Silencing Phytoene Desaturase Causes Alteration in Monoterpene Volatiles Belonging to the Methylerythritol Phosphate Pathway
Abstract
:1. Introduction
2. Results
2.1. The Photobleaching Phenotype Is Correlated to the CTV Titer
2.2. VOCs Released from C. Macrophylla
2.3. Prencipal Component Analysis (PCA)
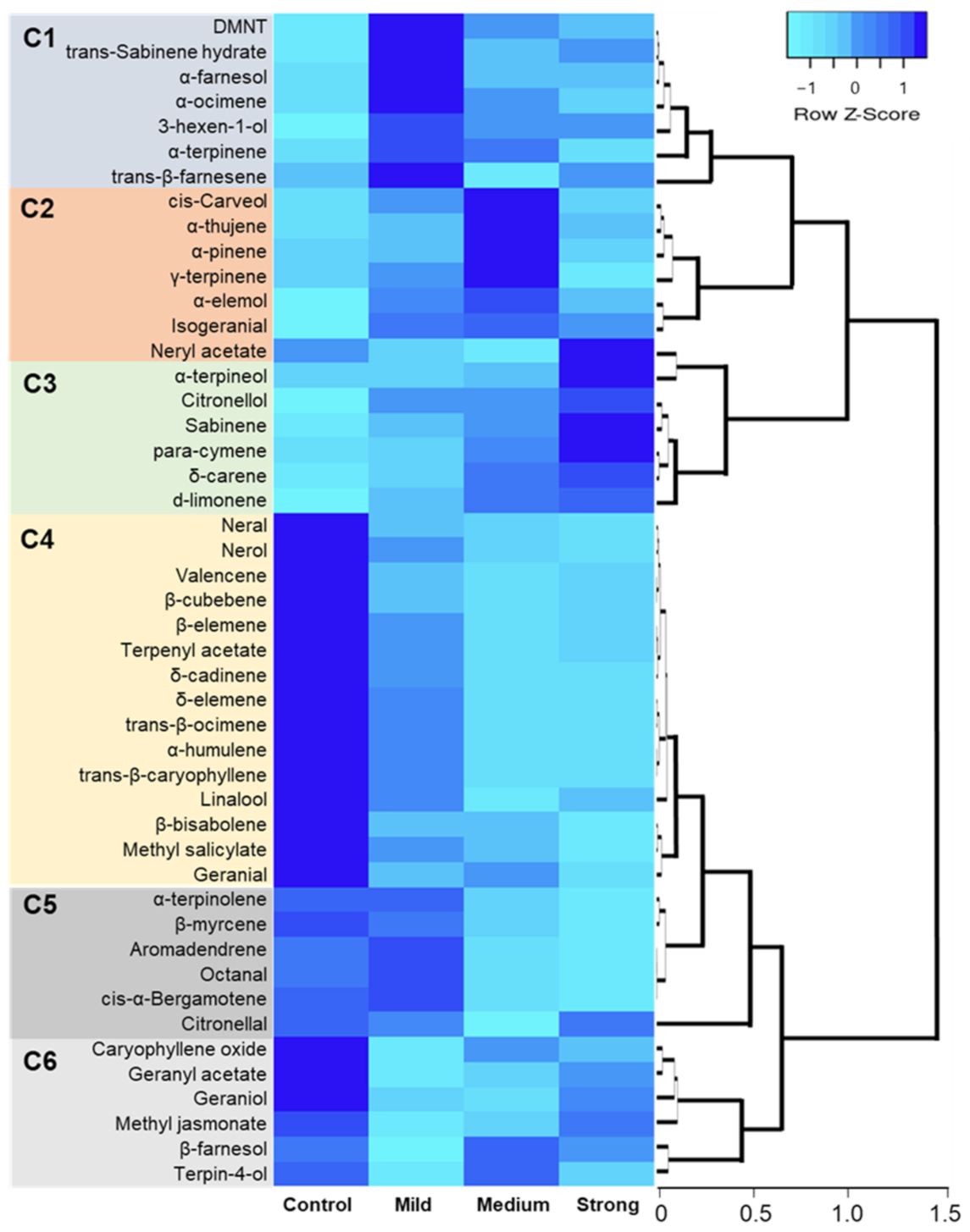
2.4. Hierarchical Clustering, Heat Map, and Simple Linear Regression
3. Discussion
4. Materials and Methods
4.1. Production of CTV-wt and CTV-tPDS Plants
4.2. Plant Maintenance
4.3. In Vivo VOC Collection
4.4. GS
4.5. Statistical Analysis
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hajeri, S.; Killiny, N.; El-Mohtar, C.; Dawson, W.O.; Gowda, S. Citrus tristeza virus-based RNAi in citrus plants induces gene silencing in Diaphorina citri, a phloem-sap sucking insect vector of citrus greening disease (Huanglongbing). J. Biotechnol. 2014, 176, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Killiny, N.; Gonzalez-Blanco, P.; Gowda, S.; Martini, X.; Etxeberria, E. Plant Functional Genomics in A Few Days: Laser-Assisted Delivery of Double-Stranded RNA to Higher Plants. Plants 2021, 10, 93. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, M.T.; Voinnet, O.; Baulcombe, D.C. Initiation and maintenance of virus-induced gene silencing. Plant Cell 1998, 10, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Maffei, M.E. Sites of synthesis, biochemistry and functional role of plant volatiles. S. Afr. J. Bot. 2010, 76, 612–631. [Google Scholar] [CrossRef] [Green Version]
- DellaPenna, D.; Pogson, B.J. Vitamin synthesis in plants: Tocopherols and carotenoids. Annu. Rev. Plant Biol. 2006, 57, 711–738. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartley, G.E.; Scolnik, P.A. Plant carotenoids—Pigments for photoprotection, visual attraction, and human health. Plant Cell 1995, 7, 1027–1038. [Google Scholar] [CrossRef] [PubMed]
- Ratcliff, F.; Martin-Hernandez, A.M.; Baulcombe, D.C. Tobacco rattle virus as a vector for analysis of gene function by silencing. Plant J. 2001, 25, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Killiny, N.; Nehela, Y.; Hijaz, F.; Ben-Mahmoud, S.K.; Hajeri, S.; Gowda, S. Citrus tristeza virus-based induced gene silencing of phytoene desaturase is more efficient when antisense orientation is used. Plant Biotechnol. Rep. 2019, 13, 179–192. [Google Scholar] [CrossRef]
- Killiny, N. Shrink the giant: Scale down the citrus tree to a model system to investigate the RNA interference efficiency. Plant Signal. Behav. 2019, 14, 1612681. [Google Scholar] [CrossRef] [PubMed]
- Killiny, N.; Nehela, Y.; George, J.; Rashidi, M.; Stelinski, L.L.; Lapointe, S.L. Phytoene desaturase-silenced citrus as a trap crop with multiple cues to attract Diaphorina citri, the vector of Huanglongbing. Plant Sci. 2021, 308, 110930. [Google Scholar] [CrossRef]
- Engelberth, J.; Alborn, H.T.; Schmelz, E.A.; Tumlinson, J.H. Airborne signals prime plants against insect herbivore attack. Proc. Natl. Acad. Sci. USA 2004, 101, 1781–1785. [Google Scholar] [CrossRef] [Green Version]
- Dudareva, N.; Klempien, A.; Muhlemann, J.K.; Kaplan, I. Biosynthesis, function and metabolic engineering of plant volatile organic compounds. New Phytol. 2013, 198, 16–32. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, M.H.; Donson, J.; Dellacioppa, G.; Harvey, D.; Hanley, K.; Grill, L.K. Cytoplasmic inhibition of carotenoid biosynthesis with virus-derived RNA. Proc. Natl. Acad. Sci. USA 1995, 92, 1679–1683. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romero, I.; Tikunov, Y.; Bovy, A. Virus-induced gene silencing in detached tomatoes and biochemical effects of phytoene desaturase gene silencing. J. Plant Physiol. 2011, 168, 1129–1135. [Google Scholar] [CrossRef]
- Zhang, X.H.; Ji, N.N.; Min, D.D.; Shu, P.; Cui, X.X.; Zhou, J.X.; Dong, L.L.; Ren, C.T.; Li, F.J.; Li, J.; et al. A co-silencing system for functional analysis of genes without visible phenotype in tomato plant development and fruit ripening using tobacco rattle virus. Sci. Hortic. 2018, 241, 100–106. [Google Scholar] [CrossRef]
- Agüero, J.; Vives, M.d.C.; Velázquez, K.; Pina, J.A.; Navarro, L.; Moreno, P.; Guerri, J. Effectiveness of gene silencing induced by viral vectors based on Citrus leaf blotch virus is different in Nicotiana benthamiana and citrus plants. Virology 2014, 460–461, 154–164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lewinsohn, E.; Sitrit, Y.; Bar, E.; Azulay, Y.; Meir, A.; Zamir, D.; Tadmor, Y. Carotenoid Pigmentation Affects the Volatile Composition of Tomato and Watermelon Fruits, As Revealed by Comparative Genetic Analyses. J. Agric. Food Chem. 2005, 53, 3142–3148. [Google Scholar] [CrossRef] [PubMed]
- Killiny, N.; Jones, S.E. Profiling of volatile organic compounds released from individual intact juvenile and mature citrus leaves. J. Plant Physiol. 2017, 208, 47–51. [Google Scholar] [CrossRef] [PubMed]




| Compound | RT | Compound | Identifier ions (m/z) | RI | Chemical formula | Pathway | Terpene class | SLR equation | p-value |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 6.00 | 3-Hexen-1-ol a | 67, 82, (100) | 838 | C6H12O | LOX | Green leaf volatile | Y = 0.6615007 + 1.5039 × 10−6 × X | 0.6371 |
| 2 | 7.66 | α-thujene a | 93, (136) | 900 | C10H16 | MEP | Monoterpene | Y = −1.124171 + 4.2505 × 10−8 × X | 0.5255 |
| 3 | 7.83 | α-pinene b | 77, (136) | 946 | C10H16 | MEP | Monoterpene | Y = 0.5354597 + 5.293 × 10−9 × X | 0.8134 |
| 4 | 8.74 | sabinene b | 93, (136) | 962 | C10H16 | MEP | Monoterpene | Y = −2.043718 + 6.3962 × 10−8 × X | 0.0239 |
| 5 | 9.12 | β-myrcene b | 77, (136) | 989 | C10H16 | MEP | Monoterpene | Y = 5.4066127 − 5.6089× 10−9 × X | 0.0151 |
| 6 | 9.40 | octanal b | 56, 84, (128) | 1008 | C8H16O | LOX | Aliphatic aldehyde | Y = 9.7890063 − 1.1892 × 10−7 × X | 0.1339 |
| 7 | 9.58 | δ-carene b | 93, (136) | 1009 | C10H16 | MEP | Monoterpene | Y = −1.024644 + 4.9396 × 10−8 × X | 0.0046 |
| 8 | 9.78 | α-terpinene b | 121, (136) | 1018 | C10H16 | MEP | Monoterpene | Y = 1.6879193 − 3.3425 × 10−9 × X | 0.9665 |
| 9 | 9.95 | para-cymene b | 119, (134) | 1026 | C10H14 | MEP | Monoterpene | Y = −0.309639 + 9.4336 × 10−9 × X | 0.0256 |
| 10 | 10.08 | d-limonene b | 68, (136) | 1038 | C10H16 | MEP | Monoterpene | Y = −5.256629 + 1.2465 × 10−9 × X | 0.0296 |
| 11 | 10.42 | trans-β-ocimene b | 107, (136) | 1055 | C10H16 | MEP | Monoterpene | Y = 5.4322736 − 6.2774 × 10−9 × X | 0.0303 |
| 12 | 10.72 | γ-terpinene b | 93, 121, (136) | 1071 | C10H16 | MEP | Monoterpene | Y = 1.6124691 − 7.942 × 10−11 × X | 0.9824 |
| 13 | 10.99 | trans-sabinene hydrate a | 93, (154) | 1081 | C10H18O | MEP | Monoterpene | Y = 0.9934156 + 4.3591 × 10−8 × X | 0.5838 |
| 14 | 11.34 | α-terpinolene b | 93, (136) | 1101 | C10H16 | MEP | Monoterpene | Y = 5.3820149 − 3.3693 × 10−8 × X | 0.0450 |
| 15 | 11.62 | linalool b | 71, 80, (154) | 1114 | C10H18O | MEP | Monoterpene alcohol | Y = 5.8499642 − 1.2174 × 10−8 × X | 0.1903 |
| 16 | 11.92 | DMNT b | 135, (150) | 1131 | C11H18 | MVA | Homoterpene | Y = 1.3459995 + 8.853 × 10−10 × X | 0.9179 |
| 17 | 12.25 | α-ocimene a | 93, (136) | 1152 | C10H16 | MEP | Monoterpene | Y = 2.0591518 − 1.3416 × 10−8 × X | 0.8609 |
| 18 | 12.75 | citronellal b | 69, (154) | 1178 | C10H18O | MEP | Monoterpene aldehyde | Y = 2.8260106 − 9.765 × 10−10 × X | 0.6888 |
| 19 | 13.44 | isogeranial a | 81, (152) | 1211 | C10H16O | MEP | Monoterpene aldehyde | Y = −1.524911 + 6.9897 × 10−8 × X | 0.7376 |
| 20 | 13.55 | Terpin-4-ol b | 71, 111, (154) | 1225 | C10H18O | MEP | Monoterpene alcohol | Y = 3.0069503 − 1.3479 × 10−7 × X | 0.7797 |
| 21 | 13.71 | methyl salicylate b | 120, (152) | 1228 | C8H8O3 | Shikimate | Benzenoid | Y = 5.6708351 − 1.4192 × 10−7 × X | 0.0363 |
| 22 | 13.82 | α-terpineol b | 59, (136) | 1233 | C10H18O | MEP | Monoterpene aldehyde | Y = 0.5691447 + 1.0031 × 10−7 × X | 0.2207 |
| 23 | 14.31 | nerol b | 69, (154) | 1264 | C10H18O | MEP | Monoterpene alcohol | Y = 4.1787856 − 1.3466 × 10−7 × X | 0.0885 |
| 24 | 14.35 | citronellol b | 69, (156) | 1267 | C10H20O | MEP | Monoterpene alcohol | Y = −2.214841 + 8.4327 × 10−8 × X | 0.0467 |
| 25 | 14.42 | cis-carveol b | 109, (152) | 1275 | C10H16O | MEP | Monoterpene alcohol | Y = 0.6619045 + 4.0657 × 10−8 × X | 0.6612 |
| 26 | 14.56 | neral b | 69, (152) | 1280 | C10H16O | MEP | Monoterpene aldehyde | Y = 4.2283559 − 3.3036 × 10−9 × X | 0.1167 |
| 27 | 14.86 | geraniol b | 69, (154) | 1292 | C10H18O | MEP | Monoterpene alcohol | Y = 2.1508954 − 2.2447 × 10−8 × X | 0.4888 |
| 28 | 15.16 | geranial b | 69, (152) | 1308 | C10H16O | MEP | Monoterpene aldehyde | Y = 4.2872926 − 2.9944 × 10−9 × X | 0.1342 |
| 29 | 16.56 | δ-elemene a | 121, (204) | 1360 | C15H24 | MVA | Sesquiterpene | Y = 3.5948159 − 1.1139 × 10−8 × X | 0.0405 |
| 30 | 16.75 | terpenyl acetate b | 121, 136, (181) | 1366 | C12H20O2 | MEP | Monoterpene Ester | Y = 4.5742907 − 1.2065 × 10−7 × X | 0.1008 |
| 31 | 16.91 | neryl acetate b | 69, (196) | 1373 | C12H20O2 | MEP | Monoterpene Ester | Y = 0.6446098 + 8.1427 × 10−8 × X | 0.5523 |
| 32 | 17.30 | geranyl acetate b | 69, (196) | 1378 | C12H20O2 | MEP | Monoterpene Ester | Y = 3.2687338 − 3.1068 × 10−8 × X | 0.5486 |
| 33 | 17.59 | β-elemene b | 93, (189) | 1384 | C15H24 | MVA | Sesquiterpene | Y = 3.7802501 − 3.4001 × 10−8 × X | 0.1234 |
| 34 | 18.22 | trans-β-caryophyllene b | 133, (204) | 1461 | C15H24 | MVA | Sesquiterpene | Y = 4.7685504 − 2.6421 × 10−9 × X | 0.0789 |
| 35 | 18.35 | aromadendrene a | 161, (204) | 1476 | C15H24 | MVA | Sesquiterpene | Y = 4.4307055 − 2.1659 × 10−8 × X | 0.1442 |
| 36 | 18.40 | cis-α-bergamotene a | 119, (204) | 1482 | C15H24 | MVA | Sesquiterpene | Y = 4.4877318 − 8.7324 × 10−9 × X | 0.1192 |
| 37 | 18.77 | trans-β-farnesene b | 93, (204) | 1505 | C15H24 | MVA | Sesquiterpene | Y = 1.9219621 − 4.4732 × 10−8 × X | 0.8137 |
| 38 | 18.85 | α-humulene b | 93, (204) | 1511 | C15H24 | MVA | Sesquiterpene | Y = 4.0697399 − 2.3463 × 10−8 × X | 0.0814 |
| 39 | 19.30 | β-cubebene a | 161, (204) | 1526 | C15H24 | MVA | Sesquiterpene | Y = 3.184197 − 1.9774 × 10−8 × X | 0.1491 |
| 40 | 19.55 | valencene b | 189, (204) | 1535 | C15H24 | MVA | Sesquiterpene | Y = 3.2005192 − 1.1617 × 10−8 × X | 0.1518 |
| 41 | 19.69 | β-bisabolene b | 93, (204) | 1541 | C15H24 | MVA | Sesquiterpene | Y = 4.3461062 − 3.8601 × 10−8 × X | 0.0665 |
| 42 | 19.91 | δ-cadinene a | 93, (204) | 1549 | C15H24 | MVA | Sesquiterpene | Y = 4.5764693 − 1.2446 × 10−7 × X | 0.0784 |
| 43 | 20.44 | α-elemol a | 59, 93, (204) | 1563 | C15H26O | MVA | Sesquiterpene alcohol | Y = −2.631255 + 1.2806 × 10−6 × X | 0.5189 |
| 44 | 21.07 | caryophyllene oxide b | 79, 93, (220) | 1589 | C15H24O | MVA | Sesquiterpenoid | Y = 4.5729864 − 3.0942 × 10−7 × X | 0.5161 |
| 45 | 21.89 | methyl jasmonate b | 84, 151, (224) | 1665 | C13H20O3 | LOX | Jasmonates | Y = 1.8891837 − 1.0704 × 10−7 × X | 0.9031 |
| 46 | 22.69 | β-farnesol b | 69, (222) | 1725 | C15H26O | MVA | Sesquiterpene alcohol | Y = 0.5641273 + 5.0138 × 10−7 × X | 0.9155 |
| 47 | 23.19 | α-farnesol b | 69, (222) | 1740 | C15H26O | MVA | Sesquiterpene alcohol | Y = 1.5524536 − 1.7063 × 10−7 × X | 0.9731 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Killiny, N. Silencing Phytoene Desaturase Causes Alteration in Monoterpene Volatiles Belonging to the Methylerythritol Phosphate Pathway. Plants 2022, 11, 276. https://doi.org/10.3390/plants11030276
Killiny N. Silencing Phytoene Desaturase Causes Alteration in Monoterpene Volatiles Belonging to the Methylerythritol Phosphate Pathway. Plants. 2022; 11(3):276. https://doi.org/10.3390/plants11030276
Chicago/Turabian StyleKilliny, Nabil. 2022. "Silencing Phytoene Desaturase Causes Alteration in Monoterpene Volatiles Belonging to the Methylerythritol Phosphate Pathway" Plants 11, no. 3: 276. https://doi.org/10.3390/plants11030276
APA StyleKilliny, N. (2022). Silencing Phytoene Desaturase Causes Alteration in Monoterpene Volatiles Belonging to the Methylerythritol Phosphate Pathway. Plants, 11(3), 276. https://doi.org/10.3390/plants11030276






