Variation of Residual Sexuality Rates along Reproductive Development in Apomictic Tetraploids of Paspalum
Abstract
1. Introduction
2. Results
2.1. Proportions of Sexual and Aposporic Megagametophytes in Mature Ovules
2.2. Reproductive Origin of Seeds
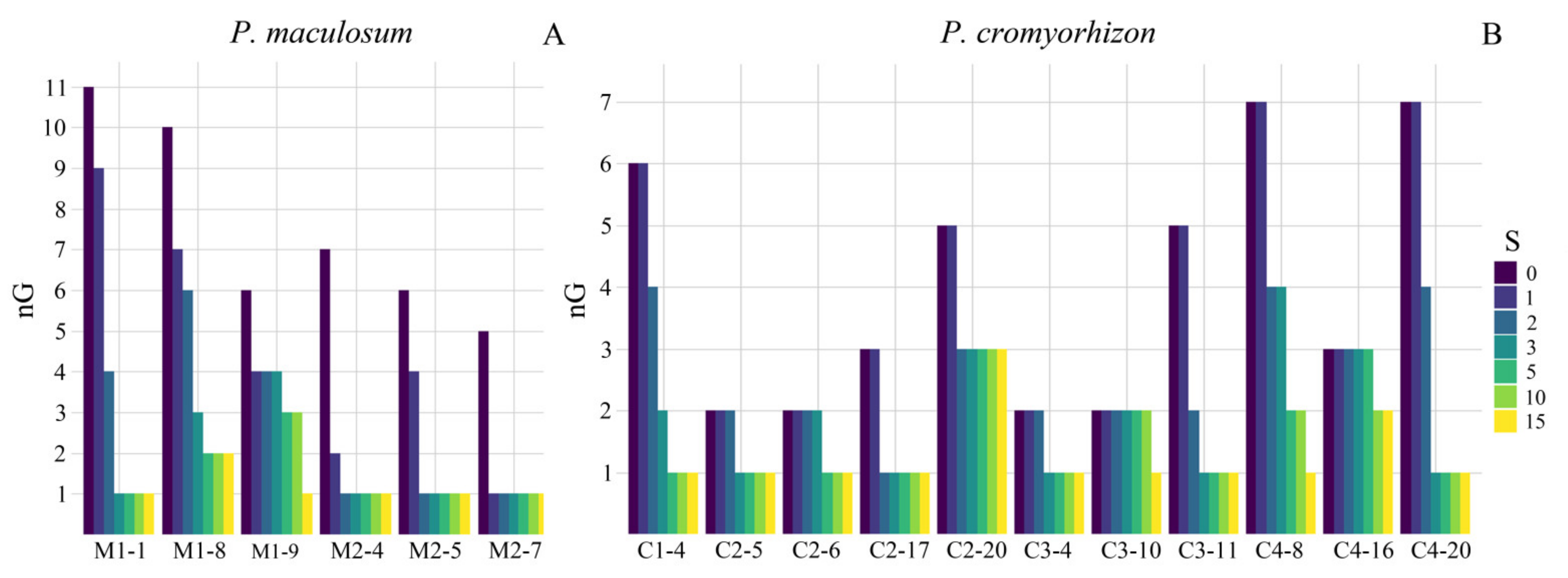
2.3. Proportions of Residual Sexuality Assessed by Progeny Tests
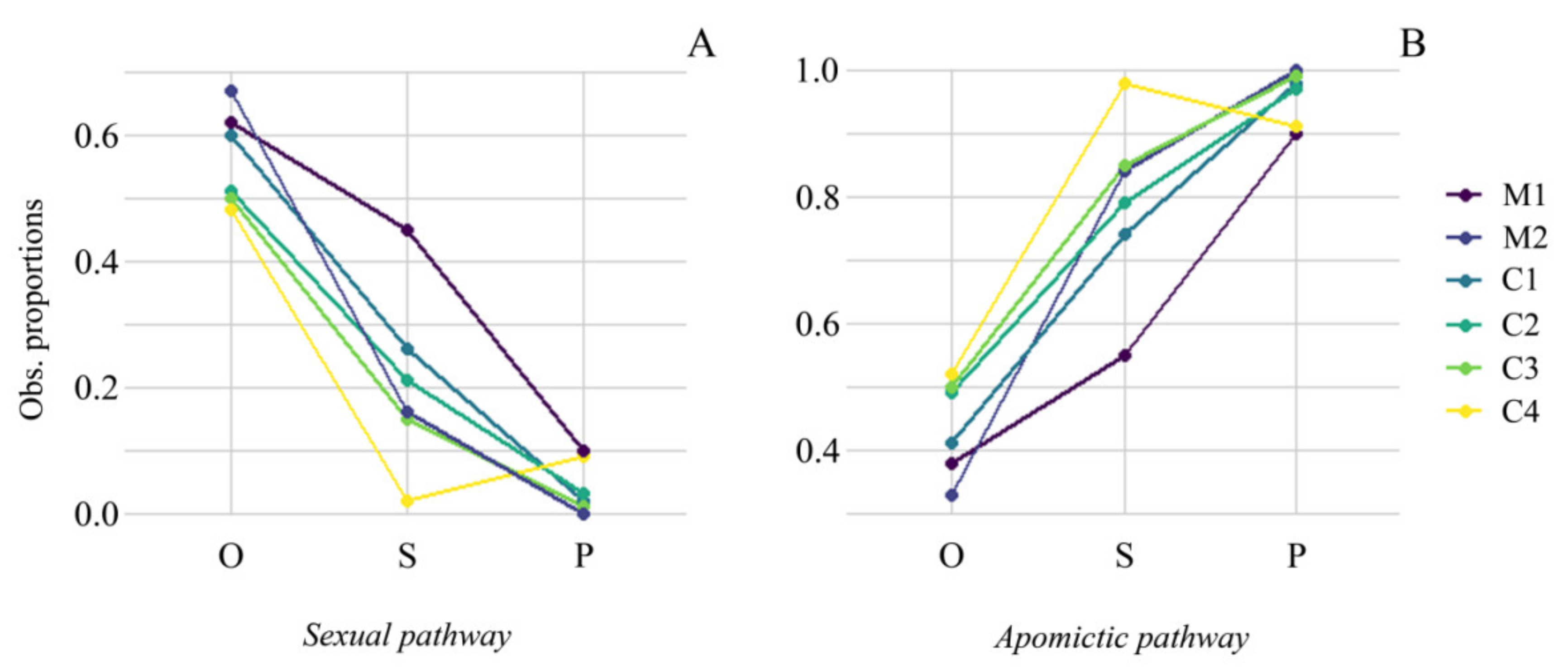
2.4. Competition among Reproductive Pathways through the Reproductive Stages
3. Discussion
3.1. Becoming a Seed: The Advantages of Aposporic Embryo Sacs
3.2. Non-Clonal Progeny and Their Evolutive Role in Apomictic Populations
4. Materials and Methods
4.1. Plant Material
4.2. Cytoembryological Analyses of Female Gametophytes
4.3. Flow Cytometry Seed Analysis
4.4. Progeny Test and Molecular Profile Analyses
4.5. Reproductive Pathway Efficiency Assessment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Asker, S.E.; Jerling, L. Apomixis in Plants; CRC Press: Boca Raton, FL, USA, 1992. [Google Scholar]
- Carman, J.G. Asynchronous expression of duplicate genes in angiosperms may cause apomixis, bispory, tetraspory, and polyembryony. Biol. J. Linn. Soc. 1997, 61, 51–94. [Google Scholar] [CrossRef]
- Hojsgaard, D.; Klatt, S.; Baier, R.; Carman, J.G.; Hörandl, E. Taxonomy and biogeography of apomixis in angiosperms and associated biodiversity characteristics. Crit. Rev. Plant Sci. 2014, 33, 414–427. [Google Scholar] [CrossRef] [PubMed]
- Nogler, G. Genetics of apospory in apomictic Ranunculus auricomus. V: Conclusion. Bot. Helv. 1984, 94, 411–422. [Google Scholar]
- Asker, S.E. Gametophytic apomixis: Elements and genetic regulation. Hereditas 1980, 93, 277–293. [Google Scholar] [CrossRef]
- Ortiz, J.P.A.; Quarin, C.L.; Pessino, S.C.; Acuña, C.; Martínez, E.J.; Espinoza, F.; Hojsgaard, D.H.; Sartor, M.E.; Cáceres, M.E.; Pupilli, F. Harnessing apomictic reproduction in grasses: What we have learned from Paspalum. Ann. Bot. 2013, 112, 767–787. [Google Scholar] [CrossRef]
- Noyes, R.D.; Givens, A.D. Quantitative assessment of megasporogenesis for the facultative apomicts Erigeron annuus and Erigeron strigosus (Asteraceae). Int. J. Plant Sci. 2013, 174, 1239–1250. [Google Scholar] [CrossRef]
- Hand, M.L.; Koltunow, A.M. The genetic control of apomixis: Asexual seed formation. Genetics 2014, 197, 441–450. [Google Scholar] [CrossRef]
- Rodrigo, J.M.; Zappacosta, D.C.; Selva, J.P.; Garbus, I.; Albertini, E.; Echenique, V. Apomixis frequency under stress conditions in weeping lovegrass (Eragrostis curvula). PLoS ONE 2017, 12, e0175852. [Google Scholar] [CrossRef]
- Karunarathne, P.; Reutemann, A.V.; Schedler, M.; Glücksberg, A.; Martínez, E.J.; Honfi, A.I.; Hojsgaard, D. Sexual modulation in a polyploid grass: A reproductive contest between environmentally inducible sexual and genetically dominant apomictic pathways. Sci. Rep. 2020, 10, 8319. [Google Scholar] [CrossRef]
- Ulum, F.B.; Castro, C.C.; Hörandl, E. Ploidy-dependent effects of light stress on the mode of reproduction in the Ranunculus auricomus complex (Ranunculaceae). Front. Plant Sci. 2020, 11, 104. [Google Scholar] [CrossRef]
- Hojsgaard, D.; Hörandl, E. A little bit of sex matters for genome evolution in asexual plants. Front. Plant Sci. 2015, 6, 82. [Google Scholar] [CrossRef]
- Karunarathne, P.; Hojsgaard, D. Single independent autopolyploidization events from distinct diploid gene pools and residual sexuality support range expansion of locally adapted tetraploid genotypes in a South American grass. Front. Genet. 2021, 12, 736088. [Google Scholar] [CrossRef]
- Grimanelli, D. Epigenetic regulation of reproductive development and the emergence of apomixis in angiosperms. Curr. Opin. Plant Biol. 2012, 15, 57–62. [Google Scholar] [CrossRef]
- Hojsgaard, D.; Hörandl, E. The rise of apomixis in natural plant populations. Front. Plant Sci. 2019, 10, 358. [Google Scholar] [CrossRef]
- Ozias-Akins, P.; Van Dijk, P. Mendelian genetics of apomixis in plants. Annu. Rev. Genet. 2007, 41, 509–537. [Google Scholar] [CrossRef]
- Grimanelli, D.; Leblanc, O.; Perotti, E.; Grossniklaus, U. Developmental genetics of gametophytic apomixis. Trends Genet. 2001, 17, 597–604. [Google Scholar] [CrossRef]
- Koltunow, A.M.; Grossniklaus, U. Apomixis: A developmental perspective. Annu. Rev. Plant Biol. 2003, 54, 547–574. [Google Scholar] [CrossRef]
- Tucker, M.R.; Koltunow, A.M.G. Sexual and asexual (apomictic) seed development in flowering plants: Molecular, morphological and evolutionary relationships. Funct. Plant Biol. 2009, 36, 490–504. [Google Scholar] [CrossRef]
- Hojsgaard, D.; Schartl, M. Skipping sex: A non-recombinant genomic assemblage of complementary reproductive modules. Bioessays 2021, 43, e2000111. [Google Scholar] [CrossRef]
- Carman, J.G.; Jamison, M.; Elliott, E.; Dwivedi, K.K.; Naumova, T.N. Apospory appears to accelerate onset of meiosis and sexual embryo sac formation in sorghum ovules. BMC Plant Biol. 2011, 11, 9. [Google Scholar] [CrossRef]
- Albertini, E.; Barcaccia, G.; Carman, J.G.; Pupilli, F. Did apomixis evolve from sex or was it the other way around? J. Exp. Bot. 2019, 70, 2951–2964. [Google Scholar] [CrossRef] [PubMed]
- Quarin, C.L. Seasonal changes in the incidence of apomixis of diploid, triploid, and tetraploid plants of Paspalum cromyorrhizon. Euphytica 1986, 35, 515–522. [Google Scholar] [CrossRef]
- Rebozzio, R.N.; Sartor, M.E.; Quarin, C.L.; Espinoza, F. Residual sexuality and its seasonal variation in natural apomictic Paspalum notatum accessions. Biol. Plant. 2011, 55, 391–395. [Google Scholar] [CrossRef]
- Selva, J.P.; Zappacosta, D.; Carballo, J.; Rodrigo, J.M.; Bellido, A.; Gallo, C.A.; Gallardo, J.; Echenique, V. Genes modulating the increase in sexuality in the facultative diplosporous grass Eragrostis curvula under water stress conditions. Genes 2020, 11, 969. [Google Scholar] [CrossRef]
- Naumova, T.N.; Hayward, M.D.; Wagenvoort, M. Apomixis and sexuality in diploid and tetraploid accessions of Brachiaria decumbens. Sex. Plant Reprod. 1999, 12, 43–52. [Google Scholar] [CrossRef]
- Quarin, C.L.; Espinoza, F.; Martínez, E.J.; Pessino, S.C.; Bovo, O.A. A rise of ploidy level induces the expression of apomixis in Paspalum notatum. Sex. Plant Reprod. 2001, 13, 243–249. [Google Scholar] [CrossRef]
- Espinoza, F.; Pessino, S.C.; Quarin, C.L.; Valle, E.M. Effect of pollination timing on the rate of apomictic reproduction reveald by RAPD Markers in Paspalum notatum. Ann. Bot. 2002, 89, 165–170. [Google Scholar] [CrossRef]
- Krahulcová, A.; Papoušková, S.; Krahulec, F. Reproduction mode in the allopolyploid facultatively apomictic hawkweed Hieracium rubrum (Asteraceae, H. subgen. Pilosella). Hereditas 2004, 141, 19–30. [Google Scholar] [CrossRef]
- Sharbel, T.F.; Voigt, M.L.; Corral, J.M.; Galla, G.; Kumlehn, J.; Klukas, C.; Schreiber, F.; Vogel, H.; Rotter, B. Apomictic and sexual ovules of Boechera display heterochronic global gene expression patterns. Plant Cell 2010, 22, 655–671. [Google Scholar] [CrossRef]
- Hojsgaard, D.H.; Martínez, E.J.; Quarin, C.L. Competition between meiotic and apomictic pathways during ovule and seed development results in clonality. New Phytol. 2013, 197, 336–347. [Google Scholar] [CrossRef]
- Harlan, J.R.; Brooks, M.H.; Borgaonkar, D.S.; De Wet, J.M.J. Nature and inheritance of apomixis in Bothriochloa and Dichanthium. Bot. Gaz. 1964, 125, 41–46. [Google Scholar] [CrossRef]
- Mazzucato, A.; Barcaccia, G.; Pezzotti, M.; Falcinelli, M. Biochemical and molecular markers for investigating the mode of reproduction in the facultative apomict Poa pratensis L. Sex. Plant Reprod. 1995, 8, 133–138. [Google Scholar] [CrossRef]
- Cosendai, A.C.; Hörandl, E. Cytotype stability, facultative apomixis and geographical parthenogenesis in Ranunculus kuepferi (Ranunculaceae). Ann. Bot. 2010, 105, 457–470. [Google Scholar] [CrossRef][Green Version]
- Aliyu, O.M.; Schranz, M.E.; Sharbel, T.F. Quantitative variation for apomictic reproduction in the genus Boechera (Brassicaceae). Am. J. Bot. 2010, 97, 1719–1731. [Google Scholar] [CrossRef]
- Vallejo-Marín, M.; Dorken, M.E.; Barrett, S.C.H. The ecological and evolutionary consequences of clonality for plant mating. Annu. Rev. Ecol. Evol. Syst. 2010, 41, 193–213. [Google Scholar] [CrossRef]
- Hamston, T.J.; de Vere, N.; King, R.A.; Pellicer, J.; Fay, M.F.; Cresswell, J.E.; Stevens, J.R. Apomixis and hybridization drives reticulate evolution and phyletic differentiation in Sorbus L.: Implications for conservation. Front. Plant Sci. 2018, 9, 1796. [Google Scholar] [CrossRef]
- Quarin, C.L.; Hanna, W.W.; Fernandez, A. Genetic studies in diploid and tetraploid Paspalum species: Embryo sac development, chromosome behavior, and fertility in P. cromyorrhizon, P. laxum, and P. proliferum. J. Hered. 1982, 73, 254–256. [Google Scholar] [CrossRef]
- Norrmann, G.A.; Quarin, C.L.; Burson, B.L. Cytogenetics and reproductive behavior of different chromosome races in six Paspalum species. J. Hered. 1989, 80, 24–28. [Google Scholar] [CrossRef]
- Reutemann, A.V.; Martínez, E.J.; Daviña, J.R.; Hojsgaard, D.H.; Honfi, A.I. El cariotipo de Paspalum cromyorrhizon diploide y tetraploide (Poaceae, Panicoideae, Paspaleae). Darwiniana 2021, 9, 375–386. [Google Scholar] [CrossRef]
- Soliman, M.; Bocchini, M.; Stein, J.; Ortiz, J.P.A.; Albertini, E.; Delgado, L. Environmental and genetic factors affecting apospory expressivity in diploid Paspalum rufum. Plants 2021, 10, 2100. [Google Scholar] [CrossRef]
- Quarin, C.L.; Norrmann, G.A. Cytology and reproductive behavior of Paspalum equitans, P. ionanthum, and their hybrids with diploid and tetraploid cytotypes of P. cromyorrhizon. Bot. Gaz. 1987, 148, 386–391. Available online: http://www.jstor.org/stable/2995357 (accessed on 1 June 2022). [CrossRef]
- Sartor, M.E.; Quarin, C.L.; Urbani, M.H.; Espinoza, F. Ploidy levels and reproductive behaviour in natural populations of five Paspalum species. Plant Syst. Evol. 2011, 293, 31–41. [Google Scholar] [CrossRef]
- Galdeano, F.; Urbani, M.H.; Sartor, M.E.; Honfi, A.I.; Espinoza, F.; Quarin, C.L. Relative DNA content in diploid, polyploid, and multiploid species of Paspalum (Poaceae) with relation to reproductive mode and taxonomy. J. Plant Res. 2016, 129, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Siena, L.A.; Sartor, M.E.; Espinoza, F.; Quarin, C.L.; Ortiz, J.P.A. Genetic and embryological evidences of apomixis at the diploid level in Paspalum rufum support recurrent auto-polyploidization in the species. Sex. Plant Reprod. 2008, 21, 205–215. [Google Scholar] [CrossRef]
- Reutemann, A.V. Diversidad Genética en Especies del Género Paspalum (Poaceae) con Diferentes Sistemas Genéticos. Ph.D. Thesis, Universidad Nacional de Córdoba, Córdoba, Argentina, 4 March 2020. [Google Scholar]
- Koltunow, A.M. Apomixis: Embryo sacs and embryos formed without meiosis or fertilization in ovules. Plant Cell 1993, 5, 1425. [Google Scholar] [CrossRef] [PubMed]
- Hojsgaard, D.; Greilhuber, J.; Pellino, M.; Paun, O.; Sharbel, T.F.; Hörandl, E. Emergence of apospory and bypass of meiosis via apomixis after sexual hybridisation and polyploidisation. New Phytol. 2014, 204, 1000–1012. [Google Scholar] [CrossRef] [PubMed]
- Grimanelli, D.; García, M.; Kaszas, E.; Perotti, E.; Leblanc, O. Heterochronic expression of sexual reproductive programs during apomictic development in Tripsacum. Genetics 2003, 165, 1521–1531. [Google Scholar] [CrossRef]
- Colono, C.; Ortiz, J.P.A.; Permingeat, H.R.; Souza Canada, E.D.; Siena, L.A.; Spoto, N.; Galdeano, F.; Espinoza, F.; Leblanc, O.; Pessino, S.C. A plant-specific TGS1 homolog influences gametophyte development in sexual tetraploid Paspalum notatum ovules. Front. Plant Sci. 2019, 10, 1566. [Google Scholar] [CrossRef]
- Quarin, C.L. Effect of pollen source and pollen ploidy on endosperm formation and seed set in pseudogamous apomictic Paspalum notatum. Sex. Plant Reprod. 1999, 11, 331–335. [Google Scholar] [CrossRef]
- Birchler, J.A. Dosage analysis of maize endosperm development. Annu. Rev. Genet. 1993, 27, 181–204. [Google Scholar] [CrossRef]
- Grossniklaus, U.; Spillane, C.; Page, D.R.; Köhler, C. Genomic imprinting and seed development: Endosperm formation with and without sex. Curr. Opin. Plant Biol. 2001, 4, 21–27. [Google Scholar] [CrossRef]
- Martínez, E.J.; Espinoza, F.; Quarin, C.L. BIII Progeny (2n + n) from apomictic Paspalum notatum obtained through early pollination. J. Hered. 1994, 85, 295–297. [Google Scholar] [CrossRef]
- Kojima, A.; Nagato, Y. Pseudogamous embryogenesis and the degree of parthenogenesis in Allium tuberosum. Sex. Plant Reprod. 1992, 5, 79–85. [Google Scholar] [CrossRef]
- Curtis, M.D.; Grossniklaus, U. Molecular control of autonomous embryo and endosperm development. Sex. Plant Reprod. 2008, 21, 79–88. [Google Scholar] [CrossRef]
- Hörandl, E.; Paun, O. Patterns and sources of genetic diversity in apomictic plants: Implications for evolutionary potentials. In Apomixis: Evolution, Mechanisms and Perspectives, 1st ed.; Hörandl, E., Grossniklaus, U., Sharbel, T., van Dijk, P., Eds.; A.R.G. Gantner Verlag: Ruggell, Liechtenstein; Koenigstein, Germany, 2007; Volume 147, pp. 169–194. [Google Scholar]
- Spillane, C.; Steimer, A.; Grossniklaus, U. Apomixis in agriculture: The quest for clonal seeds. Sex. Plant Reprod. 2001, 14, 179–187. [Google Scholar] [CrossRef]
- Nygren, A. Some interspecific crosses in Calamagrostis and their evolutionary consequences. Hereditas 1948, 34, 387–413. [Google Scholar] [CrossRef]
- Savidan, Y.; Pernès, J. Diploid-tetraploid-dihaploid cycles and the evolution of Panicum maximum Jacq. Evolution 1982, 36, 596–600. [Google Scholar] [CrossRef]
- Burson, B.L.; Bennett, H.W. Cytology and reproduction of three Paspalum species. J. Hered. 1970, 61, 129–132. [Google Scholar] [CrossRef]
- Burson, B.L.; Bennett, H.W. Chromosome numbers, microsporogenesis, and mode of reproduction of seven Paspalum species. Crop Sci. 1971, 11, 292–294. [Google Scholar] [CrossRef]
- Quarin, C.L.; Pozzobon, M.T.; Valls, J.F.M. Cytology and reproductive behavior of diploid, tetraploid and hexaploid germplasm accessions of a wild forage grass: Paspalum compressifolium. Euphytica 1996, 90, 345–349. [Google Scholar] [CrossRef]
- Espinoza, F.; Urbani, M.H.; Martínez, E.J.; Quarin, C.L. The breeding system of three Paspalum species with forage potential. Trop. Grassl. 2001, 35, 211–217. [Google Scholar]
- Hojsgaard, D.; Schegg, E.; Valls, J.F.; Martínez, E.J.; Quarin, C.L. Sexuality, apomixis, ploidy levels, and genomic relationships among four Paspalum species of the subgenus Anachyris (Poaceae). Flora Morphol. Distrib. Funct. Ecol. Plants 2008, 203, 535–547. [Google Scholar] [CrossRef]
- Quarin, C.L.; Norrmann, G.A. Interspecific hybrids between five Paspalum species. Bot. Gaz. 1990, 151, 366–369. [Google Scholar] [CrossRef]
- Naumova, T.; Den Nijs, A.P.M.; Willemse, M.T.M. Quantitative analysis of aposporous parthenogenesis in Poa pratensis genotypes. Acta Bot. Neerl. 1993, 42, 299–312. [Google Scholar] [CrossRef]
- Hörandl, E. The complex causality of geographical parthenogenesis. New Phytol. 2006, 171, 525–538. [Google Scholar] [CrossRef]
- Charlesworth, D.; Charlesworth, B. Inbreeding depression and its evolutionary consequences. Annu. Rev. Ecol. Evol. Syst. 1987, 18, 237–268. [Google Scholar] [CrossRef]
- Husband, B.C.; Schemske, D.W. Evolution of the magnitude and timing of inbreeding depression in plants. Evolution 1996, 50, 54–70. [Google Scholar] [CrossRef]
- Comai, L. The advantages and disadvantages of being polyploid. Nat. Rev. Genet. 2005, 6, 836–846. [Google Scholar] [CrossRef]
- Hojsgaard, D. Transient activation of apomixis in sexual neotriploids may retain genomically altered states and enhance polyploid establishment. Front. Plant Sci. 2018, 9, 230. [Google Scholar] [CrossRef]
- Lin, B.Y. Ploidy barrier to endosperm development in maize. Genetics 1984, 107, 103–115. [Google Scholar] [CrossRef]
- Richards, A.J. Eutriploid facultative agamospermy in Taraxacum. New Phytol. 1970, 69, 761–774. [Google Scholar] [CrossRef]
- Van Dijk, P.J. Ecological and evolutionary opportunities of apomixis: Insights from Taraxacum and Chondrilla. Philos. Trans. R. Soc. B 2003, 358, 1113–1121. [Google Scholar] [CrossRef]
- Bicknell, R.A.; Lambie, S.C.; Butler, R.C. Quantification of progeny classes in two facultatively apomictic accessions of Hieracium. Hereditas 2003, 138, 11–20. [Google Scholar] [CrossRef]
- Sorensen, A.M.; Rouse, D.; Clements, M.; John, P.; Perotti, E. Description of a fertilization-independent obligate apomictic species: Corunastylis apostasioides Fitzg. Sex. Plant Reprod. 2009, 22, 153–165. [Google Scholar] [CrossRef]
- Kant, R.; Verma, J. Obligate apomixis in Zeuxine strateumatica (Lindl.) Schltr. (Orchidaceae). Vegetos 2012, 25, 274–277. [Google Scholar]
- Xiao, H.; Luo, H.; Liu, N.; Turner, C.; Chen, X.; Ding, H.; Liang, Y.; Tan, S.; Tang, J.; Xiong, D.; et al. High fruit setting rate without male participation: A case study of obligate apomixis in Rhomboda tokioi (Orchidaceae). Flora 2021, 283, 151920. [Google Scholar] [CrossRef]
- Kollmann, J.; Steinger, T.; Roy, B.A. Evidence of sexuality in European Rubus (Rosaceae) species based on AFLP and allozyme analysis. Am. J. Bot. 2000, 87, 1592–1598. [Google Scholar] [CrossRef]
- Houliston, G.J.; Chapman, H.M. Reproductive strategy and population variability in the facultative apomict Hieracium pilosella (Asteraceae). Am. J. Bot. 2004, 91, 37–44. [Google Scholar] [CrossRef]
- Paun, O.; Greilhuber, J.; Temsch, E.M.; Hörandl, E. Patterns, sources and ecological implications of clonal diversity in apomictic Ranunculus carpaticola (Ranunculus auricomus complex, Ranunculaceae). Mol. Ecol. 2006, 15, 897–910. [Google Scholar] [CrossRef]
- Martínez, E.J.; Urbani, M.H.; Quarin, C.L.; Ortiz, J.P. Inheritance of apospory in bahiagrass, Paspalum notatum. Hereditas 2001, 135, 19–25. [Google Scholar] [CrossRef]
- Noirot, M. Allelic ratios and sterility in the agamic complex of the Maximae (Panicoideae): Evolutionary role of the residual sexuality. J. Evol. Biol. 1993, 6, 95–101. [Google Scholar] [CrossRef]
- Stebbins, G.L. Variation and Evolution in Plants; Columbia University Press: New York, NY, USA, 1950. [Google Scholar]
- Grant, V. Plant Speciation; Columbia University Press: New York, NY, USA, 1981. [Google Scholar]
- Kondrashov, A.S. Classification of hypotheses on the advantage of amphimixis. J. Hered. 1993, 84, 372–387. [Google Scholar] [CrossRef]
- Clausen, J. Partial apomixis as an equilibrium system in evolution. Carylogia 1954, 6, 469–479. [Google Scholar]
- Whitton, J.; Sears, C.J.; Bacck, E.J.; Otto, S.P. The dynamic nature of apomixis in the angiosperms. Int. J. Plant Sci. 2008, 169, 169–182. [Google Scholar] [CrossRef]
- Hörandl, E.; Hojsgaard, D. The evolution of apomixis in angiosperms: A reappraisal. Plant Biosyst. 2012, 146, 681–693. [Google Scholar] [CrossRef]
- Chapman, H.; Brown, J. ‘Thawing’ of ‘frozen’ variation in an adventive, facultatively apomictic, clonal weed. Plant Species Biol. 2001, 16, 107–118. [Google Scholar] [CrossRef]
- Richards, A.J. Plant Breeding Systems; Chapman & Hall: London, UK, 1997. [Google Scholar]
- Hodač, L.; Klatt, S.; Hojsgaard, D.; Sharbel, T.F.; Hörandl, E. A little bit of sex prevents mutation accumulation even in apomictic polyploid plants. BMC Evol. Biol. 2019, 19, 170. [Google Scholar] [CrossRef] [PubMed]
- Reutemann, A.V.; Martínez, E.J.; Schedler, M.; Daviña, J.R.; Hojsgaard, D.; Honfi, A.I. Uniparentality: Advantages for range expansion in diploid and diploid- autopolyploid species. Bot. J. Linn. Soc. 2022, in press. [Google Scholar]
- Young, B.A.; Sherwood, R.T.; Bashaw, E.C. Cleared-pistil and thick-sectioning techniques for detecting aposporous apomixis in grasses. Can. J. Bot. 1979, 57, 1668–1672. [Google Scholar] [CrossRef]
- Zilli, A.L.; Brugnoli, E.A.; Marcón, F.; Billa, M.B.; Rios, E.F.; Martínez, E.J.; Acuña, C.A. Heterosis and expressivity of apospory in tetraploid Bahiagrass hybrids. Crop Sci. 2015, 55, 1189–1201. [Google Scholar] [CrossRef]
- Otto, F. DAPI staining of fixed cells for high-resolution flow cytometry of nuclear DNA. In Methods in Cell Biology, 1st ed.; Darzynkiewicz, Z., Crissman, H.A., Eds.; Academic Press: Cambridge, MA, USA, 1990; Volume 33, pp. 105–110. [Google Scholar] [CrossRef]
- Matzk, F.; Meister, A.; Schubert, I. An efficient screen for reproductive pathways using mature seeds of monocots and dicots. Plant J. 2000, 21, 97–108. [Google Scholar] [CrossRef]
- Doležel, J.; Greilhuber, J.; Suda, J. Estimation of nuclear DNA content in plants using flow cytometry. Nat. Protoc. 2007, 2, 2233–2244. [Google Scholar] [CrossRef]
- Meirmans, P.G.; Van Tienderen, P.H. GENOTYPE and GENODIVE: Two programs for the analysis of genetic diversity of asexual organisms. Mol. Ecol. Notes 2004, 4, 792–794. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019; Available online: https://www.R-project.org/ (accessed on 7 March 2019).
| Species | Pop | n | Number of Ovules Bearing (%) | Proportions | |||||
|---|---|---|---|---|---|---|---|---|---|
| MES | AES | MES + AES | IES | SP | AP | p | |||
| P. maculosum | M1 | 146 | 51 (34.9) | - | 81 (55.5) | 14 (9.6) | 0.62 | 0.38 | <0.001 |
| M2 | 179 | 83 (46.4) | 3 (1.7) | 72 (40.2) | 21 (11.7) | 0.67 | 0.33 | <0.001 | |
| P. cromyorhizon | C1 | 60 | 19 (31.7) | 5 (8.3) | 25 (41.7) | 11 (18.3) | 0.59 | 0.41 | 0.13 |
| C2 | 160 | 8 (5.0) | 3 (1.9) | 144 (90.0) | 5 (3.1) | 0.51 | 0.49 | 0.82 | |
| C3 | 150 | 4 (2.7) | 6 (4.0) | 138 (92.0) | 2 (1.3) | 0.50 | 0.50 | 0.95 | |
| C4 | 148 | 8 (5.4) | 17 (11.5) | 119 (80.4) | 4 (2.7) | 0.48 | 0.52 | 0.62 | |
| Species | Pop | n | n (%) Seeds with | Proportions | ||||
|---|---|---|---|---|---|---|---|---|
| 2C:3C | 2C:5C | 2C:6C | SP | AP | p | |||
| P. maculosum | M1 | 152 | 68 (44.7) | 84 (55.3) | - | 0.45 | 0.55 | 0.22 |
| M2 | 120 | 19 (15.8) | 101 (84.2) | - | 0.16 | 0.84 | <0.001 | |
| P. cromyorhizon | C1 | 62 | 16 (25.8) | 46 (74.2) | - | 0.26 | 0.74 | <0.001 |
| C2 | 162 | 34 (21.0) | 126 (77.8) | 2 (1.2) | 0.21 | 0.79 | <0.001 | |
| C3 | 154 | 23 (14.9) | 130 (84.4) | 1 (0.7) | 0.15 | 0.85 | <0.001 | |
| C4 | 140 | 3 (2.1) | 137 (97.9) | - | 0.02 | 0.98 | <0.001 | |
| Species | Pop | n | n (%) of Progeny | Proportions | |||
|---|---|---|---|---|---|---|---|
| C | nC | SP | AP | p | |||
| P. maculosum | M1 | 60 | 54 (90.0) | 6 (10.0) | 0.10 | 0.90 | <0.001 |
| M2 | 52 | 52 (100) | 0 (0.0) | 0.00 | 1.00 | <0.001 | |
| P. cromyorhizon | C1 | 46 | 45 (97.8) | 1 (2.2) | 0.02 | 0.98 | <0.001 |
| C2 | 106 | 103 (97.2) | 3 (2.8) | 0.03 | 0.97 | <0.001 | |
| C3 | 105 | 104 (99.1) | 1 (0.9) | 0.01 | 0.99 | <0.001 | |
| C4 | 105 | 96 (91.4) | 9 (8.6) | 0.09 | 0.91 | <0.001 | |
| Species | Pop | Proportions | χ2 | p | Reproductive Efficiency | ||||
|---|---|---|---|---|---|---|---|---|---|
| Sexual Pathway | Apomictic Pathway | ||||||||
| Ei | Oi | Ei | Oi | Sex | Apo | ||||
| P. maculosum | M1 | 0.62 | 0.45 | 0.38 | 0.55 | 10.0 | <0.001 | 0.73 | 1.45 |
| M2 | 0.67 | 0.16 | 0.33 | 0.84 | 81.8 | <0.001 | 0.24 | 2.55 | |
| P. cromyorhizon | C1 | 0.59 | 0.26 | 0.41 | 0.74 | 12.5 | <0.001 | 0.43 | 1.8 |
| C2 | 0.51 | 0.21 | 0.49 | 0.79 | 37.7 | <0.001 | 0.42 | 1.61 | |
| C3 | 0.50 | 0.15 | 0.50 | 0.85 | 50.0 | <0.001 | 0.3 | 1.7 | |
| C4 | 0.48 | 0.02 | 0.52 | 0.98 | 86.9 | <0.001 | 0.04 | 1.88 | |
| Species | Pop | Proportions | χ2 | p | Reproductive Efficiency | ||||
|---|---|---|---|---|---|---|---|---|---|
| Sexual Pathway | Apomictic Pathway | ||||||||
| Ei | Oi | Ei | Oi | Sex | Apo | ||||
| P. maculosum | M1 | 0.45 | 0.10 | 0.55 | 0.90 | 21.3 | <0.001 | 0.22 | 1.64 |
| M2 | 0.16 | 0.00 | 0.84 | 1.00 | 7.7 | <0.001 | 0.0 | 1.19 | |
| P. cromyorhizon | C1 | 0.26 | 0.02 | 0.74 | 0.98 | 9.4 | <0.001 | 0.08 | 1.32 |
| C2 | 0.21 | 0.03 | 0.79 | 0.97 | 16.3 | <0.001 | 0.14 | 1.23 | |
| C3 | 0.15 | 0.01 | 0.85 | 0.99 | 12.9 | <0.001 | 0.07 | 1.16 | |
| C4 | 0.02 | 0.09 | 0.98 | 0.91 | 4.0 | <0.001 | 4.5 | 0.93 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reutemann, A.V.; Honfi, A.I.; Karunarathne, P.; Eckers, F.; Hojsgaard, D.H.; Martínez, E.J. Variation of Residual Sexuality Rates along Reproductive Development in Apomictic Tetraploids of Paspalum. Plants 2022, 11, 1639. https://doi.org/10.3390/plants11131639
Reutemann AV, Honfi AI, Karunarathne P, Eckers F, Hojsgaard DH, Martínez EJ. Variation of Residual Sexuality Rates along Reproductive Development in Apomictic Tetraploids of Paspalum. Plants. 2022; 11(13):1639. https://doi.org/10.3390/plants11131639
Chicago/Turabian StyleReutemann, Anna Verena, Ana Isabel Honfi, Piyal Karunarathne, Fabiana Eckers, Diego Hernan Hojsgaard, and Eric Javier Martínez. 2022. "Variation of Residual Sexuality Rates along Reproductive Development in Apomictic Tetraploids of Paspalum" Plants 11, no. 13: 1639. https://doi.org/10.3390/plants11131639
APA StyleReutemann, A. V., Honfi, A. I., Karunarathne, P., Eckers, F., Hojsgaard, D. H., & Martínez, E. J. (2022). Variation of Residual Sexuality Rates along Reproductive Development in Apomictic Tetraploids of Paspalum. Plants, 11(13), 1639. https://doi.org/10.3390/plants11131639










