Detection of Banana Mild Mosaic Virus in Musa In Vitro Plants: High-Throughput Sequencing Presents Higher Diagnostic Sensitivity Than (IC)-RT-PCR and Identifies a New Betaflexiviridae Species
Abstract
:1. Introduction
2. Materials and Methods
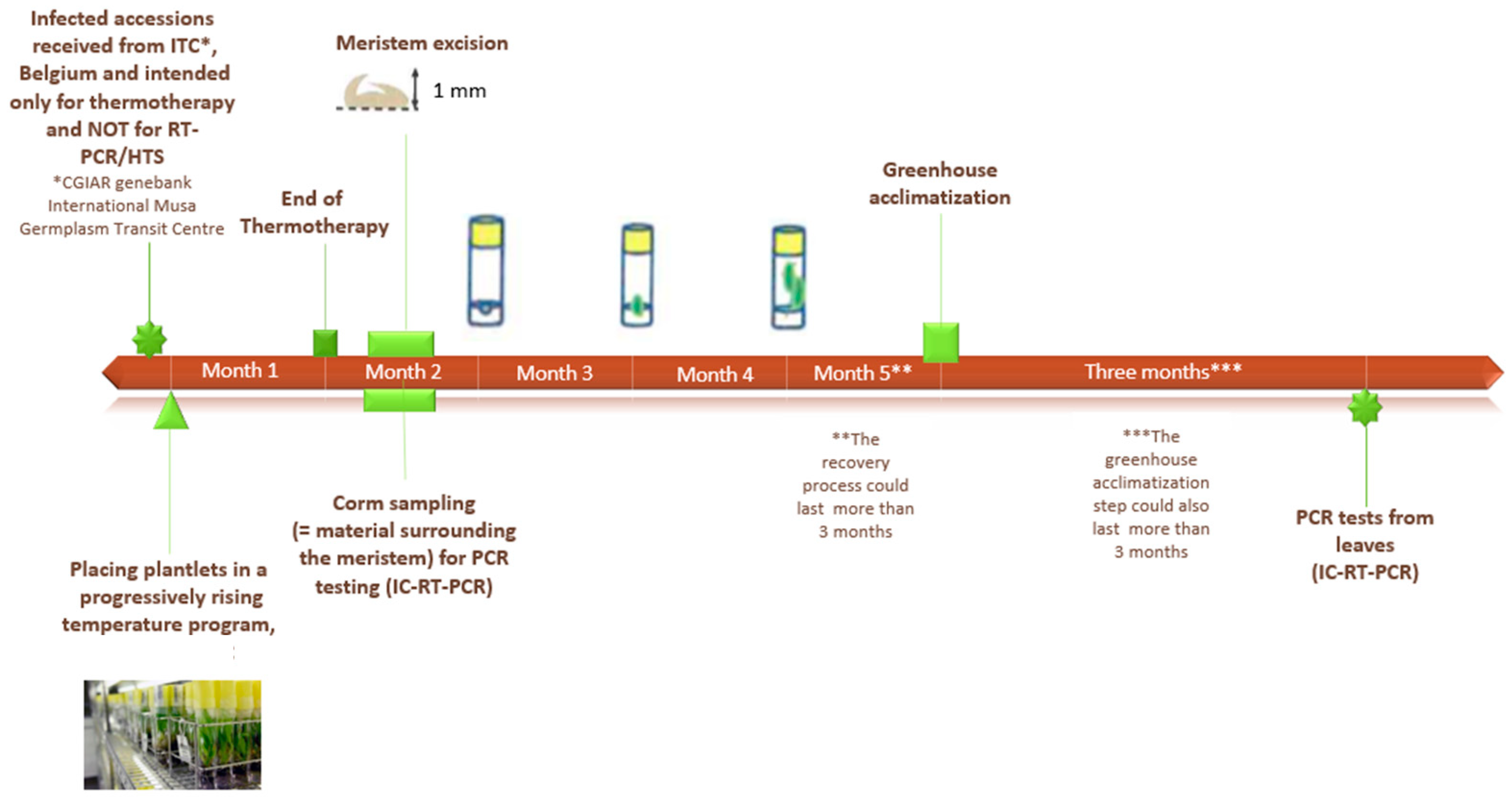
2.1. Plant Material and Tissue Culture Conditions
2.2. Sanitation Process
2.3. Sampling of In Vitro Plants
2.3.1. Meristem Sampling
2.3.2. Corm Sampling
2.3.3. The Base Sampling
2.3.4. Leaf Sampling
2.4. Sampling of Plants in Greenhouse
2.5. RNA Extraction
2.6. Molecular Assays
2.6.1. Targeted Molecular Diagnostic on In Vitro Plants
2.6.2. Targeted Molecular Diagnostic on Greenhouse Plants
2.7. Library Preparation and High-Throughput Sequencing
2.8. Statistical Analyses
2.9. Bioinformatics Analysis
2.10. Validation of Detection of the New Species
3. Results
3.1. Evaluation of Diagnostic Sensitivity of BanMMV Detection from Corm Tissues of Banana In Vitro Plants by IC-RT-PCR
3.1.1. Results on In Vitro Plants
3.1.2. Comparison of Virus Detection after Thermotherapy from Corm Tissues of In Vitro Plants and from Leaves of Greenhouse Plants Using an IC-RT-PCR Assay
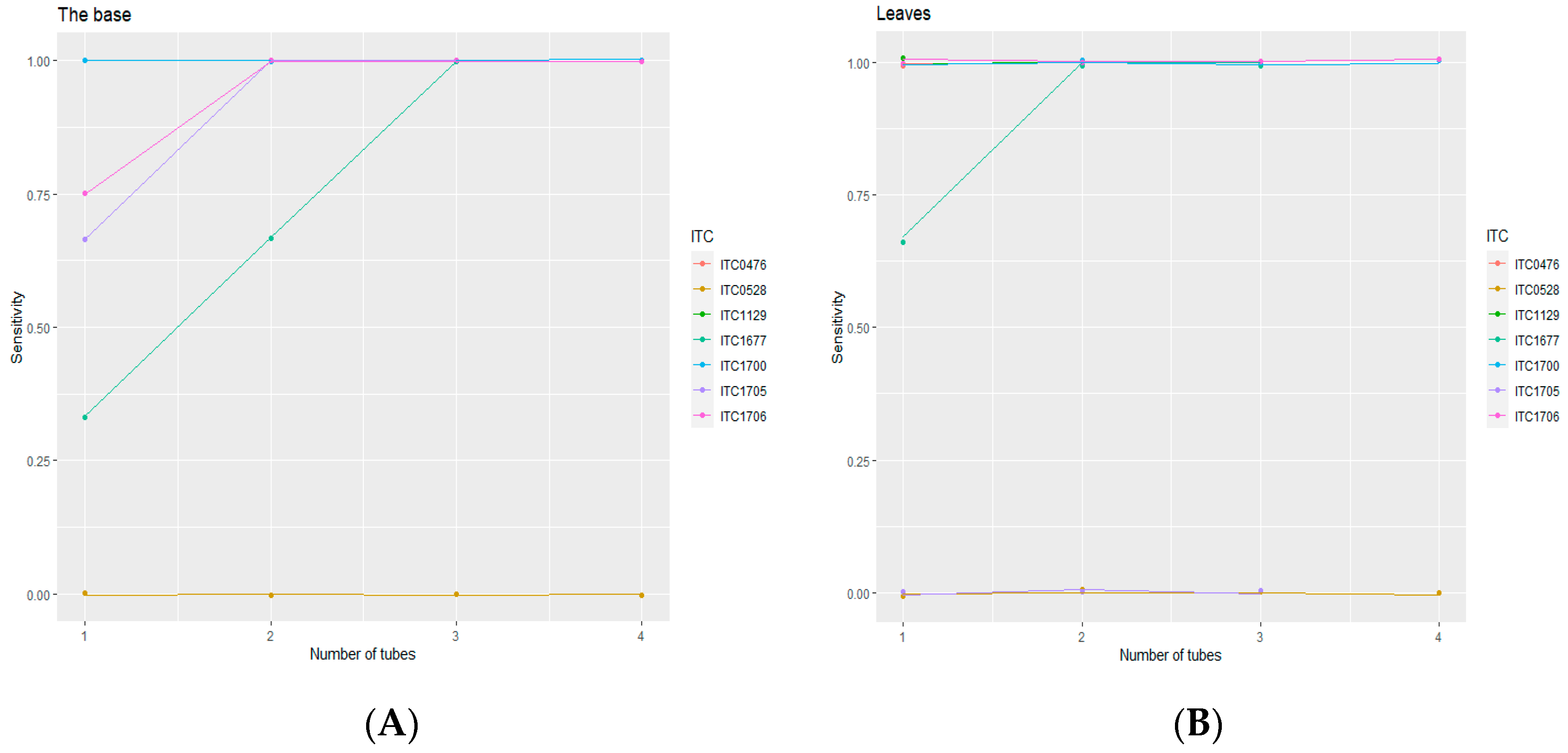
3.2. Diagnostic Sensitivity of RT-PCR from Corms and Leaves of In Vitro Plants
3.2.1. PCR Results of Infected and Healthy Banana Accessions
3.2.2. Detection of BanMMV by High-Throughput Sequencing Test on In Vitro Plants
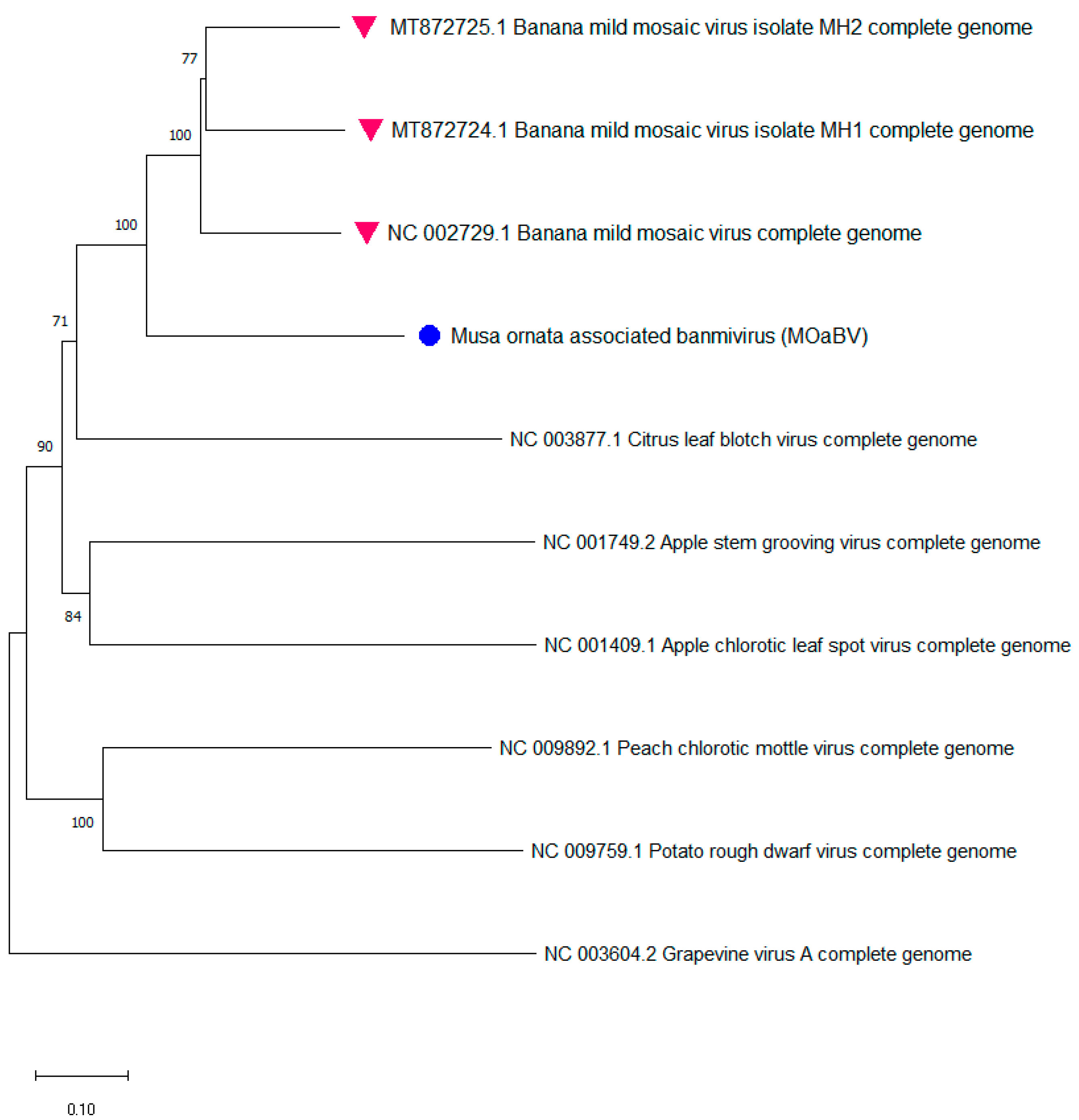
3.2.3. Identification of a New Betaflexiviridae Species Infecting Musa from ITC0528
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tripathi, L.; Ntui, V.O.; Tripathi, J.N. CRISPR/Cas9-based genome editing of banana for disease resistance. Curr. Opin. Plant Biol. 2020, 56, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Bebber, D.P. Climate change effects on Black Sigatoka disease of banana. Philos. Trans. R. Soc. B Biol. Sci. 2019, 374, 20180269. [Google Scholar] [CrossRef]
- Gambley, C.F.; Thomas, J.E. Molecular characterisation of Banana mild mosaic virus, a new filamentous virus in Musa spp. Arch. Virol. 2001, 146, 1369–1379. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.L.; Cuervo, M.; Kreuze, J.F.; Muller, G.; Kulkarni, G.; Kumari, S.G.; Massart, S.; Mezzalama, M.; Alakonya, A.; Muchugi, A.; et al. Phytosanitary Interventions for Safe Global Germplasm Exchange and the Prevention of Transboundary Pest Spread: The Role of CGIAR Germplasm Health Units. Plants 2021, 10, 328. [Google Scholar] [CrossRef] [PubMed]
- Schrader, G.; Unger, J. Plant quarantine as a measure against invasive alien species: The framework of the International Plant Protection Convention and the plant health regulations in the European Union. Biol. Invasions 2003, 5, 357–364. [Google Scholar] [CrossRef]
- Ntui, V.O.; Tripathi, J.N.; Tripathi, L. Current Plant Biology Robust CRISPR / Cas9 mediated genome editing tool for banana and plantain (Musa spp.). Curr. Plant Biol. 2020, 21, 100128. [Google Scholar] [CrossRef]
- Blomme, G.; Ocimati, W.; Felde, A.Z.; Amwonya, D. Banana pest risk assessment along banana trade axes running from low to high altitude sites, in the Eastern DR Congo and in Burundi. Afr. J. Agric. Res. 2020, 16, 1253–1269. [Google Scholar] [CrossRef]
- Tchatchambe, N.B.J.; Ibanda, N.; Adheka, G.; Onautshu, O.; Swennen, R.; Dhed, D. Production of banana bunchy top virus (BBTV) -free plantain plants by in vitro culture. Search IITA Bibliogr. 2020, 15, 361–366. [Google Scholar] [CrossRef]
- Kumar, P.L.; Selvarajan, R.; Iskra-Caruana, M.L.; Chabannes, M.; Hanna, R. Biology, Etiology, and Control of Virus Diseases of Banana and Plantain, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2015; Volume 91. [Google Scholar]
- De Clerck, C.; Crew, K.; Van den Houwe, I.; McMichael, L.; Berhal, C.; Lassois, L.; Haissam Jijakli, M.; Roux, N.; Thomas, J.; Massart, S. Lessons learned from the virus indexing of Musa germplasm: Insights from a multiyear collaboration. Ann. Appl. Biol. 2017, 171, 15–27. [Google Scholar] [CrossRef] [Green Version]
- Singh, V.; Koche, V.; Quraishi, A. Viral Elimination Strategies for Musa spp. Res. Rev. J. Microbiol. Virol. 2018, 8, 7–14. [Google Scholar]
- Vinodhini, J.; Rajendran, L.; Abirami, R.; Karthikeyan, G. Co-existence of chlorosis inducing strain of Cucumber mosaic virus with tospoviruses on hot pepper (Capsicum annuum). India. Sci. Rep. 2021, 11, 1–9. [Google Scholar] [CrossRef]
- Teycheney, P.Y.; Laboureau, N.; Iskra-Caruana, M.L.; Candresse, T. High genetic variability and evidence for plant-to-plant transfer of Banana mild mosaic virus. J. Gen. Virol. 2005, 86, 3179–3187. [Google Scholar] [CrossRef] [PubMed]
- Yéo, E.F.; Kouassi, M.K.; Pita, J.S.; Kouassi, N.K.; Koné, D.; N’guetta, S.P.A. Using thermotherapy and meristem tip culture for producing virus-free cassava planting material from six varieties cultivated in Côte d’ivoire. Int. J. Sci. Technol. Res. 2020, 9, 1607–1612. [Google Scholar]
- Umber, M.; Filloux, D.; Suzia, G.; Gomez, R.; Marais, A.; Gallet, S.; Gamiette, F.; Pavis, C.; Teycheney, P. Molecular Viral Diagnosis and Sanitation of Yam Genetic Resources: Implications for Safe Yam Germplasm Exchange. Viruses 2020, 12, 1101. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.; Barsukova, E.; Fisenko, P.; Chekushkina, T.; Chibizova, A.; Volkov, D.; Klykov, A. Applying methods of replication and recovery of potato microplants (Solanum tuberosum L.) in seed production. E3S Web Conf. 2020, 203, 02003. [Google Scholar] [CrossRef]
- Wondimu, T.; Feyissa, T.; Bedada, G. Meristem culture of selected sweet potato (Ipomoea batatas L. Lam.) cultivars to produce virus-free planting material. J. Hortic. Sci. Biotechnol. 2012, 87, 255–260. [Google Scholar] [CrossRef]
- Massart, S.; Antonio, O.; Jijakli, H.; Candresse, T. Diagnostic des viroses et séquençage haut débit: Vers un changement de paradigme? Virologie 2014, 18, 29. [Google Scholar] [CrossRef]
- Olmos, A.; Boonham, N.; Candresse, T.; Gentit, P.; Giovani, B.; Kutnjak, D.; Liefting, L.; Maree, H.J.; Minafra, A.; Moreira, A.; et al. High-throughput sequencing technologies for plant pest diagnosis: Challenges and opportunities. EPPO Bull. 2018, 48, 219–224. [Google Scholar] [CrossRef] [Green Version]
- Lassois, L.; Lepoivre, P.; Swennen, R.; Van Den Houwe, I.; Panis, B. Thermotherapy, chemotherapy, and meristem culture in Banana. Methods Mol. Biol. 2013, 11013, 419–433. [Google Scholar]
- Ruas, M.; Guignon, V.; Sempere, G.; Sardos, J.; Hueber, Y.; Duvergey, H.; Andrieu, A.; Chase, R.; Jenny, C.; Hazekamp, T.; et al. MGIS: Managing banana (Musa spp.) genetic resources information and high-throughput genotyping data. Database 2017, 2017, bax046. [Google Scholar] [CrossRef] [Green Version]
- Hanafi, M.; Tahzima, R.; Kaab, S.B.; Tamisier, L.; Roux, N.; Massart, S. Identification of Divergent Isolates of Banana Mild Mosaic Virus and Development of a New Diagnostic Primer to Improve Detection. Pathogens 2020, 9, 1045. [Google Scholar] [CrossRef] [PubMed]
- Soltani, N.; Stevens, K.A.; Klaassen, V.; Hwang, M.S.; Golino, D.A.; Al Rwahnih, M. Quality assessment and validation of high-throughput sequencing for grapevine virus diagnostics. Viruses 2021, 13, 1130. [Google Scholar] [CrossRef]
- Thomas, J.E. Technical Guidelines for the Safe Movement of Musa Germplasm; Bioversity International: Rome, Italy, 2015; ISBN 9789292550349. [Google Scholar]
- Ii, P.; Positive, T.; Single, S.; Rna, S. Betaflexiviridae. Virus Taxon. 2012, 920–941. [Google Scholar] [CrossRef]
- Helliot, B.; Panis, B.; Poumay, Y.; Swennen, R.; Lepoivre, P.; Frison, E. Cryopreservation for the elimination of cucumber mosaic and banana streak viruses from banana (Musa spp.). Plant Cell Rep. 2002, 20, 1117–1122. [Google Scholar] [CrossRef]
- Spiegel, S. Uneven distribution of tobacco streak virus in strawberry plantlets grown in vitro. Acta Hort. 1995, 385, 122–125. [Google Scholar] [CrossRef]
- Plotnikov, K.; Ryabinina, V.; Khodakova, A.; Blazhko, N. Viral Load Distribution of Cucumber Green Mottle Mosaic Virus in Leaves. Adv. Soc. Sci. Educ. Humanit. Res. 2020, 393, 210–212. [Google Scholar] [CrossRef] [Green Version]
- Jones, S.; Baizan-Edge, A.; MacFarlane, S.; Torrance, L. Viral diagnostics in plants using next generation sequencing: Computational analysis in practice. Front. Plant Sci. 2017, 8, 1770. [Google Scholar] [CrossRef]
- Oh, Y.; Kim, H.; Kim, S.G. Virus-induced plant genome editing. Curr. Opin. Plant Biol. 2021, 60, 101992. [Google Scholar] [CrossRef]
- Mathew, L.; Tiffin, H.; Erridge, Z.; McLachlan, A.; Hunter, D.; Pathirana, R. Efficiency of eradication of Raspberry bushy dwarf virus from infected raspberry (Rubus idaeus) by in vitro chemotherapy, thermotherapy and cryotherapy and their combinations. Plant Cell. Tissue Organ Cult. 2020, 144, 133–141. [Google Scholar] [CrossRef]
- Azad, A.K.; Khatun, Z.; Eaton, T.E.; Hossen, I. Generation of Virus Free Potato Plantlets through Meristem Culture and Their Field Evaluation. Am. J. Plant Sci. 2020, 11, 1827–1846. [Google Scholar] [CrossRef]
- Arif, M.; Ibrahim, M.; Ahmad, A.; Hassan, S. Elimination of citrus tristeza closterovirus from citrus bud-wood through thermotherapy. Pak. J. Bot. 2005, 37, 423–430. [Google Scholar]
- Al Rwahnih, M.; Daubert, S.; Golino, D.; Islas, C.; Rowhani, A. Comparison of next-generation sequencing versus biological indexing for the optimal detection of viral pathogens in grapevine. Phytopathology 2015, 105, 758–763. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubio, L.; Galipienso, L.; Ferriol, I. Detection of Plant Viruses and Disease Management: Relevance of Genetic Diversity and Evolution. Front. Plant Sci. 2020, 11, 1092. [Google Scholar] [CrossRef] [PubMed]

| Accession Code | Number of Tested Plants | Plants Tested Positive | Plants Tested Negative | Diagnostic Sensitivity |
|---|---|---|---|---|
| ITC0099 | 10 | 7 | 3 | 70% |
| ITC0240 | 7 | 3 | 4 | ≈43% |
| ITC0519 | 5 | 2 | 3 | 40% |
| ITC1171 | 5 | 1 | 4 | 20% |
| ITC1434 | 10 | 4 | 6 | 40% |
| ITC1460 | 10 | 2 | 8 | 20% |
| ITC1541 | 7 | 1 | 6 | ≈14% |
| ITC1564 | 7 | 2 | 5 | ≈29% |
| ITC1664 | 10 | 8 | 2 | 80% |
| ITC1681 | 6 | 5 | 1 | ≈83% |
| ITC1691 | 5 | 0 | 5 | 0% |
| ITC1758 | 10 | 10 | 0 | 100% |
| ITC1767 | 5 | 0 | 5 | 0% |
| ITC1768 | 5 | 0 | 5 | 0% |
| ITC1776 | 11 | 5 | 6 | ≈45% |
| ITC1792 | 9 | 2 | 7 | ≈22% |
| ITC1808 | 5 | 0 | 5 | 0% |
| ITC1832 | 5 | 0 | 5 | 0% |
| ITC1833 | 5 | 0 | 5 | 0% |
| Total | 137 | 52 | 85 | 38% |
| Accession Code | Tested Plant | Corm Results | Leaf Results (in Greenhouse) |
|---|---|---|---|
| ITC0099 | 1 | − | − |
| ITC0240 | 1 | − | − |
| ITC0321 | 1 | − | − |
| ITC0519 | 1 | − | − |
| 2 | − | − | |
| ITC1380 | 1 | + | − |
| 2 | + | − | |
| 3 | + | − | |
| ITC1434 | 1 | + | − |
| 2 | − | − | |
| ITC1460 | 1 | + | − |
| 2 | − | − | |
| ITC1564 | 1 | − | − |
| 2 | + | − | |
| 3 | + | − | |
| ITC1664 | 1 | + | + |
| 2 | + | − | |
| 3 | + | + | |
| ITC1681 | 1 | + | + |
| 2 | − | + | |
| 3 | − | + | |
| ITC1734 | 1 | − | − |
| 2 | − | − | |
| ITC1748 | 1 | − | − |
| 2 | − | − | |
| 3 | − | + | |
| ITC1752 | 1 | − | + |
| ITC1758 | 1 | − | + |
| ITC1767 | 1 | − | − |
| ITC1768 | 1 | + | − |
| 2 | − | − | |
| 3 | − | − | |
| ITC1776 | 1 | − | + |
| ITC1792 | 1 | + | − |
| 2 | + | − | |
| 3 | + | − | |
| ITC1808 | 1 | + | + |
| 2 | − | − | |
| ITC1831 | 1 | − | + |
| 2 | − | − | |
| ITC1857 | 1 | − | − |
| Accession Code | Nb. of Tested Plants | Status | The Base of the Plant | Leaves of the Same Plant | ||||
|---|---|---|---|---|---|---|---|---|
| Plants Tested + | Plants Tested − | DSE | Plants Tested + | Plants Tested − | DSE | |||
| ITC0476 | 2 | Infected | 2 | 0 | 100% | 2 | 0 | 100% |
| ITC0528 | 4 | Infected * | 0 | 4 | 0% | 0 | 4 | 0% |
| ITC1129 | 3 | Infected | 3 | 0 | 100% | 3 | 0 | 100% |
| ITC1677 | 3 | Infected | 1 | 2 | 33% | 2 | 1 | 67% |
| ITC1700 | 4 | Infected | 4 | 0 | 100% | 4 | 0 | 100% |
| ITC1705 | 3 | Infected | 2 | 1 | 67% | 0 | 3 | 0% |
| ITC1706 | 4 | Infected | 3 | 1 | 75% | 4 | 0 | 100% |
| ITC0245 | 4 | Healthy | 0 | 4 | - | 0 | 4 | - |
| ITC0654 | 4 | Healthy | 0 | 4 | - | 0 | 4 | - |
| ITC1120 | 4 | Healthy | 0 | 4 | - | 0 | 4 | - |
| ITC1586 | 4 | Healthy | 0 | 4 | - | 0 | 4 | - |
| Accession | Tissue | Individual | RT−PCR Result | Number of Reads Mapped to BanMMV Sequences (with 20% of Mismatches) | 1st Alien Control (BYDV) | 2nd Alien Control (PepMV) | Total Number of Reads |
|---|---|---|---|---|---|---|---|
| ITC0476 | Base | Mix | + | 5135 | 0 | 2 | 9,007,880 |
| Leaves | Mix | + | 1822 | 0 | 3 | 6,499,298 | |
| ITC0528 | Base | Pl(1) Pl(2) Pl(3) Mix | − − − − | 49 67 46 311 | 0 1 0 0 | 2 0 0 0 | 9,211,966 10,346,472 8,606,200 10,357,840 |
| Leaves | Pl(1) Pl(2) Pl(3) Mix | − − − − | 162 193 171 715 | 0 0 0 0 | 2 1 1 3 | 9,419,300 8,544,882 7,473,540 9,139,050 | |
| ITC1129 | Base | Mix | + | 667 | 0 | 2 | 9,791,204 |
| Leaves | Mix | + | 683 | 0 | 3 | 8,717,902 | |
| ITC1677 | Base | Mix | + | 282 | 0 | 2 | 9,671,458 |
| Leaves | Mix | + | 109 | 1 | 18 | 8,755,638 | |
| ITC1700 | Base | Mix | + | 2260 | 0 | 1 | 9,777,910 |
| Leaves | Mix | + | 3499 | 0 | 0 | 8,245,604 | |
| ITC1705 | Base | Mix | + | 477 | 0 | 0 | 8,759,412 |
| Leaves | Mix | + | 384 | 1 | 4 | 8,711,232 | |
| ITC1706 | Base | Mix | + | 1620 | 0 | 5 | 9,822,322 |
| Leaves | Mix | + | 625 | 0 | 15 | 6,335,434 | |
| ITC1586 * | Base | Mix | − | 0 | 0 | 0 | 8,414,752 |
| ITC0654 * | Leaves | Mix | − | 0 | 0 | 0 | 8,443,436 |
| BYDV−infected alien | Leaves | Pl(1) Pl(2) Pl(3) | − − − | 0 0 0 | 49,563 41,868 25,845 | 2 60 2 | 10,623,038 10,151,628 9,080,280 |
| PepMV−infected alien | Leaves | Pl(1) Pl(2) Pl(3) | − − − | 0 0 0 | 2 0 0 | 53,331 54,681 73,675 | 10,563,714 7,808,992 10,736,536 |
| Name | Accession | Description | Interval | E-value |
|---|---|---|---|---|
| Vmethyltransf | pfam01660 | Viral methyltransferase | 181–1125 | 2.18 × 10−65 |
| RdRP_2 super family | cl03049 | RNA dependent RNA polymerase | 4096–5229 | 1.86 × 10 −37 |
| Viral_helicase1 super family | cl26263 | Viral (Superfamily 1) RNA helicase | 2911–3630 | 3.54 × 10 −09 |
| Peptidase_C23 super family | cl05111 | Carlavirus endopeptidase | 2398–2655 | 1.28 × 10 −03 |
| Viral_helicase1 | pfam01443 | Viral (Superfamily 1) RNA helicase | 5372–5956 | 7.06 × 10 −36 |
| Plant_vir_prot | pfam01307 | Plant viral movement protein | 5945–6238 | 2.25 × 10 −23 |
| Flexi_CP super family | cl02836 | Viral coat protein | 6690–7094 | 6.96 × 10 −41 |
| 7kD_coat | pfam02495 | 7kD viral coat protein | 6216–6380 | 1.26 × 10 −04 |
| Interval | Maximum Protein Identity (Protein-Protein BLAST) | Organism | Accession | |
|---|---|---|---|---|
| ORF1 | 52 -> 5268 | 64.6% | RNA-dependant RNA polymerase (Banana mild mosaic virus) | QVD99720.1 |
| ORF2 | 5261 -> 5935 | 46% | Triple gene block protein 2 (Banana mild mosaic virus) | QVD99726.1 |
| ORF3 | 5936 -> 6274 | 58% | Triple gene block protein 3 (Banana mild mosaic virus) | QVD99727.1 |
| ORF4 | 6192 -> 6398 | 64% | Triple gene block protein 4 (Banana mild mosaic virus) | QVD99723.1 |
| ORF5 | 6471 -> 7238 | 60.8% | Coat protein (Banana mild mosaic virus) | ACN91624.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanafi, M.; Rong, W.; Tamisier, L.; Berhal, C.; Roux, N.; Massart, S. Detection of Banana Mild Mosaic Virus in Musa In Vitro Plants: High-Throughput Sequencing Presents Higher Diagnostic Sensitivity Than (IC)-RT-PCR and Identifies a New Betaflexiviridae Species. Plants 2022, 11, 226. https://doi.org/10.3390/plants11020226
Hanafi M, Rong W, Tamisier L, Berhal C, Roux N, Massart S. Detection of Banana Mild Mosaic Virus in Musa In Vitro Plants: High-Throughput Sequencing Presents Higher Diagnostic Sensitivity Than (IC)-RT-PCR and Identifies a New Betaflexiviridae Species. Plants. 2022; 11(2):226. https://doi.org/10.3390/plants11020226
Chicago/Turabian StyleHanafi, Marwa, Wei Rong, Lucie Tamisier, Chadi Berhal, Nicolas Roux, and Sebastien Massart. 2022. "Detection of Banana Mild Mosaic Virus in Musa In Vitro Plants: High-Throughput Sequencing Presents Higher Diagnostic Sensitivity Than (IC)-RT-PCR and Identifies a New Betaflexiviridae Species" Plants 11, no. 2: 226. https://doi.org/10.3390/plants11020226
APA StyleHanafi, M., Rong, W., Tamisier, L., Berhal, C., Roux, N., & Massart, S. (2022). Detection of Banana Mild Mosaic Virus in Musa In Vitro Plants: High-Throughput Sequencing Presents Higher Diagnostic Sensitivity Than (IC)-RT-PCR and Identifies a New Betaflexiviridae Species. Plants, 11(2), 226. https://doi.org/10.3390/plants11020226







