A Review of the Chemistry and Biological Activities of Acmella oleracea (“jambù”, Asteraceae), with a View to the Development of Bioinsecticides and Acaricides
Abstract
1. Introduction
2. Systematics
3. Habitat and Distribution
4. Morphological and Anatomical Features
5. Cultivation and Micropropagation
6. Ethnobotanical Uses
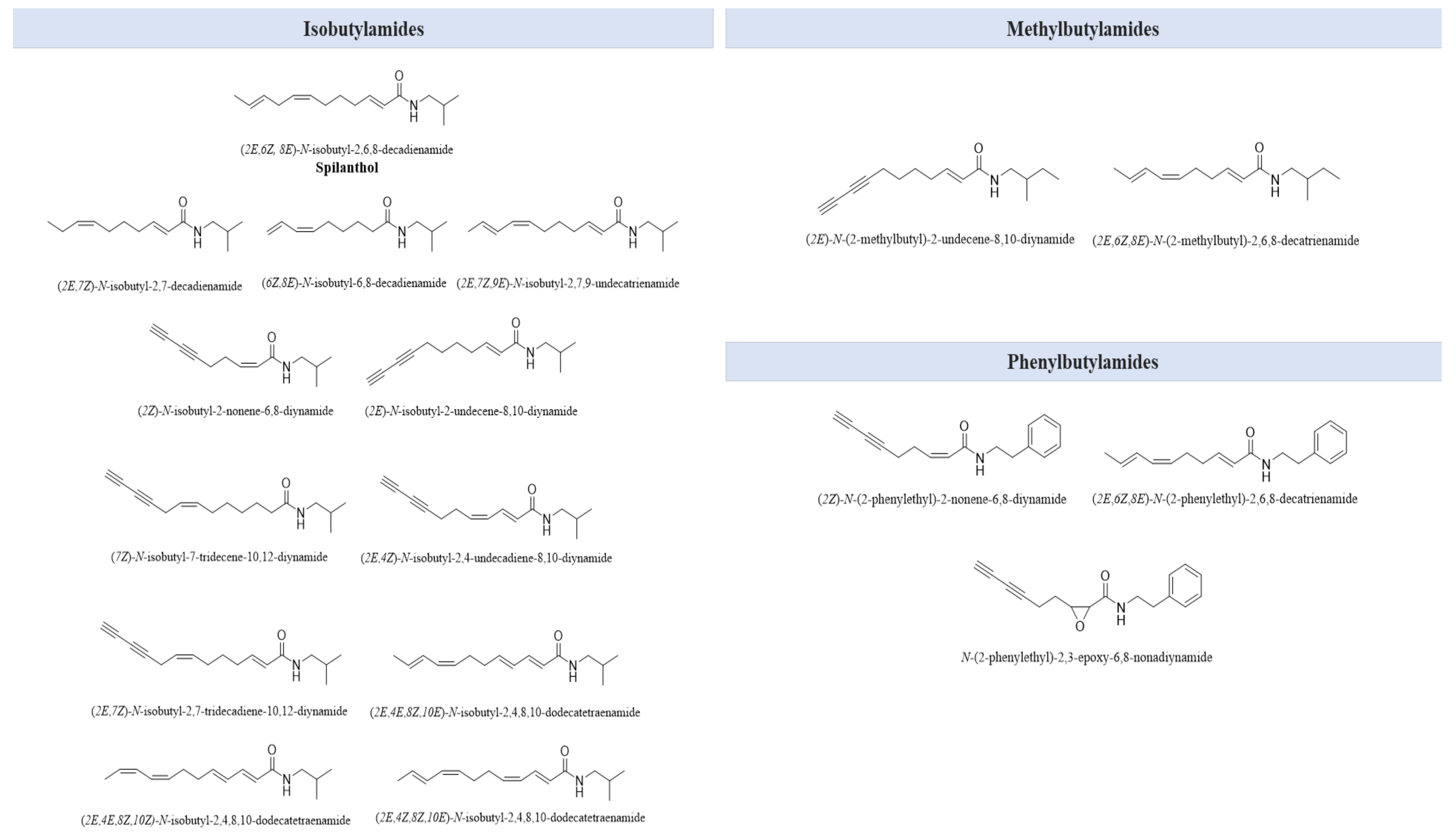
7. Secondary Metabolites
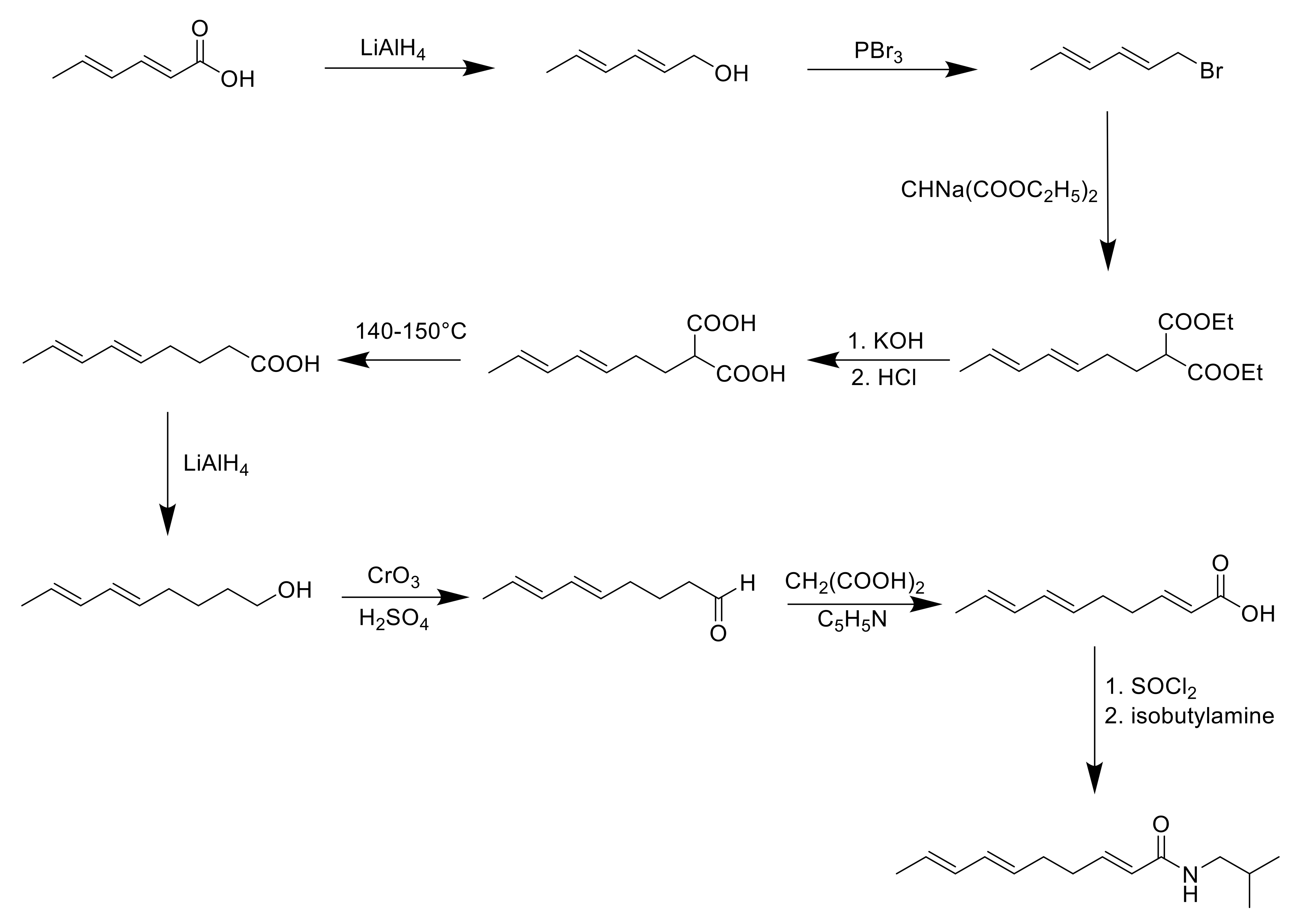
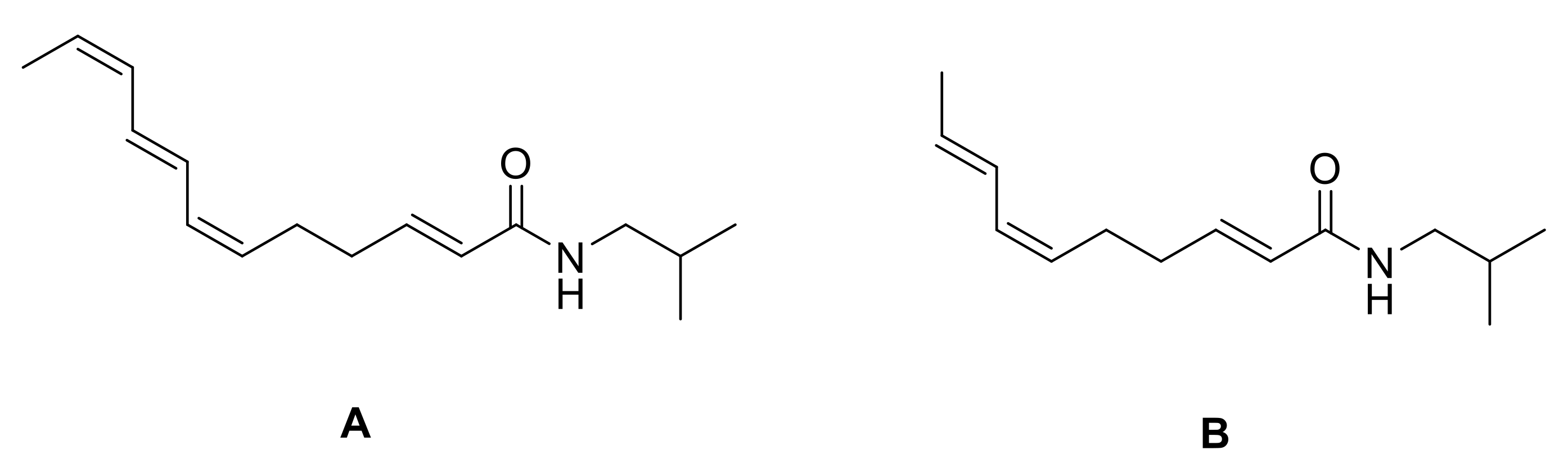
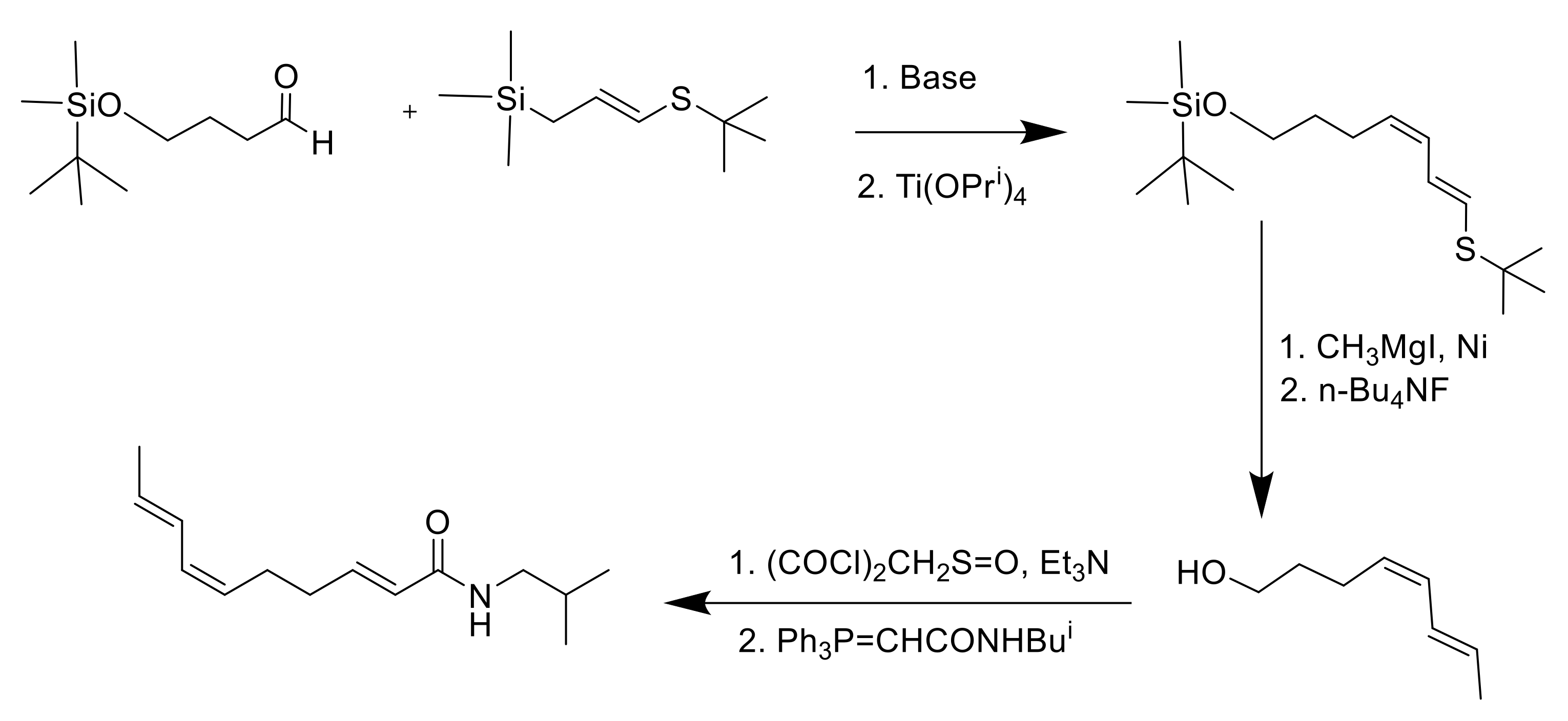
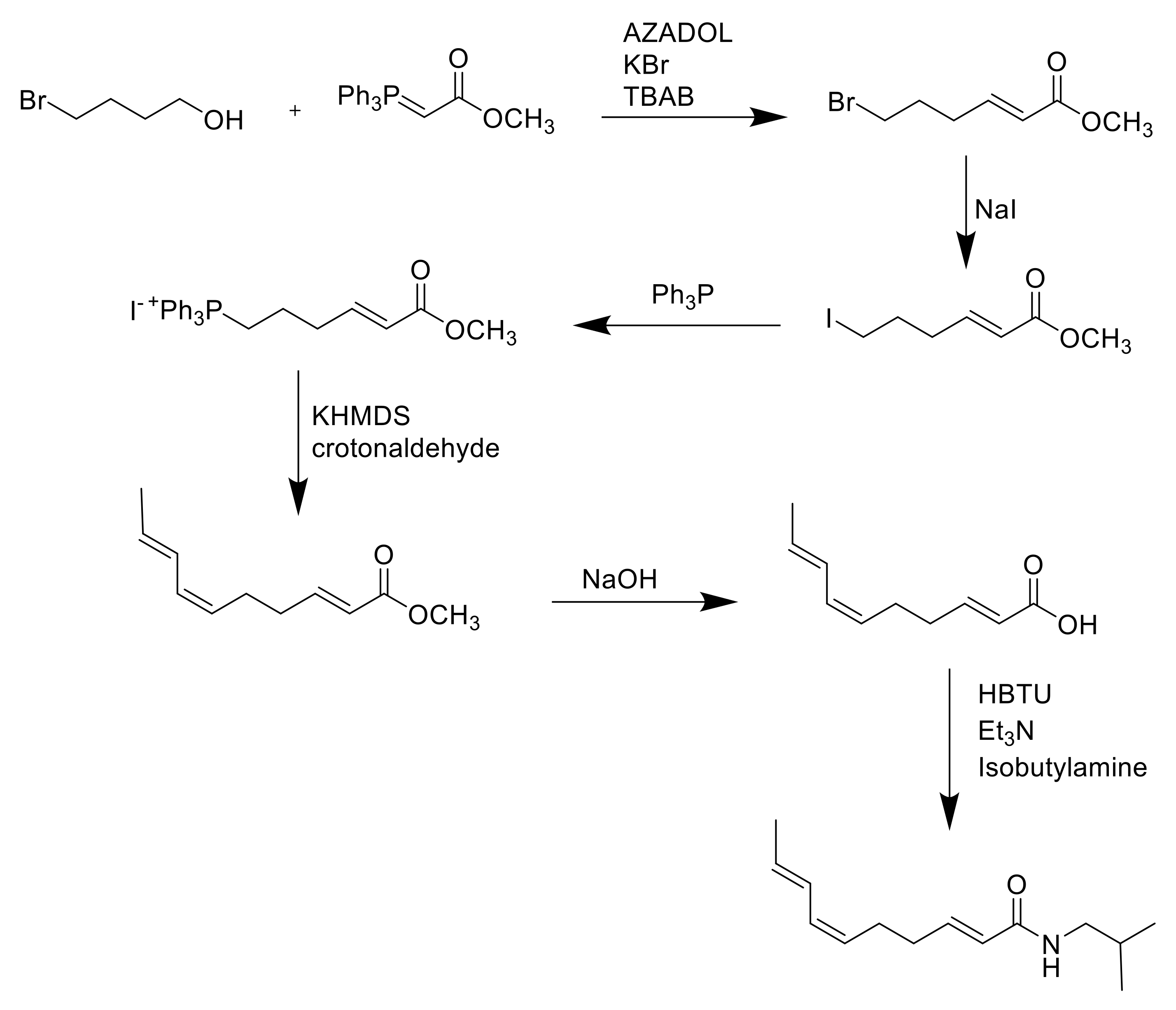
8. Synthesis of Spilanthol
9. Modes of Action of Spilanthol
10. Biological Activities
11. Insecticidal Activity
11.1. Insect Vectors
11.2. Insect Pests
12. Acaricidal Activity
12.1. Lethal Effects against Ticks
12.2. Sublethal Effects against Ticks
12.3. Side Effects on Non-Target Species
13. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pavela, R.; Morshedloo, M.R.; Mumivand, H.; Khorsand, G.J.; Karami, A.; Maggi, F.; Desneux, N.; Benelli, G. Phenolic monoterpene-rich essential oils from Apiaceae and Lamiaceae species: Insecticidal activity and safety evaluation on non-target earthworms. Entomologia 2020, 40, 421–435. [Google Scholar] [CrossRef]
- Benelli, G.; Wilke, A.B.; Bloomquist, J.R.; Desneux, N.; Beier, J.C. Overexposing mosquitoes to insecticides under global warming: A public health concern? Sci. Total Environ. 2021, 762, 143069. [Google Scholar] [CrossRef] [PubMed]
- Lucia, A.; Guzmán, E. Emulsions containing essential oils, their components or volatile semiochemicals as promising tools for insect pest and pathogen management. Adv. Colloid Interface Sci. 2021, 287, 102330. [Google Scholar] [CrossRef] [PubMed]
- Menossi, M.; Ollier, R.P.; Casalongué, C.A.; Alvarez, V.A. Essential oil-loaded bio-nanomaterials for sustainable agricultural applications. J. Chem. Technol. Biotechnol. 2021, 96, 2109–2122. [Google Scholar] [CrossRef]
- Desneux, N.; Decourtye, A.; Delpuech, J.M. The sublethal effects of pesticides on beneficial arthropods. Annu. Rev. Entomol. 2007, 52, 81–106. [Google Scholar] [CrossRef] [PubMed]
- Chloe, D.P.; Karl, W.J.W.; Jon, E.S.; Bryan, G.; Tracye, M.M.; Michael, J.L. Assessing the fate and effects of an insecticidal formulation. Environ. Toxicol. Chem. 2015, 34, 197–207. [Google Scholar] [CrossRef]
- Benelli, G.; Ceccarelli, C.; Zeni, V.; Rizzo, R.; Verde, G.L.; Sinacori, M.; Boukouvalad, M.C.; Kavallieratos, N.G.; Ubaldi, M.; Tomassoni, D.; et al. Lethal and behavioural effects of a green insecticide against an invasive polyphagous fruit fly pest and its safety to mammals. Chemosphere 2022, 287, 132089. [Google Scholar] [CrossRef]
- Czaja, K.; Góralczyk, K.; Struciński, P.; Hernik, A.; Korcz, W.; Minorczyk, M.; Łyczewska, M.; Ludwicki, J.K. Biopesticides–towards increased consumer safety in the European Union. Pest Manag. Sci. 2015, 71, 3–6. [Google Scholar] [CrossRef]
- Damalas, C.A.; Koutroubas, S.D. Current status and recent developments in biopesticide use. Agriculture 2018, 8, 13. [Google Scholar] [CrossRef]
- Seiber, J.N.; Coats, J.; Duke, S.O.; Gross, A.D. Biopesticides: State of the art and future opportunities. J. Agric. Food Chem. 2014, 62, 11613–11619. [Google Scholar] [CrossRef]
- Rondanelli, M.; Fossari, F.; Vecchio, V.; Braschi, V.; Riva, A.; Allegrini, P.; Petrangolini, G.; Iannello, G.; Faliva, M.A.; Peroni, G.; et al. Acmella oleracea for pain management. Fitoterapia 2020, 140, 104419. [Google Scholar] [CrossRef]
- Spinozzi, E.; Pavela, R.; Bonacucina, G.; Perinelli, D.R.; Cespi, M.; Petrelli, R.; Cappellacci, L.; Fiorini, D.; Scortichini, S.; Garzoli, S.; et al. Spilanthol-rich essential oil obtained by microwave-assisted extraction from Acmella oleracea (L.) R.K. Jansen and its nanoemulsion: Insecticidal, cytotoxic and anti-inflammatory activities. Ind. Crops Prod. 2021, 172, 114027. [Google Scholar] [CrossRef]
- Dubey, S.; Maity, S.; Singh, M.; Saraf, S.A.; Saha, S. Phytochemistry, Pharmacology and Toxicology of Spilanthes acmella: A Review. Adv. Pharmacol. Sci. 2013, 2013, 423750. [Google Scholar] [CrossRef]
- Benelli, G.; Pavela, R.; Drenaggi, E.; Maggi, F. Insecticidal efficacy of the essential oil of jambú (Acmella oleracea (L.) RK Jansen) cultivated in central Italy against filariasis mosquito vectors, houseflies and moth pests. J Ethnopharmacol. 2019, 229, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Gouvêa, S.M.; Carvalho, G.A.; Fidelis, E.G.; Ribeiro, A.V.; Farias, E.S.; Picanço, M.C. Effects of paracress (Acmella oleracea) extracts on the aphids Myzus persicae and Lipaphis erysimi and two natural enemies. Ind. Crops Prod. 2019, 128, 399–404. [Google Scholar] [CrossRef]
- De Araújo, I.F.; De Araújo, P.H.F.; Ferreira, R.M.A.; Sena, I.D.S.; Lima, A.L.; Carvalho, J.C.T.; Ferreina, I.M.; Souto, R.N.P. Larvicidal effect of hydroethanolic extract from the leaves of Acmella oleracea L. R. K. Jansen in Aedes aegypti and Culex quinquefasciatus. S. Afr. J. Bot. 2018, 117, 134–140. [Google Scholar] [CrossRef]
- Araújo, I.F.; Loureiro, H.A.; Marinho, V.H.S.; Neves, F.B.; Sarquis, R.S.F.; Faustino, S.M.M.; Yoshioka, S.A.; Ferreira, R.M.A.; Souto, R.N.P.; Ferreira, I.M. Larvicidal activity of the methanolic, hydroethanolic and hexanic extracts from Acmella oleracea, solubilized with silk fibroin, against Aedes aegypti. Biocatal. Agric. Biotechnol. 2020, 24, 101550. [Google Scholar] [CrossRef]
- Marchesini, P.; Barbosa, A.F.; Sanches, M.N.G.; do Nascimento, R.M.; Vale, F.L.; Fabri, R.L.; Maturano, R.; de Carvalho, M.G.; Monteiro, C. Acaricidal activity of Acmella oleracea (Asteraceae) extract against Rhipicephalus microplus: What is the influence of spilanthol? Vet 2020, 283, 109170. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, P.R.; Santos Monteiro, O.D.; da Rocha, C.Q.; Costa-Júnior, L.M.; Pinheiro Camara, M.B.; da Silva Pereira, T.C.; Soares Maia, J.G. Exposure of Rhipicephalus sanguineus sensu lato Latreille, 1806 (Acari: Ixodidae) to hexane extract of Acmella oleracea (jambù): Semi-engorged and engorged ticks. Ticks Tick Borne Dis. 2021, 12, 101705. [Google Scholar] [CrossRef] [PubMed]
- Paulraj, J.; Govindarajan, R.; Palpu, P. The genus Spilanthes ethnopharmacology, phytochemistry, and pharmacological properties: A review. Adv. Pharmacol. Sci. 2013, 2013, 510298. [Google Scholar] [CrossRef] [PubMed]
- Simas, N.K.; Dellamora, E.D.C.L.; Schripsema, J.; Lage, C.L.S.; de Oliveira Filho, A.M.; Wessjohann, L.; Porzel, A.; Kuster, R.M. Acetylenic 2-phenylethylamides and new isobutylamides from Acmella oleracea (L.) R. K. Jansen, a Brazilian spice with larvicidal activity on Aedes aegypti. Phytochemistry 2013, 6, 67–72. [Google Scholar] [CrossRef]
- Martins, C.P.S.; Melo, M.T.P.; Honório, I.C.G.; D’Ávila, V.A.; Carvalho Júnior, W.G.O. Caracterização morfológica e agronômica de acessos de jambù (Spilanthes oleracea L.) nas condições do Norte de Minas Gerais. Rev. Bras De Plantas Med. 2012, 14, 410–413. [Google Scholar] [CrossRef]
- Lee, S.H. Intestinal permeability regulation by tight junction: Implication on inflammatory bowel diseases. Intest. Res. 2015, 13, 11–18. [Google Scholar] [CrossRef]
- Mabberley, D.J.; Corner, E.J.H. A Portable Dictionary of Plants, Their Classification and Uses: Utilizing Kubitzki’s the Families and Genera of Tascular Plants. In Mabberley’s Plant-Book; Mabberley, D.J., Ed.; University Press: Cambridge, UK, 2017. [Google Scholar]
- Jansen, R.K. The systematics of Acmella (Asteraceae-Heliantheae). Syst. Bot. Monogr. 1985, 8, 1–115. [Google Scholar] [CrossRef]
- Lim, T.K. Edible Medicinal and Non-Medicinal Plants; Springer: Cham, The Netherlands, 2016; pp. 469–560. [Google Scholar] [CrossRef]
- Prachayasittikul, V.; Prachayasittikul, S.; Ruchirawat, S.; Prachayasittikul, V. High therapeutic potential of Spilanthes acmella: A review. EXCLI J. 2013, 12, 291. [Google Scholar]
- Lim, T.K. Edible medicinal and non-medicinal plants: Flowers. Edible Med. Non-Med. Plants 2014, 7, 1–1102. [Google Scholar] [CrossRef]
- Hind, N.; Biggs, N. Acmella oleracea compositae. Curtis’s Bot. Mag. 2003, 20, 31–39. [Google Scholar] [CrossRef]
- Hossan, M.S.; Hanif, A.; Bipasha, A.; Sarwar, M.S.; Karim, M.; Rahman, T.; Jahan, R.; Rahmatullah, M. Traditional Use of Medicinal Plants in Bangladesh to Treat Urinary Tract Infections and Sexually Transmitted Diseases Bay of Bengal. Ethnobot. Res. 2010, 8, 61–74. [Google Scholar] [CrossRef]
- Lalthanpuii, P.B.; Lalawmpuii, R.; Vanlaldinpuia, K.; Lalchhandama, K. Phytochemical investigations on the medicinal plant Acmella oleracea cultivated in Mizoram. India Sci. Vis. 2016, 16, 177–183. [Google Scholar]
- Sahu, J.; Jain, K.; Jain, B.; Sahu, R.K. A Review O Phytopharmacology a Micropropagatio of Spilanthes acmella. Pharmacologyonline 2011, 2, 1105–1110. [Google Scholar]
- Srinath, J.; Laksmi, T. Therapeutic potential of Spilanthes acmella-A dental note. Int. J. Pharm. Sci. Rev. Res. 2014, 25, 151–156. [Google Scholar]
- Nascimento, L.E.S.; Arriola, N.D.A.; da Silva, L.A.L.; Faqueti, L.G.; Sandjo, L.P.; de Araújo, C.E.S.; Biavatti, M.W.; Barcelos-Oliveira, J.L.; Dias de Mello Castanho Amboni, R. Phytochemical profile of different anatomical parts of jambù (Acmella oleracea (L.) R.K. Jansen): A comparison between hydroponic and conventional cultivation using PCA and cluster analysis. Food Chem. 2020, 332, 127393. [Google Scholar] [CrossRef] [PubMed]
- Uthpala, T.G.G.; Navaratne, S.B. Acmella oleracea Plant; Identification, Applications and Use as an Emerging Food Source—Review. Food Rev. Int. 2021, 37, 399–414. [Google Scholar] [CrossRef]
- Silveira, N.; Sandjo, L.P.; Biavatti, M.W. Spilanthol-containing products: A patent review (1996–2016). Trends Food Sci. Technol. 2018, 74, 107–111. [Google Scholar] [CrossRef]
- Da Borges, L.S.; Guerrero, A.C.; Goto, R.; Lima, G.P.P. Produtividade e acúmulo de nutrientes em plantas de jambù, sob adubação orgânica e mineral. Semin. Agrar. 2013, 34, 83–94. [Google Scholar] [CrossRef][Green Version]
- Souto, G.C.; Grangeiro, L.C.; de Gusmão, S.A.L.; de Sousa, V.F.L.; Cavalcante, A.E.C.; de França, F.D. Agronomic performance of jambù (Acmella oleracea) using organic fertilization. Aust. J. Crops Sci. 2018, 12, 151–156. [Google Scholar] [CrossRef]
- Da Borges, L.S.; Vieira, M.A.R.; Marques, M.O.M.; Vianello, F.; Lima, G.P.P. Influence of organic and mineral soil fertilization on essential oil of Spilanthes oleracea cv. jambuarana. J. Plant Physiol. 2012, 7, 135–142. [Google Scholar] [CrossRef]
- Da Silva Borges, L.; de Souza Vieira, M.C.; Vianello, F.; Goto, R.; Lima, G.P. Antioxidant compounds of organically and conventionally fertilized jambù (Acmella oleracea). Biol. Agric. Hortic. 2016, 32, 149–158. [Google Scholar] [CrossRef]
- Sut, S.; Ferrarese, I.; Shrestha, S.S.; Kumar, G.; Slaviero, A.; Sello, S.; Altissimo, A.; Pagni, L.; Gattesco, F.; Dall’Acqua, S. Comparison of biostimulant treatments in Acmella oleracea cultivation for alkylamides production. Plants 2020, 9, 818. [Google Scholar] [CrossRef]
- Sampaio, I.M.G.; da Silva Júnior, M.L.; de Moraes Palha Bittencourt, R.F.; dos Santos, G.A.M.; de Souza Lemos Neto, H. Production and postharvest quality of jambu in hydroponics under nitrogen application in nutrient solution1. Rev. Cienc. Agron. 2021, 52, 1–8. [Google Scholar] [CrossRef]
- Haw, A.B.; Keng, C.L. Micropropagation of Spilanthes acmella L., a bio-insecticide plant, through proliferation of multiple shoots. J. Appl. Hortic. 2003, 5, 65–68. [Google Scholar]
- Malosso, M.G.; Barbosa, E.P.; Nagao, E.O. Micropropagação de jambu [Acmella oleracea (L.) R.K. Jansen]. Rev. Bras. Plantas Med. 2008, 10, 91–95. [Google Scholar]
- Singh, S.K.; Rai, M.K.; Asthana, P.; Sahoo, L. An improved micropropagation of Spilanthes acmella L. through transverse thin cell layer culture. Acta Physiol. Plant 2009, 31, 693–698. [Google Scholar] [CrossRef]
- Singh, M.; Chaturvedi, R. Optimization of Spilanthes acmella L. cultivation by in vitro nodal segment culture. Acta Hortic. 2010, 865, 109–114. [Google Scholar] [CrossRef]
- Leng, T.C.; Ping, N.S.; Lim, B.P.; Keng, C.L. Detection of bioactive compounds from Spilanthes acmella (L.) plants and its various in vitro culture products. J. Med. Plant Res. 2011, 5, 371–378. [Google Scholar]
- Franca, J.V.; Queiroz, M.S.R.; do Amaral, B.P.; Simas, N.K.; da Silva, N.C.B.; Leal, I.C.R. Distinct growth and extractive methods of Acmella oleracea (L.) R.K. Jansen rising different concentrations of spilanthol: An important bioactive compound in human dietary. Food Res. Int. 2016, 89, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Almeida, S.P.; Souza, J.M.; Amorim, A.M.; de Gusmao, S.A.L.; Souza, R.O.; Santos, A.S. In vitro culture of jambu with different growth regulators. Hortic. Bras. 2020, 38, 134–138. [Google Scholar] [CrossRef]
- Joseph, B.; George, J.; MV, J. The role of Acmella oleracea in medicine-a review. World J. Pharm. Res. 2013, 2, 2781–2788. [Google Scholar]
- Lamarck, J.B.A.P.M. Encyclopédie Méthodique, Botanique; Chez Panckoucke: Paris, France, 1785; Volume 1, p. 563. [Google Scholar]
- Ochse, J.J.; van den Brink, R.C.B. Vegetables of the Dutch; Indies Ascher & Co.: Amsterdam, The Netherlands, 1980. [Google Scholar]
- Bailey, L.H. Manual of Cultivated Plants: Most Commonly Grown in the Continental United Stated and Canada; Macmillan: New York, NY, USA, 1949. [Google Scholar]
- Chung, K.F.; Kono, Y.; Wang, C.M.; Peng, C.I. Notes on Acmella (Asteraceae: Heliantheae) in Taiwan. Bot. Stud. 2008, 49, 73–82. [Google Scholar]
- Burdock, G.A. Fenaroli’s Handbook of Flavor Ingredients, 5th ed.; CRC Press: Boca Raton, FL, USA, 2010; p. 1864. [Google Scholar]
- Ramsewak, R.S.; Erickson, A.J.; Nair, M.G. Bioactive N-isobutylamides from the flower buds of Spilanthes acmella. Phytochemistry 1999, 51, 729–732. [Google Scholar] [CrossRef]
- Lalthanpuii, P.B.; Lalchhandama, K. Anticancer and DNA-protecting potentials of Spilanthes acmella (toothache plant) grown in Mizoram, India. J. Nat. Remedies 2019, 19, 57–63. [Google Scholar] [CrossRef]
- Lalthanpuii, P.B.; Lalchhandama, K. Chemical composition and broad-spectrum anthelmintic activity of a cultivar of toothache plant, Acmella oleracea, from Mizoram, India. Pharm. Biol. 2020, 58, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Purushothaman, Y.; Gunaseelan, S.; Vijayakumar, S.D. Spilanthes acmella and its medicinal uses—A review. Asian J. Pharm. Clin. Res. 2018, 11, 45–49. [Google Scholar] [CrossRef][Green Version]
- Favoreto, R.; Gilbert, B. Acmella oleracea (L.) R. K. Jansen (Asteraceae)—Jambu Rev. Fitos 2010, 5, 83–91. [Google Scholar]
- Yamane, L.T.; De Paula, E.; Jorge, M.P.; De Freitas-Blanco, V.S.; Junior, Í.M.; Figueira, G.M.; Anholeto, L.A.; De Oliveira, P.R.; Rodrigues, R.A.F. Acmella oleracea and Achyrocline satureioides as Sources of Natural Products in Topical Wound Care. Evidence-based Complement. Altern. Med. 2016, 2016, 3606820. [Google Scholar] [CrossRef]
- Araújo, C.A.; Sant, C.; Morgado, A.; Katherine, A.; Gomes, C.; Caroline, A.; Gomes, C.; Simas, N.K. Asteraceae family: A review of its allelopathic potential and the case of Acmella oleracea and Sphagneticola trilobata. Rodriguésia 2021, 72. [Google Scholar] [CrossRef]
- Abeysiri, G.R.P.I.; Dharmadasa, R.M.; Abeysinghe, D.C.; Samarasinghe, K. Screening of phytochemical, physico-chemical and bioactivity of different parts of Acmella oleraceae Murr. (Asteraceae), a natural remedy for toothache. Ind. Crops Prod. 2013, 50, 852–856. [Google Scholar] [CrossRef]
- Prachayasittikul, S.; Suphapong, S.; Worachartcheewan, A.; Lawung, R.; Ruchirawat, S.; Prachayasittikul, V. Bioactive metabolites from Spilanthes acmella Murr. Molecules 2009, 14, 850–867. [Google Scholar] [CrossRef] [PubMed]
- Baruah, R.N.; Leclercq, P.A. Characterization of the essential oil from flower heads of Spilanthes acmella. J. Essent. 1993, 5, 693–695. [Google Scholar] [CrossRef]
- Sharma, R.; Arumugam, N. N-alkylamides of Spilanthes (syn: Acmella): structure, purification, characterization, biological activities and applications–A review. Future Foods 2021, 3, 100022. [Google Scholar] [CrossRef]
- Cheng, Y.B.; Liu, R.H.; Ho, M.C.; Wu, T.Y.; Chen, C.Y.; Lo, I.W.; Chang, F.R. Alkylamides of Acmella oleracea. Molecules 2015, 20, 6970–6977. [Google Scholar] [CrossRef] [PubMed]
- Savic, S.; Petrovic, S.; Savic, S.; Cekic, N. Identification and photostability of N-alkylamides from Acmella oleracea extract. J. Pharm. Biomed. 2021, 195, 113819. [Google Scholar] [CrossRef] [PubMed]
- Gerber, E. Uber die chemischen Bestandteile der Parakresse (Spilanthes olearacea, Jacquin). Arch. Pharm. 1903, 241, 270–289. [Google Scholar] [CrossRef]
- Jacobson, M. Constituents of Heliopsis Species. IV. The Total Synthesis of trans-Affinin. J. Am. Chem. Soc. 1955, 77, 2461–2463. [Google Scholar] [CrossRef]
- Crombie, L.; Krasinski, A.H.A.; Manzoor-i-Khuda, M. Amides of vegetable origin. Part X. The stereochemistry and synthesis of affinin. J. Chem. Soc. 1963, 4970–4976. [Google Scholar] [CrossRef]
- Ikeda, Y.; Ukai, J.; Ikeda, N.; Yamamoto, H. Facile routes to natural acyclic polyenes syntheses of spilanthol and trail pheromone for termite. Tetrahedron Lett. 1984, 25, 5177–5180. [Google Scholar] [CrossRef]
- Wang, Z.; Lu, X.; Lei, A.; Zhang, Z. Efficient preparation of functionalized (E,Z) dienes using acetylene as the building block. J. Org. Chem. 1998, 63, 3806–3807. [Google Scholar] [CrossRef]
- Tanaka, S.; Yagi, K.; Ujihara, H.; Ishida, K. WO2009/091040A1.
- Lombardo, L.; Lankin, M.E.; Ishida, K.; Tanaka, S.; Ujihara, H.; Yagi, K.; Mei, J.B.; Green, C.B.; Mankoo, A.S. US2010/0184863A1.
- Tanaka, S.; Yagi, K.; Ujihara, H.; Ishida, K. US2011/0105773A1.
- Tanaka, S.; Yagi, K.; Ujihara, H.; Ishida, K. WO2011/007807A1.
- Tanaka, S.; Ishida, K.; Yagi, K.; Ujihara, H. US2012/0116116A1.
- Alonso, I.G.; Yamane, L.T.; de Freitas-Blanco, V.S.; Novaes, L.F.; Franz-Montan, M.; de Paula, E.; Rodrigues, M.V.N.; Rodrigues, R.A.F.; Pastre, J.C. A new approach for the total synthesis of spilanthol and analogue with improved anesthetic activity. Tetrahedron 2018, 74, 5192–5199. [Google Scholar] [CrossRef]
- Nakamura, A.; Mimaki, K.; Tanigami, K.I.; Maegawa, T. An improved and practical method for synthesizing of α-sanshools and spilanthol. Front. Chem. 2020, 8, 187. [Google Scholar] [CrossRef]
- Boonen, J.; Baert, B.; Roche, N.; Burvenich, C.; de Spiegeleer, B. Transdermal behaviour of the N-alkylamide spilanthol (affinin) from Spilanthes acmella (Compositae) extracts. J. Ethnopharmacol. 2010, 127, 77–84. [Google Scholar] [CrossRef]
- Veryser, L.; Taevernier, L.; Joshi, T.; Tatke, P.; Wynendaele, E.; Bracke, N.; Stalmans, S.; Peremans, K.; Burvenich, C.; Risseeuw, M.; et al. Mucosal and blood-brain barrier transport kinetics of the plant N-alkylamide spilanthol using in vitro and in vivo models. BMC Complement. Altern. Med. 2016, 16, 177. [Google Scholar] [CrossRef] [PubMed]
- Déciga-Campos, M.; Rios, M.Y.; Aguilar-Guadarrama, A.B. Antinociceptive effect of Heliopsis longipes extract and affinin in mice. Planta Med. 2010, 76, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Bakondi, E.; Singh, S.B.; Hajnády, Z.; Nagy-Pénzes, M.; Regdon, Z.; Kovács, K.; Hegedűs, C.; Madácsy, T.; Maléth, J.; Hegyi, P.; et al. Spilanthol inhibits inflammatory transcription factors and iNOS expression in macrophages and exerts anti-inflammatory effects in dermatitis and pancreatitis. Int. J. Mol. Sci. 2019, 20, 4308. [Google Scholar] [CrossRef]
- Huang, W.C.; Wu, L.Y.; Hu, S.; Wu, S.J. Spilanthol inhibits COX-2 and ICAM-1 expression via suppression of NF-κB and MAPK signaling in interleukin-1β-stimulated human lung epithelial cells. Inflammation 2018, 41, 1934–1944. [Google Scholar] [CrossRef] [PubMed]
- Buitimea-Cantúa, G.V.; Buitimea-Cantúa, N.E.; del Refugio Rocha-Pizaña, M.; Rosas-Burgos, E.C.; Hernández-Morales, A.; Molina-Torres, J. Antifungal and anti-aflatoxigenic activity of Heliopsis longipes roots and affinin/spilanthol against Aspergillus parasiticus by downregulating the expression of alf D and afl R genes of the aflatoxins biosynthetic pathway. J. Environ. 2020, 55 Part B, 210–219. [Google Scholar] [CrossRef]
- Arriaga-Alba, M.; Rios, M.Y.; Déciga-Campos, M. Antimutagenic properties of affinin isolated from Heliopsis longipes extract. Pharm. Biol. 2013, 51, 1035–1039. [Google Scholar] [CrossRef]
- Barbosa, A.F.; Carvalho, M.G.D.; Smith, R.E.; Sabaa-Srur, A.U. Spilanthol: Occurrence, extraction, chemistry and biological activities. Rev. Bras Farmacogn. 2016, 26, 128–133. [Google Scholar] [CrossRef]
- Pavela, R.; Maggi, F.; Iannarelli, R.; Benelli, G. Plant extracts for developing mosquito larvicides: From laboratory to the field, with insights on the modes of action. Acta Trop. 2019, 193, 236–271. [Google Scholar] [CrossRef]
- Vishwanathan, S.; Nandan, N.; Anitha, C.; Manjushree, R. The Buzz Button to Your Toothache–Spilanthes acmella: A Review. J. Ayurveda Integr. Med. Sci. 2021, 6, 77–81. [Google Scholar]
- De Freitas-Blanco, V.S.; Monteiro, K.M.; de Oliveira, P.R.; de Oliveira, E.C.S.; de Oliveira Braga, L.E.; de Carvalho, J.E.; Rodrigues, R.A.F. Spilanthol, the principal alkylamide from Acmella oleracea, attenuates 5-fluorouracil-induced intestinal mucositis in mice. Planta Med. 2019, 85, 203–209. [Google Scholar] [CrossRef]
- Rios, M.Y.; Aguilar-Guadarrama, A.B.; del Carmen Gutiérrez, M. Analgesic activity of affinin, an alkamide from Heliopsis longipes (Compositae). J. Ethnopharmacol. 2007, 110, 364–367. [Google Scholar] [CrossRef] [PubMed]
- Nomura, E.C.O.; da Silva, C.F.; Rodrigues, M.R.A.; Hamm, L.A. Antinociceptive effects of ethanolic extract from the flowers of Acmella oleracea (L.) R.K. Jansen in mice. J. Ethnopharmacol. 2013, 150, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.M.; Mohamad, A.S.; Makhtar, N.A.; Khalid, M.H.; Khalid, S.; Perimal, E.K.; Mastuki, S.N.; Zakariaa, Z.A.; Lajis, N.; Israf, D.A.; et al. Antinociceptive activity of methanolic extract of Acmella uliginosa (Sw.) Cass. J. Ethnopharmacol. 2011, 133, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Madrid, I.I.; Castañeda-Hernández, G.; Cilia-López, V.G.; Cariño-Cortés, R.; Pérez-Hernández, N.; Fernández-Martínez, E.; Ortiz, M.I. Interaction between Heliopsis Longipes Extract and Diclofenac on the Thermal Hyperalgesia Test. Phytomedicine 2009, 16, 336–341. [Google Scholar] [CrossRef]
- Lalthanpuii, P.B.; Lalruatfela, B.; Vanlaldinpuia, K.; Lalremsanga, H.T.; Lalchhandama, K. Antioxidant and cytotoxic properties of Acmella oleracea. Int. J. Phytomed. Ind. 2018, 10, 353–358. [Google Scholar] [CrossRef]
- Stein, R.; Berger, M.; Santana de Cecco, B.; Mallmann, L.P.; Terraciano, P.B.; Driemeier, D.; Rodrigues, E.; Beys-da-Silva, W.O.; Konrath, E.L. Chymase inhibition: A key factor in the anti-inflammatory activity of ethanolic extracts and spilanthol isolated from Acmella oleracea. J. Ethnopharmacol. 2021, 270, 113610. [Google Scholar] [CrossRef]
- Abdul Rahim, R.; Jayusman, P.A.; Muhammad, N.; Mohamed, N.; Lim, V.; Ahmad, N.H.; Mohamad, S.; Hamid, Z.A.A.; Ahmad, F.; Mokhtar, N.; et al. Potential Antioxidant and Anti-Inflammatory Effects of Spilanthes acmella and Its Health Beneficial Effects: A Review. Int. J. Environ. 2021, 18, 3532. [Google Scholar] [CrossRef]
- De Alcantara, B.N.; Kobayashi, Y.T.; Barroso, K.F.; da Silva, I.D.R.; de Almeida, M.B.; Barbosa, W.L.R. Pharmacognostic analyses and evaluation of the in vitro antimicrobial activity of Acmella oleracea (L.) RK Jansen (jambù) floral extract and fractions. J. Med. Plant Res. 2015, 9, 91–96. [Google Scholar] [CrossRef]
- Onoriode, O.; Oshomoh, E.O. Antibacterial Activity of Methanol and Chloroform Extracts of Spilanthes oleracea Plant on Isolated Pathogenic Oral Bacteria. JASEM 2018, 22, 237. [Google Scholar] [CrossRef]
- Uthpala, T.G.G.; Munasinghe, H.H.; Peiris, L.D.C.; Navaratne, S.B. Evaluation of antimicrobial potential and phytochemicals in Acmella (A. oleracea) flower pod extracts subjected to different drying techniques. J. Food Process. Preserv. 2021, 45, e15570. [Google Scholar] [CrossRef]
- Peretti, P.; Rodrigues, E.T.; de Souza Junior, B.M.; Bezerra, R.M.; Fernandez, E.G.; de Sousa, F.F.O.; Pinheiro, M.T. Spilanthol content of Acmella oleracea subtypes and their bactericide and antibiofilm activities against Streptococcus mutans. S. Afr. J. Bot. 2021, 143, 17–24. [Google Scholar] [CrossRef]
- Moro, S.D.D.; de Oliveira Fujii, L.; Rodrigues, L.F.; Katleen Frauz, T.; Fernandes Mazoni, A.; Maretto Esquisatto, M.A.; Ferreira Rodrigues, R.A.; Pimentel, E.R.; Aparecida de Aro, A. Acmella oleracea Extract Increases Collagen Content and Organization in Partially Transected Tendons. Microsc 2021, 84, 2588–2597. [Google Scholar] [CrossRef] [PubMed]
- Regadas, R. Efeito Do Creme De Jambu (Acmella oleracea) Sobre a Função Sexual Masculina E Feminina. 2008, 72. Available online: http://www.repositorio.ufc.br/handle/riufc/7634 (accessed on 1 September 2022).
- Sharma, V.; Boonen, J.; Chauhan, N.S.; Thakur, M.; De Spiegeleer, B.; Dixit, V.K. Spilanthes acmella ethanolic 2021flower extract: LC–MS alkylamide profiling and its effects on sexual behavior in male rats. Phytomedicine 2011, 18, 1161–1169. [Google Scholar] [CrossRef]
- Sharma, V.; Boonen, J.; Spiegeleer, B.D.; Dixit, V.K. Androgenic and spermatogenic activity of alkylamide-rich ethanol solution extract of Anacyclus pyrethrum DC. Phytother. Res. 2013, 27, 99–106. [Google Scholar] [CrossRef]
- Lira Batista, L.; Corrêa do Nascimento, L.; Francisconi Guimarães, G.; Matias Pereira, A.C.; Koga, R.; de Carvalho, R.; Teixeira dos Santos, A.V.T.L.; Fernandes, C.P.; Teixeira, T.A.; Hu, Y.; et al. A Review of Medicinal Plants Traditionally used to Treat Male Sexual Dysfunctions-the Overlooked Potential of Acmella oleracea (L.) RK Jansen. PhCog Rev. 2021, 15, 1–11. [Google Scholar] [CrossRef]
- Savic, S.M.; Cekic, N.D.; Savic, S.R.; Ilic, T.M.; Savic, S.D. All-natural’anti-wrinkle emulsion serum with Acmella oleracea extract: A design of experiments (DoE) formulation approach, rheology and in vivo skin performance/efficacy evaluation. Int. J. Cosmeti. Sci. 2021, 43, 530–546. [Google Scholar] [CrossRef]
- Duangjai, A.; Phiphitphibunsuk, W.; Klomkiao, N.; Rodjanaudomwuttikul, P.; Ruangpoom, P.; Autthakitmongkol, S.; Ontawong, A.; Kamkaew, N.; Utsinthong, M.; Saokaew, S. Spasmolytic effect of Acmella oleracea flowers extract on isolated rat ileum. J. Herb. Med. Pharmacol. 2021, 10, 109–115. [Google Scholar] [CrossRef]
- Barbosa, A.F.; Silva, K.C.; de Oliveira, M.C.; Carvalho, M.G.D.; Srur, A.U. Effects of Acmella oleracea methanolic extract and fractions on the tyrosinase enzyme. Rev. Bras. Farmacogn. 2016, 26, 321–325. [Google Scholar] [CrossRef]
- Chaniad, P.; Phuwajaroanpong, A.; Techarang, T.; Viriyavejakul, P.; Chukaew, A.; Punsawad, C. Antiplasmodial activity and cytotoxicity of plant extracts from the Asteraceae and Rubiaceae families. Heliyon 2022, 8, e08848. [Google Scholar] [CrossRef]
- Gerbino, A.; Schena, G.; Milano, S.; Milella, L.; Barbosa, A.F.; Armentano, F.; Procino, G.; Svelto, M.; Carmosino, M. Spilanthol from Acmella oleracea lowers the intracellular levels of cAMP impairing NKCC2 phosphorylation and water channel AQP2 membrane expression in mouse kidney. PLoS ONE 2016, 11, e0156021. [Google Scholar] [CrossRef]
- Maria-Ferreira, D.; da Silva, L.M.; Mendes, D.A.G.B.; Cabrini, D.; Nascimento, A.M.; Iacomini, M. Rhamnogalacturonan from Acmella oleracea (L.) R.K. Jansen: Gastroprotective and Ulcer Healing Properties in Rats. PLoS ONE 2014, 9, e84762. [Google Scholar] [CrossRef] [PubMed]
- Maria-Ferreira, D.; Dallazen, J.L.; Corso, C.R.; Nascimento, A.M.; Cipriani, T.R.; da Silva Watanabe, P.; de Mello Gonçales Sant’Ana, D.; Baggio, C.H.; de Paula Werner, M.F. Rhamnogalacturonan polysaccharide inhibits inflammation and oxidative stress and alleviates visceral pain. J. Funct. Foods 2021, 82, 104483. [Google Scholar] [CrossRef]
- Benelli, G.; Jeffries, C.L.; Walker, T. Biological Control of Mosquito Vectors: Past, Present, and Future. Insects 2016, 7, 52. [Google Scholar] [CrossRef]
- Pandey, V.; Agrawal, V.; Raghavendra, K.; Dash, A.P. Strong larvicidal activity of three species of Spilanthes (Akarkara) against malaria (Anopheles stephensi Liston, Anopheles culicifacies, species C) and filaria vector (Culex quinquefasciatus Say). Parasitol. Res. 2007, 102, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Benelli, G.; Pavela, R.; Petrelli, R.; Cappellacci, L.; Santini, G.; Fiorini, D.; Sut, S.; Dall’Acqua, S.; Canale, A.; Maggi, F. The essential oil from industrial hemp (Cannabis sativa L.) by-products as an effective tool for insect pest management in organic crops. Ind. Crops Prod. 2018, 122, 308–315. [Google Scholar] [CrossRef]
- Kadir, H.A.; Zakaria, M.B.; Kechil, A.A.; Azirun, M.D.S. Toxicity and electrophysiological effects of Spilanthes amella Murr. extracts on Periplaneta americana L. Pestic. Sci. 1989, 25, 329–335. [Google Scholar] [CrossRef]
- Moreno, S.C.; Carvalho, G.A.; Picanço, M.C.; Morais, E.G.; Pereira, R.M. Bioactivity of compounds from Acmella oleracea against Myzus absoluta (Meyrick) (Lepidoptera: Gelechiidae) and selectivity to two non-target species. Pest Manag. 2012, 68, 386–393. [Google Scholar] [CrossRef]
- Sharma, A.; Kumar, V.; Rattan, R.S.; Kumar, N.; Singh, B. Insecticidal toxicity of spilanthol from Spilanthes acmella Murr. Against Plutella xylostella L. Am. J. Plant Sci. 2012, 3, 1568–1572. [Google Scholar] [CrossRef]
- Ogban, E.; Ukpong, I.; Oku, E.; Usua, E.; Udo, S.; Ogbeche, J.; Ajang, R. Potentials of two indigenous plants powder for the control of stored maize weevil, Sitophilus zeamais (Motschulsky). Am. J. Exp. Agri. 2015, 5, 12–17. [Google Scholar] [CrossRef]
- Gasparin, G.; Miyata, M.; Coutinho, L.L.; Martinez, M.L.; Teodoro, R.L.; Furlong, J.; Regitano, L.C.A. Mapping of quantitative trait loci controlling tick [Riphicephalus (Boophilus) microplus] resistance on bovine chromosomes 5, 7 and 14. Anim. Genet. 2007, 38, 453–459. [Google Scholar] [CrossRef]
- Mendes, M.C.; Lima, C.K.P.; Nogueira, A.H.C.; Yoshihara, E.; Chiebao, D.P.; Gabriel, F.H.L.; Klafke, G.M. Resistance to cypermethrin, deltamethrin and chlorpyriphos in populations of Rhipicephalus (Boophilus) microplus (Acari: Ixodidae) from small farms of the State of São Paulo, Brazil. Vet 2011, 178, 383–388. [Google Scholar] [CrossRef]
- Castro, K.N.; Lima, D.F.; Vasconcelos, L.C.; Leite, J.R.; Santos, R.C.; Paz Neto, A.A.; Costa-Júnior, L.M. Acaricide activity in vitro of Acmella oleracea against Rhipicephalus microplus. Parasitol 2014, 113, 3697–3701. [Google Scholar] [CrossRef] [PubMed]
- Cruz, P.B.; Barbosa, A.F.; Zeringóta, V.; Melo, D.; Novato, T.; Fidelis, Q.C.; Fabri, R.L.; de Carvalho, M.G.; Oliveira Sabaa-Srur, A.U.; Daemon, E.; et al. Acaricidal activity of methanol extract of Acmella oleracea L. (Asteraceae) and spilanthol on Rhipicephalus microplus (Acari: Ixodidae) and Dermacentor nitens (Acari: Ixodidae). Vet 2016, 228, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Marchesini, P.; Barbosa, A.F.; Franco, C.; Novato, T.; Sanches, M.; de Carvalho, M.G.; Fabri, R.L.; Daemon, E.; Monteiro, C. Activity of the extract of Acmella oleracea on immature stages of Amblyomma sculptum (Acari: Ixodidae). Vet 2018, 254, 147–150. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, P.R.; Anholeto, L.A.; Ferreira Rodrigues, R.A.; Arnosti, A.; Bechara, G.H.; de Carvalho Castro, K.N.; Camargo-Mathias, M.I. Cytotoxic Effects of Extract of Acmella oleracea in the Ovaries and Midgut of Rhipicephalus sanguineus Latreille, 1806 (Acari: Ixodidae) Female Ticks. J. Microsc. Ultrastruct. 2019, 7, 28–43. [Google Scholar] [CrossRef] [PubMed]
- Anholeto, L.A.; de Oliveira, P.R.; Rodrigues, R.A.F.; Yamane, L.T.; de Carvalho Castro, K.N.; Ferreira, A.R.F.; Camargo-Mathias, M.I. Toxic action of Acmella oleracea extract on the male reproductive system of Amblyomma cajennense ticks. Vet. Parasitol. 2017, 244, 164–171. [Google Scholar] [CrossRef]
- Anholeto, L.A.; de Oliveira, P.R.; Rodrigues, R.A.F.; Yamane, L.T.; Castro, N.; de Carvalho, K.; Camargo-Mathias, M.I. Morphological alterations in the ovaries of Amblyomma cajennense semi-engorged ticks exposed to ethanolic extract of Acmella oleracea. Microsc. Res. Tech. 2018, 81, 1347–1357. [Google Scholar] [CrossRef] [PubMed]
- Ponpornpisit, A.; Pirarat, N.; Suthikrai, W.; Binwihok, A. Toxicity Test of Kameng (Eclipta prostrate Linn.) and Kradhuawean (Spilanthes acmella (Linn.) Murr.) to Early Life Stage of Zebrafish (Danio rerio). Thai J. Vet. Med. 2011, 41, 523. [Google Scholar]
- De Souza, G.C.; Matias Pereira, A.C.; Viana, M.D.; Ferreira, A.M.; da Silva, I.; de Oliveira, M.; Barbosa, W.; Silva, L.B.; Ferreira, I.M.; Dos Santos, C.; et al. Acmella oleracea (L) R. K. Jansen Reproductive toxicity in zebrafish: An n vivo and in silico assessment. eCAM 2019, 2019, 1237301. [Google Scholar] [CrossRef]




| Cultivation Technique | Soil Treatment/Medium | Shoot Formation | Plant Productivity | Minerals/ Secondary Metabolites | References |
|---|---|---|---|---|---|
| In field | mineral fertilizer | nr a | 4.40 kg m−2 | high N and K content | [37] |
| In field | organic fertilizer | nr | 2.78 kg m−2 | high N and P content | [37] |
| In field | organic fertilizer and vermicompost | nr | 2.90–3.87 kg m−2 | nr | [38] |
| In field | organic fertilization | nr | nr | high levels of total phenolics and carotenoids | [40] |
| In field | conventional fertilizer | nr | nr | high levels of nitrates, vitamin C and organic N | [40] |
| In field | biostimulant | nr | enhanced of 22–25% | maintained levels of alkylamides and polyphenols | [41] |
| Hydroponic culture | nitrogen-rich nutrient solution | nr | enhanced (at 21 mmol L−1 of N) | nr | [42] |
| In vitro (axillary buds) | MS b medium supplemented with 2.0 mg L−1 of N6-benzyladenine | multiple-shoot formation from each axillary bud | nr | nr | [43] |
| In vitro (nodal segments) | MS medium supplemented with 0.1 mg L−1 kinetin | 93.33% rooted plants | nr | nr | [44] |
| In vitro (nodal segments) | MS medium with 5.0 mg dm−3 of BAP c | 97% of shoot regeneration | nr | nr | [45] |
| multiple-shoot regeneration | nr | nr | [46] | ||
| In vitro (nodal segments) | MS medium without growth regulators | nr | nr | spilanthol content: 0.98% | [48] |
| Acclimatized | in soil | nr | nr | spilanthol concentration: 9.54% | [48] |
| In field | organic fertilization | nr | nr | spilanthol concentration: 4.65% | [48] |
| In vitro (nodal segments) | MS medium with the use of 0.125 mg L−1 of the BAP | average of 2.2 sprouts/explant | nr | nr | [49] |
| Compound | Reference | |
|---|---|---|
| Triterpenoids | 3-acetylaleuritolic acid | [64] |
| β-sitostenone | ||
| stigmasterol | ||
| Steroidal glycosides | stigmasteryl-3-O-β-D-glucopyranosides | [64] |
| β-sitosteryl-3-O-β-D-glucopyranoside | ||
| Phenolics | vanillic acid | [64] |
| trans-ferulic acid | ||
| trans-isoferulic acid | ||
| Fatty acids | n-hexadecanoic acid | [46] |
| n-tetradecanoic acid | ||
| Coumarins | scopoletin | [64] |
| Volatile compounds | β-pinene, myrcene, (E)-caryophyllene, caryophyllene oxide, germacrene D, β-phellandrene, spilanthol and acmellonate | [65] |
| [14] | ||
| [12] | ||
| N-alkylamides | isobutylamides | [67] |
| methylbutylamides phenylbutylamides | [68] |
| Target | |||
|---|---|---|---|
| Species and Instar | Treatment | Efficacy | References |
| Periplaneta americana adults | A. oleracea Spilanthol crude extract | LC50 =2.46 μg/g | [115] |
| Anopheles culifacies late third/early fourth instar larvae | A. oleracea hexane extract | LC90 = 1.92 μg/mL LC50 = 0.87 μg/mL | [113] |
| Anopheles stephensi late third/early fourth instar larvae | A. oleracea hexane extract | LC90 = 7 μg/mL LC50 = 4.57 μg/mL | [113] |
| Culex quinquefasciatus late third/early fourth instar larvae | A. oleracea hexane extract | LC90 = 8.89 μg/mL LC50 = 3.11 μg/mL | [113] |
| Tuta absoluta second instar larvae | A. oleracea hexane extract | LC80 = 2.94 μg/mL LC50 = 1.83 μg/mL | [116] |
| Pluella xylostella second instar larvae | A. oleracea crude extract, methanol and hexane extract | LC50 = 1.49 × 10−3 μg/mL LC50 = 5.14 × 10−3 μg/mL | [117] |
| Sitophilus zeamais adults | A. oleracea plant powder | 100% mortality after 96 h of exposure to 1% (w/w) | [118] |
| Culex quinquefasciatus third instar larvae | A. oleracea hydroethanolic extract | LC90 = 1.92 μg/mL LC50 = 32.40 μg/mL | [13] |
| Aedes aegypti third instar larvae | A. oleracea hexane extract, topical application | LT90 = 57.05 h LC50 = 44.3 µg/adult | [13] |
| Culex quinquefasciatus third instar larvae | A. oleracea EO | LC90 = 68.24 μg/mL LC50 = 42.2 × 10−4 μg/mL | [11] |
| Musca domestica adult females | A. oleracea EO topical application | LC90 = 73.6 × 10−4 mg/mL LC50 = 44.3 µg/adult | [11] |
| Spodoptera littoralis third instar larvae | A. oleracea EO topical application | LC90 = 87.5 µg/adult LC50 = 68.1 µg/larva | [11] |
| Myzus persicae nymphs | A. oleracea ethanol extract on kale leaves: 0.01 g/mL | LC90 = 132.1 µg/larva LT50 = 3.29 h | [12] |
| Liphapis erysimi nymphs | A. oleracea ethanol extract on kale leaves: 0.01 g/mL | LT90 = 24.85 h LT50 = 8.85 h | [12] |
| Aedes aegypti third instar larvae | A. oleracea hydroethanolic extract (1), hexane extract (2), methanolic extract (3) | LC90 = 87.5 µg/adult LC50 =
| [14] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Spinozzi, E.; Ferrati, M.; Baldassarri, C.; Cappellacci, L.; Marmugi, M.; Caselli, A.; Benelli, G.; Maggi, F.; Petrelli, R. A Review of the Chemistry and Biological Activities of Acmella oleracea (“jambù”, Asteraceae), with a View to the Development of Bioinsecticides and Acaricides. Plants 2022, 11, 2721. https://doi.org/10.3390/plants11202721
Spinozzi E, Ferrati M, Baldassarri C, Cappellacci L, Marmugi M, Caselli A, Benelli G, Maggi F, Petrelli R. A Review of the Chemistry and Biological Activities of Acmella oleracea (“jambù”, Asteraceae), with a View to the Development of Bioinsecticides and Acaricides. Plants. 2022; 11(20):2721. https://doi.org/10.3390/plants11202721
Chicago/Turabian StyleSpinozzi, Eleonora, Marta Ferrati, Cecilia Baldassarri, Loredana Cappellacci, Margherita Marmugi, Alice Caselli, Giovanni Benelli, Filippo Maggi, and Riccardo Petrelli. 2022. "A Review of the Chemistry and Biological Activities of Acmella oleracea (“jambù”, Asteraceae), with a View to the Development of Bioinsecticides and Acaricides" Plants 11, no. 20: 2721. https://doi.org/10.3390/plants11202721
APA StyleSpinozzi, E., Ferrati, M., Baldassarri, C., Cappellacci, L., Marmugi, M., Caselli, A., Benelli, G., Maggi, F., & Petrelli, R. (2022). A Review of the Chemistry and Biological Activities of Acmella oleracea (“jambù”, Asteraceae), with a View to the Development of Bioinsecticides and Acaricides. Plants, 11(20), 2721. https://doi.org/10.3390/plants11202721











