Health Beneficial Phytochemicals in Dioscorea caucasica Lipsky Leaves and Tubers and Their Inhibitory Effects on Physiologically Important Enzymes
Abstract
:1. Introduction
2. Results
2.1. Phytochemical Composition of Extracts
2.1.1. Identification of Secondary Metabolites Using UPLC-QTOF/MS
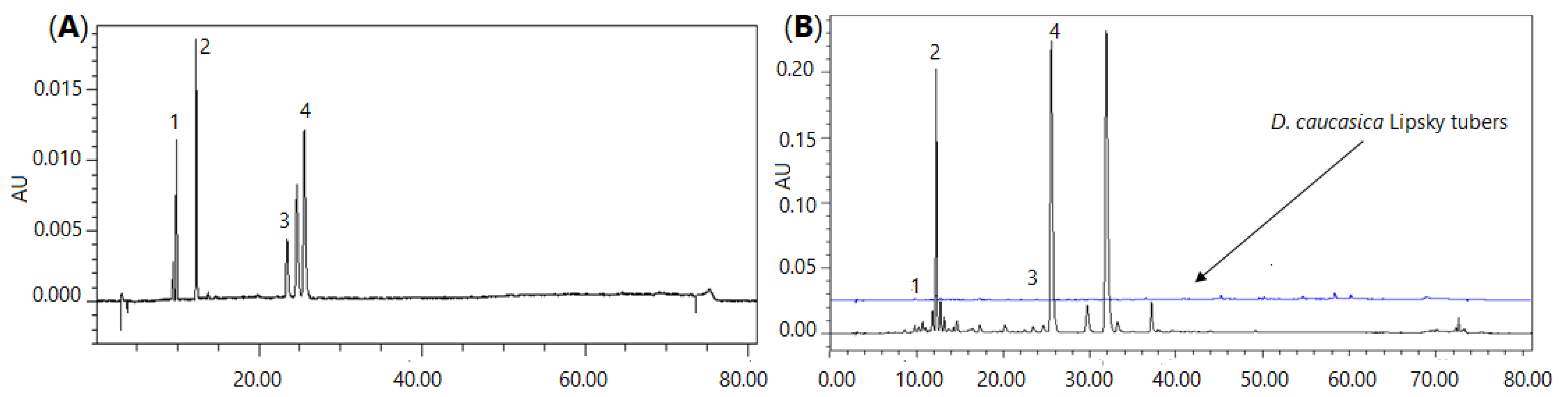
2.1.2. Quantitative Analysis of the Main Secondary Metabolites Using HPLC
2.2. Enzyme Inhibitory Properties of D. caucasica Extracts
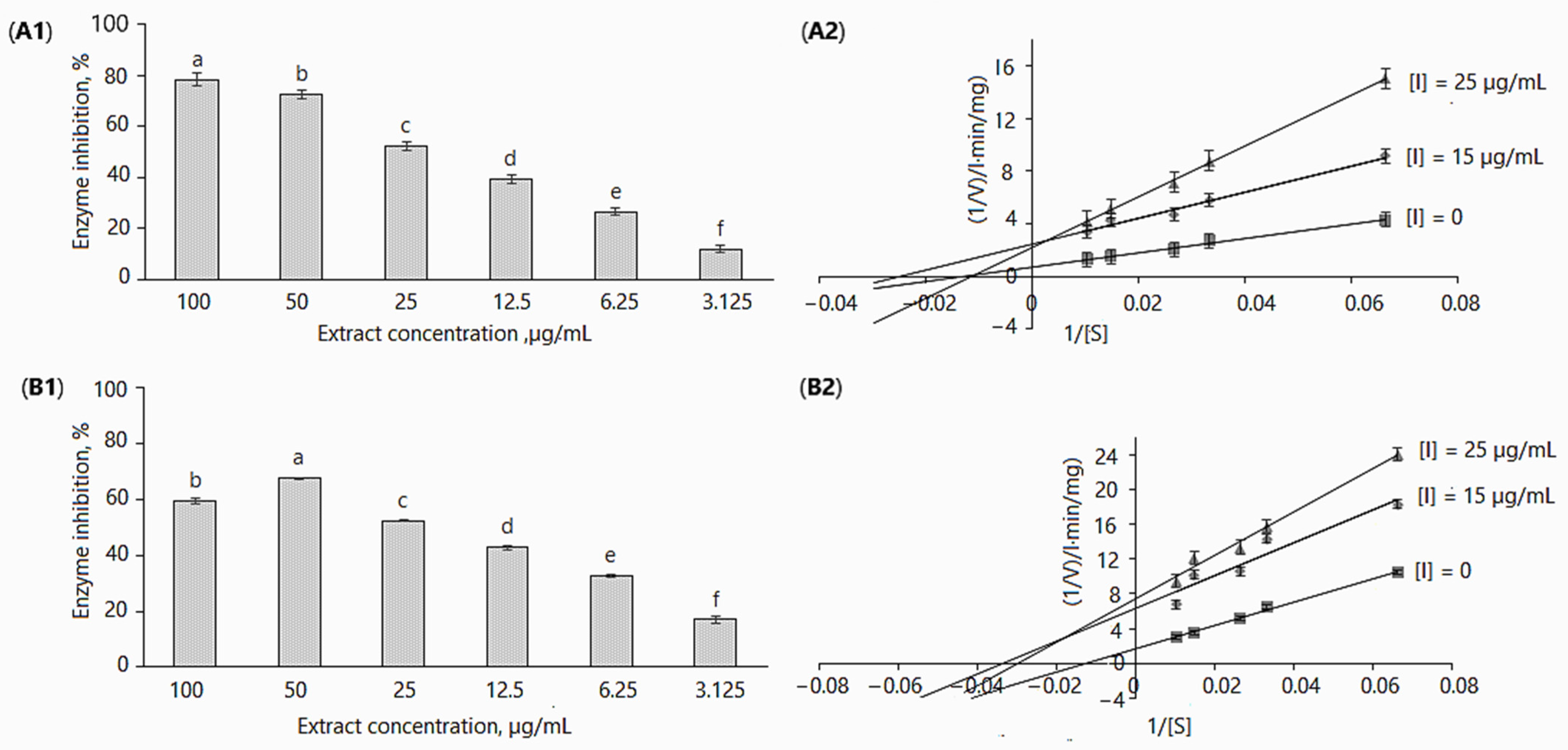
2.2.1. α-Glucosidase
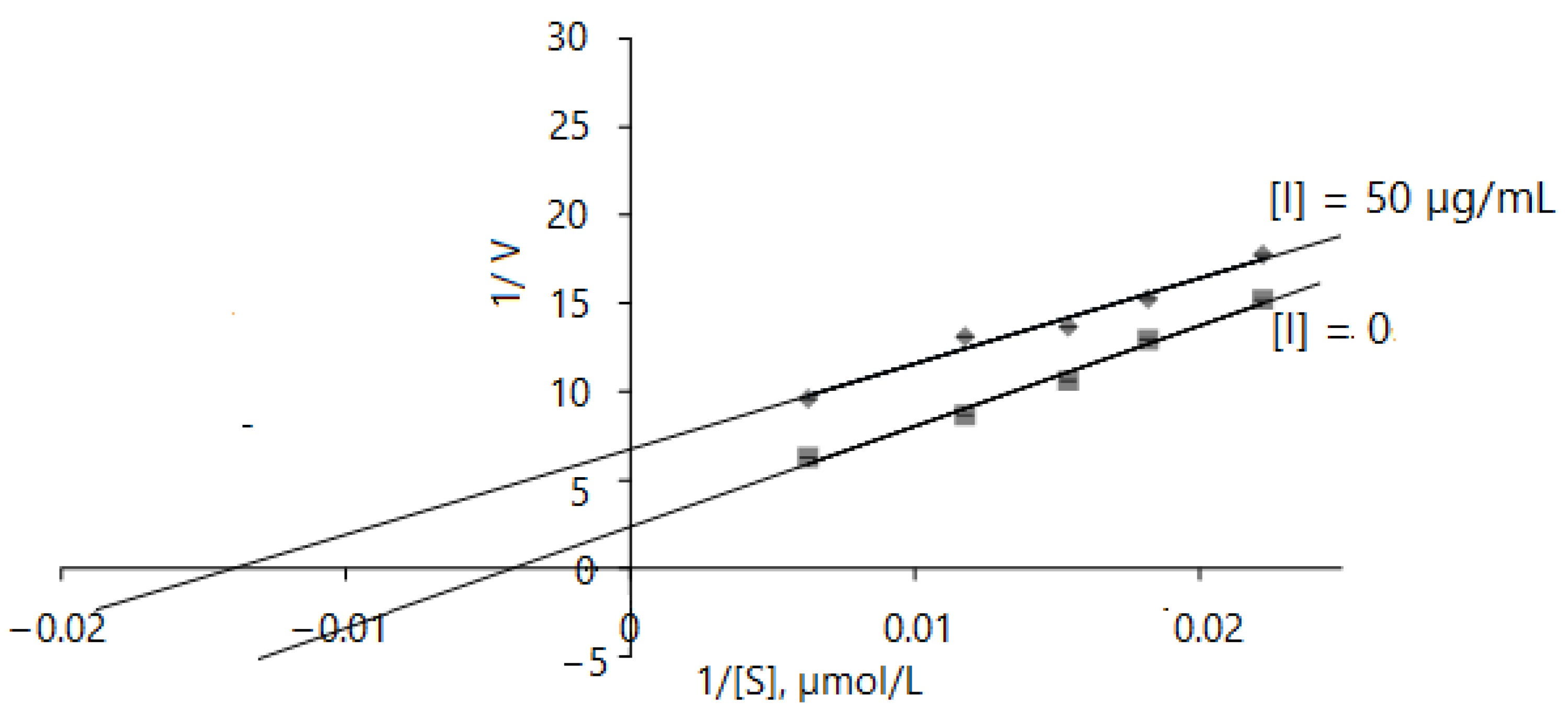
2.2.2. α-Glucosidase Inhibition Kinetics
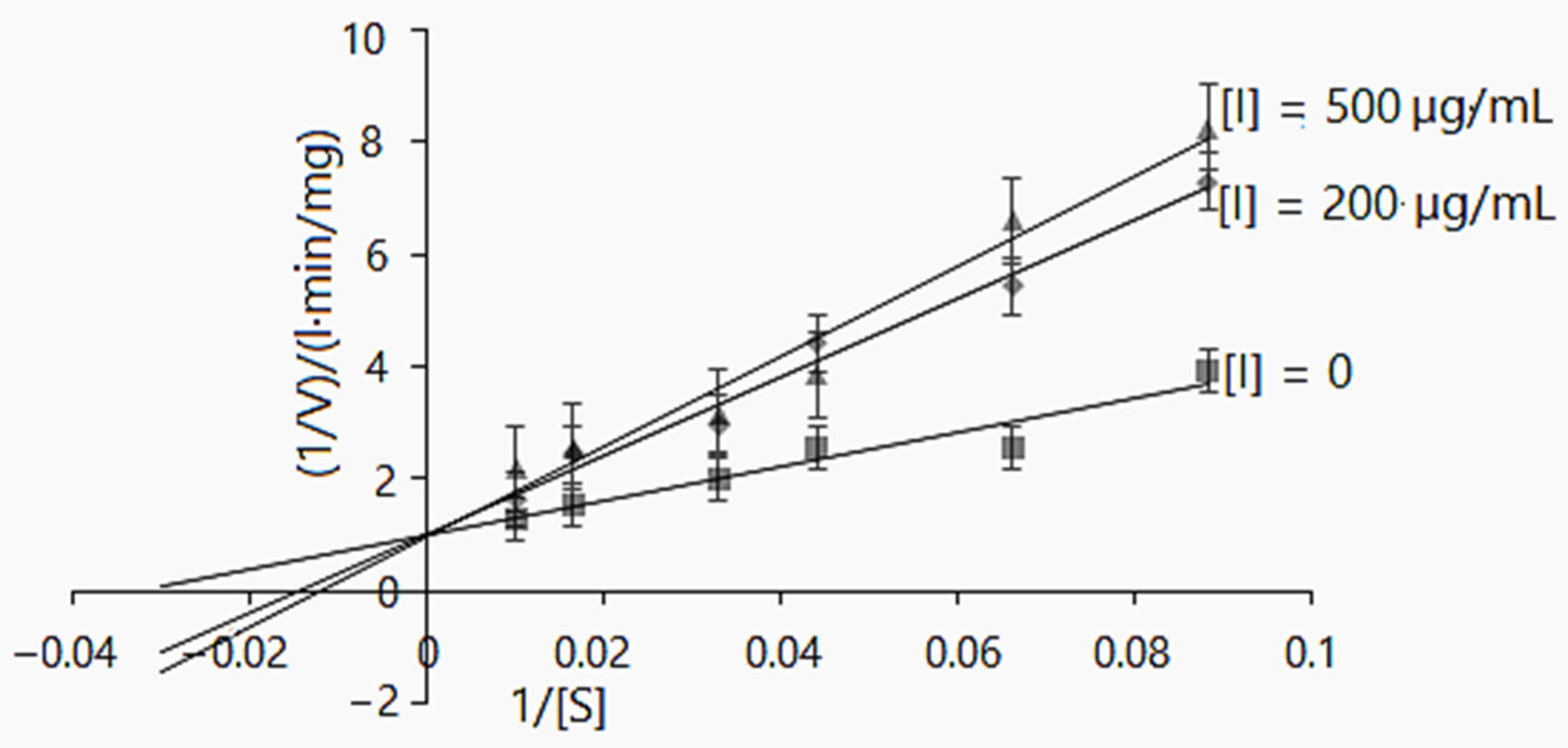
2.2.3. α-Amylase
2.2.4. Acetylcholinesterase (AChE) Inhibition Assay
2.2.5. Acetylcholinesterase Inhibition Kinetics
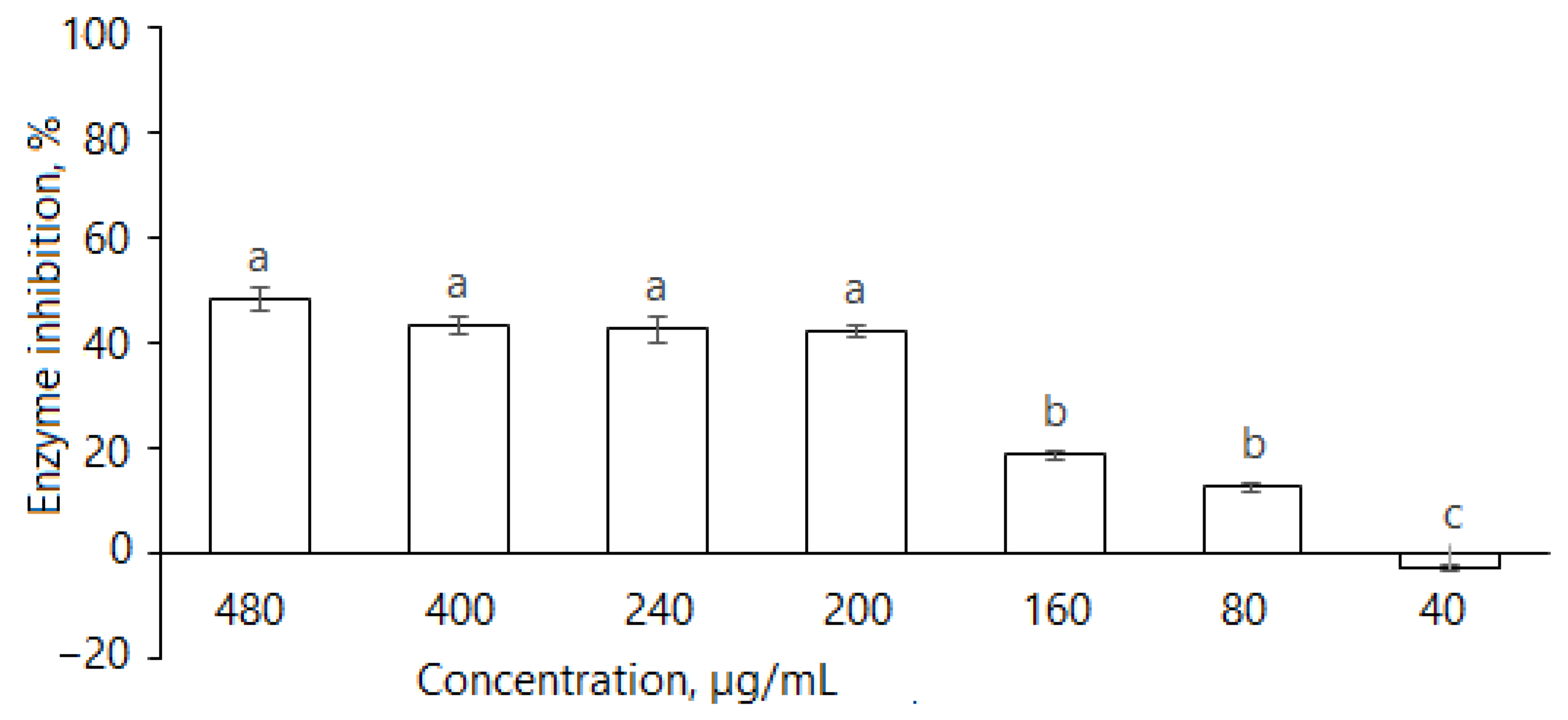
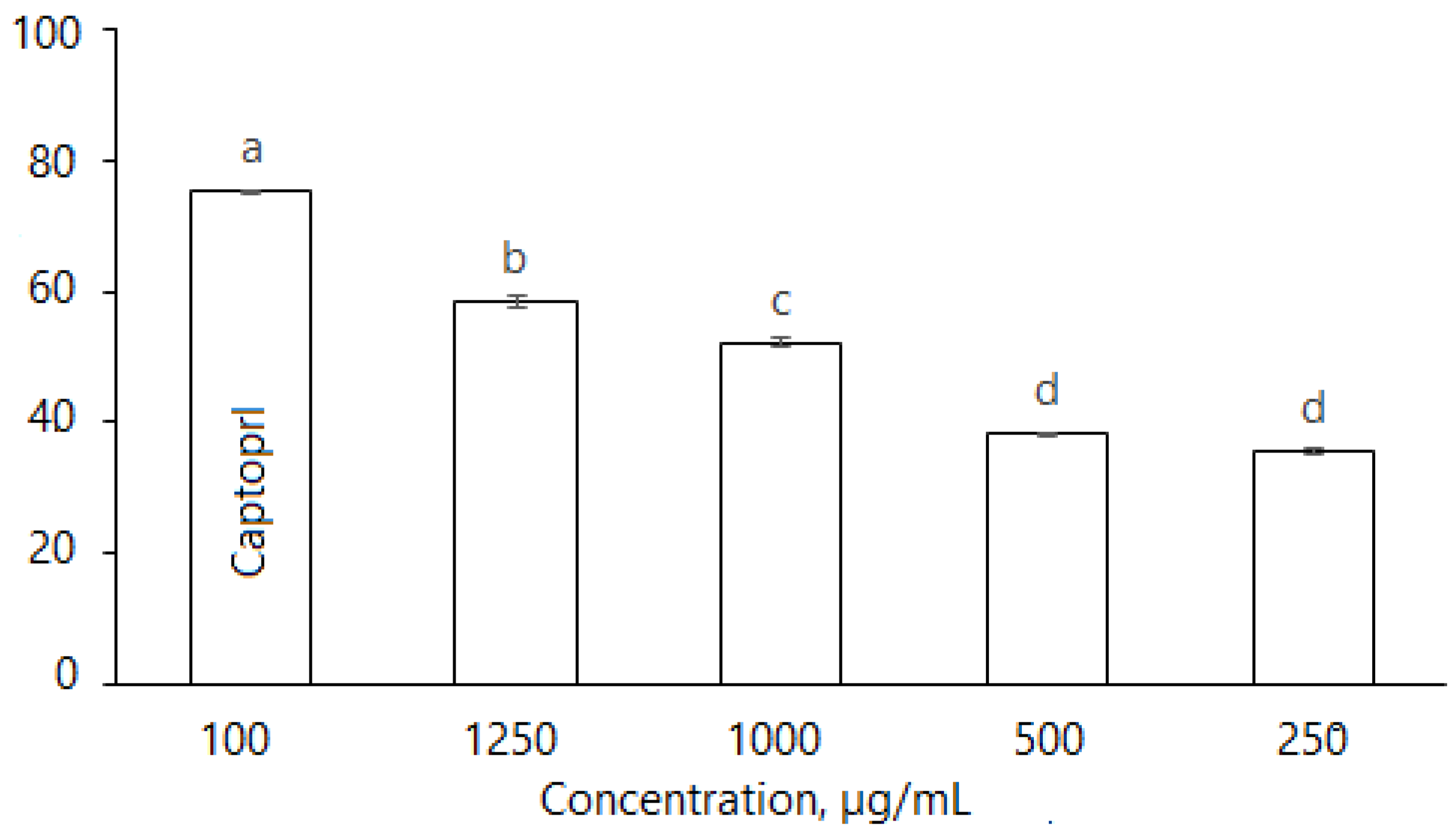
2.2.6. Angiotensin-Converting Enzyme (ACE)
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Preparation of D. caucasica Extracts
4.3. Identification and Quantitative Analysis of the Main Phytochemicals
4.3.1. Identification of Phytochemicals by UPLC-QTOF-MS
4.3.2. Quantification of the Main Phytochemicals by HPLC
4.4. Preparation of Solutions for Enzyme Inhibition Assays
4.4.1. Rabbit Lung and Captopril Solutions for Angiotensin-Converting Enzyme (ACE) Inhibition Assays
4.4.2. Donepezil HCl Solution for Acetylcholinesterase (AChE) Inhibitory Activity
4.4.3. Acarbose Solution for α-Amylase and α-Glucosidase Inhibition Assays
4.5. Determination of Enzyme Inhibition Activities
4.5.1. α-Glucosidase
4.5.2. α-Amylase
4.5.3. Acetylcholinesterase (AChE)
4.5.4. Angiotensin-Converting Enzyme (ACE)
4.6. Mathematical Modeling of Enzyme Inhibition Kinetics
4.7. Statistical Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nodland, S. Food’s Role as an Immunomodulator. Health and Nutrition Institute–Science for Healthier Food. 2021. Available online: https://khni.kerry.com/tag/nutrion/ (accessed on 21 April 2022).
- World Health Organization. Diet, nutrition, and the prevention of chronic diseases. World Health Organ. Tech. Rep. Ser. 2003, 916, 1–149. [Google Scholar]
- Boudjada, A.; Touil, A.; Bensouici, C.; Bendif, H.; Rhouati, S. Phenanthrene and dihydrophenanthrene derivatives from Dioscorea communis with anticholinesterase, and antioxidant activities. Nat. Prod. Res. 2018, 33, 3278–3282. [Google Scholar] [CrossRef] [PubMed]
- Murray, A.P.; Faraoni, M.B.; Castro, M.J.; Alza, N.P.; Cavallaro, V. Natural AChE Inhibitors from plants and their contribution to Alzheimer’s disease therapy. Curr. Neuropharmacol. 2013, 11, 388–413. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Figueiredo, E.A.d.; Alves, N.F.B.; Monteiro, M.M.d.O.; Cavalcanti, C.d.O.; Silva, T.M.S.d.; Silva, T.M.G.d.; Braga, V.d.A.; Oliveira, E.d.J. Antioxidant and antihypertensive effects of a chemically defined fraction of Syrah red wine on spontaneously hypertensive rats. Nutrients 2017, 9, 574. [Google Scholar] [CrossRef] [Green Version]
- Studzińska-Sroka, E.; Galanty, A.; Gościniak, A.; Wieczorek, M.; Kłaput, M.; Dudek-Makuch, M.; Cielecka-Piontek, J. Herbal infusions as a valuable functional food. Nutrients 2021, 13, 4051. [Google Scholar] [CrossRef]
- Zhao, C.; Li, X.; Miao, J.; Jing, S.; Li, X.; Huang, L.; Gao, W. The effect of different extraction techniques on property and bioactivity of polysaccharides from Dioscorea hemsleyi. Int. J. Biol. Macromol. 2017, 102, 847–856. [Google Scholar] [CrossRef]
- Kwon, C.S.; Sohn, H.Y.; Kim, S.H.; Kim, J.H.; Son, K.H.; Lee, J.S.; Lim, J.K.; Kim, J.S. Anti-obesity effect of Dioscorea nipponica Makino with lipase-inhibitory activity in rodents. Biosci. Biotechnol. Biochem. 2003, 67, 1451–1456. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, S.; More, P.; Derle, A.; Patil, A.B.; Markad, P.; Asok, A.; Kumbhar, N.; Shaikh, M.L.; Ramanamurthy, B.; Shinde, V.S.; et al. Diosgenin from Dioscorea bulbifera: Novel hit for treatment of Type II Diabetes Mellitus with inhibitory activity against α-amylase and α-glucosidase. PLoS ONE 2014, 9, e106039. [Google Scholar] [CrossRef] [Green Version]
- Zhao, W.X.; Wang, P.F.; Song, H.G.; Sun, N. Diosgenin attenuates neuropathic pain in a rat model of chronic constriction injury. Mol. Med. Rep. 2017, 16, 1559–1564. [Google Scholar] [CrossRef]
- Guo, X.; Sha, X.; Liu, J.; Cai, S.; Wang, Y.; Ji, B. Chinese purple yam (Dioscorea alata L.) extracts inhibit diabetes-related enzymes and protect HepG2 cells against oxidative stress and insulin resistance induced by FFA. Food Sci. Technol. Res. 2015, 21, 677–683. [Google Scholar] [CrossRef] [Green Version]
- Yadav, D.; Mishra, M.; Jadaun, P.; Choi, E.; Priyadharsini, R.; Kong, I.D. Glycemic index and glycemic load in cardiovascular disease risk. Prog. Nutr. 2016, 18, 95–101. [Google Scholar]
- Goyal, A.; Cusick, A.S.; Thielemier, B. ACE Inhibitors. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK430896/ (accessed on 22 April 2022).
- Christenhusz, M.J.; Byng, J.W. The number of known plants species in the world and its annual increase. Phytotaxa 2016, 261, 201–217. [Google Scholar] [CrossRef] [Green Version]
- Lebot, V. Tropical Root and Tuber Crops: Cassava, Sweet Potato, Yams and Aroids, 2nd ed.; CABI: Wallingford, UK; Boston, MA, USA, 2020; pp. 218–220. [Google Scholar]
- FAOASTAT Food and Agricultural Organization. Available online: https://www.fao.org/ (accessed on 20 August 2021).
- Kim, M.J.; Son, S.Y.; Jeon, S.G.; Kim, J.G.; Lee, C.-H. Metabolite Profiling of Dioscorea (Yam) leaves to identify bioactive compounds reveals their potential as renewable resources. Plants 2021, 10, 1751. [Google Scholar] [CrossRef] [PubMed]
- GBIF. Global Biodiversity Information Facility. Available online: https://www.gbif.org/ (accessed on 20 August 2021).
- Korochinsky, A.V.; Korochinskaya, V.V.; Zilfikarov, I.N.; Daironas, J.V. Innovative technology of tablets «Diosklephyt» based on Dioscorea caucasica Lypsky. Drug Dev. Regist. 2015, 4, 74–81. [Google Scholar]
- Guo, X.; Ding, X. Dioscin suppresses the viability of ovarian cancer cells by regulating the VEGFR2 and PI3K/AKT/MAPK signaling pathways. Oncol. Lett. 2018, 15, 9537–9542. [Google Scholar] [CrossRef] [Green Version]
- Yang, B.; Xu, B.; Zhao, H.; Wang, Y.; Zhang, J.; Li, C.; Wu, Q.; Cao, Y.; Li, Y.; Cao, F. Dioscin protects against coronary heart disease by reducing oxidative stress and inflammation via Sirt1/Nrf2 and p38 MAPK pathways. Mol. Med. Rep. 2018, 18, 973–980. [Google Scholar] [CrossRef] [Green Version]
- Son, I.S.; Kim, J.H.; Sohn, H.Y.; Son, K.H.; Kim, J.S.; Kwon, C.S. Antioxidative and hypolipidemic effects of diosgenin, a steroidal saponin of yam (Dioscorea spp.), on high-cholesterol fed rats. Biosci. Biotechnol. Biochem. 2007, 71, 3063–3071. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Q.; Sun, H.J.; Liu, S.M.; Jiang, X.H.; Wang, Q.Y.; Zhang, S.; Yu, D.H. Anti-inflammation effects of the total saponin fraction from Dioscorea nipponica Makino on rats with gouty arthritis by influencing MAPK signalling pathway. BMC Complement. Med. Ther. 2020, 20, 261. [Google Scholar] [CrossRef]
- Al Shukor, N.; Van Camp, J.; Gonzales, G.B.; Staljanssens, D.; Struijs, K.; Zotti, M.J.; Raes, K.; Smagghe, G. Angiotensin-converting enzyme inhibitory effects by plant phenolic compounds: A study of structure activity relationships. J. Agric. Food Chem. 2013, 61, 11832–11839. [Google Scholar] [CrossRef]
- Szakiel, A.; Grabarczyk, M.; Pączkowski, C.; Mieczkowski, A. Comparison of the profiles of non-glycosylated triterpenoids from leaves of plants of selected species of genus Dioscorea. Phytochem. Lett. 2017, 20, 350–355. [Google Scholar] [CrossRef]
- Price, E.J.; Wilkin, P.; Sarasan, V.; Fraser, P.D. Metabolite profiling of Dioscorea (yam) species reveals underutilised biodiversity and renewable sources for high-value compounds. Sci. Rep. 2016, 6, 29136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bj, H.; Zy, L.; Gs, J.; Ym, L. Studies on an aqueous soluble active constituent of Chuan-Shan-Long (Dioscorea nipponica Makino). I. Isolation and identification of p-hydroxy benzyl tartaric acid (piscidic acid) (author’s transl). Acta Pharm. Sin. 1980, 15, 764–765. [Google Scholar]
- Zeng, X.; Liu, D.; Huang, L. Metabolome Profiling of Eight Chinese Yam (Dioscorea polystachya Turcz.) Varieties Reveals Metabolite Diversity and Variety Specific Uses. Life 2021, 11, 687. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Jia, Z.; Zhang, Z.; Wang, Y.; Liu, X.; Wang, L.; Lin, R. Analysis of chemical constituents of Melastoma dodecandrum Lour. by UPLC-ESI-Q-Exactive Focus-MS/MS. Molecules 2017, 22, 476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, W.; Zhang, C.; Zhao, D.; Wang, L.; Li, P.; Li, H. Discovery of hepatotoxic equivalent combinatorial markers from Dioscorea bulbifera tuber by fingerprint-toxicity relationship modeling. Sci. Rep. 2018, 8, 462. [Google Scholar] [CrossRef] [PubMed]
- Parveen, I.; Winters, A.; Threadgill, M.D.; Hauck, B.; Morris, P. Extraction, structural characterisation and evaluation of hydroxycinnamate esters of orchard grass (Dactylis glomerata) as substrates for polyphenol oxidase. Phytochem 2008, 69, 2799–2806. [Google Scholar] [CrossRef]
- Xue, M.; Shi, H.; Zhang, J.; Liu, Q.Q.; Guan, J.; Zhang, J.Y.; Ma, Q. Stability and degradation of caffeoylquinic acids under different storage conditions studied by high-performance liquid chromatography with photo diode array detection and high-performance liquid chromatography with electrospray ionization collision-induced dissociation tandem mass spectrometry. Molecules 2016, 21, 948. [Google Scholar]
- Zhou, L.; Shi, X.M.; Ren, X.M.; Zhang, J.P.; Qin, Z.H. Identification of phenolic components in the root and leaf of purple yam (Dioscorea alata) by UHPLC-DAD-ESI-MS/MS. Mod. Food Sci. Technol. 2016, 32, 310–315. [Google Scholar]
- Ammar, S.; Contreras, M.; del Mar Contreras, M.; Belguith-Hadrich, O.; Bouaziz, M.; Segura-Carretero, A. New insights into the qualitative phenolic profile of Ficus carica L. fruits and leaves from Tunisia using ultra-high-performance liquid chromatography coupled to quadrupole-time-of-flight mass spectrometry and their antioxidant activity. RSC Adv. 2015, 5, 20035–20050. [Google Scholar] [CrossRef]
- Ablajan, K.; Tuoheti, A. Fragmentation characteristics and isomeric differentiation of flavonol O -rhamnosides using negative ion electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2012, 27, 451–460. [Google Scholar] [CrossRef]
- Lebot, V.; Malapa, R.; Molisalé, T. Development of HP-TLC method for rapid quantification of sugars, catechins, phenolic acids and saponins to assess yam (Dioscorea spp.) tuber flour quality. Plant Genet. Resour. 2019, 17, 62–72. [Google Scholar] [CrossRef]
- Wan, D.; Yang, H.; Yan, C.; Song, F.; Liu, Z.; Liu, S. Differentiation of glucose-containing disaccharides isomers by fragmentation of the deprotonated non-covalent dimers using negative electrospray ionization tandem mass spectrometry. Talanta. 2013, 115, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Khoo, H.E.; Azlan, A.; Abd Kadir, N.A.A. Fatty acid profile, phytochemicals, and other substances in Canarium odontophyllum fat extracted using supercritical carbon dioxide. Front. Chem. 2019, 7, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- LIPID MAPS. Available online: https://www.lipidmaps.org (accessed on 12 December 2021).
- Xie, T.; Liang, Y.; Hao, H.; A, J.; Xie, L.; Gong, P.; Dai, C.; Liu, L.; Kang, A.; Zheng, X.; et al. Rapid identification of ophiopogonins and ophiopogonones in Ophiopogon japonicus extract with a practical technique of mass defect filtering based on high resolution mass spectrometry. J. Chromatogr. A 2012, 1227, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, H.Y.; Chao, L.P.; Qu, L.; Ruan, J.Y.; Liu, Y.X.; Dong, Y.Z.; Han, L.F.; Wang, T. Anti-inflammatory steroids from the rhizomes of Dioscorea septemloba Thunb. Steroids 2016, 112, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Qin, H.; Yu, D. 3.11 Sterodal saponins. In ¹H NMR Handbook of Natural Products: Saponins; De Gruyter: Berlin, Germany; Boston, MA, USA, 2021; Volume 2, pp. 349–486. [Google Scholar]
- Osagie, A.U.; Opute, F.I. Major lipid constituents of Dioscorea rotundata tuber during growth and maturation. J. Exp. Bot. 1981, 32, 737–740. [Google Scholar] [CrossRef]
- Shajeela, P.S.; Tresina, P.S.; Mohan, V.R. Fatty acid composition of wild yam (Dioscorea spp.). Trop. Subtrop. Agroecosyst. 2013, 16, 35–38. [Google Scholar]
- IUBMB. Enzyme Nomenclature: Recommendations (1992) of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology. EC 3.2.1.20. Available online: https://www.qmuil.ac.uk/enzyme/EC3/2/1/20.html (accessed on 20 April 2022).
- Carbohydrate-Active Enzymes Database (CAZy). Available online: www.cazy.org/GH13.html (accessed on 20 April 2022).
- ExplorEnz–The Enzyme Database. ExplorEnz: EC 3.2.1.20. Available online: Enzyme-database.org (accessed on 20 April 2022).
- Sigma-Aldrich protocol. Enzymatic Assay of α-GLUCOSIDASE (EC 3.2.1.20) p-Nitrophenyl α-D-Glucoside as Substrate. Sigma-Aldrich. [Online]. Available online: http://www.sigmaaldric.com/technical-documents/protocol/enzymatic-assay-of-a-glucosidase.html (accessed on 19 April 2022).
- Lineweaver, H.; Burk, D. The determination of enzyme dissociation constants. J. Am. Chem. Soc. 1934, 56, 658–666. [Google Scholar] [CrossRef]
- Page, M.I.; Engel, P.C. (Eds.) Chapter 3 Enzyme kinetics. In The Chemnistry of Enzyme Action; Reprinted from New Comprehensive Biochemistry; Elsevier: Amsterdam, The Netherlands, 1984; Volume 6, pp. 73–110. [Google Scholar]
- Berg, J.M.; Stryer, T.J.L. Appendix: Vmax and Km can be determined by double-reciprocal plots. In Biochemistryi, 5th ed.; W H Freeman: New York, NY, USA, 2002. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V., Jr.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]
- Ghosh, S.; Ahire, M.; Patil, S.; Jabgunde, A.; Bhat Dusane, M.; Joshi, B.N.; Pardesi, K.; Jachak, S.; Dhavale, D.D.; Chopade, B.A. Antidiabetic activity of Gnidia glaucaand and Dioscorea bulbifera: Potent amylase and glucosidase inhibitors. Evid. Based Complement. Alter. Med. 2012, 2012, 929051. [Google Scholar]
- Avula, B.; Wang, Y.H.; Ali, Z.; Smillie, T.J.; Khan, I.A. Chemical fingerprint analysis and quantitative determination of steroidal compounds from Dioscorea villosa, Dioscorea species and dietary supplements using UHPLC-ELSD. Biomed. Chromatogr. 2014, 28, 281–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vendl, O.; Wawrosch, C.; Noe, C.; Molina, C.; Kahl, G.; Kopp, B. Diosgenin contents and DNA fingerprint screening of various yam (Dioscorea sp.) genotypes. Z. Nat. C 2006, 61, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Teng, H.; Chen, L. α-Glucosidase and α-amylase inhibitors from seed oil: A review of liposoluble substance to treat diabetes. Crit. Rev. Food Sci. Nutr. 2017, 57, 3438–3448. [Google Scholar] [CrossRef] [PubMed]
- Navarro del Hierroa, J.; Herrera, T.; Fornari, T.; Reglero, G.; Martin, D. The gastrointestinal behavior of saponins and its significance for their bioavailability and bioactivities. J. Funct. Foods. 2018, 40, 484–497. [Google Scholar] [CrossRef]
- Li, Y.Q.; Zhou, F.C.; Gao, F.; Bian, J.S.; Shan, F. Comparative evaluation of quercetin, isoquercetin and rutin as inhibitors of α-glucosidase. J. Agric. Food Chem. 2009, 57, 11463–11468. [Google Scholar] [CrossRef]
- Meng, S.; Cao, J.; Feng, Q.; Peng, J.; Hu, Y. Roles of chlorogenic acid on regulating glucose and lipids metabolism: A review. Evid. Based Complement. Alter. Med. 2013, 2013, 801457. [Google Scholar] [CrossRef]
- Šola, I.; Poljuha, D.; Mikulic-Petkovsek, M.; Davosir, D.; Pinterić, M.; Bilić, J.; Veberic, R.; Hudina, M.; Rusak, G. Biopotential of underutilized Rosaceae inflorescences: LC-DAD-MS phytochemical profiles associated with antioxidant, antidiabetic, anti-inflammatory and antiproliferative activity in vitro. Plants 2022, 11, 271. [Google Scholar] [CrossRef]
- Zhang, Y.; Bai, B.; Yan, Y.; Liang, J.; Guan, X. Bound polyphenols from red quinoa prevailed over free polyphenols in reducing postprandial blood glucose rises by inhibiting α-glucosidase activity and starch digestion. Nutrients 2022, 14, 728. [Google Scholar] [CrossRef]
- Ostberg-Potthoff, J.J.; Berger, K.; Richling, E.; Winterhalter, P. Activity-guided fractionation of red fruit extracts for the identification of compounds influencing glucose metabolism. Nutrients 2019, 11, 1166. [Google Scholar] [CrossRef] [Green Version]
- Rondanelli, M.; Riva, A.; Petrangolini, G.; Allegrini, P.; Bernardinelli, L.; Fazia, T.; Peroni, G.; Gasparri, C.; Nichetti, M.; Faliva, M.A.; et al. The metabolic effects of Cynara supplementation in overweight and obese class I subjects with newly detected impaired fasting glycemia: A double-blind, placebo-controlled, randomized clinical trial. Nutrients 2020, 12, 3298. [Google Scholar] [CrossRef]
- Chempedia. Available online: http://chempedia.info/info/eadie_hofstee (accessed on 23 April 2022).
- Cho, Y.S.; Lim, H.S. Comparison of various estimation methods for the parameters of Michaelis–Menten equation based on in vitro elimination kinetic simulation data. Transl. Clin. Pharmacol. 2018, 26, 39–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Owusu-Apenten, R.K. Food Enzymes. In Introduction to Food Chemistry, 1st ed.; CRC Press: Boca Raton, FL, USA, 2004; pp. 174–185. [Google Scholar]
- Marasović, M.; Marasović, T.; Miloš, M. Robust nonlinear regression in enzyme kinetic parameters estimation. J. Chem. 2017, 2017, 6560983. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, N.; Nagashima, T. Autolysate and enzymatic hydrolysates from yam (Dioscorea opposita Thunb) tuber mucilage tororo have antioxidant and angiotensin I-converting enzyme inhibitory activities. J. Food Agric. Environ. 2007, 5, 39–43. [Google Scholar]
- López-Fernández-Sobrino, R.; Soliz-Rueda, J.R.; Margalef, M.; Arola-Arnal, A.; Suárez, M.; Bravo, F.I.; Muguerza, B. ACE Inhibitory and antihypertensive activities of wine lees and relationship among bioactivity and phenolic profile. Nutrients 2021, 13, 679. [Google Scholar] [CrossRef] [PubMed]
- Amato, A.; Caldara, G.-F.; Nuzzo, D.; Baldassano, S.; Picone, P.; Rizzo, M.; Mulè, F.; Di Carlo, M. NAFLD and atherosclerosis are prevented by a natural dietary supplement containing curcumin, silymarin, guggul, chlorogenic acid and inulin in mice fed a high-fat diet. Nutrients 2017, 9, 492. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.J.; Yang, L.; Zhao, Q.; Caen, J.P.; He, H.Y.; Jin, Q.H.; Guo, L.H.; Alemany, M.; Zhang, L.Y.; Shi, Y.F. Induction of acetylcholinesterase expression during apoptosis in various cell types. Cell Death Differ. 2002, 9, 790–800. [Google Scholar] [CrossRef] [Green Version]
- Orhan, I.; Kartal, M.; Tosun, F.; Şener, B. Screening of various phenolic acids and flavonoid derivatives for their anticholinesterase potential. Z. Nat. C 2007, 62, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Cai, J.; Fang, Z.; Li, S.; Huang, Z.; Tang, Z.; Luo, Q.; Chen, H. The composition and anti-aging activities of polyphenol extract from Phyllanthus emblica L. fruit. Nutrients 2022, 14, 857. [Google Scholar] [CrossRef]
- Tenfen, A.; Vechi, G.; Cechinel-Zanchett, C.C.; Lorenzett, T.S.; Reginato-Couto, C.E.; Siebert, D.A.; Vitali, L.; Micke, G.; Klein-Júnior, L.C.; Cechinel Filho, V. Phenolic profile by HPLC-ESI-MS/MS of six Brazilian Eugenia species and their potential as cholinesterase inhibitors. Nat. Prod. Res. 2021, 35, 2608–2611. [Google Scholar] [CrossRef]
- Sigma-Aldrich Product. α-Glucosidase from Saccharomyces cerevisiae. Sigma-Aldrich. Available online: https://www.sigmaaldrich.com/LT/en/product/sigma/g5003 (accessed on 20 April 2022).
- Sigma-Aldrich Product. α-Amylase from Porcine Pancreas. Sigma-Aldrich. Available online: https://www.sigmaaldrich.com/product/sigma/a6255 (accessed on 19 April 2022).
- Sigma-Aldrich Product. Lung Acetone Powder from Rabbit. Sigma-Aldrich. Available online: https://www.sigmaaldrich.com/product/sigma/l0756 (accessed on 20 April 2022).
- Sigma-Aldrich Product. Acetylcholinesterase from Electrophorus electricus. Sigma-Aldrich. Available online: https://www.sigmaaldrich.com/product/sigma/c3389 (accessed on 20 April 2022).
- Vytautas Magnus University Botanical Garden in Kaunas, Lithuania. Available online: http://www.bgci.org/garden.php?id=119 (accessed on 23 April 2022).
- Waters ACQUITY UPLC System. Ultra Perfomance LC Separation Science Redefined. (Part Number 720001136EN). Available online: https://www.waters.com/webassets/cms/library/docs/720001136en.pdf (accessed on 24 April 2022).
- Swartz, M.E. UPLC: An introduction and review. J. Liq. Chrom. 2005, 28, 1253–1263. [Google Scholar] [CrossRef]
- Waters Corporation. Waters, the Science of What’s Possible. ACQUITY UPLC and ACQUITY PREMIER BEH Columns (Part Number 715001371). Available online: www.waters.com (accessed on 24 April 2022).
- Vermeirssen, V.; Van Camp, J.; Verstraete, W. Optimisation and validation of an angiotensin-converting enzyme inhibition assay for the screening of bioactive peptides. J. Biochem. Biophys. Methods 2002, 51, 75–87. [Google Scholar] [CrossRef]
- Donath-Nagy Gabriella, Vancea Szende, Imre Silvia Comparative study if captopril Derivatization Reaction by LC-UV, LC-MS and CE-UV methods. Croatica Chemica Acta 2011, 84, 423–427. [CrossRef]
- Sigma-Aldrich Protocol. Enzymatic Assay of α-Amylase (EC 3.2.1.1) Sigma-Aldrich. Available online: http://www.sigmaaldric.com/technical-documents/protocol/enzymatic-assay-of-a-amylase.html (accessed on 19 April 2022).
- Eyer, P.; Worek, F.; Kiderlen, D.; Sinko, G.; Stuglin, A.; Simeon-Rudolf, V.; Reiner, E. Molar absorption coefficients for the reduced Ellman reagent: Reassessment. Anal. Biochem. 2003, 312, 224–227. [Google Scholar] [CrossRef]
| ID No. | tR, min | m/z [M − H]− Found | m/z [M − H]− Calculated | Detected Ion(s) | Mass Error (ppm) | Compound | Leaves | Tubers | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Organic Acids | |||||||||
| 1 | 0.4 | 191.0561 | 191.0556 | 191[C7H11O6]− | −0.5 | Quinic acid | + | ||
| 0.7 # | 191.0562 | −0.6 | + | MS data | |||||
| 2 | 0.5 | 133.0142 | 133.0137 | 133[C4H5O5]− | −0.5 | Malic acid | + | [26,30] | |
| 0.8 # | 133.0142 | −0.5 | + | ||||||
| 3 | 0.5 | 173.0454 | 173.0450 | 173[C7H9O5]− | −0.4 | Shikimic acid | + | [26] | |
| 4 | 0.8 | 191.0196 | 191.0191 | 191[C6H7O7]− | −0.4 | Citric acid/isocitric acid | + | ||
| 1.2 # | 191.0200 | −0.8 | + | MS data | |||||
| 5 | 1.8 | 255.0512 | 255.0505 | 255[C11H11O7]− | −0.7 | Piscidic acid | + | [27] | |
| Hydroxycinnamates | |||||||||
| 6 | 1.5 | 707.1822 | 707.1823 | 707[C32H35O18]− [2M − H]− | −0.1 | + | |||
| 353.0875 | 353.0872 | 353[C16H17O9]− | −0.3 | 3-Caffeoylquinic acid * | [31] | ||||
| 1.6 | 707.1822 | 707.1823 | 707[C32H35O18]− [2M − H]− | −0.1 | + | ||||
| 8 | 355.0664 | 355.0665 | 355[C15H15O10]− | −0.1 | Caffeic acid O-glucuronide | ||||
| 9 | 297.0612 | 297.0610 | 297[C13H13O8]− | −0.2 | Caffeoylthreonic acid | + | [31] | ||
| 10 | 1.7 | 707.1827 | 707.1823 | 707[C32H35O18]− [2M − H]− | −0.4 | + | |||
| 353.0876 | 353.0872 | 353[C16H17O9]− | −0.4 | 5-Caffeoylquinic acid * | [26] | ||||
| 191.0559 | 191.0556 | 191[C7H11O6]− | −0.5 | ||||||
| 11 | 1.9 | 337.0930 | 337.0923 | 337[C16H17O8]− | −0.4 | Coumaroylquinic acid | + | ||
| 12 | 353.0877 | 353.0872 | 353[C16H17O9]− | −0.5 | 4-Caffeoylquinic acid | + | [31,32] | ||
| 191.0564 | 191[C7H11O6]− | −0.7 | + | ||||||
| 13 | 2.0 | 335.0771 | 335.0766 | 335[C16H15O8]− | −0.5 | Caffeoylshikimic acid | + | [33] | |
| 14 | 2.1 | 367.1031 | 367.1029 | 367[C17H19O9]− | −0.2 | Feruloylquinic acid | + | [31] | |
| Flavonoids | |||||||||
| 15 | 2.3 | 609.1463 | 609.1455 | 609[C27H29O16]− | −0.8 | Quercetin-3-O-rutinoside (rutin) * | + | ||
| 16 | 2.4 | 463.0879 | 463.0876 | 463[C21H19O12]− | −0.3 | Quercetin-3-O-glucoside (isoquercitrin) * | + | ||
| 17 | 505.0995 | 505.0982 | 505[C23H21O13]− | −1.3 | Quercetin-O-acetylhexoside | + | [34] | ||
| 18 | 2.5 | 549.0883 | 549.0880 | 549[C24H21O15]− | −0.3 | Quercetin 3-O-(6′′-malonyl) glucoside | + | ||
| 19 | 2.6 | 447.0930 | 447.0927 | 447[C21H19O11]− | −0.3 | Quercetin-3-O-rhamnoside (quercitrin) | + | [35] | |
| 20 | 2.7 | 533.0934 | 533.0931 | 533[C24H21O14]− | −0,27 | Quercetin-3-O-malonyl(rhamnoside) | + | MS data | |
| 21 | 489.1036 | 489.1033 | 489[C23H21O12]− | −0.3 | Quercetin-3-O-acetyl(rhamnoside) | + | |||
| Sugars | |||||||||
| 22 | 0.6 | 179.0561 | 179.0555 | 179[C6H11O6]− | −0.6 | Hexose | + | ||
| 23 | 0.7 | 341.1089 | 341.1083 | 341[C12H21O11]− | −0.6 | Sucrose | + | [26,36,37] | |
| 24 | 0.7 | 683.2252 | 683.2246 | 683[C24H43O22]− [2M − H]− | −0.6 | Unseparated sugars | + | [37] | |
| 25 | 0.7 | 1025.3414 | 1025.3408 | 1025[C36H63O33]− [3M − H]− | −0.6 | Unseparated sugars | + | [37] | |
| Fatty Acids and Conjugates | |||||||||
| 26 | 4.6 | 329.2335 | 329.2328 | 329[C18H33O5]− | −0.7 | Trihydroxy octadecenoic acid | + | [38] | |
| 27 | 5.8 | 293.2120 | 293.2117 | 293[C18H29O3]− | −0.3 | Hydroxy octadecatrienoic acid | + | [38] | |
| 28 | 6.8 | 358.2601 | 358.2593 | 358[C19H36NO5]− | −0.8 | Hydroxy dodecanoylcarnitine | + | LMFA07070032 [39] | |
| 29 | 6.9 8.0 | 295.2280 295.2281 | 295.2273 | 295[C18H31O3]− | −0.7 −0.8 | Hydroxy octadecadienoic acid (linolenic acid) | + | [38] | |
| 30 | 8.4 | 271.2279 | 271.2273 | 271[C16H31O3]− | −0.6 | Hydroxy hexadecenoic acid | + | [38] | |
| 31 | 8.8 | 279.2330 | 279.2324 | 279[C18H31O2]− | −0.6 | Octadecadienoic acid (Linoleic acid) | + | [38] | |
| Other Compounds | |||||||||
| 32 | 0.3 | 225.0617 | 225.0610 | 225[C7H13O8]− | −0.7 | Glucoheptonic acid | + | [29] | |
| 33 | 0.9 | 290.0881 | 290.0875 | 290[C11H16NO8]− | −0.6 | Neu5Ac2en | + | CID65309 | |
| 34 | 128.0352 | 128.0347 | 128 [C5H6NO3]− | −0.5 | Pyroglutamic acid | + | [28] | ||
| 35 | 1.4 | 345.1188 | 345.1186 | 345[C15H21O9]− | −0.2 | Aucubin | + | ||
| 36 | 3.3 | 1109.5383 | 1109.5379 | 1109[C52H85O25]− | −0.4 | Steroidal glycoside | + | [40] | |
| 37 | 3.7 | 1093.5431 | 1093.5431 | 1093[C52H85O24]− | −0.0 | Steroidal glycoside | + | [40,41] | |
| 38 | 4.3 | 1075.5327 | 1075.5325 | 1075[C52H83O23]− [M − H-146]− | −0.2 | Steroidal glycoside (ophiopogonin derivative?) | + | [40] | |
| 39 | 6.5 | 929.4754 | 929.4746 | 929[C46H73O19]− | −0.1 | Steroidal glycoside | + | [40] | |
| 40 | 6.6 | 913.4798 | 913.4797 | 913[C46H73O18]− | −0.1 | Steroidal glycoside | + | [40] | |
| 41 | 6.8 | 767.4228 | 767.4217 | 767[C40H63O14]− | −1.1 | Steroidal saponin (pentandroside B?) | + | [42] | |
| Unidentified Compounds | |||||||||
| 42 | 6.1 | 559.3117 | 559.3118 | 559[C28H47O11]− | −0.1 | Unidentified | + | MS data | |
| 43 | 6.6 | 483.2727 | 483.2719 | 483[C25H35N6O4]− | −0.8 | Unidentified peptide | + | MS data | |
| 44 | 5.6 | 721.3652 | 721.3646 | 721[C34H57O16]− | −0.6 | Unidentified galactolipid | + | MS data | |
| 45 | 6.8 | 723.3809 | 723.3803 | 723[C34H59O16]− | −0.6 | Unidentified galactolipid | + | MS data | |
| Conc., µg/mL | With 0.1 M Na2CO3 | Without 0.1 M Na2CO3 | Percentage Difference | p-Value |
|---|---|---|---|---|
| 3.125 | 16.93 ± 1.43 | 11.94 ± 1.22 | −4.990 | 0.01 |
| 6.25 | 32.73 ± 0.54 | 26.47 ± 1.43 | −6.260 | 0.0021 |
| 12.5 | 42.81 ± 0.67 | 39.32 ± 1.75 | −3.490 | 0.0321 |
| 25 | 52.49 ± 0.47 | 52.31 ± 1.64 | −0.180 | 0.8639 |
| 50 | 67.69 ± 0.26 | 72.57 ± 1.52 | 4.880 | 0.0054 |
| 100 | 59.30 ± 1.1 | 78.49 ± 2.39 | 19.19 | 0.0002 |
| Inhibitor | Parameters | Enzymatic Reaction I | Enzymatic Reaction II | Mode |
|---|---|---|---|---|
| None | Km, mg/L | 80.873 ± 0.26 | 78.55 ± 0.11 | |
| Vmax, mg/L·min | 0.597 ± 0.07 | 1.43 ± 0.183 | ||
| CE | 0.007 | 0.018 | ||
| Leaf extract, | Km(app), mg/L | 29.761 ± 1.54 | 39.984 ± 0.31 | |
| 15 µg/mL | Vmax(app), mg/L·min | 0.157 ± 0.03 | 0.402 ± 0.041 | Mixed-type |
| Ki, µg/mL | 5.35 | 5.86 | ||
| CE | 0.005 | 0.01 | ||
| Leaf extract, | Km(app), mg/L | 33.615 ± 0.75 | 86.909 ± 0.16 | |
| 25 µg/mL | Vmax(app), mg/L·min | 0.134 ± 0.004 | 0.449 ± 0.026 | Mixed-type |
| Ki, µg/mL | 7.23 | 11.44 | ||
| CE | 0.004 | 0.005 | ||
| None | Km, mg/L | 31.446 ± 0.28 | - | |
| Vmax, mg/L·min | 1.023 ± 0.23 | - | ||
| CE | 0.032 | |||
| Km(app), mg/L | 69.34 ± 0.31 | - | ||
| Tuber extract, | Vmax(app), mg/L·min | 0.991 ± 0.22 | - | Competitive |
| 200 µg/mL | Ki, µg/mL | 165.97 | - | |
| CE | 0.014 | - | ||
| Km(app), mg/L | 83.125 ± 0.51 | - | ||
| Tuber extract, | Vmax(app), mg/L·min | 1.034 ± 0.38 | - | |
| 500 µg/mL | Ki, µg/mL | 304.32 | - | |
| CE | 0.011 | - |
| No Inhibitor | D. caucasica, µg/mL | ||||
|---|---|---|---|---|---|
| - | 25 | 40 | 50 | 80 | 100 |
| 0.138 ± 0.001 a (0) | 0.1 ± 0.002 b (27.55) | 0.082 ± 0.002 c (40.58) | 0.078 ± 0.002 d (43.48) | 0.076 ± 0.002 e (45.31) | 0.074 ± 0.002 e (45.85) |
| Type of Inhibition | Effect on Vmax | Effect on Km | Effect on Slope of L–B plot | Position of Intersection of L–B plots |
|---|---|---|---|---|
| Competitive | No change | Increase | Increase | Ordinate axis |
| Uncompetitive | Decrease | Decrease | No change | None |
| Non-competitive (a) Simple | Decrease | No change | Increase | Abscissa axis |
| (b) Mixed: Ki < Ki(A) | Decrease | Increase | Increase | Second quadrant |
| (c) Mixed: Ki > Ki(A) | Decrease | Decrease | Increase | Third quadrant |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adomėnienė, A.; Pukalskas, A.; Ragažinskienė, O.; Venskutonis, P.R. Health Beneficial Phytochemicals in Dioscorea caucasica Lipsky Leaves and Tubers and Their Inhibitory Effects on Physiologically Important Enzymes. Plants 2022, 11, 1341. https://doi.org/10.3390/plants11101341
Adomėnienė A, Pukalskas A, Ragažinskienė O, Venskutonis PR. Health Beneficial Phytochemicals in Dioscorea caucasica Lipsky Leaves and Tubers and Their Inhibitory Effects on Physiologically Important Enzymes. Plants. 2022; 11(10):1341. https://doi.org/10.3390/plants11101341
Chicago/Turabian StyleAdomėnienė, Aušra, Audrius Pukalskas, Ona Ragažinskienė, and Petras Rimantas Venskutonis. 2022. "Health Beneficial Phytochemicals in Dioscorea caucasica Lipsky Leaves and Tubers and Their Inhibitory Effects on Physiologically Important Enzymes" Plants 11, no. 10: 1341. https://doi.org/10.3390/plants11101341
APA StyleAdomėnienė, A., Pukalskas, A., Ragažinskienė, O., & Venskutonis, P. R. (2022). Health Beneficial Phytochemicals in Dioscorea caucasica Lipsky Leaves and Tubers and Their Inhibitory Effects on Physiologically Important Enzymes. Plants, 11(10), 1341. https://doi.org/10.3390/plants11101341







