Developing Oligo Probes for Chromosomes Identification in Hemp (Cannabis sativa L.)
Abstract
:1. Introduction
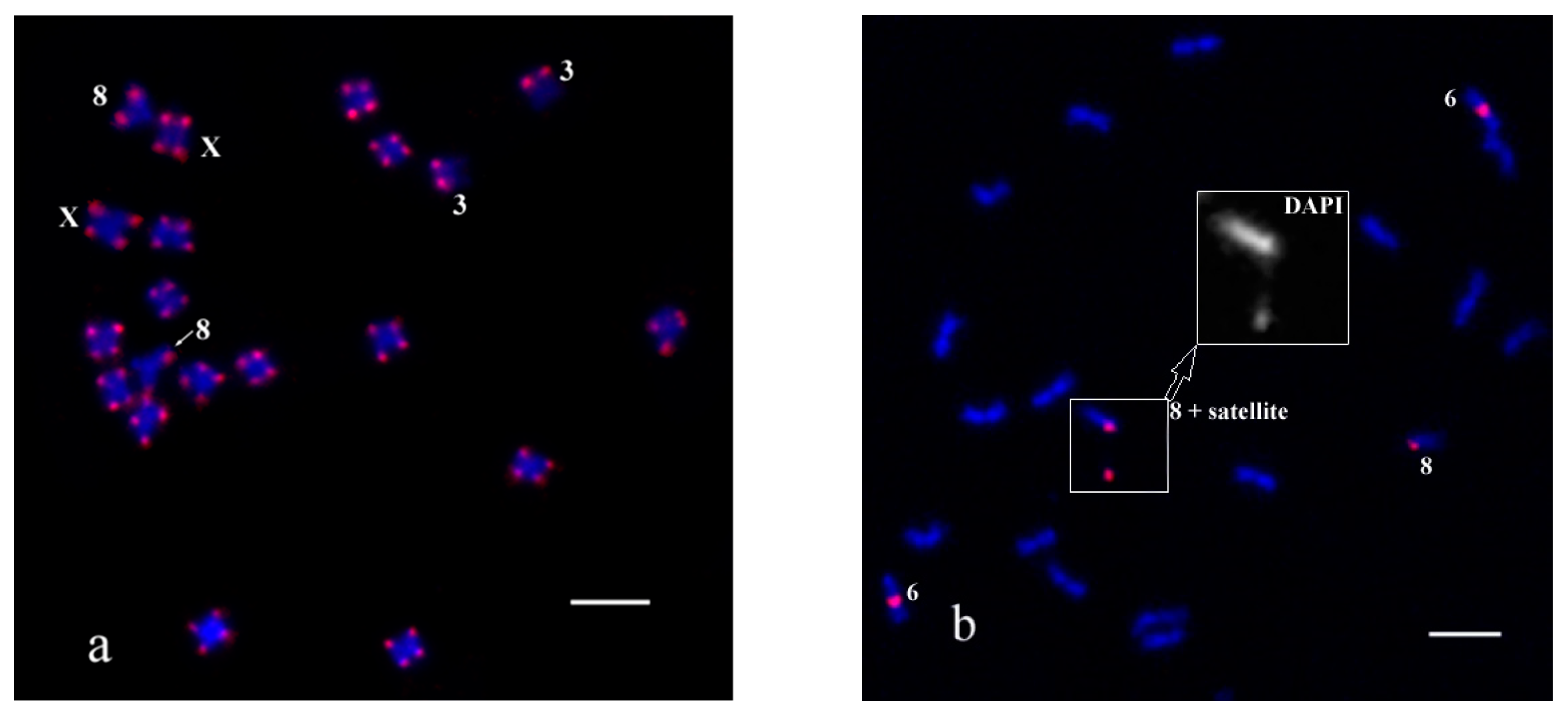
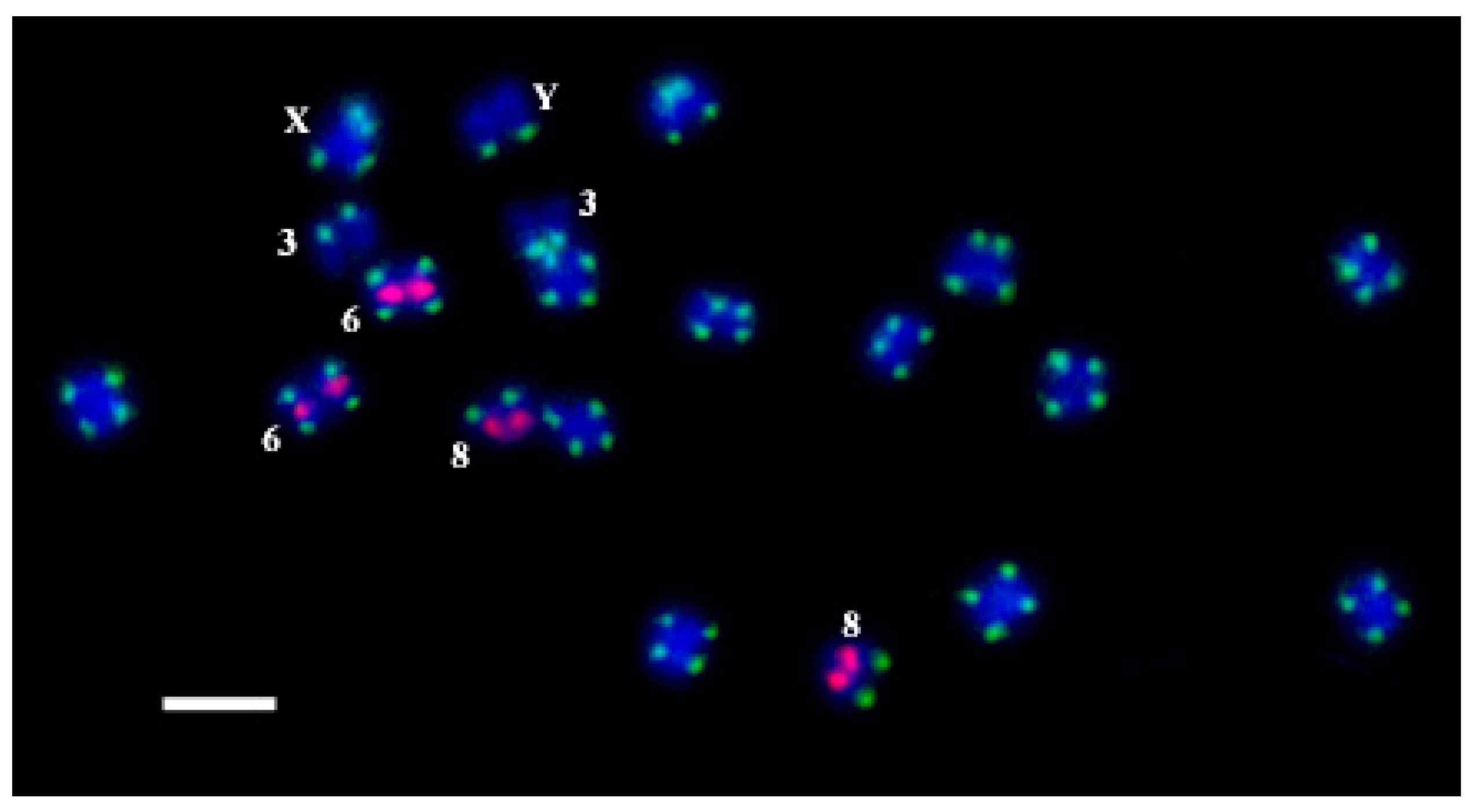
2. Results and Discussion
3. Conclusions
4. Materials and Methods
4.1. Plant Material
4.2. Chromosome Preparation
4.3. DNA Probes and Labeling
4.4. Fluorescence In Situ Hybridization (FISH) and Oligo-FISH
4.5. Microscopy and Image Analysis
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- ElSohly, M.A.; Radwan, M.M.; Gul, W.; Chandra, S.; Galal, A. Phytochemistry of Cannabis sativa L. Prog. Chem. Org. Nat. Prod. 2017, 103, 1–36. [Google Scholar] [CrossRef]
- Cherney, J.H.; Small, E. Industrial Hemp in North America: Production, Politics and Potential. Agronomy 2016, 6, 58. [Google Scholar] [CrossRef] [Green Version]
- Crini, G.; Lichtfouse, E.; Chanet, G.; Morin-Crini, N. Applications of hemp in textiles, paper industry, insulation and building materials, horticulture, animal nutrition, food and beverages, nutraceuticals, cosmetics and hygiene, medicine, agrochemistry, energy production and environment: A review. Environ. Chem. Lett. 2020, 18, 1451–1476. [Google Scholar] [CrossRef]
- Jiang, H.E.; Li, X.; Zhao, Y.X.; Ferguson, D.K.; Hueber, F.; Bera, S.; Wang, Y.F.; Zhao, L.C.; Liu, C.J.; Li, C.S. A new insight into Cannabis sativa (Cannabaceae) utilization from 2500-year-old Yanghai Tombs, Xinjiang, China. J. Ethnopharmacol. 2006, 108, 414. [Google Scholar] [CrossRef]
- Kriese, U.; Schumann, E.; Weber, W.E.; Beyer, M.; Brühl, L.; Matthus, B. Oil content, tocopherol composition and fatty acid patterns of the seeds of 51 C. sativa L. genotypes. Euphytica 2004, 137, 339. [Google Scholar] [CrossRef]
- Werf, H.V.D.; Mathussen, E.W.J.M.; Haverkort, A.J. The potential of hemp (Cannabis sativa L.) for sustainable fibre production: A crop physiological appraisal. Ann. Appl. Biol. 1996, 129, 109–123. [Google Scholar] [CrossRef]
- Abrams, D.I.; Jay, C.A.; Shade, S.B.; Vizoso, H.; Reda, H.; Press, S.; Kelly, M.E.; Rowbotham, M.C.; Petersen, K.L. Cannabis in painful HIV-associated sensory neuropathy: A randomized placebo-controlled trial. Neurology 2007, 68, 515. [Google Scholar] [CrossRef]
- Andre, C.M.; Hausman, J.F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [Green Version]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef]
- Guindon, J.; Hohmann, A.G. The endocannabinoid system and pain. Curr. Drug Targets CNS Neurol. Disord. 2009, 8, 403. [Google Scholar] [CrossRef]
- Merlin, M.D. Archaeological evidence for the tradition of psychoactive plant use in the old world. Econ. Bot. 2003, 57, 295–323. [Google Scholar] [CrossRef]
- Potter, D.J. A review of the cultivation and processing of cannabis (Cannabis sativa L.) for production of prescription medicines in the UK. Drug Test. Anal. 2014, 6, 31–38. [Google Scholar] [CrossRef]
- Russo, E. Hemp for headache: An in-depth historical and scientific review of cannabis in migraine treatment. J. Cannabis Ther. 2001, 1, 21–92. [Google Scholar] [CrossRef]
- Slatkin, N.E. Cannabinoids in the treatment of chemotherapy-induced nausea and vomiting: Beyond prevention of acute emesis. J. Support. Oncol. 2007, 5, 1–9. [Google Scholar]
- Zuardi, A.W. History of Cannabis as a medicine: A review. Braz. J. Psychiat. 2006, 28, 153. [Google Scholar] [CrossRef] [Green Version]
- Renner, S.S. The relative and absolute frequencies of angiosperm sexual systems: Dioecy, monoecy, gynodioecy, and an updated online database. Am. J. Bot. 2014, 101, 1588–1596. [Google Scholar] [CrossRef] [Green Version]
- Muyle, A.; Shearn, R.; Marais, G.A. The evolution of sex chromosomes and dosage compensation in plants. Genome Biol. Evol. 2017, 9, 627–645. [Google Scholar] [CrossRef] [Green Version]
- Fruchard, C.; Marais, G.A. Evolutionary Developmental Biology: A Reference Guide; Nuno de la Rosa, L., Müller, G., Eds.; Springer: Cham, Switzerland, 2021; pp. 683–696. [Google Scholar] [CrossRef] [Green Version]
- Divashuk, M.G.; Alexandrov, O.S.; Kroupin, P.Y.; Karlov, G.I. Molecular cytogenetic mapping of Humulus lupulus sex chromosomes. Cytogenet. Genome Res. 2011, 134, 213–219. [Google Scholar] [CrossRef]
- Grabowska-Joachimiak, A.; Mosiolek, M.; Lech, A.; Góralski, G. C-Banding/DAPI and in situ hybridization reflect karyotype structure and sex chromosome differentiation in Humulus japonicus Siebold & Zucc. Cytogenet. Genome Res. 2011, 132, 203–211. [Google Scholar] [CrossRef]
- Matsunaga, S.; Kawano, S. Sex determination by sex chromosomes in dioecious plants. Plant Biol. 2001, 3, 481–488. [Google Scholar] [CrossRef]
- Ming, R.; Bendahmane, A.; Renner, S.S. Sex chromosomes in land plants. Annu. Rev. Plant Biol. 2011, 62, 485–514. [Google Scholar] [CrossRef] [Green Version]
- Razumova, O.V.; Alexandrov, O.S.; Divashuk, M.G.; Sukhorada, T.I.; Karlov, G.I. Molecular cytogenetic analysis of monoecious hemp (Cannabis sativa L.) cultivars reveals its karyotype variations and sex chromosomes constitution. Protoplasma 2016, 253, 895–901. [Google Scholar] [CrossRef]
- Hesami, M.; Pepe, M.; Alizadeh, M.; Rakei, A.; Baiton, A.; Jones, A.M.P. Recent advances in cannabis biotechnology. Ind. Crops Prod. 2020, 158, 113026. [Google Scholar] [CrossRef]
- Hurgobin, B.; Tamiru-Oli, M.; Welling, M.T.; Doblin, M.S.; Bacic, A.; Whelan, J.; Lewsey, M.G. Recent advances in Cannabis sativa genomics research. New Phytol. 2021, 230, 73–89. [Google Scholar] [CrossRef]
- Prentout, D.; Razumova, O.; Rhoné, B.; Badouin, H.; Henri, H.; Feng, C.; Käfer, J.; Karlov, G.; Marais, G.A. A high-throughput segregation analysis identifies the sex chromosomes of Cannabis sativa. bioRxiv 2019. [Google Scholar] [CrossRef] [Green Version]
- Razumova, O.V.; Divashuk, M.G.; Alexandrov, O.S.; Karlov, G.I. GISH painting of the Y chromosomes suggests advanced phases of sex chromosome evolution in three dioecious Cannabaceae species (Humulus lupulus, H. japonicus, and Cannabis sativa). Protoplasma 2022, 1774, 1–8. [Google Scholar] [CrossRef]
- Faeti, V.; Mandolino, G.; Ranalli, P. Genetic diversity of Cannabis sativa germplasm based on RAPD markers. Plant Breed. 1996, 115, 367–370. [Google Scholar] [CrossRef]
- Forapani, S.; Carboni, A.; Paoletti, C.; Moliterni, V.C.; Ranalli, P.; Mandolino, G. Comparison of hemp varieties using random amplified polymorphic DNA markers. Crop Sci. 2001, 41, 1682–1689. [Google Scholar] [CrossRef] [Green Version]
- Kroupin, P.Y.; Kuznetsova, V.M.; Nikitina, E.A.; Martirosyan, Y.T.; Karlov, G.I.; Divashuk, M.G. Development of new cytogenetic markers for Thinopyrum ponticum (Podp.) Z.-W. Liu & R.-C. Wang. Comp. Cytogenet. 2019, 13, 231–243. [Google Scholar] [CrossRef]
- Alexandrov, O.S.; Romanov, D.V.; Divshuk, M.G.; Razumova, O.V.; Ulyanov, D.S.; Karlov, G.I. Studying and physical mapping of the 45S IGS linked CS-237 DNA repeat in Cannabis sativa L. Plants 2022, 11, 1396. [Google Scholar] [CrossRef]
- Chao, Y.T.; Chen, W.C.; Chen, C.Y.; Ho, H.Y.; Yeh, C.H.; Kuo, Y.T.; Su, C.L.; Yen, S.H.; Hsueh, H.Y.; Yeh, J.H.; et al. Chromosome-level assembly, genetic and physical mapping of Phalaenopsis aphrodite genome provides new insights into species adaptation and resources for orchid breeding. Plant Biotechnol. J. 2018, 16, 2027–2041. [Google Scholar] [CrossRef] [Green Version]
- Bray-Ward, P. FISH Probes and Labelling Techniques. FISH. A Practical Approach; Oxford University Press: Oxford, UK, 2002; pp. 5–28. [Google Scholar]
- Giorgetti, L.; Piolot, T.; Heard, E. High-resolution 3D DNA FISH using plasmid probes and computational correction of optical aberrations to study chromatin structure at the sub-megabase scale. In Nuclear Bodies and Noncoding RNAs; Humana Press: New York, NY, USA, 2015; Volume 1262, pp. 37–53. [Google Scholar] [CrossRef]
- Huber, D.; von Voithenberg, L.V.; Kaigala, G.V. Fluorescence in situ hybridization (FISH): History, limitations and what to expect from micro-scale FISH? Micro Nano Eng. 2018, 1, 15–24. [Google Scholar] [CrossRef]
- Jiang, J. Fluorescence in situ hybridization in plants: Recent developments and future applications. Chromosome Res. 2019, 27, 153–165. [Google Scholar] [CrossRef]
- Kroupin, P.; Kuznetsova, V.; Romanov, D.; Kocheshkova, A.; Karlov, G.; Dang, T.X.; Khuat, T.M.L.; Kirov, I.; Alexandrov, O.; Polkhovskiy, A.; et al. Pipeline for the rapid development of cytogenetic markers using genomic data of related species. Genes 2019, 10, 113. [Google Scholar] [CrossRef] [Green Version]
- Lang, T.; Li, G.; Wang, H.; Yu, Z.; Chen, Q.; Yang, E.; Fu, S.; Tang, Z.; Yang, Z. Physical location of tandem repeats in the wheat genome and application for chromosome identification. Planta 2019, 249, 663–675. [Google Scholar] [CrossRef]
- Tang, Z.; Yang, Z.; Fu, S. Oligonucleotides replacing the roles of repetitive sequences pAs1, pSc119. 2, pTa-535, pTa71, CCs1, and pAWRC. 1 for FISH analysis. J. Appl. Genet. 2014, 55, 313–318. [Google Scholar] [CrossRef]
- Fu, S.; Chen, L.; Wang, Y.; Li, M.; Yang, Z.; Qiu, L.; Yan, B.; Ren, Z.; Tang, Z. Oligonucleotide probes for ND-FISH analysis to identify rye and wheat chromosomes. Sci. Rep. 2015, 5, 10552. [Google Scholar] [CrossRef] [Green Version]
- Tang, S.; Qiu, L.; Xiao, Z.; Fu, S.; Tang, Z. New oligonucleotide probes for ND-FISH analysis to identify barley chromosomes and to investigate polymorphisms of wheat chromosomes. Genes 2016, 7, 118. [Google Scholar] [CrossRef] [Green Version]
- Tang, S.; Tang, Z.; Qiu, L.; Yang, Z.; Li, G.; Lang, T.; Zhu, W.; Zhang, J.; Fu, S. Developing new oligo probes to distinguish specific chromosomal segments and the A, B, D genomes of wheat (Triticum aestivum L.) using ND-FISH. Front. Plant Sci. 2018, 9, 1104. [Google Scholar] [CrossRef]
- Xi, W.; Tang, S.; Du, H.; Luo, J.; Tang, Z.; Fu, S. ND-FISH-positive oligonucleotide probes for detecting specific segments of rye (Secale cereale L.) chromosomes and new tandem repeats in rye. Crop J. 2020, 8, 171–181. [Google Scholar] [CrossRef]
- Cuadrado, Á.; Golczyk, H.; Jouve, N. A novel, simple and rapid nondenaturing FISH (ND-FISH) technique for the detection of plant telomeres. Potential used and possible target structures detected. Chromosome Res. 2009, 17, 755–762. [Google Scholar] [CrossRef]
- Cuadrado, Á.; Jouve, N. Chromosomal detection of simple sequence repeats (SSRs) using nondenaturing FISH (ND-FISH). Chromosoma 2010, 119, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Kuznetsova, V.M.; Razumova, O.V.; Karlov, G.I.; Dang, T.X.; Kroupin, P.Y.; Divashuk, M.G. Some peculiarities in application of denaturating and non-denaturating in situ hybridization on chromosomes of cereals. Mosc. Univ. Biol. Sci. Bull. 2019, 74, 75–80. [Google Scholar] [CrossRef]
- Li, Z.; Ren, Z.; Tan, F.; Tang, Z.; Fu, S.; Yan, B.; Ren, T. Molecular cytogenetic characterization of new wheat-rye 1R (1B) substitution and translocation lines from a Chinese Secale cereal L. Aigan with resistance to stripe rust. PLoS ONE 2016, 11, e0163642. [Google Scholar]
- Xiao, Z.; Tang, S.; Qiu, L.; Tang, Z.; Fu, S. Oligonucleotides and ND-FISH displaying different arrangements of tandem repeats and identification of Dasypyrum villosum chromosomes in wheat backgrounds. Molecules 2017, 22, 973. [Google Scholar] [CrossRef] [Green Version]
- Ren, T.; He, M.; Sun, Z.; Tan, F.; Luo, P.; Tang, Z.; Fu, S.; Yan, B.; Ren, Z.; Li, Z. The polymorphisms of oligonucleotide probes in wheat cultivars determined by ND-FISH. Molecules 2019, 24, 1126. [Google Scholar] [CrossRef] [Green Version]
- Jiang, M.; Xaio, Z.Q.; Fu, S.L.; Tang, Z.X. FISH karyotype of 85 common wheat cultivars/lines displayed by ND-FISH using oligonucleotide probes. Cereal Res. Commun. 2017, 45, 549–563. [Google Scholar] [CrossRef] [Green Version]
- Li, G.; Zhang, T.; Yu, Z.; Wang, H.; Yang, E.; Yang, Z. An efficient Oligo-FISH painting system for revealing chromosome rearrangements and polyploidization in Triticeae. Plant J. 2021, 105, 978–993. [Google Scholar] [CrossRef]
- Liu, X.; Sun, S.; Wu, Y.; Zhou, Y.; Gu, S.; Yu, H.; Yi, C.; Gu, M.; Jiang, J.; Liu, B.; et al. Dual-color oligo-FISH can reveal chromosomal variations and evolution in Oryza species. Plant J. 2020, 101, 112–121. [Google Scholar] [CrossRef]
- Fu, L.; Wang, Q.; Li, L.; Lang, T.; Guo, J.; Wang, S.; Sun, Z.; Han, S.; Huang, B.; Dong, W.; et al. Physical mapping of repetitive oligonucleotides facilitates the establishment of a genome map-based karyotype to identify chromosomal variations in peanut. BMC Plant Biol. 2021, 21, 107. [Google Scholar] [CrossRef]
- Luo, X.; He, Z. Distribution of FISH oligo-5S rDNA and oligo-(AGGGTTT) 3 in Hibiscus mutabilis L. Genome 2021, 64, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Liu, J.; He, Z. Oligo-FISH Can Identify Chromosomes and Distinguish Hippophaë rhamnoides L. Taxa. Genes 2022, 13, 195. [Google Scholar] [CrossRef] [PubMed]
- Divashuk, M.G.; Alexandrov, O.S.; Razumova, O.V.; Kirov, I.V.; Karlov, G.I. Molecular cytogenetic characterization of the dioecious Cannabis sativa with an XY chromosome sex determination system. PLoS ONE 2014, 9, e85118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Untergasser, A.; Cutcutache, I.; Koressaar, T.; Ye, J.; Faircloth, B.C.; Remm, M.; Rozen, S.G. Primer3—new capabilities and interfaces. Nucleic Acids Res. 2012, 40, e115. [Google Scholar] [CrossRef] [Green Version]
- Romanov, D.V.; Shirnin, S.Y.; Karlov, G.I.; Divashuk, M.G. Cytogenetic Study of Aegopodium podagraria (Umbelliferae) for Use in Breeding. Mosc. Univ. Biol. Sci. Bull. 2020, 75, 65–70. [Google Scholar] [CrossRef]
- Rogers, S.O.; Bendich, A.J. Extraction of DNA from plant tissues. In Plant Molecular Biology Manual; Springer: Dordrecht, The Netherlands, 1989; Volume 1, pp. 73–83. [Google Scholar] [CrossRef]
- Karlov, G.I.; Danilova, T.V.; Horlemann, C.; Weber, G. Molecular cytogenetics in hop (Humulus lupulus L.) and identification of sex chromosomes by DAPI-banding. Euphytica 2003, 132, 185–190. [Google Scholar] [CrossRef]
| Labeled Oligo-Probe | Repeat | Oligonucleotide | Dye |
|---|---|---|---|
| oligoCS-1-FAM | CS-1 | 5′-TAGTTATCTGTTAAAATCTCAACCTACACA-3′ | 5`-6-FAM |
| oligoCS-1-Cy5 | CS-1 | 5′-TAGTTATCTGTTAAAATCTCAACCTACACA-3′ | 5`-Cy5 |
| oligoCS-1-TAMRA | CS-1 | 5′-ATCGTTTTAATCGAAATAGTGAAAATCTCA-3′ | 5`-TAMRA |
| oligoCS-237-FAM | CS-237 | 5′-ATGTATTGCTGACACTCATTTGAAATCATC-3′ | 5`-6-FAM |
| oligoCS-237-Cy5 | CS-237 | 5′-ATGTATTGCTGACACTCATTTGAAATCATC-3′ | 5`-Cy5 |
| oligoCS-237-TAMRA | CS-237 | 5′-TACGTTGGTTGATTGAGGATGTTTGAAA-3′ | 5`-TAMRA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romanov, D.V.; Karlov, G.I.; Divashuk, M.G. Developing Oligo Probes for Chromosomes Identification in Hemp (Cannabis sativa L.). Plants 2022, 11, 1900. https://doi.org/10.3390/plants11151900
Romanov DV, Karlov GI, Divashuk MG. Developing Oligo Probes for Chromosomes Identification in Hemp (Cannabis sativa L.). Plants. 2022; 11(15):1900. https://doi.org/10.3390/plants11151900
Chicago/Turabian StyleRomanov, Dmitry V., Gennady I. Karlov, and Mikhail G. Divashuk. 2022. "Developing Oligo Probes for Chromosomes Identification in Hemp (Cannabis sativa L.)" Plants 11, no. 15: 1900. https://doi.org/10.3390/plants11151900
APA StyleRomanov, D. V., Karlov, G. I., & Divashuk, M. G. (2022). Developing Oligo Probes for Chromosomes Identification in Hemp (Cannabis sativa L.). Plants, 11(15), 1900. https://doi.org/10.3390/plants11151900






