Essential Oils and Neuropathic Pain
Abstract
:1. Introduction
1.1. Neuropathic Pain
1.1.1. Background: Definition, Etiology, and Pathogenesis
1.1.2. Impact
1.1.3. Focus on Natural Remedies
2. Essential Oils
2.1. Definition
2.2. Purported Uses
2.3. Global Market
2.4. Lack of Evidence
3. Lavender (Lavendula) for Neuropathic Pain
| Essential Oil | KEY Volatile Component(s) | Chemical Structure | Plant |
|---|---|---|---|
| LAVENDER | Linalyl acetate LINALOOL Tannins Caryophyllene |  |  |
| BERGAMOT | D-Limonene LINALYL ACETATE Linalool α-Terpinene/β- Pinene |  |  [29] |
| NUTMEG | TERPENEHYDROCARBONS Oxygenated Terpenes (LINALOOL) Aromatic ethers |  |  [30] |
| ROSEMARY | 1,8 CINEOLE Camphor α−Pinene |  |  |
| BILLY GOAT WEED | Terpinine-4-ol Bornyl acetate E-Caryophyllene γ -Muroleno δ -Cadinene α-Muroleno Caryophyllene-oxide LONGIFOLENE α-Humulene Precocene I & II | 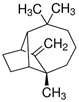 |  [31] |
| EUCALYPTUS | 1,8 CINEOL α-Pinene | 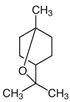 |  [32] |
4. Bergamot (Citrus bergamia) for Neuropathic Pain
5. Nutmeg (Myristica fragrans) for Neuropathic Pain
6. Rosemary (Salvia rosmarinus) for Neuropathic Pain
7. Billy Goat Weed (Ageratum conyzoides) for Neuropathic Pain
8. Eucalyptus (Eucalyptus globulus) for Neuropathic Pain
9. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Giovannini, S.; Coraci, D.; Brau, F.; Galluzzo, V.; Loreti, C.; Caliandro, P.; Padua, L.; Maccauro, G.; Biscotti, L.; Bernabei, R. Neuropathic Pain in the Elderly. Diagnostics 2021, 11, 613. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Pandhi, P.; Dutta, P. Painful diabetic neuropathy: An update. Ann. Neurosci. 2011, 18, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Kocot-Kępska, M.; Zajączkowska, R.; Mika, J.; Kopsky, D.; Wordliczek, J.; Dobrogowski, J.; Przeklasa-Muszyńska, A. Topical Treatments and Their Molecular/Cellular Mechanisms in Patients with Peripheral Neuropathic Pain—Narrative Review. Pharmaceutics 2021, 13, 450. [Google Scholar] [CrossRef] [PubMed]
- Gustorff, B.; Dorner, T.E.; Likar, R.; Grisold, W.; Lawrence, K.; Schwarz, F.; Rieder, A. Prevalence of self-reported neuropathic pain and impact on quality of life: A prospective representative survey. Acta Anaesthesiol. Scand. 2007, 52, 132–136. [Google Scholar] [CrossRef]
- Gonçalves, L.; Silva, R.; Pinto-Ribeiro, F.; Pêgo, J.M.; Bessa, J.M.; Pertovaara, A.; Sousa, N.; Almeida, A. Neuropathic pain is associated with depressive behaviour and induces neuroplasticity in the amygdala of the rat. Exp. Neurol. 2008, 213, 48–56. [Google Scholar] [CrossRef] [Green Version]
- Stroemel-Scheder, C.; Kundermann, B.; Lautenbacher, S. The effects of recovery sleep on pain perception: A systematic review. Neurosci. Biobehav. Rev. 2020, 113, 408–425. [Google Scholar] [CrossRef]
- Yawn, B.P.; Wollan, P.C.; Weingarten, T.N.; Watson, J.C.; Hooten, W.M.; Melton, L.J. The Prevalence of Neuropathic Pain: Clinical Evaluation Compared with Screening Tools in a Community Population. Pain Med. 2009, 10, 586–593. [Google Scholar] [CrossRef] [Green Version]
- McCarberg, B.H.; Billington, R. Consequences of neuropathic pain: Quality-of-life issues and associated costs. Am. J. Manag. Care 2006, 12, S263–S268. [Google Scholar]
- Berger, A.; Dukes, E.M.; Oster, G. Clinical characteristics and economic costs of patients with painful neuropathic disorders. J. Pain 2004, 5, 143–149. [Google Scholar] [CrossRef]
- Schaefer, C.; Sadosky, A.; Mann, R.; Daniel, S.; Parsons, B.; Tuchman, M.; Anschel, A.; Stacey, B.R.; Nalamachu, S.; Nieshoff, E. Pain severity and the economic burden of neuropathic pain in the United States: BEAT Neuropathic Pain Observational Study. Clin. Outcomes Res. 2014, 6, 483–496. [Google Scholar] [CrossRef] [Green Version]
- Sadosky, A.; Schaefer, C.; Mann, R.; Daniel, S.; Parsons, B.; Bergstrom, F.; Nieshoff, E.; Tuchman, M.; Nalamachu, S.; Anschel, A.; et al. Burden of neuropathic pain on quality of life in the United States: BEAT neuropathic pain observational study. J. Pain 2013, 14, S31. [Google Scholar] [CrossRef]
- Forouzanfar, F.; Hosseinzadeh, H. Medicinal herbs in the treatment of neuropathic pain: A review. Iran J. Basic Med. Sci. 2018, 21, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Chandran, H.; Meena, M.; Sharma, K. Microbial Biodiversity and Bioremediation Assessment Through Omics Approaches. Front. Environ. Chem. 2020, 1, 570326. [Google Scholar] [CrossRef]
- Aziz, Z.A.A.; Ahmad, A.; Setapar, S.H.M.; Karakucuk, A.; Azim, M.M.; Lokhat, D.; Rafatullah, M.; Ganash, M.; Kamal, M.A.; Ashraf, G. Essential Oils: Extraction Techniques, Pharmaceutical And Therapeutic Potential—A Review. Curr. Drug Metab. 2018, 19, 1100–1110. [Google Scholar] [CrossRef]
- Nirmala, M.J.; Durai, L.; Gopakumar, V.; Nagarajan, R. Anticancer and antibacterial effects of a clove bud essential oil-based nanoscale emulsion system. Int. J. Nanomed. 2019, 14, 6439–6450. [Google Scholar] [CrossRef] [Green Version]
- Abers, M.; Schroeder, S.; Goelz, L.; Sulser, A.; Rose, T.S.; Puchalski, K.; Langland, J. Antimicrobial activity of the volatile substances from essential oils. BMC Complement. Med. Ther. 2021, 21, 124. [Google Scholar] [CrossRef]
- Alam, A.; Rehman, N.; Ansari, M.; Palla, A. Effects of Essential Oils of Elettaria cardamomum Grown in India and Guatemala on Gram-Negative Bacteria and Gastrointestinal Disorders. Molecules 2021, 26, 2546. [Google Scholar] [CrossRef]
- Hong, S.J.; Cho, J.; Boo, C.G.; Youn, M.Y.; Pan, J.H.; Kim, J.K.; Shin, E.-C. Inhalation of Patchouli (Pogostemon Cablin Benth.) Essential Oil Improved Metabolic Parameters in Obesity-Induced Sprague Dawley Rats. Nutrients 2020, 12, 2077. [Google Scholar] [CrossRef]
- Mieres-Castro, D.; Ahmar, S.; Shabbir, R.; Mora-Poblete, F. Antiviral Activities of Eucalyptus Essential Oils: Their Effectiveness as Therapeutic Targets against Human Viruses. Pharmaceuticals 2021, 14, 1210. [Google Scholar] [CrossRef]
- Horváth, A.; Pandur, E.; Sipos, K.; Micalizzi, G.; Mondello, L.; Böszörményi, A.; Birinyi, P.; Horváth, G. Anti-inflammatory effects of lavender and eucalyptus essential oils on the in vitro cell culture model of bladder pain syndrome using T24 cells. BMC Complement. Med. Ther. 2022, 22, 119. [Google Scholar] [CrossRef]
- Matulyte, I.; Jekabsone, A.; Jankauskaite, L.; Zavistanaviciute, P.; Sakiene, V.; Bartkiene, E.; Ruzauskas, M.; Kopustinskiene, D.M.; Santini, A.; Bernatoniene, J. The Essential Oil and Hydrolats from Myristica fragrans Seeds with Magnesium Aluminometasilicate as Excipient: Antioxidant, Antibacterial, and Anti-inflammatory Activity. Foods 2020, 9, 37. [Google Scholar] [CrossRef] [Green Version]
- Grand View Research. Essential Oils Market Size, Share & Trends Analysis Report by Product (Orange, Cornmint, Eucalyptus), By Application (Medical, Food & Beverages, Spa & Relaxation), By Sales Channel, By Region, And Segment Forecasts, 2021–2028; Market Analysis Report; Grand View Research: San Francisco, CA, USA, 2021. [Google Scholar]
- Nasiri, A.; Mahmodi, M.A. Aromatherapy massage with lavender essential oil and the prevention of disability in ADL in patients with osteoarthritis of the knee: A randomized controlled clinical trial, Complementary Therapies in Clinical Practice. Complement. Ther. Clin. Pr. 2018, 30, 116–121. [Google Scholar] [CrossRef] [PubMed]
- Metin, G.; Arikan Donmez, Z.; Izgu, A.; Ozdemir, N.L.; Arslan, I.E. Aromatherapy Massage for Neuropathic Pain and Quality of Life in Diabetic Patients. Jrnl. Nurs. Schol. 2017, 49, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Rivaz, M.; Rahpeima, M.; Khademian, Z.; Dabbaghmanesh, M.H. The effects of aromatherapy massage with lavender essential oil on neuropathic pain and quality of life in diabetic patients: A randomized clinical trial. Complement. Ther. Clin. Pr. 2021, 44, 101430. [Google Scholar] [CrossRef] [PubMed]
- Sanna, M.D.; Les, F.; Lopez, V.; Galeotti, N. Lavender (Lavandula angustifolia Mill.) Essential Oil Alleviates Neuropathic Pain in Mice with Spared Nerve Injury. Front. Pharmacol. 2019, 10, 472. [Google Scholar] [CrossRef]
- e Soares, G.A.B.; Bhattacharya, T.; Chakrabarti, T.; Tagde, P.; Cavalu, S. Exploring Pharmacological Mechanisms of Essential Oils on the Central Nervous System. Plants 2021, 11, 21. [Google Scholar] [CrossRef]
- Tashiro, S.; Yamaguchi, R.; Ishikawa, S.; Sakurai, T.; Kajiya, K.; Kanmura, Y.; Kuwaki, T.; Kashiwadani, H. Odour-induced analgesia mediated by hypothalamic orexin neurons in mice. Sci. Rep. 2016, 6, 37129. [Google Scholar] [CrossRef] [Green Version]
- Available online: https://commons.wikimedia.org/wiki/File:Citrus_bergamia_-_Bergamot.jpg (accessed on 4 June 2022).
- Available online: https://commons.wikimedia.org/wiki/File:Myris_fragr_Fr_080112-3290_ltn.jpg (accessed on 4 June 2022).
- Available online: https://commons.wikimedia.org/wiki/File:Ageratum_conyzoides_1.jpg (accessed on 4 June 2022).
- Available online: https://commons.wikimedia.org/wiki/File:Eucalyptus_globulus_subsp._pseudoglobulus.jpg (accessed on 4 June 2022).
- Komatsu, T.; Katsuyama, S.; Uezono, Y.; Sakurada, C.; Tsuzuki, M.; Hamamura, K.; Bagetta, G.; Sakurada, S.; Sakurada, T. Possible involvement of the peripheral Mu-opioid system in antinociception induced by bergamot essential oil to allodynia after peripheral nerve injury. Neurosci. Lett. 2018, 686, 127–132. [Google Scholar] [CrossRef]
- Kuwahata, H.; Komatsu, T.; Katsuyama, S.; Sakurada, S.; Sakurada, C. Inhibitory effect of bergamot essential oil injected into the mouse hind-paw on capsaicin-induced nociceptive behaviors. Aroma Res. 2009, 10, 128–133. [Google Scholar]
- Katsuyama, S.; Mizoguchi, H.; Komatsu, T.; Sakurada, C.; Tsuzuki, M.; Sakurada, S.; Sakurada, T. Antinociceptive effects of spinally administered nociceptin/orphanin FQ and its N-terminal fragments on capsaicin-induced nociception. Peptides 2011, 32, 1530–1535. [Google Scholar] [CrossRef]
- Hamamura, K.; Katsuyama, S.; Komatsu, T.; Scuteri, D.; Bagetta, G.; Aritake, K.; Sakurada, T. Behavioral Effects of Continuously Administered Bergamot Essential Oil on Mice with Partial Sciatic Nerve Ligation. Front. Pharmacol. 2020, 11, 1310. [Google Scholar] [CrossRef] [PubMed]
- Abourashed, E.A.; El-Alfy, A.T. Chemical diversity and pharmacological significance of the secondary metabolites of nutmeg (Myristica fragrans Houtt.). Phytochem. Rev. 2016, 15, 1035–1056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Motilal, S.; Maharaj, R.G. Nutmeg Extracts for Painful Diabetic Neuropathy: A Randomized, Double-Blind, Controlled Study. J. Altern. Complement. Med. 2013, 19, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Rašković, A.; Milanović, I.; Pavlović, N.; Ćebović, T.; Vukmirović, S.; Mikov, M. Antioxidant activity of rosemary (Rosmarinus officinalis L.) essential oil and its hepatoprotective potential. BMC Complement. Altern. Med. 2014, 14, 225. [Google Scholar] [CrossRef] [Green Version]
- Rahbardar, M.G.; Hosseinzadeh, H. Therapeutic effects of rosemary (Rosmarinus officinalis L.) and its active constituents on nervous system disorders. Iran J. Basic Med. Sci. 2020, 23, 1100–1112. [Google Scholar] [CrossRef]
- Sukmawan, Y.P.; Anggadiredja, K.; Adnyana, I.K. Anti-Neuropathic Pain Activity of Ageratum conyzoides L due to the Essential Oil Components. CNS Neurol. Disord.-Drug Targets 2021, 20, 181–189. [Google Scholar] [CrossRef]
- Sukmawan, Y.P.; Anggadiredja, K.; Adnyana, I.K. Anti-neuropathic pain mechanistic study of A. conyzoides essential oil, Precocene II, Caryophyllene, or Longifolene as single agent and in combination with pregabalin. CNS Neurol. Disord. Drug Targets 2022. [Google Scholar] [CrossRef]
- Sebei, K.; Sakouhi, F.; Herchi, W.; Khouja, M.L.; Boukhchina, S. Chemical composition and antibacterial activities of seven Eucalyptus species essential oils leaves. Biol. Res. 2015, 48, 7. [Google Scholar] [CrossRef] [Green Version]
- Jung, Y.-H.; Kim, Y.O.; Lin, H.; Cho, J.-H.; Park, J.-H.; Lee, S.-D.; Bae, J.; Kang, K.M.; Kim, Y.-G.; Pae, A.N.; et al. Discovery of Potent Antiallodynic Agents for Neuropathic Pain Targeting P2X3 Receptors. ACS Chem. Neurosci. 2017, 8, 1465–1478. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Liu, Y.-G.; Li, Q.; Wang, X.-D.; Zheng, X.-B.; Yang, B.-L.; Wan, B.; Ma, J.-M.; Liu, Z.-X. 1,8-cineole decreases neuropathic pain probably via a mechanism mediating P2X3 receptor in the dorsal root ganglion. Neurochem. Int. 2018, 121, 69–74. [Google Scholar] [CrossRef]
- Zheng, X.-B.; Zhang, Y.-L.; Li, Q.; Liu, Y.-G.; Wang, X.-D.; Yang, B.-L.; Zhu, G.-C.; Zhou, C.-F.; Gao, Y.; Liu, Z.-X. Effects of 1,8-cineole on neuropathic pain mediated by P2X2 receptor in the spinal cord dorsal horn. Sci. Rep. 2019, 9, 7909. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.; Lau, B.; Ngai, S.; Tsang, H. Therapeutic Effect and Mechanisms of Essential Oils in Mood Disorders: Interaction between the Nervous and Respiratory Systems. Int. J. Mol. Sci. 2021, 22, 4844. [Google Scholar] [CrossRef] [PubMed]
- Paula-Freire, L.; Molska, G.R.; Andersen, M.L.; Carlini, E.L. Ocimum gratissimum Essential Oil and its Isolated Compounds (Eusenol and Myrcene) Reduce Neuropathic Pain in Mice. Planta Med. 2016, 82, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Ahmadimoghaddam, D.; Zarei, M.; Mohammadi, S.; Izadidastenaei, Z.; Salehi, I. Bupleurum falcatum L. alleviates nociceptive and neuropathic pain: Potential mechanisms of action. J. Ethnopharmacol. 2021, 273, 113990. [Google Scholar] [CrossRef] [PubMed]
- Borgonetti, V.; López, V.; Galeotti, N. Ylang-ylang (Cananga odorata (Lam.) Hook. f. & Thomson) essential oil reduced neuropathic-pain and associated anxiety symptoms in mice. J. Ethnopharmacol. 2022, 294, 115362. [Google Scholar] [CrossRef] [PubMed]
| Essential oil | Human Studies | Significance | Comments | Animal Studies | Significance | Comments |
|---|---|---|---|---|---|---|
| LAVENDER | Y | Y | PD | Y | Y | Mice |
| BERGAMOT | N | - | - | Y | Y | |
| NUTMEG | Y | NS | 74 subj (DPN) | N | - | - |
| ROSEMARY | N | - | - | N | - | - |
| BILLY GOAT WEED | N | - | - | Y | +/− | Rat (Poorly described) |
| EUCALYPTUS | N | - | - | Y | Y | Rat |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ridouh, I.; Hackshaw, K.V. Essential Oils and Neuropathic Pain. Plants 2022, 11, 1797. https://doi.org/10.3390/plants11141797
Ridouh I, Hackshaw KV. Essential Oils and Neuropathic Pain. Plants. 2022; 11(14):1797. https://doi.org/10.3390/plants11141797
Chicago/Turabian StyleRidouh, Imane, and Kevin V. Hackshaw. 2022. "Essential Oils and Neuropathic Pain" Plants 11, no. 14: 1797. https://doi.org/10.3390/plants11141797
APA StyleRidouh, I., & Hackshaw, K. V. (2022). Essential Oils and Neuropathic Pain. Plants, 11(14), 1797. https://doi.org/10.3390/plants11141797






