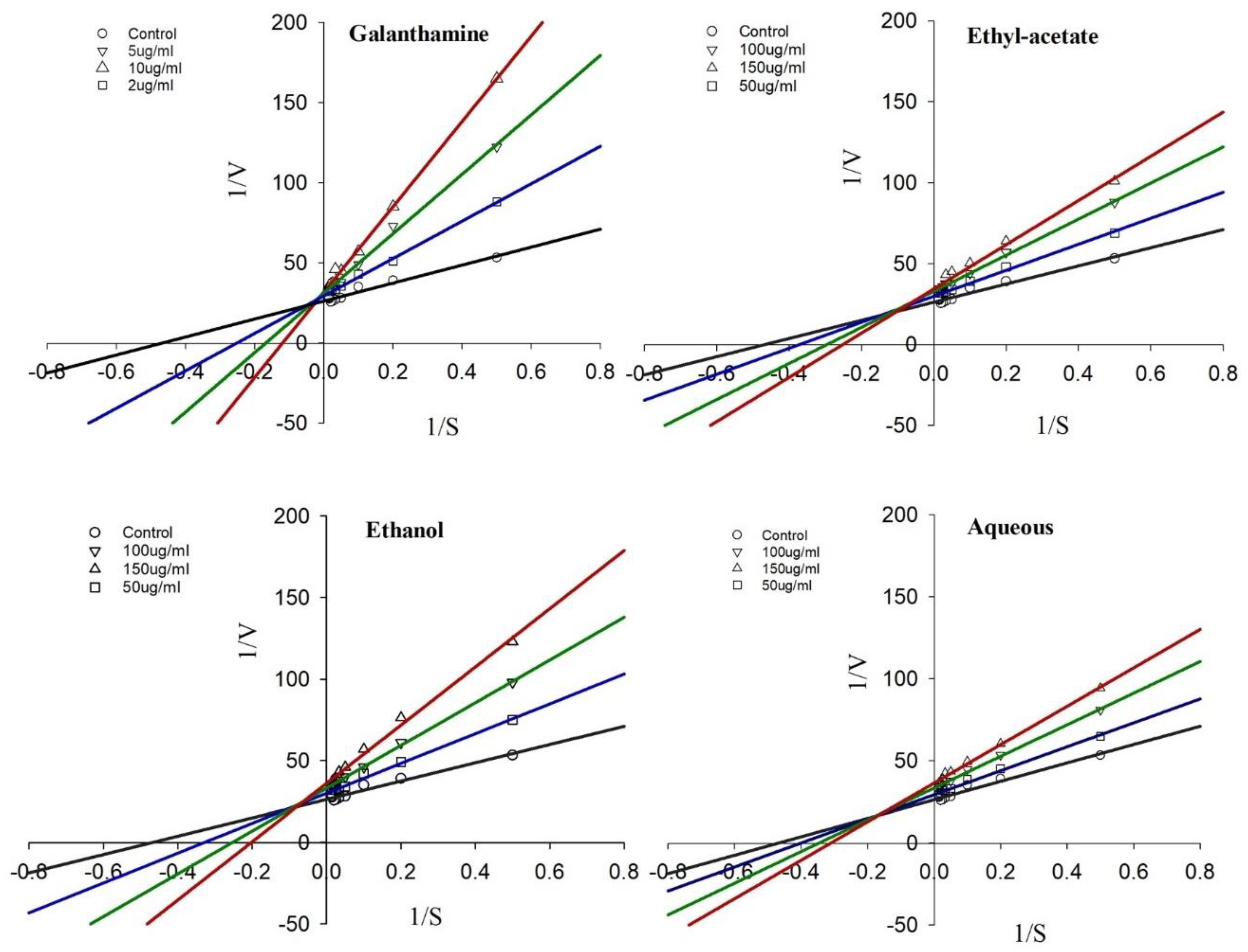
2.4. Kinetics of AChE Inhibition Observed by the R. officinalis Extracts
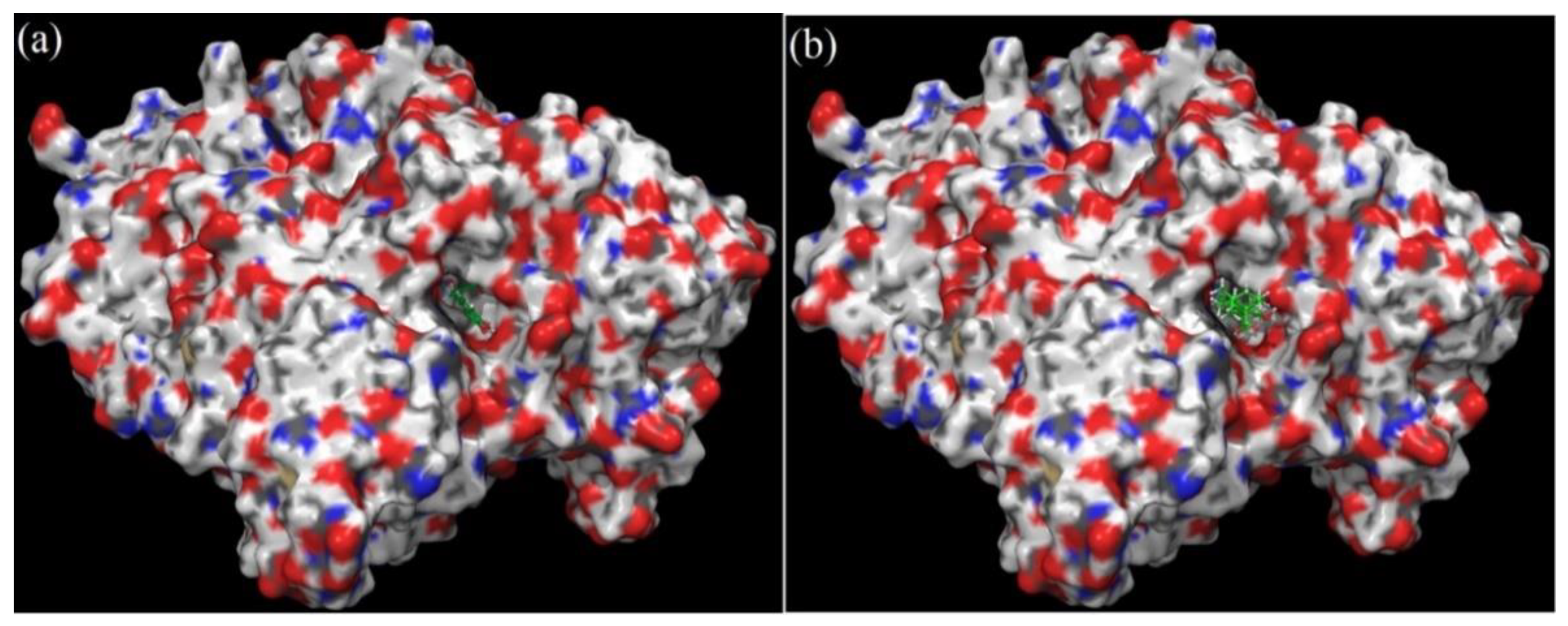
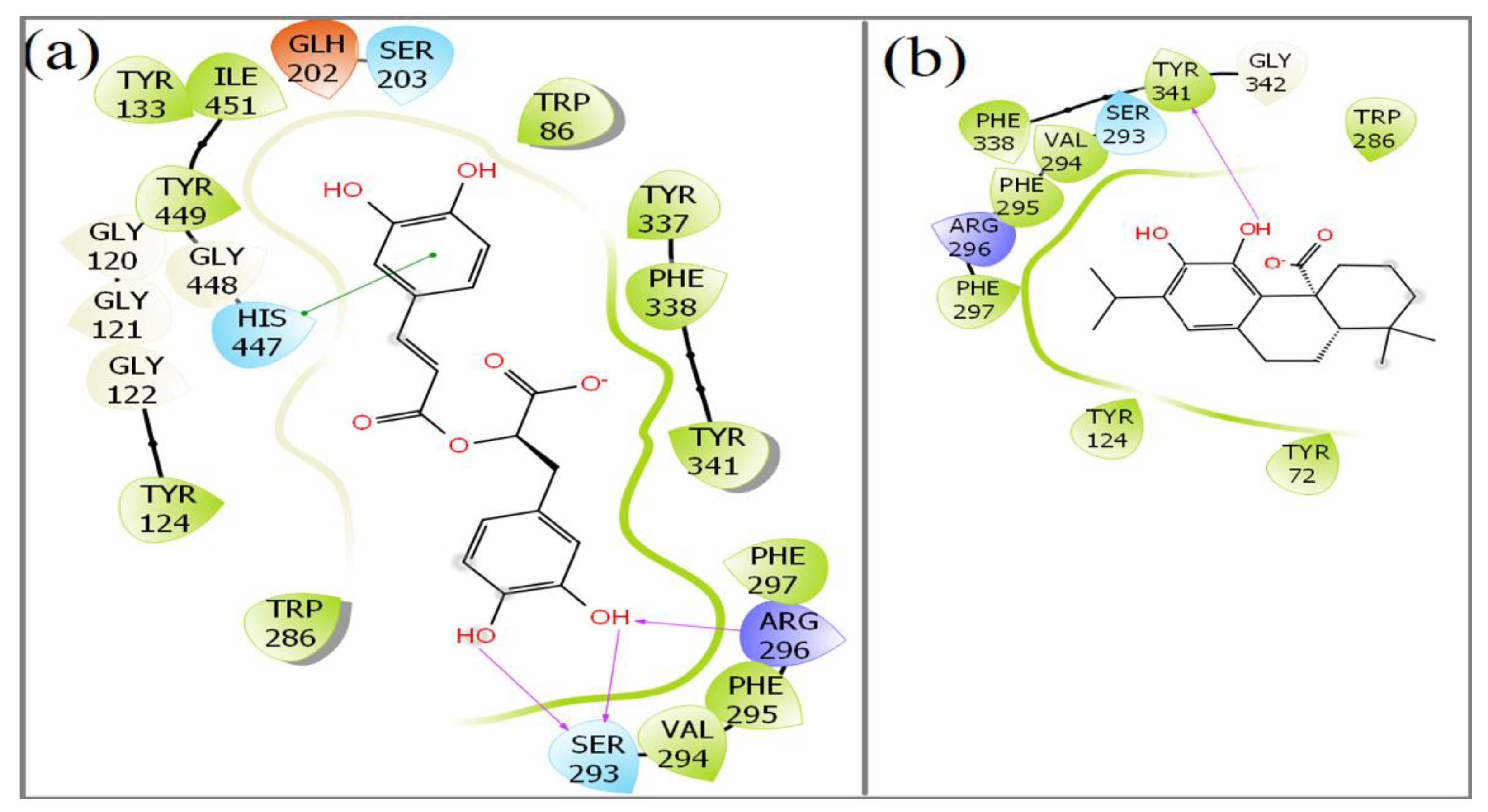
2.5. Molecular Docking of Rosmarinic Acid and Carnosic Acid with Human AChE
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Collection of Plant Material and Preparation of Extract
4.3. Estimation of Total Phenolic Content of the Prepared Rosmarinus Officinalis L. Extracts
4.4. Measurement of DPPH Radical Scavenging Activity
4.5. Cell Culture and Cell-Viability Assay
4.6. Determination of AChE Enzyme Activity
4.7. Estimation of Inhibition Kinetic Parameters
4.8. Induced Fit Docking
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- NIH. Alzheimer’s Disease Fact Sheet. 8 July 2021. Available online: https://www.nia.nih.gov/health/alzheimers-disease-fact-sheet (accessed on 16 October 2021).
- Prince, M.; Bryce, R.; Albanese, E.; Wimo, A.; Ribeiro, W.; Ferri, C.P. The global prevalence of dementia: A systematic review and metaanalysis. Alzheimers Dement. 2013, 9, 63–75. [Google Scholar] [CrossRef]
- Matthews, K.A.; Xu, W.; Gaglioti, A.H.; Holt, J.B.; Croft, J.B.; Mack, D.; McGuire, L.C. Racial and ethnic estimates of Alzheimer’s disease and related dementias in the United States (2015–2060) in adults aged ≥65 years. Alzheimers Dement. 2019, 1, 17–24. [Google Scholar] [CrossRef]
- Howes, M.J.R.; Perry, E. The Role of Phytochemicals in the Treatment and Prevention of Dementia. Drug Aging 2011, 28, 439–468. [Google Scholar] [CrossRef]
- Yiannopoulou, K.G.; Papageorgiou, S.G. Current and Future Treatments in Alzheimer Disease: An Update. J. Cent. Nerv. Syst. Dis. 2020, 29, 12. [Google Scholar] [CrossRef] [Green Version]
- Geula, C.; Darvesh, S. Butyrylcholinesterase, cholinergic neurotransmission and the pathology of Alzheimer’s disease. Drugs Today 2004, 40, 711–721. [Google Scholar] [CrossRef]
- Perry, E.; Walker, M.; Grace, J.; Perry, R. Acetylcholine in mind: A neurotransmitter correlate of consciousness? Trends Neurosci. 1999, 22, 273–280. [Google Scholar] [CrossRef]
- Schneider, L.S. A critical review of cholinesterase inhibitors as a treatment modality in Alzheimer’s disease. Dialogues Clin. Neurosci. 2000, 2, 111–128. [Google Scholar]
- Giacobini, E. Cholinesterase inhibitors stabilize Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2000, 920, 321–327. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Kumar, V.; Houghton, P.J. Screening of Indian medicinal plants for acetylcholinesterase inhibitory activity. Phytother. Res. 2007, 21, 1142–1145. [Google Scholar] [CrossRef]
- Rollinger, J.M.; Schuster, D.; Baier, E.; Ellmerer, E.P.; Langer, T.; Stuppner, H. Taspine: Bioactivity-guided isolation and molecular ligand-target insight of a potent acetylcholinesterase inhibitor from Magnolia x soulangiana. J. Nat. Prod. 2006, 69, 1341–1346. [Google Scholar] [CrossRef] [Green Version]
- Konstantina, G.Y.; Sokratis, G.P. Current and future treatments for Alzheimer’s disease. Ther. Adv. Neurol. Disord. 2013, 61, 19–33. [Google Scholar]
- Mehta, M.; Adem, A.; Sabbagh, M. New Acetylcholinesterase Inhibitors for Alzheimer’s Disease. Int. J. Alzheimers Dis. 2012, 2012, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Akhondzadeh, S.; Abbasi, S.H. Herbal medicine in the treatment of Alzheimer’s disease. Am. J. Alzheimers Dis. Other Dement. 2006, 21, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Mayeux, R.; Sano, M. Treatment of Alzheimer’s disease. N. Engl. J. Med. 1999, 341, 1670–1679. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, M.; Kukreja, H.; Chugh, R.; Silakari, O.; Singh, D. Acetylcholinesterase inhibitors as Alzheimer therapy: From nerve toxins to neuroprotection. Eur. J. Med. Chem. 2013, 70, 165–188. [Google Scholar] [CrossRef]
- Tian, J.; Shi, J.; Zhang, X.; Wang, Y. Herbal therapy: A new pathway for the treatment of Alzheimer’s disease. Alzheimers Res. Ther. 2010, 2, 30. [Google Scholar] [CrossRef] [Green Version]
- Li-Min, F.; Ju-Tzu, L. A Systematic Review of Single Chinese Herbs for Alzheimer’s Disease Treatment. Evid. Based Complement. Altern. Med. 2011, 2011, 640284. [Google Scholar] [CrossRef] [Green Version]
- González-Minero, F.J.; Bravo-Díaz, L.; Ayala-Gómez, A. Rosmarinus officinalis L. (Rosemary): An Ancient Plant with Uses in Personal Healthcare and Cosmetics. Cosmetics 2020, 7, 77. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Carvalho-Costa, D.; Cavaleiro, C.; Costa, H.S.; Albuquerque, T.G.; Castilho, M.C.; Ramos, F.; Melo, N.R.; Sanches-Silva, A. A novel insight on an ancient aromatic plant: The rosemary (Rosmarinus officinalis L.). Trends Food Sci. Technol. 2015, 45, 355–368. [Google Scholar] [CrossRef]
- Amaral, G.P.; Mizdal, C.R.; Stefanello, S.T.; Mendez, A.S.L.; Puntel, R.L.; de Campos, M.M.A.; Soares, F.A.A.; Fachinetto, R. Antibacterial and antioxidant effects of Rosmarinus officinalis L. extract and its fractions. J. Tradit. Complement. Med. 2018, 9, 383–392. [Google Scholar] [CrossRef]
- Rašković, A.; Milanović, I.; Pavlović, N.; Ćebović, T.; Vukmirović, S.; Mikov, M. Antioxidant activity of rosemary (Rosmarinus officinalis L.) essential oil and its hepatoprotective potential. BMC Complement. Altern. Med. 2014, 14, 225. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Li, N.; Luo, M.; Zu, Y.; Efferth, T. Antibacterial activity and anticancer activity of Rosmarinus officinalis L. essential oil compared to that of its main components. Molecules 2012, 17, 2704–2713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allegra, A.; Tonacci, A.; Pioggia, G.; Musolino, C.; Gangemi, S. Anticancer Activity of Rosmarinus officinalis L.: Mechanisms of Action and Therapeutic Potentials. Nutrients 2020, 12, 1739. [Google Scholar] [CrossRef] [PubMed]
- Bao, T.Q.; Li, Y.; Qu, C.; Zheng, Z.G.; Yang, H.; Li, P. Antidiabetic Effects and Mechanisms of Rosemary (Rosmarinus officinalis L.) and its Phenolic Components. Am. J. Chin. Med. 2020, 48, 1353–1368. [Google Scholar] [CrossRef] [PubMed]
- Ghasemzadeh Rahbardar, M.; Amin, B.; Mehri, S.; Mirnajafi-Zadeh, S.J.; Hosseinzadeh, H. Anti-inflammatory effects of ethanolic extract of Rosmarinus officinalis L. and rosmarinic acid in a rat model of neuropathic pain. Biomed. Pharmacother. 2017, 86, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, R.; Alvarado, J.L.; Presno, M.; Pérez-Veyna, O.; Serrano, C.J.; Yahuaca, P. Oxidative stress modulation by Rosmarinus officinalis in CCl4-induced liver cirrhosis. Phytother. Res. 2010, 4, 595–601. [Google Scholar] [CrossRef]
- Singhal, A.K.; Naithani, V.; Bangar, O.P. Medicinal plants with a potential to treat Alzheimer and associated symptoms. Int. J. Nutr. Pharmacol. Neurol. Dis. 2012, 2, 84–91. [Google Scholar] [CrossRef]
- Habtemariam, S. Natural products in Alzheimer’s disease therapy: Would old therapeutic approaches fix the broken promise of modern medicines? Molecules 2019, 24, 1519. [Google Scholar] [CrossRef] [Green Version]
- Ozarowski, M.; Mikolajczak, P.L.; Bogacz, A.; Gryszczynska, A.; Kujawska, M.; Jodynis-Liebert, J.; Piasecka, A.; Napieczynska, H.; Szulc, M.; Kujawski, R.; et al. Rosmarinus officinalis L. leaf extract improves memory impairment and affects acetylcholinesterase and butyrylcholinesterase activities in rat brain. Fitoterapia 2013, 91, 261–271. [Google Scholar] [CrossRef]
- Sharma, K. Cholinesterase inhibitors as Alzheimer’s therapeutics (Review). Mol. Med. Rep. 2019, 20, 1479–1487. [Google Scholar] [CrossRef] [Green Version]
- Lane, R.M.; Kivipelto, M.; Greig, N.H. Acetylcholinesterase and its inhibition in Alzheimer disease. Clin. Neuropharmacol. 2004, 27, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Guzior, N.; Wieckowska, A.; Panek, D.; Malawska, B. Recent development of multifunctional agents as potential drug candidates for the treatment of Alzheimer’s disease. Curr. Med. Chem. 2015, 22, 373–404. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, T.C.; Gomes, T.M.; Pinto, B.A.S.; Camara, A.L.; Paes, A.M.A. Naturally Occurring Acetylcholinesterase Inhibitors and Their Potential Use for Alzheimer’s Disease Therapy. Front Pharmacol. 2018, 18, 1192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef]
- Tanase, C.; Coșarcă, S.; Muntean, D.L. A Critical Review of Phenolic Compounds Extracted from the Bark of Woody Vascular Plants and Their Potential Biological Activity. Molecules 2019, 24, 1182. [Google Scholar] [CrossRef] [Green Version]
- Roseiro, L.B.; Rauter, A.P.; Serralheiro, M.L.M. Polyphenols as acetylcholinesterase inhibitors: Structural specificity and impact on human disease. Nutr. Aging. 2012, 1, 99–111. [Google Scholar] [CrossRef] [Green Version]
- Houghton, P.J.; Ren, Y.; Howes, M.J. Acetylcholinesterase inhibitors from plants and fungi. Nat. Prod. Rep. 2006, 23, 181–199. [Google Scholar] [CrossRef]
- Orhan, I.E.; Orhan, G.; Gurkas, E. An overview on natural cholinesterase inhibitors—A multi-targeted drug class—And their mass production. Mini Rev. Med. Chem. 2011, 11, 836–842. [Google Scholar] [CrossRef]
- Rosenfeld, C.A.; Sultatos, L.G. Concentration-dependent kinetics of acetylcholinesterase inhibition by the organophosphate paraoxon. Toxicol. Sci. 2006, 90, 460–469. [Google Scholar] [CrossRef] [Green Version]
- Rahman, A.-U.; Khalid, A.; Sultana, N.; Nabeel Ghayur, M.; Ahmed Mesaik, M.; Khan, M.R.; Gilani, A.H.; Choudhary, M.I. New natural cholinesterase inhibiting and calcium channel blocking quinoline alkaloids. J. Enzyme Inhib. Med. Chem. 2006, 21, 703–710. [Google Scholar] [CrossRef] [Green Version]
- Mena, P.; Cirlini, M.; Tassotti, M.; Herrlinger, K.A.; Dall’Asta, C.; del Rio, D. Phytochemical Profiling of Flavonoids, Phenolic Acids, Terpenoids, and Volatile Fraction of a Rosemary (Rosmarinus officinalis L.) Extract. Molecules 2019, 21, 1576. [Google Scholar] [CrossRef] [PubMed]
- Szwajgier, D. Anticholinesterase activity of phenolic acids and their derivatives. Z. Nat. C J. Biosci. 2013, 68, 125–132. [Google Scholar]
- Loussouarn, M.; Krieger-Liszkay, A.; Svilar, L.; Bily, A.; Birtić, S.; Havaux, M. Carnosic Acid and Carnosol, Two Major Antioxidants of Rosemary, Act through Different Mechanisms. Plant Physiol. 2017, 175, 1381–1394. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borrás-Linares, I.; Pérez-Sánchez, A.; Lozano-Sánchez, J.; Barrajón-Catalán, E.; Arráez-Román, D.; Cifuentes, A.; Micol, V.; Carretero, A.S. A bioguided identification of the active compounds that contribute to the antiproliferative/cytotoxic effects of rosemary extract on colon cancer cells. Food Chem. Toxicol. 2015, 80, 215–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munné-Bosch, S.; Schwarz, K.; Alegre, L. Response of Abietane Diterpenes to Stress in Rosmarinus officinalis L.: New Insights into the Function of Diterpenes in Plants. Free Radic. Res. 1999, 3, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Demirezer, L.Ö.; Gürbüz, P.; Kelicen Uğur, E.P.; Bodur, E.; Özener, N.; Uz, A.; Güvenalp, Z. Molecular docking and ex vivo and in vitro anticholinesterase activity studies of Salvia sp. and highlighted rosmarinic acid. Turk. J. Med. Sci. 2015, 45, 1141–1148. [Google Scholar] [CrossRef]
- Merad, M.; Soufi, W.; Ghalem, S.; Boukli, F.; Baig, M.H.; Ahmad, K.; Kamal, M.A. Molecular interaction of acetylcholinesterase with carnosic acid derivatives: A neuroinformatics study. CNS Neurol. Disord. Drug Targets 2014, 13, 440–446. [Google Scholar] [CrossRef]
- Mannelli, L.d.; Micheli, L.; Maresca, M.; Cravotto, G.; Bellumori, M.; Innocenti, M.; Mulinacci, N.; Ghelardini, C. Anti-neuropathic effects of Rosmarinus officinalis L. terpenoid fraction: Relevance of nicotinic receptors. Sci. Rep. 2016, 6, 34832. [Google Scholar] [CrossRef] [Green Version]
- Braca, A.; De Tommasi, N.; Di Bari, L.; Pizza, C.; Politi, M.; Morelli, I. Antioxidant Principles from Bauhinia t arapotensis. J. Nat. Prod. 2001, 64, 892–895. [Google Scholar] [CrossRef]
- Albano, S.M.; Lima, A.S.; Miguel, M.G.; Pedro, L.G.; Barroso, J.G.; Figueiredo, A.C. Antioxidant, anti-5-lipoxygenase and antiacetylcholinesterase activities of essential oils and decoction waters of some aromatic plants. Rec. Nat. Prod. 2012, 6, 35–48. [Google Scholar]
- Palmer, T. Enzymes Biochemistry, Biotechnology, Clinical Chemistry; East-West Press Pvt Ltd.: Delhi, India, 2004; ISBN 978-8176710466. [Google Scholar]
- Sheikh, I.A. Stereoselectivity and potential endocrine disrupting activity of Bis-(2-ethylhexyl)phthalate (DEHP) against human progesterone receptor: A computational perspective. J. Appl. Toxicol. 2016, 36, 741–774. [Google Scholar] [CrossRef] [PubMed]
- Ganaie, A.A.; Siddiqui, H.R.; Sheikh, I.A. A novel terpenoid class for prevention and treatment of KRAS-driven cancers: Comprehensive analysis using in situ, in vitro and in vivo model systems. Mol. Carcinog. 2020, 59, 886–896. [Google Scholar] [CrossRef] [PubMed]
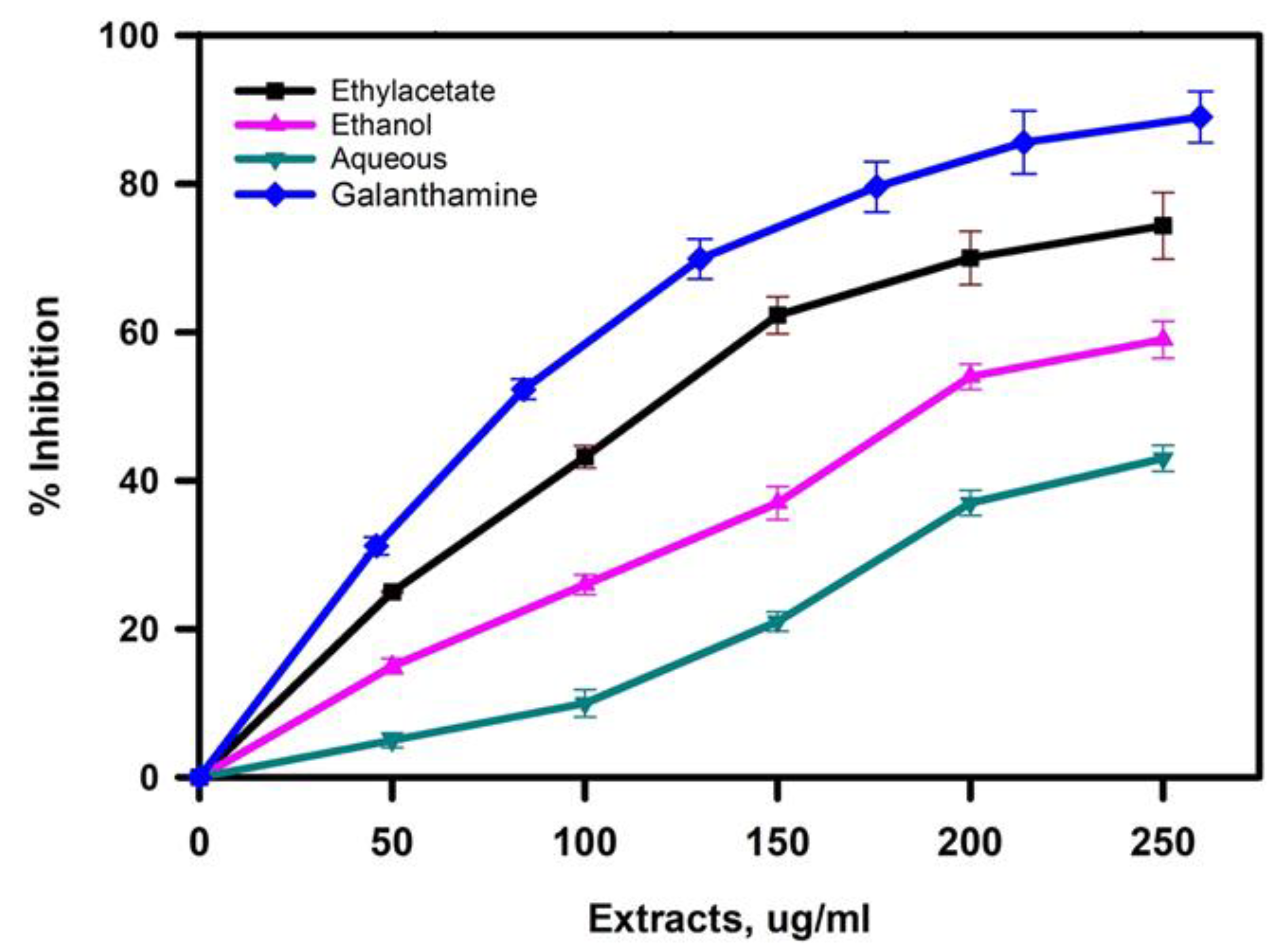

| Extract | DPPH Activity (µg/mL) | p Value **** | Anti-Cholinesterase Activity (µg/mL) | p Value **** |
|---|---|---|---|---|
| Catechin | 173 ± 8 | 0.0001 | 0.0001 | |
| Galanthamine | 0.0001 | 4.73 ± 0.13 | 0.0001 | |
| Ethyl-acetate | 272 ± 7 | 0.0001 | 101.2 ± 5.5 | 0.0001 |
| Ethanol | 387 ± 11 | 0.0001 | 202.3 ± 8.7 | 0.0001 |
| Aqueous | 584 ± 8 | 247 ± 12 |
| Inhibitor | Concentration (µM; µg/mL) | Km (µM) | Vmax |
|---|---|---|---|
| Galanthamine | 0 | 2.14 ± 0.13 | 0.38 ± 0.09 |
| 2 | 5.47 ± 0.17 | 0.29 ± 0.06 | |
| 5 | 9.76 ± 0.25 | 0.21 ± 0.07 | |
| 10 | 17.38 ± 0.33 | 0.15 ± 0.05 | |
| Ethyl-acetate | 50 | 4.21 ± 0.28 | 0.32 ± 0.08 |
| 100 | 7.19 ± 0.41 | 0.28 ± 0.10 | |
| 150 | 8.28 ± 0.63 | 0.23 ± 0.08 | |
| Ethanol | 50 | 3.05 ± 0.21 | 0.35 ± 0.05 |
| 100 | 4.65 ± 0.12 | 0.30 ± 0.06 | |
| 150 | 6.24 ± 0.53 | 0.28 ± 0.08 | |
| Aqueous | 50 | 2.50 ± 0.09 | 0.34 ± 0.09 |
| 100 | 3.67 ± 0.11 | 0.31 ± 0.04 | |
| 150 | 4.22 ± 0.29 | 0.27 ± 0.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamli, M.R.; Sharaf, A.A.M.; Sabir, J.S.M.; Rather, I.A. Phytochemical Screening of Rosmarinus officinalis L. as a Potential Anticholinesterase and Antioxidant–Medicinal Plant for Cognitive Decline Disorders. Plants 2022, 11, 514. https://doi.org/10.3390/plants11040514
Kamli MR, Sharaf AAM, Sabir JSM, Rather IA. Phytochemical Screening of Rosmarinus officinalis L. as a Potential Anticholinesterase and Antioxidant–Medicinal Plant for Cognitive Decline Disorders. Plants. 2022; 11(4):514. https://doi.org/10.3390/plants11040514
Chicago/Turabian StyleKamli, Majid Rasool, Abeer Abdullah M. Sharaf, Jamal S. M. Sabir, and Irfan A. Rather. 2022. "Phytochemical Screening of Rosmarinus officinalis L. as a Potential Anticholinesterase and Antioxidant–Medicinal Plant for Cognitive Decline Disorders" Plants 11, no. 4: 514. https://doi.org/10.3390/plants11040514
APA StyleKamli, M. R., Sharaf, A. A. M., Sabir, J. S. M., & Rather, I. A. (2022). Phytochemical Screening of Rosmarinus officinalis L. as a Potential Anticholinesterase and Antioxidant–Medicinal Plant for Cognitive Decline Disorders. Plants, 11(4), 514. https://doi.org/10.3390/plants11040514







