Seaweed Extracts as an Effective Gateway in the Search for Novel Antibiofilm Agents against Staphylococcus aureus
Abstract
:1. Introduction
2. Results
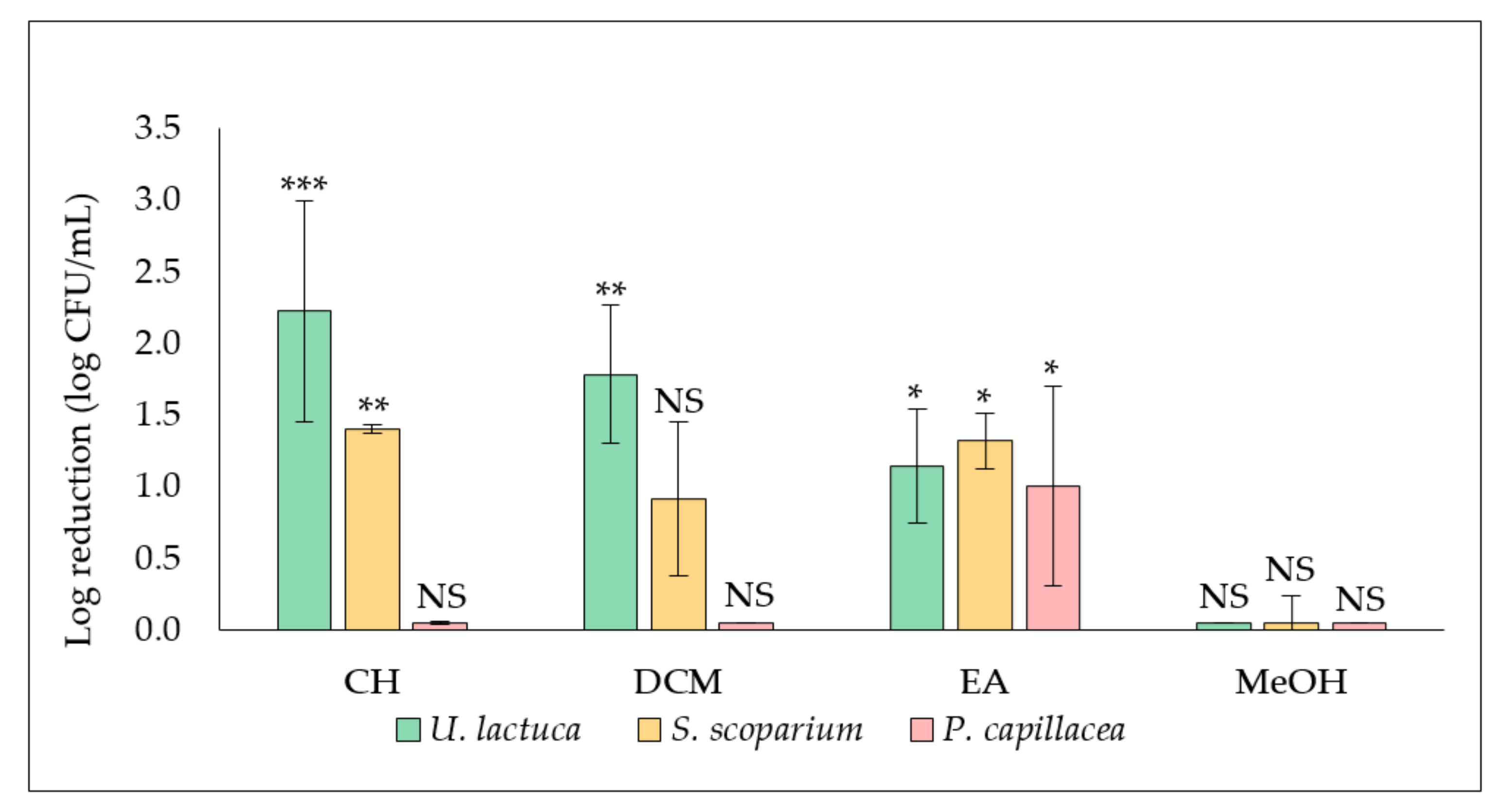
2.1. Evaluation of the Antibiofilm Activity of Algal Extracts
2.1.1. Effect of Extracts on S. aureus Biofilm Formation and Development (Extract Added at t0)
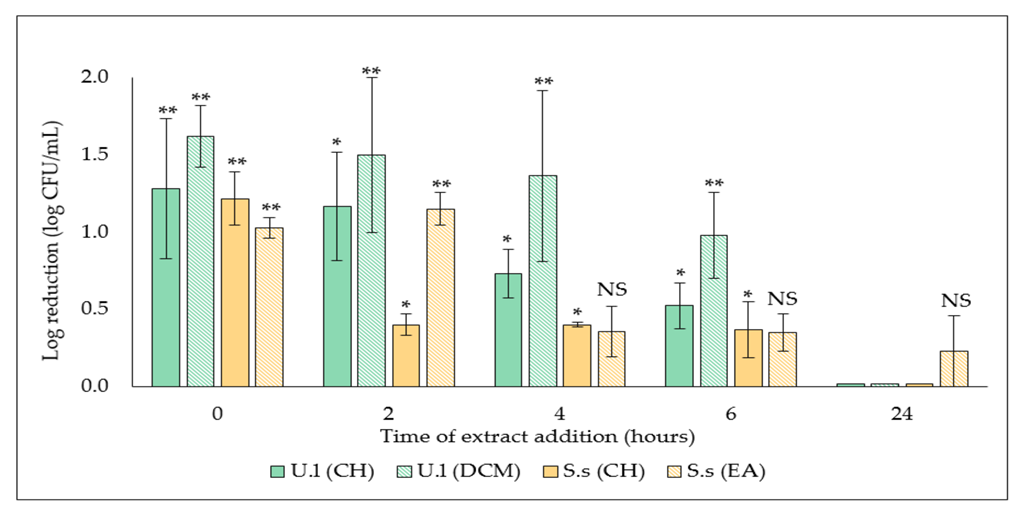
2.1.2. Determination of Biofilm Development Stage Targeted by the Selected Extracts
2.2. Checking the Potential Bactericidal Effect of the Selected Extracts
2.3. Effect of the Selected Extracts on S. aureus Hydrophobicity—Contact Angle Measurement Method
3. Discussion
4. Materials and Methods
4.1. Collection of Algal Materials and Extract Preparation
4.2. Bacterial Strain and Culture Media
4.3. Evaluation of the Antibiofilm Activity of Extracts
4.3.1. Effect of Extracts on S. aureus Biofilm Formation and Development (Extract Added at t0)
4.3.2. Epifluorescence Microscopic Analysis of Treated Biofilms (Extract Added at t0)
4.3.3. Determination of Biofilm Development Stage Targeted by the Selected Extracts
4.4. Checking the Potential Bactericidal Activity of the Selected Extracts
4.5. Effect of the Selected Extract on S. aureus Hydrophobicity—Contact Angle Measurement Method
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Kiuru, P.; D’Auria, M.V.; Muller, C.D.; Tammela, P.; Vuorela, H.; Yli-Kauhaluoma, J. Exploring marine resources for bioactive compounds. Planta Med. 2014, 80, 1234–1246. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, C. Marine Natural Products in Medicinal Chemistry. ACS Med. Chem. Lett. 2018, 9, 959–961. [Google Scholar] [CrossRef] [PubMed]
- Leandro, A.; Pereira, L.; Goncalves, A.M.M. Diverse Applications of Marine Macroalgae. Mar. Drugs 2019, 18, 17. [Google Scholar] [CrossRef]
- Shannon, E.; Abu-Ghannam, N. Antibacterial Derivatives of Marine Algae: An Overview of Pharmacological Mechanisms and Applications. Mar. Drugs 2016, 14, 81. [Google Scholar] [CrossRef] [PubMed]
- Bhowmick, S.; Mazumdar, A.; Moulick, A.; Adam, V. Algal metabolites: An inevitable substitute for antibiotics. Biotechnol. Adv. 2020, 43, 107571. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.-C.; Wingender, J.; Szewzyk, U.; Steinberg, P.; Rice, S.A.; Kjelleberg, S. Biofilms: An emergent form of bacterial life. Nat. Rev. Microbiol. 2016, 14, 563–575. [Google Scholar] [CrossRef]
- Uruen, C.; Chopo-Escuin, G.; Tommassen, J.; Mainar-Jaime, R.C.; Arenas, J. Biofilms as Promoters of Bacterial Antibiotic Resistance and Tolerance. Antibiotics 2020, 10, 3. [Google Scholar] [CrossRef]
- Pendleton, J.N.; Gorman, S.P.; Gilmore, B.F. Clinical relevance of the ESKAPE pathogens. Expert Rev. Anti Infect. Ther. 2013, 11, 297–308. [Google Scholar] [CrossRef] [PubMed]
- WHO. Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Tong, S.Y.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef] [PubMed]
- Archer, N.K.; Mazaitis, M.J.; Costerton, J.W.; Leid, J.G.; Powers, M.E.; Shirtliff, M.E. Staphylococcus aureus biofilms: Properties, regulation, and roles in human disease. Virulence 2011, 2, 445–459. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tolker-Nielsen, T. Biofilm Development. Microbiol. Spectr. 2015, 3, MB-0001-2014. [Google Scholar] [CrossRef] [PubMed]
- Rumbaugh, K.P.; Sauer, K. Biofilm dispersion. Nat. Rev. Microbiol. 2020, 18, 571–586. [Google Scholar] [CrossRef] [PubMed]
- Suresh, M.K.; Biswas, R.; Biswas, L. An update on recent developments in the prevention and treatment of Staphylococcus aureus biofilms. Int. J. Med. Microbiol. 2019, 309, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Panda, A.K.; De Mandal, S.; Shakeel, M.; Bisht, S.S.; Khan, J. Natural Anti-biofilm Agents: Strategies to Control Biofilm-Forming Pathogens. Front. Microbiol. 2020, 11, 566325. [Google Scholar] [CrossRef] [PubMed]
- Guzzo, F.; Scognamiglio, M.; Fiorentino, A.; Buommino, E.; D’Abrosca, B. Plant Derived Natural Products against Pseudomonas aeruginosa and Staphylococcus aureus: Antibiofilm Activity and Molecular Mechanisms. Molecules 2020, 25, 5024. [Google Scholar] [CrossRef]
- Rima, M.; Trognon, J.; Latapie, L.; Chbani, A.; Roques, C.; El Garah, F. Seaweed Extracts: A Promising Source of Antibiofilm Agents with Distinct Mechanisms of Action against Pseudomonas aeruginosa. Mar. Drugs 2022, 20, 92. [Google Scholar] [CrossRef]
- Preda, V.G.; Sandulescu, O. Communication is the key: Biofilms, quorum sensing, formation and prevention. Discoveries 2019, 7, e100. [Google Scholar] [CrossRef]
- Campanac, C.; Pineau, L.; Payard, A.; Baziard-Mouysset, G.; Roques, C. Interactions between Biocide Cationic Agents and Bacterial Biofilms. Antimicrob. Agents Chemother. 2002, 46, 1469–1474. [Google Scholar] [CrossRef]
- Bjarnsholt, T.; Ciofu, O.; Molin, S.; Givskov, M.; Hoiby, N. Applying insights from biofilm biology to drug development—Can a new approach be developed? Nat. Rev. Drug Discov. 2013, 12, 791–808. [Google Scholar] [CrossRef]
- Koo, H.; Allan, R.N.; Howlin, R.P.; Stoodley, P.; Hall-Stoodley, L. Targeting microbial biofilms: Current and prospective therapeutic strategies. Nat. Rev. Microbiol. 2017, 15, 740–755. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liang, E.; Cheng, Y.; Mahmood, T.; Ge, F.; Zhou, K.; Bao, M.; Lv, L.; Li, L.; Yi, J.; et al. Is combined medication with natural medicine a promising therapy for bacterial biofilm infection? Biomed. Pharmacother. 2020, 128, 110184. [Google Scholar] [CrossRef] [PubMed]
- Melander, R.J.; Basak, A.K.; Melander, C. Natural products as inspiration for the development of bacterial antibiofilm agents. Nat. Prod. Rep. 2020, 37, 1454–1477. [Google Scholar] [CrossRef] [PubMed]
- Dahms, H.; Dobretsov, S. Antifouling Compounds from Marine Macroalgae. Mar. Drugs 2017, 15, 265. [Google Scholar] [CrossRef] [PubMed]
- Stowe, S.D.; Richards, J.J.; Tucker, A.T.; Thompson, R.; Melander, C.; Cavanagh, J. Anti-biofilm compounds derived from marine sponges. Mar. Drugs 2011, 9, 2010–2035. [Google Scholar] [CrossRef]
- Saeed, A.; Abotaleb, S.; Gheda, S.; Alam, N.; Elmehalawy, A. In vitro Assessment of Antimicrobial, Antioxidant and Anticancer Activities of Some Marine Macroalgae. Egypt. J. Bot. 2019, 60, 81–96. [Google Scholar] [CrossRef]
- Anjali, K.P.; Sangeetha, B.M.; Devi, G.; Raghunathan, R.; Dutta, S. Bioprospecting of seaweeds (Ulva lactuca and Stoechospermum marginatum): The compound characterization and functional applications in medicine-a comparative study. J. Photochem. Photobiol. B 2019, 200, 111622. [Google Scholar] [CrossRef]
- Rima, M.; Chbani, A.; Roques, C.; El Garah, F. Comparative study of the insecticidal activity of a high green plant (Spinacia oleracea) and a chlorophytae algae (Ulva lactuca) extracts against Drosophila melanogaster fruit fly. Ann. Pharm. Fr. 2021, 79, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Guner, A.; Nalbantsoy, A.; Sukatar, A.; Karabay Yavasoglu, N.U. Apoptosis-inducing activities of Halopteris scoparia L. Sauvageau (Brown algae) on cancer cells and its biosafety and antioxidant properties. Cytotechnology 2019, 71, 687–704. [Google Scholar] [CrossRef]
- Campos, A.M.; Matos, J.; Afonso, C.; Gomes, R.; Bandarra, N.M.; Cardoso, C. Azorean macroalgae (Petalonia binghamiae, Halopteris scoparia and Osmundea pinnatifida) bioprospection: A study of fatty acid profiles and bioactivity. Int. J. Food Sci. Technol. 2018, 54, 880–890. [Google Scholar] [CrossRef]
- Ismail, G.; Gheda, S.; Abo-Shady, A.; Abdel-Karim, O. In vitro potential activity of some seaweeds as antioxidants and inhibitors of diabetic enzymes. Food Sci. Technol. 2020, 40, 681–691. [Google Scholar] [CrossRef] [Green Version]
- Shobier, A.H.; Abdel Ghani, S.A.; Barakat, K.M. GC/MS spectroscopic approach and antifungal potential of bioactive extracts produced by marine macroalgae. Egypt. J. Aquat. Res. 2016, 42, 289–299. [Google Scholar] [CrossRef]
- Pushparaj, A.; Raubbin, R.; Balasankar, T. Antibacterial activity of Kappaphycus alvarezii and Ulva lactuca extracts against human pathogenic bacteria. Int. J. Curr. Microbiol. Appl. Sci. 2014, 3, 432–436. [Google Scholar]
- De Alencar, D.B.; de Carvalho, F.C.T.; Reboucas, R.H.; Dos Santos, D.R.; Dos Santos Pires-Cavalcante, K.M.; de Lima, R.L.; Baracho, B.M.; Bezerra, R.M.; Viana, F.A.; Dos Fernandes Vieira, R.H.S.; et al. Bioactive extracts of red seaweeds Pterocladiella capillacea and Osmundaria obtusiloba (Floridophyceae: Rhodophyta) with antioxidant and bacterial agglutination potential. Asian Pac. J. Trop. Med. 2016, 9, 372–379. [Google Scholar] [CrossRef]
- Dulger, B.; Hacioglu, N.; Erdugan, H.; Aysel, V. Antimicrobial Activity of Some Brown Algae from Turkey. Asian J. Chem. 2009, 21, 4113–4117. [Google Scholar]
- Yuvaraj, N.; Arul, V. Preliminary Screening of Anti-Biofilm, Anti-Larval Settlement and Cytotoxic Potential of Seaweeds and Seagrasses Collected from Pondicherry and Rameshwaram Coastal Line, India. WJFMS 2014, 6, 169–175. [Google Scholar] [CrossRef]
- Pantanella, F.; Valenti, P.; Natalizi, T. Analytical techniques to study microbial biofilm on abiotic surfaces: Pros and cons of the main techniques currently in use. Ann. Ig 2013, 25, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Fontaine, B.M.; Nelson, K.; Lyles, J.T.; Jariwala, P.B.; Garcia-Rodriguez, J.M.; Quave, C.L.; Weinert, E.E. Identification of Ellagic Acid Rhamnoside as a Bioactive Component of a Complex Botanical Extract with Anti-biofilm Activity. Front. Microbiol. 2017, 8, 496. [Google Scholar] [CrossRef]
- Liu, M.; Wu, X.; Li, J.; Liu, L.; Zhang, R.; Shao, D.; Du, X. The specific anti-biofilm effect of gallic acid on Staphylococcus aureus by regulating the expression of the ica operon. Food Control 2017, 73, 613–618. [Google Scholar] [CrossRef]
- Allkja, J.; van Charante, F.; Aizawa, J.; Reigada, I.; Guarch-Perez, C.; Vazquez-Rodriguez, J.A.; Cos, P.; Coenye, T.; Fallarero, A.; Zaat, S.A.J.; et al. Interlaboratory study for the evaluation of three microtiter plate-based biofilm quantification methods. Sci. Rep. 2021, 11, 13779. [Google Scholar] [CrossRef] [PubMed]
- Xiang, H.; Cao, F.; Ming, D.; Zheng, Y.; Dong, X.; Zhong, X.; Mu, D.; Li, B.; Zhong, L.; Cao, J.; et al. Aloe-emodin inhibits Staphylococcus aureus biofilms and extracellular protein production at the initial adhesion stage of biofilm development. Appl. Microbiol. Biotechnol. 2017, 101, 6671–6681. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Ravaioli, S.; Montanaro, L. Polysaccharide intercellular adhesin in biofilm: Structural and regulatory aspects. Front. Cell. Infect. Microbiol. 2015, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Foster, T.J.; Geoghegan, J.A.; Ganesh, V.K.; Hook, M. Adhesion, invasion and evasion: The many functions of the surface proteins of Staphylococcus aureus. Nat. Rev. Microbiol. 2014, 12, 49–62. [Google Scholar] [CrossRef] [Green Version]
- Bauer, J.; Siala, W.; Tulkens, P.M.; Van Bambeke, F. A combined pharmacodynamic quantitative and qualitative model reveals the potent activity of daptomycin and delafloxacin against Staphylococcus aureus biofilms. Antimicrob. Agents Chemother. 2013, 57, 2726–2737. [Google Scholar] [CrossRef]
- Krasowska, A.; Sigler, K. How microorganisms use hydrophobicity and what does this mean for human needs? Front. Cell. Infect. Microbiol. 2014, 4, 112. [Google Scholar] [CrossRef] [PubMed]
- Heilmann, C. Adhesion mechanisms of staphylococci. Adv. Exp. Med. Biol. 2011, 715, 105–123. [Google Scholar] [CrossRef]
- Forson, A.M.; van der Mei, H.C.; Sjollema, J. Impact of solid surface hydrophobicity and micrococcal nuclease production on Staphylococcus aureus Newman biofilms. Sci. Rep. 2020, 10, 12093. [Google Scholar] [CrossRef]
- Kouidhi, B.; Zmantar, T.; Hentati, H.; Bakhrouf, A. Cell surface hydrophobicity, biofilm formation, adhesives properties and molecular detection of adhesins genes in Staphylococcus aureus associated to dental caries. Microb. Pathog. 2010, 49, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Braga, P.; Reggio, S. Correlation between reduction of surface hydrophobicity of and the decrease in its adhesiveness induced by subinhibitory concentrations of brodimoprim. Pharmacol. Res. 1995, 32, 315–319. [Google Scholar] [CrossRef]
- Allegrone, G.; Ceresa, C.; Rinaldi, M.; Fracchia, L. Diverse Effects of Natural and Synthetic Surfactants on the Inhibition of Staphylococcus aureus Biofilm. Pharmaceutics 2021, 13, 1172. [Google Scholar] [CrossRef]
- Zhao, F.; Wang, P.; Lucardi, R.D.; Su, Z.; Li, S. Natural Sources and Bioactivities of 2,4-Di-Tert-Butylphenol and Its Analogs. Toxins 2020, 12, 35. [Google Scholar] [CrossRef]
- Khalilzadeh, P.; Lajoie, B.; El Hage, S.; Furiga, A.; Baziard, G.; Berge, M.; Roques, C. Growth inhibition of adherent Pseudomonas aeruginosa by an N-butanoyl-L-homoserine lactone analog. Can. J. Microbiol. 2010, 56, 317–325. [Google Scholar] [CrossRef] [PubMed]
- Feuillolay, C.; Pecastaings, S.; Le Gac, C.; Fiorini-Puybaret, C.; Luc, J.; Joulia, P.; Roques, C. A Myrtus communis extract enriched in myrtucummulones and ursolic acid reduces resistance of Propionibacterium acnes biofilms to antibiotics used in acne vulgaris. Phytomedicine 2016, 23, 307–315. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elabed, S.; Elabed, A.; Sadiki, M.; Elfarricha, O.; Ibnsouda, S. Assessment of the Salvia officinalis and Myrtus communis Aqueous Extracts Effect on Cell Surface Tension Parameters and Hydrophobicity of Staphylococcus aureus CIP54354 and Bacillus subtilis ILP142B. J. Appl. Sci. 2017, 17, 246–252. [Google Scholar] [CrossRef]

| Initial Bacterial Suspension | Green Alga U. lactuca | Brown Alga S. scoparium | ||
|---|---|---|---|---|
| CH | DCM | CH | EA | |
| 105 CFU/mL | 0.98 ± 0.00 | 0.96 ± 0.01 | 0.99 ± 0.02 | 0.95 ± 0.00 |
| 102 CFU/mL | 1.00 ± 0.00 | 0.99 ± 0.00 | 0.99 ± 0.00 | 1.02 ± 0.00 |
| Sample | Contact Angle ϴ° | Water Droplet Deposited on the Bacterial Layers |
|---|---|---|
| Control | 94.2 ± 3.8° |  |
| U.l (CH) | 85.6 ± 0.9° ** | |
| U.l (DCM) | 57.9 ± 8.1° *** | |
| S.s (CH) | 94.1 ± 4.1° NS | |
| S.s (EA) | 90.8 ± 6.3° NS |
| Stage of Biofilm Formation | Time Point of Extract Addition | ||||
|---|---|---|---|---|---|
| 0 | 2 h | 4 h | 6 h | 24 h | |
| 0 | ↓ + | ↓ | ↓ | ↓ | ↓ |
| 2 h | + | ||||
| 4 h | + | ||||
| 6 h | + | ||||
| 24 h | Scraping time | + | |||
| 48 h | _ | Scraping time | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rima, M.; Chbani, A.; Roques, C.; El Garah, F. Seaweed Extracts as an Effective Gateway in the Search for Novel Antibiofilm Agents against Staphylococcus aureus. Plants 2022, 11, 2285. https://doi.org/10.3390/plants11172285
Rima M, Chbani A, Roques C, El Garah F. Seaweed Extracts as an Effective Gateway in the Search for Novel Antibiofilm Agents against Staphylococcus aureus. Plants. 2022; 11(17):2285. https://doi.org/10.3390/plants11172285
Chicago/Turabian StyleRima, Maya, Asma Chbani, Christine Roques, and Fatima El Garah. 2022. "Seaweed Extracts as an Effective Gateway in the Search for Novel Antibiofilm Agents against Staphylococcus aureus" Plants 11, no. 17: 2285. https://doi.org/10.3390/plants11172285
APA StyleRima, M., Chbani, A., Roques, C., & El Garah, F. (2022). Seaweed Extracts as an Effective Gateway in the Search for Novel Antibiofilm Agents against Staphylococcus aureus. Plants, 11(17), 2285. https://doi.org/10.3390/plants11172285







