Variation of Macro- and Microelements, and Trace Metals in Spring Wheat Genetic Resources in Siberia
Abstract
:1. Introduction
2. Results
2.1. Variation for Agronomic Traits
2.2. Adjustment of Grain Element Concentration
2.3. Variation for Element Concentrations
2.4. Elements’ Suitability for Genetic Enhancement
2.5. Genetic Resources Characterization for Grain Ionome
2.6. Superior Germplasm
3. Discussion
4. Materials and Methods
4.1. Panel Composition and Evaluation
4.2. Grain Ionomics Analysis
4.3. Data Processing and Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Morgounov, A.; Sonder, K.; Abugalieva, A.; Bhadauria, V.; Cuthbert, R.D.; Shamanin, V.; Zelenskiy, Y.; DePauw, R.M. Effect of climate change on spring wheat yields in North America and Eurasia in 1981–2015 and implications for breeding. PLoS ONE 2018, 13, 1–16. [Google Scholar] [CrossRef]
- Salt, D.E.; Baxter, I.; Lahner, B. Ionomics and the study of the plant ionome. Annu. Rev. Plant Biol. 2008, 59, 709–733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- White, P.J.; Broadley, M.R. Biofortification of crops with seven mineral elements often lacking in human diets—Iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef]
- Bhatt, R.; Hossain, A.; Sharma, P. Zinc biofortification as an innovative technology to alleviate the zinc deficiency in human health: A review. Open Agric. 2020, 5, 176–187. [Google Scholar] [CrossRef]
- Velu, G.; Singh, R.P.; Joshi, A.K. A Decade of Progress on Genetic Enhancement of Grain Zinc and Iron in CIMMYT Wheat Germplasm. In Wheat and Barley Grain Biofortification; Om Gupta, P., Pandey, V., Narwal, S., Sharma, P., Ram, S., Singh, G.P., Eds.; Woodhead Publishing: Sawston, UK, 2020; pp. 129–138. [Google Scholar]
- Sazawal, S.; Dhingra, U.; Dhingra, P.; Dutta, A.; Deb, S.; Kumar, J.; Devi, P.; Prakash, A. Efficacy of high zinc biofortified wheat in improvement of micronutrient status, and prevention of morbidity among preschool children and women—A double masked, randomized, controlled trial. Nutr. J. 2018, 17, 8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bityutskii, N.; Yakkonen, K.; Loskutov, I. Content of iron, zinc and manganese in grains of Triticum aestivum, Secale cereale, Hordeum vulgare and Avena sativa cultivars registered in Russia. Genet. Resour. Crop Evol. 2017, 64, 1955–1961. [Google Scholar] [CrossRef]
- Morgounov, A.; Savin, T.; Flis, P.; Babkenov, A.; Chudinov, V.; Kazak, A.; Koksel, H.; Likhenko, I.; Sharma, R.; Shelaeva, T.; et al. Effects of environments and cultivars on grain ionome of spring wheat grown in Kazakhstan and Russia. Crop Pasture 2021. accepted, early view on-line. [Google Scholar]
- Sharma, R.C.; Duveiller, E. Selection Index for Improving Helminthosporium Leaf Blight Resistance, Maturity, and Kernel Weight in Spring Wheat. Crop Sci. 2003, 43, 2031–2036. [Google Scholar] [CrossRef]
- Guzman, C.; Medina-Larqué, A.S.; Velu, G.; González-Santoyo, H.; Singh, R.P.; Huerta-Espino, J.; Ortiz-Monasterio, I.; Peña, R.J. Use of wheat genetic resources to develop biofortified wheat with enhanced grain zinc and iron concentrations and desirable processing quality. J. Cereal Sci. 2014, 60, 617–622. [Google Scholar] [CrossRef]
- Velu, G.; Crespo, H.L.; Guzman, C.; Huerta, J.; Payne, T.; Singh, R.P. Assessing genetic diversity to breed competitive biofortified wheat with enhanced grain Zn and Fe concentrations. Front Plant Sci. 2019, 9, 1971. [Google Scholar] [CrossRef]
- Trethowan, R.M.; Morgounov, A.; He, Z.; De Pauw, R.; Crossa, J.; Warburton, M.; Baytasov, A.; Zhang, C.; Mergoum, M.; Alvarado, G. The global adaptation of bread wheat at high latitudes. Euphytica 2006, 152, 303–316. [Google Scholar] [CrossRef]
- Shamanin, V.; Salina, E.; Wanyera, R.; Zelenskiy, Y.; Olivera, P.; Morgounov, A. Genetic diversity of spring wheat from Kazakhstan and Russia for resistance to stem rust Ug99. Euphytica 2016, 212, 287–296. [Google Scholar] [CrossRef]
- Morgounov, A.; Pozherukova, V.; Kolmer, J.; Gultyaeva, E.; Abugalieva, A.; Chudinov, V.; Kuzmin, O.; Rasheed, A.; Rsymbetov, A.; Shepelev, S.; et al. Genetic basis of spring wheat resistance to leaf rust (Puccinia triticina) in Kazakhstan and Russia. Euphytica 2020, 216, 1–15. [Google Scholar] [CrossRef]
- Bhatta, M.; Shamanin, V.; Shepelev, S.; Baenziger, P.S.; Pozherukova, V.; Pototskaya, I.; Morgounov, A. Genetic diversity and population structure analysis of synthetic and bread wheat accessions in Western Siberia. J. Appl. Genet. 2019, 60, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Bhatta, M.; Shamanin, V.; Shepelev, S.; Baenziger, P.S.; Pozherukova, V.; Pototskaya, I.; Morgounov, A. Marker-Trait Associations for Enhancing Agronomic Performance, Disease Resistance, and Grain Quality in Synthetic and Bread Wheat Accessions in Western Siberia. G3 2019, 9, 4209–4222. [Google Scholar] [CrossRef] [Green Version]
- Hazard, B.; Trafford, K.; Lovegrove, A.; Griffiths, S.; Uauy, C.; Shewry, P. Strategies to improve wheat for human health. Nat. Food 2020, 1, 475–480. [Google Scholar] [CrossRef]
- El-Ramady, H.; Abdalla, N.; Elbasiouny, H.; Elbehiry, F.; Elsakhawy, T.; Omara, A.E.-D.; Amer, M.; Bayoumi, Y.; Shalaby, T.A.; Eid, Y.; et al. Nano-biofortification of different crops to immune against COVID-19: A review. Ecotoxicol. Environ. Saf. 2021, 222, 112500. [Google Scholar] [CrossRef]
- Praharaj, S.; Skalicky, M.; Maitra, S.; Bhadra, P.; Shankar, T.; Brestic, M.; Hejnak, V.; Vachova, P.; Hossain, A. Zinc Biofortification in Food Crops Could Alleviate the Zinc Malnutrition in Human Health. Molecules 2021, 26, 3509. [Google Scholar] [CrossRef]
- Shi, Y.; Li, J.; Sun, Z. Success to iron biofortification of wheat grain by combining both plant and microbial genetics. Rhizosphere 2020, 15, 100218. [Google Scholar] [CrossRef]
- Galić, L.; Vinković, T.; Ravnjak, B.; Lončarić, Z. Agronomic Biofortification of Significant Cereal Crops with Selenium—A Review. Agronomy 2021, 11, 1015. [Google Scholar] [CrossRef]
- Finkelstein, J.L.; Haas, J.D.; Mehta, S. Iron-biofortified staple food crops for improving iron status: A review of the current evidence. Curr. Opin. Biotechnol. 2017, 44, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Loskutov, I.G.; Khlestkina, E.K. Wheat, Barley, and Oat Breeding for Health Benefit Components in Grain. Plants 2021, 10, 86. [Google Scholar] [CrossRef]
- Shelenga, T.; Kerv, Y.; Perchuk, I.; Solovyeva, A.E.; Khlestkina, E.; Loskutov, I.G.; Konarev, A. The Potential of Small Grains Crops in Enhancing Biofortification Breeding Strategies for Human Health Benefit. Agronomy 2021, 11, 1420. [Google Scholar] [CrossRef]
- Abugalieva, A.I.; Flis, P.; Shamanin, V.P.; Savin, T.; Morgounov, A. Ionomic analysis of spring wheat grain produced in Kazakhstan and Russia. Comm. Soil. Sci. Plant Anal. 2021, 52, 704–7011. [Google Scholar] [CrossRef]
- Fatiukha, A.; Klymiuk, V.; Peleg, Z.; Saranga, Y.; Cakmak, I.; Krugman, T.; Korol, A.B.; Fahima, T. Variation in phosphorus and sulfur content shapes the genetic architecture and phenotypic associations within the wheat grain ionome. Plant J. 2019, 101, 555–572. [Google Scholar] [CrossRef]
- Aberkane, H.; Payne, T.; Kishi, M.; Smale, M.; Amri, A.; Jamora, N. Transferring diversity of goat grass to farmers’ fields through the development of synthetic hexaploid wheat. Food Secur. 2020, 12, 1017–1033. [Google Scholar] [CrossRef]
- Gupta, P.K.; Balyan, H.S.; Sharma, S.; Kumar, R. Biofortification and bioavailability of Zn, Fe and Se in wheat: Present status and future prospects. Theor. Appl. Genet. 2020, 134, 1–35. [Google Scholar] [CrossRef]
- Shamanin, V.; Shepelev, S.; Pozherukova, V.; Gultyaeva, E.; Kolomiets, T.; Pakholkova, E.; Morgounov, A. Primary hexaploid synthetics: Novel sources of wheat disease resistance. Crop Prot. 2019, 121, 7–10. [Google Scholar] [CrossRef]
- Simmonds, N.W. Principles of Crop Improvement, 2nd ed.; Longman Group: New York, NY, USA, 1981. [Google Scholar]
- Crossa, J.; Fritsche-Neto, R.; Montesinos-Lopez, O.A.; Costa-Neto, G.; Dreisigacker, S.; Montesinos-Lopez, A.; Bentley, A.R. The Modern Plant Breeding Triangle: Optimizing the Use of Genomics, Phenomics, and Enviromics Data. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef]
- Bhatta, M.; Baenziger, P.S.; Waters, B.M.; Poudel, R.; Belamkar, V.; Poland, J.; Morgounov, A. Genome-Wide Association Study Reveals Novel Genomic Regions Associated with 10 Grain Minerals in Synthetic Hexaploid Wheat. Int. J. Mol. Sci. 2018, 19, 3237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rai, P.K.; Lee, S.S.; Zhang, M.; Tsang, Y.F.; Kim, K.-H. Heavy metals in food crops: Health risks, fate, mechanisms, and management. Environ. Int. 2019, 125, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Morgounov, A.; Abugalieva, A.; Akan, K.; Akın, B.; Baenziger, S.; Bhatta, M.; Dababat, A.A.; Demir, L.; Dutbayev, Y.; El Bouhssini, M.; et al. High-yielding winter synthetic hexaploid wheats resistant to multiple diseases and pests. Plant Genet. Resour. 2017, 16, 273–278. [Google Scholar] [CrossRef]
- Matsuoka, Y.; Takumi, S.; Kawahara, T. Natural variation for fertile triploid F1 hybrid formation in allohexaploid wheat speciation. Appl. Genet. 2007, 115, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Pask, A.J.D.; Pietragalla, J.; Mullan, D.M.; Reynolds, M.P. Physiological Breeding II: A Field Guide to Wheat Phenotyping; CIMMYT: Veracruz, Mexico, 2012. [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
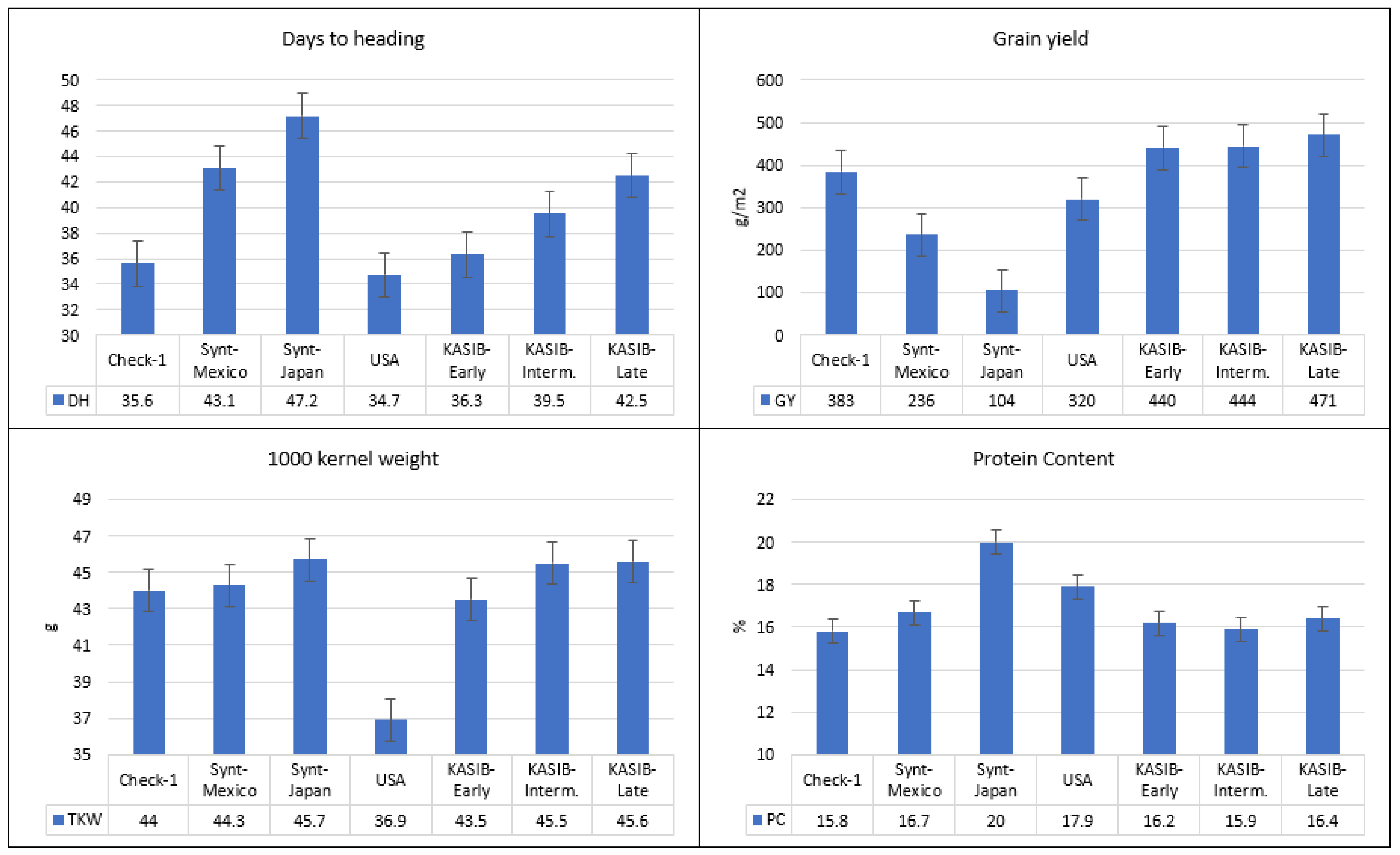
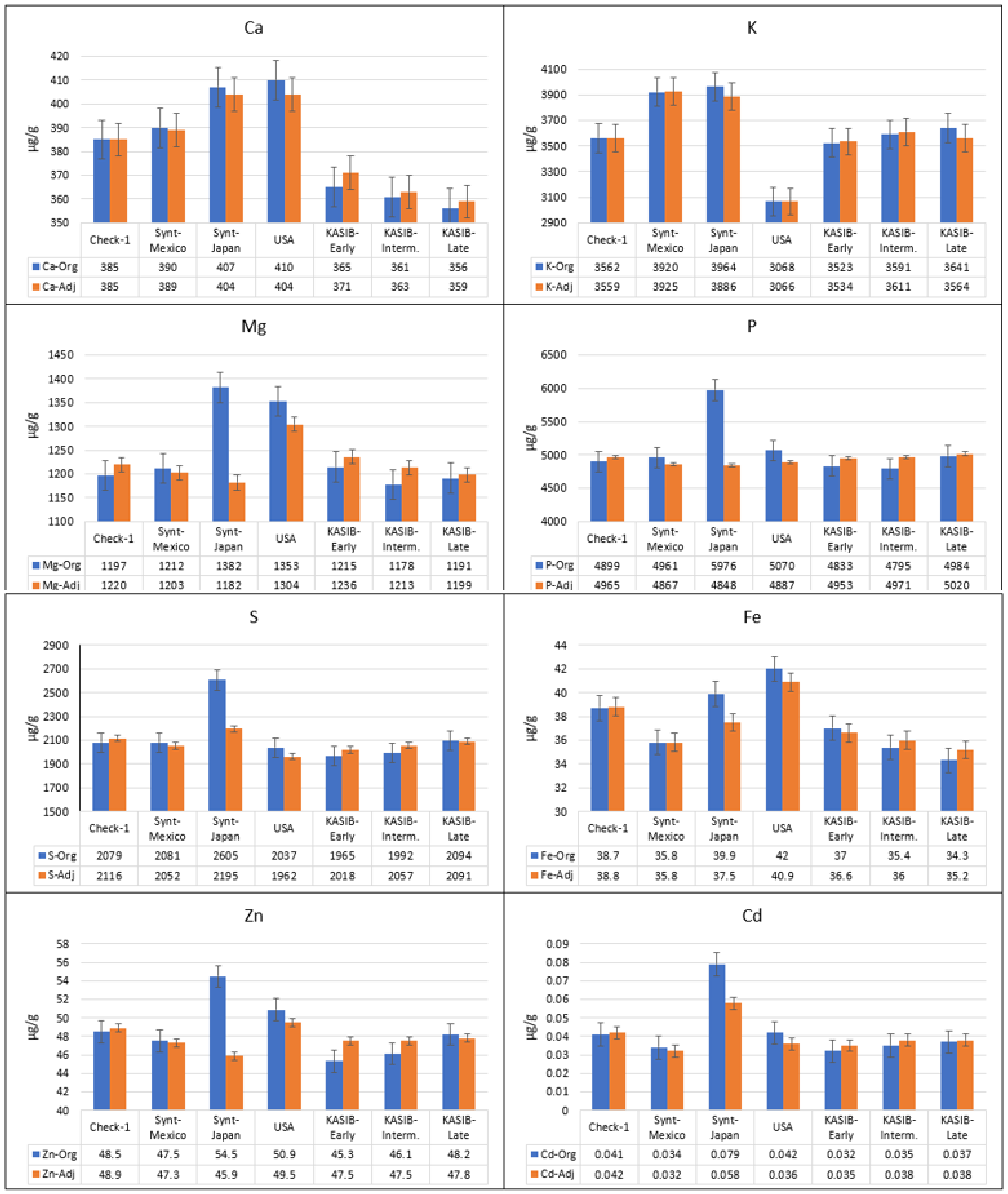
| Element | Year | Yield | PC | TKW | Ca | K | Mg | P | S |
|---|---|---|---|---|---|---|---|---|---|
| Ca | 2017 | −0.11 | 0.06 | −0.14 | - | 0.16 | 0.15 | 0.01 | 0.09 |
| 2018 | −0.14 | 0.06 | −0.15 | - | −0.03 | 0.22 * | 0.03 | 0.11 | |
| K | 2017 | −0.13 | 0.03 | 0.10 | 0.16 | - | −0.07 | −0.07 | 0.30 * |
| 2018 | −0.19 | 0.16 | 0.17 | −0.03 | - | 0.07 | 0.07 | 0.09 | |
| Mg | 2017 | −0.24 * | 0.43 * | −0.07 | 0.15 | −0.07 | - | 0.79 * | 0.60 * |
| 2018 | −0.19 | 0.29 * | −0.06 | 0.22 * | 0.07 | - | 0.76 * | 0.55 * | |
| P | 2017 | −0.14 | 0.32 * | 0.08 | 0.01 | 0.20 * | 0.79 * | - | 0.68 * |
| 2018 | −0.22 * | 0.30 * | 0.05 | 0.03 | 0.48 * | 0.76 * | - | 0.20 * | |
| S | 2017 | −0.23 * | 0.45 * | 0.13 | 0.09 | 0.30 * | 0.60 * | 0.68 * | - |
| 2018 | −0.34 * | 0.47 * | 0.12 | 0.11 | 0.09 | 0.55 * | 0.20 * | - | |
| Cu | 2017 | −0.14 | 0.28 * | −0.02 | −0.03 | 0.10 | 0.62 * | 0.68 * | 0.54 * |
| 2018 | −0.21 * | 0.25 * | 0.01 | 0.08 | 0.21 * | 0.60 * | 0.60 * | 0.47 * | |
| Fe | 2017 | −0.10 | 0.23 * | 0.29 * | −0.01 | −0.14 | 0.23 * | −0.06 | −0.09 |
| 2018 | −0.13 | 0.17 | −0.14 | −0.03 | −0.12 | 0.05 | −0.04 | −0.10 | |
| Mn | 2017 | −0.13 | 0.35 * | −0.02 | 0.11 | −0.10 | 0.68 * | 0.57 * | 0.49 * |
| 2018 | −0.09 | 0.05 | 0.09 | 0.22 * | −0.10 | 0.63 * | 0.46 * | 0.38 * | |
| Zn | 2017 | −0.03 | 0.20 * | 0.04 | −0.08 | 0.02 | 0.53 * | 0.73 * | 0.48 * |
| 2018 | −0.30 * | 0.26 * | 0.02 | 0.09 | 0.27 * | 0.49 * | 0.63 * | 0.50 * | |
| Cd | 2017 | −0.20 * | 0.29 * | 0.01 | 0.00 | −0.02 | 0.46 * | 0.44 * | 0.49 * |
| 2018 | −0.22 * | 0.38 * | 0.10 | 0.09 | 0.05 | 0.37 * | 0.32 * | 0.40 * | |
| Co | 2017 | 0.01 | 0.07 | −0.19 | 0.12 | 0.14 | 0.17 | 0.14 | 0.04 |
| 2018 | 0.01 | 0.05 | 0.04 | 0.19 | 0.12 | 0.11 | 0.15 | 0.15 | |
| Ni | 2017 | −0.08 | 0.15 | −0.01 | −0.01 | 0.09 | 0.25 * | 0.25 * | 0.23 * |
| 2018 | −0.24 * | 0.36 * | 0.01 | −0.03 | 0.14 | 0.23 * | 0.28 * | 0.31 * | |
| Mo | 2017 | −0.07 | 0.20 * | 0.05 | 0.01 | 0.17 | 0.12 | 0.20 * | 0.24 * |
| 2018 | −0.02 | 0.15 | 0.18 | 0.05 | 0.27 * | 0.21 * | 0.33 * | 0.26 * | |
| Rb | 2017 | −0.05 | 0.06 | 0.16 | −0.02 | 0.12 | 0.04 | 0.08 | 0.07 |
| 2018 | −0.18 | 0.12 | 0.01 | 0.07 | 0.18 | −0.01 | 0.00 | 0.16 | |
| Sr | 2017 | −0.34 * | 0.22 * | −0,08 | 0.60 * | 0.11 | 0.17 | −0.00 | 0.16 |
| 2018 | −0.34 * | 0.31 * | −0.22 * | 0.65 * | 0.04 | 0.17 | 0.04 | 0.16 | |
| Number of significant correlations | 11 | 18 | 3 | 4 | 6 | 16 | 14 | 15 | |
| Element-Year | Data | Adjustment Variables 1 | Mean | Min | Max | CV, % | H2 | r (Original–Adjusted) |
|---|---|---|---|---|---|---|---|---|
| Macroelements | ||||||||
| Ca-2017 | Original | - | 362 | 237 | 518 | 15.1 | 0.59 | - |
| Ca-2018 | Original | - | 397 | 266 | 531 | 13.8 | 0.49 | 0.98 |
| Adjusted | Mg | 273 | 523 | 13.6 | 0.50 | |||
| K-2017 | Original | - | 3652 | 2654 | 5453 | 10.2 | 0.67 | 0.99 |
| Adjusted | S | 2715 | 5314 | 10.1 | 0.69 | |||
| K-2018 | Original | - | 3642 | 2298 | 4992 | 15.7 | 0.79 | - |
| Mg-2017 | Original | - | 1208 | 922 | 1532 | 9.1 | 0.60 | 0.56 |
| Adjusted | YLD, PC, P, S | 997 | 1400 | 5.4 | 0.62 | |||
| Mg-2018 | Original | - | 1225 | 926 | 1524 | 8.5 | 0.58 | 0.66 |
| Adjusted | PC, Ca, P, S | 1007 | 1461 | 5.6 | 0.69 | |||
| P-2017 | Original | - | 5180 | 3906 | 6701 | 10.2 | 0.43 | 0.54 |
| Adjusted | PC, K, Mg, S | 4401 | 5995 | 5.6 | 0.62 | |||
| P-2018 | Original | - | 4699 | 3428 | 6993 | 12.3 | 0.57 | 0.48 |
| Adjusted | YLD, PC, K, Mg, S | 3835 | 5500 | 5.8 | 0.68 | |||
| S-2017 | Original | - | 2059 | 1710 | 2682 | 9.5 | 0.69 | 0.60 |
| Adjusted | YLD, PC, K, Mg, P | 1768 | 2386 | 5.8 | 0.59 | |||
| S-2018 | Original | - | 2050 | 1572 | 2698 | 9.6 | 0.71 | 0.64 |
| Adjusted | YLD, PC, Mg, P | 1762 | 2433 | 6.1 | 0.66 | |||
| Microelements | ||||||||
| Cu-2017 | Original | - | 4.66 | 3.09 | 7.08 | 13.8 | 0.54 | 0.76 |
| Adjusted | PC, Mg, P, S | 3.53 | 6.29 | 10.4 | 0.63 | |||
| Cu-2018 | Original | - | 3.72 | 2.30 | 6.06 | 15.3 | 0.57 | 0.74 |
| Adjusted | YLD, PC, Mg, P, S | 2.67 | 5.22 | 11.3 | 0.55 | |||
| Fe-2017 | Original | - | 37.7 | 21.1 | 53.6 | 14.2 | 0.50 | 0.90 |
| Adjusted | PC, Mg | 21.4 | 51.8 | 12.9 | 0.40 | |||
| Fe-2018 | Original | - | 35.8 | 22.5 | 47.2 | 12.9 | 0.25 | - |
| Mn-2017 | Original | - | 43.1 | 31.9 | 60.0 | 11.2 | 0.58 | 0.72 |
| Adjusted | PC, Mg, P, S | 31.0 | 52.4 | 8.2 | 0.58 | |||
| Mn-2018 | Original | - | 44.8 | 31.9 | 59.1 | 11.7 | 0.63 | 0.81 |
| Adjusted | Mg, P, S | 32.6 | 52.4 | 8,2 | 0.70 | |||
| Zn-2017 | Original | - | 53.3 | 31.1 | 77.7 | 15.6 | 0.16 | 0.74 |
| Adjusted | PC, Mg, P, S | 40.0 | 69.3 | 10.9 | 0.27 | |||
| Zn-2018 | Original | - | 41.8 | 23.9 | 76.3 | 19.1 | 0.42 | 0.67 |
| Adjusted | PC, K, Mg, P, S | 26.4 | 62.0 | 13.1 | 0.39 | |||
| Toxic trace elements | ||||||||
| Cd-2017 | Original | - | 0.044 | 0.019 | 0.117 | 35.8 | 0.66 | 0.75 |
| Adjusted | YLD, PC, Mg, P, S | 0.024 | 0.099 | 27.7 | 0.57 | |||
| Cd-2018 | Original | - | 0.031 | 0.011 | 0.088 | 35.6 | 0.68 | 0.77 |
| Adjusted | YLD, PC, Mg, P, S | 0.013 | 0.071 | 27.7 | 0.55 | |||
| Co-2017 | Original | - | 0.010 | 0.006 | 0.021 | 27.0 | 0.72 | - |
| Co-2018 | Original | - | 0.009 | 0.006 | 0.017 | 17.1 | 0.27 | - |
| Ni-2017 | Original | - | 0.212 | 0.113 | 0.356 | 23.6 | 0.44 | 0.94 |
| Adjusted | Mg, P, S | 0.113 | 0.360 | 22.4 | 0.44 | |||
| Ni-2018 | Original | - | 0.148 | 0.078 | 0.312 | 26.6 | 0.57 | 0.85 |
| Adjusted | YLD, PC, Mg, P, S | 0.075 | 0.224 | 22.6 | 0.47 | |||
| Trace elements | ||||||||
| Mo-2017 | Original | - | 0.347 | 0.209 | 0.579 | 22.4 | 0.71 | 0.94 |
| Adjusted | PC, P, S | 0.208 | 0.554 | 22.6 | 0.69 | |||
| Mo-2018 | Original | - | 0.308 | 0.195 | 0.592 | 24.0 | 0.78 | 0.91 |
| Adjusted | K, Mg, P, S | 0.183 | 0.554 | 21.4 | 0.74 | |||
| Rb-2017 | Original | - | 4.35 | 2.29 | 6.75 | 21.7 | 0.25 | - |
| Rb-2018 | Original | - | 3.51 | 2.20 | 5.34 | 18.9 | 0.25 | - |
| Sr-2017 | Original | - | 2.09 | 0.914 | 3.627 | 30.3 | 0.81 | 0.91 |
| Adjusted | YLD, PC, TKW | 0.909 | 3628 | 27.7 | 0.77 | |||
| Sr-2018 | Original | - | 2.42 | 1.175 | 4.307 | 27.2 | 0.75 | 0.82 |
| Adjusted | YLD, PC, TKW | 1.366 | 4.053 | 22.2 | 0.63 | |||
| Element | Coefficient of Phenotypic Variation for 2017–2018 Means | Number of Significant Correlations with Agronomic Traits and Other Elements | H2 Based on ANOVA of Original Data | Overall Sum of Ranks | ||||
|---|---|---|---|---|---|---|---|---|
| % | Rank | Value | Rank | Value | Rank | Value | Rank | |
| Ca | 14.5 | 9 | 1 | 3.5 | 0.73 | 5 | 17.5 | 4 |
| K | 13.0 | 11 | 1 | 3.5 | 0.61 | 8 | 22.5 | 7 |
| Mg | 8.8 | 15 | 8 | 9.5 | 0.67 | 6 | 30.5 | 12 |
| P | 11.3 | 13 | 9 | 12.5 | 0.60 | 10 | 34.5 | 14 |
| S | 9.6 | 14 | 9 | 12.5 | 0.81 | 3 | 28.5 | 11 |
| Cu | 14.6 | 8 | 10 | 11 | 0.61 | 9 | 28.0 | 10 |
| Fe | 13.6 | 10 | 2 | 5 | 0.58 | 11 | 26.0 | 9 |
| Mn | 11.5 | 12 | 7 | 7 | 0.55 | 12 | 31.0 | 13 |
| Zn | 17.4 | 7 | 10 | 14 | 0.27 | 15 | 36.0 | 15 |
| Cd | 35.7 | 1 | 10 | 14 | 0.76 | 4 | 19.0 | 5 |
| Co | 22.1 | 5 | 0 | 1.5 | 0.65 | 7 | 13.5 | 3 |
| Ni | 25.1 | 3 | 8 | 9.5 | 0.49 | 13 | 25.5 | 8 |
| Mo | 23.2 | 4 | 7 | 7 | 0.84 | 2 | 13.0 | 2 |
| Rb | 20.3 | 6 | 0 | 1.5 | 0.48 | 14 | 21.5 | 6 |
| Sr | 28.8 | 2 | 7 | 7 | 0.87 | 1 | 10.0 | 1 |
| Entry | Genotype | Group * | Highest/Lowest Elements | Grain Yield | Protein Content | ||
|---|---|---|---|---|---|---|---|
| g/m2 | Rank | % | Rank | ||||
| - | Pamyati Azieva (Check-1) | - | 399 | - | 16.5 | - | |
| - | Serebristaya (Check-2) | - | 471 | - | 14.9 | - | |
| 151 | Lutestsens KS 963 | K-E | Ca, Mg | 572 | 1 | 17.2 | 66 |
| 112 | Lutestsens 15-14 | K-L | Mn, Zn | 557 | 2 | 16.5 | 101 |
| 91 | Lutestsens 7-04-4 | K-I | - | 541 | 3 | 17.5 | 54 |
| 164 | Silach | K-L | Ca, Mg, Rb | 541 | 4 | 16.7 | 95 |
| 94 | Element 22 | K-L | Ca, P, S, Ni | 535 | 5 | 17.0 | 75 |
| 152 | Lutestsens 1296 | K-I | Ca, K, Cd | 521 | 6 | 15.6 | 125 |
| 157 | OmGAU-100 | K-L | Ca, P, Cu | 518 | 7 | 16.4 | 103 |
| 116 | Uralosibirskaya | K-I | - | 515 | 8 | 17.6 | 52 |
| 85 | Lutescens 310-00-1 | K-I | P, Rb | 514 | 9 | 17.5 | 54 |
| 89 | Aestivum 947 | K-I | Ni | 514 | 10 | 15.8 | 119 |
| 156 | Novosibirskaya 41 | K-E | P, Cu, Cd, Ni | 482 | 23 | 19.0 | 16 |
| 143 | Lutestsens 1103 | K-I | Ca, Mg, Mn, Ni, Mo | 469 | 30 | 16.1 | 111 |
| 125 | Stepnaya 253 | K-I | Ca, Fe, Zn | 468 | 31 | 14.1 | 133 |
| 114 | OmGAU-90 | K-I | Ca, Mg, S | 466 | 33 | 15.7 | 121 |
| 132 | Lutestsens 248-01 | K-I | Mn, Zn, Mo, Sr | 394 | 58 | 15.9 | 117 |
| 66 | RBOT | USA | Cu, Fe, Rb | 386 | 63 | 18.4 | 28 |
| 87 | L 485 | K-E | Ca, P, Mn | 378 | 66 | 17.0 | 73 |
| 103 | Lutestsens 15-12 | K-I | Cu, Co, Ni | 372 | 71 | 17.6 | 51 |
| 73 | Freyr | USA | Mg, Zn, Ni, Mo, Rb | 354 | 75 | 19.0 | 15 |
| 61 | Pandur/Ae. Squarrosa(409) | S-M | K, S, Cu, Co | 330 | 79 | 17.4 | 57 |
| 78 | Alpine | USA | Mg, Fe, Zn, Rb | 321 | 81 | 17.9 | 43 |
| 13 | Ukr-Od 1530.94/Ae. Squarrosa(392) | S-M | Fe, Mn, Sr | 313 | 82 | 17.2 | 71 |
| 77 | Brennan | USA | Mg, Fe, Mn | 297 | 87 | 18.7 | 19 |
| 36 | Aisberg/Ae. Squarrosa (369)//Demir | S-M | Cu, Zn, Cd, Co, Mo | 273 | 94 | 16.3 | 107 |
| 3 | Ukr-Od 952.92/Ae. Squarrosa(1031) | S-M | Cd, Co, Rb | 255 | 100 | 17.0 | 76 |
| 12 | Aisberg/Ae. Squarrosa(511) | S-M | Ca, S, Cu, Mo | 245 | 103 | 16.8 | 87 |
| 6 | Ukr-Od 1530.94/Ae. Squarrosa(458) | S-M | P, Cd, Co | 213 | 115 | 17.7 | 47 |
| 57 | Ukr-Od 1530.94/Ae. Squarrosa(392) | S-M | K, P, S, Zn, Cd, Mo | 209 | 116 | 18.2 | 34 |
| 16 | Ukr-Od 1530.94/Ae. Squarrosa(458) | S-M | Ca, Cd, Rb | 206 | 118 | 17.2 | 69 |
| 22 | Langdon/IG 48042 | S-J | Ca, S, Fe | 141 | 125 | 20.7 | 6 |
| 51 | Langdon/IG 131606 | S-J | Ca, Co, Ni | 97 | 130 | 20.6 | 7 |
| LSD 0.05 | 15 | - | 0.3 | - | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shepelev, S.; Morgounov, A.; Flis, P.; Koksel, H.; Li, H.; Savin, T.; Sharma, R.; Wang, J.; Shamanin, V. Variation of Macro- and Microelements, and Trace Metals in Spring Wheat Genetic Resources in Siberia. Plants 2022, 11, 149. https://doi.org/10.3390/plants11020149
Shepelev S, Morgounov A, Flis P, Koksel H, Li H, Savin T, Sharma R, Wang J, Shamanin V. Variation of Macro- and Microelements, and Trace Metals in Spring Wheat Genetic Resources in Siberia. Plants. 2022; 11(2):149. https://doi.org/10.3390/plants11020149
Chicago/Turabian StyleShepelev, Sergey, Alexey Morgounov, Paulina Flis, Hamit Koksel, Huihui Li, Timur Savin, Ram Sharma, Jingxin Wang, and Vladimir Shamanin. 2022. "Variation of Macro- and Microelements, and Trace Metals in Spring Wheat Genetic Resources in Siberia" Plants 11, no. 2: 149. https://doi.org/10.3390/plants11020149
APA StyleShepelev, S., Morgounov, A., Flis, P., Koksel, H., Li, H., Savin, T., Sharma, R., Wang, J., & Shamanin, V. (2022). Variation of Macro- and Microelements, and Trace Metals in Spring Wheat Genetic Resources in Siberia. Plants, 11(2), 149. https://doi.org/10.3390/plants11020149






