Antigiardial Activity of Foeniculum vulgare Hexane Extract and Some of Its Constituents
Abstract
:1. Introduction
2. Results
3. Discussions
4. Materials and Methods
4.1. Plant Material and Extraction
4.2. Chemicals
4.3. Parasite Culture
4.4. Cytotoxic Activity on Vero Cell Line
4.5. Antigiardial Activity
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leung, A.K.C.; Leung, A.A.M.; Wong, A.H.C.; Sergi, C.M.; Kam, J.K.M. Giardiasis: An overview. Recent Pat. Inflamm. Allergy Drug Discov. 2019, 13, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Ryan, U.; Hijjawi, N.; Feng, Y.; Xiao, L. Giardia: An under-reported foodborne parasite. Int. J. Parasitol. 2019, 49, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Argüello-García, R.; Leitsch, D.; Skinner-Adams, T.; Ortega-Pierres, M.G. Drug resistance in Giardia: Mechanisms and alternative treatments for Giardiasis. Adv. Parasitol. 2020, 107, 201–282. [Google Scholar]
- CCernikova, L.; Faso, C.; Hehl, A.B. Five facts about Giardia lamblia. PLoS Pathog. 2018, 14, e1007250. [Google Scholar] [CrossRef]
- Ryan, U.; Cacciò, S.M. Zoonotic potential of Giardia. Int. J. Parasitol. 2013, 43, 943–956. [Google Scholar] [CrossRef] [PubMed]
- Mørch, K.; Hanevik, K. Giardiasis treatment: An update with a focus on refractory disease. Curr. Opin. Infect. Dis. 2020, 33, 355–364. [Google Scholar] [CrossRef]
- Tejman-Yarden, N.; Eckmann, L. New approaches to the treatment of giardiasis. Curr. Opin. Infect. Dis. 2011, 24, 451–456. [Google Scholar] [CrossRef]
- Leitsch, D. Drug Resistance in the Microaerophilic Parasite Giardia lamblia. Curr. Trop. Med. Rep. 2015, 2, 128–135. [Google Scholar] [CrossRef]
- Nash, T.E. Treatment of Giardia lamblia Infections. Pediatr. Infect. Dis. J. 2001, 20, 193–195. [Google Scholar] [CrossRef]
- Hernández Ceruelos, A.; Romero-Quezada, L.C.; Ruvalcaba Ledezma, J.C.; López Contreras, L. Therapeutic uses of metronidazole and its side effects: An update. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 397–401. [Google Scholar]
- He, W.; Huang, B. A review of chemistry and bioactivities of a medicinal spice: Foeniculum vulgare. J. Med. Plants Res. 2011, 5, 3595–3600. [Google Scholar]
- Argueta Villamar, A.; Cano Asseleih, L.M. Atlas de las Plantas de la Medicina Tradicional Mexicana, 1st ed.; Instituto Nacional Indigenista: Mexico City, Mexico, 1994; 650p. [Google Scholar]
- Esquivel-Ferriño, P.C.; Favela-Hernández, J.M.J.; Garza-González, E.; Waksman, N.; Ríos, M.Y.; Del Rayo Camacho-Corona, M. Antimycobacterial activity of constituents from Foeniculum vulgare var. Dulce Grown in Mexico. Molecules 2012, 17, 8471–8482. [Google Scholar] [CrossRef] [PubMed]
- Ikeura, H.; Kohara, K.; Li, X.-X.; Kobayashi, F.; Hayata, Y. Identification of (E,E)-2,4-Undecadienal from Coriander (Coriandrum sativum L.) as a Highly Effective Deodorant Compound against the Offensive Odor of Porcine Large Intestine. J. Agric. Food Chem. 2010, 58, 11014–11017. [Google Scholar] [CrossRef] [PubMed]
- Leitsch, D.; Kolarich, D.; Wilson, I.B.H.; Altmann, F.; Duchêne, M. Nitroimidazole action in Entamoeba histolytica: A central role for thioredoxin reductase. PLoS Biol. 2007, 5, 1820–1834. [Google Scholar] [CrossRef] [PubMed]
- Leitsch, D.; Kolarich, D.; Binder, M.; Stadlmann, J.; Altmann, F.; Duchêne, M. Trichomonas vaginalis: Metronidazole and other nitroimidazole drugs are reduced by the flavin enzyme thioredoxin reductase and disrupt the cellular redox system. Implications for nitroimidazole toxicity and resistance. Mol. Microbiol. 2009, 72, 518–536. [Google Scholar] [CrossRef]
- Williams, C.F.; Lloyd, D.; Kolarich, D.; Alagesan, K.; Duchêne, M.; Cable, J.; Williams, D.; Leitsch, D. Disrupted intracellular redox balance of the diplomonad fish parasite Spironucleus vortens by 5-nitroimidazoles and garlic-derived compounds. Vet. Parasitol. 2012, 190, 62–73. [Google Scholar] [CrossRef] [PubMed]
- Bisignano, G.; Laganà, M.G.; Trombetta, D.; Arena, S.; Nostro, A.; Uccella, N.; Mazzanti, G.; Saija, A. In vitro antibacterial activity of some aliphatic aldehydes from Olea europaea L. FEMS Microbiol. Lett. 2001, 198, 9–13. [Google Scholar] [CrossRef]
- Carvalho, V.M.; Di Mascio, P.; de Arruda Campos, I.P.; Douki, T.; Cadet, J.; Medeiros, M.H.G. Formation of 1,N6-Etheno-2‘-deoxyadenosine Adducts by trans, trans-2,4-Decadienal. Chem. Res. Toxicol. 1998, 11, 1042–1047. [Google Scholar] [CrossRef]
- Carvalho, V.M.; Asahara, F.; Di Mascio, P.; De Arruda Campos, I.P.; Cadet, J.; Medeiros, M.H.G. Novel 1,N6-etheno-2′-deoxyadenosine adducts from lipid peroxidation products. Chem. Res. Toxicol. 2000, 13, 397–405. [Google Scholar] [CrossRef]
- Sigolo, C.A.O.; Di Mascio, P.; Medeiros, M.H.G. Covalent modification of cytochrome c exposed to trans,trans-2,4-decadienal. Chem. Res. Toxicol. 2007, 20, 1099–1110. [Google Scholar] [CrossRef]
- Brandelli, C.L.C.; Giordani, R.B.; De Carli, G.A.; Tasca, T. Indigenous traditional medicine: In vitro anti-giardial activity of plants used in the treatment of diarrhea. Parasitol. Res. 2009, 104, 1345–1349. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, K.A.L.; De Melo, N.I.; Aguiar, D.P.; De Oliveira, P.F.; Groppo, M.; Da Silva Filho, A.A.; Rodrigues, V.; Cunha, W.R.; Tavares, D.C.; Magalhães, L.G.; et al. Anthelmintic Effects of the Essential Oil of Fennel (Foeniculum vulgare Mill., Apiaceae) against Schistosoma mansoni. Chem. Biodivers. 2015, 12, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Méabed, E.M.; El Sayed, N.M.; Abou-Sreea, A.I.; Roby, M.H. Chemical analysis of aqueous extracts of Origanum majorana and Foeniculum vulgare and their efficacy on Blastocystis spp. cysts. Phytomedicine 2018, 43, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Karami, F.; Dastan, D.; Fallah, M.; Matini, M. In vitro activity of foeniculum vulgare and its main essential oil component trans-anethole on trichomonas vaginalis. Iran. J. Parasitol. 2019, 14, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Keister, D.B. Axenic culture of Giardia lamblia in TYI-S-33 medium supplemented with bile. Trans. R. Soc. Trop. Med. Hyg. 1983, 77, 487–488. [Google Scholar] [CrossRef]
- Uc-Cachón, A.H.; Borges-Argáez, R.; Said-Fernández, S.; Vargas-Villarreal, J.; González-Salazar, F.; Méndez-González, M.; Cáceres-Farfán, M.; Molina-Salinas, G.M. Naphthoquinones isolated from Diospyros anisandra exhibit potent activity against pan-resistant first-line drugs Mycobacterium tuberculosis strains. Pulm. Pharmacol. Ther. 2014, 27, 114–120. [Google Scholar] [CrossRef]
- Adeyemi, O.S.; Atolani, O.; Awakan, O.J.; Olaolu, T.D.; Nwonuma, C.O.; Alejolowo, O.; Otohinoyi, D.A.; Rotimi, D.; Owolabi, A.; Batiha, G.E.-S. In vitro screening to identify anti-toxoplasma compounds and in silico modeling for bioactivities and toxicity. Yale J. Biol. Med. 2019, 92, 369–383. [Google Scholar]
- Mata-Cárdenas, B.D.; Vargas-Villarreal, J.; González-Salazar, F.; Palacios-Corona, R.; Said-Fernández, S. A new vial microassay to screen antiprotozoal drugs. Pharmacologyonline 2008, 1, 529–537. [Google Scholar]
| Extract/Compound | % Inhibition at 300 μg/mL |
|---|---|
| Hexane extract | 94 |
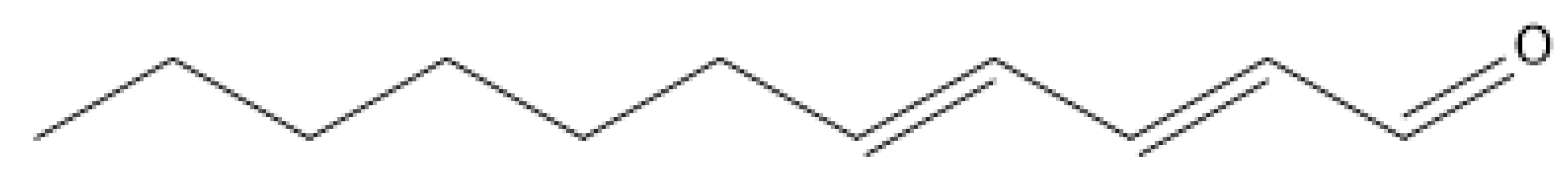
| Trans, trans-2,4-undecadienal c | 97 |
| p-Anisaldehyde b | 67 |
| (R) (−)-Carvone a | 66 |
| (1R,4S) (+)-Camphene a | 61 |
| 1,3-Benzenediol b | 56 |
| Estragole b | 49 |
| Fenchylacetate a | 38.5 |
| (R)- (+)-β-Citronellol a | 38 |
| o-Cymene b | 30 |
| Terpinolene a | 30 |
| Undecanal c | 25 |
| γ-Terpinene a | 24 |
| Oleic acid d | 24 |
| Methylchavicol b | 20 |
| Trans-anethol b | 17 |
| (S) (+)-Fenchone a | 16 |
| (−)-α-Tujone a | 14 |
| (R) (−)-Fenchone a | 6.5 |
| Pinacol e | 2 |
| Metronidazol | 100 |
| Extract/Compound/Drug | G. duodenalis IC50 (μg/mL) (IC95%) | Vero Cells IC50 (μg/mL) (IC95%) | Selectivity Index (SI) |
|---|---|---|---|
| Hexane extract | 89.3 (66.6–116.7) | nd | nd |
| (1R,4S) (+)-Camphene | 181.1 (158.1–207.9) | 494.3 (492.1–496.4) | 2.7 |
| (R) (−)-Carvone | 207.0 (162.2–267.3) | 350.7 (348.4–353.0) | 1.6 |
| Estragole | 150.0 (130.8–175.9) | 258.7 (245–265) | 1.7 |
| p-Anisaldehyde | 196.8 (165.6–234.4) | 540.7 (538.6–542.9) | 2.7 |
| 1,3-Benzenediol | 168.7 (140.5–182.5) | 437.90 (410.7–470) | 2.5 |
| Trans, trans-2,4-undecadienal | 72.1 (57.7–90.1) | 588.8 (586.7–591.0) | 8.1 |
| Metronidazole | 0.5 (0.4–0.6) | 83.5 (81–86.1) | 160.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Domínguez-Vigil, I.G.; Mata-Cárdenas, B.D.; Esquivel-Ferriño, P.C.; Avalos-Alanís, F.G.; Vargas-Villarreal, J.; Camacho-Corona, M.d.R. Antigiardial Activity of Foeniculum vulgare Hexane Extract and Some of Its Constituents. Plants 2022, 11, 2212. https://doi.org/10.3390/plants11172212
Domínguez-Vigil IG, Mata-Cárdenas BD, Esquivel-Ferriño PC, Avalos-Alanís FG, Vargas-Villarreal J, Camacho-Corona MdR. Antigiardial Activity of Foeniculum vulgare Hexane Extract and Some of Its Constituents. Plants. 2022; 11(17):2212. https://doi.org/10.3390/plants11172212
Chicago/Turabian StyleDomínguez-Vigil, Irma G., Benito D. Mata-Cárdenas, Patricia C. Esquivel-Ferriño, Francisco G. Avalos-Alanís, Javier Vargas-Villarreal, and María del Rayo Camacho-Corona. 2022. "Antigiardial Activity of Foeniculum vulgare Hexane Extract and Some of Its Constituents" Plants 11, no. 17: 2212. https://doi.org/10.3390/plants11172212
APA StyleDomínguez-Vigil, I. G., Mata-Cárdenas, B. D., Esquivel-Ferriño, P. C., Avalos-Alanís, F. G., Vargas-Villarreal, J., & Camacho-Corona, M. d. R. (2022). Antigiardial Activity of Foeniculum vulgare Hexane Extract and Some of Its Constituents. Plants, 11(17), 2212. https://doi.org/10.3390/plants11172212






