Hippeastrum stapfianum (Kraenzl.) R.S.Oliveira & Dutilh (Amaryllidaceae) Ethanol Extract Activity on Acetylcholinesterase and PPAR-α/γ Receptors
Abstract
1. Introduction
2. Results
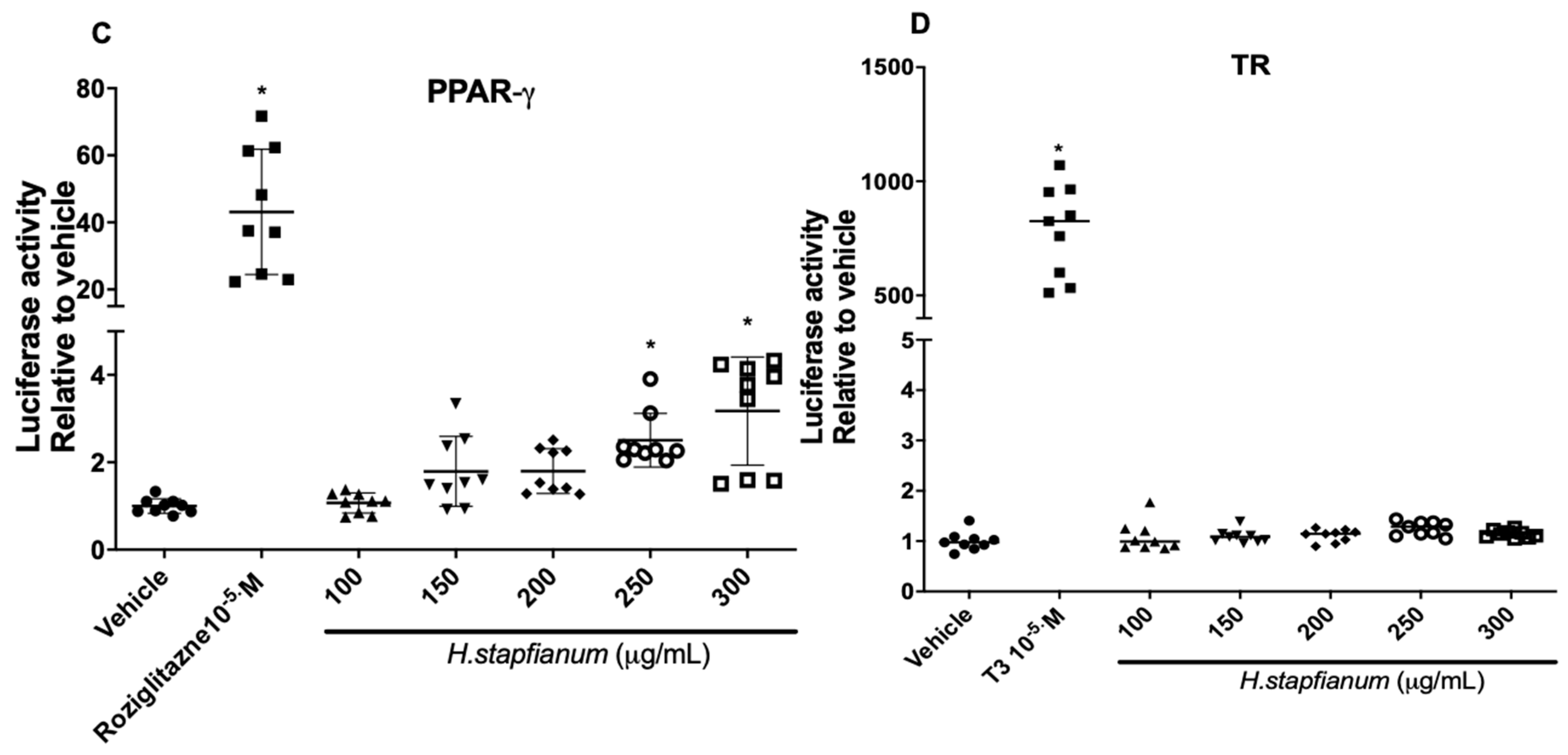
2.1. EE Upregulates the PPAR-α and PPAR-γ Receptor
2.2. EE Performs Antioxidant Capacity and Promotes Acetylcholinesterase Inhibition In Vitro
2.3. EE and Fractions Chemical Characterization by GC-MS Analysis and HPLC
3. Discussion
4. Materials and Methods
4.1. Hippeastrum stapfianum Plant Material
4.2. Hippeastrum stapfianum Extracts and Fractions
4.3. HeLa Cell Culture
4.4. Cell Viability by MTT Assay (Mitochondrial Activity)
4.5. Transfection and Luciferase Gene Reporter Assay
4.6. Acetylcholinesterase Inhibition
4.7. 2,2-Diphenyl-1-picrylhydrazyl (DPPH) Scavenging Assay
4.8. Fosfomolybdenium Reduction Assay
4.9. GC-MS Analysis
4.10. HPLC-DAD-UV Analysis
4.11. Statistical and Data Analysis
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Habartová, K.; Cahlíková, L.; Řezáčová, M.; Havelek, R. The biological activity of alkaloids from the Amaryllidaceae: From cholinesterases inhibition to anticancer activity. Nat. Prod. Commun. 2016, 11, 1587–1594. [Google Scholar] [CrossRef] [PubMed]
- Tallini, L.R.; Osorio, E.H.; Santos, V.D.D.; Borges, W.S.; Kaiser, M.; Viladomat, F.; Zuanazzi, J.A.S.; Bastida, J. Hippeastrum reticulatum (Amaryllidaceae): Alkaloid profiling, biological activities and molecular docking. Molecules 2017, 22, 2191. [Google Scholar] [CrossRef] [PubMed]
- Costa, G.G.P.; Silva, C.A.G.; Gomes, J.V.D.; Torres, A.G.; Santos, I.R.I.; Almeida, F.T.C.; Fagg, C.W.; Simeoni, L.A.; Silveira, D.; Gomes-Copeland, K.K.P. Influence of in vitro micropropagation on lycorine biosynthesis and anticholinesterase activity in Hippeastrum goianum. Rev. Bras. Farmacogn. 2019, 29, 262–265. [Google Scholar] [CrossRef]
- Colović, M.B.; Krstić, D.Z.; Lazarević-Pašti, T.D.; Bondžić, A.M.; Vasić, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013, 11, 315–335. [Google Scholar] [CrossRef]
- Paoletti, F.; Mocali, A.; Vannucchi, A. Acetylcholinesterase in murine erythroleukemia (Friend) cells: Evidence for megakaryocyte-like expression and potential growth-regulatory role of enzyme activity. Blood 1992, 79, 2873–2879. [Google Scholar] [CrossRef]
- Trang, A.; Khandhar, P.B. Physiology, Acetylcholinesterase; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Walczak-Nowicka, Ł.J.; Herbet, M. Acetylcholinesterase inhibitors in the treatment of neurodegenerative diseases and the role of acetylcholinesterase in their pathogenesis. Int. J. Mol. Sci. 2021, 22, 9290. [Google Scholar] [CrossRef]
- Khan, H.; Marya; Amin, S.; Kamal, M.A.; Patel, S. Flavonoids as acetylcholinesterase inhibitors: Current therapeutic standing and future prospects. Biomed. Pharmacother. 2018, 101, 860–870. [Google Scholar] [CrossRef] [PubMed]
- Katz, N.K.; Barohn, R.J. The history of acetylcholinesterase inhibitors in the treatment of myasthenia gravis. Neuropharmacology 2021, 182, 108303. [Google Scholar] [CrossRef]
- Ribaudo, G.; Memo, M.; Gianoncelli, A. Multi-target natural and nature-inspired compounds against neurodegeneration: A focus on dual cholinesterase and phosphodiesterase inhibitors. Appl. Sci. 2021, 11, 5044. [Google Scholar] [CrossRef]
- Saxena, M.; Dubey, R. Target enzyme in Alzheimer’s disease: Acetylcholinesterase inhibitors. Curr. Top. Med. Chem. 2019, 19, 264–275. [Google Scholar] [CrossRef]
- Lane, R.M.; Kivipelto, M.; Greig, N.H. Acetylcholinesterase and its inhibition in Alzheimer disease. Clin. Neuropharmacol. 2004, 27, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Wilcock, G.K.; Lilienfeld, S.; Gaens, E. Efficacy and safety of galantamine in patients with mild to moderate Alzheimer’s disease: Multicentre randomised controlled trial. BMJ 2000, 321, 1445–1449. [Google Scholar] [CrossRef] [PubMed]
- Maelicke, A.; Samochocki, M.; Jostock, R.; Fehrenbacher, A.; Ludwig, J.; Albuquerque, E.X.; Zerlin, M. Allosteric sensitization of nicotinic receptors by galantamine, a new treatment strategy for Alzheimer’s disease. Biol. Psychiatry 2001, 49, 279–288. [Google Scholar] [CrossRef]
- WHO. Global Status Report on the Public Health Response to Dementia; World Health Organization: Geneva, Switzerland, 2021; p. 137.
- ALZ. 2018 Alzheimer’s disease facts and figures. Alzheimer’s Dement. 2018, 14, 367–429. [Google Scholar] [CrossRef]
- Neels, J.G.; Grimaldi, P.A. Physiological functions of peroxisome proliferator-activated receptor β. Physiol. Rev. 2014, 94, 795–858. [Google Scholar] [CrossRef] [PubMed]
- Govindarajulu, M.; Pinky, P.D.; Bloemer, J.; Ghanei, N.; Suppiramaniam, V.; Amin, R. Signaling Mechanisms of selective PPARgamma modulators in Alzheimer’s disease. PPAR Res. 2018, 2018, 2010675. [Google Scholar] [CrossRef] [PubMed]
- Hamano, T.; Shirafuji, N.; Makino, C.; Yen, S.H.; Kanaan, N.M.; Ueno, A.; Suzuki, J.; Ikawa, M.; Matsunaga, A.; Yamamura, O.; et al. Pioglitazone prevents tau oligomerization. Biochem. Biophys. Res. Commun. 2016, 478, 1035–1042. [Google Scholar] [CrossRef]
- Wójtowicz, S.; Strosznajder, A.K.; Jeżyna, M.; Strosznajder, J.B. The novel role of PPAR alpha in the brain: Promising target in therapy of Alzheimer’s disease and other neurodegenerative disorders. Neurochem. Res. 2020, 45, 972–988. [Google Scholar] [CrossRef]
- Lemberger, T.; Desvergne, B.; Wahli, W. Peroxisome proliferator-activated receptors: A nuclear receptor signaling pathway in lipid physiology. Annu. Rev. Cell Dev. Biol. 1996, 12, 335–363. [Google Scholar] [CrossRef]
- Kleiboeker, B.; Lodhi, I.J. Peroxisomal regulation of energy homeostasis: Effect on obesity and related metabolic disorders. Mol. Metabol. 2022, 65, 101577. [Google Scholar] [CrossRef]
- Amato, A.A.; Rajagopalan, S.; Lin, J.Z.; Carvalho, B.M.; Figueira, A.C.; Lu, J.; Ayers, S.D.; Mottin, M.; Silveira, R.L.; Souza, P.C.; et al. GQ-16, a novel peroxisome proliferator-activated receptor gamma (PPARgamma) ligand, promotes insulin sensitization without weight gain. J. Biol. Chem. 2012, 287, 28169–28179. [Google Scholar] [CrossRef] [PubMed]
- Lima, C.L.; Amorim, B.R.; Royer, C.; Resende, A.P.; Borin, M.F.; Neves, F.A.R.; Acevedo, A.C. Investigation of PPARbeta/delta within human dental pulp cells: A preliminary in vitro study. PPAR Res. 2021, 2021, 8854921. [Google Scholar] [CrossRef] [PubMed]
- Cheignon, C.; Tomas, M.; Bonnefont-Rousselot, D.; Faller, P.; Hureau, C.; Collin, F. Oxidative stress and the amyloid beta peptide in Alzheimer’s disease. Redox Biol. 2018, 14, 450–464. [Google Scholar] [CrossRef] [PubMed]
- Papuć, E.; Rejdak, K. The role of myelin damage in Alzheimer’s disease pathology. Arch. Med. Sci. 2020, 16, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Ma, Z.; Qin, H.; Yao, Z. Thyroid hormone potentially benefits multiple sclerosis via facilitating remyelination. Mol. Neurobiol. 2016, 53, 4406–4416. [Google Scholar] [CrossRef] [PubMed]
- Gomes, J.V.D.; Tosta, C.L.; Cunha Neto, Á.; Fagg, C.W.; Silva, C.A.G.; Gomes-Copeland, K.K.P.; Magalhães, P.O.; Fonseca-Bazzo, Y.M.; Jamal, C.M.; Silveira, D. Chemical profile and biological activity of Crinum americanum L. (Amaryllidaceae). SAJB 2022, 146, 25–35. [Google Scholar] [CrossRef]
- Lianza, M.; Verdan, M.H.; Andrade, J.P.; Poli, F.; Almeida, L.C.; Costa-Lotufo, L.V.; Neto, Á.C.; Oliveira, S.C.C.; Bastida, J.; Batista, A.N.L.; et al. Isolation, absolute configuration and cytotoxic activities of alkaloids from Hippeastrum goianum (Ravenna) meerow (Amaryllidaceae). J. Braz. Chem. Soc. 2020, 31, 2135–2145. [Google Scholar] [CrossRef]
- Ortiz, J.E.; Garro, A.; Pigni, N.B.; Aguero, M.B.; Roitman, G.; Slanis, A.; Enriz, R.D.; Feresin, G.E.; Bastida, J.; Tapia, A. Cholinesterase-inhibitory effect and in silico analysis of alkaloids from bulbs of Hieronymiella species. Phytomedicine 2018, 39, 66–74. [Google Scholar] [CrossRef]
- Scheltens, P.; De Strooper, B.; Kivipelto, M.; Holstege, H.; Chételat, G.; Teunissen, C.E.; Cummings, J.; van der Flier, W.M. Alzheimer’s disease. Lancet 2021, 397, 1577–1590. [Google Scholar] [CrossRef]
- Mandrekar-Colucci, S.; Karlo, J.C.; Landreth, G.E. Mechanisms underlying the rapid peroxisome proliferator-activated receptor-gamma-mediated amyloid clearance and reversal of cognitive deficits in a murine model of Alzheimer’s disease. J. Neurosci. 2012, 32, 10117–10128. [Google Scholar] [CrossRef]
- Ramsay, R.R.; Popovic-Nikolic, M.R.; Nikolic, K.; Uliassi, E.; Bolognesi, M.L. A perspective on multi-target drug discovery and design for complex diseases. Clin. Translat. Med. 2018, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Reyes-Irisarri, E.; Hüll, M.; Kummer, M.P. Impact and therapeutic potential of PPARs in Alzheimer’s disease. Curr. Neuropharmacol. 2011, 9, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Reich, D.; Gallucci, G.; Tong, M.; de la Monte, S.M. Therapeutic advantages of dual targeting of PPAR-delta and PPAR-gamma in an experimental model of sporadic Alzheimer’s disease. J. Park. Dis. Alzheimer’s Dis. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Cortes, N.; Alvarez, R.; Osorio, E.H.; Alzate, F.; Berkov, S.; Osorio, E. Alkaloid metabolite profiles by GC/MS and acetylcholinesterase inhibitory activities with binding-mode predictions of five Amaryllidaceae plants. J. Pharm. Biomed. Anal. 2015, 102, 222–228. [Google Scholar] [CrossRef]
- Gasca, C.A.; Moreira, N.C.S.; Almeida, F.C.; Gomes, J.V.D.; Castillo, W.O.; Fagg, C.W.; Magalhaes, P.O.; Fonseca-Bazzo, Y.M.; Sakamoto-Hojo, E.; Medeiros, Y.K.; et al. Acetylcholinesterase inhibitory activity, anti-inflammatory, and neuroprotective potential of Hippeastrum psittacinum (Ker Gawl.) herb (Amaryllidaceae). Food Chem. Toxicol. 2020, 145, 111703. [Google Scholar] [CrossRef]
- Castillo, W.O.; Aristizabal-Pachon, A.F.; Sakamoto-Hojo, E.; Gasca, C.A.; Cabezas-Fajardo, F.A.; Takahashi, C. Caliphruria subedentata (Amaryllidaceae) decreases genotoxicity and cell death induced by β-amyloid peptide in SH-SY5Y cell line. Mutat. Res. Genet. Toxicol. Env. Mutagen. 2018, 836, 54–61. [Google Scholar] [CrossRef]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- López, S.; Bastida, J.; Viladomat, F.; Codina, C. Acetylcholinesterase inhibitory activity of some Amaryllidaceae alkaloids and Narcissus extracts. Life Sci. 2002, 71, 2521–2529. [Google Scholar] [CrossRef]
- Locatelli, M.; Gindro, R.; Travaglia, F.; Coïsson, J.D.; Rinaldi, M.; Arlorio, M. Study of the DPPH-scaveging activity. Development of a free software for the correct interpretation of data. Food Chem. 2009, 114, 889–897. [Google Scholar] [CrossRef]
- Prieto, P.; Pineda, M.; Aguilar, M. Spectrophotometric quantitation of antioxidant capacity through the formation of a phosphomolybdenum complex: Specific application to the determination of vitamin E. Anal. Biochem. 1999, 269, 337–341. [Google Scholar] [CrossRef]
- Rodrigues, P.M.; Dutra Gomes, J.V.; Jamal, C.M.; Cunha Neto, A.; Santos, M.L.; Fagg, C.W.; Fonseca-Bazzo, Y.M.; Magalhaes, P.O.; Sales, P.M.; Silveira, D. Triterpenes from Pouteria ramiflora (Mart.) Radlk. Leaves (Sapotaceae). Food Chem. Toxicol. 2017, 109, 1063–1068. [Google Scholar] [CrossRef] [PubMed]
- Leite, C.F.M.; Leite, B.H.M.; Barros, I.M.C.; Gomes, S.M.; Fagg, C.W.; Simeoni, L.A.; Silveira, D.; Fonseca, Y.M. Determination of rutin in Erythroxylum suberosum extract by liquid chromatography: Applicability in standardization of herbs and stability studies. Bol. Lat. Caribe. Plant Med. Aromat. 2014, 13, 135–143. [Google Scholar]



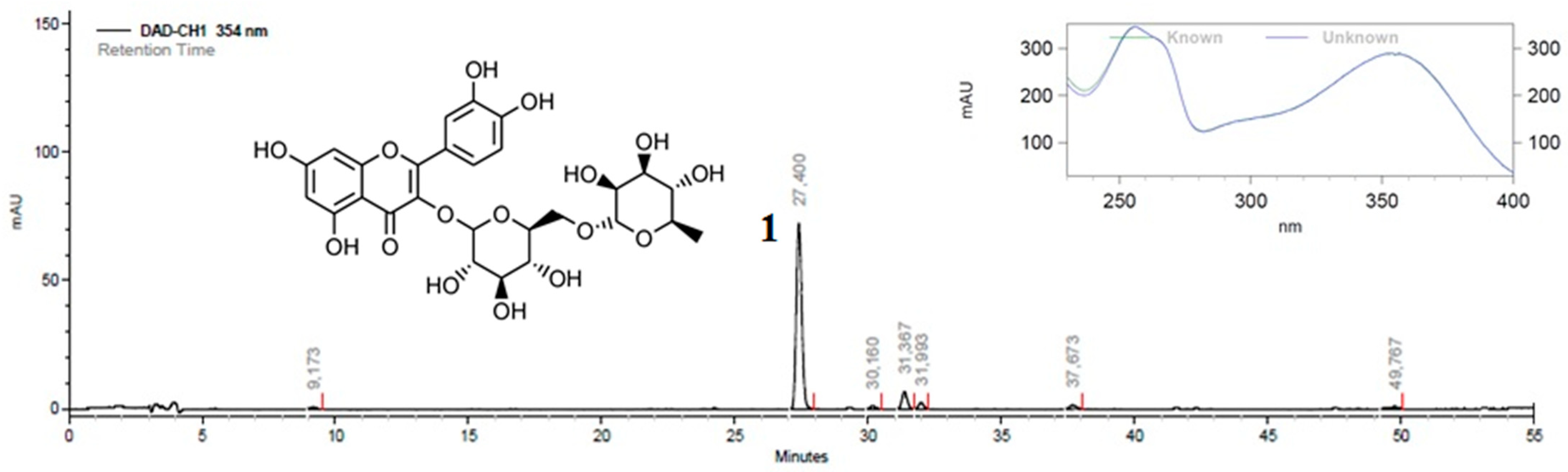

| Peak | %TIC | RT | Compound | Mass Fragmentation (Relative Intensity) | Reference |
|---|---|---|---|---|---|
| 1 | 0.8 | 34.9 | Lycorine | 226 (100), 227 (59), 250 (19), 27 (187), 286 (13), 147 (10), 228 (9), 119 (9), 248 (9) | [28] |
| 2 | 0.6 | 35.0 | Non-identified (NI-1) | 343 (100), 341 (71), 344 (36), 266 (33), 40 (29), 252 (28), 196 (21), 282 (19), 310 (19), 283 (18) | - |
| 3 | 94.6 | 38.0 | 7-Demethoxy-9-O-methylhostasine | 125 (100), 96 (21), 309 (7), 311 (5), 94 (5), 126 (4), 123 (4), 124 (3), 193 (3), 82 (3) | [29] |
| 4 | 1.3 | 38.4 | Non-identified (NI-2) | 311 (100), 40 (27), 310 (22), 294 (13), 309 (13), 312 (13), 296 (11), 251 (11), 208 (10), 268 (9) | [30] |
| 5 | 2.7 | 43.0 | Non-identified (NI-3) | 297 (100), 254 (39), 296 (26), 252 (23), 148 (20), 298 (16), 77 (11), 295 (10), 236 (9), 196 (9) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomes-Copeland, K.K.P.; Meireles, C.G.; Gomes, J.V.D.; Torres, A.G.; Sinoti, S.B.P.; Fonseca-Bazzo, Y.M.; Magalhães, P.d.O.; Fagg, C.W.; Simeoni, L.A.; Silveira, D. Hippeastrum stapfianum (Kraenzl.) R.S.Oliveira & Dutilh (Amaryllidaceae) Ethanol Extract Activity on Acetylcholinesterase and PPAR-α/γ Receptors. Plants 2022, 11, 3179. https://doi.org/10.3390/plants11223179
Gomes-Copeland KKP, Meireles CG, Gomes JVD, Torres AG, Sinoti SBP, Fonseca-Bazzo YM, Magalhães PdO, Fagg CW, Simeoni LA, Silveira D. Hippeastrum stapfianum (Kraenzl.) R.S.Oliveira & Dutilh (Amaryllidaceae) Ethanol Extract Activity on Acetylcholinesterase and PPAR-α/γ Receptors. Plants. 2022; 11(22):3179. https://doi.org/10.3390/plants11223179
Chicago/Turabian StyleGomes-Copeland, Kicia Karinne Pereira, Cinthia Gabriel Meireles, João Victor Dutra Gomes, Amanda Gomes Torres, Simone Batista Pires Sinoti, Yris Maria Fonseca-Bazzo, Pérola de Oliveira Magalhães, Christopher William Fagg, Luiz Alberto Simeoni, and Dâmaris Silveira. 2022. "Hippeastrum stapfianum (Kraenzl.) R.S.Oliveira & Dutilh (Amaryllidaceae) Ethanol Extract Activity on Acetylcholinesterase and PPAR-α/γ Receptors" Plants 11, no. 22: 3179. https://doi.org/10.3390/plants11223179
APA StyleGomes-Copeland, K. K. P., Meireles, C. G., Gomes, J. V. D., Torres, A. G., Sinoti, S. B. P., Fonseca-Bazzo, Y. M., Magalhães, P. d. O., Fagg, C. W., Simeoni, L. A., & Silveira, D. (2022). Hippeastrum stapfianum (Kraenzl.) R.S.Oliveira & Dutilh (Amaryllidaceae) Ethanol Extract Activity on Acetylcholinesterase and PPAR-α/γ Receptors. Plants, 11(22), 3179. https://doi.org/10.3390/plants11223179








