Cannabinol: History, Syntheses, and Biological Profile of the Greatest “Minor” Cannabinoid
Abstract
1. Introduction
2. History
3. Synthesis
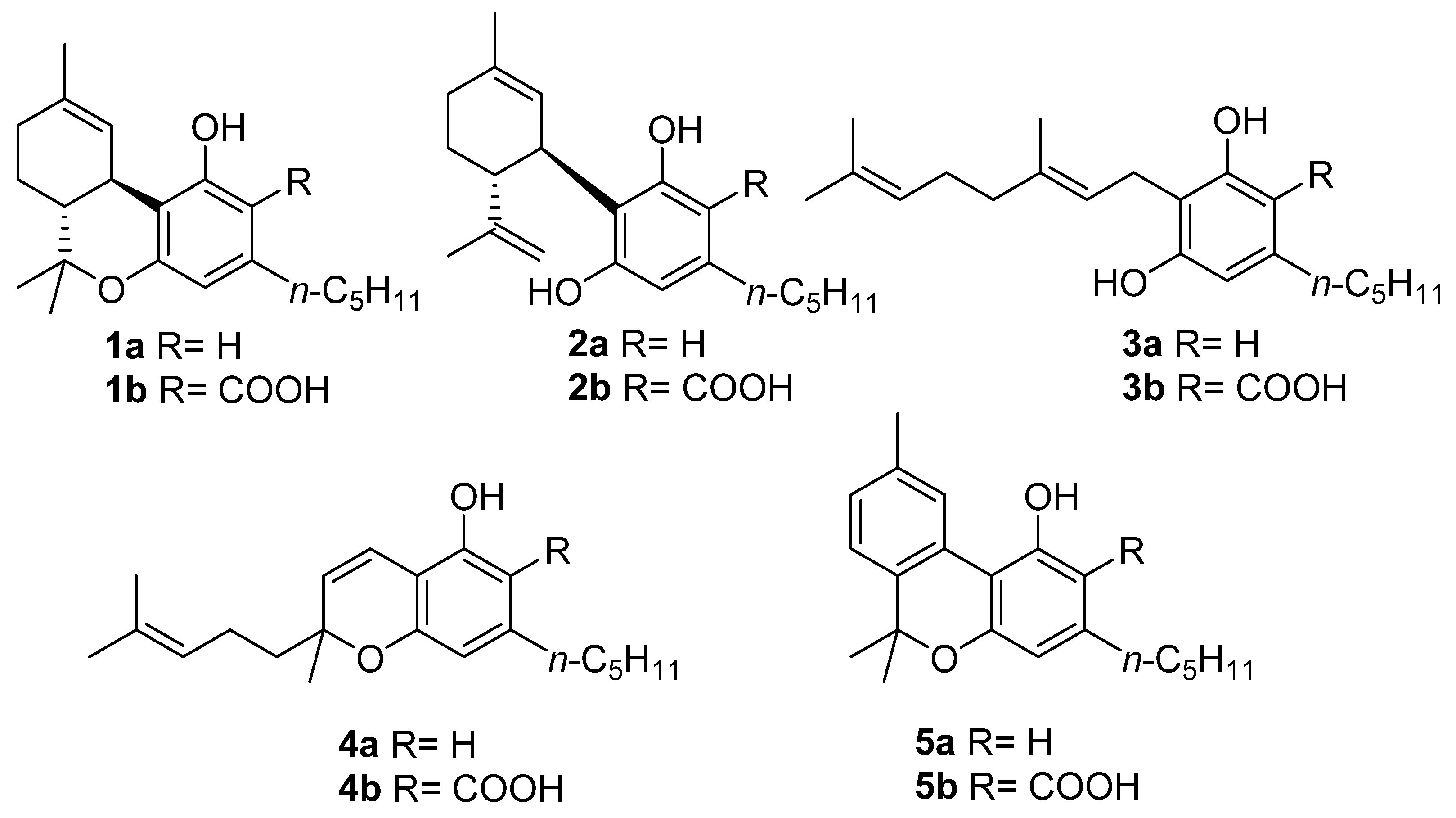
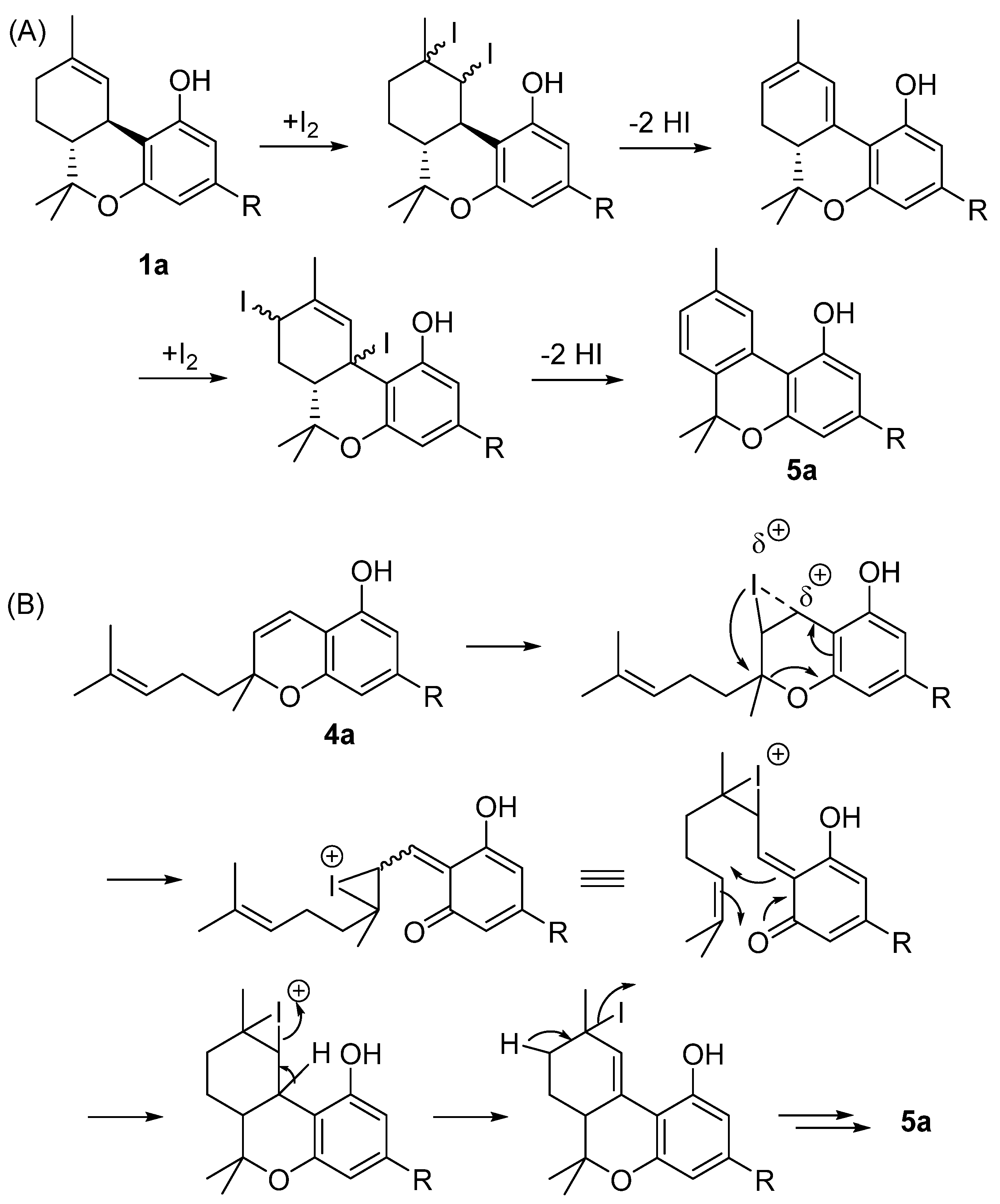
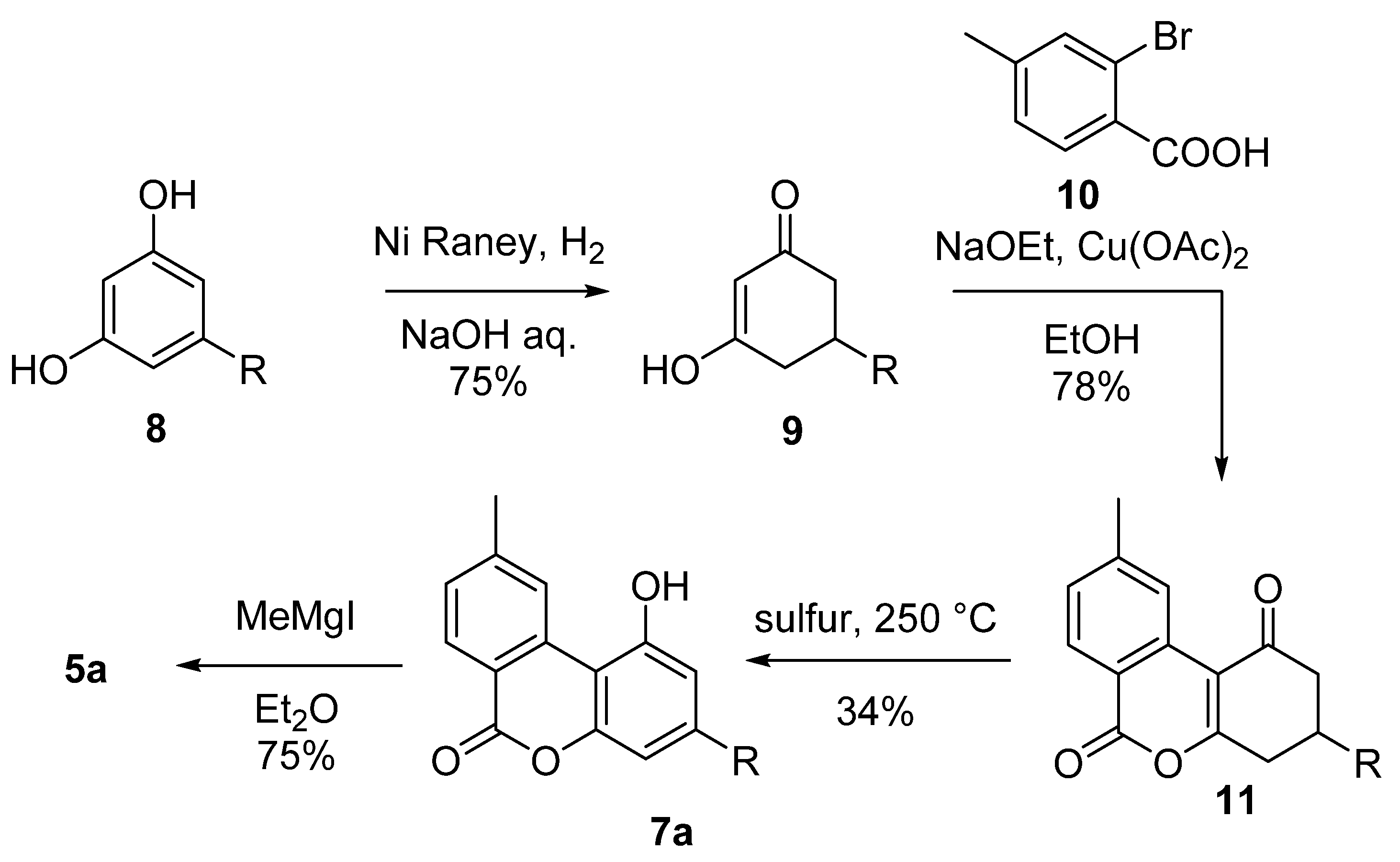
3.1. Semisynthesis
3.2. Total Synthesis through Lactonic Intermediate
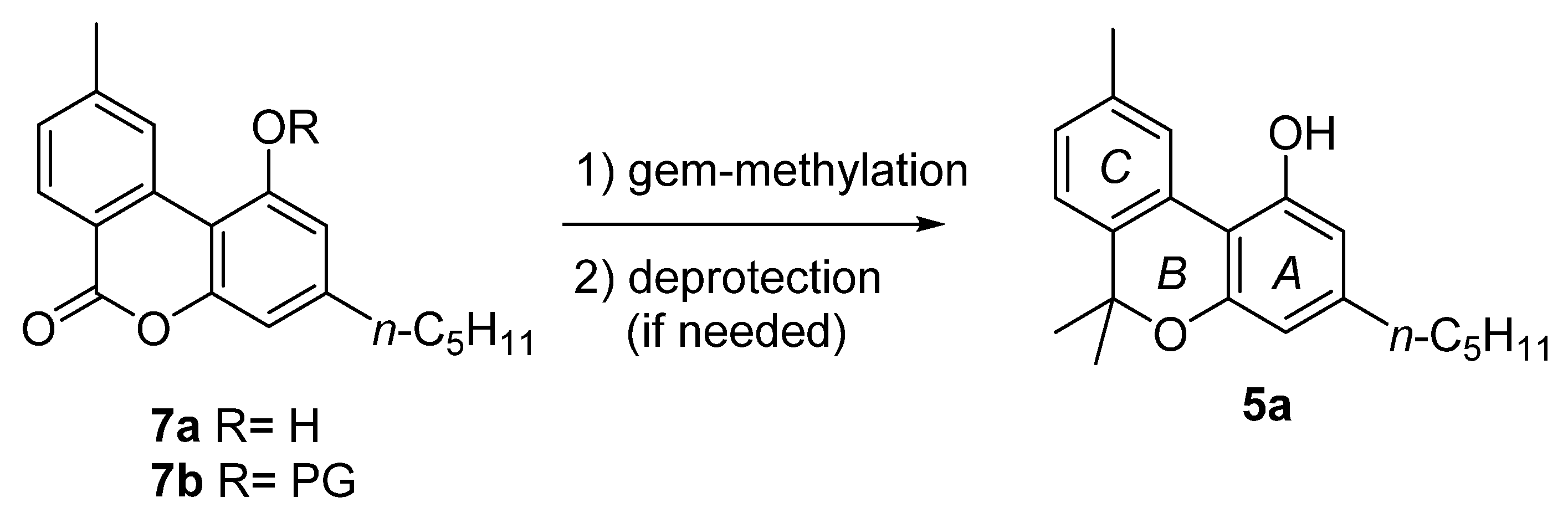
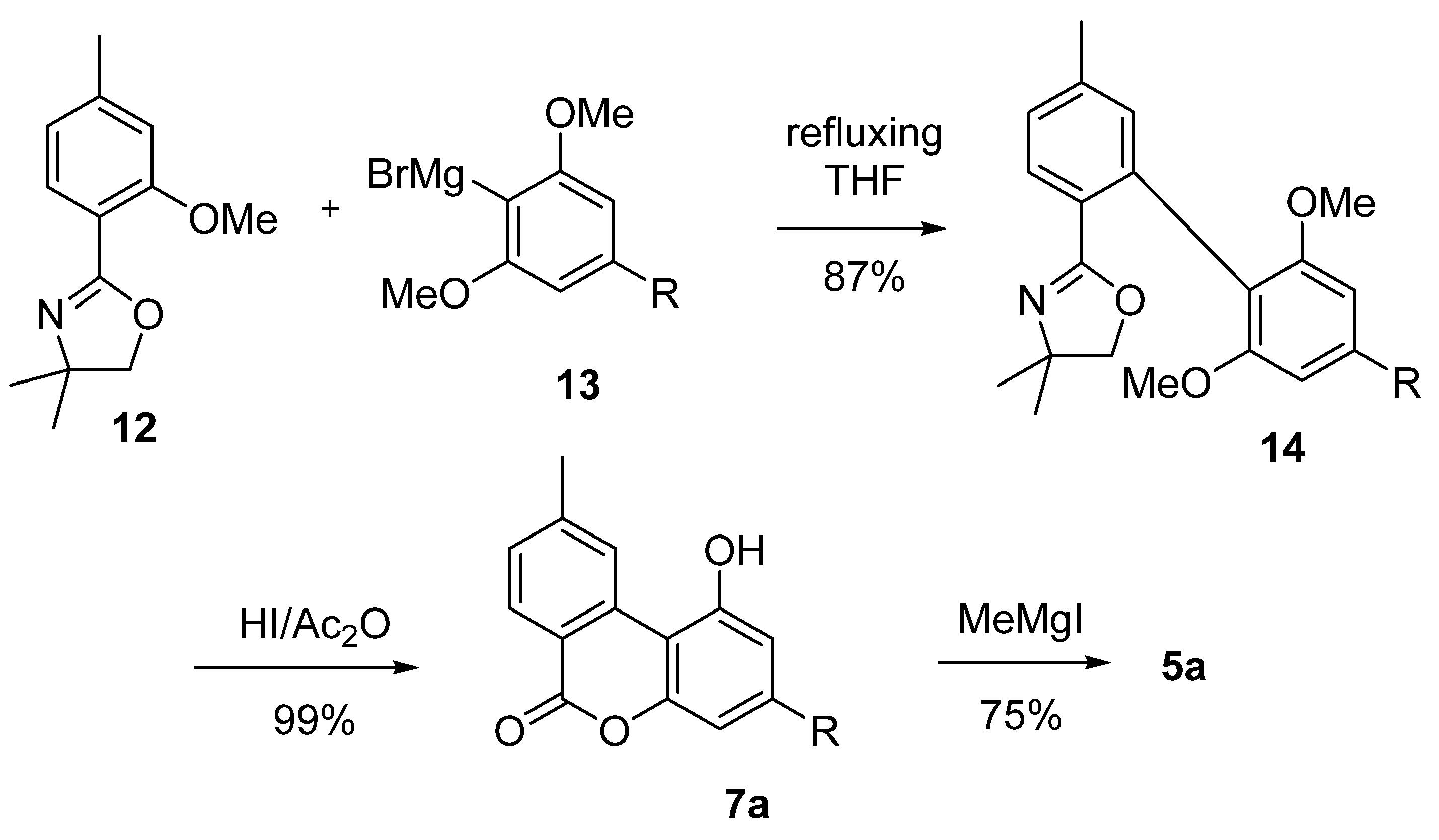
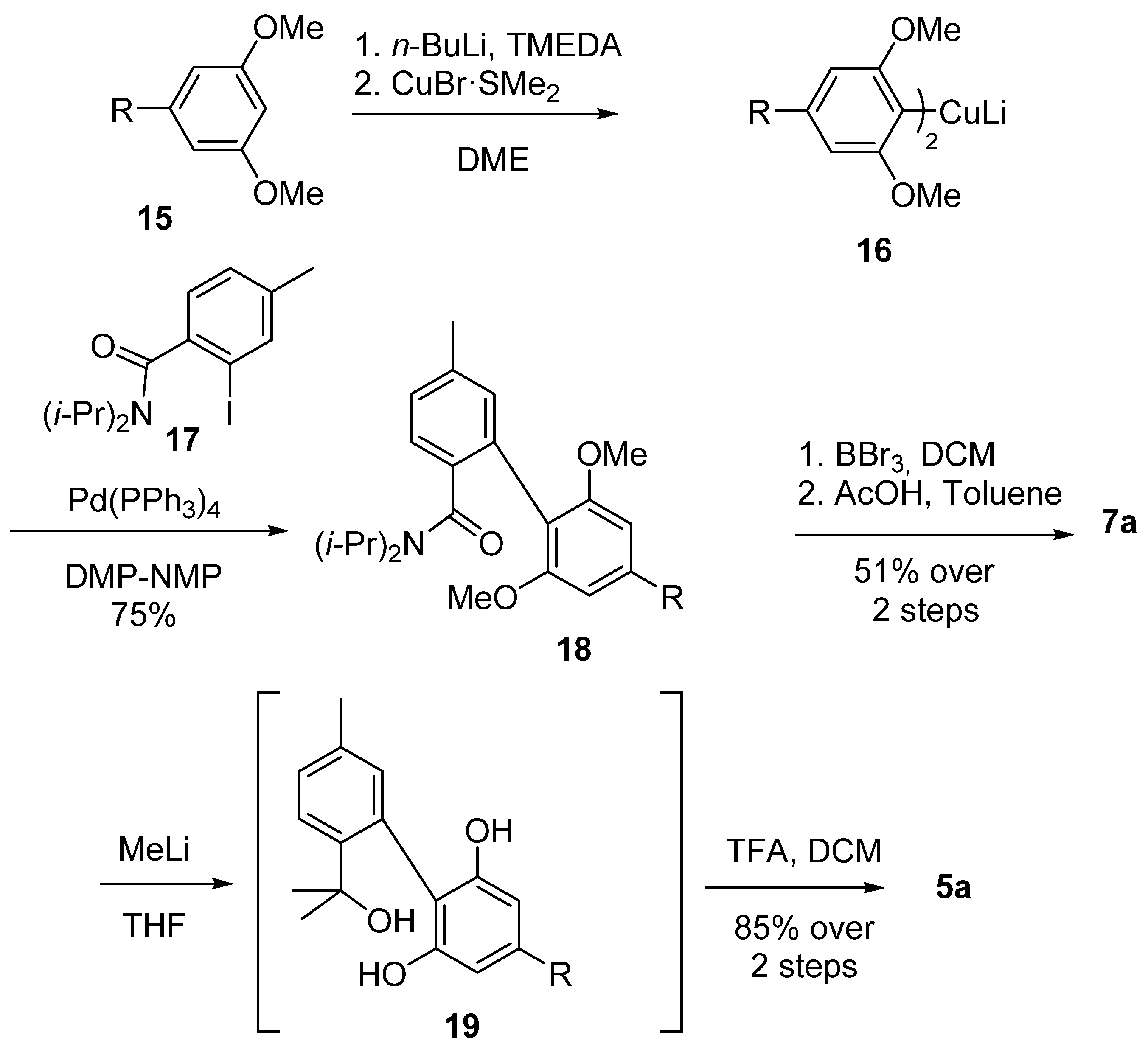
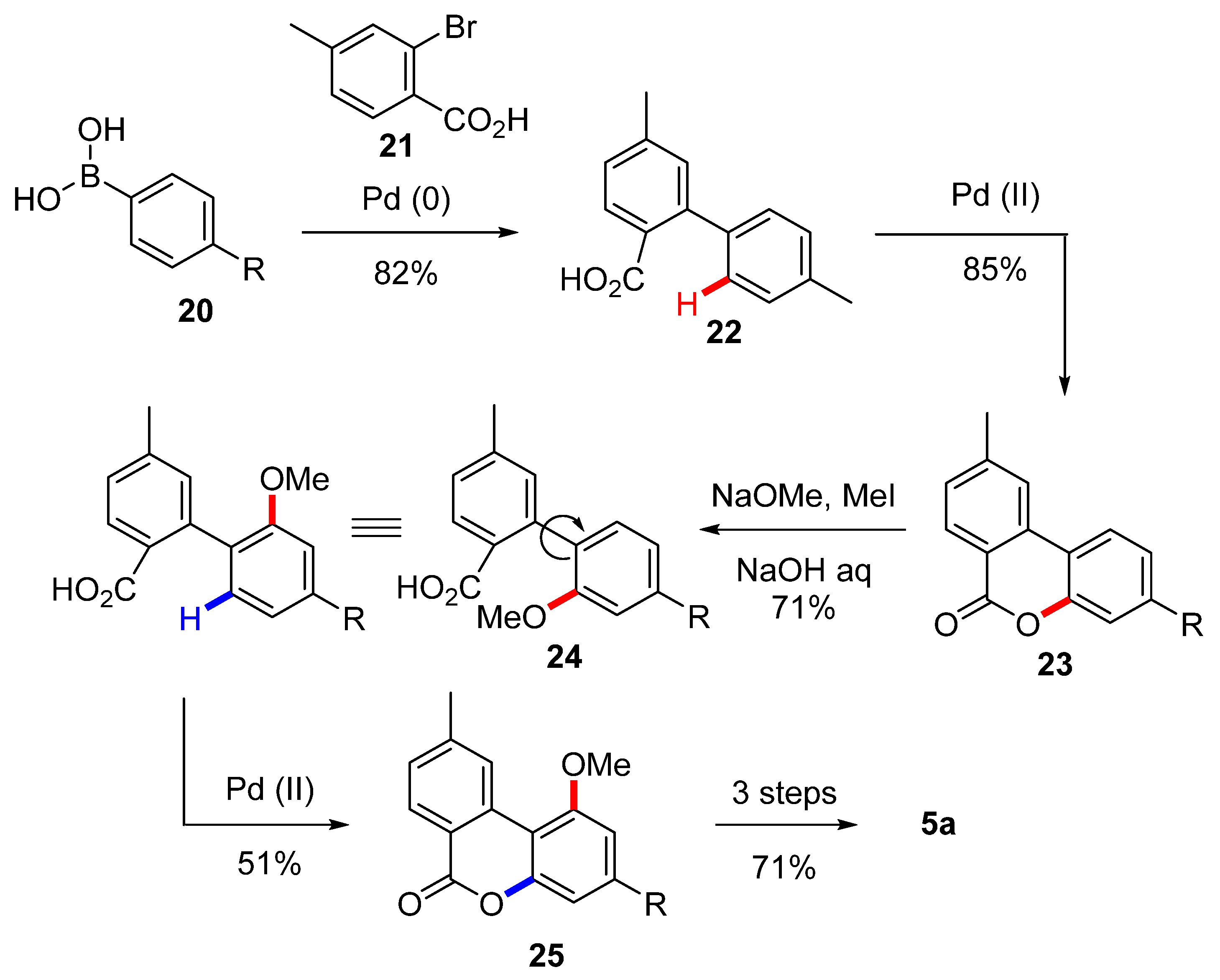
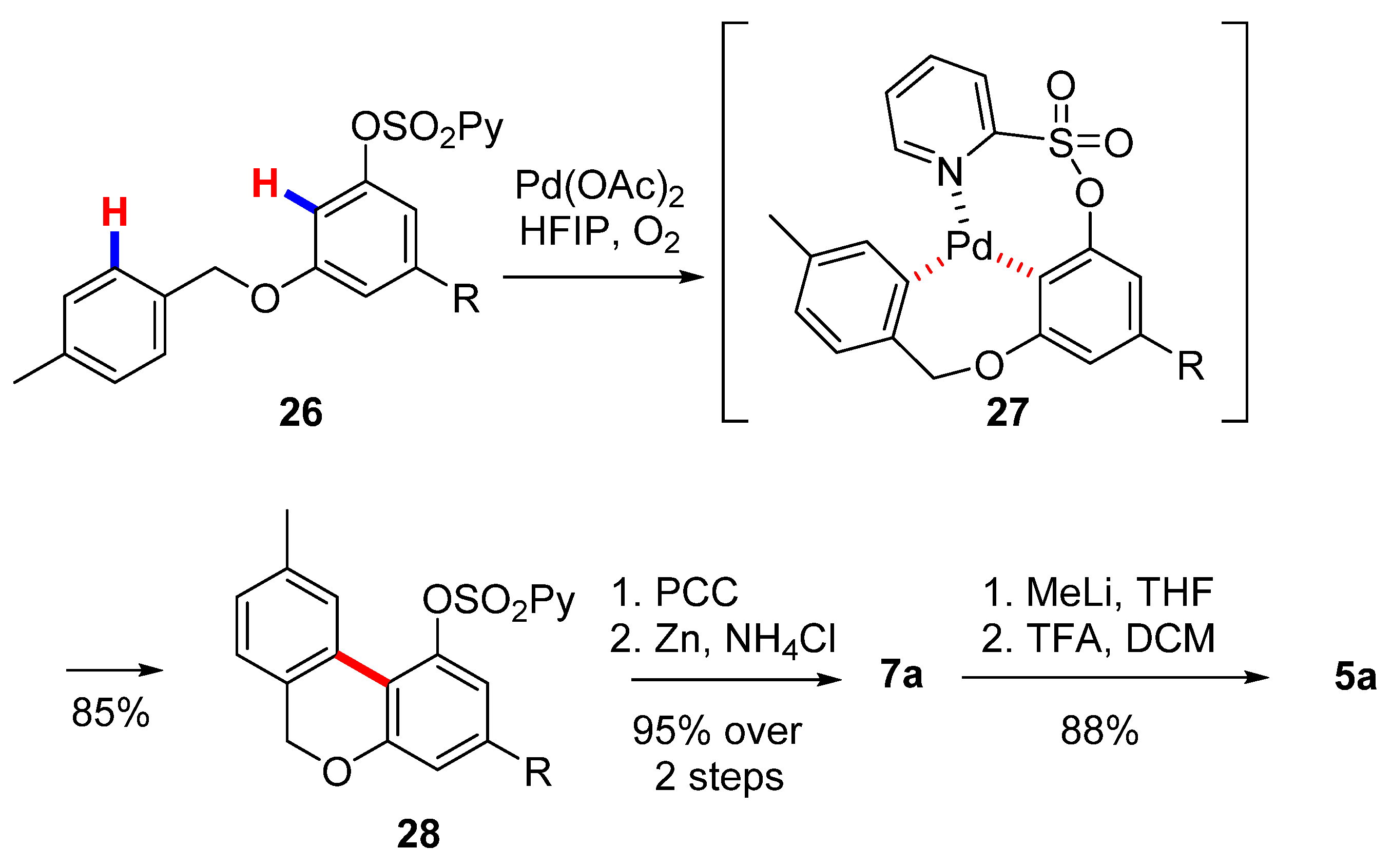
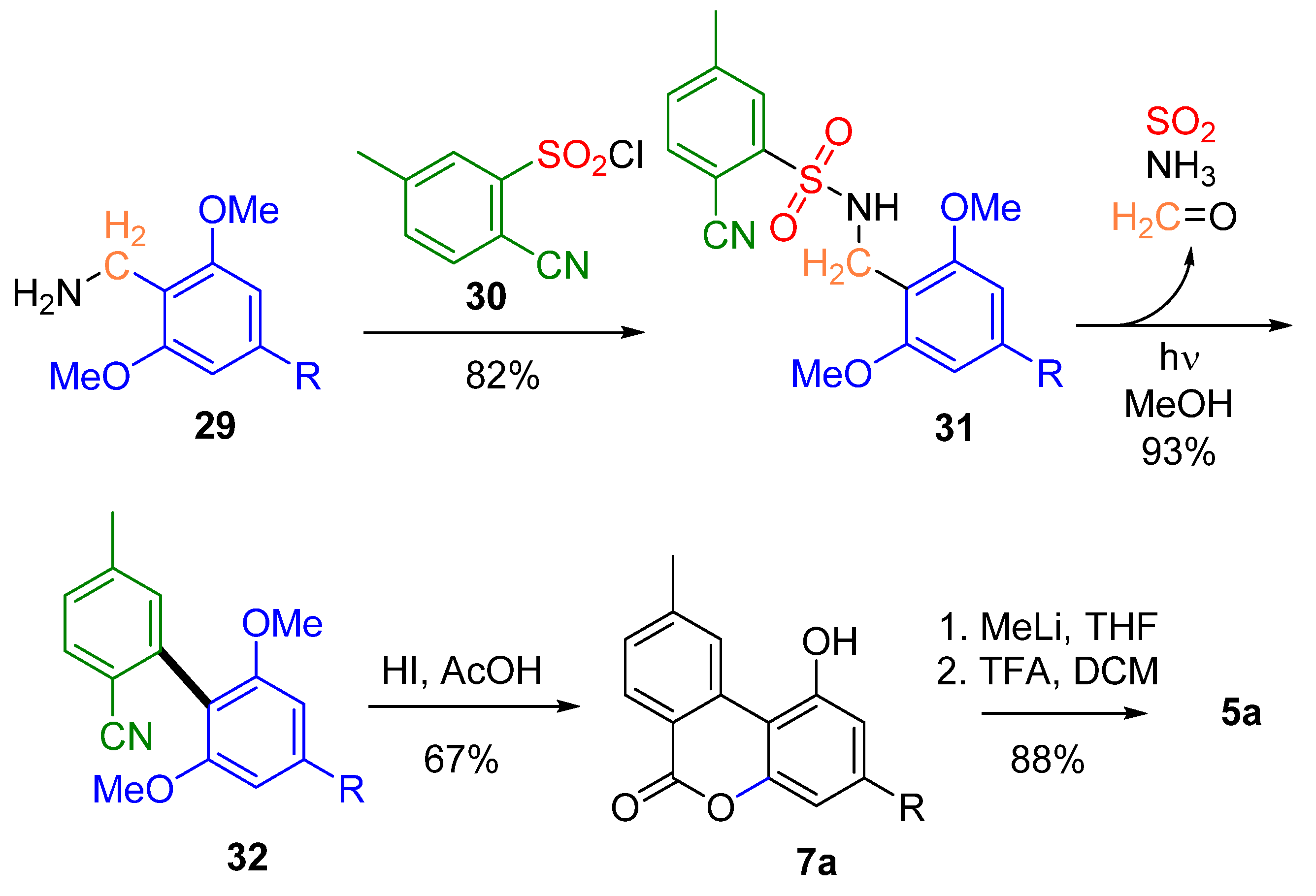
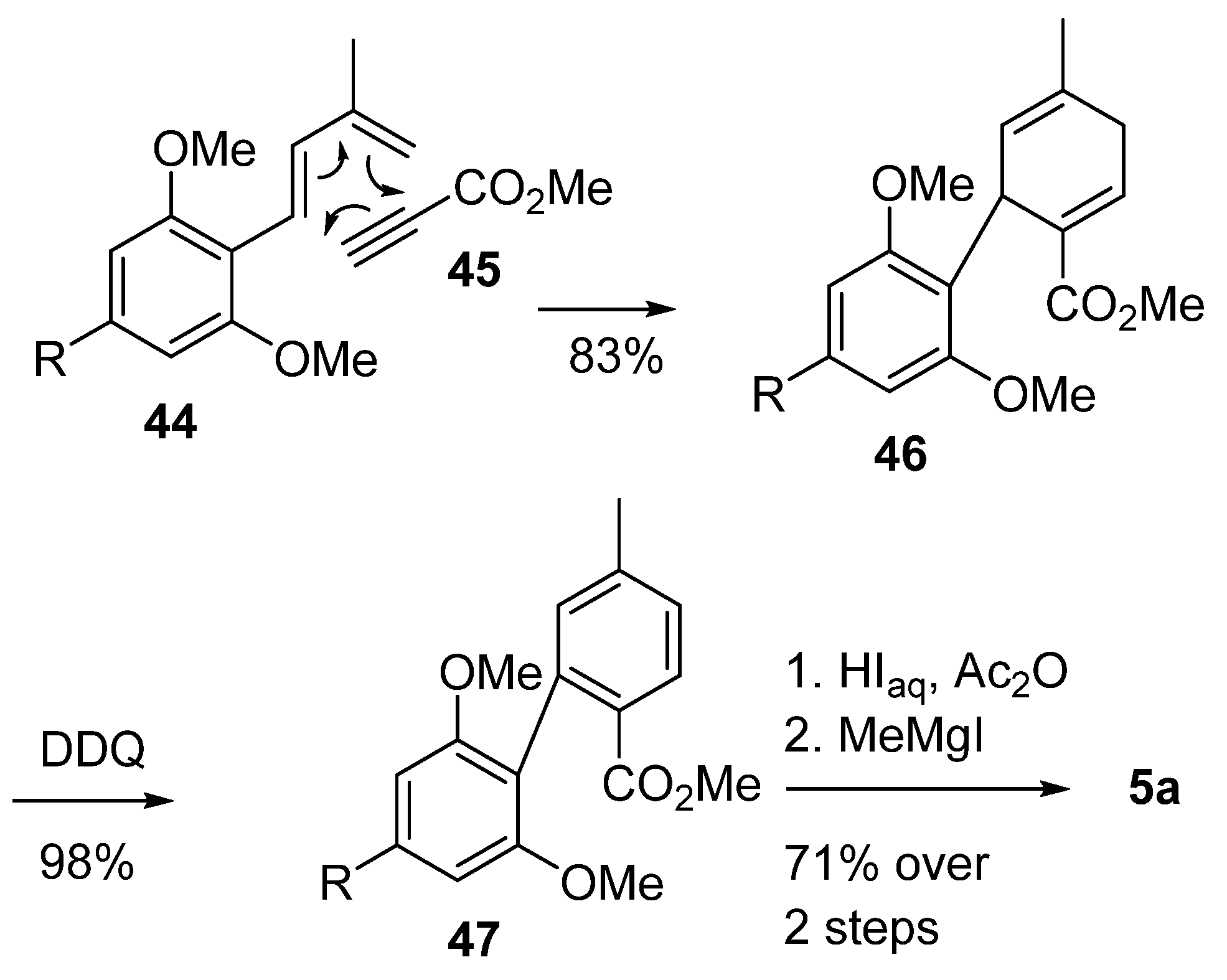
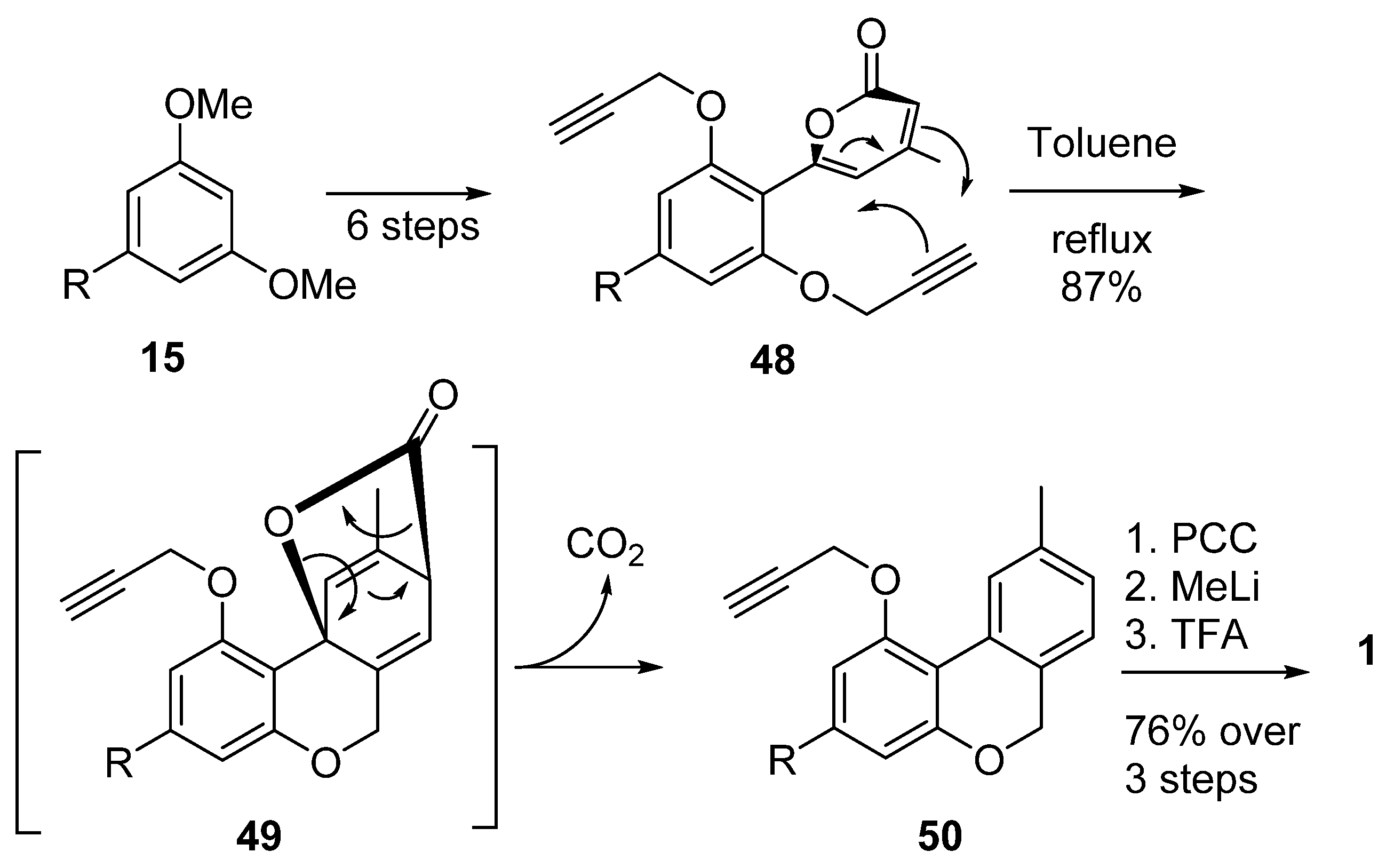
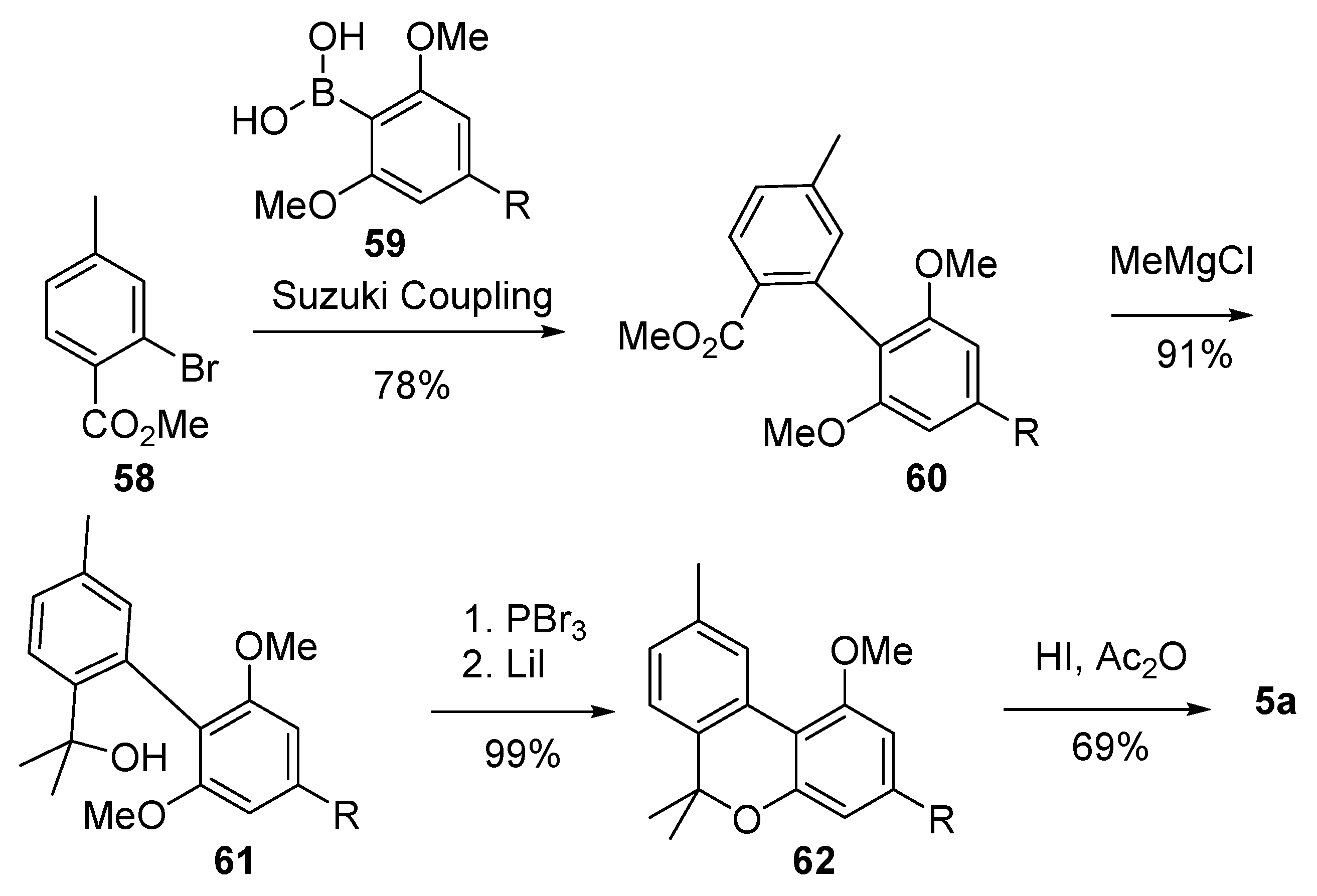
3.2.1. Synthesis through a Biphenyl Coupling Approach
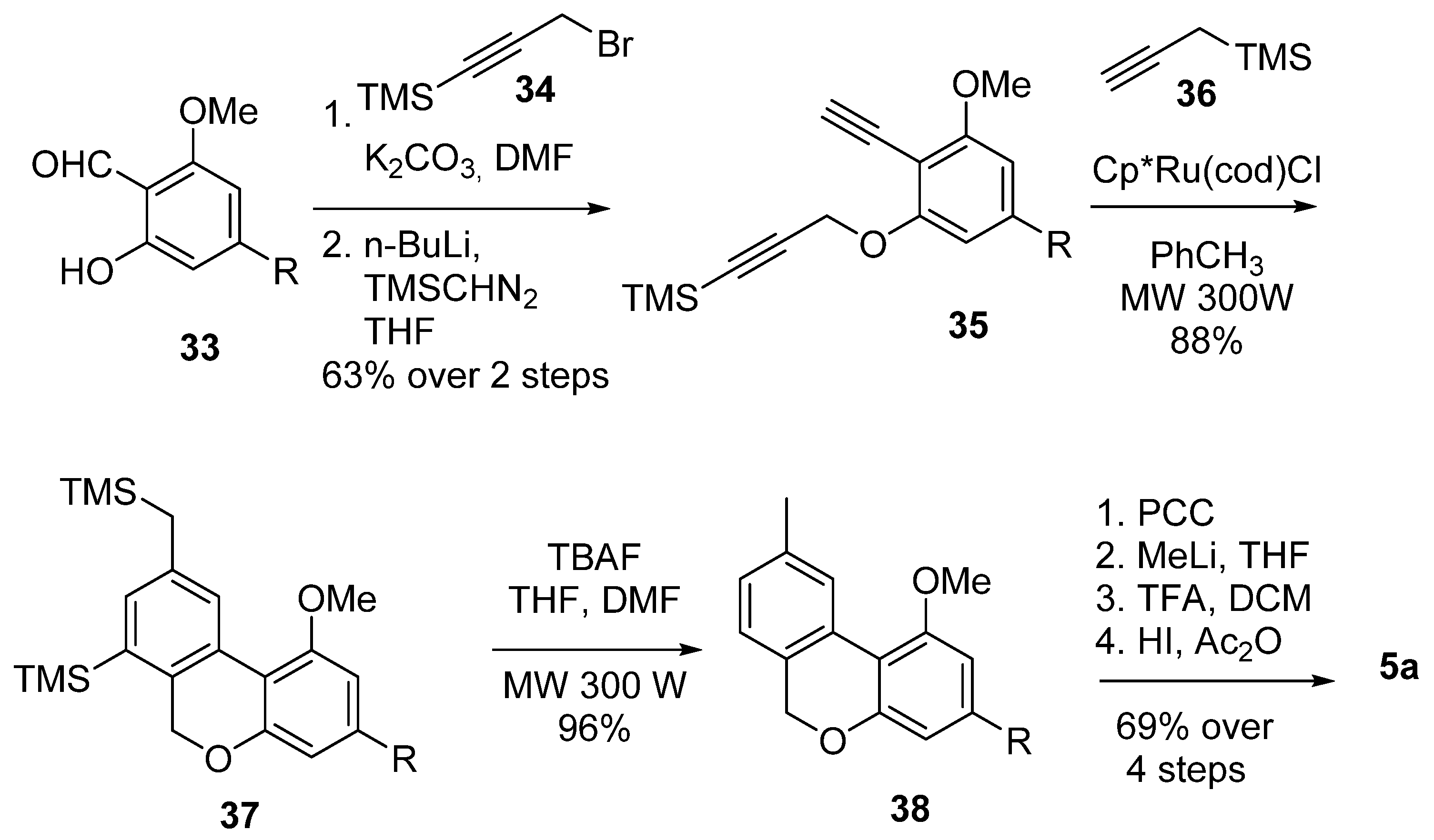
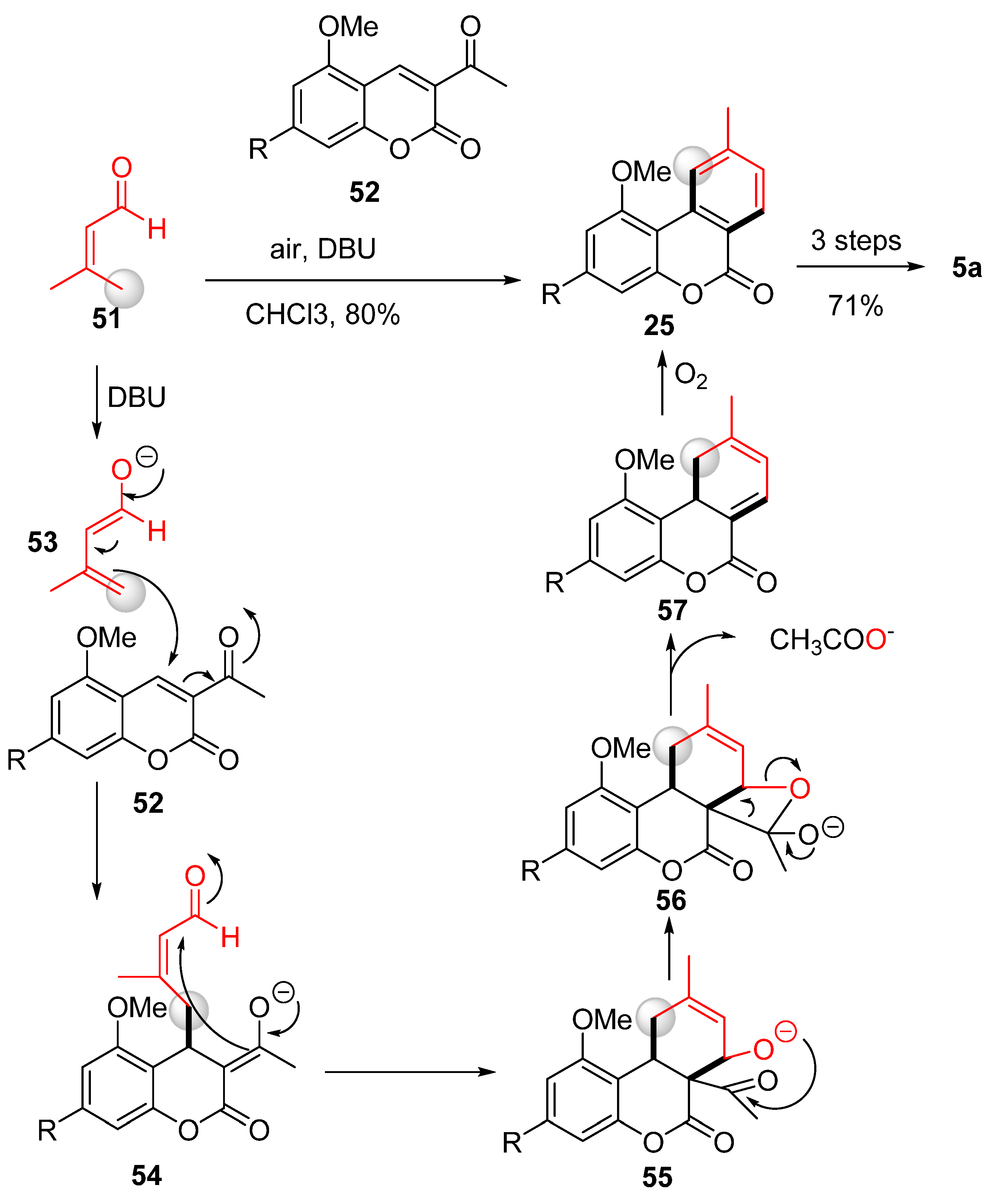
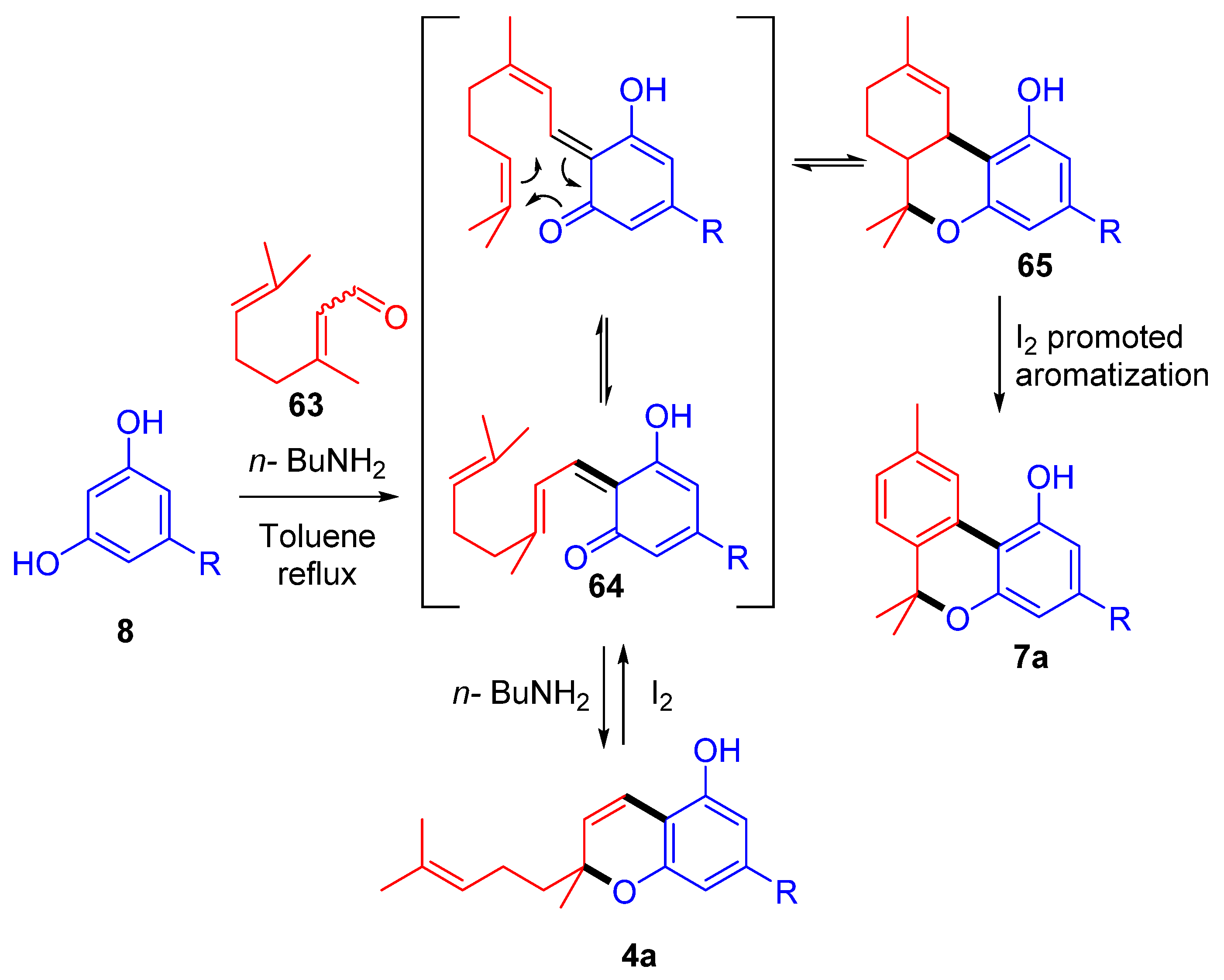
3.2.2. Synthesis through a Cyclization Approach
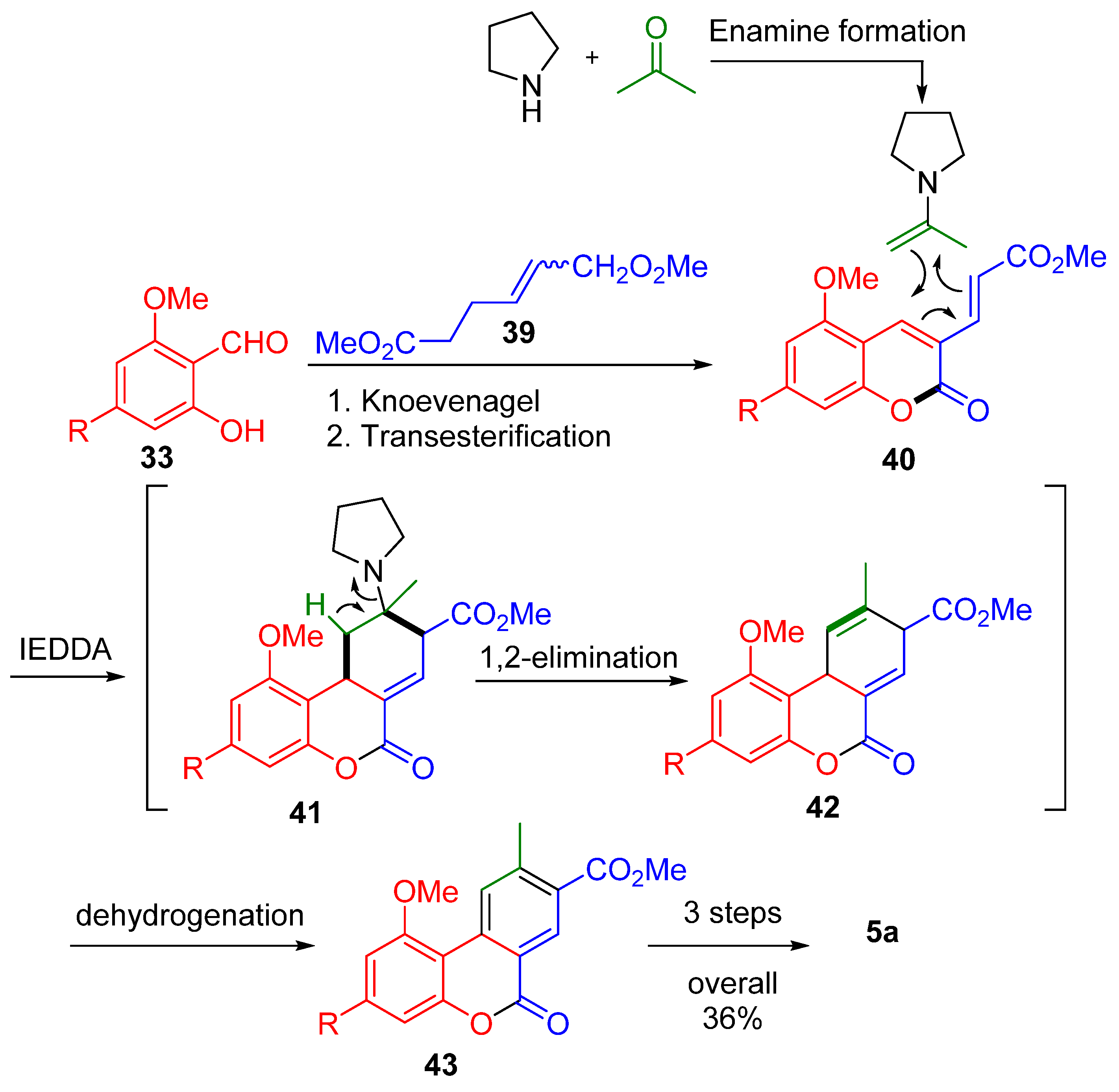
3.3. Total Synthesis through Non-Lactonic Intermediate
4. Biological Profile
4.1. Biochemical Assays
4.1.1. Cannabinoid Receptors (CBs)
4.1.2. Transient Receptor Potential Channels (TRPs)
4.2. Pre-Clinical and Clinical Assays
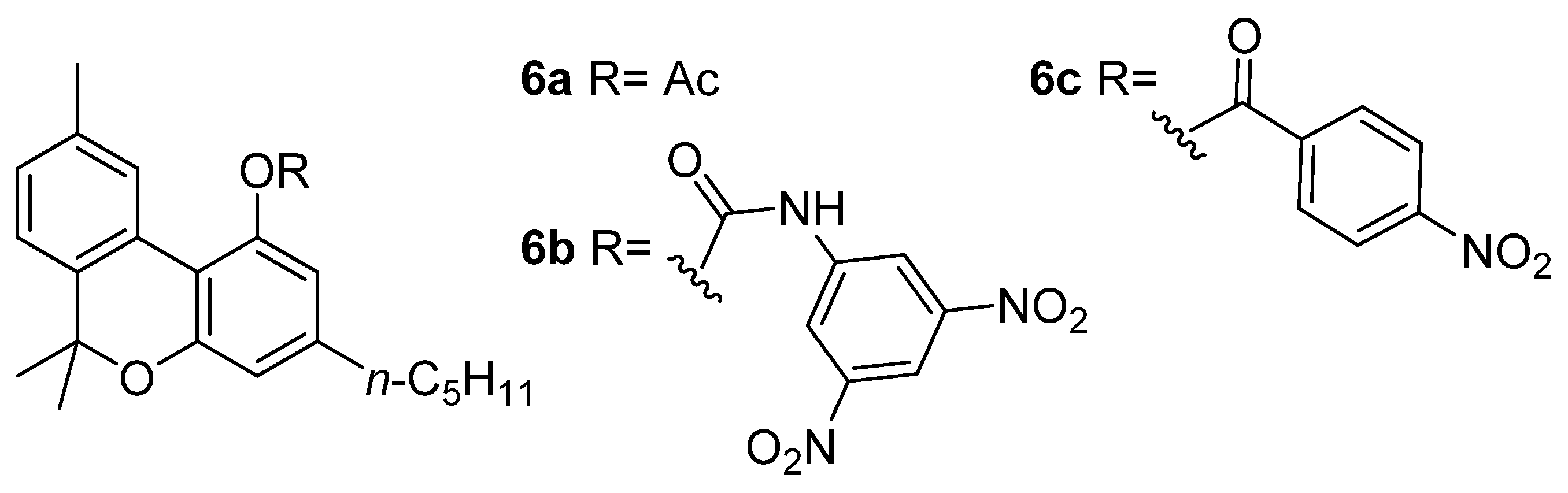
4.2.1. Analgesic and Anti-Inflammatory Activity
4.2.2. Antibacterial Activity
4.2.3. Orexigenic Activity
4.2.4. Treatment of Epidermolysis Bullosa
4.2.5. Sleep Induction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Karche, T.; Singh, M.R. The Application of Hemp (Cannabissativa L.) for a Green Economy: A Review. Turk. J. Bot. 2019, 43, 710–723. [Google Scholar] [CrossRef]
- Hesami, M.; Pepe, M.; Baiton, A.; Salami, S.A.; Jones, A.M.P. New Insight into Ornamental Applications of Cannabis: Perspectives and Challenges. Plants 2022, 11, 2383. [Google Scholar] [CrossRef] [PubMed]
- Pisanti, S.; Bifulco, M. Medical Cannabis: A Plurimillennial History of an Evergreen. J. Cell. Physiol. 2019, 234, 8342–8351. [Google Scholar] [CrossRef] [PubMed]
- De Souza, M.R.; Henriques, A.T.; Limberger, R.P. Medical Cannabis Regulation: An Overview of Models around the World with Emphasis on the Brazilian Scenario. J. Cannabis Res. 2022, 4, 33. [Google Scholar] [CrossRef]
- Fraguas-Sánchez, A.I.; Torres-Suárez, A.I. Medical Use of Cannabinoids. Drugs 2018, 78, 1665–1703. [Google Scholar] [CrossRef] [PubMed]
- Romero-Sandoval, E.A.; Fincham, J.E.; Kolano, A.L.; Sharpe, B.N.; Alvarado-Vázquez, P.A. Cannabis for Chronic Pain: Challenges and Considerations. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2018, 38, 651–662. [Google Scholar] [CrossRef] [PubMed]
- Hesami, M.; Pepe, M.; Alizadeh, M.; Rakei, A.; Baiton, A.; Phineas Jones, A.M. Recent Advances in Cannabis Biotechnology. Ind. Crops Prod. 2020, 158, 113026. [Google Scholar] [CrossRef]
- Nguyen, G.-N.; Jordan, E.N.; Kayser, O. Synthetic Strategies for Rare Cannabinoids Derived from Cannabis sativa. J. Nat. Prod. 2022, 85, 1555–1568. [Google Scholar] [CrossRef]
- Atakan, Z. Cannabis, a Complex Plant: Different Compounds and Different Effects on Individuals. Ther. Adv. Psychopharmacol. 2012, 2, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, V. New Approaches and Challenges to Targeting the Endocannabinoid System. Nat. Rev. Drug Discov. 2018, 17, 623–639. [Google Scholar] [CrossRef] [PubMed]
- Anokwuru, C.P.; Makolo, F.L.; Sandasi, M.; Tankeu, S.Y.; Elisha, I.L.; Agoni, C.; Combrinck, S.; Viljoen, A. Cannabigerol: A Bibliometric Overview and Review of Research on an Important Phytocannabinoid. Phytochem. Rev. 2022, 21, 1523–1547. [Google Scholar] [CrossRef]
- Pollastro, F.; Caprioglio, D.; Del Prete, D.; Rogati, F.; Minassi, A.; Taglialatela-Scafati, O.; Munoz, E.; Appendino, G. Cannabichromene. Nat. Prod. Commun. 2018, 13, 1934578X1801300. [Google Scholar] [CrossRef]
- Iversen, L. The Pharmacology of Delta-9-Tetrahydrocannabinol (THC); Oxford University Press: Oxford, UK, 2018; Volume 1. [Google Scholar]
- Sholler, D.J.; Schoene, L.; Spindle, T.R. Therapeutic Efficacy of Cannabidiol (CBD): A Review of the Evidence From Clinical Trials and Human Laboratory Studies. Curr. Addict. Rep. 2020, 7, 405–412. [Google Scholar] [CrossRef]
- Maiocchi, A.; Barbieri, J.; Fasano, V.; Passarella, D. Stereoselective Synthetic Strategies to (−)-Cannabidiol. ChemistrySelect 2022, 7, e202202400. [Google Scholar] [CrossRef]
- Nachnani, R.; Raup-Konsavage, W.M.; Vrana, K.E. The Pharmacological Case for Cannabigerol. J. Pharmacol. Exp. Ther. 2021, 376, 204–212. [Google Scholar] [CrossRef]
- Bloemendal, V.R.L.J.; van Hest, J.C.M.; Rutjes, F.P.J.T. Synthetic Pathways to Tetrahydrocannabinol (THC): An Overview. Org. Biomol. Chem. 2020, 18, 3203–3215. [Google Scholar] [CrossRef]
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A Unified Critical Inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef]
- Caprioglio, D.; Amin, H.I.M.; Taglialatela-Scafati, O.; Muñoz, E.; Appendino, G. Minor Phytocannabinoids: A Misleading Name but a Promising Opportunity for Biomedical Research. Biomolecules 2022, 12, 1084. [Google Scholar] [CrossRef]
- Walsh, K.B.; McKinney, A.E.; Holmes, A.E. Minor Cannabinoids: Biosynthesis, Molecular Pharmacology and Potential Therapeutic Uses. Front. Pharmacol. 2021, 12, 777804. [Google Scholar] [CrossRef]
- Tahir, M.N.; Shahbazi, F.; Rondeau-Gagné, S.; Trant, J.F. The Biosynthesis of the Cannabinoids. J. Cannabis Res. 2021, 3, 7. [Google Scholar] [CrossRef]
- Garrett, E.R.; Gouyette, A.J.; Roseboom, H. Stability of Tetrahydrocannabinols II. J. Pharm. Sci. 1978, 67, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Bastola, K.; Hazekamp, A.; Verpoorte, R. Synthesis and Spectroscopic Characterization of Cannabinolic Acid. Planta Med. 2007, 73, 273–275. [Google Scholar] [CrossRef] [PubMed]
- Pain, S. A Potted History. Nature 2015, 525, S10–S11. [Google Scholar] [CrossRef] [PubMed]
- Lavrieux, M.; Jacob, J.; Disnar, J.-R.; Bréheret, J.-G.; Le Milbeau, C.; Miras, Y.; Andrieu-Ponel, V. Sedimentary Cannabinol Tracks the History of Hemp Retting. Geology 2013, 41, 751–754. [Google Scholar] [CrossRef]
- Russo, E.B. History of Cannabis and Its Preparations in Saga, Science, and Sobriquet. Chem. Biodivers. 2007, 4, 1614–1648. [Google Scholar] [CrossRef]
- Appendino, G. The Early History of Cannabinoid Research. Rendiconti Lincei. Scienze Fisiche e Naturali 2020, 31, 919–929. [Google Scholar] [CrossRef]
- Pertwee, R.G. Cannabinoid Pharmacology: The First 66 Years: Cannabinoid Pharmacology. Br. J. Pharmacol. 2006, 147, S163–S171. [Google Scholar] [CrossRef]
- Wood, T.B.; Spivey, W.T.N.; Easterfield, T.H. XL.—Charas. The Resin of Indian Hemp. J. Chem. Soc. Trans. 1896, 69, 539–546. [Google Scholar] [CrossRef]
- Caprioglio, D.; Mattoteia, D.; Taglialatela-Scafati, O.; Muñoz, E.; Appendino, G. Cannabinoquinones: Synthesis and Biological Profile. Biomolecules 2021, 11, 991. [Google Scholar] [CrossRef]
- Wood, T.B.; Spivey, W.T.N.; Easterfield, T.H. III.—Cannabinol. Part I. J. Chem. Soc. Trans. 1899, 75, 20–36. [Google Scholar] [CrossRef]
- Madan, H.G.; Randall, W.B.; Shaw, S.; Simpson, M.; Newton Spivey, W.T. Obituary notices: Sir Joseph Henry Gilbert, Ph.D., M.A., L.L.D., Sc.D., F.R.S., 1817–1901. J. Chem. Soc. Trans. 1902, 81, 625–636. [Google Scholar] [CrossRef]
- Walton, R. Marihuana, America’s New Drug Problem; Lippincott: New York, NY, USA, 1938. [Google Scholar]
- Brian, R. Davis “Easterfield, Thomas Hill”, Dictionary of New Zealand Biography, First Published in 1996. Te Ar—The Encyclopedia of New Zealand. Available online: https://teara.govt.nz/en/biographies/3e1/easterfield-thomas-hill (accessed on 15 September 2022).
- Mills, J.H. Cannabis Britannica: Empire, Trade, and Prohibition, 1800–1928; Oxford University Press: Oxford, UK, 2005; ISBN 978-0-19-927881-7. [Google Scholar]
- Cahn, R.S.; Ingold, C.; Prelog, V. Specification of Molecular Chirality. Angew. Chem. Int. Ed. Engl. 1966, 5, 385–415. [Google Scholar] [CrossRef]
- Cahn, R.S. 326. Cannabis Indica Resin. Part IV. The Synthesis of Some 2: 2-Dimethyldibenzopyrans, and Confirmation of the Structure of Cannabinol. J. Chem. Soc. Resumed 1933, 1400–1405. [Google Scholar] [CrossRef]
- Mechoulam, R. Todd’s Achievement. Nature 1997, 386, 755. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.; Pease, D.C.; Clark, J.H. Isolation of Cannabinol, Cannabidiol and Quebrachitol from Red Oil of Minnesota Wild Hemp. J. Am. Chem. Soc. 1940, 62, 2194–2196. [Google Scholar] [CrossRef]
- Work, T.S.; Bergel, F.; Todd, A.R. The Active Principles of Cannabis Indica Resin. I. Biochem. J. 1939, 33, 123–127. [Google Scholar] [CrossRef]
- Jacob, A.; Todd, A.R. 119. Cannabis Indica. Part II. Isolation of Cannabidiol from Egyptian Hashish. Observations on the Structure of Cannabinol. J. Chem. Soc. Resumed 1940, 649–653. [Google Scholar] [CrossRef]
- Adams, R.; Baker, B.R.; Wearn, R.B. Structure of Cannabinol. III. Synthesis of Cannabinol, 1-Hydroxy-3-n-Amyl-6,6,9-Trimethyl-6-Dibenzopyran 1. J. Am. Chem. Soc. 1940, 62, 2204–2207. [Google Scholar] [CrossRef]
- Hanessian, S.; Margarita, R.; Hall, A.; Johnstone, S.; Tremblay, M.; Parlanti, L. New and Old Challenges in Total Synthesis. From Concept to Practice. Pure Appl. Chem. 2003, 75, 209–221. [Google Scholar] [CrossRef][Green Version]
- Wianowska, D.; Dawidowicz, A.L.; Kowalczyk, M. Transformations of Tetrahydrocannabinol, Tetrahydrocannabinolic Acid and Cannabinol during Their Extraction from Cannabis sativa L. J. Anal. Chem. 2015, 70, 920–925. [Google Scholar] [CrossRef]
- Hidayati, N.; Saefumillah, A.; Cahyana, A.H. Simple Isolation Method of Cannabinol from Cannabis sativa to Produce Secondary Reference Standard Analysis Material. IOP Conf. Ser. Mater. Sci. Eng. 2020, 902, 012063. [Google Scholar] [CrossRef]
- Meltzer, P.C.; Dalzell, H.C.; Razdan, R.K. An Improved Synthesis of Cannabinol and Cannabiorcol. Synthesis 1981, 1981, 985–987. [Google Scholar] [CrossRef]
- Mechoulam, R.; Yagnitinsky, B.; Gaoni, Y. Hashish. XII. Stereoelectronic Factor in the Chloranil Dehydrogenation of Cannabinoids. Total Synthesis of Dl-Cannabichromene. J. Am. Chem. Soc. 1968, 90, 2418–2420. [Google Scholar] [CrossRef] [PubMed]
- Pollastro, F.; Caprioglio, D.; Marotta, P.; Moriello, A.S.; De Petrocellis, L.; Taglialatela-Scafati, O.; Appendino, G. Iodine-Promoted Aromatization of p-Menthane-Type Phytocannabinoids. J. Nat. Prod. 2018, 81, 630–633. [Google Scholar] [CrossRef] [PubMed]
- Caprioglio, D.; Mattoteia, D.; Minassi, A.; Pollastro, F.; Lopatriello, A.; Muňoz, E.; Taglialatela-Scafati, O.; Appendino, G. One-Pot Total Synthesis of Cannabinol via Iodine-Mediated Deconstructive Annulation. Org. Lett. 2019, 21, 6122–6125. [Google Scholar] [CrossRef] [PubMed]
- Costantino, L.; Barlocco, D. Privileged Structures as Leads in Medicinal Chemistry. Curr. Med. Chem. 2006, 13, 65–85. [Google Scholar] [CrossRef]
- Novák, J.; Salemink, C.A. Cannabis Xxiv. A New Convenient Synthesis of Cannabinol. Tetrahedron Lett. 1982, 23, 253–254. [Google Scholar] [CrossRef]
- Hattori, T.; Suzuki, T.; Miyano, S. A Practical and Efficient Method for the Construction of the Biphenyl Framework; Nucleophilic Aromatic Substitution on 2-Methoxybenzoates with Aryl Grignard Reagents. J. Chem. Soc. Chem. Commun. 1991, 1375–1376. [Google Scholar] [CrossRef]
- Nüllen, M.; Göttlich, R. Synthesis of Cannabinol by a Modified Ullmann-Ziegler Cross-Coupling. Synlett 2013, 24, 1109–1112. [Google Scholar] [CrossRef]
- Li, Y.; Ding, Y.-J.; Wang, J.-Y.; Su, Y.-M.; Wang, X.-S. Pd-Catalyzed C–H Lactonization for Expedient Synthesis of Biaryl Lactones and Total Synthesis of Cannabinol. Org. Lett. 2013, 15, 2574–2577. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.-D.; Li, B.; Wang, D.-Y.; Gao, Y.-R.; Guo, S.-H.; Pan, G.-F.; Wang, Y.-Q. Synthesis of 6 H -Benzo[ c ]Chromenes via Palladium-Catalyzed Intramolecular Dehydrogenative Coupling of Two Aryl C–H Bonds. Org. Lett. 2017, 19, 798–801. [Google Scholar] [CrossRef] [PubMed]
- Kloss, F.; Neuwirth, T.; Haensch, V.G.; Hertweck, C. Metal-Free Synthesis of Pharmaceutically Important Biaryls by Photosplicing. Angew. Chem. Int. Ed. 2018, 57, 14476–14481. [Google Scholar] [CrossRef]
- Teske, J.A.; Deiters, A. A Cyclotrimerization Route to Cannabinoids. Org. Lett. 2008, 10, 2195–2198. [Google Scholar] [CrossRef] [PubMed]
- Nandaluru, P.R.; Bodwell, G.J. Multicomponent Synthesis of 6 H -Dibenzo[b,d]Pyran-6-Ones and a Total Synthesis of Cannabinol. Org. Lett. 2012, 14, 310–313. [Google Scholar] [CrossRef]
- Minuti, L.; Temperini, A.; Ballerini, E. High-Pressure-Promoted Diels–Alder Approach to Biaryls: Application to the Synthesis of the Cannabinols Family. J. Org. Chem. 2012, 77, 7923–7931. [Google Scholar] [CrossRef] [PubMed]
- Fan, F.; Dong, J.; Wang, J.; Song, L.; Song, C.; Chang, J. An Intramolecular Pyranone Diels-Alder Cycloaddition Approach to Cannabinol. Adv. Synth. Catal. 2014, 356, 1337–1342. [Google Scholar] [CrossRef]
- Mou, C.; Zhu, T.; Zheng, P.; Yang, S.; Song, B.-A.; Chi, Y.R. Green and Rapid Access to Benzocoumarins via Direct Benzene Construction through Base-Mediated Formal [4+2] Reaction and Air Oxidation. Adv. Synth. Catal. 2016, 358, 707–712. [Google Scholar] [CrossRef]
- Norseeda, K.; Tummatorn, J.; Krajangsri, S.; Thongsornkleeb, C.; Ruchirawat, S. Synthesis of 6-Alkyl-6 H -Benzo[ c ]Chromene Derivatives by Cyclization/Selective Ether Cleavage in One Pot: Total Synthesis of Cannabinol. Asian J. Org. Chem. 2016, 5, 792–800. [Google Scholar] [CrossRef]
- Lopatriello, A.; Caprioglio, D.; Minassi, A.; Schiano Moriello, A.; Formisano, C.; De Petrocellis, L.; Appendino, G.; Taglialatela-Scafati, O. Iodine-Mediated Cyclization of Cannabigerol (CBG) Expands the Cannabinoid Biological and Chemical Space. Bioorg. Med. Chem. 2018, 26, 4532–4536. [Google Scholar] [CrossRef] [PubMed]
- De Petrocellis, L.; Di Marzo, V. An Introduction to the Endocannabinoid System: From the Early to the Latest Concepts. Best Pract. Res. Clin. Endocrinol. Metab. 2009, 23, 1–15. [Google Scholar] [CrossRef]
- Moreno, E.; Cavic, M.; Canela, E.I. Functional Fine-Tuning of Metabolic Pathways by the Endocannabinoid System—Implications for Health and Disease. Int. J. Mol. Sci. 2021, 22, 3661. [Google Scholar] [CrossRef] [PubMed]
- Rhee, M.-H.; Vogel, Z.; Barg, J.; Bayewitch, M.; Levy, R.; Hanuš, L.; Breuer, A.; Mechoulam, R. Cannabinol Derivatives: Binding to Cannabinoid Receptors and Inhibition of Adenylylcyclase. J. Med. Chem. 1997, 40, 3228–3233. [Google Scholar] [CrossRef] [PubMed]
- Riedel, G.; Fadda, P.; McKillop-Smith, S.; Pertwee, R.G.; Platt, B.; Robinson, L. Synthetic and Plant-Derived Cannabinoid Receptor Antagonists Show Hypophagic Properties in Fasted and Non-Fasted Mice: Cannabinoid Antagonists as Anorectic Agents. Br. J. Pharmacol. 2009, 156, 1154–1166. [Google Scholar] [CrossRef] [PubMed]
- Rock, E.M.; Kopstick, R.L.; Limebeer, C.L.; Parker, L.A. Tetrahydrocannabinolic Acid Reduces Nausea-Induced Conditioned Gaping in Rats and Vomiting in Suncus murinus: THCA, Emesis and Nausea. Br. J. Pharmacol. 2013, 170, 641–648. [Google Scholar] [CrossRef]
- Munro, S.; Thomas, K.L.; Abu-Shaar, M. Molecular Characterization of a Peripheral Receptor for Cannabinoids. Nature 1993, 365, 61–65. [Google Scholar] [CrossRef]
- Ryberg, E.; Larsson, N.; Sjögren, S.; Hjorth, S.; Hermansson, N.-O.; Leonova, J.; Elebring, T.; Nilsson, K.; Drmota, T.; Greasley, P.J. The Orphan Receptor GPR55 Is a Novel Cannabinoid Receptor: GPR55, a Novel Cannabinoid Receptor. Br. J. Pharmacol. 2007, 152, 1092–1101. [Google Scholar] [CrossRef]
- De Petrocellis, L.; Ligresti, A.; Moriello, A.S.; Allarà, M.; Bisogno, T.; Petrosino, S.; Stott, C.G.; Di Marzo, V. Effects of Cannabinoids and Cannabinoid-Enriched Cannabis Extracts on TRP Channels and Endocannabinoid Metabolic Enzymes: Novel Pharmacology of Minor Plant Cannabinoids. Br. J. Pharmacol. 2011, 163, 1479–1494. [Google Scholar] [CrossRef]
- Wong, H.; Cairns, B.E. Cannabidiol, Cannabinol and Their Combinations Act as Peripheral Analgesics in a Rat Model of Myofascial Pain. Arch. Oral Biol. 2019, 104, 33–39. [Google Scholar] [CrossRef] [PubMed]
- Jan, T.-R.; Farraj, A.K.; Harkema, J.R.; Kaminski, N.E. Attenuation of the Ovalbumin-Induced Allergic Airway Response by Cannabinoid Treatment in A/J Mice ☆. Toxicol. Appl. Pharmacol. 2003, 188, 24–35. [Google Scholar] [CrossRef]
- Elsohly, M.A.; Harland, E.; Murphy, J.C.; Wirth, P.; Waller, C.W. Cannabinoids in Glaucoma: A Primary Screening Procedure. J. Clin. Pharmacol. 1981, 21, 472S–478S. [Google Scholar] [CrossRef]
- Aiken, C.T.; Tobin, A.J.; Schweitzer, E.S. A Cell-Based Screen for Drugs to Treat Huntington’s Disease. Neurobiol. Dis. 2004, 16, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Appendino, G.; Gibbons, S.; Giana, A.; Pagani, A.; Grassi, G.; Stavri, M.; Smith, E.; Rahman, M.M. Antibacterial Cannabinoids from Cannabis sativa: A Structure−Activity Study. J. Nat. Prod. 2008, 71, 1427–1430. [Google Scholar] [CrossRef]
- Farrimond, J.A.; Whalley, B.J.; Williams, C.M. Cannabinol and Cannabidiol Exert Opposing Effects on Rat Feeding Patterns. Psychopharmacology 2012, 223, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Farrimond, J.A.; Whalley, B.J.; Williams, C.M. Non-Δ9tetrahydrocannabinol Phytocannabinoids Stimulate Feeding in Rats. Behav. Pharmacol. 2012, 23, 113–117. [Google Scholar] [CrossRef]
- InMed Announces Update on Phase 2 Clinical Trial Investigating INM-755 Cannabinol Cream for Epidermolysis Bullosa. Available online: https://investors.inmedpharma.com/news-releases/news-release-details/inmed-announces-update-phase-2-clinical-trial-investigating-inm (accessed on 15 September 2022).
- InMed Announces Results of Second Phase 1 Clinical Trial of INM-755 CBN Cream in Healthy Subjects. Available online: https://investors.inmedpharma.com/2021-01-08-InMed-Announces-Results-of-Second-Phase-1-Clinical-Trial-of-INM-755-CBN-Cream-in-Healthy-Subjects?_ga=2.241590278.568845992.1663078970-2025599024.1663078970 (accessed on 15 September 2022).
- Corroon, J. Cannabinol and Sleep: Separating Fact from Fiction. Cannabis Cannabinoid Res. 2021, 6, 366–371. [Google Scholar] [CrossRef]
- Yoshida, H.; Usami, N.; Ohishi, Y.; Watanabe, K.; Yamamoto, I.; Yoshimura, H. Synthesis and Pharmacological Effects in Mice of Halogenated Cannabinol Derivatives. Chem. Pharm. Bull. 1995, 43, 335–337. [Google Scholar] [CrossRef]
- Chesher, G.B.; Jackson, D.M.; Starmer, G.A. Interaction of Cannabis and General Anaesthetic Agents in Mice. Br. J. Pharmacol. 1974, 50, 593–599. [Google Scholar] [CrossRef]
- Takahashi, R.N.; Karniol, I.G. Pharmacological interaction between cannabinol and δ9-tetrahydrocannabinol. Psychopharmacologia 1975, 41, 277–284. [Google Scholar] [CrossRef]
- Karniol, I.G.; Shirakawa, I.; Takahashi, R.N.; Knobel, E.; Musty, R.E. Effects of ∆9-Tetrahydrocannabinol and Cannabinol in Man. Pharmacology 1975, 13, 502–512. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maioli, C.; Mattoteia, D.; Amin, H.I.M.; Minassi, A.; Caprioglio, D. Cannabinol: History, Syntheses, and Biological Profile of the Greatest “Minor” Cannabinoid. Plants 2022, 11, 2896. https://doi.org/10.3390/plants11212896
Maioli C, Mattoteia D, Amin HIM, Minassi A, Caprioglio D. Cannabinol: History, Syntheses, and Biological Profile of the Greatest “Minor” Cannabinoid. Plants. 2022; 11(21):2896. https://doi.org/10.3390/plants11212896
Chicago/Turabian StyleMaioli, Chiara, Daiana Mattoteia, Hawraz Ibrahim M. Amin, Alberto Minassi, and Diego Caprioglio. 2022. "Cannabinol: History, Syntheses, and Biological Profile of the Greatest “Minor” Cannabinoid" Plants 11, no. 21: 2896. https://doi.org/10.3390/plants11212896
APA StyleMaioli, C., Mattoteia, D., Amin, H. I. M., Minassi, A., & Caprioglio, D. (2022). Cannabinol: History, Syntheses, and Biological Profile of the Greatest “Minor” Cannabinoid. Plants, 11(21), 2896. https://doi.org/10.3390/plants11212896







