Green Revolution to Gene Revolution: Technological Advances in Agriculture to Feed the World
Abstract
:1. Introduction
2. Mutation Breeding to Increase Plant Varieties
3. Breeding Strategies to Increase Crop Yield
4. The Rise of Recombinant DNA Technology and Genetically Modified (GM) Crops
5. A New Era of Genome Editing Using Sequence-Specific Nuclease (SSN)-Based Tools
6. The Rapid Emergence of the CRISPR/Cas System for High Specificity Gene Editing
6.1. Variety of CRISPR Enzymes and Current Applications in Crops
6.2. CRISPR Reagents Delivery Systems
7. SSN-based Genome Editing: Good or Bad?
8. SSN-based Genome Editing: A Modern Technology within a Conventional Regulatory Framework
9. Benefits of Agricultural Biotechnology
9.1. Improved Crop Yield and Efficient Land Use
9.2. Economic Benefits to Farmers and Consumers
9.3. Reduced Environmental Impacts of Agriculture
9.4. Increased Tolerance to Crop Diseases
9.5. Nutrient Enhancement of Staple Crops
9.6. Production of Plant-Based Pharmaceuticals
10. Concerns about the Effects of Agricultural Biotechnology on Human Health and the Environment
10.1. Effects on Human Health
10.2. Long Term Effects on Genetic Diversity
10.3. Over-Reliance on Mega-Companies in the Agricultural Market
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- The Food and Agriculture Organization (FAO). The Future of Food and Agriculture: Alternative Pathways to 2050; Summary Version; FAO: Rome, Italy, 2018; pp. 1–60. [Google Scholar]
- The Food and Agriculture Organization (FAO). Small Family Farmers Produce a Third of the World’s Food. Available online: https://www.fao.org/newsroom/detail/Small-family-farmers-produce-a-third-of-the-world-s-food/en (accessed on 5 March 2022).
- The Food and Agriculture Organization (FAO). Staple Foods: What Do People Eat? Available online: https://www.fao.org/3/u8480e/u8480e07.htm#:~:text=The%20world%20has%20over%2050,animal%20products%20(7%20percent) (accessed on 5 March 2022).
- The International Service for the Acquisition of Agri-biotech Applications (ISAAA). Pocket K No. 13: Conventional Plant Breeding. Available online: https://www.isaaa.org/resources/publications/pocketk/13/default.asp (accessed on 5 March 2022).
- Ania, W.; Mark, W. History of Agricultural Biotechnology: How Crop Development has Evolved. Available online: https://www.nature.com/scitable/knowledge/library/history-of-agricultural-biotechnology-how-crop-development-25885295/ (accessed on 5 March 2022).
- Orton, T.J. Chapter 12—protection of proprietary plant germplasm. In Horticultural Plant Breeding; Orton, T.J., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 221–230. [Google Scholar]
- Pingali, P.L. Green revolution: Impacts, limits, and the path ahead. Proc. Natl. Acad. Sci. USA 2012, 109, 12302–12308. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Dijk, M.; Morley, T.; Rau, M.L.; Saghai, Y. A meta-analysis of projected global food demand and population at risk of hunger for the period 2010–2050. Nat. Food 2021, 2, 494–501. [Google Scholar] [CrossRef]
- United Nations (UN). World Population Prospects 2019: Highlights; (ST/ESA/SER.A/423); UN: New York, NY, USA, 2019. [Google Scholar]
- Karunarathna, N.L.; Patiranage, D.S.R.; Harloff, H.J.; Sashidhar, N.; Jung, C. Genomic background selection to reduce the mutation load after random mutagenesis. Sci. Rep. 2021, 11, 19404. [Google Scholar] [CrossRef] [PubMed]
- Bailey-Serres, J.; Parker, J.E.; Ainsworth, E.A.; Oldroyd, G.E.D.; Schroeder, J.I. Genetic strategies for improving crop yields. Nature 2019, 575, 109–118. [Google Scholar] [CrossRef] [Green Version]
- Viana, V.E.; Pegoraro, C.; Busanello, C.; Costa de Oliveira, A. Mutagenesis in rice: The basis for breeding a new super plant. Front. Plant Sci. 2019, 10, 1326. [Google Scholar] [CrossRef] [Green Version]
- Muller, H.J. Artificial transmutation of the gene. Science 1927, 66, 84–87. [Google Scholar] [CrossRef]
- Stadler, L.J. Mutations in barley induced by X-rays and radium. Science 1928, 68, 186–187. [Google Scholar] [CrossRef]
- Stadler, L.J. Recovery following genetic deficiency in maize. Proc. Natl. Acad. Sci. USA 1930, 16, 714–720. [Google Scholar] [CrossRef] [Green Version]
- Pacher, M.; Puchta, H. From classical mutagenesis to nuclease-based breeding—directing natural DNA repair for a natural end-product. Plant J. 2017, 90, 819–833. [Google Scholar] [CrossRef]
- Mei, M.; Deng, H.; Lu, Y.; Zhuang, C.; Liu, Z.; Qiu, Q.; Qiu, Y.; Yang, T.C. Mutagenic effects of heavy ion radiation in plants. Adv. Space Res. 1994, 14, 363–372. [Google Scholar] [CrossRef]
- Dhaliwal, A.K.; Mohan, A.; Sidhu, G.; Maqbool, R.; Gill, K.S. An ethylmethane sulfonate mutant resource in pre-green revolution hexaploid wheat. PLoS ONE 2015, 10, e0145227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumawat, S.; Rana, N.; Bansal, R.; Vishwakarma, G.; Mehetre, S.T.; Das, B.K.; Kumar, M.; Kumar Yadav, S.; Sonah, H.; Sharma, T.R.; et al. Expanding avenue of fast neutron mediated mutagenesis for crop improvement. Plants 2019, 8, 164. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kambhampati, S.; Aznar-Moreno, J.A.; Hostetler, C.; Caso, T.; Bailey, S.R.; Hubbard, A.H.; Durrett, T.P.; Allen, D.K. On the inverse correlation of protein and oil: Examining the effects of altered central carbon metabolism on seed composition using soybean fast neutron mutants. Metabolites 2019, 10, 18. [Google Scholar] [CrossRef] [Green Version]
- Islam, N.; Krishnan, H.B.; Natarajan, S. Proteomic profiling of fast neutron-induced soybean mutant unveiled pathways associated with increased seed protein content. J. Proteome Res. 2020, 19, 3936–3944. [Google Scholar] [CrossRef] [PubMed]
- Holme, I.B.; Gregersen, P.L.; Brinch-Pedersen, H. Induced genetic variation in crop plants by random or targeted mutagenesis: Convergence and differences. Front. Plant Sci. 2019, 10, 1468. [Google Scholar] [CrossRef]
- Kaiser, N.; Douches, D.; Dhingra, A.; Glenn, K.C.; Herzig, P.R.; Stowe, E.C.; Swarup, S. The role of conventional plant breeding in ensuring safe levels of naturally occurring toxins in food crops. Trends Food Sci. Technol. 2020, 100, 51–66. [Google Scholar] [CrossRef]
- López-Caamal, A.; Tovar-Sánchez, E. Genetic, morphological, and chemical patterns of plant hybridization. Rev. Chil. Hist. Nat. 2014, 87, 16. [Google Scholar] [CrossRef] [Green Version]
- Charlesworth, D. Evolution of plant breeding systems. Curr. Biol. 2006, 16, R726–R735. [Google Scholar] [CrossRef] [Green Version]
- Vallejo-Marin, M.; Walker, C.; Friston-Reilly, P.; Solis-Montero, L.; Igic, B. Recurrent modification of floral morphology in heterantherous solanum reveals a parallel shift in reproductive strategy. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 2014, 369, 20130256. [Google Scholar] [CrossRef] [Green Version]
- Erickson, L.R.; Atnaseo, C. 4.10—transgenic crops with producer-oriented traits: Development, application, and impact. In Comprehensive Biotechnology, 2nd ed.; Moo-Young, M., Ed.; Academic Press: Oxford, UK, 2011; pp. 121–131. [Google Scholar]
- Ghețe, A.B.; Haș, V.; Vidican, R.; Copândean, A.; Ranta, O.; Moldovan, C.M.; Crișan, I.; Duda, M.M. Influence of detasseling methods on seed yield of some parent inbred lines of turda maize hybrids. Agronomy 2020, 10, 729. [Google Scholar] [CrossRef]
- Yu, D.; Gu, X.; Zhang, S.; Dong, S.; Miao, H.; Gebretsadik, K.; Bo, K. Molecular basis of heterosis and related breeding strategies reveal its importance in vegetable breeding. Hortic. Res. 2021, 8, 120. [Google Scholar] [CrossRef] [PubMed]
- Leducq, J.B.; Gosset, C.C.; Gries, R.; Calin, K.; Schmitt, E.; Castric, V.; Vekemans, X. Self-incompatibility in Brassicaceae: Identification and characterization of SRK-like sequences linked to the S-locus in the tribe Biscutelleae. G3 Genes Genomes Genet. 2014, 4, 983–992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, F.; Yang, X.; Zhao, N.; Hu, Z.; Mackenzie, S.A.; Zhang, M.; Yang, J. Exploiting sterility and fertility variation in cytoplasmic male sterile vegetable crops. Hortic. Res. 2022, 9, uhab039. [Google Scholar] [CrossRef] [PubMed]
- Munoz-Sanz, J.V.; Zuriaga, E.; Cruz-Garcia, F.; McClure, B.; Romero, C. Self-(in)compatibility systems: Target traits for crop-production, plant breeding, and biotechnology. Front. Plant Sci. 2020, 11, 195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, F.; Butz, P.W. The gene revolution genetically modified crops. In The Future of Genetically Modified Crops, 1st ed.; Lessons from the Green Revolution; RAND Corporation: Santa Monica, CA, USA, 2004; pp. 39–64. [Google Scholar]
- Cohen, S.N.; Chang, A.C.; Boyer, H.W.; Helling, R.B. Construction of biologically functional bacterial plasmids in vitro. Proc. Natl. Acad. Sci. USA 1973, 70, 3240–3244. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kado, C.I. Historical account on gaining insights on the mechanism of crown gall tumorigenesis induced by Agrobacterium Tumefaciens Front. Microbiol. 2014, 5, 340. [Google Scholar] [CrossRef]
- Bevan, M.W.; Flavell, R.B.; Chilton, M.D. A chimaeric antibiotic resistance gene as a selectable marker for plant cell transformation. Biotechnology 1983, 24, 367–370. [Google Scholar] [CrossRef]
- Abel, P.P.; Nelson, R.S.; De, B.; Hoffmann, N.; Rogers, S.G.; Fraley, R.T.; Beachy, R.N. Delay of disease development in transgenic plants that express the tobacco mosaic virus coat protein gene. Science 1986, 232, 738–743. [Google Scholar] [CrossRef]
- Vaeck, M.; Reynaerts, A.; Höfte, H.; Jansens, S.; De Beuckeleer, M.; Dean, C.; Zabeau, M.; Montagu, M.V.; Leemans, J. Transgenic plants protected from insect attack. Nature 1987, 328, 33–37. [Google Scholar] [CrossRef]
- Rottenberg, A. Has agriculture dispersed worldwide from a single origin? Genet. Resour. Crop Evol. 2017, 64, 1107–1113. [Google Scholar] [CrossRef]
- Mendel, G. Verhandlungen des naturforschenden vereines zu brünn. Verh. Naturf. Ver. Brünn 1866, 4, 3–47. [Google Scholar]
- Cavara, F. Eziologia di alcune malattie di piante coltivate. Le Stazioni Sper. Agrar. Ital. 1897, 30, 482–509. [Google Scholar]
- Cavara, F. Tuberculosi della vite. Intorno alla eziologia di alcune malattie di piante cultivate. Le Stazioni Sper. Agrar. Ital. 1897, 30, 483–487. [Google Scholar]
- Sankaran, N. On the historical significance of Beijerinck and his contagium vivum fluidum for modern virology. Hist. Philos. Life Sci. 2018, 40, 41. [Google Scholar] [CrossRef] [PubMed]
- Smith, E.F.; Townsend, C.O. A plant-tumor of bacterial origin. Science 1907, 25, 671–673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Napoli, C.; Lemieux, C.; Jorgensen, R. Introduction of a chimeric chalcone synthase gene into petunia results in reversible co-suppression of homologous genes in trans. Plant Cell 1990, 2, 279–289. [Google Scholar] [CrossRef] [Green Version]
- Bawa, A.S.; Anilakumar, K.R. Genetically modified foods: Safety, risks and public concerns-a review. J. Food Sci. Technol. 2013, 50, 1035–1046. [Google Scholar] [CrossRef] [Green Version]
- Kramer, M.G.; Redenbaugh, K. Commercialization of a tomato with an antisense polygalacturonase gene: The FLAVR SAVR™ tomato story. Euphytica 1994, 79, 293–297. [Google Scholar] [CrossRef]
- Abbas, M.S.T. Genetically engineered (modified) crops (Bacillus thuringiensis crops) and the world controversy on their safety. Egypt. J. Biol. Pest Control 2018, 28, 52. [Google Scholar] [CrossRef]
- Sanahuja, G.; Banakar, R.; Twyman, R.M.; Capell, T.; Christou, P. Bacillus thuringiensis: A century of research, development and commercial applications. Plant Biotechnol. J. 2011, 9, 283–300. [Google Scholar] [CrossRef] [Green Version]
- Dill, G.M. Glyphosate-resistant crops: History, status and future. Pest Manag Sci. 2005, 61, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Al-Babili, S.; Klöti, A.; Zhang, J.; Lucca, P.; Beyer, P.; Potrykus, I. Engineering the provitamin a (beta-carotene) biosynthetic pathway into (carotenoid-free) rice endosperm. Science 2000, 287, 303–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adjusted Gross Income (AGI). Analysis of the genome sequence of the flowering plant Arabidopsis thaliana. Nature 2000, 408, 796–815. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The International Rice Genome Sequencing Project (IRGSP). The map-based sequence of the rice genome. Nature 2005, 436, 793–800. [Google Scholar] [CrossRef]
- Paine, J.A.; Shipton, C.A.; Chaggar, S.; Howells, R.M.; Kennedy, M.J.; Vernon, G.; Wright, S.Y.; Hinchliffe, E.; Adams, J.L.; Silverstone, A.L.; et al. Improving the nutritional value of golden rice through increased pro-vitamin a content. Nat. Biotechnol. 2005, 23, 482–487. [Google Scholar] [CrossRef]
- Shukla, V.K.; Doyon, Y.; Miller, J.C.; DeKelver, R.C.; Moehle, E.A.; Worden, S.E.; Mitchell, J.C.; Arnold, N.L.; Gopalan, S.; Meng, X.; et al. Precise genome modification in the crop species Zea mays using zinc-finger nucleases. Nature 2009, 459, 437–441. [Google Scholar] [CrossRef]
- Li, T.; Liu, B.; Spalding, M.H.; Weeks, D.P.; Yang, B. High-efficiency TALEN-based gene editing produces disease-resistant rice. Nat. Biotechnol. 2012, 30, 390–392. [Google Scholar] [CrossRef]
- Shan, Q.; Wang, Y.; Li, J.; Zhang, Y.; Chen, K.; Liang, Z.; Zhang, K.; Liu, J.; Xi, J.J.; Qiu, J.L.; et al. Targeted genome modification of crop plants using a CRISPR-Cas system. Nat. Biotechnol. 2013, 31, 686–688. [Google Scholar] [CrossRef]
- Waltz, E. GABA-enriched tomato is first CRISPR-edited food to enter market. Nat. Biotechnol. 2022, 40, 9–11. [Google Scholar] [CrossRef]
- Lindbo, J.A.; Silva-Rosales, L.; Proebsting, W.M.; Dougherty, W.G. Induction of a highly specific antiviral state in transgenic plants: Implications for regulation of gene expression and virus resistance. Plant Cell 1993, 5, 1749–1759. [Google Scholar] [CrossRef]
- Vega Rodríguez, A.; Rodríguez-Oramas, C.; Sanjuán Velázquez, E.; Hardisson de la Torre, A.; Rubio Armendáriz, C.; Carrascosa Iruzubieta, C. Myths and realities about genetically modified food: A risk-benefit analysis. Appl. Sci. 2022, 12, 2861. [Google Scholar] [CrossRef]
- James, C.; Krattiger, A.F. Global Review of the Field Testing and Commercialization of Transgenic Plants, 1986 to 1995: The First Decade of Crop Biotechnology; Isaaa Briefs No. 1; Isaaa: Ithaca, NY, USA, 1996; pp. 1–31. [Google Scholar]
- The International Service for the Acquisition of Agri-biotech Applications (ISAAA). Japan Starts Sale of Genome-Edited High-GABA Tomato. Available online: https://www.isaaa.org/kc/cropbiotechupdate/article/default.asp?ID=19024 (accessed on 5 March 2022).
- Que, Q.; Chilton, M.D.; de Fontes, C.M.; He, C.; Nuccio, M.; Zhu, T.; Wu, Y.; Chen, J.S.; Shi, L. Trait stacking in transgenic crops: Challenges and opportunities. GM Crops 2010, 1, 220–229. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Ow, D.W. Precise, flexible and affordable gene stacking for crop improvement. Bioengineered 2017, 8, 451–456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Das, G.; Patra, J.K.; Baek, K.H. Insight into mas: A molecular tool for development of stress resistant and quality of rice through gene stacking. Front. Plant Sci. 2017, 8, 985. [Google Scholar] [CrossRef] [Green Version]
- Collier, R.; Thomson, J.G.; Thilmony, R. A versatile and robust Agrobacterium-based gene stacking system generates high-quality transgenic Arabidopsis plants. Plant J. 2018, 95, 573–583. [Google Scholar] [CrossRef] [Green Version]
- Affoh, R.; Zheng, H.; Dangui, K.; Dissani, B.M. The impact of climate variability and change on food security in sub-saharan africa: Perspective from panel data analysis. Sustainability 2022, 14, 759. [Google Scholar] [CrossRef]
- Dasgupta, S.; Robinson, E.J.Z. Attributing changes in food insecurity to a changing climate. Sci. Rep. 2022, 12, 4709. [Google Scholar] [CrossRef]
- Agrimonti, C.; Lauro, M.; Visioli, G. Smart agriculture for food quality: Facing climate change in the 21st century. Crit. Rev. Food Sci. Nutr. 2021, 61, 971–981. [Google Scholar] [CrossRef]
- Kassaye, A.Y.; Shao, G.; Wang, X.; Shifaw, E.; Wu, S. Impact of climate change on the staple food crops yield in ethiopia: Implications for food security. Theor. Appl. Climatol. 2021, 145, 327–343. [Google Scholar] [CrossRef]
- Gao, C. Genome engineering for crop improvement and future agriculture. Cell 2021, 184, 1621–1635. [Google Scholar] [CrossRef]
- Jaganathan, D.; Ramasamy, K.; Sellamuthu, G.; Jayabalan, S.; Venkataraman, G. CRISPR for crop improvement: An update review. Front. Plant Sci. 2018, 9, 985. [Google Scholar] [CrossRef] [PubMed]
- Dong, O.X.; Ronald, P.C. Targeted DNA insertion in plants. Proc. Natl. Acad. Sci. USA 2021, 118, e2004834117. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, A.; Gill, R.A.; Hassan, M.U.; Mahmood, A.; Qari, S.; Zaman, Q.U.; Ilyas, M.; Aamer, M.; Batool, M.; Li, H.; et al. A critical review: Recent advancements in the use of CRISPR/Cas9 technology to enhance crops and alleviate global food crises. Curr. Issues Mol. Biol. 2021, 43, 1950–1976. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, Z.; Iqbal, M.S.; Ahmad, A.; Memon, A.G.; Ansari, M.I. New prospects on the horizon: Genome editing to engineer plants for desirable traits. Curr. Plant Biol. 2020, 24, 100171. [Google Scholar] [CrossRef]
- Karlson, C.K.S.; Mohd-Noor, S.N.; Nolte, N.; Tan, B.C. CRISPR/dcas9-based systems: Mechanisms and applications in plant sciences. Plants 2021, 10, 2055. [Google Scholar] [CrossRef]
- Ranjha, L.; Howard, S.M.; Cejka, P. Main steps in DNA double-strand break repair: An introduction to homologous recombination and related processes. Chromosoma 2018, 127, 187–214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van Gent, D.C.; van der Burg, M. Non-homologous end-joining, a sticky affair. Oncogene 2007, 26, 7731–7740. [Google Scholar] [CrossRef] [Green Version]
- Lusser, M.; Parisi, C.; Plan, D.; Rodríguez-Cerezo, E. New Plant Breeding Techniques: State-of-the-Art and Prospects for Commercial Development; Publications Office of the European Union: Luxembourg, 2011; pp. 1–217. [Google Scholar]
- Eckerstorfer, M.F.; Dolezel, M.; Heissenberger, A.; Miklau, M.; Reichenbecher, W.; Steinbrecher, R.A.; Wassmann, F. An EU perspective on biosafety considerations for plants developed by genome editing and other new genetic modification techniques (nGMs). Front. Bioeng. Biotechnol. 2019, 7, 31. [Google Scholar] [CrossRef] [Green Version]
- Kieu, N.P.; Lenman, M.; Wang, E.S.; Petersen, B.L.; Andreasson, E. Mutations introduced in susceptibility genes through CRISPR/Cas9 genome editing confer increased late blight resistance in potatoes. Sci. Rep. 2021, 11, 4487. [Google Scholar] [CrossRef]
- Oliva, R.; Ji, C.; Atienza-Grande, G.; Huguet-Tapia, J.C.; Perez-Quintero, A.; Li, T.; Eom, J.S.; Li, C.; Nguyen, H.; Liu, B.; et al. Broad-spectrum resistance to bacterial blight in rice using genome editing. Nat. Biotechnol. 2019, 37, 1344–1350. [Google Scholar] [CrossRef] [Green Version]
- Xie, K.; Yang, Y. RNA-guided genome editing in plants using a CRISPR-Cas system. Mol. Plant 2013, 6, 1975–1983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, M.; Li, X.; Zhou, Z.; Wu, P.; Fang, M.; Pan, X.; Lin, Q.; Luo, W.; Wu, G.; Li, H. Reassessment of the four yield-related genes Gn1a, DEP1, GS3, and IPA1 in rice using a CRISPR/Cas9 system. Front. Plant Sci. 2016, 7, 377. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortigosa, A.; Gimenez-Ibanez, S.; Leonhardt, N.; Solano, R. Design of a bacterial speck resistant tomato by CRISPR/Cas9-mediated editing of SlJAZ2. Plant Biotechnol. J. 2019, 17, 665–673. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodriguez-Leal, D.; Lemmon, Z.H.; Man, J.; Bartlett, M.E.; Lippman, Z.B. Engineering quantitative trait variation for crop improvement by genome editing. Cell 2017, 171, 470–480.e8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.R.; Jeon, I.; Yu, H.; Kim, S.G.; Kim, H.S.; Ahn, S.J.; Lee, J.; Lee, S.K.; Kim, H.U. Increasing monounsaturated fatty acid contents in hexaploid camelina sativa seed oil by FAD2 gene knockout using CRISPR-Cas9. Front. Plant Sci. 2021, 12, 702930. [Google Scholar] [CrossRef] [PubMed]
- Braatz, J.; Harloff, H.J.; Mascher, M.; Stein, N.; Himmelbach, A.; Jung, C. CRISPR-Cas9 targeted mutagenesis leads to simultaneous modification of different homoeologous gene copies in polyploid oilseed rape (Brassica napus). Plant Physiol. 2017, 174, 935–942. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waltz, E. Gene-edited CRISPR mushroom escapes us regulation. Nature 2016, 532, 293. [Google Scholar] [CrossRef] [Green Version]
- Shan, Q.; Zhang, Y.; Chen, K.; Zhang, K.; Gao, C. Creation of fragrant rice by targeted knockout of the OsBADH2 gene using TALEN technology. Plant Biotechnol. J. 2015, 13, 791–800. [Google Scholar] [CrossRef]
- Wang, F.; Wang, C.; Liu, P.; Lei, C.; Hao, W.; Gao, Y.; Liu, Y.G.; Zhao, K. Enhanced rice blast resistance by CRISPR/Cas9-targeted mutagenesis of the ERF transcription factor gene OsERF922. PLoS ONE 2016, 11, e0154027. [Google Scholar] [CrossRef]
- Jia, H.; Zhang, Y.; Orbovic, V.; Xu, J.; White, F.F.; Jones, J.B.; Wang, N. Genome editing of the disease susceptibility gene CsLOB1 in citrus confers resistance to citrus canker. Plant Biotechnol. J. 2017, 15, 817–823. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Cheng, X.; Shan, Q.; Zhang, Y.; Liu, J.; Gao, C.; Qiu, J.L. Simultaneous editing of three homoeoalleles in hexaploid bread wheat confers heritable resistance to powdery mildew. Nat. Biotechnol. 2014, 32, 947–951. [Google Scholar] [CrossRef] [PubMed]
- Andersson, M.; Turesson, H.; Nicolia, A.; Falt, A.S.; Samuelsson, M.; Hofvander, P. Efficient targeted multiallelic mutagenesis in tetraploid potato (Solanum tuberosum) by transient CRISPR-Cas9 expression in protoplasts. Plant Cell Rep. 2017, 36, 117–128. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, C.; Bortesi, L.; Baysal, C.; Twyman, R.M.; Fischer, R.; Capell, T.; Schillberg, S.; Christou, P. Characteristics of genome editing mutations in cereal crops. Trends Plant Sci. 2017, 22, 38–52. [Google Scholar] [CrossRef] [PubMed]
- Ran, Y.; Patron, N.; Kay, P.; Wong, D.; Buchanan, M.; Cao, Y.Y.; Sawbridge, T.; Davies, J.P.; Mason, J.; Webb, S.R.; et al. Zinc finger nuclease-mediated precision genome editing of an endogenous gene in hexaploid bread wheat (Triticum aestivum) using a DNA repair template. Plant Biotechnol. J. 2018, 16, 2088–2101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tian, S.; Jiang, L.; Cui, X.; Zhang, J.; Guo, S.; Li, M.; Zhang, H.; Ren, Y.; Gong, G.; Zong, M.; et al. Engineering herbicide-resistant watermelon variety through CRISPR/Cas9-mediated base-editing. Plant Cell Rep. 2018, 37, 1353–1356. [Google Scholar] [CrossRef]
- Wang, F.; Xu, Y.; Li, W.; Chen, Z.; Wang, J.; Fan, F.-j.; Tao, Y.; Jiang, Y.; Zhu, Q.-H.; Yang, J. Creating a novel herbicide-tolerance OsALS allele using CRISPR/Cas9-mediated gene editing. Crop J. 2021, 9, 305–312. [Google Scholar] [CrossRef]
- Li, T.; Liu, B.; Chen, C.Y.; Yang, B. TALEN-mediated homologous recombination produces site-directed DNA base change and herbicide-resistant rice. J. Genet. Genom. 2016, 43, 297–305. [Google Scholar] [CrossRef] [Green Version]
- Danilo, B.; Perrot, L.; Mara, K.; Botton, E.; Nogue, F.; Mazier, M. Efficient and transgene-free gene targeting using Agrobacterium-mediated delivery of the CRISPR/Cas9 system in tomato. Plant Cell Rep. 2019, 38, 459–462. [Google Scholar] [CrossRef]
- Yu, Q.H.; Wang, B.; Li, N.; Tang, Y.; Yang, S.; Yang, T.; Xu, J.; Guo, C.; Yan, P.; Wang, Q.; et al. CRISPR/Cas9-induced targeted mutagenesis and gene replacement to generate long-shelf life tomato lines. Sci. Rep. 2017, 7, 11874. [Google Scholar] [CrossRef]
- Takatsuka, A.; Kazama, T.; Arimura, S.I.; Toriyama, K. TALEN-mediated depletion of the mitochondrial gene orf312 proves that it is a Tadukan-type cytoplasmic male sterility-causative gene in rice. Plant J. 2022, in press. [Google Scholar] [CrossRef]
- Shi, J.; Gao, H.; Wang, H.; Lafitte, H.R.; Archibald, R.L.; Yang, M.; Hakimi, S.M.; Mo, H.; Habben, J.E. ARGOS8 variants generated by CRISPR-Cas9 improve maize grain yield under field drought stress conditions. Plant Biotechnol. J. 2017, 15, 207–216. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Y.; Zhang, X.; Wu, C.; He, Y.; Ma, Y.; Hou, H.; Guo, X.; Du, W.; Zhao, Y.; Xia, L. Engineering herbicide-resistant rice plants through CRISPR/Cas9-mediated homologous recombination of acetolactate synthase. Mol. Plant 2016, 9, 628–631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hummel, A.W.; Chauhan, R.D.; Cermak, T.; Mutka, A.M.; Vijayaraghavan, A.; Boyher, A.; Starker, C.G.; Bart, R.; Voytas, D.F.; Taylor, N.J. Allele exchange at the EPSPS locus confers glyphosate tolerance in cassava. Plant Biotechnol. J. 2018, 16, 1275–1282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, O.X.; Yu, S.; Jain, R.; Zhang, N.; Duong, P.Q.; Butler, C.; Li, Y.; Lipzen, A.; Martin, J.A.; Barry, K.W.; et al. Marker-free carotenoid-enriched rice generated through targeted gene insertion using CRISPR-Cas9. Nat. Commun. 2020, 11, 1178. [Google Scholar] [CrossRef] [Green Version]
- Ainley, W.M.; Sastry-Dent, L.; Welter, M.E.; Murray, M.G.; Zeitler, B.; Amora, R.; Corbin, D.R.; Miles, R.R.; Arnold, N.L.; Strange, T.L.; et al. Trait stacking via targeted genome editing. Plant Biotechnol. J. 2013, 11, 1126–1134. [Google Scholar] [CrossRef]
- Bonawitz, N.D.; Ainley, W.M.; Itaya, A.; Chennareddy, S.R.; Cicak, T.; Effinger, K.; Jiang, K.; Mall, T.K.; Marri, P.R.; Samuel, J.P.; et al. Zinc finger nuclease-mediated targeting of multiple transgenes to an endogenous soybean genomic locus via non-homologous end joining. Plant Biotechnol. J. 2019, 17, 750–761. [Google Scholar] [CrossRef]
- Ali, Z.; Shami, A.; Sedeek, K.; Kamel, R.; Alhabsi, A.; Tehseen, M.; Hassan, N.; Butt, H.; Kababji, A.; Hamdan, S.M.; et al. Fusion of the Cas9 endonuclease and the vird2 relaxase facilitates homology-directed repair for precise genome engineering in rice. Commun. Biol. 2020, 3, 44. [Google Scholar] [CrossRef]
- Mishra, R.; Joshi, R.K.; Zhao, K. Base editing in crops: Current advances, limitations and future implications. Plant Biotechnol. J. 2020, 18, 20–31. [Google Scholar] [CrossRef]
- Zhu, H.; Li, C.; Gao, C. Applications of CRISPR-Cas in agriculture and plant biotechnology. Nat. Rev. Mol. Cell. Biol. 2020, 21, 661–677. [Google Scholar] [CrossRef]
- Kang, B.C.; Yun, J.Y.; Kim, S.T.; Shin, Y.; Ryu, J.; Choi, M.; Woo, J.W.; Kim, J.S. Precision genome engineering through adenine base editing in plants. Nat. Plants 2018, 4, 427–431. [Google Scholar] [CrossRef]
- Komor, A.C.; Kim, Y.B.; Packer, M.S.; Zuris, J.A.; Liu, D.R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 2016, 533, 420–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kuang, Y.; Li, S.; Ren, B.; Yan, F.; Spetz, C.; Li, X.; Zhou, X.; Zhou, H. Base-editing-mediated artificial evolution of OsALS1 in planta to develop novel herbicide-tolerant rice germplasms. Mol. Plant 2020, 13, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhu, J.; Wu, H.; Liu, C.; Huang, C.; Lan, J.; Zhao, Y.; Xie, C. Precise base editing of non-allelic acetolactate synthase genes confers sulfonylurea herbicide resistance in maize. Crop J. 2020, 8, 449–456. [Google Scholar] [CrossRef]
- Cheng, H.; Hao, M.; Ding, B.; Mei, D.; Wang, W.; Wang, H.; Zhou, R.; Liu, J.; Li, C.; Hu, Q. Base editing with high efficiency in allotetraploid oilseed rape by A3A-PBE system. Plant Biotechnol. J. 2021, 19, 87–97. [Google Scholar] [CrossRef]
- Veillet, F.; Chauvin, L.; Kermarrec, M.P.; Sevestre, F.; Merrer, M.; Terret, Z.; Szydlowski, N.; Devaux, P.; Gallois, J.L.; Chauvin, J.E. The Solanum tuberosum GBSSI gene: A target for assessing gene and base editing in tetraploid potato. Plant Cell Rep. 2019, 38, 1065–1080. [Google Scholar] [CrossRef]
- Bharat, S.S.; Li, S.; Li, J.; Yan, L.; Xia, L. Base editing in plants: Current status and challenges. Crop J. 2020, 8, 384–395. [Google Scholar] [CrossRef]
- Park, S.-C.; Park, S.; Jeong, Y.J.; Lee, S.B.; Pyun, J.W.; Kim, S.; Kim, T.H.; Kim, S.W.; Jeong, J.C.; Kim, C.Y. DNA-free mutagenesis of GIGANTEA in Brassica oleracea var. capitata using CRISPR/Cas9 ribonucleoprotein complexes. Plant Biotechnol. Rep. 2019, 13, 483–489. [Google Scholar] [CrossRef]
- Liang, Z.; Chen, K.; Li, T.; Zhang, Y.; Wang, Y.; Zhao, Q.; Liu, J.; Zhang, H.; Liu, C.; Ran, Y.; et al. Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat. Commun. 2017, 8, 14261. [Google Scholar] [CrossRef]
- Murovec, J.; Gucek, K.; Bohanec, B.; Avbelj, M.; Jerala, R. DNA-free genome editing of Brassica oleracea and B. rapa protoplasts using CRISPR-Cas9 ribonucleoprotein complexes. Front. Plant Sci. 2018, 9, 1594. [Google Scholar] [CrossRef]
- Xu, J.; Hua, K.; Lang, Z. Genome editing for horticultural crop improvement. Hortic. Res. 2019, 6, 113. [Google Scholar] [CrossRef] [Green Version]
- Cong, L.; Ran, F.A.; Cox, D.; Lin, S.; Barretto, R.; Habib, N.; Hsu, P.D.; Wu, X.; Jiang, W.; Marraffini, L.A.; et al. Multiplex genome engineering using CRISPR/Cas systems. Science 2013, 339, 819–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mali, P.; Yang, L.; Esvelt, K.M.; Aach, J.; Guell, M.; DiCarlo, J.E.; Norville, J.E.; Church, G.M. RNA-guided human genome engineering via Cas9. Science 2013, 339, 823–826. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guo, M.; Chen, H.; Dong, S.; Zhang, Z.; Luo, H. CRISPR-Cas gene editing technology and its application prospect in medicinal plants. Chin. Med. 2022, 17, 33. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Zhou, H.; Bi, H.; Fromm, M.; Yang, B.; Weeks, D.P. Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res. 2013, 41, e188. [Google Scholar] [CrossRef]
- Nekrasov, V.; Staskawicz, B.; Weigel, D.; Jones, J.D.; Kamoun, S. Targeted mutagenesis in the model plant Nicotiana benthamiana using Cas9 RNA-guided endonuclease. Nat. Biotechnol. 2013, 31, 691–693. [Google Scholar] [CrossRef]
- Svitashev, S.; Young, J.K.; Schwartz, C.; Gao, H.; Falco, S.C.; Cigan, A.M. Targeted mutagenesis, precise gene editing, and site-specific gene insertion in maize using Cas9 and guide RNA. Plant Physiol. 2015, 169, 931–945. [Google Scholar] [CrossRef] [Green Version]
- Čermák, T.; Baltes, N.J.; Čegan, R.; Zhang, Y.; Voytas, D.F. High-frequency, precise modification of the tomato genome. Genome Biol. 2015, 16, 232. [Google Scholar] [CrossRef] [Green Version]
- Jacobs, T.B.; LaFayette, P.R.; Schmitz, R.J.; Parrott, W.A. Targeted genome modifications in soybean with CRISPR/Cas9. BMC Biotechnol. 2015, 15, 16. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Zhang, S.; Wang, W.; Xiong, X.; Meng, F.; Cui, X. Efficient targeted mutagenesis in potato by the CRISPR/Cas9 system. Plant Cell Rep. 2015, 34, 1473–1476. [Google Scholar] [CrossRef] [Green Version]
- Varkonyi-Gasic, E.; Wang, T.; Voogd, C.; Jeon, S.; Drummond, R.S.M.; Gleave, A.P.; Allan, A.C. Mutagenesis of kiwifruit centroradialis-like genes transforms a climbing woody perennial with long juvenility and axillary flowering into a compact plant with rapid terminal flowering. Plant Biotechnol. J. 2019, 17, 869–880. [Google Scholar] [CrossRef] [Green Version]
- Tripathi, J.N.; Ntui, V.O.; Ron, M.; Muiruri, S.K.; Britt, A.; Tripathi, L. CRISPR/Cas9 editing of endogenous banana streak virus in the B genome of Musa spp. Overcomes a major challenge in banana breeding. Commun. Biol. 2019, 2, 46. [Google Scholar] [CrossRef] [Green Version]
- Fan, D.; Liu, T.; Li, C.; Jiao, B.; Li, S.; Hou, Y.; Luo, K. Efficient CRISPR/Cas9-mediated targeted mutagenesis in populus in the first generation. Sci. Rep. 2015, 5, 12217. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Unver, T.; Zhang, B. A high-efficiency CRISPR/Cas9 system for targeted mutagenesis in cotton (Gossypium hirsutum L.). Sci. Rep. 2017, 7, 43902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, Y.; Xin, S.; Dai, X.; Yang, X.; Huang, H.; Hua, Y. Efficient genome editing of rubber tree (Hevea brasiliensis) protoplasts using CRISPR/Cas9 ribonucleoproteins. Ind. Crops Prod. 2020, 146, 112146. [Google Scholar] [CrossRef]
- Yeap, W.C.; Norkhairunnisa Che Mohd, K.; Norfadzilah, J.; Muad, M.R.; Appleton, D.R.; Harikrishna, K. An efficient clustered regularly interspaced short palindromic repeat (CRISPR)/CRISPR-associated protein 9 mutagenesis system for oil palm (Elaeis guineensis). Front. Plant Sci. 2021, 12, 773656. [Google Scholar] [CrossRef] [PubMed]
- Huang, B.; Zhuo, R.; Fan, H.; Wang, Y.; Xu, J.; Jin, K.; Qiao, G. An efficient genetic transformation and CRISPR/Cas9-based genome editing system for moso bamboo (Phyllostachys edulis). Front. Plant Sci. 2022, 13, 822022. [Google Scholar] [CrossRef]
- Kishi-Kaboshi, M.; Aida, R.; Sasaki, K. Generation of gene-edited Chrysanthemum morifolium using multicopy transgenes as targets and markers. Plant Cell Physiol. 2017, 58, 216–226. [Google Scholar] [CrossRef] [Green Version]
- Shibuya, K.; Watanabe, K.; Ono, M. CRISPR/Cas9-mediated mutagenesis of the EPHEMERAL1 locus that regulates petal senescence in Japanese morning glory. Plant Physiol. Biochem. 2018, 131, 53–57. [Google Scholar] [CrossRef]
- Yan, R.; Wang, Z.; Ren, Y.; Li, H.; Liu, N.; Sun, H. Establishment of efficient genetic transformation systems and application of CRISPR/Cas9 genome editing technology in Lilium pumilum DC. Fisch. and Lilium longiflorum white heaven. Int. J. Mol. Sci. 2019, 20, 2920. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.; Xu, X.; Huang, R.; Yang, S.; Li, M.; Guo, Y. CRISPR/Cas9-mediated targeted mutation reveals a role for AN4 rather than DPL in regulating venation formation in the corolla tube of Petunia hybrida. Hortic. Res. 2021, 8, 116. [Google Scholar] [CrossRef]
- Tong, C.G.; Wu, F.H.; Yuan, Y.H.; Chen, Y.R.; Lin, C.S. High-efficiency CRISPR/Cas-based editing of Phalaenopsis orchid MADS genes. Plant Biotechnol. J. 2020, 18, 889–891. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Li, H.; Liu, C.; He, Y.; Wang, H.; Zhao, T.; Xu, X.; Li, J.; Yang, H.; Jiang, J. CRISPR-Cas9-mediated mutagenesis of the SlSRM1-like gene leads to abnormal leaf development in tomatoes. BMC Plant Biol. 2022, 22, 13. [Google Scholar] [CrossRef] [PubMed]
- Krenek, P.; Chubar, E.; Vadovic, P.; Ohnoutkova, L.; Vlcko, T.; Bergougnoux, V.; Capal, P.; Ovecka, M.; Samaj, J. CRISPR/Cas9-induced loss-of-function mutation in the barley Mitogen-activated protein kinase 6 gene causes abnormal embryo development leading to severely reduced grain germination and seedling shootless phenotype. Front. Plant Sci. 2021, 12, 670302. [Google Scholar] [CrossRef]
- Galli, M.; Martiny, E.; Imani, J.; Kumar, N.; Koch, A.; Steinbrenner, J.; Kogel, K.H. CRISPR/SpCas9-mediated double knockout of barley Microrchidia MORC1 and MORC6a reveals their strong involvement in plant immunity, transcriptional gene silencing and plant growth. Plant Biotechnol. J. 2022, 20, 89–102. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lin, Z.; Yue, Y.; Zhao, H.; Fei, X.; E, L.; Liu, C.; Chen, S.; Lai, J.; Song, W. Loss-of-function alleles of ZmPLD3 cause haploid induction in maize. Nat. Plants 2021, 7, 1579–1588. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhang, R.; Jia, X.; Tang, X.; Guo, Y.; Yang, H.; Zheng, X.; Qian, Q.; Qi, Y.; Zhang, Y. CRISPR-Cas9 mediated OsMIR168a knockout reveals its pleiotropy in rice. Plant Biotechnol. J. 2022, 20, 310–322. [Google Scholar] [CrossRef]
- Le Rhun, A.; Escalera-Maurer, A.; Bratovic, M.; Charpentier, E. CRISPR-Cas in Streptococcus pyogenes. RNA Biol. 2019, 16, 380–389. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Manghwar, H.; Sun, L.; Wang, P.; Wang, G.; Sheng, H.; Zhang, J.; Liu, H.; Qin, L.; Rui, H.; et al. Whole genome sequencing reveals rare off-target mutations and considerable inherent genetic or/and somaclonal variations in CRISPR/Cas9-edited cotton plants. Plant Biotechnol. J. 2019, 17, 858–868. [Google Scholar] [CrossRef]
- Young, J.; Zastrow-Hayes, G.; Deschamps, S.; Svitashev, S.; Zaremba, M.; Acharya, A.; Paulraj, S.; Peterson-Burch, B.; Schwartz, C.; Djukanovic, V.; et al. CRISPR-Cas9 editing in maize: Systematic evaluation of off-target activity and its relevance in crop improvement. Sci. Rep. 2019, 9, 6729. [Google Scholar] [CrossRef] [Green Version]
- Jin, S.; Zong, Y.; Gao, Q.; Zhu, Z.; Wang, Y.; Qin, P.; Liang, C.; Wang, D.; Qiu, J.L.; Zhang, F.; et al. Cytosine, but not adenine, base editors induce genome-wide off-target mutations in rice. Science 2019, 364, 292–295. [Google Scholar] [CrossRef]
- Jinek, M.; Chylinski, K.; Fonfara, I.; Hauer, M.; Doudna, J.A.; Charpentier, E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 2012, 337, 816–821. [Google Scholar] [CrossRef] [PubMed]
- Lino, C.A.; Harper, J.C.; Carney, J.P.; Timlin, J.A. Delivering CRISPR: A review of the challenges and approaches. Drug Deliv. 2018, 25, 1234–1257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ran, F.A.; Cong, L.; Yan, W.X.; Scott, D.A.; Gootenberg, J.S.; Kriz, A.J.; Zetsche, B.; Shalem, O.; Wu, X.; Makarova, K.S.; et al. In vivo genome editing using Staphylococcus aureus Cas9. Nature 2015, 520, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Schiml, S.; Fauser, F.; Puchta, H. The CRISPR/Cas system can be used as nuclease for in planta gene targeting and as paired nickases for directed mutagenesis in Arabidopsis resulting in heritable progeny. Plant J. 2014, 80, 1139–1150. [Google Scholar] [CrossRef]
- Walton, R.T.; Christie, K.A.; Whittaker, M.N.; Kleinstiver, B.P. Unconstrained genome targeting with near-pamless engineered CRISPR-Cas9 variants. Science 2020, 368, 290–296. [Google Scholar] [CrossRef]
- Ren, B.; Liu, L.; Li, S.; Kuang, Y.; Wang, J.; Zhang, D.; Zhou, X.; Lin, H.; Zhou, H. Cas9-NG greatly expands the targeting scope of the genome-editing toolkit by recognizing NG and other atypical PAMs in rice. Mol. Plant 2019, 12, 1015–1026. [Google Scholar] [CrossRef]
- Ge, Z.; Zheng, L.; Zhao, Y.; Jiang, J.; Zhang, E.J.; Liu, T.; Gu, H.; Qu, L.J. Engineered xCas9 and SpCas9-NG variants broaden PAM recognition sites to generate mutations in Arabidopsis plants. Plant Biotechnol. J. 2019, 17, 1865–1867. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Luo, J.; Xu, M.; Li, S.; Zhang, J.; Li, H.; Yan, L.; Zhao, Y.; Xia, L. Plant genome editing using xCas9 with expanded PAM compatibility. J. Genet. Genom. 2019, 46, 277–280. [Google Scholar] [CrossRef]
- Sretenovic, S.; Yin, D.; Levav, A.; Selengut, J.D.; Mount, S.M.; Qi, Y. Expanding plant genome-editing scope by an engineered iSpyMacCas9 system that targets A-rich PAM sequences. Plant Commun. 2021, 2, 100101. [Google Scholar] [CrossRef]
- Ha, D.I.; Lee, J.M.; Lee, N.E.; Kim, D.; Ko, J.H.; Kim, Y.S. Highly efficient and safe genome editing by CRISPR-Cas12a using CRISPR RNA with a ribosyl-2′-o-methylated uridinylate-rich 3′-overhang in mouse zygotes. Exp. Mol. Med. 2020, 52, 1823–1830. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, M.; Zheng, T.; Hou, Y.; Zhang, P.; Tang, T.; Wei, J.; Du, Q. Specificity profiling of CRISPR system reveals greatly enhanced off-target gene editing. Sci. Rep. 2020, 10, 2269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swarts, D.C.; van der Oost, J.; Jinek, M. Structural basis for guide RNA processing and seed-dependent DNA targeting by CRISPR-Cas12a. Mol. Cell 2017, 66, 221–233.e4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Cheng, Y.; Fang, H.; Roberts, N.; Zhang, L.; Vakulskas, C.A.; Niedz, R.P.; Culver, J.N.; Qi, Y. Highly efficient genome editing in plant protoplasts by ribonucleoprotein delivery of CRISPR-Cas12a nucleases. Front. Genome Ed. 2022, 4, 780238. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zuris, J.A.; Viswanathan, R.; Edelstein, J.N.; Turk, R.; Thommandru, B.; Rube, H.T.; Glenn, S.E.; Collingwood, M.A.; Bode, N.M.; et al. AsCas12a ultra nuclease facilitates the rapid generation of therapeutic cell medicines. Nat. Commun. 2021, 12, 3908. [Google Scholar] [CrossRef] [PubMed]
- Toth, E.; Varga, E.; Kulcsar, P.I.; Kocsis-Jutka, V.; Krausz, S.L.; Nyeste, A.; Welker, Z.; Huszar, K.; Ligeti, Z.; Talas, A.; et al. Improved LbCas12a variants with altered PAM specificities further broaden the genome targeting range of Cas12a nucleases. Nucleic Acids Res. 2020, 48, 3722–3733. [Google Scholar] [CrossRef]
- Malzahn, A.A.; Tang, X.; Lee, K.; Ren, Q.; Sretenovic, S.; Zhang, Y.; Chen, H.; Kang, M.; Bao, Y.; Zheng, X.; et al. Application of CRISPR-Cas12a temperature sensitivity for improved genome editing in rice, maize, and Arabidopsis. BMC Biol. 2019, 17, 9. [Google Scholar] [CrossRef] [Green Version]
- Bernabe-Orts, J.M.; Casas-Rodrigo, I.; Minguet, E.G.; Landolfi, V.; Garcia-Carpintero, V.; Gianoglio, S.; Vazquez-Vilar, M.; Granell, A.; Orzaez, D. Assessment of Cas12a-mediated gene editing efficiency in plants. Plant Biotechnol. J. 2019, 17, 1971–1984. [Google Scholar] [CrossRef] [Green Version]
- Schindele, P.; Puchta, H. Engineering CRISPR/LbCas12a for highly efficient, temperature-tolerant plant gene editing. Plant Biotechnol. J. 2020, 18, 1118–1120. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Mao, Y.; Lu, Y.; Tao, X.; Zhu, J.K. Multiplex gene editing in rice using the CRISPR-Cpf1 system. Mol. Plant 2017, 10, 1011–1013. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Li, J.; He, Y.; Xu, M.; Zhang, J.; Du, W.; Zhao, Y.; Xia, L. Precise gene replacement in rice by RNA transcript-templated homologous recombination. Nat. Biotechnol. 2019, 37, 445–450. [Google Scholar] [CrossRef]
- Zhong, Z.; Zhang, Y.; You, Q.; Tang, X.; Ren, Q.; Liu, S.; Yang, L.; Wang, Y.; Liu, X.; Liu, B.; et al. Plant genome editing using FnCpf1 and LbCpf1 nucleases at redefined and altered PAM sites. Mol. Plant 2018, 11, 999–1002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.; Zhang, Y.; Kleinstiver, B.P.; Guo, J.A.; Aryee, M.J.; Miller, J.; Malzahn, A.; Zarecor, S.; Lawrence-Dill, C.J.; Joung, J.K.; et al. Activities and specificities of CRISPR/Cas9 and Cas12a nucleases for targeted mutagenesis in maize. Plant Biotechnol. J. 2019, 17, 362–372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.; Kim, S.T.; Ryu, J.; Kang, B.C.; Kim, J.S.; Kim, S.G. CRISPR/Cpf1-mediated DNA-free plant genome editing. Nat. Commun. 2017, 8, 14406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, B.; Liang, S.; Alariqi, M.; Wang, F.; Wang, G.; Wang, Q.; Xu, Z.; Yu, L.; Naeem Zafar, M.; Sun, L.; et al. The application of temperature sensitivity CRISPR/LbCpf1 (LbCas12a) mediated genome editing in allotetraploid cotton (G. hirsutum) and creation of nontransgenic, gossypol-free cotton. Plant Biotechnol. J. 2021, 19, 221–223. [Google Scholar] [CrossRef]
- Li, B.; Rui, H.; Li, Y.; Wang, Q.; Alariqi, M.; Qin, L.; Sun, L.; Ding, X.; Wang, F.; Zou, J.; et al. Robust CRISPR/Cpf1 (Cas12a)-mediated genome editing in allotetraploid cotton (Gossypium hirsutum). Plant Biotechnol. J. 2019, 17, 1862–1864. [Google Scholar] [CrossRef] [Green Version]
- Jia, H.; Orbovic, V.; Wang, N. CRISPR-LbCas12a-mediated modification of citrus. Plant Biotechnol. J. 2019, 17, 1928–1937. [Google Scholar] [CrossRef] [Green Version]
- Park, H.M.; Liu, H.; Wu, J.; Chong, A.; Mackley, V.; Fellmann, C.; Rao, A.; Jiang, F.; Chu, H.; Murthy, N.; et al. Extension of the crRNA enhances Cpf1 gene editing in vitro and in vivo. Nat. Commun. 2018, 9, 3313. [Google Scholar] [CrossRef]
- Begemann, M.B.; Gray, B.N.; January, E.; Gordon, G.C.; He, Y.; Liu, H.; Wu, X.; Brutnell, T.P.; Mockler, T.C.; Oufattole, M. Precise insertion and guided editing of higher plant genomes using Cpf1 CRISPR nucleases. Sci. Rep. 2017, 7, 11606. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Zhang, Y.; Xia, L.; Qi, Y. CRISPR-Cas12a enables efficient biallelic gene targeting in rice. Plant Biotechnol. J. 2020, 18, 1351–1353. [Google Scholar] [CrossRef]
- Li, S.; Li, J.; Zhang, J.; Du, W.; Fu, J.; Sutar, S.; Zhao, Y.; Xia, L. Synthesis-dependent repair of Cpf1-induced double strand DNA breaks enables targeted gene replacement in rice. J. Exp. Bot. 2018, 69, 4715–4721. [Google Scholar] [CrossRef] [Green Version]
- Wolter, F.; Puchta, H. In planta gene targeting can be enhanced by the use of CRISPR/Cas12a. Plant J. 2019, 100, 1083–1094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kleinstiver, B.P.; Sousa, A.A.; Walton, R.T.; Tak, Y.E.; Hsu, J.Y.; Clement, K.; Welch, M.M.; Horng, J.E.; Malagon-Lopez, J.; Scarfo, I.; et al. Engineered CRISPR-Cas12a variants with increased activities and improved targeting ranges for gene, epigenetic and base editing. Nat. Biotechnol. 2019, 37, 276–282. [Google Scholar] [CrossRef]
- Li, X.; Wang, Y.; Liu, Y.; Yang, B.; Wang, X.; Wei, J.; Lu, Z.; Zhang, Y.; Wu, J.; Huang, X.; et al. Base editing with a Cpf1-cytidine deaminase fusion. Nat. Biotechnol. 2018, 36, 324–327. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Lowder, L.G.; Zhang, T.; Malzahn, A.A.; Zheng, X.; Voytas, D.F.; Zhong, Z.; Chen, Y.; Ren, Q.; Li, Q.; et al. A CRISPR-Cpf1 system for efficient genome editing and transcriptional repression in plants. Nat. Plants 2017, 3, 17103. [Google Scholar] [CrossRef] [PubMed]
- Tak, Y.E.; Kleinstiver, B.P.; Nunez, J.K.; Hsu, J.Y.; Horng, J.E.; Gong, J.; Weissman, J.S.; Joung, J.K. Inducible and multiplex gene regulation using CRISPR-Cpf1-based transcription factors. Nat. Methods 2017, 14, 1163–1166. [Google Scholar] [CrossRef] [Green Version]
- Breinig, M.; Schweitzer, A.Y.; Herianto, A.M.; Revia, S.; Schaefer, L.; Wendler, L.; Cobos Galvez, A.; Tschaharganeh, D.F. Multiplexed orthogonal genome editing and transcriptional activation by Cas12a. Nat. Methods 2019, 16, 51–54. [Google Scholar] [CrossRef]
- Campa, C.C.; Weisbach, N.R.; Santinha, A.J.; Incarnato, D.; Platt, R.J. Multiplexed genome engineering by Cas12a and CRISPR arrays encoded on single transcripts. Nat. Methods 2019, 16, 887–893. [Google Scholar] [CrossRef]
- Abudayyeh, O.O.; Gootenberg, J.S.; Essletzbichler, P.; Han, S.; Joung, J.; Belanto, J.J.; Verdine, V.; Cox, D.B.T.; Kellner, M.J.; Regev, A.; et al. RNA targeting with CRISPR-cas13. Nature 2017, 550, 280–284. [Google Scholar] [CrossRef] [Green Version]
- Mahas, A.; Aman, R.; Mahfouz, M. CRISPR-cas13d mediates robust RNA virus interference in plants. Genome Biol. 2019, 20, 263. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Chen, Z.; Chen, F.; Xie, G.; Ling, Y.; Peng, Y.; Lin, Y.; Luo, N.; Chiang, C.-M.; Wang, H. Targeted mrna demethylation using an engineered dcas13b-alkbh5 fusion protein. Nucleic Acids Res. 2020, 48, 5684–5694. [Google Scholar] [CrossRef]
- Abudayyeh, O.O.; Gootenberg, J.S.; Franklin, B.; Koob, J.; Kellner, M.J.; Ladha, A.; Joung, J.; Kirchgatterer, P.; Cox, D.B.T.; Zhang, F. A cytosine deaminase for programmable single-base RNA editing. Science 2019, 365, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Zhu, H. Modified gene editing systems: Diverse bioengineering tools and crop improvement. Front. Plant Sci. 2022, 13, 847169. [Google Scholar] [CrossRef] [PubMed]
- Laforest, L.C.; Nadakuduti, S.S. Advances in delivery mechanisms of CRISPR gene-editing reagents in plants. Front. Genome Ed. 2022, 4, 830178. [Google Scholar] [CrossRef] [PubMed]
- Maher, M.F.; Nasti, R.A.; Vollbrecht, M.; Starker, C.G.; Clark, M.D.; Voytas, D.F. Plant gene editing through de novo induction of meristems. Nat. Biotechnol. 2020, 38, 84–89. [Google Scholar] [CrossRef]
- Xu, C.L.; Ruan, M.Z.C.; Mahajan, V.B.; Tsang, S.H. Viral delivery systems for CRISPR. Viruses 2019, 11, 28. [Google Scholar] [CrossRef] [Green Version]
- Lau, C.H.; Suh, Y. In vivo genome editing in animals using aav-CRISPR system: Applications to translational research of human disease. F1000Research 2017, 6, 2153. [Google Scholar] [CrossRef] [Green Version]
- Kantor, B.; Bailey, R.M.; Wimberly, K.; Kalburgi, S.N.; Gray, S.J. Methods for gene transfer to the central nervous system. Adv. Genet. 2014, 87, 125–197. [Google Scholar] [CrossRef] [Green Version]
- Pausch, P.; Al-Shayeb, B.; Bisom-Rapp, E.; Tsuchida, C.A.; Li, Z.; Cress, B.F.; Knott, G.J.; Jacobsen, S.E.; Banfield, J.F.; Doudna, J.A. CRISPR-casphi from huge phages is a hypercompact genome editor. Science 2020, 369, 333–337. [Google Scholar] [CrossRef]
- Kujur, S.; Senthil-Kumar, M.; Kumar, R. Plant viral vectors: Expanding the possibilities of precise gene editing in plant genomes. Plant Cell Rep. 2021, 40, 931–934. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, Q.; Chen, Q.J. Agrobacterium-mediated delivery of CRISPR/Cas reagents for genome editing in plants enters an era of ternary vector systems. Sci. China Life Sci. 2020, 63, 1491–1498. [Google Scholar] [CrossRef]
- Zhang, Y.; Iaffaldano, B.; Qi, Y. CRISPR ribonucleoprotein-mediated genetic engineering in plants. Plant Commun. 2021, 2, 100168. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Brant, E.; Budak, H.; Zhang, B. CRISPR/Cas: A nobel prize award-winning precise genome editing technology for gene therapy and crop improvement. J. Zhejiang Univ. Sci. B 2021, 22, 253–284. [Google Scholar] [CrossRef] [PubMed]
- Zafar, K.; Sedeek, K.E.M.; Rao, G.S.; Khan, M.Z.; Amin, I.; Kamel, R.; Mukhtar, Z.; Zafar, M.; Mansoor, S.; Mahfouz, M.M. Genome editing technologies for rice improvement: Progress, prospects, and safety concerns. Front. Genome Ed. 2020, 2, 5. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Tang, L.; Shahzad, R.; Mawia, A.M.; Rao, G.S.; Jamil, S.; Wei, C.; Sheng, Z.; Shao, G.; Wei, X.; et al. CRISPR-based crop improvements: A way forward to achieve zero hunger. J. Agric. Food Chem. 2021, 69, 8307–8323. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, C.; Lillemo, M.; Hvoslef-Eide, T.A.K. Global regulation of genetically modified crops amid the gene edited crop boom—A review. Front. Plant Sci. 2021, 12, 630396. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, H.; Zhu, H. CRISPR technology is revolutionizing the improvement of tomato and other fruit crops. Hortic. Res. 2019, 6, 77. [Google Scholar] [CrossRef] [Green Version]
- van de Wiel, C.C.M.; Schaart, J.G.; Lotz, L.A.P.; Smulders, M.J.M. New traits in crops produced by genome editing techniques based on deletions. Plant Biotechnol. Rep. 2017, 11, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Krasileva, K.V.; Vasquez-Gross, H.A.; Howell, T.; Bailey, P.; Paraiso, F.; Clissold, L.; Simmonds, J.; Ramirez-Gonzalez, R.H.; Wang, X.; Borrill, P.; et al. Uncovering hidden variation in polyploid wheat. Proc. Natl. Acad. Sci. USA 2017, 114, E913–E921. [Google Scholar] [CrossRef] [Green Version]
- Ahmar, S.; Gill, R.A.; Jung, K.H.; Faheem, A.; Qasim, M.U.; Mubeen, M.; Zhou, W. Conventional and molecular techniques from simple breeding to speed breeding in crop plants: Recent advances and future outlook. Int. J. Mol. Sci. 2020, 21, 2590. [Google Scholar] [CrossRef] [Green Version]
- Chen, K.; Wang, Y.; Zhang, R.; Zhang, H.; Gao, C. CRISPR/Cas genome editing and precision plant breeding in agriculture. Annu. Rev. Plant Biol. 2019, 70, 667–697. [Google Scholar] [CrossRef]
- The European Court of Justice (ECJ). Judgement of the Court in Case C-528/16: Court of Justice of the European Union. Available online: http://curia.europa.eu/juris/document/document.jsf?text=&docid=204387&pageIndex=0&doclang=EN&mode=lst&dir=&occ=first&part=1&cid=138460 (accessed on 1 January 2020).
- Waltz, E. With a free pass, CRISPR-edited plants reach market in record time. Nat. Biotechnol. 2018, 36, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Waltz, E. A face-lift for biotech rules begins. Nat. Biotechnol. 2015, 33, 1221–1222. [Google Scholar] [CrossRef] [PubMed]
- The Food and Agriculture Organization (FAO). Principles for the Risk Analysis of Foods Derived from Modern Biotechnology. Available online: https://www.fao.org/fao-who-codexalimentarius/sh-proxy/fr/?lnk=1&url=https%253A%252F%252Fworkspace.fao.org%252Fsites%252Fcodex%252FStandards%252FCXG%2B44-2003%252FCXG_044e.pdf (accessed on 1 January 2021).
- The International Service for the Acquisition of Agri-biotech Applications (ISAAA). Pocket K No. 56: Substantial Equivalence of GM and Non-GM Crops. Available online: https://www.isaaa.org/resources/publications/pocketk/56/default.asp#:~:text=In%20other%20words%2C%20substantial%20equivalence,or%20removed%20through%20genetic%20engineering (accessed on 1 January 2021).
- United Nations (UN). Parties to the Cartagena Protocol and Its Supplementary Protocol on Liability and Redress. Available online: https://bch.cbd.int/protocol/parties/ (accessed on 1 January 2021).
- European Commission (EC). GMO Legislation. Available online: https://ec.europa.eu/food/plants/genetically-modified-organisms/gmo-legislation_en (accessed on 1 January 2021).
- Bayer. Annual Monitoring Report on the Cultivation of Mon 810 in 2020; Bayer: Leverkusen, Germany, 2021; pp. 1–50. [Google Scholar]
- Belder, T.D. Biotechnology and Other New Production Technologies Annual; USDA Foreign Agricultural Service: The Hague, The Netherlands, 2021; pp. 1–68. [Google Scholar]
- The International Service for the Acquisition of Agri-biotech Applications (ISAAA). Pocket K No. 42: Stacked Traits in Biotech Crops. Available online: https://www.isaaa.org/resources/publications/pocketk/42/default.asp (accessed on 1 January 2021).
- Vesprini, F.; Whelan, A.I.; Goberna, M.F.; Murrone, M.L.; Barros, G.E.; Frankow, A.; Godoy, P.; Lewi, D.M. Update of argentina’s regulatory policies on the environmental risk assessment. Front. Bioeng. Biotechnol. 2021, 9, 834589. [Google Scholar] [CrossRef]
- Taverniers, I.; Papazova, N.; Bertheau, Y.; De Loose, M.; Holst-Jensen, A. Gene stacking in transgenic plants: Towards compliance between definitions, terminology, and detection within the EU regulatory framework. Environ. Biosaf. Res. 2008, 7, 197–218. [Google Scholar] [CrossRef] [PubMed]
- Sato, S. Agricultural Biotechnology Annual; USDA Foreign Agricultural Service: Tokyo, Japan, 2021; pp. 1–21. [Google Scholar]
- Ahmad, A.; Munawar, N.; Khan, Z.; Qusmani, A.T.; Khan, S.H.; Jamil, A.; Ashraf, S.; Ghouri, M.Z.; Aslam, S.; Mubarik, M.S.; et al. An outlook on global regulatory landscape for genome-edited crops. Int. J. Mol. Sci. 2021, 22, 11753. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Applicability of the efsa opinion on site-directed nucleases type 3 for the safety assessment of plants developed using site-directed nucleases type 1 and 2 and oligonucleotide-directed mutagenesis. EFSA J. 2020, 18, e06299. [Google Scholar] [CrossRef]
- Strauss, S.H.; Sax, J.K. Ending event-based regulation of GMO crops. Nat. Biotechnol. 2016, 34, 474–477. [Google Scholar] [CrossRef]
- Eckerstorfer, M.F.; Engelhard, M.; Heissenberger, A.; Simon, S.; Teichmann, H. Plants developed by new genetic modification techniques-comparison of existing regulatory frameworks in the EU and non-EU countries. Front. Bioeng. Biotechnol. 2019, 7, 26. [Google Scholar] [CrossRef] [Green Version]
- Grassini, P.; Eskridge, K.M.; Cassman, K.G. Distinguishing between yield advances and yield plateaus in historical crop production trends. Nat. Commun. 2013, 4, 2918. [Google Scholar] [CrossRef]
- Alexandratos, N.; Bruinsma, J. World Agriculture Towards 2030/2050: The 2012 Revision; FAO: Rome, Italy, 2012. [Google Scholar]
- Brookes, G.; Barfoot, P. GM crop technology use 1996-2018: Farm income and production impacts. GM Crops Food 2020, 11, 242–261. [Google Scholar] [CrossRef]
- Klumper, W.; Qaim, M. A meta-analysis of the impacts of genetically modified crops. PLoS ONE 2014, 9, e111629. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Xu, J.; Gao, Z.; Tian, H.; Gao, Y.; Kariman, K. Genetically modified crops are superior in their nitrogen use efficiency-a meta-analysis of three major cereals. Sci. Rep. 2020, 10, 8568. [Google Scholar] [CrossRef] [PubMed]
- Beatty, P.H.; Shrawat, A.K.; Carroll, R.T.; Zhu, T.; Good, A.G. Transcriptome analysis of nitrogen-efficient rice over-expressing alanine aminotransferase. Plant Biotechnol. J. 2009, 7, 562–576. [Google Scholar] [CrossRef] [PubMed]
- Good, A.G.; Johnson, S.J.; De Pauw, M.; Carroll, R.T.; Savidov, N.; Vidmar, J.; Lu, Z.; Taylor, G.; Stroeher, V. Engineering nitrogen use efficiency with alanine aminotransferase. Canad. J. Bot. 2007, 85, 252–262. [Google Scholar] [CrossRef]
- Pellegrino, E.; Bedini, S.; Nuti, M.; Ercoli, L. Impact of genetically engineered maize on agronomic, environmental and toxicological traits: A meta-analysis of 21 years of field data. Sci. Rep. 2018, 8, 3113. [Google Scholar] [CrossRef] [Green Version]
- Qaim, M. Bt cotton, yields and farmers’ benefits. Nat. Plants 2020, 6, 1318–1319. [Google Scholar] [CrossRef]
- Alvarez, F.; Manalo, A.; Clarete, R. Economic assessment of GM corn use in the Philippines. Int. J. Food Sci. Agric. 2021, 5, 115–128. [Google Scholar] [CrossRef]
- Sexton, S.E.; Zilberman, D. Land for food and fuel production: The role of agricultural biotechnology. In The Intended and Unintended Effects of U.S. Agricultural and Biotechnology Policies; Zivin, J.S.G., Perloff, J.M., Eds.; University of Chicago Press: Chicago, IL, USA, 2011; pp. 269–288. [Google Scholar]
- Brookes, G.; Barfoot, P. Environmental impacts of genetically modified (GM) crop use 1996–2018: Impacts on pesticide use and carbon emissions. GM Crops Food 2020, 11, 215–241. [Google Scholar] [CrossRef]
- Savary, S.; Willocquet, L.; Pethybridge, S.J.; Esker, P.; McRoberts, N.; Nelson, A. The global burden of pathogens and pests on major food crops. Nat. Ecol. Evol. 2019, 3, 430–439. [Google Scholar] [CrossRef]
- van Esse, H.P.; Reuber, T.L.; van der Does, D. Genetic modification to improve disease resistance in crops. New Phytol. 2020, 225, 70–86. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, S.A.; Pitz, K.Y.; Manshardt, R.; Zee, F.; Fitch, M.; Gonsalves, D. Virus coat protein transgenic papaya provides practical control of Papaya ringspot virus in Hawaii. Plant Dis. 2002, 86, 101–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonsalves, C.; Lee, D.; Gonsalves, D. Transgenic virus-resistant papaya: The Hawaiian ’rainbow’ was rapidly adopted by farmers and is of major importance in Hawaii today. APSnet Feature Artic. Available online: https://doi.org/10.1094/APSnetFeature-2004-0804 (accessed on 5 March 2022). [CrossRef] [Green Version]
- Kasote, D.; Sreenivasulu, N.; Acuin, C.; Regina, A. Enhancing health benefits of milled rice: Current status and future perspectives. Crit. Rev. Food Sci. Nutr. 2021, 1–21. [Google Scholar] [CrossRef] [PubMed]
- FAO; UNICEF; WFP; WHO. The State of Food Security and Nutrition in the World 2021: Transforming Food Systems for Food Security, Improved Nutrition and Affordable Healthy Diets for All; FAO: Rome, Italy, 2021; Volume 1, pp. 1–240. [Google Scholar]
- De Steur, H.; Mehta, S.; Gellynck, X.; Finkelstein, J.L. GM biofortified crops: Potential effects on targeting the micronutrient intake gap in human populations. Curr. Opin. Biotechnol. 2017, 44, 181–188. [Google Scholar] [CrossRef]
- Zhu, Q.; Wang, B.; Tan, J.; Liu, T.; Li, L.; Liu, Y.G. Plant synthetic metabolic engineering for enhancing crop nutritional quality. Plant Commun. 2020, 1, 100017. [Google Scholar] [CrossRef]
- Mallikarjuna Swamy, B.P.; Marundan, S., Jr.; Samia, M.; Ordonio, R.L.; Rebong, D.B.; Miranda, R.; Alibuyog, A.; Rebong, A.T.; Tabil, M.A.; Suralta, R.R.; et al. Development and characterization of GR2E Golden rice introgression lines. Sci. Rep. 2021, 11, 2496. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Prevalence of Vitamin a Deficiency in Populations at Risk 1995–2005: WHO Global Database on Vitamin a Deficiency; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- Garg, M.; Sharma, N.; Sharma, S.; Kapoor, P.; Kumar, A.; Chunduri, V.; Arora, P. Biofortified crops generated by breeding, agronomy, and transgenic approaches are improving lives of millions of people around the world. Front. Nutr. 2018, 5, 12. [Google Scholar] [CrossRef]
- Naqvi, S.; Zhu, C.; Farre, G.; Ramessar, K.; Bassie, L.; Breitenbach, J.; Perez Conesa, D.; Ros, G.; Sandmann, G.; Capell, T.; et al. Transgenic multivitamin corn through biofortification of endosperm with three vitamins representing three distinct metabolic pathways. Proc. Natl. Acad. Sci. USA 2009, 106, 7762–7767. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.P.; Gruissem, W.; Bhullar, N.K. Single genetic locus improvement of iron, zinc and beta-carotene content in rice grains. Sci. Rep. 2017, 7, 6883. [Google Scholar] [CrossRef]
- Blancquaert, D.; Van Daele, J.; Strobbe, S.; Kiekens, F.; Storozhenko, S.; De Steur, H.; Gellynck, X.; Lambert, W.; Stove, C.; Van Der Straeten, D. Improving folate (vitamin B9) stability in biofortified rice through metabolic engineering. Nat. Biotechnol. 2015, 33, 1076–1078. [Google Scholar] [CrossRef]
- Storozhenko, S.; De Brouwer, V.; Volckaert, M.; Navarrete, O.; Blancquaert, D.; Zhang, G.F.; Lambert, W.; Van Der Straeten, D. Folate fortification of rice by metabolic engineering. Nat. Biotechnol. 2007, 25, 1277–1279. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Immunization Coverage. Available online: https://www.who.int/news-room/fact-sheets/detail/immunization-coverage (accessed on 4 January 2021).
- Kurup, V.M.; Thomas, J. Edible vaccines: Promises and challenges. Mol. Biotechnol. 2020, 62, 79–90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rukavtsova, E.B.; Rudenko, N.V.; Puchko, E.N.; Zakharchenko, N.S.; Buryanov, Y.I. Study of the immunogenicity of hepatitis B surface antigen synthesized in transgenic potato plants with increased biosafety. J. Biotechnol. 2015, 203, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.B.; Ganapathi, T.R.; Revathi, C.J.; Srinivas, L.; Bapat, V.A. Expression of hepatitis B surface antigen in transgenic banana plants. Planta 2005, 222, 484–493. [Google Scholar] [CrossRef] [PubMed]
- Davod, J.; Fatemeh, D.N.; Honari, H.; Hosseini, R. Constructing and transient expression of a gene cassette containing edible vaccine elements and shigellosis, anthrax and cholera recombinant antigens in tomato. Mol. Biol. Rep. 2018, 45, 2237–2246. [Google Scholar] [CrossRef]
- Karasev, A.V.; Foulke, S.; Wellens, C.; Rich, A.; Shon, K.J.; Zwierzynski, I.; Hone, D.; Koprowski, H.; Reitz, M. Plant based HIV-1 vaccine candidate: Tat protein produced in spinach. Vaccine 2005, 23, 1875–1880. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, M.; Li, Y.; Zhao, Y.; He, H.; Yang, G.; Zheng, C. Oral immunogenicity and protective efficacy in mice of a carrot-derived vaccine candidate expressing UreB subunit against Helicobacter pylori. Protein Expr. Purif. 2010, 69, 127–131. [Google Scholar] [CrossRef]
- Lossl, A.G.; Waheed, M.T. Chloroplast-derived vaccines against human diseases: Achievements, challenges and scopes. Plant Biotechnol. J. 2011, 9, 527–539. [Google Scholar] [CrossRef]
- Ali, Q.; Yu, C.; Hussain, A.; Ali, M.; Ahmar, S.; Sohail, M.A.; Riaz, M.; Ashraf, M.F.; Abdalmegeed, D.; Wang, X.; et al. Genome engineering technology for durable disease resistance: Recent progress and future outlooks for sustainable agriculture. Front. Plant Sci. 2022, 13, 860281. [Google Scholar] [CrossRef]
- Bucchini, L.; Goldman, L.R. Starlink corn: A risk analysis. Environ. Health Perspect. 2002, 110, 5–13. [Google Scholar] [CrossRef] [Green Version]
- Bruhn, C.M. Starlink Corn: What Happened. Available online: https://ccr.ucdavis.edu/biotechnology/starlink-corn-what-happened (accessed on 4 January 2022).
- Seegerweiss. Starlink Genetically Modified Corn Contamination. Available online: https://www.seegerweiss.com/commercial-litigation/star-link-genetically-modified-corn-seed-settlement/ (accessed on 4 January 2022).
- Centers for Disease Control and Prevention (CDC). Investigation of Human Health Effects Associated with Potential Exposure to Genetically Modified Corn; CDC: Atlanta, GA, USA, 2001; pp. 1–24. [Google Scholar]
- Ladics, G.S.; Bartholomaeus, A.; Bregitzer, P.; Doerrer, N.G.; Gray, A.; Holzhauser, T.; Jordan, M.; Keese, P.; Kok, E.; Macdonald, P.; et al. Genetic basis and detection of unintended effects in genetically modified crop plants. Transgenic Res. 2015, 24, 587–603. [Google Scholar] [CrossRef] [Green Version]
- Gu, X.; Liu, L.; Zhang, H. Transgene-free genome editing in plants. Front. Genome Ed. 2021, 3, 805317. [Google Scholar] [CrossRef] [PubMed]
- Lu, H.P.; Liu, S.M.; Xu, S.L.; Chen, W.Y.; Zhou, X.; Tan, Y.Y.; Huang, J.Z.; Shu, Q.Y. CRISPR-s: An active interference element for a rapid and inexpensive selection of genome-edited, transgene-free rice plants. Plant Biotechnol. J. 2017, 15, 1371–1373. [Google Scholar] [CrossRef] [PubMed]
- Aliaga-Franco, N.; Zhang, C.; Presa, S.; Srivastava, A.K.; Granell, A.; Alabadi, D.; Sadanandom, A.; Blazquez, M.A.; Minguet, E.G. Identification of transgene-free CRISPR-edited plants of rice, tomato, and Arabidopsis by monitoring dsred fluorescence in dry seeds. Front. Plant Sci. 2019, 10, 1150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, X.; Chen, J.; Dai, X.; Zhang, D.; Zhao, Y. An effective strategy for reliably isolating heritable and Cas9-free Arabidopsis mutants generated by CRISPR/Cas9-mediated genome editing. Plant Physiol. 2016, 171, 1794–1800. [Google Scholar] [CrossRef] [Green Version]
- He, Y.; Zhu, M.; Wang, L.; Wu, J.; Wang, Q.; Wang, R.; Zhao, Y. Programmed self-elimination of the CRISPR/Cas9 construct greatly accelerates the isolation of edited and transgene-free rice plants. Mol. Plant 2018, 11, 1210–1213. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The Food and Agriculture Organization (FAO). Women-Users, Preservers and Managers of Agro-Biodiversity. Available online: https://www.fao.org/3/x0171e/x0171e03.htm (accessed on 1 January 2020).
- Buiatti, M.; Christou, P.; Pastore, G. The application of GMOs in agriculture and in food production for a better nutrition: Two different scientific points of view. Genes Nutr. 2013, 8, 255–270. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, B.B. Resilience in agriculture through crop diversification: Adaptive management for environmental change. BioScience 2011, 61, 183–193. [Google Scholar] [CrossRef] [Green Version]
- Rubio, L.; Galipienso, L.; Ferriol, I. Detection of plant viruses and disease management: Relevance of genetic diversity and evolution. Front. Plant Sci. 2020, 11, 1092. [Google Scholar] [CrossRef]
- Plumer, B. Why Bayer’s Massive Deal to Buy Monsanto Is so Worrisome. Available online: https://www.vox.com/2016/9/14/12916344/monsanto-bayer-merger (accessed on 5 January 2022).
- Bratspies, R. Owning all the seeds: Consolidation and control in agbiotech. J. Environ. Law 2017, 47, 583–608. [Google Scholar]
- Demont, M.; Dillen, K.; Mathijs, E.; Tollens, E. GM crops in europe: How much value and for whom? EuroChoices 2007, 6, 46–53. [Google Scholar] [CrossRef]
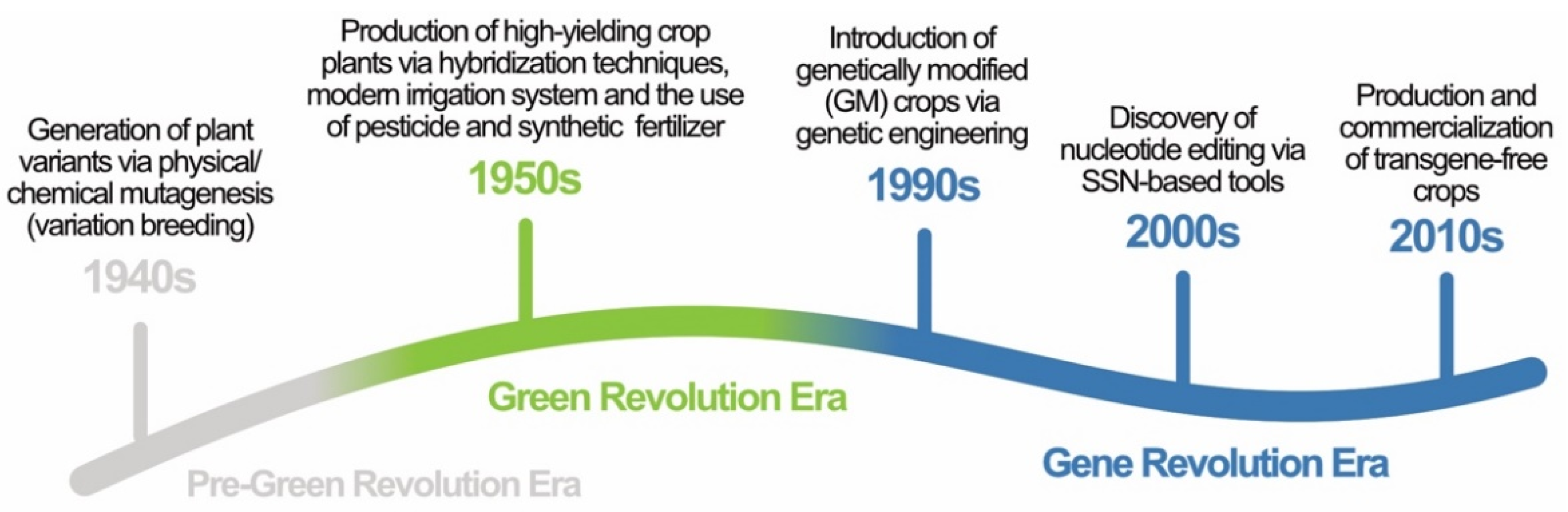
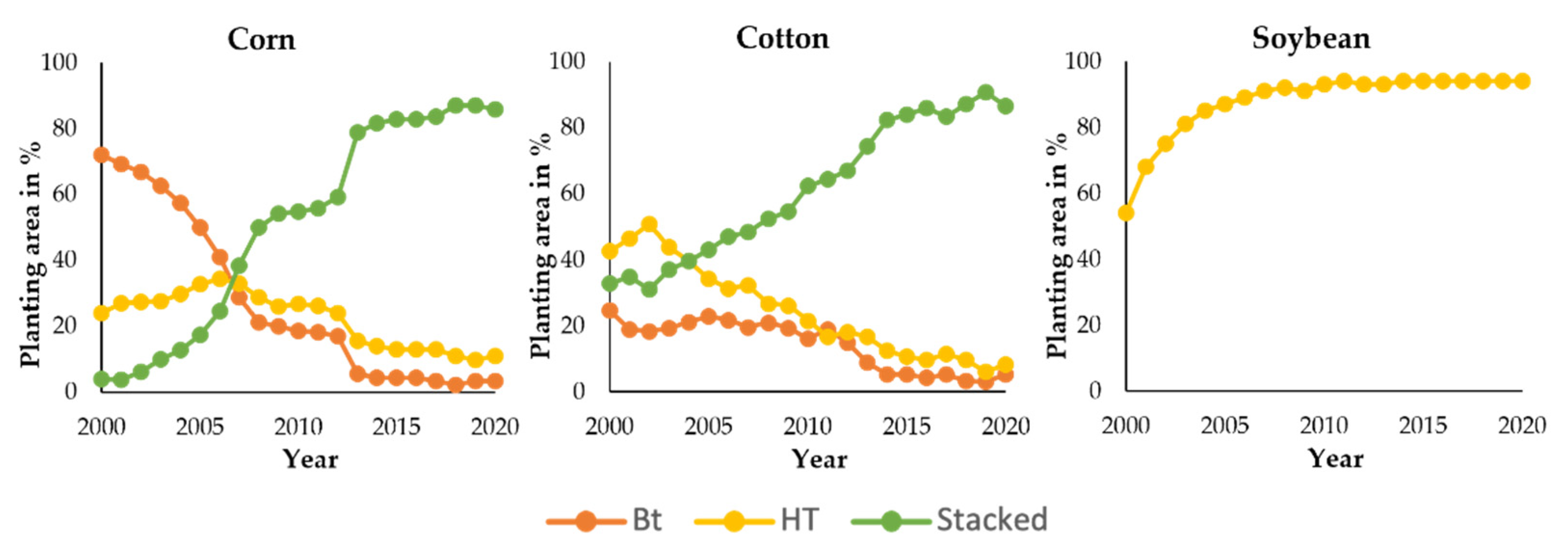
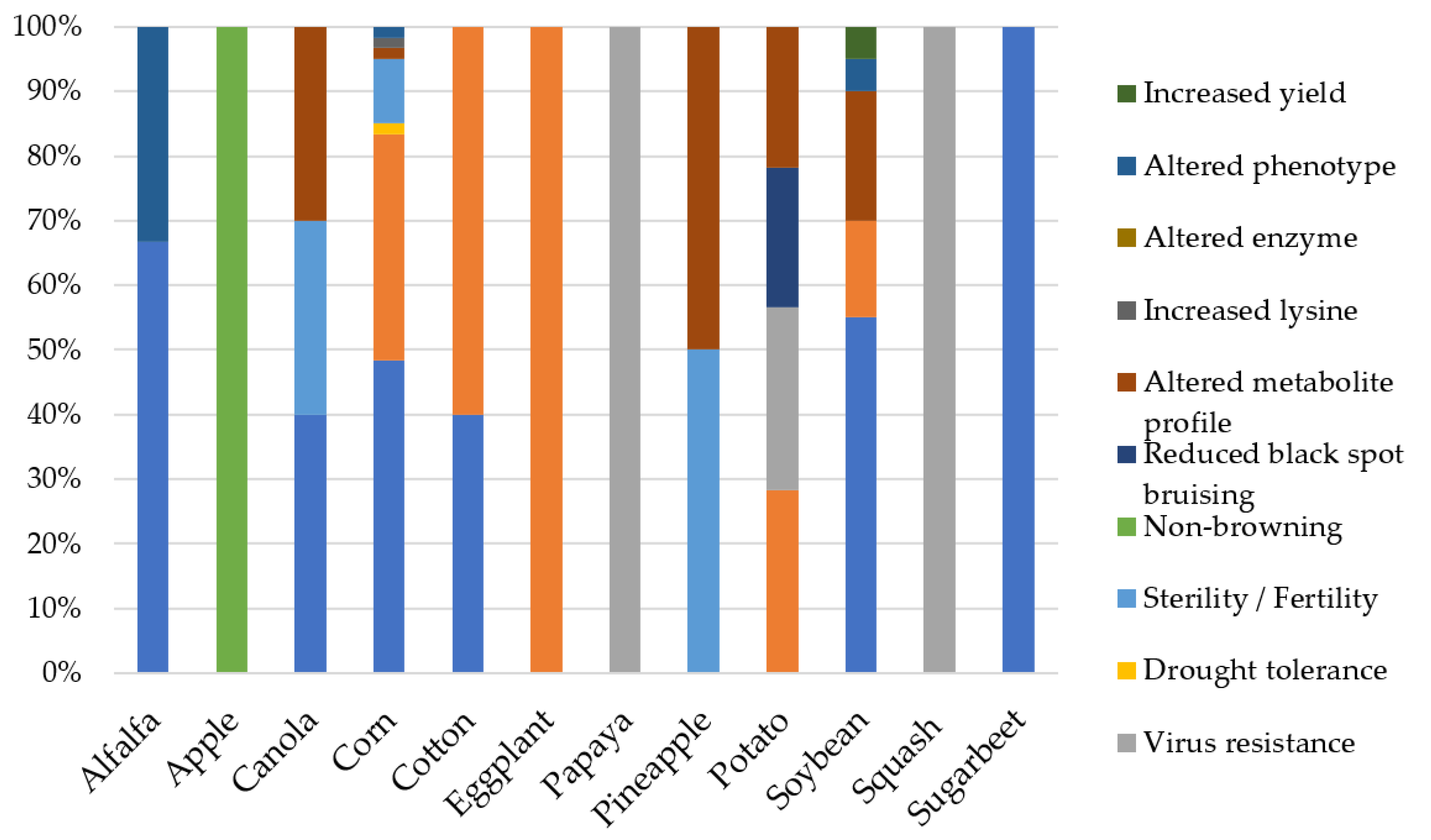
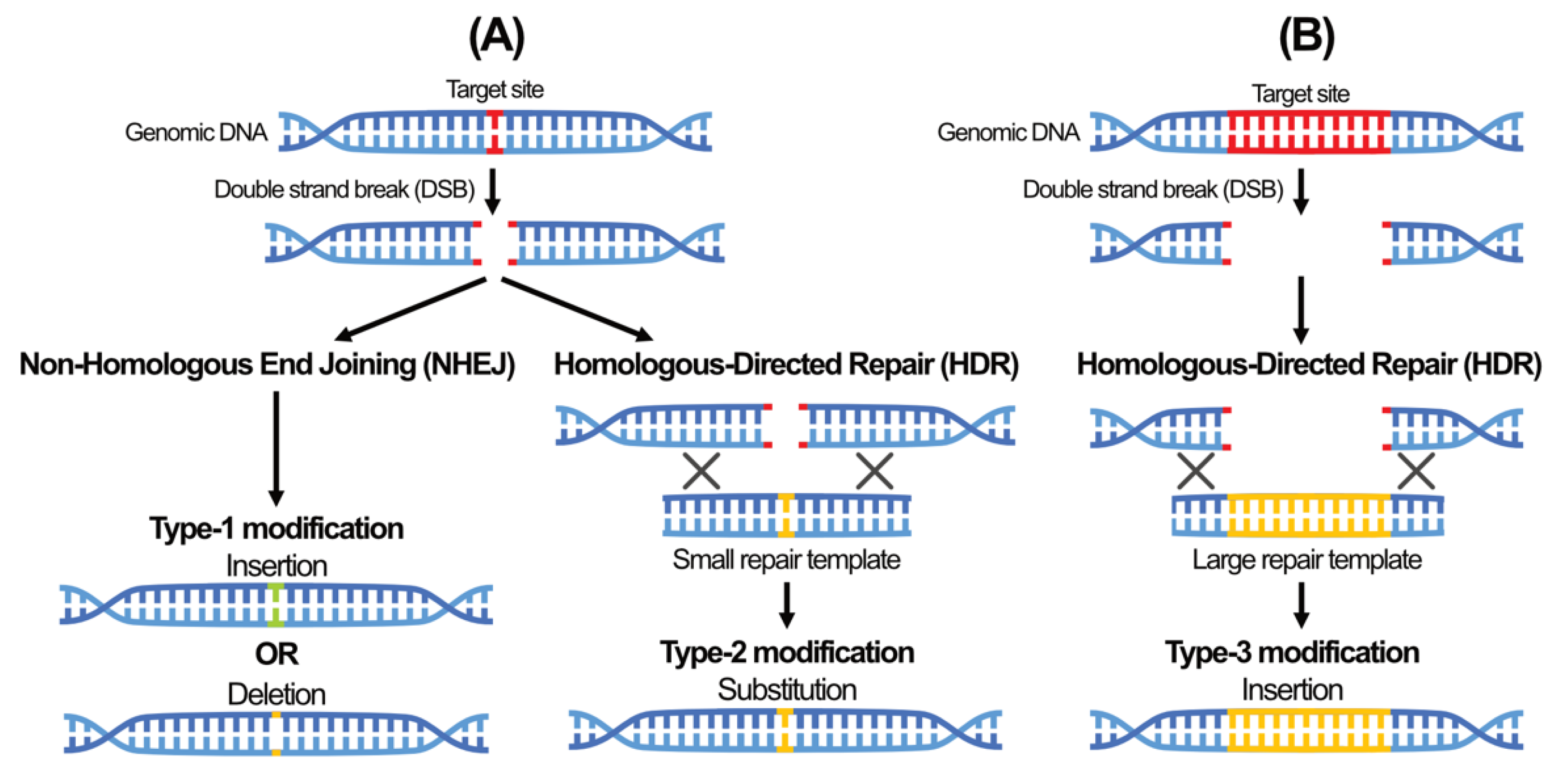
| Year | Milestone | References |
|---|---|---|
| ~11,000 years ago | The oldest evidence of domestication of ‘founder crops’ (einkorn wheat, emmer wheat, barley, lentil, pea, chickpea, bitter vetch, flax). | [39] |
| 1865 | Gregor Mendel discovers the foundational principles of inheritance in a living organism by studying the common pea plant (Pisum sativum). | [40] |
| 1897 | Agrobacterium was first isolated from a crown gall tumor. | [41,42] |
| 1898 | The first documented study on the tobacco mosaic virus (TMV), laying the foundation of virology. | [43] |
| 1907 | The causative agent of the crown gall tumor was discovered and named Bacterium tumefaciens. | [44] |
| 1940–1970 s | An in-depth study on the mechanism of crown gall tumorigenesis induced by Agrobacterium tumefaciens. | [35] |
| 1983 | The first transgenic plant was reported in tobacco (Nicotiana tabacum) harboring an antibiotic resistance gene. | [36] |
| 1986 | TMV-resistant transgenic tobacco was reported. | [37] |
| 1987 | Transgenic insect-resistant tobacco plant was reported. | [38] |
| 1990 | A ‘co-suppression’ phenomenon was observed in petunia (Petunia hybrida) that was genetically engineered to overexpress chalcone synthase (CHS). This started a new frontier in RNA interference (RNAi) research in living organisms. | [45] |
| 1992 | China became the first country to commercialize transgenic plants by introducing virus-resistant tobacco. | [46] |
| 1993 | The US Food and Drug Administration (FDA) approved the commercialization of the first transgenic food product, an RNAi-based ‘Flavr Savr’ tomato (cherry tomato; Lycopersicon esculentum). | [47] |
| 1993 | The European Union (EU) approved herbicide-resistant tobacco as the first genetically engineered crop to be commercialized in Europe. | [48] |
| 1995 | The US Environmental Protection Agency (EPA) approved the first pesticide-producing food crop (Bacillus thuringiensis [Bt] potato and Bt corn) and non-food crop (Bt cotton). | [49] |
| 1996 | Glyphosate-resistant soybean (Glycine max) became the first herbicide-resistant crop to be marketed for the consumer market in the US. | [50] |
| 2000 | Biofortified rice, known as ‘Golden Rice’, successfully demonstrated that engineering an entire biosynthetic pathway in an organism was possible. | [51] |
| 2000 | The first plant genome sequence was reported in Arabidopsis. | [52] |
| 2005 | The rice genome became the first crop plant to be sequenced. | [53] |
| 2005 | Golden Rice 2 with an increase in total carotenoids of up to 23-fold was reported. | [54] |
| 2009 | The first report of zinc-finger nuclease (ZFN) application in plants (corn). | [55] |
| 2012 | The first report of transcription activator-like effector (TALENS) application in plants (rice). | [56] |
| 2013 | The first report of clustered regularly interspersed short palindromic repeats (CRISPR) application in plants (rice and common wheat). | [57] |
| 2021 | First commercialization of a CRISPR-edited crop (tomato). | [58] |
| Crop | Target Site | Result | Reference |
|---|---|---|---|
| Model plants | |||
| Arabidopsis (Arabidopsis thaliana) | Transgene mutant GFP | Insertion and deletion mutations at the targeted 20 bp sequences; restoration of GFP functionality | [126] |
| Rice (Oryza sativa) | Promoter region of the bacterial blight susceptibility genes, OsSWEET14 and OsSWEET11 | Deletion and substitution mutations | [126] |
| Tobacco (Nicotiana tabacum) | Transgene mutant GFP | Insertion and deletion mutations at the targeted 20 bp sequences; restoration of GFP functionality | [126] |
| Benthi (Nicotiana benthamiana) | Nuclear-localization (PDS locus) of GFP-Cas9 expression | Deletion and substitution mutations | [127] |
| Food crops | |||
| Corn (maize; Zea mays) | Upstream of the LIGULELESS1 (LIG1), male fertility genes (Ms26 and Ms45), and acetolactate synthase genes (ALS1 and ALS2) | ALS2 editing yielded chlorsulfuron-resistant plants | [128] |
| Tomato (Solanum lycopersicum) | ANTHOCYANIN MUTANT1 (ANT1) | ANT1-overexpression, which encodes a Myb transcription factor, results in intensely purple plant tissue due to anthocyanin accumulation | [129] |
| Sorghum (Sorghum bicolor) | An out-of-frame red fluorescence protein gene (DsRED2) | Restoration of DsRED2 fluorescence | [126] |
| Soybean (Glycine max) | Transgene GFP; single-copy soybean gene, Glyma07g14530; homoeologous gene-pair, Glyma01g38150 and Glyma11g07220; homoeologous gene pair Glyma04g36150 and Glyma06g18790; soybean miRNAs, miR1514 and miR1509 | Loss of GFP fluorescence; variety of mutations, including deletions, SNPs, insertions, and replacements (two or more bases inserted after a deletion event) | [130] |
| Potato (Solanum tuberosum) | StIAA2 encoding an Aux/IAA protein involved in petiole hyponasty and shoot morphogenesis | Deletion, insertion, and substitution mutations | [131] |
| Kiwifruit (Actinidia chinensis) | CENTRORADIALIS (CEN)-like genes, AcCEN4 and AcCEN | Transformed a climbing woody perennial into a compact plant with rapid terminal flower and fruit development | [132] |
| Banana (Musa balbisiana) | Integrated endogenous banana streak virus (eBSV) sequences | eBSV knockout | [133] |
| Wheat (Tricium aestivum) | TaMLO | Insertion and deletion mutations frequencies of 26.5–38.0% | [57] |
| Industrial crops | |||
| Poplar (Populus tomentosa) | Populus tomentosa PHYTOENE DESATURASE GENE8 (PtoPDS) required for chlorophyll biosynthesis | Mutants with albino phenotype | [134] |
| Canola or rapeseed (Brassica napus) | ALCATRAZ (ALC). ALC is involved in valve margin development, therefore contributes to seed shattering from mature fruits | Increased shatter resistance (avoid seed loss during mechanical harvest) | [88] |
| Cotton (Gossypium hirsutum) | GhMYB25-like A and GhMYB25-like D | Deletion mutations of −1bp/−3bp/−7bp nucleotides and +1 bp insertion mutation; an indication of efficient genomic editing in the allotetraploid cotton genome | [135] |
| Rubber tree (Hevea brasiliensis) | FLOWERING LOCUS T (FT) and TERMINAL FLOWER1 (TFL1) | Mutation frequencies ranging from 3.74% to 20.11% at five target sites; Insertion and deletion patterns | [136] |
| Oil palm (Elaeis guineensis) | Elaeis guineensis PHYTOENE DESATURASE (EgPDS) | Insertions, deletions, and nucleotide substitutions, with a mutation efficiency of 62.5–83.33%; chimeric albino phenotypes | [137] |
| Moso bamboo (Phyllostachys edulis) | PePDS1 and PePDS2 | Insertion and deletion mutations; mutants with albino shoot phenotype | [138] |
| Ornamental plants | |||
| Indian chrysanthemum (Chrysanthemum nankingense) | Integrated Chiridius poppei (CpYGFP) expressing yellowish-green fluorescent protein | Mostly small deletions (1 bp); a large deletion (−1020 bp) was also observed | [139] |
| Japanese morning glory (Ipomoea nil) | EPHEMERAL1 (EPH1) crucial in petal senescence | 1-bp and/or 2-bp deletions occurred at the target sites | [140] |
| Coral lily (Lilium pumilum) and Easter lily (Lilium longiflorum) | PDS | Insertion, deletion and substitution; Mutants with completely albino, pale yellow and albino–green chimeric phenotypes | [141] |
| Petunia (Petunia hybrida) | DEEP PURPLE (DPL) | Insertion and deletion mutations; absence of the vein-associated anthocyanin pattern above the abaxial surface of the flower bud, but not corolla tube venation | [142] |
| Orchid (Phalaenopsis equestris) | MADS44, MADS36 and MADS8 | Insertion and deletion | [143] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamdan, M.F.; Mohd Noor, S.N.; Abd-Aziz, N.; Pua, T.-L.; Tan, B.C. Green Revolution to Gene Revolution: Technological Advances in Agriculture to Feed the World. Plants 2022, 11, 1297. https://doi.org/10.3390/plants11101297
Hamdan MF, Mohd Noor SN, Abd-Aziz N, Pua T-L, Tan BC. Green Revolution to Gene Revolution: Technological Advances in Agriculture to Feed the World. Plants. 2022; 11(10):1297. https://doi.org/10.3390/plants11101297
Chicago/Turabian StyleHamdan, Mohd Fadhli, Siti Nurfadhlina Mohd Noor, Nazrin Abd-Aziz, Teen-Lee Pua, and Boon Chin Tan. 2022. "Green Revolution to Gene Revolution: Technological Advances in Agriculture to Feed the World" Plants 11, no. 10: 1297. https://doi.org/10.3390/plants11101297
APA StyleHamdan, M. F., Mohd Noor, S. N., Abd-Aziz, N., Pua, T.-L., & Tan, B. C. (2022). Green Revolution to Gene Revolution: Technological Advances in Agriculture to Feed the World. Plants, 11(10), 1297. https://doi.org/10.3390/plants11101297






