Arbuscular Mycorrhizal Fungi and Fertilization Influence Yield, Growth and Root Colonization of Different Tomato Genotype
Abstract
1. Introduction
2. Results
2.1. Soil Sample Results and Differences of Soil Components before and after Cultures
2.2. Parameters of Plant Growth, Fruit Production and Quality
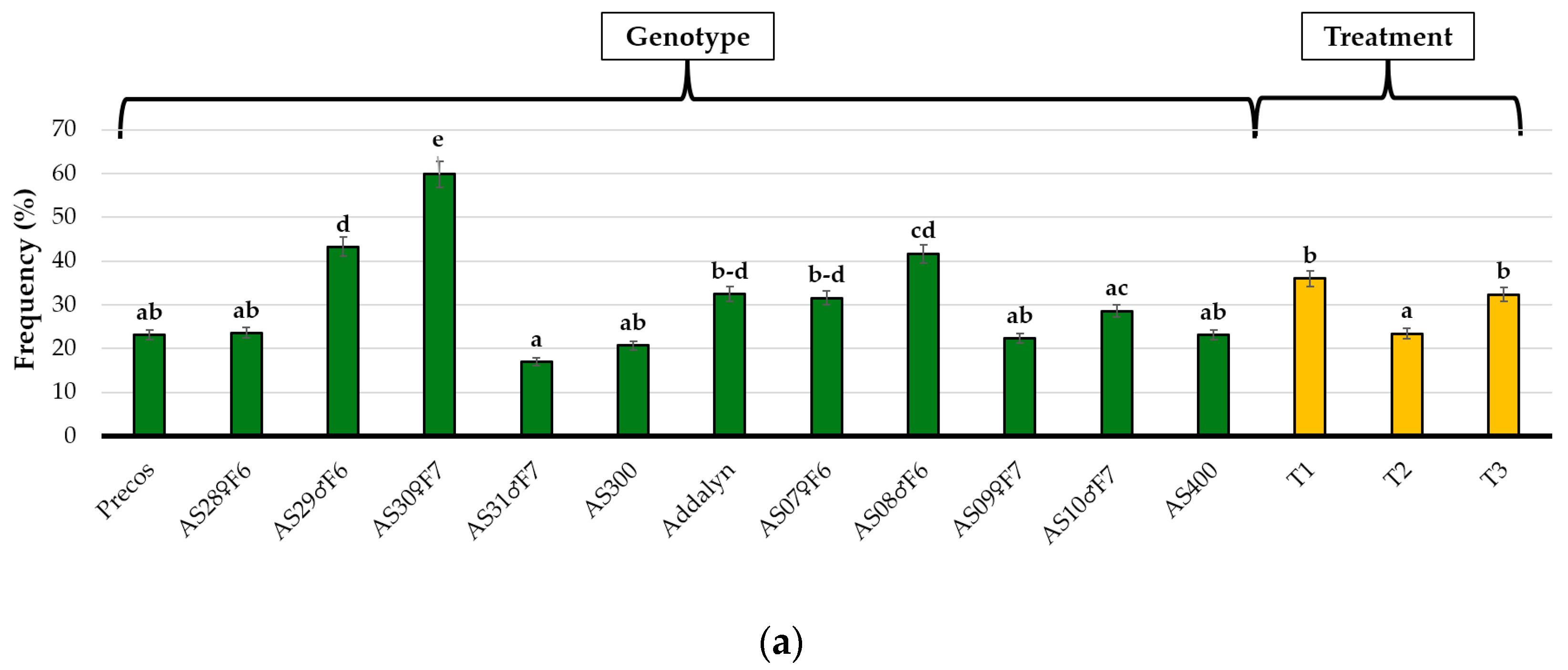
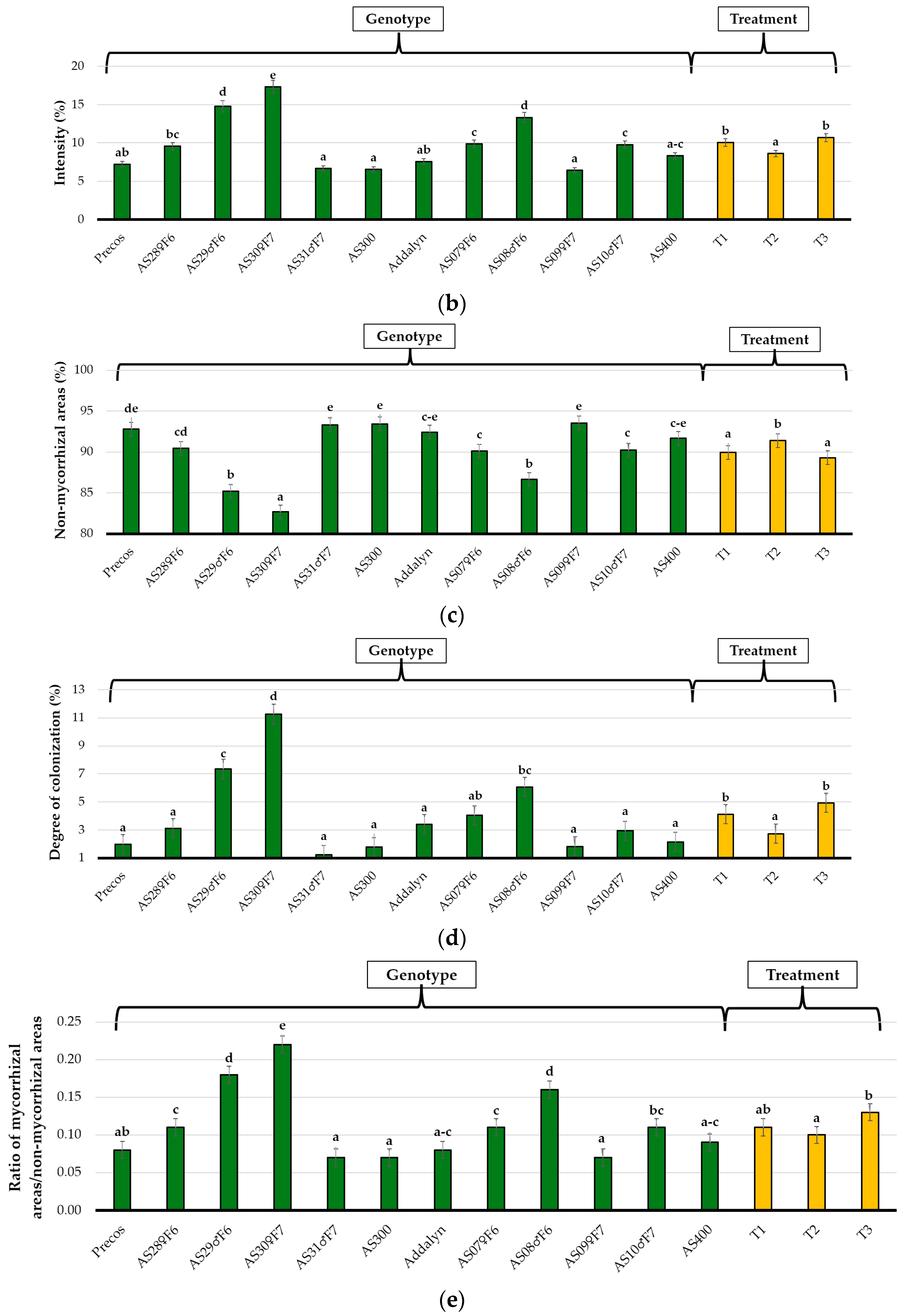
2.3. Colonization Parameters As a Genotype Reaction to Mycorrhizal Inoculation and Treatment Influence
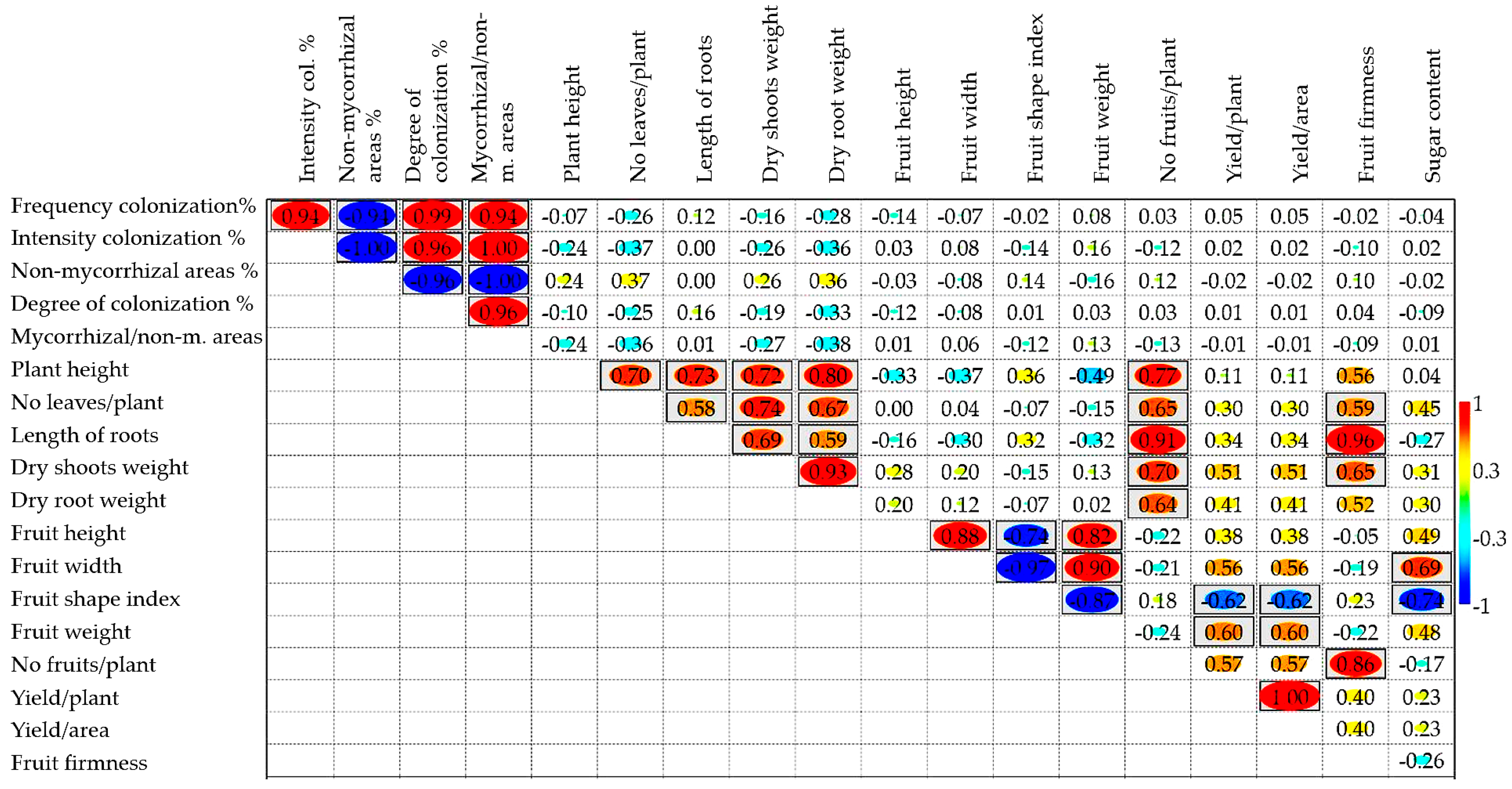
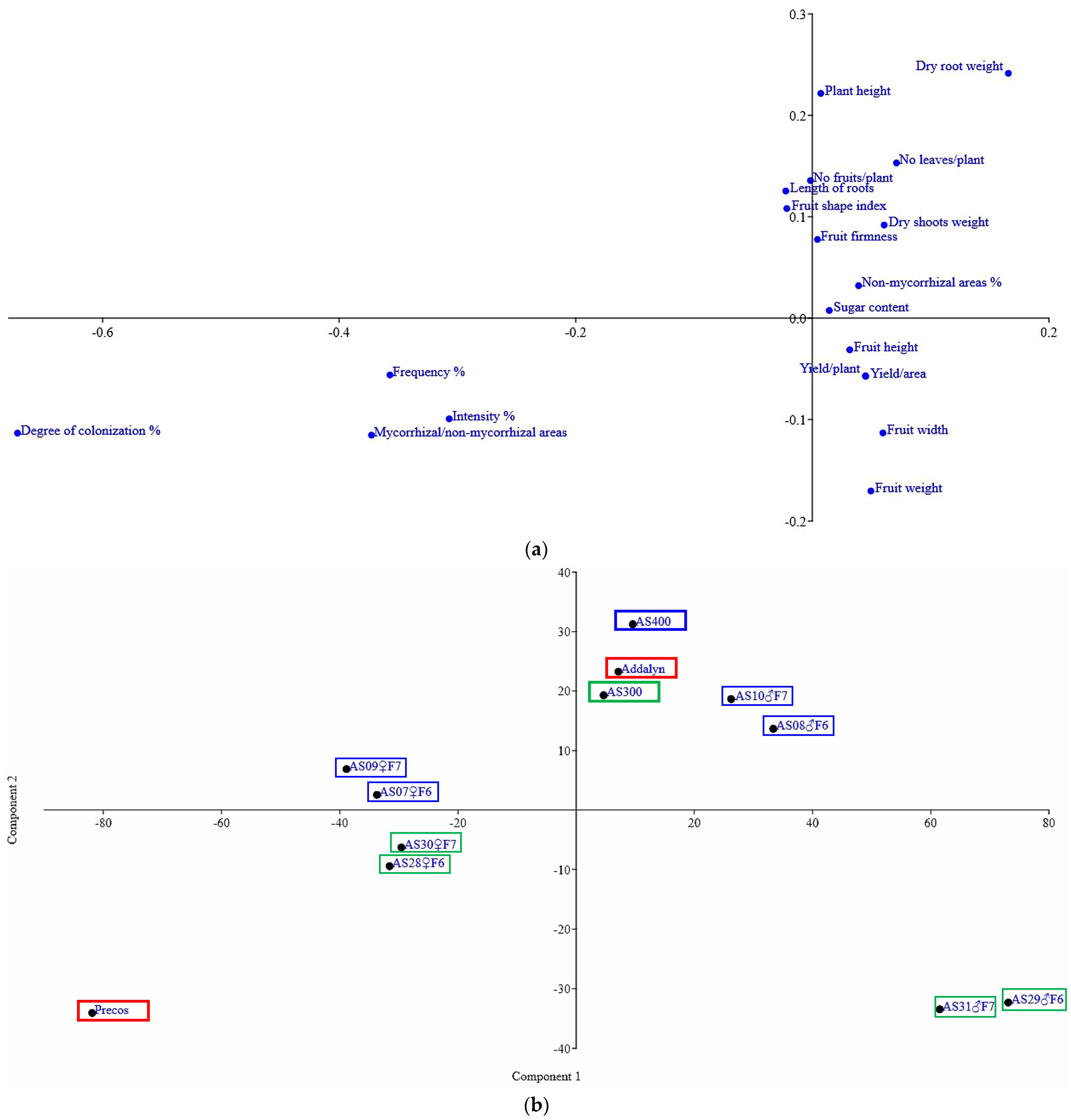
2.4. Correlations between the Analyzed Characteristics and the Multivariate Analysis
3. Discussion
4. Materials and Methods
4.1. Microclimatological Measurements Using Climate Stations
4.2. Field Location and Experimental Design
4.3. Biological Material
4.4. Fertigation Plan and Nutrition Data
4.5. Laboratory and Microscopic Analysis
4.6. Soil Sample Collection and Preparation for Analysis for Agrotechnical Purposes
4.7. Growth Parameters
4.8. Determination of Sugar Content (°Brix) and Firmness
4.9. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. 2019. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 15 January 2022).
- Abu-Alrub, I.; Sameer, S.A.; Aziz, A. Effect of Different Rates of Nitrogen and Phosphorus Fertilizers on Yield and Quality of Greenhouse Tomato under the UAE Condition. EC Agric. 2019, 5, 139–146. [Google Scholar]
- Li, B.; Boiarkina, I.; Young, B.; Yu, W.; Singhal, N. Prediction of Future Phosphate Rock: A Demand Based Model. J. Environ. Inform. 2018, 31, 41–53. [Google Scholar] [CrossRef]
- Abdul-Baki, A.A.; Teasdale, J.; Korcak, R.; Chitwood, D.; Huettel, R. Fresh-market Tomato Production in a Low-input Alternative System Using Cover-crop Mulch. HortScience 1996, 31, 65–69. [Google Scholar] [CrossRef]
- Roberts, D.P.; Mattoo, A.K. Sustainable Agriculture—Enhancing Environmental Benefits, Food Nutritional Quality and Building Crop Resilience to Abiotic and Biotic Stresses. Agriculture 2018, 8, 8. [Google Scholar] [CrossRef]
- Mattoo, A.K.; Teasdale, J.R. Ecological and Genetic Systems Underlying Sustainable Horticulture. Hortic. Rev. 2010, 37, 331–362. [Google Scholar] [CrossRef]
- Fatima, T.; Sobolev, A.P.; Teasdale, J.R.; Kramer, M.; Bunce, J.; Handa, A.K.; Mattoo, A.K. Fruit metabolite networks in engineered and non-engineered tomato genotypes reveal fluidity in a hormone and agroecosystem specific manner. Metabolomics 2016, 12, 103. [Google Scholar] [CrossRef]
- Adetunjia, A.T.; Ncubeb, B.; Muliddzic, R.; Lewu, F.B. Management impact and benefit of cover crops on soil quality: A review. Soil Tillage Res. 2020, 204, 104707. [Google Scholar] [CrossRef]
- Kocira, A.; Staniak, M.; Tomaszewska, M.; Kornas, R.; Cymerman, J.; Panasiewicz, K.; Lipińska, H. Legume Cover Crops as One of the Elements of Strategic Weed Management and Soil Quality Improvement. A Review. Agriculture 2020, 10, 394. [Google Scholar] [CrossRef]
- Soane, B.D.; Ball, B.C.; Arvidsson, J.; Basch, G.; Moreno, F.; Roger-Estrade, J. No-till in northern, western and south-western Europe: A review of problems and opportunities for crop production and the environment. Soil Tillage Res. 2012, 118, 66–87. [Google Scholar] [CrossRef]
- Abdollahi, I.; Munkholm, I.J. Tillage system and cover crop effects on soil quality: I. Chemical, mechanical and biological properties. J. Soil Water Conserv. 2014, 78, 262–270. [Google Scholar] [CrossRef]
- Tebrügge, F. No-tillage visions—Protection of soil, water and climate and influence on management and farm income. Conserv. Agric. 2001, 1, 303–316. [Google Scholar]
- Moretti, C.; Mattos, L.; Calbo, A.; Sargent, S. Climate changes and potential impacts on postharvest quality of fruit and vegetable crops: A review. Food Res. Int. 2010, 43, 1824–1832. [Google Scholar] [CrossRef]
- Singh, R.; Singh, G.S. Traditional agriculture: A climate-smart approach for sustainable food production. Energy Ecol. Environ. 2017, 2, 296–316. [Google Scholar] [CrossRef]
- Hillel, D.; Hatfield, J.L.; Powlson, D.S.; Rosenzweig, C.; Scow, K.M.; Singer, M.J. Encyclopedia of Soils in the Environment; Sparks, D.L., Ed.; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; Volume 4, 2200p. [Google Scholar]
- Sharma, N.; Singhvi, R. Effects of Chemical Fertilizers and Pesticides on Human Health and Environment: A Review. Int. J. Agric. Environ. Biotechnol. 2017, 10, 675. [Google Scholar] [CrossRef]
- Gardi, C.; Jeffery, S.; Saltelli, A. An estimate of potential threats levels to soil biodiversity in EU. Glob. Chang. Biol. 2013, 19, 1538–1548. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A.; McCulley, L.; Roh, R.; Lopez-Ridaura, S.; Palacios-Rojas, N.; Gunaratna, N.S. Maize agro-food systems to ensure food and nutrition security in reference to the Sustainable Development Goals. Glob. Food Secur. 2019, 25, 100327. [Google Scholar] [CrossRef]
- Igiehon, O.; Babalola, O. Biofertilizers and Sustainable Agriculture: Exploring Arbuscular Mycorrhizal Fungi. Appl. Microbiol. Biotechnol. 2017, 101, 4871–4881. [Google Scholar] [CrossRef]
- Barazetti, A.R.; Simionato, A.S.; Navarro, M.O.P.; Santos, I.M.O.d.; Modolon, F.; Andreata, M.F.d.L.; Liuti, G.; Cely, M.V.T.; Chryssafidis, A.L.; Dealis, M.L.; et al. Formulations of Arbuscular Mycorrhizal Fungi Inoculum Applied to Soybean and CornPlants under Controlled and Field Conditions. Appl. Soil Ecol. 2019, 142, 25–33. [Google Scholar] [CrossRef]
- Jiang, Y.N.; Wang, W.X.; Xie, Q.J.; Liu, N.; Liu, L.X.; Wang, D.P.; Zhang, X.; Yang, C.; Chen, X.; Tang, D.; et al. Plants transfer lipids to sustain colonization by mutualistic mycorrhizal and parasitic fungi. Science 2017, 356, 1172–1175. [Google Scholar] [CrossRef]
- Mahmood, I.; Rizvi, R. Mycorrhiza and Organic Farming. Asian J. Plant Sci. 2010, 9, 241–248. [Google Scholar] [CrossRef][Green Version]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008. [Google Scholar]
- Keymer, A.; Pimprikar, P.; Wewer, V.; Huber, C.; Brands, M.; Bucerius, S.L.; Delaux, P.-M.; Klingl, V.; von Röpenack-Lahaye, E.; Wang, T.L.; et al. Lipid transfer from plants to arbuscular mycorrhiza fungi. eLife 2017, 6, e29107. [Google Scholar] [CrossRef] [PubMed]
- Pozo, M.J.; Azcón-Aguilar, C. Unraveling mycorrhiza-induced resistance. Curr. Opin. Plant Biol. 2007, 10, 393–398. [Google Scholar] [CrossRef] [PubMed]
- Evelin, H.; Kapoor, R.; Giri, B. Arbuscular mycorrhizal fungi in alleviation of salt stress: A review. Ann. Bot. 2009, 104, 1263–1280. [Google Scholar] [CrossRef]
- Miransari, M. Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant Biol. 2010, 12, 563–569. [Google Scholar] [CrossRef]
- Bona, E.; Cantamessa, S.; Massa, N.; Manassero, P.; Marsano, F.; Copetta, A.; Lingua, G.; D’Agostino, G.; Gamalero, E.; Berta, G. Arbuscular mycorrhizal fungi and plant growth-promoting pseudomonads improve yield, quality and nutritional value of tomato: A field study. Mycorrhiza 2016, 27, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Guo, H.; Zhang, Q.; Guo, H.; Zhang, L.; Zhang, C.; Gou, Z.; Liu, Y.; Wei, J.; Chen, A.; et al. Arbuscular mycorrhizal fungi (AMF) enhanced the growth, yield, fiber quality and phosphorus regulation in upland cotton (Gossypium hirsutum L.). Sci. Rep. 2020, 10, 2084. [Google Scholar] [CrossRef] [PubMed]
- Kapulnik, Y.; Tsror (Lahkim), L.; Zipori, I.; Hazanovsky, M.; Wininger, S.; Dag, A. Effect of AMF application on growth, productivity and susceptibility to Verticillium wilt of olives grown under desert conditions. Symbiosis 2010, 52, 103–111. [Google Scholar] [CrossRef]
- Sabatino, L.; Iapichino, G.; Consentino, B.B.; D’Anna, F.; Rouphel, Y. Rootstock and Arbuscular Mycorrhiza Combinatorial Effects on Eggplant Crop Performance and Fruit Quality under Greenhouse Conditions. Agronomy 2020, 10, 693. [Google Scholar] [CrossRef]
- Wang, C.; Li, X.; Zhou, J.; Wang, G.; Dong, Y. Effects of Arbuscular Mycorrhizal Fungi on Growth and Yield of Cucumber Plants. Commun. Soil Sci. Plant Anal. 2008, 39, 499–509. [Google Scholar] [CrossRef]
- Sun, R.-T.; Zhang, Z.-Z.; Zhou, N.; Srivastava, A.; Kuča, K.; Abd-Allah, E.F.; Hashem, A.; Wu, Q.-S. A review of the interaction of medicinal plants and arbuscular mycorrhizal fungi in the rhizosphere. Not. Bot. Horti Agrobot. Cluj-Napoca 2021, 49, 12454. [Google Scholar] [CrossRef]
- Alori, E.T.; Glick, B.R.; Babalola, O.O. Microbial Phosphorus Solubilization and Its Potential for Use in Sustainable Agriculture. Front. Microbiol. 2017, 8, 971. [Google Scholar] [CrossRef] [PubMed]
- Gianinazzi, S.; Gollotte, A.; Binet, M.-N.; Van Tuinen, D.; Redecker, D.; Wipf, D. Agroecology: The key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 2010, 20, 519–530. [Google Scholar] [CrossRef]
- Crișan, I.; Stoie, A. Seasonal arbuscular mycorrhiza colonization dynamic displays genotype-specific pattern in Iris sibirica L. Not. Sci. Biol. 2021, 13, 10838. [Google Scholar] [CrossRef]
- De Vita, P.; Avio, L.; Sbrana, C.; Laidò, G.; Marone, D.; Mastrangelo, A.M.; Cattivelli, L.; Giovannetti, M. Genetic markers associated to arbuscular mycorrhizal colonization in durum wheat. Sci. Rep. 2018, 8, 10612. [Google Scholar] [CrossRef] [PubMed]
- Jacott, C.N.; Murray, J.D.; Ridout, C.J. Trade-Offs in Arbuscular Mycorrhizal Symbiosis: Disease Resistance, Growth Responses and Perspectives for Crop Breeding. Agronomy 2017, 7, 75. [Google Scholar] [CrossRef]
- Felföldi, Z.; Ranga, F.; Socaci, S.A.; Farcas, A.; Plazas, M.; Sestras, A.F.; Vodnar, D.C.; Prohens, J.; Sestras, R.E. Physico-Chemical, Nutritional, and Sensory Evaluation of Two New Commercial Tomato Hybrids and Their Parental Lines. Plants 2021, 10, 2480. [Google Scholar] [CrossRef]
- Boldt, K.; Pörs, Y.; Haupt, B.; Bitterlich, M.; Kühn, C.; Grimm, B.; Franken, P. Photochemical processes, carbon assimilation and RNA accumulation of sucrose transporter genes in tomato arbuscular mycorrhiza. J. Plant Physiol. 2011, 168, 1256–1263. [Google Scholar] [CrossRef]
- Ceballos, I.; Ruiz, M.; Fernández, C.; Peña, R.; Rodríguez, A.; Sanders, I.R. The In Vitro Mass-Produced Model Mycorrhizal Fungus, Rhizophagus irregularis, Significantly Increases Yields of the Globally Important Food Security Crop Cassava. PLoS ONE 2013, 8, e70633. [Google Scholar] [CrossRef]
- Bucher, M.; Hause, B.; Krajinski, F.; Küster, H. Through the doors of perception to function in arbuscular mycorrhizal symbioses. New Phytol. 2014, 204, 833–840. [Google Scholar] [CrossRef]
- Dey, M.; Ghosh, S. Arbuscular mycorrhizae in plant immunity and crop pathogen control. Rhizosphere 2022, 22, 100524. [Google Scholar] [CrossRef]
- Devi, N.O.; Tombisana Devi, R.K.T.; Debbarma, M.; Hajong, M.; Thokchom, S. Effect of endophytic Bacillus and arbuscular mycorrhiza fungi (AMF) against Fusarium wilt of tomato caused by Fusarium oxysporum f. sp. lycopersici. ESBCP 2022, 32, 1. [Google Scholar] [CrossRef]
- Tran, C.T.; Watts-Williams, S.J.; Smernik, R.J.; Cavagnaro, T.R. Arbuscular mycorrhizas increased tomato biomass and nu-trition but did not affect local soil P availability or 16S bacterial community in the field. Sci. Total Environ. 2022, 819, 152620. [Google Scholar] [CrossRef] [PubMed]
- Di Tomassi, I.; Chatterjee, N.; Barrios-Masias, F.H.; Zhou, Q.; Gu, C.; Margenot, A.J. Arbuscular mycorrhizae increase bio-mass and nutrient uptake of tomato fertilized with struvite compared to monoammonium phosphate. Plant Soil 2021, 464, 321–333. [Google Scholar] [CrossRef]
- Avio, L.; Turrini, A.; Giovannetti, M.; Sbrana, C. Designing the Ideotype Mycorrhizal Symbionts for the Production of Healthy Food. Front. Plant Sci. 2018, 9, 1089. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, K.S.; Santhanakrishnan, P.; Balasubramanian, P. Responses of field grown tomato plants to arbuscular mycorrhizal fungal colonization under varying intensities of drought stress. Sci. Hortic. 2006, 107, 245–253. [Google Scholar] [CrossRef]
- Dasgan, H.; Kusvuran, S.; Ortas, I. Responses of soilless grown tomato plants to arbuscular mycorrhizal fungal (Glomus fasciculatum) colonization in recycling and open systems. Afr. J. Biotechnol. 2008, 7, 3606–3613. [Google Scholar]
- Salvioli, A.; Zouari, I.; Chalot, M.; Bonfante, P. The arbuscular mycorrhizal status has an impact on the transcriptome profile and amino acid composition of tomato fruit. BMC Plant Biol. 2012, 12, 44. [Google Scholar] [CrossRef]
- Grant, C.A.; Flaten, D.N.; Tomasiewicz, D.J.; Sheppard, S.C. The importance of early season phosphorus nutrition. Can. J. Plant Sci. 2001, 81, 211–224. [Google Scholar] [CrossRef]
- Grant, C.; Bittman, S.; Montreal, M.; Plenchette, C.; Morel, C. Soil and fertilizer phosphorus: Effects on plant P supply and mycorrhizal development. Can. J. Plant Sci. 2005, 85, 3–14. [Google Scholar] [CrossRef]
- Berta, G.; Sampo, S.; Gamalero, E.; Massa, N.; Lemanceau, P. Suppression of Rhizoctonia root-rot of tomato by Glomus mossae BEG12 and Pseudomonas fluorescens A6RI is associated with their effect on the pathogen growth and on the root morphogenesis. Eur. J. Plant Pathol. 2005, 111, 279–288. [Google Scholar] [CrossRef]
- Ezzo, M.I.; Glala, A.A.; Habib, H.A.M.; Helaly, A.A. Response of sweet pepper grown in sandy and clay soil lysimeters to water regimes. Am.-Eurasian J. Agric. Environ. Sci. 2010, 8, 18–26. [Google Scholar]
- Ortas, I. Effect of mycorrhiza application on plant growth and nutrient uptake in cucumber production under field conditions. Span. J. Agric. Res. 2010, 8, S116–S122. [Google Scholar]
- Matsubara, Y.; Tamura, H.; Harada, T. Growth enhancement and verticillium wilt control by vesicular arbuscular mycorrhizal fungus inoculation in eggplant. J. Jpn. Soc. Hortic. Sci. 1995, 64, 555–561. [Google Scholar] [CrossRef]
- Gemma, J.N.; Koske, R.E.; Habte, M. Mycorrhizal dependency of some endemic and endangered Hawaiian plant species. Am. J. Bot. 2002, 89, 337–345. [Google Scholar] [CrossRef]
- Plenchette, C.; Fortin, J.A.; Furlan, V. Growth responses of several plant species to mycorrhizae in a soil of moderate P-fertility. Plant Soil 1983, 70, 199–209. [Google Scholar] [CrossRef]
- Hart, M.; Ehret, D.L.; Krumbein, A.; Leung, C.; Murch, S.; Turi, C.E.; Franken, P. Inoculation with arbuscular mycorrhizal fungi improves the nutritional value of tomatoes. Mycorrhiza 2015, 25, 359–376. [Google Scholar] [CrossRef]
- Xie, M.-M.; Wang, Y.; Li, Q.-S.; Kuča, K.; Wu, Q.-S. A friendly-environmental strategy: Application of arbuscular mycorrhizal fungi to ornamental plants for plant growth and garden landscape. Not. Bot. Horti Agrobo. Cluj-Napoca 2020, 48, 1100–1115. [Google Scholar] [CrossRef]
- Heeb, A.; Lundegårdh, B.; Savage, G.; Ericsson, T. Impact of organic and inorganic fertilizers on yield, taste, and nutritional quality of tomatoes. J. Plant Nutr. Soil Sci. 2006, 169, 535–541. [Google Scholar] [CrossRef]
- Ortas, I. The effect of mycorrhizal fungal inoculation on plant yield, nutrient uptake and inoculation effectiveness under long-term field conditions. Field Crop. Res. 2012, 125, 35–48. [Google Scholar] [CrossRef]
- Baum, C.; El-Tohamy, W.; Gruda, N. Increasing the productivity and product quality of vegetable crops using arbuscular mycorrhizal fungi: A review. Sci. Hortic. 2015, 187, 131–141. [Google Scholar] [CrossRef]
- Sanayei, S.; Barmaki, M.; Ebadi, A.; Torabi-Giglou, M. Amelioration of water deficiency stress in roselle (Hibiscus sabdariffa) by arbuscular mycorrhizal fungi and plant growth-promoting rhizobacteria. Not. Bot. Horti Agrobo. Cluj-Napoca 2021, 49, 11987. [Google Scholar] [CrossRef]
- Bowles, T.M.; Barrios-Masias, F.H.; Carlisle, E.A.; Cavagnaro, T.R.; Jackson, L.E. Effects of arbuscular mycorrhizae on tomato yield, nutrient uptake, water relations, and soil carbon dynamics under deficit irrigation in field conditions. Sci. Total Environ. 2016, 566–567, 1223–1234. [Google Scholar] [CrossRef] [PubMed]
- Helyes, L.; Pék, Z.; Daood, H.G.; Posta, K. Simultaneous effect of mycorrhizae and water supply on yield formation of processing tomato. Acta Hortic. 2017, 1159, 31–36. [Google Scholar] [CrossRef]
- Njeru, E.M.; Bocci, G.; Avio, L.; Sbrana, C.; Turrini, A.; Giovannetti, M.; Bàrberi, P. Functional identity has a stronger effect than diversity on mycorrhizal symbiosis and productivity of field grown organic tomato. Eur. J. Agron. 2017, 86, 1–11. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G.; Graziani, G.; Ritieni, A.; Cardarelli, M.; De Pascale, S. Phenolic composition, antioxidant activity and mineral profile in two seed-propagated artichoke cultivars as affected by microbial inoculants and planting time. Food Chem. 2017, 234, 10–19. [Google Scholar] [CrossRef]
- Ziane, H.; Meddad-Hamza, A.; Beddiar, A.; Gianinazzi, S. Effects of Arbuscular Mycorrhizal Fungi and Fertilization Levels on Industrial Tomato Growth and Production. Int. J. Agric. Biol. 2017, 19, 341–347. [Google Scholar] [CrossRef]
- Bitterlich, M.; Rouphael, Y.; Graefe, J.; Franken, P. Arbuscular Mycorrhizas: A Promising Component of Plant Production Systems Provided Favorable Conditions for Their Growth. Front. Plant Sci. 2018, 9, 1329. [Google Scholar] [CrossRef]
- Rouphael, Y.; Colla, G. Synergistic bio stimulatory action: Designing the next generation of plant bio stimulants for sustainable agriculture. Front Plant Sci. 2018, 9, 1655. [Google Scholar] [CrossRef]
- Candido, V.; Campanelli, G.; D’Addabbo, T.; Castronuovo, D.; Perniola, M.; Camele, I. Growth and yield promoting effect of artificial mycorrhization on field tomato at different irrigation regimes. Sci. Hortic. 2015, 187, 35–43. [Google Scholar] [CrossRef]
- Nzanza, B.; Marais, D.; Soundy, P. Effect of arbuscular mycorrhizal fungal inoculation and biochar amendment on growth and yield of tomato. Int. J. Agric. Biol. 2012, 14, 965–969. [Google Scholar]
- Colella, T.; Candido, V.; Campanelli, G.; Camele, I.; Battaglia, D. Effect of irrigation regimes and artificial mycorrhization on insect pest infestations and yield in tomato crop. Phytoparasitica 2014, 42, 235–246. [Google Scholar] [CrossRef]
- Fujita, M.; Kusajima, M.; Fukagawa, M.; Okumura, Y.; Nakajima, M.; Akiyama, K.; Asami, T.; Yoneyama, K.; Kato, H.; Nakashita, H. Response of tomatoes primed by mycorrhizal colonization to virulent and avirulent bacterial pathogens. Sci. Rep. 2022, 12, 4686. [Google Scholar] [CrossRef] [PubMed]
- Volpe, V.; Chitarra, W.; Cascone, P.; Volpe, M.G.; Bartolini, P.; Moneti, G.; Pieraccini, G.; Di Serio, C.; Maserti, B.; Guerrieri, E.; et al. The Association with Two Different Arbuscular Mycorrhizal Fungi Differently Affects Water Stress Tolerance in Tomato. Front. Plant Sci. 2018, 9, 1480. [Google Scholar] [CrossRef] [PubMed]
- Al-Karaki, G.N. Nursery inoculation of tomato with arbuscular mycorrhizal fungi and subsequent performance under irrigation with saline water. Sci. Hortic. 2006, 109, 1–7. [Google Scholar] [CrossRef]
- Song, Y.; Chen, D.; Lu, K.; Sun, Z.; Zeng, R. Enhanced tomato disease resistance primed by arbuscular mycorrhizal fungus. Front. Plant Sci. 2015, 6, 786. [Google Scholar] [CrossRef]
- Hashem, A.; Akhter, A.; Alqarawi, A.A.; Singh, G.; Almutairi, K.F.; Abd_Allah, E.F. Mycorrhizal fungi induced activation of tomato defense system mitigates Fusarium wilt stress. Saudi J. Biol. Sci. 2021, 28, 5442–5450. [Google Scholar] [CrossRef]
- Chitarra, W.; Pagliarani, C.; Maserti, B.; Lumini, E.; Siciliano, I.; Cascone, P.; Schubert, A.; Gambino, G.; Balestrini, R.; Guerrieri, E. Insights on the Impact of Arbuscular Mycorrhizal Symbiosis on Tomato Tolerance to Water Stress. Plant Physiol. 2016, 171, 1009–1023. [Google Scholar] [CrossRef]
- Caradonia, F.; Francia, E.; Morcia, C.; Ghizzoni, R.; Moulin, L.; Terzi, V.; Ronga, D. Arbuscular Mycorrhizal Fungi and Plant Growth Promoting Rhizobacteria Avoid Processing Tomato Leaf Damage during Chilling Stress. Agronomy 2019, 9, 299. [Google Scholar] [CrossRef]
- Frey-Klett, P.; Garbaye, J.; Tarkka, M. The mycorrhiza helper bacteria revisited. New Phytol. 2007, 176, 22–36. [Google Scholar] [CrossRef]
- Makoboko, M.M.; Bertling, I.; Du Plooy, D. Arbuscular mycorrhiza has limited effects on yield and quality of tomatoes grown under soilless cultivation. Acta Agric. Scand. Sect. B–Soil Plant Sci. 2013, 63, 261–270. [Google Scholar]
- Cwala, Y.; Laubscher, C.P.; Ndakidemi, P.A.; Meyer, A.H. Mycorrhizal root colonization and the subsequent host plant response of soil less grown tomato plants in the presence and absence of the mycorrhizal stimulant, Mycotech. Afr. J. Microbiol. Res. 2010, 4, 414–419. [Google Scholar]
- Kowalska, I.; Konieczny, A.; Gastol, M.; Sady, W.; Hanus-Fajerska, E. Effect of mycorrhiza and phosphorus content in nutrient solution on the yield and nutritional status of tomato plants grown on rockwool or coconut coir. Agric. Food Sci. 2015, 24, 39–51. [Google Scholar] [CrossRef]
- Treseder, K.K. The extent of mycorrhizal colonization of roots and its influence on plant growth and phosphorus content. Plant Soil 2013, 371, 1–13. [Google Scholar] [CrossRef]
- Vierheilig, H.; Piché, Y. A modified procedure for staining arbuscular mycorrhizal fungi in roots. Z. Pflanz. Bodenkd. 1998, 161, 601–602. [Google Scholar] [CrossRef]
- Stoian, V.; Florian, V. Mycorrhiza–Benefits, Influence, Diagnostic Method. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca. Agric. 2009, 66, 2009. [Google Scholar] [CrossRef]
- Luo, X.; Shi, S.; Liu, Y.; Yang, H.; Li, N.; Dong, Z.; Zhu, B.; He, X. Arbuscular mycorrhizal fungal communities of topsoil and subsoil of an annual maize-wheat rotation after 15-years of differential mineral and organic fertilization. Agric. Ecosyst. Environ. 2021, 315, 107442. [Google Scholar] [CrossRef]
- Stoian, V.; Vidican, R.; Crişan, I.; Puia, C.; Şandor, M.; Stoian, V.A.; Păcurar, F.; Vaida, I. Sensitive approach and future perspectives in microscopic patterns of mycorrhizal roots. Sci. Rep. 2019, 9, 10233. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 9. Available online: https://palaeo-electronica.org/2001_1/past/issue1_01.htm (accessed on 12 March 2022).

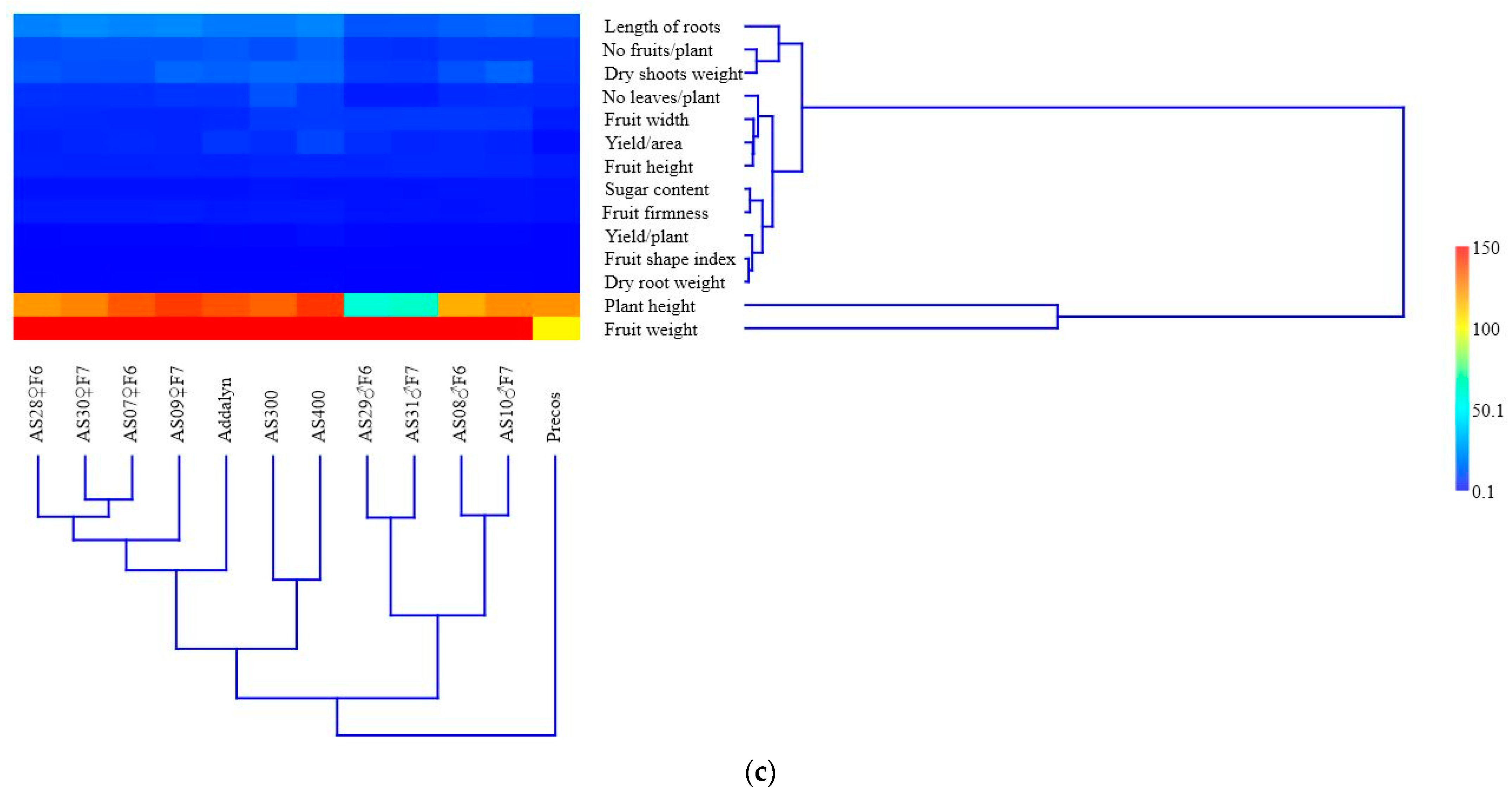
| Elements and Units of Measurement | Before Culture | After Culture | ||||
|---|---|---|---|---|---|---|
| Treatment * | Treatment * | |||||
| T1 | T2 | T3 | T1 | T2 | T3 | |
| pH | 8.02 | 7.51 | 7.70 | 7.91 | 7.58 | 7.84 |
| Humus (%) | 1.62 | 3.88 | 2.98 | 2.64 | 4.31 | 2.38 |
| Nitrogen—N (%) | 0.11 | 0.25 | 0.17 | 0.14 | 0.26 | 0.22 |
| Phosphorus—P (ppm) | 59 | 272 | 211 | 114 | 360 | 264 |
| Potassium—K (ppm) | 154 | 552 | 264 | 266 | 576 | 376 |
| Calcium—Ca (mg/100 g soil) | 6 | 8 | 7 | 9 | 6.4 | 6 |
| Magnesium—Mg (mg/100 g soil) | 1.82 | 4.86 | 4.25 | 1.21 | 2.18 | 3.64 |
| Sodium—Na (mg/100 g soil) | 24.5 | 15.75 | 21.5 | 25 | 17.75 | 16.75 |
| Potassium—K (mg/100 g soil) | 2 | 11 | 3 | 3 | 9 | 5 |
| HCO3 (mg/100 g soil) | 24.4 | 21.35 | 21.35 | 27.45 | 24.4 | 18.3 |
| Chlorine—Cl (mg/100 g soil) | 39.05 | 28.4 | 40.82 | 40.82 | 28.4 | 31.95 |
| SO4 (mg/100 g soil) | 12.8 | 44.8 | 32 | 32 | 38.4 | 32 |
| CaCO3 (%) | 2.2 | 3.2 | 4.8 | 1.8 | 1 | 2.2 |
| Cadmium—Cd (mg/kg) | 12.24 | 13.18 | 21.75 | 8.61 | 12.22 | 21.24 |
| Copper—Cu (mg/kg) | 58.78 | 71.35 | 67.13 | 71.69 | 66.02 | 66.9 |
| Nickel—Ni (mg/kg) | 43.71 | 45.84 | 55.7 | 77.82 | 53.62 | 44.92 |
| Zinc—Zn (mg/kg) | 104.24 | 151.68 | 120.43 | 114.9 | 147.61 | 111.71 |
| Chromium—Cr (mg/kg) | 68.6 | 57.17 | 92.5 | 133.83 | 73.17 | 55.05 |
| Lead—Pb (mg/kg) | 20.63 | 43.4 | 22.65 | 31.86 | 24.35 | 32.66 |
| Manganese—Mn (mg/kg) | 889.91 | 1197.5 | 942.2 | 953.1 | 1201.9 | 902.9 |
| Cobalt—Co (mg/kg) | 4.27 | 23.02 | 1.75 | 8.76 | 22.69 | 16.15 |
| Iron—Fe (ppm) | 143 | 206 | 325 | 156 | 168 | 237 |
| Humidity (%) | 16.48 | 31.02 | 17.16 | 16.96 | 27.76 | 20.68 |
| Salt content (mS) | 0.33 | 0.3 | 0.33 | 0.34 | 0.3 | 0.25 |
| N-NO3 | 0.75 | 2.55 | 0.15 | 0.9 | 1.05 | 1.35 |
| N-NH4 | 0.84 | 0.63 | 0.21 | 0.84 | 0.42 | 0.42 |
| Elements and Units of Measurement. | Before Culture (B) | After Culture (A) | Difference (A-B) (±) | Significance (‘p’ Value) |
|---|---|---|---|---|
| pH | 7.74 ± 0.26 | 7.78 ± 0.17 | 0.04 | 0.698 |
| Humus (%) | 2.83 ± 1.14 | 3.11 ± 1.05 | 0.28 | 0.610 |
| Nitrogen—N (%) | 0.18 ± 0.07 | 0.21 ± 0.06 | 0.03 | 0.122 |
| Phosphorus—P (ppm) | 180.67 ± 109.69 | 246.00 ± 123.98 | 65.33 | 0.029 * |
| Potassium—K (ppm) | 323.33 ± 205.53 | 406.00 ± 157.16 | 82.67 | 0.106 |
| Calcium—Ca (mg/100 g soil) | 7.00 ± 1.00 | 7.13 ± 1.63 | 0.13 | 0.935 |
| Magnesium—Mg (mg/100 g soil) | 3.64 ± 1.61 | 2.34 ± 1.22 | −1.30 | 0.200 |
| Sodium—Na (mg/100 g soil) | 20.58 ± 4.45 | 19.83 ± 4.50 | −0.75 | 0.749 |
| Potassium—K (mg/100 g soil) | 5.33 ± 4.93 | 5.67 ± 3.06 | 0.33 | 0.808 |
| HCO3 (mg/100 g soil) | 22.37 ± 1.76 | 23.38 ± 4.66 | 1.02 | 0.667 |
| Chlorine—Cl (mg/100 g soil) | 36.09 ± 6.72 | 33.72 ± 6.40 | −2.37 | 0.547 |
| SO4 (mg/100 g soil) | 29.87 ± 16.11 | 34.13 ± 3.70 | 4.27 | 0.635 |
| CaCO3 (%) | 3.40 ± 1.31 | 1.67 ± 0.61 | −1.73 | 0.125 |
| Cadmium—Cd (mg/kg) | 15.72 ± 5.24 | 14.02 ± 6.51 | −1.70 | 0.223 |
| Copper—Cu (mg/kg) | 65.75 ± 6.40 | 68.20 ± 3.05 | 2.45 | 0.696 |
| Nickel—Ni (mg/kg) | 48.42 ± 6.40 | 58.79 ± 17.05 | 10.37 | 0.509 |
| Zinc—Zn (mg/kg) | 125.45 ± 24.12 | 124.74 ± 19.87 | −0.71 | 0.914 |
| Chromium—Cr (mg/kg) | 72.76 ± 18.03 | 87.35 ± 41.26 | 14.59 | 0.671 |
| Lead—Pb (mg/kg) | 28.89 ± 12.60 | 29.62 ± 4.58 | 0.73 | 0.948 |
| Manganese—Mn (mg/kg) | 1009.86 ± 164.57 | 1019.31 ± 160.13 | 9.45 | 0.780 |
| Cobalt—Co (mg/kg) | 9.68 ± 11.62 | 15.87 ± 6.97 | 6.19 | 0.290 |
| Iron—Fe (ppm) | 224.67 ± 92.42 | 187.00 ± 43.71 | −37.67 | 0.326 |
| Humidity (%) | 21.55 ± 8.21 | 21.80 ± 5.49 | 0.25 | 0.911 |
| Salt content (mS) | 0.32 ± 0.02 | 0.30 ± 0.05 | −0.02 | 0.499 |
| N–NO3 | 1.15 ± 1.25 | 1.10 ± 0.23 | −0.05 | 0.955 |
| N–NH4 | 0.56 ± 0.32 | 0.56 ± 0.24 | 0.00 | 0.999 |
| Genotypes (G) | Length of Roots (cm) | Mean ± SD (G) | Plant Height (cm) | Mean ± SD (G) | Number of Leaves/Plant | Mean ± SD (G) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T1 | T2 | T3 | T1 | T2 | T3 | ||||
| Precos | 16.5 ab | 16.4 ab | 17.4 a–c | 16.8 ± 0.6 A | 114.0 d | 122.0 f–i | 129.5 j–m | 121.8 ± 7.8 BC | 8.0 b–d | 9.0 c–e | 10.0 e–g | 9.0 ± 1.0 B |
| AS28♀F6 | 24.4 g–k | 24.6 g–k | 26.7 k–m | 25.2 ± 1.3 CD | 114.0 d | 121.0 e–h | 127.0 i–l | 120.7 ± 6.5 BC | 9.0 c–e | 10.5 e–h | 11.0 f–i | 10.2 ± 1.0 CD |
| AS29♂F6 | 15.8 ab | 15.7 ab | 18.3 b–d | 16.6 ± 1.5 A | 56.0 a | 56.0 a | 60.5 ab | 57.5 ± 2.6 A | 6.0 a | 6.5 ab | 6.5 ab | 6.5 ± 0.3 A |
| AS30♀F7 | 25.6 i–l | 27.5 lm | 29.2 m | 27.4 ± 1.8 D | 120.0 e–h | 123.5 g–i | 129.5 j–m | 124.3 ± 4.8 CD | 9.0 c–e | 9.5 d–f | 11.0 f–i | 9.8 ± 1.0 BC |
| AS31♂F7 | 15.1 a | 15.9 ab | 19.3 cd | 16.8 ± 2.2 A | 57.5 a | 59.5 ab | 63.5 b | 60.2 ± 3.1 A | 6.0 a | 6.0 a | 7.5 a–c | 6.5 ± 0.9 A |
| AS300 | 22.5 f–h | 24.0 g–j | 25.1 h–l | 23.9 ± 1.3 C | 115.5 de | 132.0 lm | 144.0 no | 130.5 ± 14.3 DE | 10.5 e–h | 12.0 h–j | 12.5 i–j | 16.7 ± 1.0 EF |
| Addalyn | 23.3 g–i | 23.2 g–i | 24.8 g–k | 23.8 ± 0.9 C | 124.0 g–j | 134.5 m | 143.5 no | 134.0 ± 9.8 EF | 10.5 e–h | 10.0 e–g | 12.0 h–j | 10.8 ± 1.0 DE |
| AS07♀F6 | 24.3 g–k | 25.5 i–l | 27.7 lm | 25.8 ± 1.7 CD | 125.5 h–k | 133.0 m | 140.5 n | 133.0 ± 7.5 EF | 9.5 d–f | 10.5 e–h | 10.0 e–g | 10.0 ± 0.5 CD |
| AS08♂F6 | 17.5 a–c | 17.8 bc | 20.7 d–f | 18.7 ± 1.8 AB | 107.0 c | 117.0 d–f | 124.5 g–k | 116.2 ± 8.8 B | 9.0 c–e | 9.0 c–e | 9.0 c–e | 9.0 ± 0.0 B |
| AS09♀F7 | 25.2 i–l | 26.2 j–l | 28.9 m | 26.8 ± 1.9 D | 125.0 g–k | 140.0 n | 149.0 o | 138.0 ± 12.1 F | 9.5 d–f | 10.5 e–h | 12.5 i–j | 10.8 ± 1.5 DE |
| AS10♂F7 | 18.1 bc | 19.8 c–e | 22.2 e–g | 20.0 ± 2.1 B | 119.0 d–g | 119.0 d–g | 130.0 k–m | 122.7 ± 6.4 BC | 9.5 d–f | 9.5 d–f | 9.5 d–f | 9.5 ± 0.0 BC |
| AS400 | 23.9 g–j | 25.8 i–l | 27.5 lm | 25.8 ± 1.8C D | 130.0 k–m | 140.0 n | 148.0 o | 139.3 ± 9.0 F | 11.0 f–i | 11.5 g–j | 13.0 j | 11.8 ± 1.0 F |
| Mean ± SD (T) | 21.0 ± 4.0 X | 21.9 ± 4.4 Y | 24.0 ± 4.2 Z | - | 108.9 ± 25.2 X | 116.5 ± 28.5 Y | 124.1 ± 30.2 Z | - | 8.9 ± 1.6 X | 9.5 ± 1.8 Y | 10.4 ± 2.0 Z | - |
| Genotypes (G) | Dry Root Weight (g) | Mean ± SD (G) | Dry Shoot Weight (g) | Mean ± SD (G) | ||||
|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T1 | T2 | T3 | |||
| Precos | 0.66 c | 0.66 c | 0.81 cd | 0.71 ± 0.09 B | 10.5 a | 10.7 a | 11.2 ab | 10.8 ± 0.4 A |
| AS28♀F6 | 1.29 e | 1.43 e–g | 1.32 ef | 1.35 ± 0.07 C | 16.2 ef | 17.2 f–h | 17.8 g–i | 17.1 ± 0.8 B |
| AS29♂F6 | 0.37 ab | 0.41 b | 0.38 ab | 0.39 ± 0.02 A | 12.4 b | 11.1 ab | 11.9 ab | 11.8 ± 0.7 A |
| AS30♀F7 | 0.87 d | 0.71 c | 0.78 cd | 0.79 ± 0.08 B | 14.7 cd | 16.1 d–f | 16.1 d–f | 15.7 ± 0.8 B |
| AS31♂F7 | 0.26 a | 0.38 ab | 0.32 ab | 0.32 ± 0.06 A | 10.7 a | 11.7 ab | 12.2 b | 11.7 ± 0.8 A |
| AS300 | 1.70 j–l | 1.83 lm | 1.90 mn | 1.81 ± 0.10 F | 19.0 i–l | 20.6 m | 20.9 m | 20.2 ± 1.0 C |
| Addalyn | 1.59 h–j | 1.70 j–l | 1.71 j–l | 1.67 ± 0.07 E | 18.1 h–j | 19.9 k–m | 19.0 i–l | 19.0 ± 0.9 C |
| AS07♀F6 | 1.44 f–h | 1.48 g–i | 1.60 ij | 1.51 ± 0.08 D | 14.5 c | 15.5 c–e | 16.3 ef | 15.4 ± 0.9 B |
| AS08♂F6 | 1.56 g–j | 1.68 jk | 1.46 f–i | 1.57 ± 0.11 DE | 16.1 d–f | 16.6 e–g | 16.0 d–f | 16.2 ± 0.3 B |
| AS09♀F7 | 2.00 no | 2.07 o | 2.23 p | 2.10 ± 0.12 G | 20.8 m | 18.6 i–k | 20.9 m | 20.1 ± 1.3 C |
| AS10♂F7 | 1.80 k–m | 1.90 mn | 2.04 no | 1.91 ± 0.12 F | 19.1 i–l | 19.8 k–m | 19.1 i–l | 19.4 ± 0.4 C |
| AS400 | 2.06 o | 2.07 o | 2.37 q | 2.17 ± 0.18 G | 19.2 j–l | 20.1 lm | 21.1 m | 20.2 ± 1.0 C |
| Mean ± SD (T) | 1.30 ± 0.62 X | 1.36 ± 0.64 Y | 1.41 ± 0.70 Z | - | 15.97 ± 3.5 X | 16.53 ± 3.6 Y | 16.90 ± 3.6 Z | - |
| Genotypes (G) | Fruit Height (cm) | Mean ± SD (G) | Fruit Width (cm) | Mean ± SD (G) | Fruit Shape Index | Mean ± SD (G) | Fruit Weight (g) | Mean ± SD (G) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T1 | T2 | T3 | T1 | T2 | T3 | T1 | T2 | T3 | |||||
| Precos | 6.5 ab | 6.0 a | 6.5 ab | 6.3 ± 0.3 A | 6.5 a | 6.5 a | 7.0 ab | 6.7 ± 0.3 A | 1.00 k | 0.93 i–k | 0.93 i–k | 0.95 ± 0.04 D | 100.0 a | 92.5 a | 112.0 b | 101.5 ± 9.8 A |
| AS28♀F6 | 7.7 c–f | 8.2 e–g | 7.0 a–d | 7.6 ± 0.6 BC | 9.2 e | 8.5 c–e | 8.0 b–d | 8.6 ± 0.6 B | 0.83 d–j | 0.97 jk | 0.88 f–k | 0.89 ± 0.07 CD | 154.5 c | 156.0 c | 160.5 c | 157.0 ± 3.1 B |
| AS29♂F6 | 8.0 d–g | 7.7 c–f | 7.5 b–f | 7.7 ± 0.3 C | 11.2 gh | 11.0 gh | 11.0 gh | 11.1 ± 0.1 C | 0.71 a–e | 0.70 a–e | 0.68 a–c | 0.70 ± 0.02 A | 237.0 j–l | 240.0 kl | 248.5 l | 241.8 ± 6.0 F |
| AS30♀F7 | 8.0 d–g | 6.7 a–c | 6.7 a–c | 7.1 ± 0.8 B | 9.5 ef | 7.5 a–c | 8.0 b–d | 8.3 ± 1.0 B | 0.84 e–j | 0.90 g–k | 0.84 e–j | 0.86 ± 0.03 CD | 154.5 c | 160.0 c | 164.5 c | 159.7 ± 5.0 B |
| AS31♂F7 | 8.5 fg | 8.0 d–g | 8.5 fg | 8.3 ± 0.3 D | 11.2 gh | 11.0 gh | 11.5 g–i | 11.2 ± 0.3 CD | 0.76 b–g | 0.73 a–e | 0.74 a–f | 0.74 ± 0.02 AB | 225.0 h–j | 230.5 i–k | 238.5 j–l | 231.3 ± 6.8 EF |
| AS300 | 8.2 e–g | 7.7 c–f | 8.0 d–g | 8.0±0.3CD | 10.5 fg | 11.7 hi | 11.5 g–i | 11.2 ± 0.6 CD | 0.78 b–h | 0.66 ab | 0.69 a–d | 0.71 ± 0.06 A | 199.0 de | 192.0 d | 214.5 f–h | 201.8 ± 11.5 C |
| Addalyn | 7.0 a–d | 7.2 b–e | 7.2 b–e | 7.1 ± 0.1 B | 7.2 ab | 9.2 e | 9.5 ef | 8.6 ± 1.3 B | 0.96 jk | 0.78 b–h | 0.76 b–g | 0.83 ± 0.11 BC | 191.0 d | 198.0 de | 228.5 i–k | 205.8 ± 19.9 C |
| AS07♀ F6 | 7.7 c–f | 7.0 a–d | 6.7 a–c | 7.1 ± 0.5 B | 9.2 e | 7.5 a–c | 7.5 a–c | 8.1 ± 1.0 B | 0.84 e–j | 0.94 i–k | 0.90 g–k | 0.89 ± 0.05 CD | 161.5 c | 158.5 c | 160.5 c | 160.2 ± 1.5 B |
| AS08♂F6 | 9.0 g | 8.5fg | 7.7 c–f | 8.4 ± 0.7 D | 11.0 gh | 12.0 hi | 11.5 g–i | 11.5 ± 0.5 CD | 0.82 c–i | 0.71 a–e | 0.67 ab | 0.73 ± 0.08 A | 225.0 h–j | 221.0 g–i | 230.5 i–k | 225.5 ± 4.8 E |
| AS09♀F7 | 8.2e–g | 7.5 b–f | 7.0 a–d | 7.6 ± 0.6 BC | 9.0 de | 7.5 a–c | 7.5 a–c | 8.0 ± 0.9 B | 0.91 h–k | 1.00 k | 0.94i–k | 0.95 ± 0.05 D | 163.0 c | 153.0 c | 156.0 c | 157.3 ± 5.1 B |
| AS10♂F7 | 8.2 e–g | 7.7 c–f | 8.2 e–g | 8.0 ± 0.3 CD | 11.0 gh | 11.2 gh | 11.0 gh | 11.1 ± 0.1 C | 0.75 a–f | 0.69 a–c | 0.75 a–f | 0.73 ± 0.03 A | 212.5 f–h | 222.5 g–i | 229.0 i–k | 221.3 ± 8.3 DE |
| AS400 | 8.2 e–g | 7.7 c–f | 7.7 c–f | 7.9 ± 0.3 CD | 11.2 gh | 11.7 hi | 12.5 i | 11.8 ± 0.7 D | 0.73 a–f | 0.66 ab | 0.62 a | 0.67 ± 0.06 A | 203.5 d–f | 209.5 e–g | 220.5 g–i | 211.2 ± 8.6 CD |
| Mean ± SD (T) | 7.9 ± 0.7 X | 7.5 ± 0.7 Y | 7.4 ± 0.6 Z | - | 9.7 ± 1.6 X | 9.6 ± 2.0 Y | 9.7 ± 2.0 Z | - | 0.83 ± 0.09 X | 0.81 ± 0.13 Y | 0.78 ± 0.11 Z | - | 185.5 ± 39.7 X | 186.1 ± 43.0 Y | 197.0 ± 43.7 Z | - |
| Genotypes (G) | Number of Fruit/Plants | Mean ± SD (G) | Yield/Plant (kg) | Mean ± SD (G) | Yield/Area (kg) | Mean ± SD (G) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T1 | T2 | T3 | T1 | T2 | T3 | ||||
| Precos | 11.5 a–c | 12.0 a–d | 10.5 ab | 11.3 ± 0.8 AB | 1.25 a | 1.35 a | 1.20 a | 1.3 ± 0.1 A | 4.13 a | 4.45 a | 3.96 a | 4.18 ± 0.25 A |
| AS28♀F6 | 16.0 e–i | 14.5 c–f | 15.0 d–g | 15.2 ± 0.8 C | 2.35 b–d | 2.05 b | 2.20 bc | 2.2 ± 0.2 B | 7.76 b–d | 6.77 b | 7.26 bc | 7.26±0.50 B |
| AS29♂F6 | 9.5 a | 10.5 ab | 11.5 a–c | 10.5 ± 1.0 AB | 2.95 d–g | 2.70 b–f | 3.30 fg | 3.0 ± 0.3 D | 9.73 d–g | 8.91 b–f | 10.89 fg | 9.84 ± 0.99 D |
| AS30♀F7 | 18.0 g–i | 16.5 f–i | 14.5 c–f | 16.3 ± 1.8 CD | 2.60 b–f | 2.45 b–d | 2.35 b–d | 2.5 ± 0.1 BC | 8.58 b–f | 8.09 b–d | 7.76 b–d | 8.14 ± 0.41 BC |
| AS31♂F7 | 10.0 ab | 10.0ab | 10.0 ab | 10.0 ± 0.0 A | 2.40 b–d | 2.20 bc | 2.70 b–f | 2.4 ± 0.3 BC | 7.92 b–d | 7.26 bc | 8.91 b–f | 8.03 ± 0.83 BC |
| AS300 | 16.0 e–i | 15.5 e–h | 15.5 e–h | 15.7 ± 0.3 C | 2.95 d–g | 2.80 c–f | 2.95 d–g | 2.9 ± 0.1 D | 9.73 d–g | 9.24 c–f | 9.73 d–g | 9.57 ± 0.28 D |
| Addalyn | 17.0 f–i | 17.5 f–i | 18.0 g–i | 17.5 ± 0.5 DE | 3.25 e–g | 3.25 e–g | 3.65 gh | 3.4 ± 0.2 E | 10.73 e–g | 10.73 e–g | 12.05 gh | 11.17 ± 0.76 E |
| AS07♀F6 | 17.0 f–i | 17.0 f–i | 16.0 e–i | 16.7 ± 0.6 CD | 2.55 b–e | 2.95 d–g | 2.25 b–d | 2.6 ± 0.4 C | 8.41 b–e | 9.73 d–g | 7.43 b–d | 8.52 ± 1.15 C |
| AS08♂F6 | 12.0 a–d | 13.0 b–e | 11.0 ab | 12.0 ± 1.0 B | 2.25 b–d | 2.85 c–f | 2.60 b–f | 2.6 ± 0.3 C | 7.43 b–d | 9.41 c–f | 8.58 b–f | 8.47 ± 0.99 C |
| AS09♀F7 | 16.5 f–i | 16.5 f–i | 15.0 d–g | 16.0 ± 0.9 CD | 2.20 bc | 2.70 b–f | 2.30 b–d | 2.4 ± 0.3 BC | 7.26 bc | 8.91 b–f | 7.59 b–d | 7.92 ± 0.87 BC |
| AS10♂F7 | 12.0 a–d | 11.0 ab | 12.0 a–d | 11.7 ± 0.6 AB | 2.25 b–d | 2.20 bc | 2.40 b–d | 2.3 ± 0.1 BC | 7.43 b–d | 7.26 bc | 7.92 b–d | 7.54 ± 0.34 BC |
| AS400 | 19.0 i | 19.0 i | 18.5 hi | 18.8 ± 0.3 E | 4.05 hi | 4.35 i | 4.15 hi | 4.2 ± 0.2 F | 13.32 hi | 14.35 i | 13.69 hi | 13.79 ± 0.52 F |
| Mean ± SD (T) | 14.5 ± 3.3 X | 14.4 ± 3.0 Y | 13.9 ± 2.9 Z | - | 2.59 ± 0.68 X | 2.65 ± 0.73 Y | 2.67 ± 0.77 Z | - | 8.54 ± 2.24 X | 8.76 ± 2.42 Y | 8.81 ± 2.54 Z | |
| Genotypes (G) | Fruit Firmness (kg/cm2) | Mean ± SD (G) | Sugar Content (°Brix) | Mean ± SD (G) | ||||
|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T1 | T2 | T3 | |||
| Precos | 4.5 a–c | 4.0 a | 4.7 b–d | 4.4 ± 0.4 A | 4.6 ab | 4.6 ab | 5.0 c–g | 4.7 ± 0.2 A–C |
| AS28♀F6 | 5.7 f–h | 6.0 g–i | 6.5 i–k | 6.1 ± 0.4 CD | 4.6 ab | 4.6 ab | 4.7 ab | 4.6 ± 0.1 AB |
| AS29♂F6 | 4.7 b–d | 4.7 b–d | 5.0 c–e | 4.8 ± 0.2 A | 4.8 a–d | 4.7 ab | 5.2 f–h | 4.9 ± 0.3 B–D |
| AS30♀F7 | 7.0 k | 5.7 f–h | 6.2 h–j | 6.3 ± 0.7 CD | 4.6 ab | 4.6 ab | 4.7 a–c | 4.6 ± 0.1 A–C |
| AS31♂F7 | 5.0 c–e | 4.7 b–d | 5.2 d–f | 5.0 ± 0.3 AB | 4.6 ab | 4.5 a | 4.8 a–c | 4.6 ± 0.2 A–C |
| AS300 | 6.0 g–i | 6.0 g–i | 6.5 i–k | 6.2 ± 0.3 CD | 5.2 f–h | 5.3 gh | 5.4 h | 5.3 ± 0.1 E |
| Addalyn | 5.5 e–g | 5.5 e–g | 6.0 g–i | 5.7 ± 0.3 BC | 4.6 ab | 4.7 a–c | 4.7 a–c | 4.7 ± 0.1 A–C |
| AS07♀F6 | 6.2 h–j | 6.0 g–i | 6.2 h–j | 6.1 ± 0.1 CD | 4.5 a | 4.6 ab | 4.7 ab | 4.6 ± 0.1 AB |
| AS08♂F6 | 5.0 c–e | 4.2 ab | 4.7 b–d | 4.6 ± 0.4 A | 5.3 gh | 4.8 a–c | 5.1 d–h | 5.1 ± 0.3 DE |
| AS09♀F7 | 6.5 i–k | 6.2 h–j | 7.0 k | 6.6 ± 0.4 D | 4.6 ab | 4.6 ab | 4.5 a | 4.6 ± 0.1 A |
| AS10♂F7 | 5.2 d–f | 4.7 b–d | 5.0 c–e | 5.0 ± 0.3 AB | 5.1 d–h | 5.0 c–g | 5.2 e–h | 5.1 ± 0.1 DE |
| AS400 | 6.7 jk | 6.0 g–i | 6.0 g–i | 6.2 ± 0.4 CD | 4.9 a–e | 4.9 b–f | 5.0 c–g | 4.9 ± 0.1 CD |
| Mean ± SD (T) | 5.7 ± 0.8 X | 5.3 ± 0.8 Y | 5.8 ± 0.8 Z | - | 4.8 ± 0.3 X | 4.7 ± 0.2 Y | 4.9 ± 0.3 Z | - |
| Genotypes/ Treatment (G/T) | Frequency (%) | Intensity (%) | Non-Mycorrhizal Areas (%) | Degree of Colonization % | Ratio of Mycorrhizal Areas/Non-Mycorrhizal Areas | |
|---|---|---|---|---|---|---|
| Precos | T1 | 26.0 ± 14.2 a–e | 9.3 ± 2.5 b–h | 90.6 ± 2.5 e–k | 2.6 ± 2.1 a–e | 0.11 ± 0.03 a–g |
| T2 | 10.7 ± 4.6 a–c | 5.0 ± 0.6 a–b | 95.6 ± 0.6 k–l | 0.4 ± 0.3 a | 0.04 ± 0.01 ab | |
| T3 | 32.7 ± 11.4 c–h | 7.0 ± 4.4 a–e | 92.0 ± 4.4 g–l | 2.8 ± 2.3 a–f | 0.09 ± 0.05 a–f | |
| AS28♀F6 | T1 | 13.0 ± 1.7 a–c | 16.6 ± 1.0 jk | 95.0 ± 1.0 j–l | 0.6 ± 0.1 ab | 0.05 ± 0.01 a–c |
| T2 | 14.0 ± 5.3 a–c | 12.6 ± 1.0 e–k | 93.0 ± 1.0 h–l | 0.9 ± 0.4 ab | 0.08 ± 0.01 a–e | |
| T3 | 43.7 ± 21.8 d–i | 15.0 ± 3.2 h–k | 83.3 ± 3.2 bc | 7.7 ± 4.7 g–i | 0.20 ± 0.05 i–k | |
| AS29♂F6 | T1 | 41.3 ± 7.1 d–i | 16.6 ± 3.1 jk | 87.3 ± 3.1 b–h | 5.3 ± 2.2 b–i | 0.15 ± 0.04 e–k |
| T2 | 41.7 ± 18.9 d–i | 13.3 ± 3.0 f–k | 85.0 ± 3.0 b–e | 8.3 ± 1.8 hi | 0.18 ± 0.04 g–k | |
| T3 | 46.7 ± 17.6 e–i | 16.6 ± 5.5 jk | 83.3 ± 5.5 bc | 8.3 ± 5.6 hi | 0.20 ± 0.08 jk | |
| AS30♀F7 | T1 | 54.7 ± 6.1 hi | 13.3 ± 1.5 f–k | 86.6 ± 1.5 b–g | 7.3 ± 1.5 e–i | 0.16 ± 0.02 f–k |
| T2 | 44.3 ± 17.8 d–i | 14.0 ± 4.0 g–k | 86.0 ± 4.0 b–f | 6.6 ± 4.2 c–i | 0.16 ± 0.04 f–k | |
| T3 | 80.3 ± 0.6 j | 24.6 ± 5.9 l | 75.3 ± 5.9 a | 19.8 ± 4.7 j | 0.33 ± 0.10 l | |
| AS31♂F7 | T1 | 24.3 ± 8.0 a–d | 8.0 ± 2.0 a–f | 92.0 ± 2.0 g–l | 2.0 ± 1.1 a–c | 0.09 ± 0.02 a–f |
| T2 | 13.3 ± 9.5 a–c | 7.0 ± 2.6 a–e | 93.0 ± 2.6 h–l | 1.1 ± 0.5 ab | 0.07 ± 0.03 a–e | |
| T3 | 13.3 ± 9.5 a–c | 5.0 ± 1.0 a–c | 95.0 ± 1.0 j–l | 0.5 ± 0.3 ab | 0.05 ± 0.01 a–c | |
| AS300 | T1 | 25.3 ± 8.1 a–e | 6.0 ± 1.0 a–d | 94.0 ± 1.0 i–l | 1.6 ± 0.7 ab | 0.06 ± 0.01 a–d |
| T2 | 30.7 ± 9.2 c–g | 11.0 ± 2.6 d–i | 89.0 ± 2.6 d–i | 3.5 ± 1.7 a | 0.13 ± 0.03 c–j | |
| T3 | 6.0 ± 5.3 a | 2.6 ± 1.2 a | 97.3 ± 1.2 l | 0.2 ± 0.1 a–g | 0.03 ± 0.01 a | |
| Addalyn | T1 | 60.3 ± 13.9 i | 12.0 ± 2.6 e–j | 88.0 ± 2.6 c–h | 7.4 ± 3.2 f–i | 0.14 ± 0.03 d–j |
| T2 | 14.0 ± 6.9 a–c | 5.3 ± 1.2 a–d | 94.6 ± 1.2 i–l | 0.8 ± 0.2 ab | 0.05 ± 0.01 a–c | |
| T3 | 23.0 ± 8.7 a–d | 5.3 ± 4.2 a–d | 95.6 ± 4.2 i–l | 2.0 ± 0.5 a–c | 0.06 ± 0.05 a–d | |
| AS07♀F6 | T1 | 42.0 ± 15.9 d–i | 16.3 ± 2.1 i–k | 83.6 ± 2.1 b–d | 6.9 ± 3.3 d–i | 0.19 ± 0.03 h–k |
| T2 | 8.0 ± 6.9 ab | 3.0 ± 1.0 a | 97.0 ± 1.0 l | 0.2 ± 0.1 a | 0.03 ± 0.01 a | |
| T3 | 44.6 ± 14.2 d–i | 10.3 ± 2.5 c–h | 89.6 ± 2.5 e–j | 4.8 ± 2.7 a–i | 0.12 ± 0.03 b–h | |
| AS08♂F6 | T1 | 48.6 ± 8.1 f–i | 13.3 ± 4.9 f–k | 86.6 ± 4.9 b–g | 6.5 ± 2.7 c–i | 0.15 ± 0.07 e–k |
| T2 | 23.8 ± 1.5 a–d | 9.0 ± 2.0 b–g | 92.0 ± 2.0 f–k | 2.2 ± 1.5 a–d | 0.10 ± 0.02 a–g | |
| T3 | 52.7 ± 12.1 g–i | 17.6 ± 2.5 k | 82.3 ± 2.5 b | 9.4 ± 3.0 i | 0.22 ± 0.04 k | |
| AS09♀F7 | T1 | 42.0 ± 7.2 d–i | 9.6 ± 1.5 b–h | 90.3 ± 1.5 e–k | 4.3 ± 1.3 a–h | 0.11 ± 0.02 a–g |
| T2 | 12.3 ± 4.9 a–c | 4.6 ± 1.5 a–c | 95.3 ± 1.5 j–l | 0.6 ± 0.4 ab | 0.05 ± 0.02 a–c | |
| T3 | 12.6 ± 10.0 a–c | 5.0 ± 2.0 a–c | 95.0 ± 2.0 j–l | 0.7 ± 0.2 ab | 0.05 ± 0.02 a–c | |
| AS10♂F7 | T1 | 39.3 ± 9.0 d–i | 10.0 ± 2.0 b–h | 90.0 ± 2.0 e–l | 4.0 ± 1.7 a–h | 0.11 ± 0.02 b–g |
| T2 | 29.0 ± 17.3 b–f | 12.0 ± 3.6 e–j | 88.0 ± 3.6 c–h | 3.4 ± 1.9 a–g | 0.14 ± 0.05 d–j | |
| T3 | 17.3 ± 6.4 a–c | 7.3 ± 2.1 a–e | 92.6 ± 2.1 h–l | 1.3 ± 0.7 ab | 0.08 ± 0.02 a–e | |
| AS400 | T1 | 15.0 ± 5.0 a–c | 5.0 ± 0.0 a–c | 95.0 ± 0.1 j–l | 0.7 ± 0.3 ab | 0.05 ± 0.00 a–c |
| T2 | 39.0 ± 5.2 d–i | 11.0 ± 2.0 d–i | 89.0 ± 2.0 d–i | 4.2 ± 0.9 a–h | 0.12 ± 0.03 b–i | |
| T3 | 15.3 ± 3.1 a–c | 9.0 ± 2.0 b–g | 91.0 ± 2.0 f–k | 1.4 ± 0.5 ab | 0.10 ± 0.02 a–g | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Felföldi, Z.; Vidican, R.; Stoian, V.; Roman, I.A.; Sestras, A.F.; Rusu, T.; Sestras, R.E. Arbuscular Mycorrhizal Fungi and Fertilization Influence Yield, Growth and Root Colonization of Different Tomato Genotype. Plants 2022, 11, 1743. https://doi.org/10.3390/plants11131743
Felföldi Z, Vidican R, Stoian V, Roman IA, Sestras AF, Rusu T, Sestras RE. Arbuscular Mycorrhizal Fungi and Fertilization Influence Yield, Growth and Root Colonization of Different Tomato Genotype. Plants. 2022; 11(13):1743. https://doi.org/10.3390/plants11131743
Chicago/Turabian StyleFelföldi, Zoltán, Roxana Vidican, Vlad Stoian, Ioana A. Roman, Adriana F. Sestras, Teodor Rusu, and Radu E. Sestras. 2022. "Arbuscular Mycorrhizal Fungi and Fertilization Influence Yield, Growth and Root Colonization of Different Tomato Genotype" Plants 11, no. 13: 1743. https://doi.org/10.3390/plants11131743
APA StyleFelföldi, Z., Vidican, R., Stoian, V., Roman, I. A., Sestras, A. F., Rusu, T., & Sestras, R. E. (2022). Arbuscular Mycorrhizal Fungi and Fertilization Influence Yield, Growth and Root Colonization of Different Tomato Genotype. Plants, 11(13), 1743. https://doi.org/10.3390/plants11131743












