Genomic and Morphological Differentiation of Spirit Producing Agave angustifolia Traditional Landraces Cultivated in Jalisco, Mexico
Abstract
:1. Introduction
2. Results
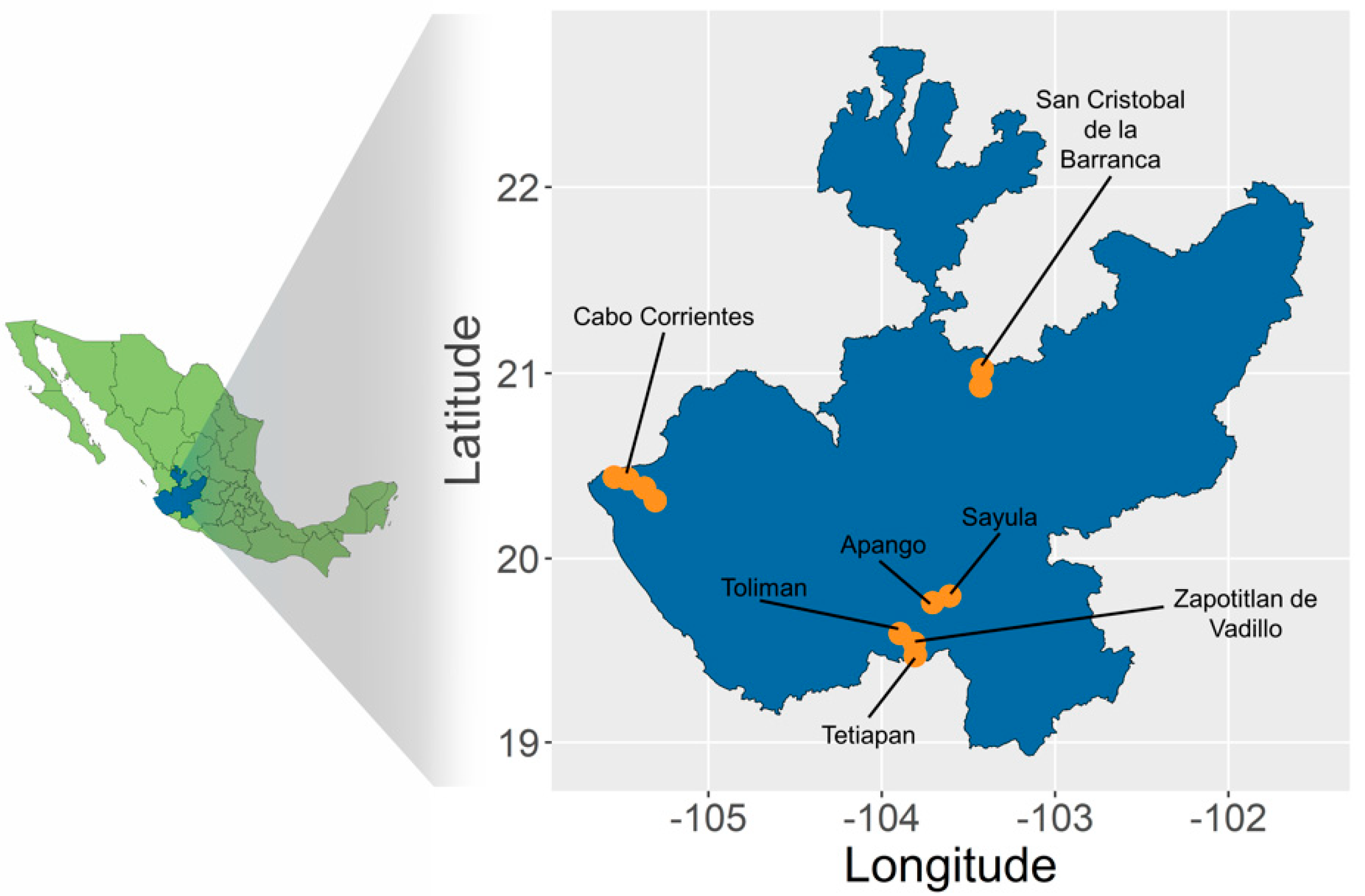
2.1. Sampling and SNP Calling
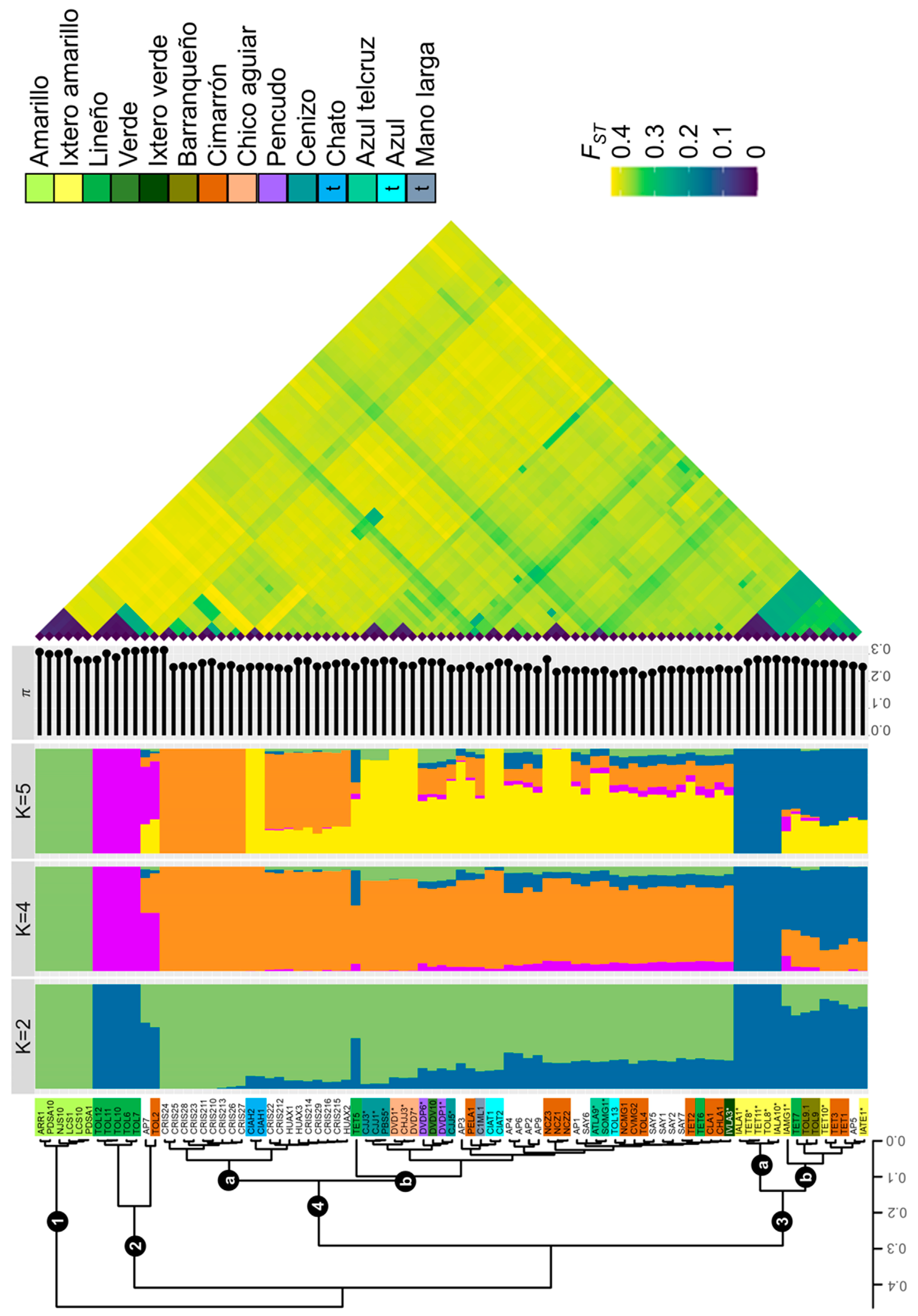
2.2. Genetic Differentiation and Population Structure
| Municipality or Locality | Species | Landrace | Code | Voucher | N |
|---|---|---|---|---|---|
| Cabo Corrientes | A. angustifolia | Amarillo | LCS1, LCS10 | MPDM-218 | 2 |
| Cabo Corrientes | A. angustifolia | Amarillo | PDSA1, PSDA10 | MPDM-243 | 2 |
| Cabo Corrientes | A. angustifolia | Amarillo | NJS10 | - | 1 |
| Cabo Corrientes | A. angustifolia | Amarillo | ARR1 | MPDM-215 | 1 |
| Tolimán | A. angustifolia | Ixtero Amarillo Barranqueño | TOL8 | - | 1 |
| Tequila | A. tequilana | Azul | CIAT1-2 | - | 2 |
| Tolimán | A. tequilana | Azul | TOL13 | - | 1 |
| Tolimán | A. rhodacantha | Azul Telcruz | SOMG1 | - | 1 |
| Zapotitlán de Vadillo | A. rhodacantha | Azul Telcruz | ATLA9 | MPDM-273 | 1 |
| Cabo Corrientes | A. rhodacantha | Cenizo | CJJ1, CJJ3, CJJ5 | MPDM-219 | 3 |
| Cabo Corrientes | A. rhodacantha | Cenizo | PBS5 | MDPM-223 | 1 |
| Tequila | A. tequilana | Chato | CIAH1-2 | - | 2 |
| Cabo Corrientes | A. rhodacantha | Chico Aguiar | CHJJ3 | PCR-9688 | 1 |
| Cabo Corrientes | A. rhodacantha | Chico Aguiar | DVD1, DVD7 | MPDM-235 | 2 |
| Zapotitlán de Vadillo (Tetiapán) | A. angustifolia | Cimarrón | TET1-3 | - | 3 |
| Tolimán | A. angustifolia | Cimarrón | TOL1-4 | - | 4 |
| Tolimán | A. angustifolia | Cimarrón Verde | CVMG | - | 1 |
| Zapotitlán de Vadillo (Tetiapán) | A. rhotacantha | Ixtero Amarillo | TET8-11 | - | 4 |
| Tolimán | A. rhodacantha | Ixtero Amarillo | IAMG1 | - | 1 |
| Zapotitlán de Vadillo | A. rhodacantha | Ixtero Amarillo | IALA1, IALA10 | MPDM-271 | 2 |
| Zapotitlán de Vadillo | A. rhodacantha | Ixtero Amarillo | IATE1 | - | 1 |
| Zapotitlán de Vadillo | A. aff. rhodacantha | Ixtero Verde | IVLA3 | MPDM-269 | 1 |
| Zapotitlán de Vadillo (Tetiapán) | A. angustifolia | Lineño | TET4-7 | - | 4 |
| Tolimán | A. angustifolia | Lineño | TOL5-6 | DCT-23, DCT-25, DCT-26 | 3 |
| Tolimán | A. angustifolia | Lineño Ixtero | TOL7 | - | 1 |
| Tolimán | A. angustifolia | Lineño silvestre | TOL9-12 | - | 4 |
| Tolimán | A. angustifolia | Negro Cimarrón | NCMG1 | DCT-24 | 1 |
| Zapotitlán de Vadillo | A. rhodacantha | Negro Cimarrón | NCZ1-3 | MPDM-276 | 3 |
| Cabo Corrientes | A. rhodacantha | Pencudo | DVDP6, DVDP1 | MPDM-238 | 2 |
| Cabo Corrientes | A. aff. rhodacantha | Verde | DVDV10 | MPDM-237 | 1 |
| San Gabriel (Apango) | A. angustifolia | wild | AP1-9 | DCT-21, DCT-22 | 9 |
| Milpillas (entronque Huaxtla) | A. angustifolia | wild | HUAX1-3 | LMCC-150 | 3 |
| San Cristobal de la Barranca | A. angustifolia | wild | CRIS2_1-16 | LMCC-151 | 16 |
| Sayula | A. angustifolia | wild | SAY1-7 | DCT-20 | 7 |
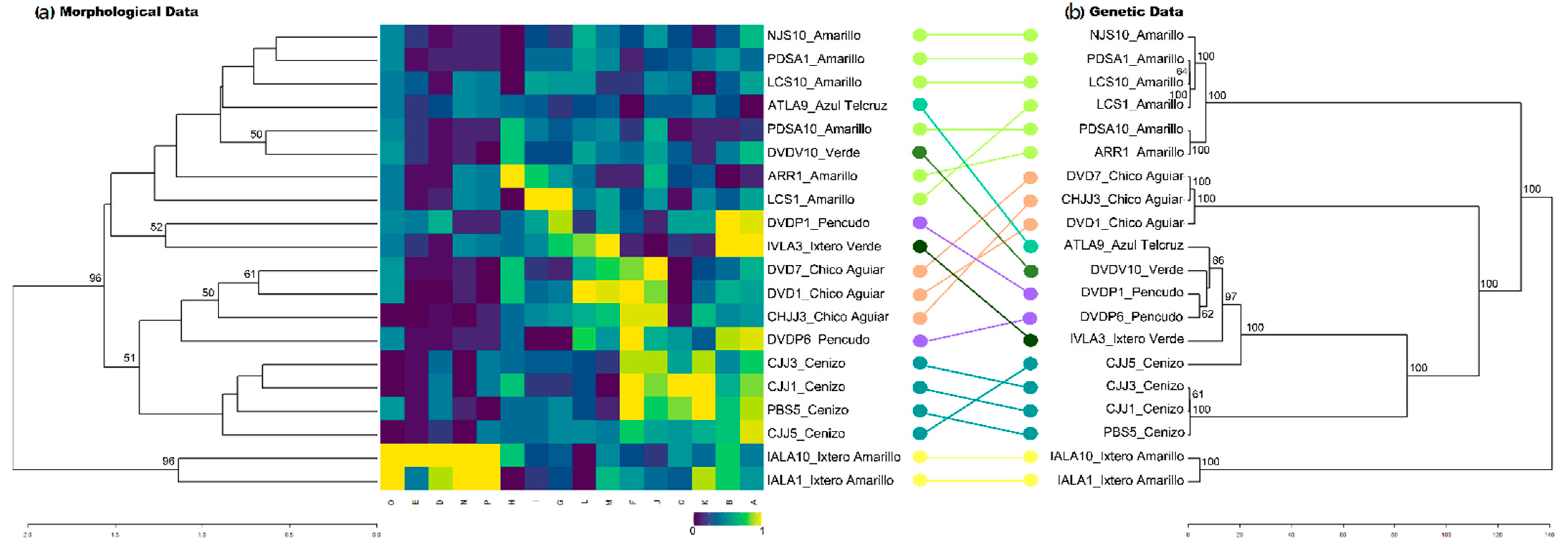
2.3. Morphological Differentiation
3. Discussion
3.1. Genetic Differentiation within the A. angustifolia Complex in Jalisco
3.2. A. tequilana Genetic Background and Domestication
3.3. Traditional Landraces and Their Morphological Identity
3.4. Genetic Differentiation of Landraces
4. Conclusions
5. Materials and Methods
5.1. Sample Collection
5.2. Morphological Data and Analyses
5.3. DNA Sequencing and Bioinformatic Analyses
5.4. Population Genetics and Statistical Analyses
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Callen, E.O. Food Habits of Some Pre-Columbian Mexican Indians. Econ. Bot. 1965, 19, 335–343. [Google Scholar] [CrossRef]
- Gentry, H.S. Agaves of Continental North America; University of Arizona Press: Tucson, AZ, USA, 1982. [Google Scholar]
- Walton, M.K. The evolution and localization of mezcal and tequila in Mexico. Geográfica 1977, 85, 113–132. [Google Scholar]
- Valenzuela-Zapata, A.G. El Agave Tequilero, Su Cultivo e Industria, 2nd ed.; Monsato: Guadalajara, México, 1997. [Google Scholar]
- Luna-Zamora, R. La Historia del Tequila, de Sus Regiones y Sus Hombres, 2nd ed.; CONACULTA: México City, México, 1999.
- Bruman, H.J. Aboriginal Drink Areas of New Spain. Ph.D. Thesis, University of California, Berkley, CA, USA, 1940. [Google Scholar]
- Bruman, H.J. Alcohol in Ancient Mexico; University of Utah Press: Salt Lake, UT, USA, 2000. [Google Scholar]
- Colunga-GarcíaMarin, P.; Zizumbo-Villarreal, D. El tequila y otros mezcales del centro-occidente de México: Domesticación, diversidad y conservación de germoplasma. In En lo Ancestral Hay Futuro: Del Tequila, los Mezcales y Otros Agaves; Centro de Investigación Científica de Yucatán: Mérida, México, 2007; pp. 113–131. [Google Scholar]
- SEGOB. Declaración de Protección a la Denominación de Origen Tequila; Diario Oficial de la Federación: Ciudad de México, México, 1999.
- CRT. Consejo Regulador del Tequila. Inf. Estadístico 1995, 1999, 2022. Available online: https://www.crt.org.mx/EstadisticasCRTweb/ (accessed on 16 June 2022).
- Tetreault, D.; McCulligh, C.; Lucio, C. Distilling Agro-Extractivism: Agave and Tequila Production in Mexico. J. Agrar. Chang. 2021, 21, 219–241. [Google Scholar] [CrossRef]
- Hostettler, S. Land Use Changes and Transnational Migration; EPFL: Lausanne, Switzerland, 2007. [Google Scholar]
- Bowen, S.; Zapata, A.V. Geographical Indications, Terroir, and Socioeconomic and Ecological Sustainability: The Case of Tequila. J. Rural Stud. 2009, 25, 108–119. [Google Scholar] [CrossRef]
- Grzybowska, N.S.; Gerritsen, P. Construyendo Poderes Locales: Microdestilerias y Agave Azul en el sur de Jalisco; Editorial Universitaria; Centro Universitario de la Costa Sur–Universidad de Guadalajara: Autlán de Navarro, México, 2013. [Google Scholar]
- Zizumbo-Villarreal, D.; Vargas-Ponce, O.; Rosales-Adame, J.J.; Colunga-GarcíaMarín, P. Sustainability of the Traditional Management of Agave Genetic Resources in the Elaboration of Mezcal and Tequila Spirits in Western Mexico. Genet. Resour. Crop Evol. 2013, 60, 33–47. [Google Scholar] [CrossRef]
- Gerritsen, P.R.W.; Martínez-Rivera, L.M. Agave Azul, Sociedad y Medio Ambiente: Una Perspectiva de La Costa Sur de Jalisco; Universidad de Guadalajara, Centro Universitario de la Costa Sur: Autlán de Navarro, México, 2010; ISBN 607-450-213-7. [Google Scholar]
- Gil-Vega, K.; González Chavira, M.; Martínez de la Vega, O.; Simpson, J.; Vandemark, G. Analysis of Genetic Diversity in Agave Tequilana Var. Azul Using RAPD Markers. Euphytica 2001, 119, 335–341. [Google Scholar] [CrossRef]
- Vargas-Ponce, O.; Zizumbo-Villarreal, D.; Martínez-Castillo, J.; Coello-Coello, J.; Colunga-GarcíaMarín, P. Diversity and Structure of Landraces of Agave Grown for Spirits under Traditional Agriculture: A Comparison with Wild Populations of A. Angustifolia (Agavaceae) and Commercial Plantations of A. tequilana. Am. J. Bot. 2009, 96, 448–457. [Google Scholar] [CrossRef]
- Ibarrola-Rivas, M.J. Sustainability Analyses of Agave Production in Mexico. Master’s Thesis, University of Groningen, Groningen, The Netherlands, 2010; p. 53. [Google Scholar]
- Torres-García, I.; Rendón-Sandoval, F.J.; Blancas, J.; Moreno-Calles, A.I. The Genus Agave in Agroforestry Systems of Mexico. Bot. Sci. 2019, 97, 263–290. [Google Scholar] [CrossRef]
- Eguiarte, L.E.; Jiménez Barrón, O.A.; Aguirre-Planter, E.; Scheinvar, E.; Gamez, N.; Gasca-Pineda, J.; Castellanos-Morales, G.; Moreno-Letelier, A.; Souza, V. Evolutionary Ecology of Agave: Distribution Patterns, Phylogeny, and Coevolution (an Homage to Howard S. Gentry). Am. J. Bot. 2021, 108, 216–235. [Google Scholar] [CrossRef]
- Colunga-GarcíaMarín, P.; Zizumbo-Villarreal, D.; Martínez-Torres, J. Tradiciones en el aprovechamiento de los agaves mexicanos: Una aportacion a la proteccion legal y conservación de su diversidad biológica y cultural. In En lo Ancestral Hay Futuro: Del Tequila, los Mezcales y Otros Agaves; Centro de Investigaciones Científicas de Yucatán: Mérida, México, 2007; p. 248. [Google Scholar]
- Torres, I.; Casas, A.; Vega, E.; Martínez-Ramos, M.; Delgado-Lemus, A. Population Dynamics and Sustainable Management of Mescal Agaves in Central Mexico: Agave potatorum in the Tehuacán-Cuicatlán Valley. Econ. Bot. 2015, 69, 26–41. [Google Scholar]
- Vargas-Ponce, O.; Zizumbo-Villarreal, D.; Colunga-García Marin, P. In Situ Diversity and Maintenance of Traditional Agave Landraces Used in Spirits Production in West-Central Mexico. Econ. Bot. 2007, 61, 362–375. [Google Scholar] [CrossRef]
- Rivera-Lugo, M.; García-Mendoza, A.; Simpson, J.; Solano, E.; Gil-Vega, K. Taxonomic Implications of the Morphological and Genetic Variation of Cultivated and Domesticated Populations of the Agave angustifolia Complex (Agavoideae, Asparagaceae) in Oaxaca, Mexico. Plant Syst. Evol. 2018, 304, 969–979. [Google Scholar] [CrossRef]
- Trejo, L.; Limones, V.; Peña, G.; Scheinvar, E.; Vargas-Ponce, O.; Zizumbo-Villarreal, D.; Colunga-GarcíaMarín, P. Genetic Variation and Relationships among Agaves Related to the Production of Tequila and Mezcal in Jalisco. Ind. Crops Prod. 2018, 125, 140–149. [Google Scholar] [CrossRef]
- El Mezcal Va Por los Mercados Internacionales. Líderes Mexicanos, October 20, 2021. Available online: https://lideresmexicanos.com/tendencias/el-mezcal-va-por-los-mercados-internacionales/ (accessed on 23 June 2022).
- Gobierno del Estado de Oaxaca. Estados Unidos, principal comprador de mezcal en el mundo. In Coordinación General de Comunicación Social y Vocería del Estado; Gobierno del Estado de Oaxaca, Oaxaca, México. LA, California, 27 de Junio de 2019. Available online: https://www.oaxaca.gob.mx/comunicacion/estados-unidos-principal-comprador-de-mezcal-en-el-mundo/ (accessed on 23 June 2022).
- Elshire, R.J.; Glaubitz, J.C.; Sun, Q.; Poland, J.A.; Kawamoto, K.; Buckler, E.S.; Mitchell, S.E. A Robust, Simple Genotyping-by-Sequencing (GBS) Approach for High Diversity Species. PLoS ONE 2011, 6, e19379. [Google Scholar] [CrossRef]
- Palomino, G.; Dolezel, J.; Mendez, I.; Rubluo, A. Nuclear Genome Size Analysis of Agave tequilana Weber. Caryologia 2003, 56, 37–46. [Google Scholar] [CrossRef]
- Klimova, A.; Ruiz Mondragón, K.Y.; Molina Freaner, F.; Aguirre-Planter, E.; Eguiarte, L.E. Genomic Analyses of Wild and Cultivated Bacanora Agave (Agave angustifolia var. pacifica) Reveal Inbreeding, Few Signs of Cultivation History and Shallow Population Structure. Plants 2022, 11, 1426. [Google Scholar] [CrossRef]
- Gil-Vega, K.; Díaz-Quezada, C.E.; Nava-Cedillo, A.; García-Mendoza, A.; Simpson, J. Análisis AFLP del género Agave refleja la clasificación taxonómica basada en caracteres morfológicos y otros métodos moleculares. In En lo Ancestral Hay Futuro: Del Tequila, los Mezcales y Otros Agaves; Centro de Investigaciones Científicas de Yucatán: Mérida, México, 2007; pp. 23–39. [Google Scholar]
- Zietkiewicz, E.; Rafalski, A.; Labuda, D. Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 1994, 20, 176–183. [Google Scholar] [CrossRef]
- Abraham-Juárez, M.J.; Ramírez-Malagón, R.; del C. Gil-Vega, K.; Simpson, J. AFLP analysis of genetic variability in three reproductive forms of Agave tequilana. Rev. Fitotec. Mex. 2009, 32, 171–175. [Google Scholar] [CrossRef]
- Barrientos Rivera, G.; Esparza Ibarra, E.L.; Segura Pacheco, H.R.; Talavera Mendoza, Ó.; Sampedro Rosas, M.L.; Hernández Castro, E. Morphological characterization of Agave angustifolia and its conservation in Guerrero, Mexico. Rev. Mex. Cienc. Agrícolas 2019, 10, 655–668. [Google Scholar] [CrossRef]
- Deschamps, S.; Llaca, V.; May, G.D. Genotyping-by-Sequencing in Plants. Biology 2012, 1, 460–483. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Galván, G. Domesticación de Agaves Productores de Fibra en el Centro-Occidente de México: Una Aproximación Etnobotánica y Morfológica. Master’s Thesis, Centro de Investigaciones Científicas de Yucatán (CICY), Mérida, México, 2011. [Google Scholar]
- Bischl, B.; Lang, M.; Bossek, J.; Horn, D.; Richter, J.; Surmann, D. BBmisc: Miscellaneous Helper Functions for B. Bischl; Version 1.12; R Package; R Foundation: Vienna, Austria, 2017. [Google Scholar]
- Hvitfeldt, E. Paletteer: Comprehensive Collection of Color Palettes. Available online: https://cran.r-project.org/web/packages/paletteer/index.html (accessed on 10 May 2022).
- Galili, T. Dendextend: An R Package for Visualizing, Adjusting and Comparing Trees of Hierarchical Clustering. Bioinformatics 2015, 31, 3718–3720. [Google Scholar] [CrossRef] [PubMed]
- Paradis, E.; Schliep, K. Ape 5.0: An Environment for Modern Phylogenetics and Evolutionary Analyses in R. Bioinformatics 2019, 35, 526–528. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’hara, R.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H. Package ‘Vegan’; Version 2.5.7; Community Ecology Package; R Foundation: Vienna, Austria, 2013; Volume 2, pp. 1–295. [Google Scholar]
- Knaus, B.J.; Grünwald, N.J. Vcfr: A Package to Manipulate and Visualize Variant Call Format Data in R. Mol. Ecol. Resour. 2017, 17, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Obenchain, V.; Lawrence, M.; Carey, V.; Gogarten, S.; Shannon, P.; Morgan, M. VariantAnnotation: A Bioconductor Package for Exploration and Annotation of Genetic Variants. Bioinformatics 2014, 30, 2076–2078. [Google Scholar] [CrossRef]
- Jombart, T.; Ahmed, I. Adegenet 1.3-1: New Tools for the Analysis of Genome-Wide SNP Data. Bioinformatics 2011, 27, 3070–3071. [Google Scholar] [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data; ScienceOpen, Inc.: Burlington, MA, USA, 2010. [Google Scholar]
- Ewels, P.; Magnusson, M.; Lundin, S.; Käller, M. MultiQC: Summarize Analysis Results for Multiple Tools and Samples in a Single Report. Bioinformatics 2016, 32, 3047–3048. [Google Scholar]
- Li, H.; Durbin, R. Fast and Accurate Short Read Alignment with Burrows–Wheeler Transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef]
- Danecek, P.; Bonfield, J.K.; Liddle, J.; Marshall, J.; Ohan, V.; Pollard, M.O.; Whitwham, A.; Keane, T.; McCarthy, S.A.; Davies, R.M. Twelve Years of SAMtools and BCFtools. Gigascience 2021, 10, giab008. [Google Scholar] [CrossRef]
- Rochette, N.C.; Rivera-Colón, A.G.; Catchen, J.M. Stacks 2: Analytical Methods for Paired-end Sequencing Improve RADseq-based Population Genomics. Mol. Ecol. 2019, 28, 4737–4754. [Google Scholar]
- Chang, C.C.; Chow, C.C.; Tellier, L.C.; Vattikuti, S.; Purcell, S.M.; Lee, J.J. Second-Generation PLINK: Rising to the Challenge of Larger and Richer Datasets. Gigascience 2015, 4, s13742-015. [Google Scholar] [CrossRef] [PubMed]
- Alexander, D.H.; Novembre, J.; Lange, K. Fast Model-Based Estimation of Ancestry in Unrelated Individuals. Genome Res. 2009, 19, 1655–1664. [Google Scholar] [CrossRef] [PubMed]
- Kopelman, N.M.; Mayzel, J.; Jakobsson, M.; Rosenberg, N.A.; Mayrose, I. Clumpak: A Program for Identifying Clustering Modes and Packaging Population Structure Inferences across K. Mol. Ecol. Resour. 2015, 15, 1179–1191. [Google Scholar] [CrossRef] [PubMed]
- Goudet, J.; Jombart, T. Package “hierfstat”, Version 1.5-11; Estimation and Tests of Hierarchical F-Statistics. Available online: https://cran.r-project.org/web/packages/hierfstat/index.html (accessed on 20 April 2022).
- Jombart, T. Adegenet: A R package for the multivariate analysis of genetic markers. Bioinformatics 2008, 24, 1403–1405. [Google Scholar] [CrossRef] [PubMed]
- Pfeifer, B.; Wittelsbürer, U.; Ramos-Onsins, S.E.; Lercher, M.J. PopGenome: An Efficient Swiss Army Knife for Population Genomic Analysis in R. Mol. Biol. Evol. 2014, 31, 1929–1936. [Google Scholar] [CrossRef] [Green Version]

| Cluster | n | Ho | He | Fis | pi |
|---|---|---|---|---|---|
| 1 | 6 | 0.2884 | 0.1546 | −0.8011 | 102.7727 |
| 2 | 7 | 0.3017 | 0.2155 | −0.2893 | 125.0220 |
| 3 | 14 | 0.3144 | 0.2667 | −0.1198 | 151.5476 |
| 4 | 60 | 0.2850 | 0.3337 | 0.1543 | 182.6654 |
| 3a | 5 | 0.3204 | 0.1708 | −0.8420 | 113.8000 |
| 3b | 9 | 0.3109 | 0.2946 | −0.0520 | 165.0458 |
| 4a | 20 | 0.2711 | 0.3157 | 0.1326 | 174.0885 |
| 4b | 40 | 0.2925 | 0.3300 | 0.1222 | 180.4139 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabrera-Toledo, D.; Mendoza-Galindo, E.; Larranaga, N.; Herrera-Estrella, A.; Vásquez-Cruz, M.; Hernández-Hernández, T. Genomic and Morphological Differentiation of Spirit Producing Agave angustifolia Traditional Landraces Cultivated in Jalisco, Mexico. Plants 2022, 11, 2274. https://doi.org/10.3390/plants11172274
Cabrera-Toledo D, Mendoza-Galindo E, Larranaga N, Herrera-Estrella A, Vásquez-Cruz M, Hernández-Hernández T. Genomic and Morphological Differentiation of Spirit Producing Agave angustifolia Traditional Landraces Cultivated in Jalisco, Mexico. Plants. 2022; 11(17):2274. https://doi.org/10.3390/plants11172274
Chicago/Turabian StyleCabrera-Toledo, Dánae, Eddy Mendoza-Galindo, Nerea Larranaga, Alfredo Herrera-Estrella, Marilyn Vásquez-Cruz, and Tania Hernández-Hernández. 2022. "Genomic and Morphological Differentiation of Spirit Producing Agave angustifolia Traditional Landraces Cultivated in Jalisco, Mexico" Plants 11, no. 17: 2274. https://doi.org/10.3390/plants11172274
APA StyleCabrera-Toledo, D., Mendoza-Galindo, E., Larranaga, N., Herrera-Estrella, A., Vásquez-Cruz, M., & Hernández-Hernández, T. (2022). Genomic and Morphological Differentiation of Spirit Producing Agave angustifolia Traditional Landraces Cultivated in Jalisco, Mexico. Plants, 11(17), 2274. https://doi.org/10.3390/plants11172274









