Molecular, Histological and Histochemical Responses of Banana Cultivars Challenged with Fusarium oxysporum f. sp. cubense with Different Levels of Virulence
Abstract
:1. Introduction
2. Results
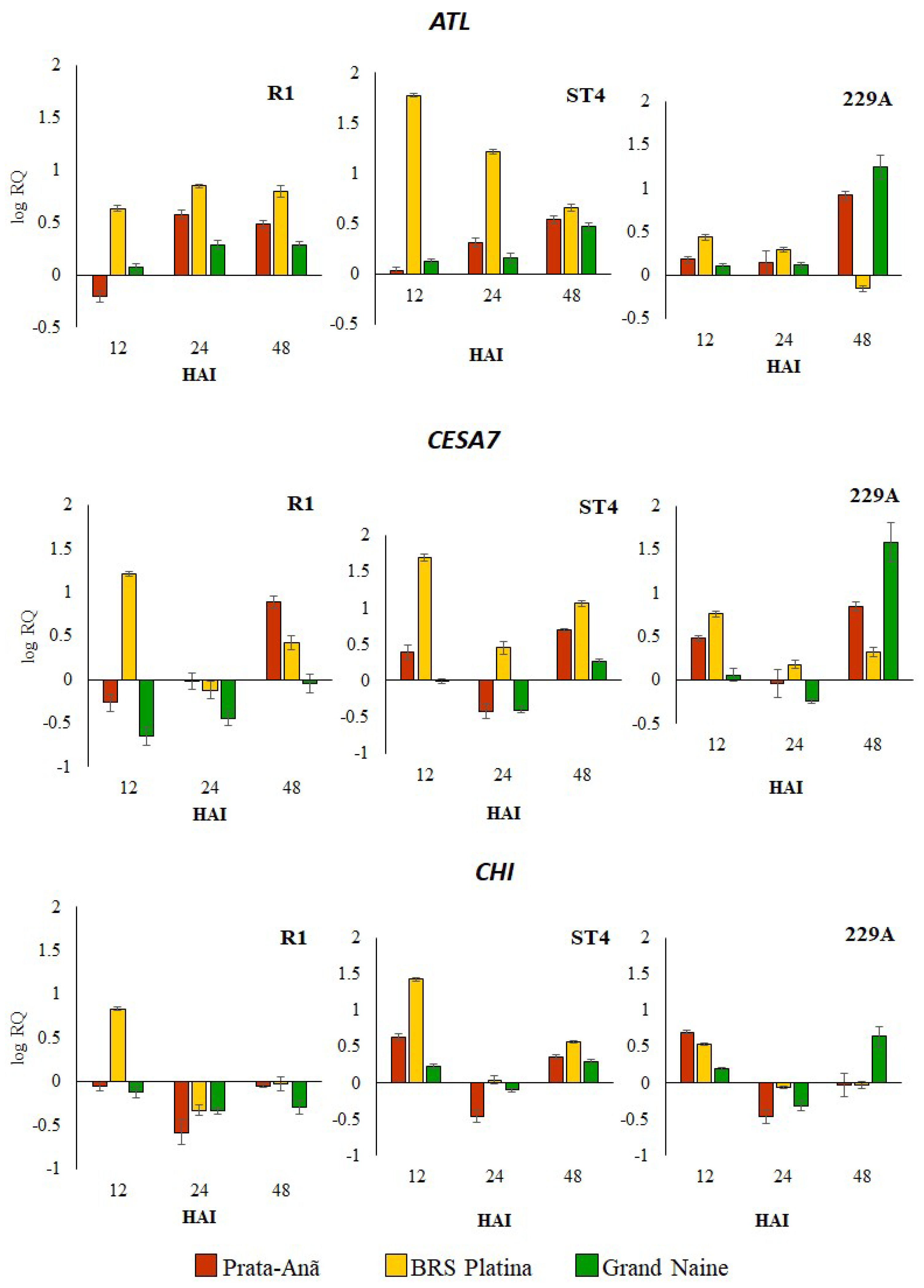
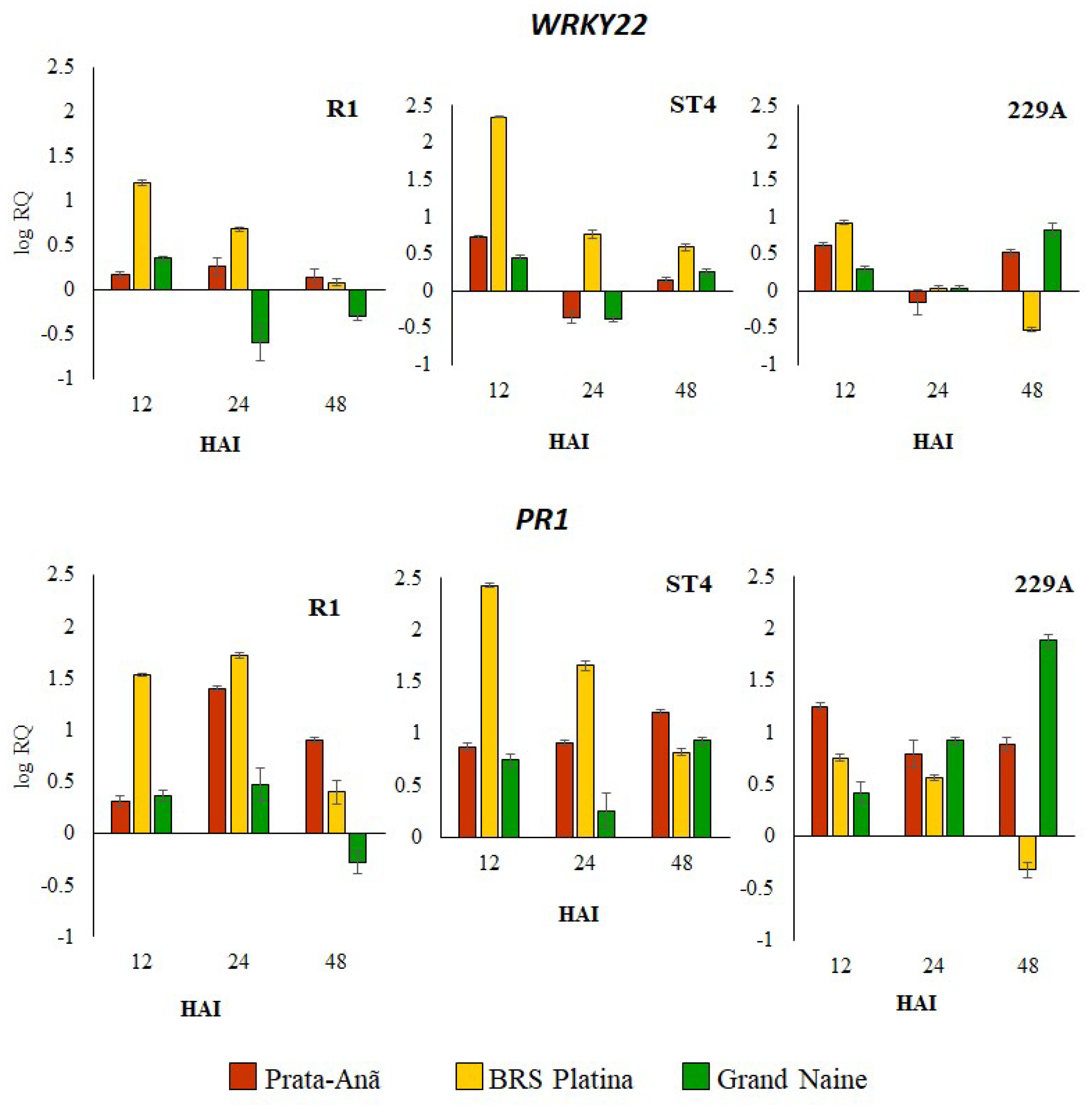
2.1. Expression Profile of Defense Genes in Response to Different Foc Isolates
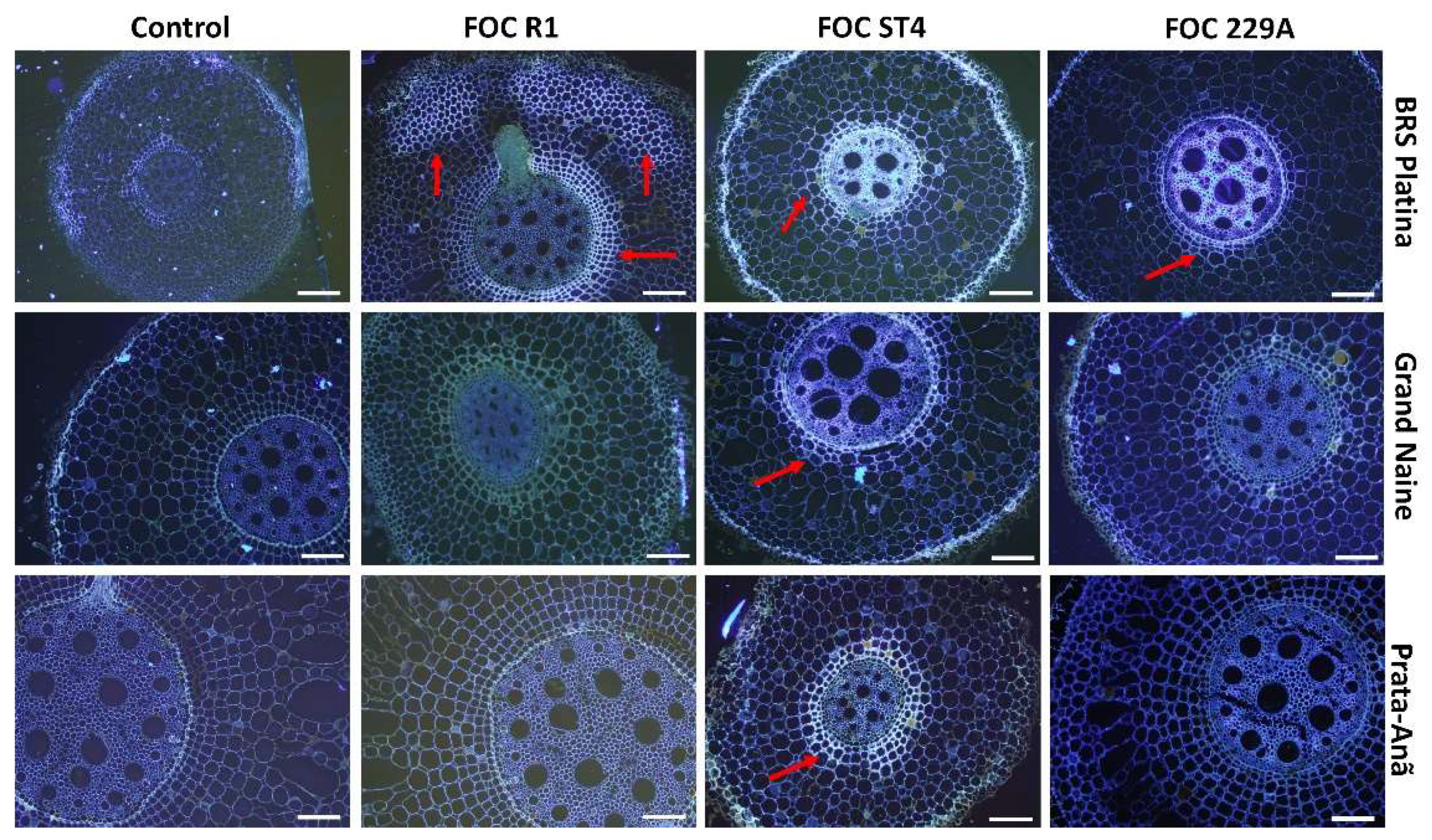
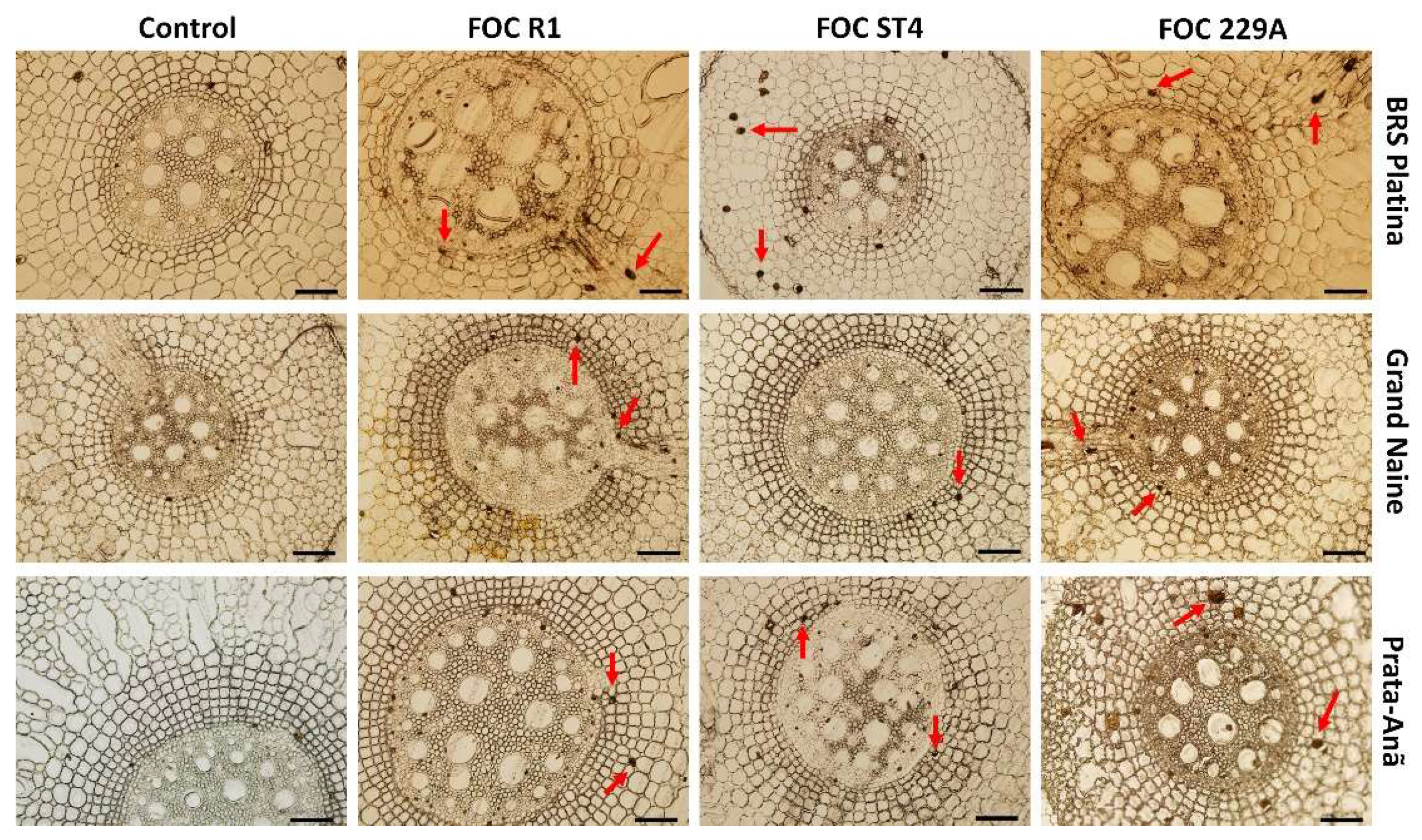
2.2. Histological and Histochemical Responses
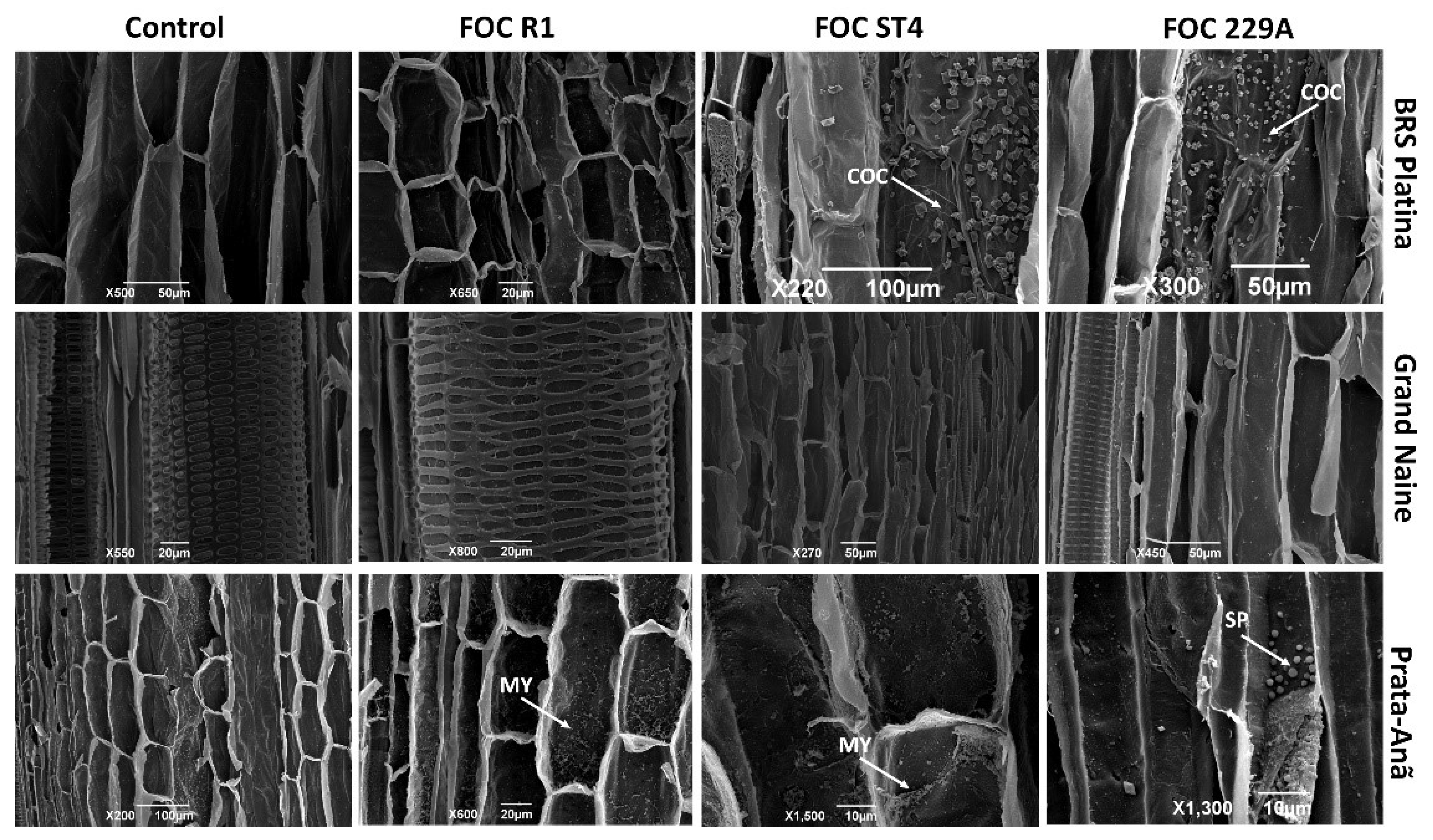
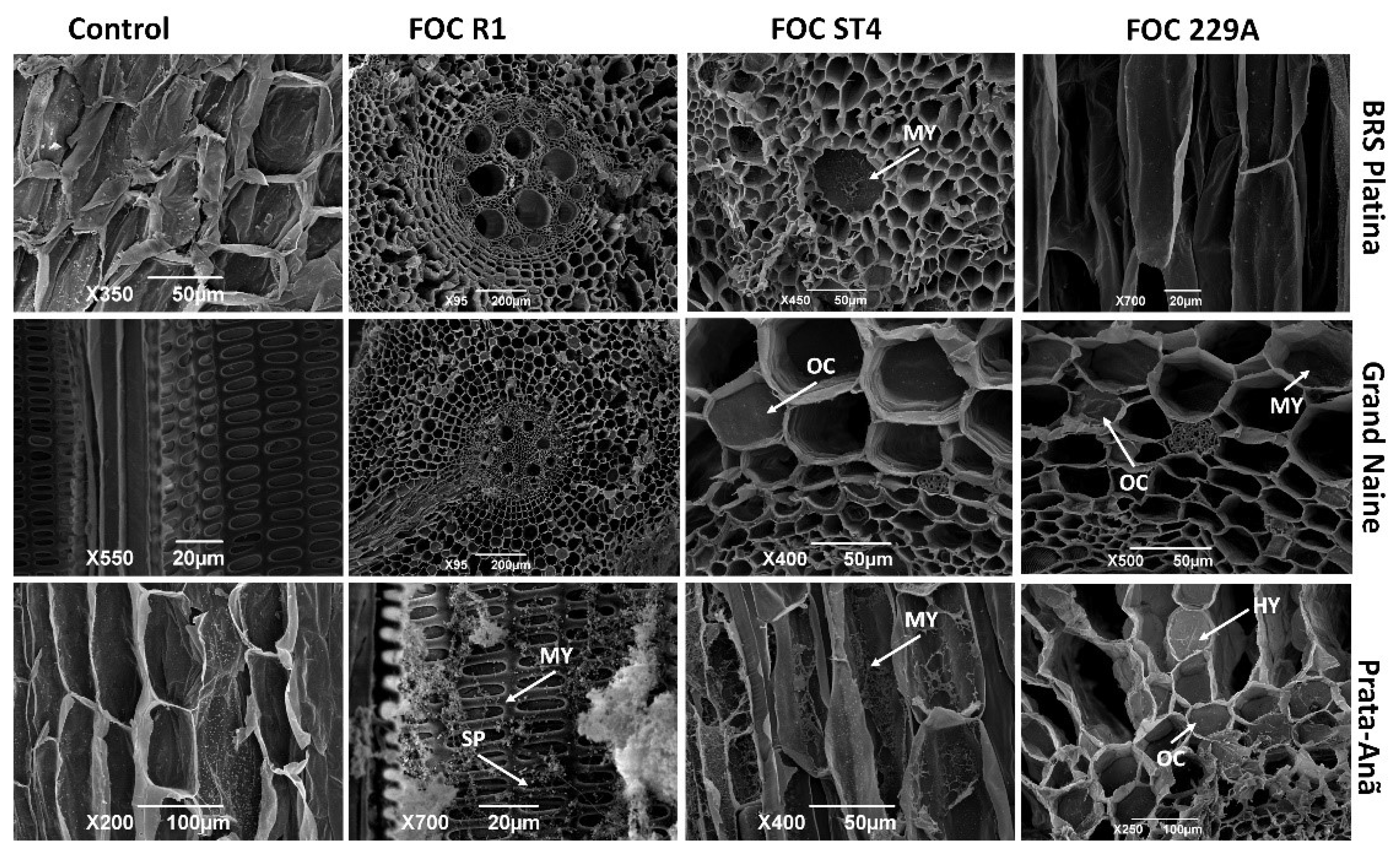
2.3. Analysis of the Interaction by Scanning Electron Microscopy
2.4. Symptoms
3. Discussion
4. Materials and Methods
4.1. Foc Isolates
4.2. Plant Material
4.3. Bioassay
4.4. RNA Extraction and cDNA Synthesis
4.5. Gene Expression Analysis by Quantitative Real-Time PCR
4.6. Histological and Histochemical Analyses
4.7. Analysis by Scanning Electron Microscopy—SEM
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ploetz, R.C.; Pegg, K. Fusarium wilt of banana and Wallace’s line: Was the disease originally restricted to his Indo-Malayan region? Australas. Plant Pathol. 1997, 26, 239–249. [Google Scholar] [CrossRef]
- Fourie, G.; Steenkamp, E.T.; Gordon, T.R.; Viljoen, A. Evolutionary relationships among the Fusarium oxysporum f. sp. cubense vegetative compatibility groups. Appl. Environ. Microbiol. 2009, 75, 4770–4781. [Google Scholar] [CrossRef]
- Maryani, N.; Lombard, L.; Poerba, Y.S.; Subandiyah, S.; Crous, P.W.; Kema, G.H.J. Phylogeny and genetic diversity of the banana Fusarium wilt pathogen Fusarium oxysporum f. sp. cubense in the Indonesian centre of origin. Stud. Mycol. 2019, 92, 155–194. [Google Scholar] [CrossRef] [PubMed]
- Stover, R. Fusarial Wilt (Panama disease) of Bananas and Other Musa Species; Kew: Commonwealth Mycological Institute: Kew, UK, 1962. [Google Scholar]
- Bancroft, J. Report of the board appointed to enquire into the cause of disease affecting livestock and plants. Votes Proc. 1876, 3, 1011–1038. [Google Scholar]
- Li, C.; Shao, J.; Wang, Y.; Li, W.; Guo, D.; Yan, B.; Xia, Y.; Peng, M. Analysis of banana transcriptome and global gene expression profiles in banana roots in response to infection by race 1 and tropical race 4 of Fusarium oxysporum f. sp. cubense. BMC Genom. 2013, 14, 851. [Google Scholar] [CrossRef] [PubMed]
- Ploetz, R.C. Fusarium wilt of banana is caused by several pathogens referred to as Fusarium oxysporum f. sp. cubense. Phytopathology 2006, 96, 653–656. [Google Scholar] [CrossRef]
- Mostert, D.; Molina, A.B.; Daniells, J.; Fourie, G.; Hermanto, C.; Chao, C.P.; Fabregar, E.; Sinohin, V.G.; Masdek, N.; Thangavelu, R.; et al. The distribution and host range of the banana Fusarium wilt fungus, Fusarium oxysporum f. sp. cubense, in Asia. PLoS ONE 2017, 12, e0181630. [Google Scholar] [CrossRef]
- Ploetz, R.; Freeman, S.; Konkol, J.; Al-Abed, A.; Naser, Z.; Shalan, K.; Barakat, R.; Israeli, Y. Tropical race 4 of Panama disease in the Middle East. Phytoparasitica 2015, 43, 283–293. [Google Scholar] [CrossRef]
- Dita, M.; Barquero, M.; Heck, D.; Mizubuti, E.S.G.; Staver, C.P. Fusarium wilt of banana: Current knowledge on epidemiology and research needs toward sustainable disease management. Front. Plant Sci. 2018, 9, 1468. [Google Scholar] [CrossRef] [PubMed]
- García-Bastidas, F.A.; Quintero-Vargas, J.C.; Ayala-Vasquez, M.; Schermer, T.; Seidl, M.F.; Santos-Paiva, M.; Noguera, A.M.; Aguilera-Galvez, C.; Wittenberg, A.; Hofstede, R.; et al. First report of Fusarium wilt Tropical race 4 in Cavendish bananas caused by Fusarium odoratissimum in Colombia. Plant Dis. 2020, 104, 994. [Google Scholar] [CrossRef]
- Rocha, A.D.J.; Soares, J.M.D.S.; Nascimento, F.D.S.; Santos, A.S.; Amorim, V.B.D.O.; Ferreira, C.F.; Haddad, F.; Santos-Serejo, J.A.D.; Amorim, E.P. Improvements in the resistance of the banana species to Fusarium Wilt: A systematic review of methods and perspectives. J. Fungi 2021, 7, 249. [Google Scholar] [CrossRef]
- Buddenhagen, I. Understanding strain diversity in Fusarium oxysporum f. sp. cubense and history of introduction of “Tropical Race 4” to better manage banana production. Acta Hortic. 2009, 828, 193–204. [Google Scholar] [CrossRef]
- Siamak, S.B.; Zheng, S. Banana Fusarium wilt (Fusarium oxysporum f. sp. cubense) control and resistance, in the context of developing wilt-resistant bananas within sustainable production systems. Hortic. Plant J. 2018, 4, 208–218. [Google Scholar] [CrossRef]
- Anderson, J.; Aitken, E. Effect of in Planta Treatment of ‘Cavendish’ Banana with Herbicides and Fungicides on the Colonisation and Sporulation by Fusarium oxysporum f. sp. cubense Subtropical Race 4. J. Fungi 2021, 7, 184. [Google Scholar] [CrossRef]
- Bubici, G.; Kaushal, M.; Prigigallo, M.I.; Gomez-Lama Cabanas, C.; Mercado-Blanco, J. Biological control agents against Fusarium wilt of banana. Front. Microbiol. 2019, 10, 616. [Google Scholar] [CrossRef]
- Catambacan, D.G.; Cumagun, C.J.R. Weed-Associated Fungal Endophytes as Biocontrol Agents of Fusarium oxysporum f. sp. cubense TR4 in Cavendish Banana. J. Fungi 2021, 7, 224. [Google Scholar] [CrossRef] [PubMed]
- D’Hont, A.; Denoeud, F.; Aury, J.M.; Baurens, F.C.; Carreel, F.; Garsmeur, O.; Noel, B.; Bocs, S.; Droc, G.; Rouard, M.; et al. The banana (Musa acuminata) genome and the evolution of monocotyledonous plants. Nature 2012, 21, 213–217. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, J.; Jia, C.; Liu, J.; Li, Y.; Yin, X.; Xu, B.; Jin, Z. De novo characterization of the banana root transcriptome and analysis of gene expression under Fusarium oxysporum f. sp. cubense tropical race 4 infection. BMC Genom. 2012, 13, 650. [Google Scholar] [CrossRef] [PubMed]
- Bai, T.T.; Xie, W.B.; Zhou, P.P.; Wu, Z.L.; Xiao, W.C.; Zhou, L.; Sun, L.; Ruan, X.L.; Li, H.P.; Tumuhimbise, R.; et al. Transcriptome and expression profile analysis of highly resistant and susceptible banana roots challenged with Fusarium oxysporum f. sp. cubense tropical race 4. PLoS ONE 2013, 8, e73945. [Google Scholar] [CrossRef]
- Niu, Y.; Hu, B.; Li, X.; Chen, H.; Takáč, T.; Šamaj, J.; Xu, C. Comparative digital gene expression analysis of tissue-cultured plantlets of highly resistant and susceptible banana cultivars in response to Fusarium oxysporum. Int. J. Mol. Sci. 2018, 19, 350. [Google Scholar] [CrossRef]
- Zhang, L.; Cenci, A.; Rouard, M.; Zhang, D.; Wang, Y.; Tang, W.; Zheng, S.J. Transcriptomic analysis of resistant and susceptible banana corms in response to infection by Fusarium oxysporum f. sp. cubense tropical race 4. Sci. Rep. 2019, 9, 8199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, H.; Ye, Y.; Guo, Y.; Li, H. Comparative transcriptome analysis revealed resistance differences of Cavendish bananas to Fusarium oxysporum f. sp. cubense race1 and race 4. BMC Genet. 2020, 21, 122. [Google Scholar] [CrossRef]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef]
- Tang, D.; Wang, G.; Zhou, J.-M. Receptor kinases in plant-pathogen interactions: More than pattern recognition. Plant Cell. 2017, 29, 618–637. [Google Scholar] [CrossRef]
- Wan, J.; He, M.; Hou, Q.; Zou, L.; Yang, Y.; Wei, Y.; Chen, X. Cell wall associated immunity in plants. Stress Biol. 2021, 1, 3. [Google Scholar] [CrossRef]
- Dong, H.; Fan, H.; Lei, Z.; Wu, C.; Zhou, D.; Li, H. Histological and gene expression analyses in banana reveals the pathogenic differences between races 1 and 4 of banana Fusarium wilt pathogen. Phytopathology 2019, 109, 1029–1042. [Google Scholar] [CrossRef]
- Dita, M.A.; Pérez, V.L.; Martinez De La Parte, E. Inoculation of Fusarium oxysporum f. sp. cubense causal agent of Fusarium wilt in banana. In Technical Manual: Prevention and Diagnostic of Fusarium Wilt (Panama Disease) of Banana Caused by Fusarium oysporum f. sp. cubense Tropical Race 4 (TR4); Vicente, L.P., Dita, M.A.R., Martínez, E., Eds.; United Nations: FAO: Rome, Italy, 2014; pp. 55–58. [Google Scholar]
- Li, W.; Wang, X.; Li, C.; Sun, J.; Li, S.; Peng, M. Dual species transcript profiling during the interaction between banana (Musa acuminata) and the fungal pathogen Fusarium oxysporum f. sp. cubense. BMC Genom. 2019, 20, 519. [Google Scholar] [CrossRef]
- Kazan, K.; Manners, J.M. Linking development to defense: Auxin in plant–pathogen interactions. Trends Plant Sci. 2009, 14, 373–382. [Google Scholar] [CrossRef]
- Cheng, C.; Liu, F.; Sun, X.; Tian, N.; Mensah, R.A.; Li, D.; Zhongxiong, L. Identification of Fusarium oxysporum f. sp. cubense tropical race 4 (Foc TR4) responsive miRNAs in banana root. Sci. Rep. 2019, 9, 13682. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.L. Estudos Histológicos e Moleculares da Interação Musa spp. x Fusarium oxysporum f. sp. cubense. Master’s Thesis, Universidade de São Paulo, Piracicaba, São Paulo, Brazil, 26 April 2013. [Google Scholar]
- Menna, A.; Dora, S.; Sancho-Andres, G.; Kashyap, A.; Meena, M.K.; Sklodowski, K.; Gasperini, D.; Coll, N.S.; Sánchez-Rodríguez, C. A primary cell wall cellulose-dependent defense mechanism against vascular pathogens revealed by time-resolved dual transcriptomics. BMC Biol. 2021, 19, 161. [Google Scholar] [CrossRef] [PubMed]
- Elieh-Ali-Komi, D.; Hamblin, M.R. Chitin and chitosan: Production and application of versatile biomedical nanomaterials. Int. J. Adv. Res. 2016, 4, 411–427. [Google Scholar]
- Lorrai, R.; Ferrari, S. Host Cell Wall damage during pathogen infection: Mechanisms of perception and role in plant-pathogen interactions. Plants 2021, 10, 399. [Google Scholar] [CrossRef]
- Persson, S.; Wei, H.; Milne, J.; Page, G.P.; Somerville, C.R. Identification of genes required for cellulose synthesis by regression analysis of public microarray data sets. Proc. Natl. Acad. Sci. USA 2005, 102, 8633–8638. [Google Scholar] [CrossRef]
- Ding, Z.; Li, M.; Sun, F.; Xi, P.; Sun, L.; Zhang, L.; Jiang, Z. Mitogen-activated protein kinases are associated with the regulation of physiological traits and virulence in Fusarium oxysporum f. sp. cubense. PLoS ONE 2015, 10, e0122634. [Google Scholar] [CrossRef]
- Slusarenko, A.J.; Meier, B.M.; Croft, K.P.C.; Eiben, H.G. Lipoxygenase in Plant Disease. In Mechanisms of Plant Defense Responses. Developments in Plant Pathology; Fritig, B., Legrand, M., Eds.; Springer: Dordrecht, The Netherlands, 1993; Volume 2. [Google Scholar] [CrossRef]
- Hwang, I.S.; Hwang, B.K. The pepper 9-lipoxygenase gene CaLOX1 functions in defense and cell death responses to microbial pathogens. Plant Physiol. 2010, 152, 948–967. [Google Scholar] [CrossRef] [PubMed]
- Umate, P. Genome-wid analysis of lipoxygenase gene family in Arabidopsis and rice. Plant Signal. Behav. 2011, 6, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.; Arif, Y.; Miszczuk, E.; Bajguz, A.; Hayat, S. Specific Roles of Lipoxygenases in Development and Responses to Stress in Plants. Plants 2022, 11, 979. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, H.; Wu, J.; Wang, B.; Tian, N.; Liu, J.; Sun, X.; Wu, H.; Huang, Y.; Lü, P.; et al. Genome-wide identification and expression pattern analysis of lipoxygenase gene family in banana. Sci. Rep. 2021, 11, 9948. [Google Scholar] [CrossRef]
- Li, C.Y.; Deng, G.M.; Yang, J.; Viljoen, A.; Jin, Y.; Kuang, R.B.; Zuo, C.W.; Lv, Z.C.; Yang, Q.S.; Sheng, O.; et al. Transcriptome profiling of resistant and susceptible Cavendish banana roots following inoculation with Fusarium oxysporum f. sp. cubense tropical race 4. BMC Genom. 2012, 13, 374. [Google Scholar] [CrossRef]
- Li, W.B.; Li, C.Q.; Sun, J.B.; Peng, M. Metabolomic, biochemical, and gene expression analyses reveal the underlying responses of resistant and susceptible banana species during early infection with Fusarium oxysporum f. sp cubense. Plant Dis. 2017, 101, 534–543. [Google Scholar] [CrossRef]
- Belkhadir, Y.; Subramaniam, R.; Dangl, J.L. Plant disease resistance protein signaling: NBS–LRR proteins and their partners. Curr. Opin. Plant Biol. 2004, 7, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-López, D.; Herrera-Valencia, V.A.; Góngora-Castillo, E.; García-Laynes, S.; Puch-Hau, C.; López-Ochoa, L.A.; Peraza-Echeverria, S. Genome-Wide Analysis of the LRR-RLP Gene Family in a Wild Banana (Musa acuminata ssp. malaccensis) Uncovers Multiple Fusarium Wilt Resistance Gene Candidates. Genes 2022, 13, 638. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.; Li, H.; Chen, H.; Qiao, F.; Zeng, H. NBS-LRR gene family in banana (Musa acuminata): Genome-wide identification and responses to Fusarium oxysporum f. sp. cubense race 1 and tropical race 4. Eur. J. Plant Pathol. 2020, 157, 549–563. [Google Scholar] [CrossRef]
- Rushton, P.J.; Somssich, I.E.; Ringler, P.; Shen, Q.J. WRKY transcription factors. Trends Plant Sci. 2010, 15, 247–258. [Google Scholar] [CrossRef]
- Phukan, U.J.; Jeena, G.S.; Shukla, R.K. WRKY transcription factors: Molecular regulation and stress responses in plants. Front. Plant Sci. 2016, 7, 760. [Google Scholar] [CrossRef]
- Mao, G.; Meng, X.; Liu, Y.; Zheng, Z.; Chen, Z.; Zhang, S. Phosphorylation of a WRKY transcription factor by two pathogen-responsive MAPKs drives phytoalexin biosynthesis in Arabidopsis. Plant Cell. 2011, 23, 1639–1653. [Google Scholar] [CrossRef] [PubMed]
- Dang, F.F.; Wang, Y.N.; Yu, L.; Eulgem, T.; Lai, Y.; Liu, Z.Q.; Wang, X.; Qiu, A.L.; Zhang, T.X.; Lin, J.; et al. CaWRKY40, a WRKY protein of pepper, plays an important role in the regulation of tolerance to heat stress and resistance to Ralstonia solanacearum infection. Plant Cell Environ. 2013, 36, 757–774. [Google Scholar] [CrossRef]
- Sarris, P.F.; Duxbury, Z.; Huh, S.U.; Ma, Y.; Segonzac, C.; Sklenar, J.; Derbyshire, P.; Cevik, V.; Rallapalli, G.; Saucet, S.B.; et al. A plant immune receptor detects pathogen effectors that target WRKY transcription factors. Cell 2015, 161, 1089–1100. [Google Scholar] [CrossRef]
- Gao, Y.F.; Liu, J.K.; Yang, F.M.; Zhang, G.Y.; Wang, D.; Zhang, L.; Ou, Y.B.; Yao, Y.A. The WRKY transcription factor WRKY8 promotes resistance to pathogen infection and mediates drought and salt stress tolerance in Solanum lycopersicum. Physiol. Plant. 2020, 168, 98–117. [Google Scholar] [CrossRef]
- Eulgem, T.; Rushton, P.J.; Robatzek, S.; Somssich, I.E. The WRKY superfamily of plant transcription factors. Trends Plant Sci. 2000, 5, 199–206. [Google Scholar] [CrossRef]
- Jiang, J.; Ma, S.; Ye, N.; Jiang, M.; Cao, J.; Zhang, J. WRKY transcription factors in plant responses to stresses. J. Integr. Plant Biol. 2017, 59, 86–101. [Google Scholar] [CrossRef] [Green Version]
- Breen, S.J.; Williams, M.; Outram, B.; Kobe, P.S.; Solomon, P.S. Emerging insights into the functions of pathogenesis-related protein 1. Trends Plant Sci. 2017, 22, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Tian, T.; Feng, L.; Yang, X.; Li, L.; Tan, X.; Wu, W.; Li, Z.; Treves, H.; Serneels, F.; et al. Pathogenesis-related protein 1 suppresses oomycete pathogen by targeting against AMPK kinase complex. J. Adv. Res. 2022, in press. [Google Scholar] [CrossRef]
- Lu, S.; Faris, J.D.; Sherwood, R.; Friesen, T.L.; Edwards, M.C. A dimeric PR-1-type pathogenesis-related protein interacts with ToxA and potentially mediates ToxA-induced necrosis in sensitive wheat. Mol. Plant Pathol. 2014, 15, 650–663. [Google Scholar] [CrossRef] [PubMed]
- Breen, S.J.; Williams, B.; Winterberg, B.; Kobe, P.S. Solomon Wheat PR-1 proteins are targeted by necrotrophic pathogen effector proteins. Plant J. 2016, 88, 13–25. [Google Scholar] [CrossRef]
- Li, W.; Ge, X.; Wu, W.; Wang, W.; Hu, Y.; Mo, Y.; Sun, D.; Shi, S.; Xie, J. Identification of defense-related genes in banana roots infected by Fusarium oxysporum f. sp. cubense tropical race 4. Euphytica 2015, 205, 837–849. [Google Scholar] [CrossRef]
- Wang, Z.; Jia, C.; Li, J.; Huang, S.; Xu, B.; Jin, Z. Activation of salicylic acid metabolism and signal transduction can enhance resistance to Fusarium wilt in banana (Musa acuminata L. AAA group, cv. Cavendish). Funct. Integr. Genom. 2015, 15, 47–62. [Google Scholar] [CrossRef]
- Thakker, J.N.; Patel, S.; Dhandhukia, P.C. Induction of defense-related enzymes in banana plants: Effect of live and dead pathogenic strain of Fusarium oxysporum f. sp. cubense. Int. Sch. Res. Not. 2013, 2013, 601303. [Google Scholar] [CrossRef]
- Berg, N.V.D.; Berger, D.K.; Hein, I.; Birch, P.R.J.; Wingfield, M.J.; Viljoen, A. Tolerance in banana to Fusarium wilt is associated with early upregulation of cell wall-strengthening genes in the roots. Mol. Plant Pathol. 2007, 8, 333–341. [Google Scholar] [CrossRef]
- Ceita, G.D.O.; Macedo, J.N.A.; Santos, T.B.; Alemanno, L.; Gesteira, A.D.; Micheli, F.; Mariano, A.C.; Gramacho, K.P.; Silva, D.D.C.; Meinhardt, L.; et al. Involvement of calcium oxalate degradation during programmed cell death in Theobroma cacao tissues triggered by the hemibiotrophic fungus Moniliophthora pemiciosa. Plant Sci. 2007, 173, 106–117. [Google Scholar] [CrossRef]
- Warman, N.M.; Aitken, E.A. The movement of Fusarium oxysporum f. sp. cubense (sub-tropical race 4) in susceptible cultivars of banana. Front. Plant. Sci. 2018, 9, 1748. [Google Scholar] [CrossRef] [Green Version]
- Rocha, A.D.J.; Ferreira, M.D.S.; Rocha, L.D.S.; Oliveira, S.A.; Amorim, E.P.; Mizubuti, E.S.; Haddad, F. Interaction between Fusarium oxysporum f. sp. cubense and Radopholus similis can lead to changes in the resistance of banana cultivars to Fusarium wilt. Eur. J. Plant Pathol. 2020, 158, 403–417. [Google Scholar] [CrossRef]
- Velame, K.V.C. Genes de Bananeira Candidatos à Resistência a Murcha de Fusarium e Caracterização da Agressividade e Virulência do Fungo. Ph.D. Thesis, Universidade Estadual de Feira de Santana, Feira de Santana, Bahia, Brazil, 31 May 2017. [Google Scholar]
- Batista, I.C.; Heck, D.W.; Santos, A.; Alves, G.; Ferro, C.G.; Dita, M.; Mizubuti, E.S. The Brazilian population of Fusarium oxysporum f. sp. cubense is not structured by VCG or by geographic origin. bioRxiv 2022, 1–31. [Google Scholar] [CrossRef]
- Costa, S.N.; Bragança, C.A.D.; Ribeiro, L.R.; Amorim, E.P.; Silva, S.O.; Dita, M.; Laranjeira, F.F.; Haddad, F. Genetic structure of Fusarium oxysporum f. sp. cubense in diferent regions from Brazil. Plant Pathol. 2015, 64, 37–146. [Google Scholar] [CrossRef]
- Chen, A.; Sun, J.; Matthews, A.; Armas-Egas, L.; Chen, N.; Hamill, S.; Mintoff, S.; Tran-Nguyen, L.T.T.; Batley, J.; Aitken, E.A.B. Assessing variations in host resistance to Fusarium oxysporum f sp. cubense race 4 in Musa Species, with a focus on the subtropical race 4. Front. Microbiol. 2019, 10, 1062. [Google Scholar] [CrossRef]
- Raman, T.; Edwin Raj, E.; Muthukathan, G.; Loganathan, M.; Periyasamy, P.; Natesh, M.; Manivasakan, P.; Kotteeswaran, S.; Rajendran, S.; Subbaraya, U. Comparative Whole-Genome Sequence Analyses of Fusarium Wilt Pathogen (Foc R1, STR4 and TR4) Infecting Cavendish (AAA) Bananas in India, with a Special Emphasis on Pathogenicity Mechanisms. J. Fungi 2021, 7, 717. [Google Scholar] [CrossRef] [PubMed]
- Castricini, A.; Coelho, E.F.; Rodrigues, M.G.V.; Coutinho, R.C. Caracterização pós-colheita de frutos de bananeira ‘BRS Platina’ de primeiro ciclo, sob regulação do déficit de irrigação. Rev. Bras. Frutic. 2012, 34, 1013–1021. [Google Scholar] [CrossRef]
- Marques, P.R.R.; Donato, S.L.R.; São José, A.R.; Rosa, R.C.C.; de Magalhães Arantes, A. Nutritional status and production of ‘Prata-Anã’(AAB) and ‘BRS Platina’(AAAB) banana plants with organic fertilization. Nativa 2022, 10, 60–68. [Google Scholar] [CrossRef]
- de Matos, A.P.; Cordeiro, Z.J.M.; Oliveira, S.S.; Amorim, E.P.; Ferreira, D.M.V. Reaction of diploid (AA) and tetraploid (AAAB) banana hybrids to Fusarium wilt under field conditions. Acta Hortic. 2011, 897, 387–390. [Google Scholar] [CrossRef]
- Ribeiro, L.R.; Silva, S.O.; Oliveira, S.A.S.; Amorim, E.P.; Serejo, J.A.S.S.; Haddad, F. Sources of resistance to Fusarium oxysporum f. sp. cubense in banana germplasm. Rev. Brasil. Fruticult. 2018, 40, e-202. [Google Scholar] [CrossRef]
- Gonçalves, Z.S.; Haddad, F.; Amorim, V.B.O.; Ferreira, C.F.; de Oliveira, S.; Amorim, E.P. Agronomic characterization and identification of banana genotypes resistant to Fusarium wilt race 1. Eur. J. Plant Pathol. 2019, 155, 1093–1103. [Google Scholar] [CrossRef]
- Santos, A.S.; Amorim, E.P.; Almeida, A.A.F.; Ferreira, C.F.; Coelho Filho, M.A.; Pirovani, C.P. Diversity of banana diploid hybrids: An assessment based on a hydroponic system. Plant Breed. 2020, 139, 1251–1265. [Google Scholar] [CrossRef]
- Better Bananas. Panama TR4 Variety Screening Trial (December 2018) Sub-Trial Results (Plant and First Ratoon). Available online: https://betterbananas.com.au/2022/03/04/panama-tr4-variety-screening-trial-december-2018-sub-trial-results-plant-and-first-ratoon/ (accessed on 3 June 2022).
- Acuña, R.; Rouard, M.; Leiva, A.M.; Marques, C.; Olortegui, A.; Ureta, C.; Cabrera-Pintado, R.M.; Rojas, J.C.; López, D.; Cenci, A.; et al. First report of Fusarium oxysporum f. sp. cubense Tropical Race 4, causing Fusarium wilt in Cavendish bananas in Peru. Plant Dis. 2021, 106, 2268. [Google Scholar] [CrossRef] [PubMed]
- Heck, D.W.; Dita, M.; Del Ponte, E.M.; Mizubuti, E.S.G. Incidence, spatial pattern and temporal progress of fusarium wilt of bananas. J. Fungi 2021, 7, 646. [Google Scholar] [CrossRef]
- McKinney, H.H. Influence of soil, temperature and moisture on infection of wheat seedlings by Helminthosporium sativum. J. Agric. Res. 1923, 26, 195–217. [Google Scholar]
- Zhao, L.; Ding, Q.; Zeng, J.; Wang, F.R.; Zhang, J.; Fan, S.J.; He, X.Q. An improved CTAB-Ammonium Acetate method for total RNA isolation from cotton. Phytochem. Anal. 2012, 23, 647–650. [Google Scholar] [CrossRef] [PubMed]
- Podevin, N.; Krauss, A.; Henry, I.; Swennen, R.; Remy, S. Selection and validation of reference genes for quantitative RT-PCR expression studies of the non-model crop Musa. Mol. Breed. 2012, 30, 1237–1252. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Pfaffl, M.W.; Horgan, G.W.; Dempfle, L. Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 2002, 30, e30–e36. [Google Scholar] [CrossRef]
- Karnovsky, M.J. A formaldehydeglutaraldehyde fixative of high osmolality for use in electron microscopy. J. Cell Biol. 1965, 27, 137. [Google Scholar]
- Johansen, D.A. Plant Microtechnique; Mc Graw Hill: New York, NY, USA, 1940; p. 523. [Google Scholar]
- Hughes, J.; McCully, M.E. The use of an optical brightener in the study of plant structures. Stain Technol. 1975, 50, 319–329. [Google Scholar] [CrossRef] [PubMed]
- Phillips, J.M.; Hayman, D.S. Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection. Trans. Br. Mycol. Soc. 1970, 55, 158–161. [Google Scholar] [CrossRef]



| ID | Gene | Description | Sequence (5′-3′) | pb | Reference |
|---|---|---|---|---|---|
| GSMUA_Achr5T15720_001 | CESA7 | Cellulose synthase A catalytic subunit 7 | F: GAGAATGGAGAACGGGTGCA | 108 | [32] |
| R: CCCCTCCATGTCTCTCTCCA | |||||
| GSMUA_Achr8T02300_001 | ATL | Auxin transporter-like protein 1 | F: GGTTCAGCTGCTCCTCCAAT | 161 | [32] |
| R: AGAACAGCTGCAGGATCACC | |||||
| GSMUA_Achr8T15700_001 | PI206 | Putative Disease resistance response protein 206 | F: AGTACAACGGGAGCAGCTTC | 128 | [32] |
| R: GATGAGCCTGCTGATGGTGT | |||||
| XM_009417035.2 | WRKY22 | Transcription factor WRKY 22 | F: CGTGACGTACGAAGGAGAGCA | 95 | [27] |
| R: GGTCAACGCGAAGTCAACCA | |||||
| XM_009417035.2 | PR1 | Pathogenesis-related proteins 1 | F: AGTTATGGACGAGCTACCCG | 77 | [27] |
| R: GTAGCTGAAGTACTTCCCCTC | |||||
| XM_009415745.2 | CHI | Chitinase | F: TACTGGAACTACAACTACGGAGC | 82 | [27] |
| R: CGTTCTGCTCGAGGTACTC | |||||
| XM_008803483.2 | LOX | Lipoxygenases | F: ACGATGCAGACGGTATTGGAGT | 94 | [27] |
| R: GGTACTGTCCGAAGTTGACG | |||||
| 25SMU | 25S | 25S rRNA | F: ACATTGTCAGGTGGGGAGTT | 106 | [83] |
| R: CCTTTTGTTCCACACGAGATT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rocha, A.d.J.; Soares, J.M.d.S.; Nascimento, F.d.S.; Rocha, A.d.S.; Amorim, V.B.O.d.; Ramos, A.P.d.S.; Ferreira, C.F.; Haddad, F.; Amorim, E.P. Molecular, Histological and Histochemical Responses of Banana Cultivars Challenged with Fusarium oxysporum f. sp. cubense with Different Levels of Virulence. Plants 2022, 11, 2339. https://doi.org/10.3390/plants11182339
Rocha AdJ, Soares JMdS, Nascimento FdS, Rocha AdS, Amorim VBOd, Ramos APdS, Ferreira CF, Haddad F, Amorim EP. Molecular, Histological and Histochemical Responses of Banana Cultivars Challenged with Fusarium oxysporum f. sp. cubense with Different Levels of Virulence. Plants. 2022; 11(18):2339. https://doi.org/10.3390/plants11182339
Chicago/Turabian StyleRocha, Anelita de Jesus, Julianna Matos da Silva Soares, Fernanda dos Santos Nascimento, Adailson dos Santos Rocha, Vanusia Batista Oliveira de Amorim, Andresa Priscila de Souza Ramos, Cláudia Fortes Ferreira, Fernando Haddad, and Edson Perito Amorim. 2022. "Molecular, Histological and Histochemical Responses of Banana Cultivars Challenged with Fusarium oxysporum f. sp. cubense with Different Levels of Virulence" Plants 11, no. 18: 2339. https://doi.org/10.3390/plants11182339
APA StyleRocha, A. d. J., Soares, J. M. d. S., Nascimento, F. d. S., Rocha, A. d. S., Amorim, V. B. O. d., Ramos, A. P. d. S., Ferreira, C. F., Haddad, F., & Amorim, E. P. (2022). Molecular, Histological and Histochemical Responses of Banana Cultivars Challenged with Fusarium oxysporum f. sp. cubense with Different Levels of Virulence. Plants, 11(18), 2339. https://doi.org/10.3390/plants11182339










