Phragmites australis (Cav.) Trin. ex Steud. Extract Induces Apoptosis-like Programmed Cell Death in Acanthamoeba castellanii Trophozoites
Abstract
1. Introduction
2. Results
2.1. PAE Showed Amoebicidal Activity against A. castellanii and A. polyphaga
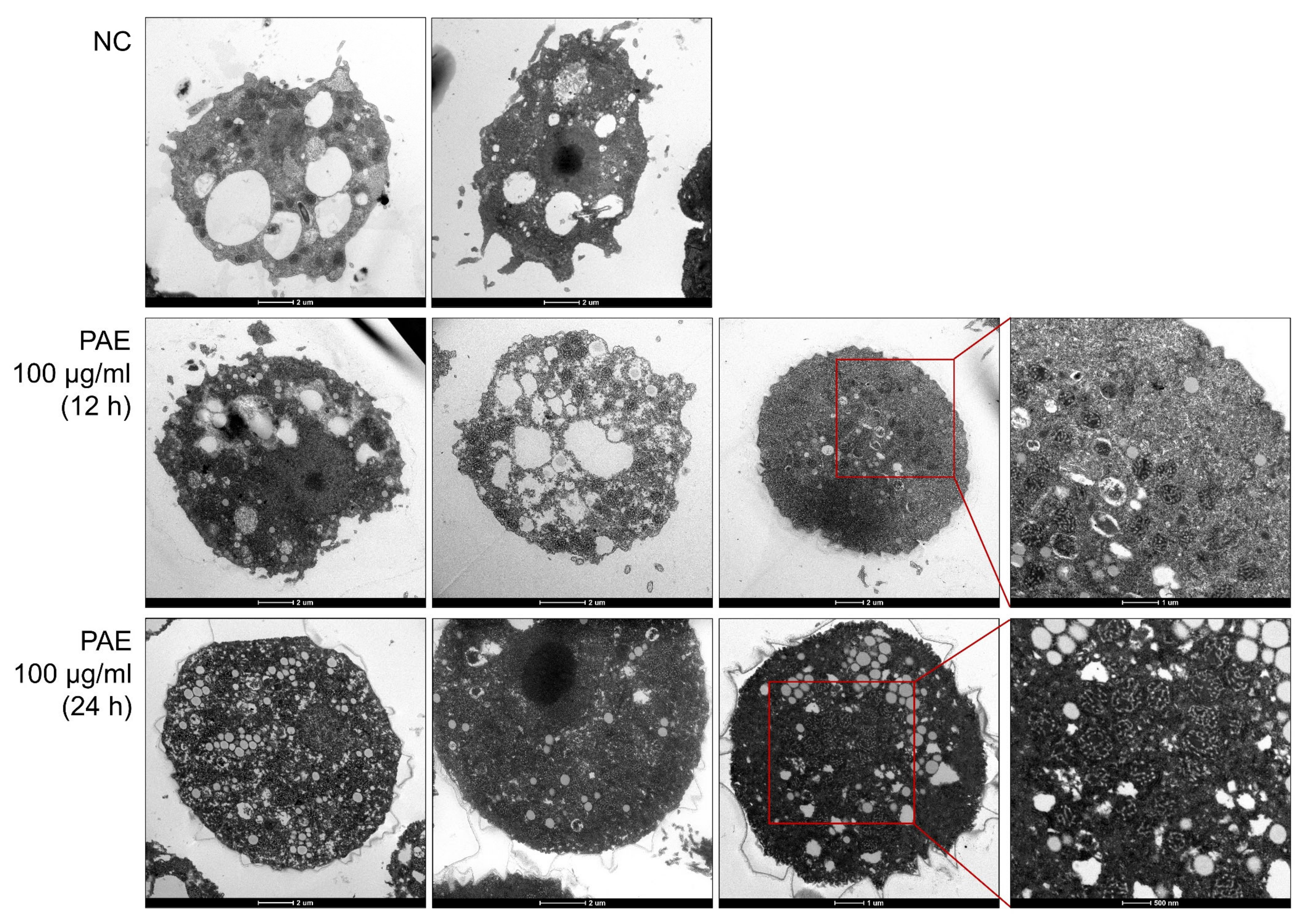
2.2. PAE Induced Changes in the Ultrastructural Morphology of A. castellanii
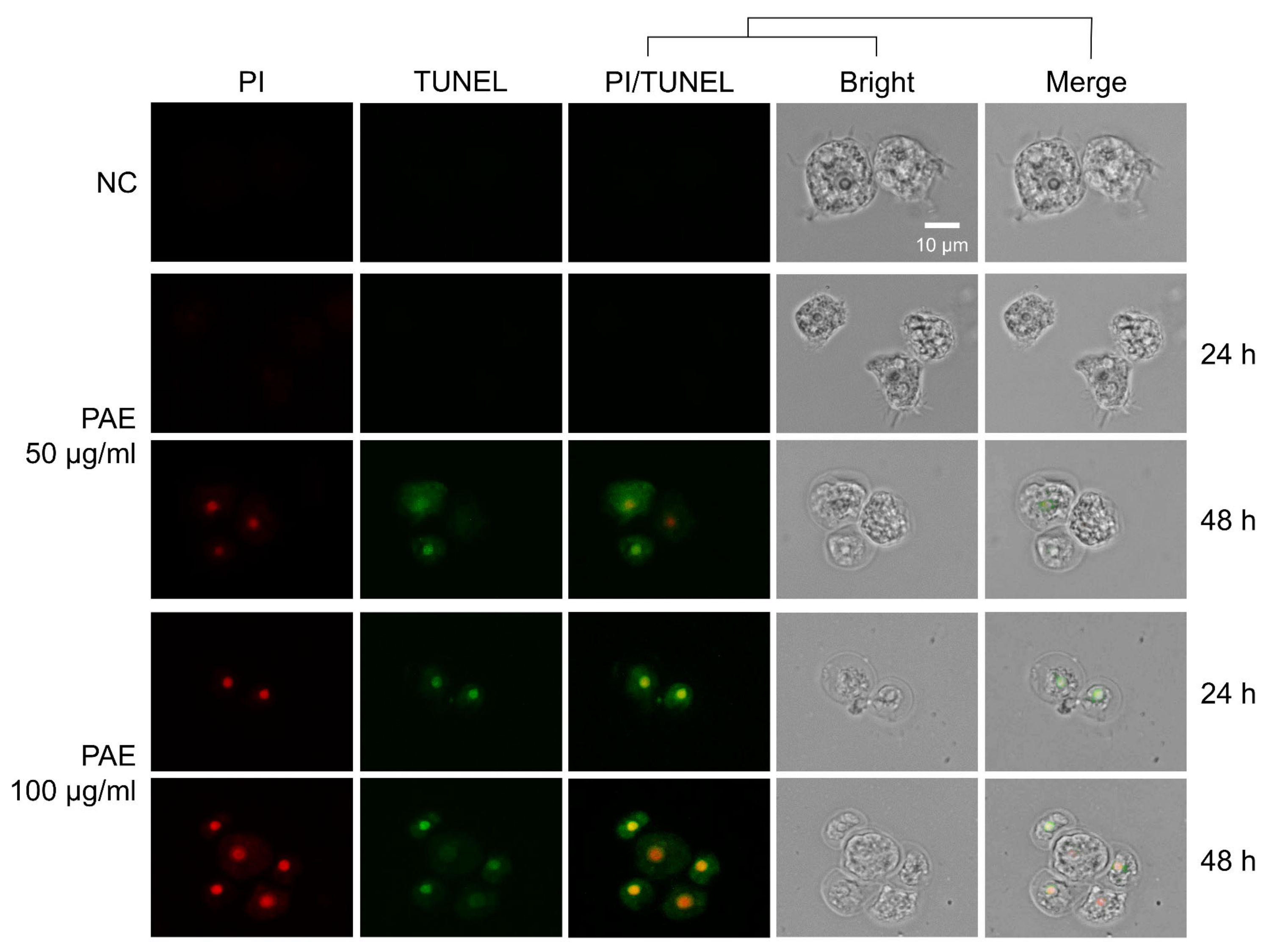
2.3. PAE Induced Apoptotic Death in A. castellanii
2.4. PAE Induced the Disruption of Mitochondrial Function in A. castellanii
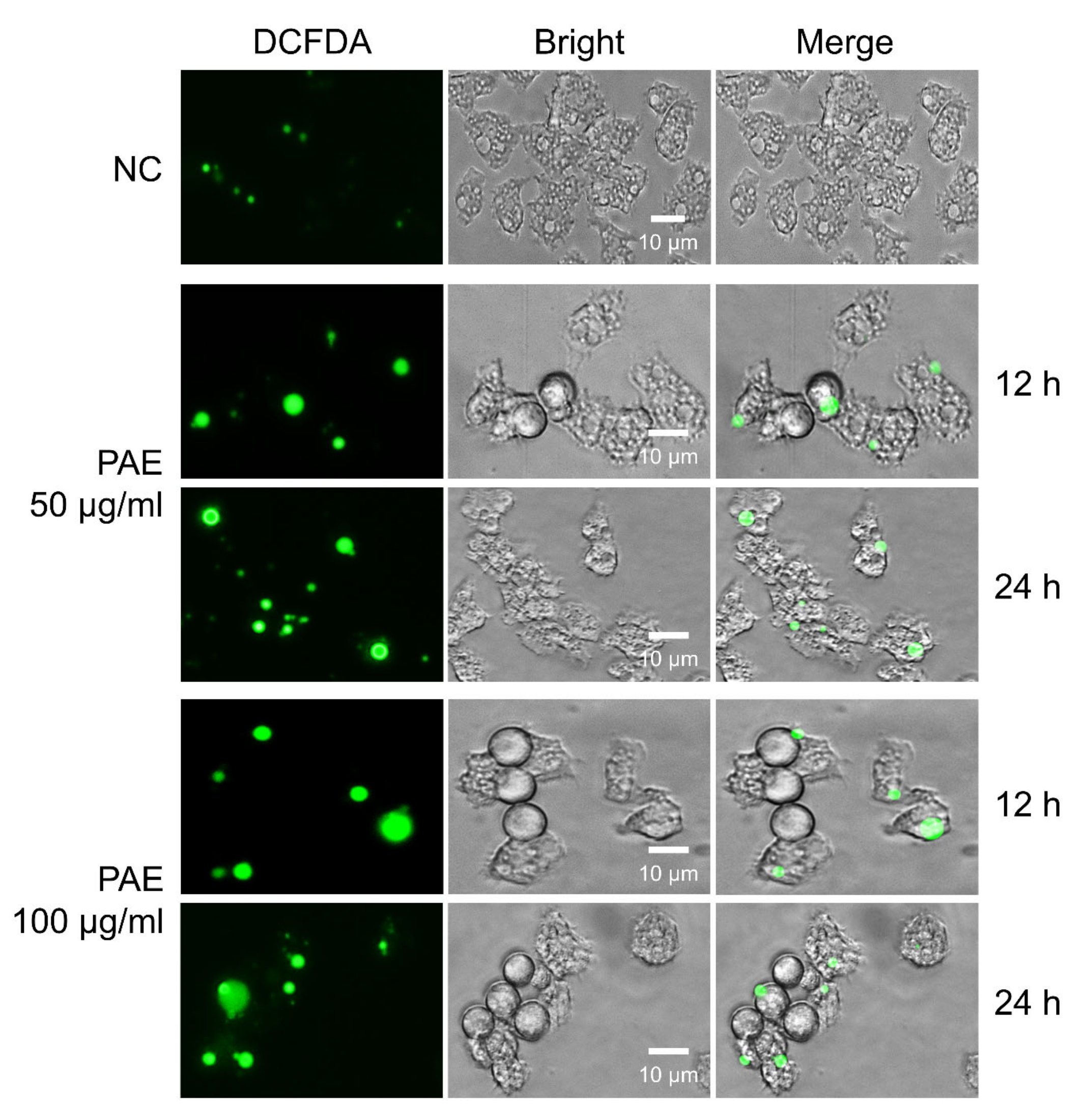
2.5. PAE Induced Increased Intracellular ROS Production in A. castellanii
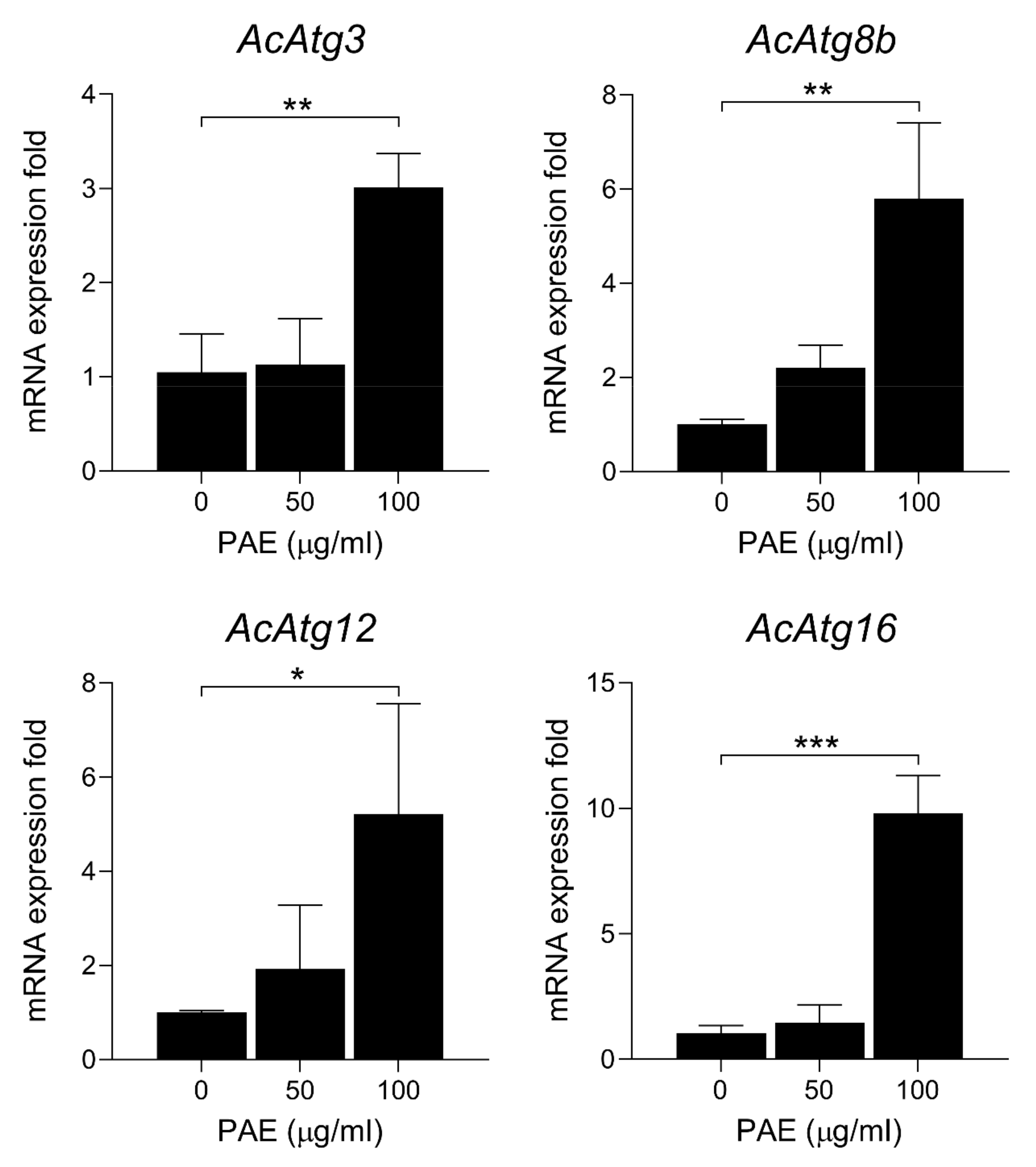
2.6. PAE Induced Increased Expressions of Autophagy Markers
3. Discussion
4. Materials and Methods
4.1. Preparation of PAE
4.2. Cultivation of Acanthamoeba and Human Cell Lines
4.3. Amoebicidal Assay
4.4. Cytotoxicity Assay for HCE-2 and ARPE-19 Cells
4.5. Transmission Electron Microscopy (TEM)
4.6. Fluorometric Apoptosis/Necrosis Assay
4.7. TUNEL Assay
4.8. Mitochondrial Membrane Potential Assay
4.9. Measurement of ATP Levels
4.10. Intracellular Reactive Oxygen Species (ROS) Assay
4.11. Quantitative Polymerase Chain Reaction (qPCR) for Transcripts of Autophagy-Related Genes
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Greub, G.; Raoult, D. Microorganisms resistant to free-living amoebae. Clin. Microbiol. Rev. 2004, 17, 413–433. [Google Scholar] [CrossRef] [PubMed]
- Samba-Louaka, A.; Robino, E.; Cochard, T.; Branger, M.; Delafont, V.; Aucher, W.; Wambeke, W.; Bannantine, J.P.; Biet, F.; Héchard, Y. Environmental Mycobacterium avium subsp. paratuberculosis hosted by free-living amoebae. Front. Cell. Infect. Microbiol. 2018, 8, 28. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo-Morales, J.; Martín-Navarro, C.M.; López-Arencibia, A.; Arnalich-Montiel, F.; Piñero, J.E.; Valladares, B. Acanthamoeba keratitis: An emerging disease gathering importance worldwide? Trends Parasitol. 2013, 29, 181–187. [Google Scholar] [CrossRef]
- De Lacerda, A.G.; Lira, M. Acanthamoeba keratitis: A review of biology, pathophysiology and epidemiology. Ophthalmic Physiol. Opt. 2021, 41, 116–135. [Google Scholar] [CrossRef]
- Jain, R.; Garg, P.; Motukupally, S.R.; Geary, M.B. Clinico-microbiological review of non-contact-lens-associated acanthamoeba keratitis. Semin. Ophthalmol. 2015, 30, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Bunsuwansakul, C.; Mahboob, T.; Hounkong, K.; Laohaprapanon, S.; Chitapornpan, S.; Jawjit, S.; Yasiri, A.; Barusrux, S.; Bunluepuech, K.; Sawangjaroen, N.; et al. Acanthamoeba in southeast Asia—Overview and challenges. Korean J. Parasitol. 2019, 57, 341–357. [Google Scholar] [CrossRef] [PubMed]
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba spp. as agents of disease in humans. Clin. Microbiol. Rev. 2003, 16, 273–307. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Gorski, M.; Henderson, T.; Lazzaro, D.; Haseeb, M.A. Clinical course of unilateral Acanthamoeba keratitis in a cosmetic contact lens wearer. Ann. Clin. Lab. Sci. 2015, 45, 366–370. [Google Scholar] [PubMed]
- Huang, F.C.; Shih, M.H.; Chang, K.F.; Huang, J.M.; Shin, J.W.; Lin, W.C. Characterizing clinical isolates of Acanthamoeba castellanii with high resistance to polyhexamethylene biguanide in Taiwan. J. Microbiol. Immunol. Infect. 2017, 50, 570–577. [Google Scholar] [CrossRef]
- Iovieno, A.; Oechsler, R.A.; Ledee, D.R.; Miller, D.; Alfonso, E.C. Drug-resistant severe acanthamoeba keratitis caused by rare T5 acanthamoeba genotype. Eye Contact Lens 2010, 36, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Yanai, R.; Yamada, N.; Ueda, K.; Tajiri, M.; Matsumoto, T.; Kido, K.; Nakamura, S.; Saito, F.; Nishida, T. Evaluation of povidone-iodine as a disinfectant solution for contact lenses: Antimicrobial activity and cytotoxicity for corneal epithelial cells. Contact Lens Anterior Eye 2006, 29, 85–91. [Google Scholar] [CrossRef]
- Lorenzo-Morales, J.; Khan, N.A.; Walochnik, J. An update on Acanthamoeba keratitis: Diagnosis, pathogenesis and treatment. Parasite 2015, 22, 10. [Google Scholar] [CrossRef]
- Szentmáry, N.; Daas, L.; Shi, L.; Laurik, K.L.; Lepper, S.; Milioti, G.; Seitz, B. Acanthamoeba keratitis—Clinical signs, differential diagnosis and treatment. J. Curr. Ophthalmol. 2019, 31, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Clardy, J.; Walsh, C. Lessons from natural molecules. Nature 2004, 432, 829–837. [Google Scholar] [CrossRef]
- Polat, Z.A.; Vural, A.; Ozan, F.; Tepe, B.; Özcelik, S.; Cetin, A. In vitro evaluation of the amoebicidal activity of garlic (Allium sativum) extract on Acanthamoeba castellanii and its cytotoxic potential on corneal cells. J. Ocul. Pharmacol. Ther. 2008, 24, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Shoaib, H.M.; Muazzam, A.G.; Mir, A.; Jung, S.Y.; Matin, A. Evaluation of inhibitory potential of some selective methanolic plants extracts on biological characteristics of Acanthamoeba castellanii using human corneal epithelial cells in vitro. Parasitol. Res. 2013, 112, 1179–1788. [Google Scholar] [CrossRef] [PubMed]
- Boonhok, R.; Sangkanu, S.; Norouzi, R.; Siyadatpanah, A.; Mirzaei, F.; Mitsuwan, W.; Charong, N.; Wisessombat, S.; Pereira, M.D.L.; Rahmatullah, M.; et al. Amoebicidal activity of Cassia angustifolia extract and its effect on Acanthamoeba triangularis autophagy-related gene expression at the transcriptional level. Parasitology 2021, 148, 1074–1082. [Google Scholar] [CrossRef] [PubMed]
- Sangkanu, S.; Mitsuwan, W.; Mahabusarakam, W.; Jimoh, T.O.; Wilairatana, P.; Girol, A.P.; Verma, A.K.; de Lourdes Pereira, M.; Rahmatullah, M.; Wiart, C.; et al. Anti-Acanthamoeba synergistic effect of chlorhexidine and Garcinia mangostana extract or α-mangostin against Acanthamoeba triangularis trophozoite and cyst forms. Sci. Rep. 2021, 11, 8053. [Google Scholar] [CrossRef] [PubMed]
- Maliński, M.P.; Budzianowski, J.; Kikowska, M.; Derda, M.; Jaworska, M.M.; Mlynarczyk, D.T.; Szukalska, M.; Florek, E.; Thiem, B. Two ecdysteroids isolated from micropropagated lychnis flos-cuculi and the biological activity of plant. Molecules 2021, 26, 904. [Google Scholar] [CrossRef] [PubMed]
- Mitsuwan, W.; Sangkanu, S.; Romyasamit, C.; Kaewjai, C.; Jimoh, T.O.; de Lourdes Pereira, M.; Siyadatpanah, A.; Kayesth, S.; Nawaz, M.; Rahmatullah, M.; et al. Curcuma longa rhizome extract and Curcumin reduce the adhesion of Acanthamoeba triangularis trophozoites and cysts in polystyrene plastic surface and contact lens. Int. J. Parasitol. Drugs Drug Resist. 2020, 14, 218–229. [Google Scholar] [CrossRef]
- Fakae, L.B.; Stevenson, C.W.; Zhu, X.Q.; Elsheikha, H.M. In vitro activity of Camellia sinensis (green tea) against trophozoites and cysts of Acanthamoeba castellanii. Int. J. Parasitol. Drugs Drug Resist. 2020, 13, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Nayeri, T.; Bineshian, F.; Khoshzaban, F.; Asl, A.D.; Ghaffarifar, F. Evaluation of the effects of Rumex obtusifolius seed and leaf extracts against Acanthamoeba: An in vitro study. Infect. Disord. Drug Targets 2020, 21, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Sifaoui, I.; Rodríguez-Expósito, R.L.; Reyes-Batlle, M.; Rizo-Liendo, A.; Piñero, J.E.; Bazzocchi, I.L.; Lorenzo-Morales, J.; Jiménez, I.A. Ursolic acid derivatives as potential agents against Acanthamoeba Spp. Pathogens 2019, 8, 130. [Google Scholar] [CrossRef] [PubMed]
- Sifaoui, I.; López-Arencibia, A.; Martín-Navarro, C.M.; Reyes-Batlle, M.; Wagner, C.; Chiboub, O.; Mejri, M.; Valladares, B.; Abderrabba, M.; Piñero, J.E.; et al. Programmed cell death in Acanthamoeba castellanii Neff induced by several molecules present in olive leaf extracts. PLoS ONE 2017, 12, e0183795. [Google Scholar] [CrossRef]
- Mahboob, T.; Azlan, A.M.; Shipton, F.N.; Boonroumkaew, P.; Nor Azman, N.S.; Sekaran, S.D.; Ithoi, I.; Tan, T.C.; Samudi, C.; Wiart, C.; et al. Acanthamoebicidal activity of periglaucine A and betulinic acid from Pericampylus glaucus (Lam.) Merr. in vitro. Exp. Parasitol. 2017, 183, 160–166. [Google Scholar] [CrossRef]
- Clevering, O.A.; Brix, H.; Lukavská, J. Geographic variation in growth responses in Phragmites australis. Aquat. Bot. 2001, 69, 89–108. [Google Scholar] [CrossRef]
- Chen, Y.; Li, L.; Jiang, L.R.; Tan, J.Y.; Guo, L.N.; Wang, X.L.; Dong, W.; Wang, W.B.; Sun, J.K.; Song, B. Alkaloids constituents from the roots of Phragmites australis (Cav.) Trin. ex Steud. with their cytotoxic activities. Nat. Prod. Res. 2022, 36, 1454–1459. [Google Scholar] [CrossRef]
- Delfino, V.; Calonico, C.; Nostro, A.L.; Castronovo, L.M.; Del Duca, S.; Chioccioli, S.; Coppini, E.; Fibbi, D.; Vassallo, A.; Fani, R. Antibacterial activity of bacteria isolated from Phragmites australis against multidrug-resistant human pathogens. Future Microbiol. 2021, 16, 291–303. [Google Scholar] [CrossRef]
- Zhu, L.; Zhang, D.; Yuan, C.; Ding, X.; Shang, Y.; Jiang, Y.; Zhu, G. Anti-Inflammatory & antiviral effects of water-soluble crude extract from Phragmites australis in vitro. Pak. J. Pharm. Sci. 2017, 30, 1357–1362. [Google Scholar]
- Moon, E.K.; Kim, S.H.; Hong, Y.; Chung, D., II; Goo, Y.K.; Kong, H.H. Autophagy inhibitors as a potential antiamoebic treatment for Acanthamoeba keratitis. Antimicrob. Agents Chemother. 2015, 59, 4020–4025. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Ruiz, A.; Alzate, J.F.; MacLeod, E.T.; Lüder, C.G.K.; Fasel, N.; Hurd, H. Apoptotic markers in protozoan parasites. Parasites Vectors 2010, 3, 104. [Google Scholar] [CrossRef] [PubMed]
- Kaczanowski, S.; Sajid, M.; Reece, S.E. Evolution of apoptosis-like programmed cell death in unicellular protozoan parasites. Parasites Vectors 2011, 4, 44. [Google Scholar] [CrossRef] [PubMed]
- Deponte, M. Programmed cell death in protists. Biochim. Biophys. Acta Mol. Cell Res. 2008, 1783, 1396–1405. [Google Scholar] [CrossRef] [PubMed]
- Reece, S.E.; Pollitt, L.C.; Colegrave, N.; Gardner, A. The meaning of death: Evolution and ecology of apoptosis in protozoan parasites. PLoS Pathog. 2011, 7, e1002320. [Google Scholar] [CrossRef] [PubMed]
- Martín-Navarro, C.M.; López-Arencibia, A.; Sifaoui, I.; Reyes-Batlle, M.; Valladares, B.; Martínez-Carretero, E.; Piñero, J.E.; Maciver, S.K.; Lorenzo-Morales, J. Statins and voriconazole induce programmed cell death in Acanthamoeba castellanii. Antimicrob. Agents Chemother. 2015, 59, 2817–2824. [Google Scholar] [CrossRef]
- Zorova, L.D.; Popkov, V.A.; Plotnikov, E.Y.; Silachev, D.N.; Pevzner, I.B.; Jankauskas, S.S.; Babenko, V.A.; Zorov, S.D.; Balakireva, A.V.; Juhaszova, M.; et al. Mitochondrial membrane potential. Anal. Biochem. 2018, 552, 50–59. [Google Scholar] [CrossRef]
- Joshi, D.C.; Bakowska, J.C. Determination of mitochondrial membrane potential and reactive oxygen species in live rat cortical neurons. J. Vis. Exp. 2011, 51, 2704. [Google Scholar] [CrossRef]
- Rajasekaran, N.S.; Connell, P.; Christians, E.S.; Yan, L.J.; Taylor, R.P.; Orosz, A.; Zhang, X.Q.; Stevenson, T.J.; Peshock, R.M.; Leopold, J.A.; et al. Human alpha B-crystallin mutation causes oxido-reductive stress and protein aggregation cardiomyopathy in mice. Cell 2007, 130, 427–439. [Google Scholar] [CrossRef]
- Cartuche, L.; Sifaoui, I.; Cruz, D.; Reyes-Batlle, M.; López-Arencibia, A.; Javier Fernández, J.; Díaz-Marrero, A.R.; Piñero, J.E.; Lorenzo-Morales, J. Staurosporine from Streptomyces sanyensis activates programmed cell death in Acanthamoeba via the mitochondrial pathway and presents low in vitro cytotoxicity levels in a macrophage cell line. Sci. Rep. 2019, 9, 11651. [Google Scholar] [CrossRef]
- Koutsogiannis, Z.; MacLeod, E.T.; Maciver, S.K. G418 induces programmed cell death in Acanthamoeba through the elevation of intracellular calcium and cytochrome c translocation. Parasitol. Res. 2019, 118, 641–651. [Google Scholar] [CrossRef]
- Moon, E.K.; Chung, D., II; Hong, Y.C.; Kong, H.H. Autophagy protein 8 mediating autophagosome in encysting Acanthamoeba. Mol. Biochem. Parasitol. 2009, 168, 43–48. [Google Scholar] [CrossRef]
- Moon, E.K.; Chung, D.I.; Hong, Y.; Kong, H.H. Atg3-mediated lipidation of atg8 is involved in encystation of Acanthamoeba. Korean J. Parasitol. 2011, 49, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Song, S.M.; Han, B.I.; Moon, E.K.; Lee, Y.R.; Yu, H.S.; Jha, B.K.; Danne, D.B.S.; Kong, H.H.; Chung, D., II; Hong, Y. Autophagy protein 16-mediated autophagy is required for the encystation of Acanthamoeba castellanii. Mol. Biochem. Parasitol. 2012, 183, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Moon, E.K.; Hong, Y.; Chung, D., II; Kong, H.H. Identification of Atg8 isoform in encysting Acanthamoeba. Korean J. Parasitol. 2013, 51, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Boonhok, R.; Sangkanu, S.; Chuprom, J.; Srisuphanunt, M.; Norouzi, R.; Siyadatpanah, A.; Mirzaei, F.; Mitsuwan, W.; Wisessombat, S.; de Lourdes Pereira, M.; et al. Peganum harmala extract has antiamoebic activity to Acanthamoeba triangularis trophozoites and changes expression of autophagy-related genes. Pathogens 2021, 10, 842. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, Y.M.; Jung, S.Y.; Jang, G.S.; Kang, W.C.; Jung, S.S.; Oh, S.H.; Yang, J.C. Illustrated Grasses of Korea (Revised and Enlarged Edition) (Korean); Korea National Arboretum: Pocheon, Republic of Korea, 2011; pp. 374–375. [Google Scholar]
- Visvesvara, G.S.; Balamuth, W. Comparative studies on related free-living and pathogenic amebae with special reference to Acanthamoeba. J. Protozool. 1975, 22, 245–256. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lê, H.-G.; Choi, J.-S.; Hwang, B.-S.; Jeong, Y.-T.; Kang, J.-M.; Võ, T.-C.; Cho, P.-Y.; Lee, Y.-K.; Yoo, W.-G.; Hong, Y.; et al. Phragmites australis (Cav.) Trin. ex Steud. Extract Induces Apoptosis-like Programmed Cell Death in Acanthamoeba castellanii Trophozoites. Plants 2022, 11, 3459. https://doi.org/10.3390/plants11243459
Lê H-G, Choi J-S, Hwang B-S, Jeong Y-T, Kang J-M, Võ T-C, Cho P-Y, Lee Y-K, Yoo W-G, Hong Y, et al. Phragmites australis (Cav.) Trin. ex Steud. Extract Induces Apoptosis-like Programmed Cell Death in Acanthamoeba castellanii Trophozoites. Plants. 2022; 11(24):3459. https://doi.org/10.3390/plants11243459
Chicago/Turabian StyleLê, Hương-Giang, Ji-Su Choi, Buyng-Su Hwang, Yong-Tae Jeong, Jung-Mi Kang, Tuấn-Cường Võ, Pyo-Yun Cho, Young-Kyung Lee, Won-Gi Yoo, Yeonchul Hong, and et al. 2022. "Phragmites australis (Cav.) Trin. ex Steud. Extract Induces Apoptosis-like Programmed Cell Death in Acanthamoeba castellanii Trophozoites" Plants 11, no. 24: 3459. https://doi.org/10.3390/plants11243459
APA StyleLê, H.-G., Choi, J.-S., Hwang, B.-S., Jeong, Y.-T., Kang, J.-M., Võ, T.-C., Cho, P.-Y., Lee, Y.-K., Yoo, W.-G., Hong, Y., Oh, Y.-T., & Na, B.-K. (2022). Phragmites australis (Cav.) Trin. ex Steud. Extract Induces Apoptosis-like Programmed Cell Death in Acanthamoeba castellanii Trophozoites. Plants, 11(24), 3459. https://doi.org/10.3390/plants11243459








