Determination of 1-Deoxynojirimycin (1-DNJ) in Leaves of Italian or Italy-Adapted Cultivars of Mulberry (Morus sp.pl.) by HPLC-MS
Abstract
1. Introduction
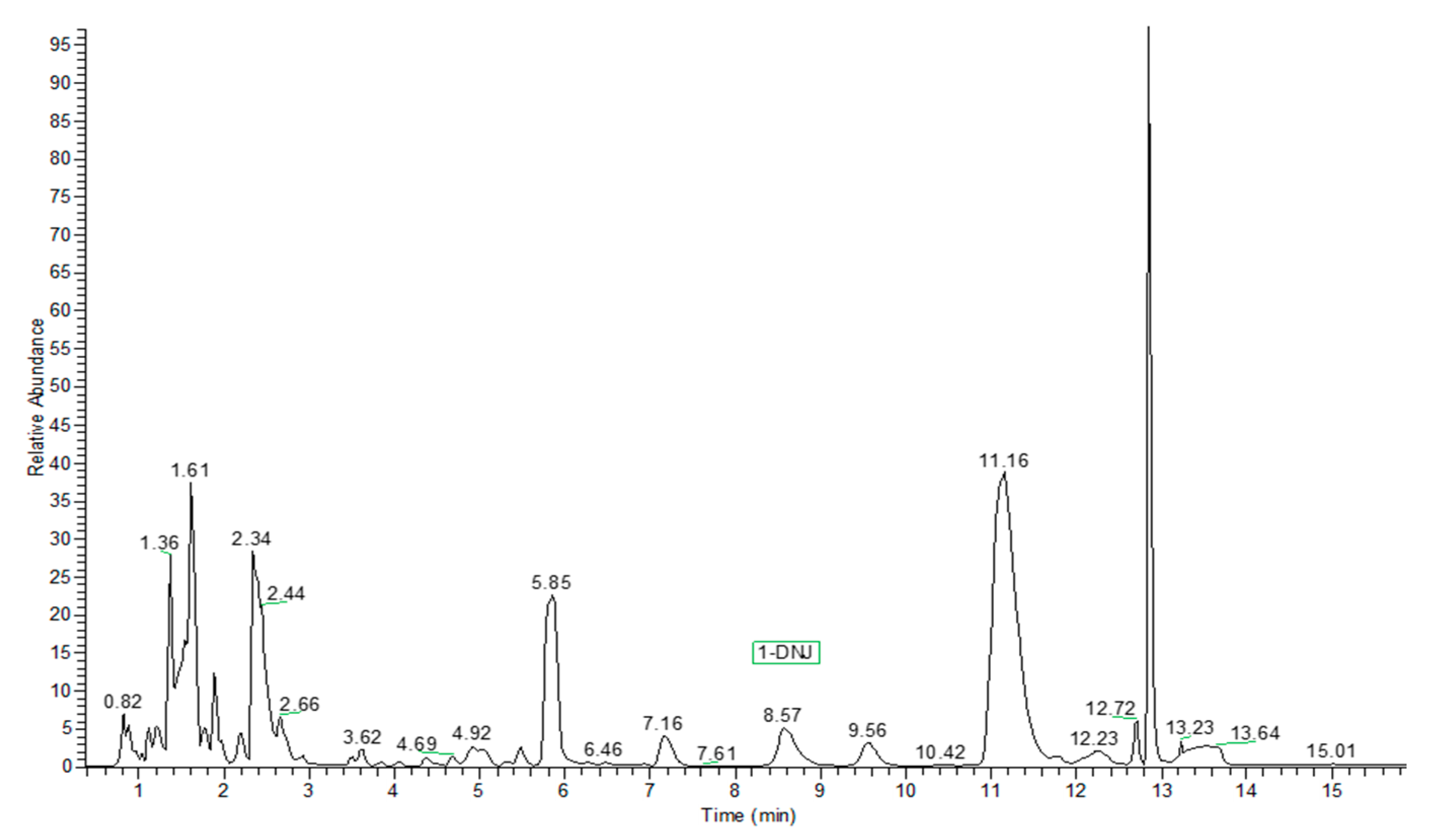
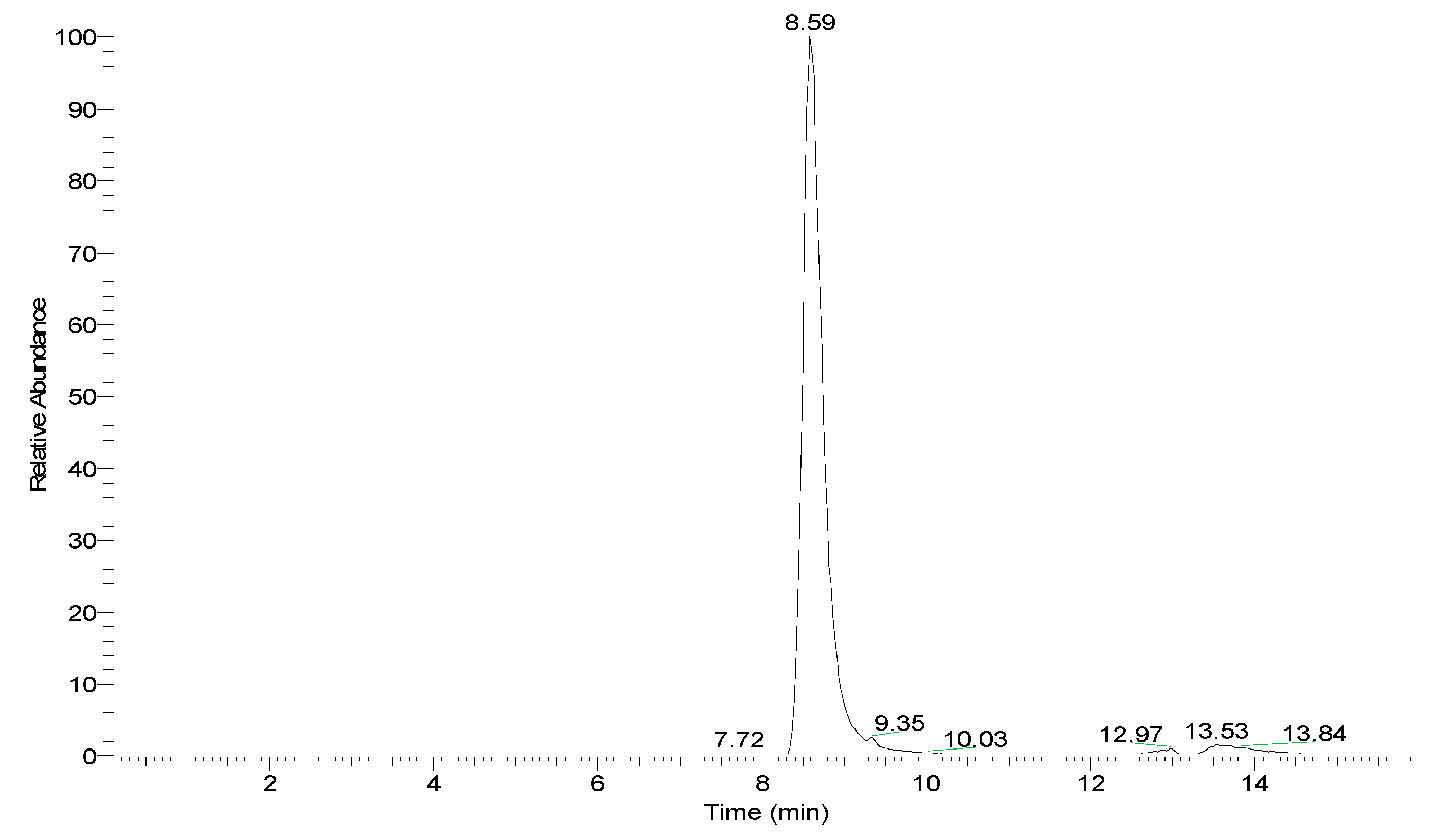
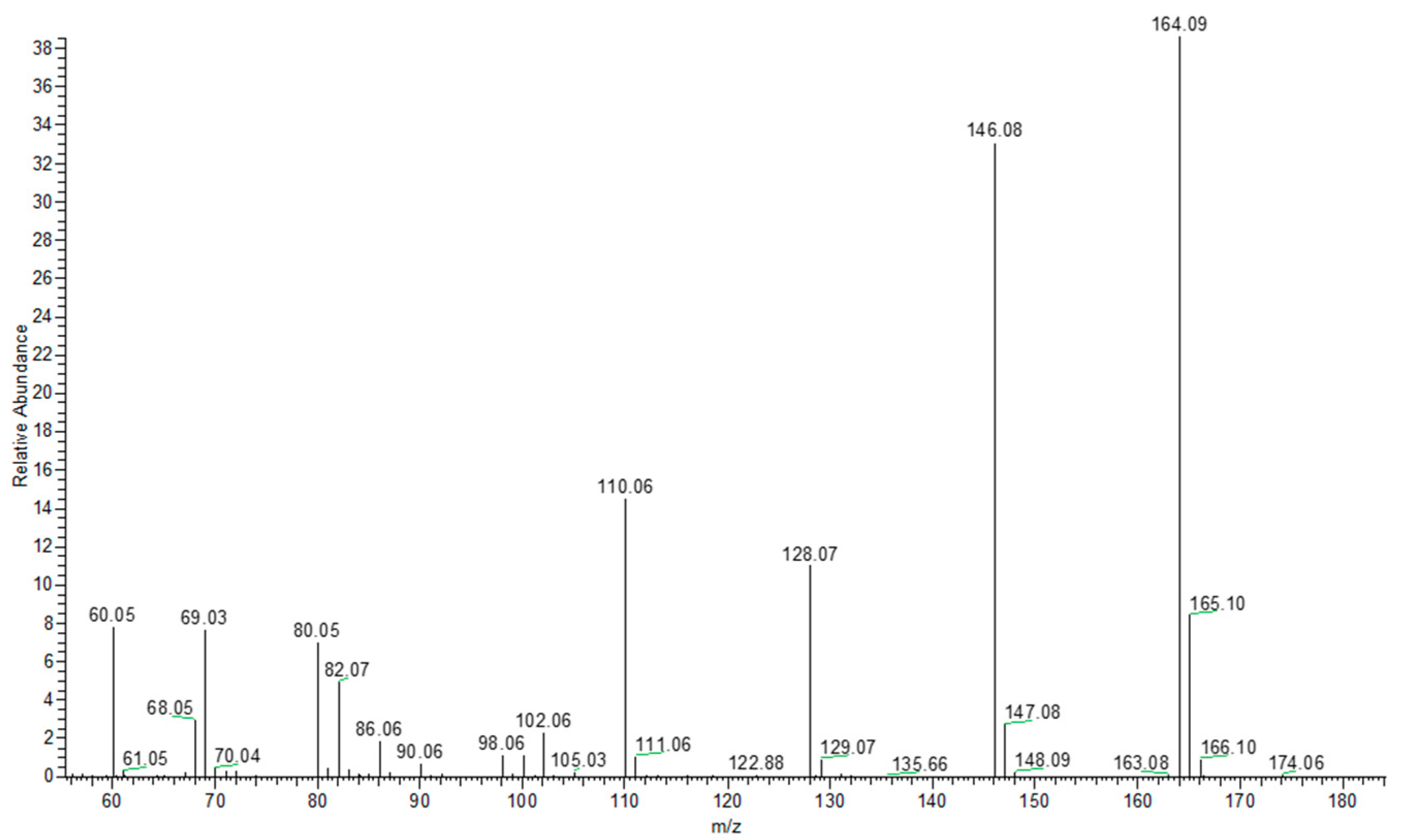
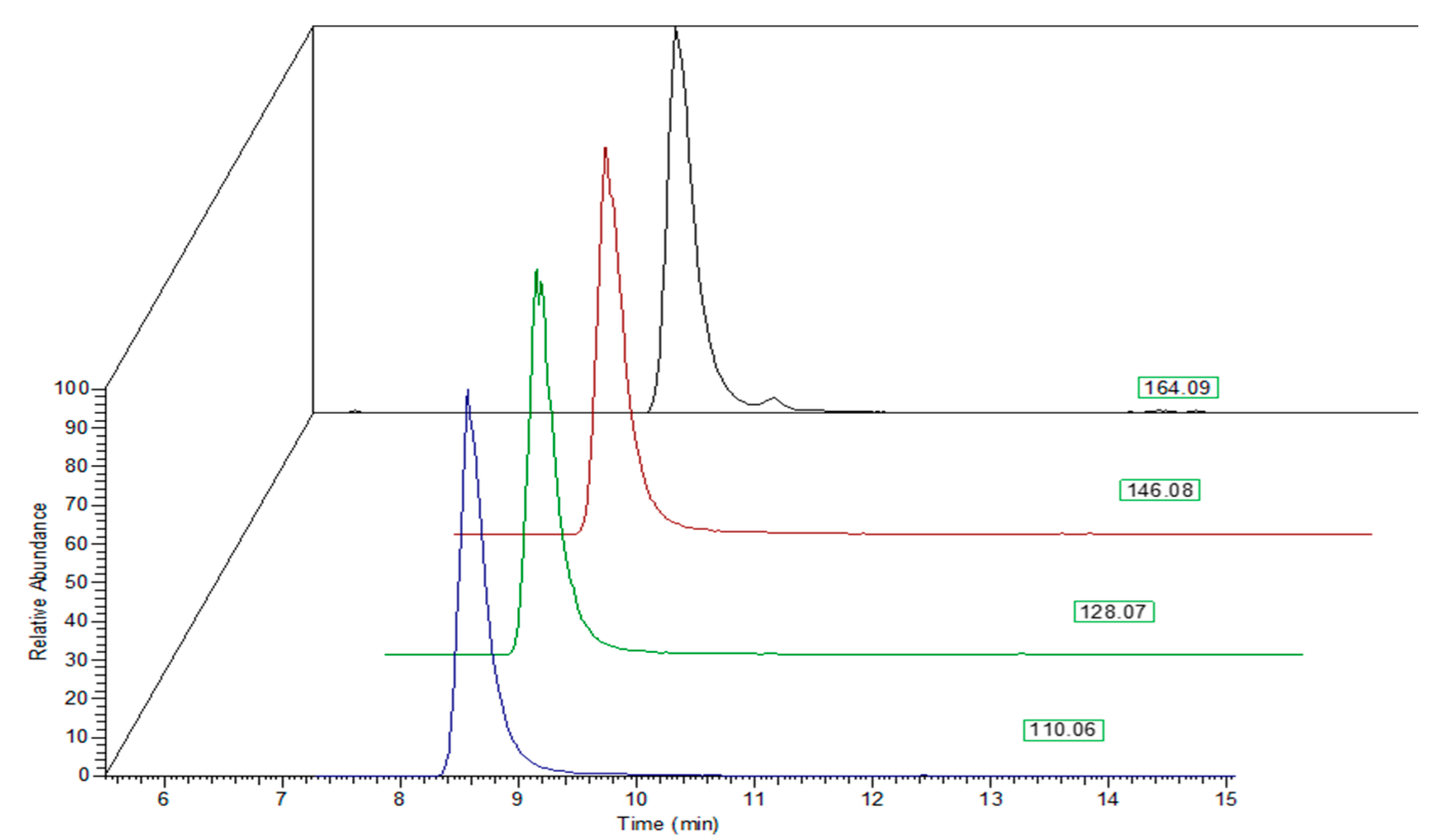
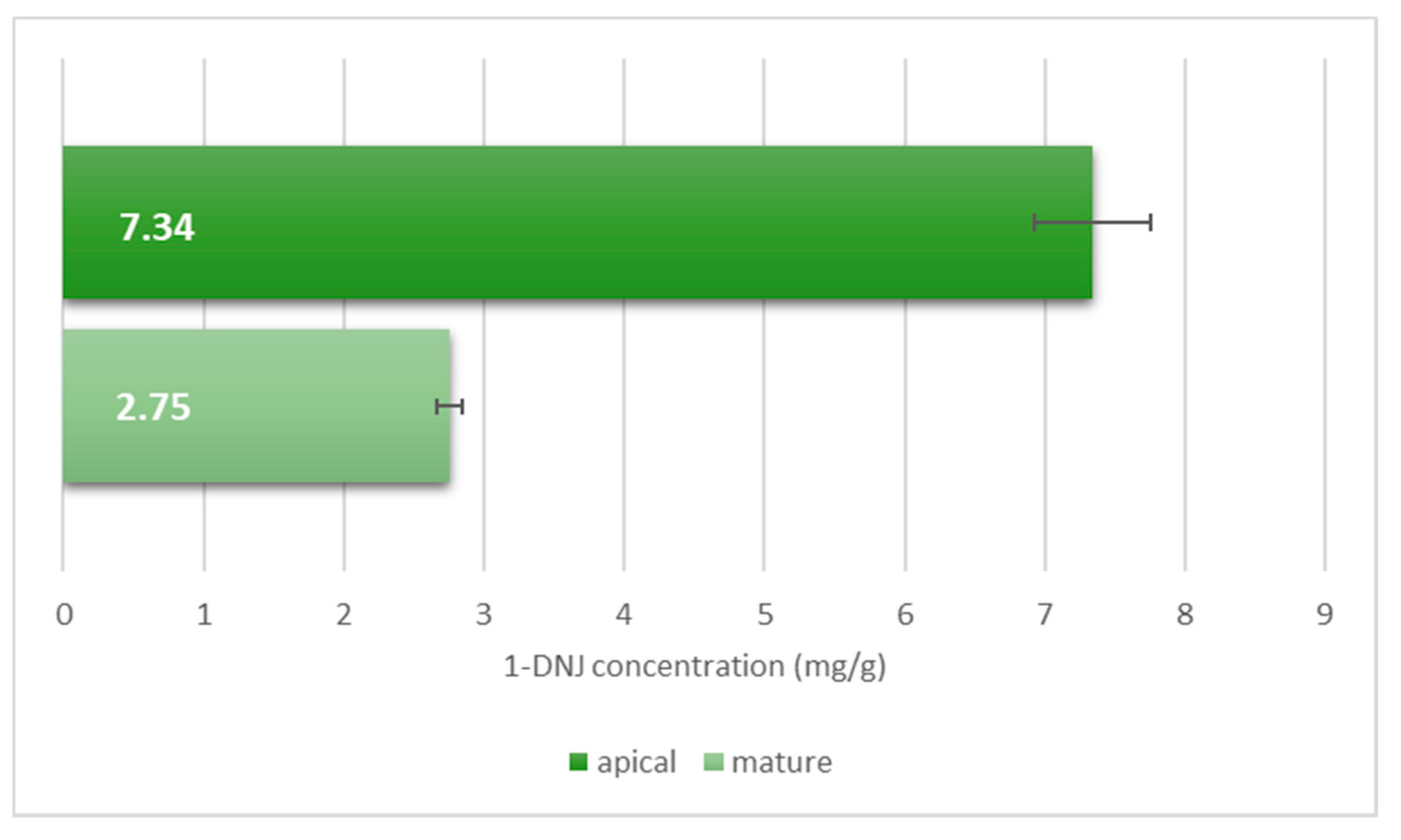
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals and Solvents
3.2. Plant Material and Extraction Procedure
3.3. HPLC-ESI-MS Analysis
3.4. Method Validation
3.5. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Nasri, H.; Baradaran, A.; Shirzad, H.; Rafieian-Kopaei, M. New Concepts in Nutraceuticals as Alternative for Pharmaceuticals. Int. J. Prev. Med. 2014, 5, 1487–1499. [Google Scholar]
- Zamoner, L.O.B.; Aragão-Leoneti, V.; Carvalho, I. Iminosugars: Effects of Stereochemistry, Ring Size, and N-Substituents on Glucosidase Activities. Pharmaceuticals 2019, 12, 108. [Google Scholar] [CrossRef]
- Yagi, Y.M. The Structure of Moranoline, a Piperidine Alkaloid from Morus Species. Nippon Nogei Kagaku Kaishi 1976, 50, 571–572. [Google Scholar] [CrossRef]
- Konno, K.; Ono, H.; Nakamura, M.; Tateishi, K.; Hirayama, C.; Tamura, Y.; Hattori, M.; Koyama, A.; Kohno, K. Mulberry Latex Rich in Antidiabetic Sugar-Mimic Alkaloids Forces Dieting on Caterpillars. Proc. Natl. Acad. Sci. USA 2006, 103, 1337–1341. [Google Scholar] [CrossRef]
- Lemus, I.; García, R.; Delvillar, E.; Knop, G. Hypoglycaemic Activity of Four Plants Used in Chilean Popular Medicine. Phytother. Res. 1999, 13, 91–94. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, J.; Chan, P. Treating Type 2 Diabetes Mellitus with Traditional Chinese and Indian Medicinal Herbs. Evid. Based Complement. Alternat. Med. 2013, 2013, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Zhu, F.; Chen, K. 1-Deoxynojirimycin: Sources, Extraction, Analysis and Biological Functions. Nat. Prod. Commun. 2017, 12, 1934578X1701200934. [Google Scholar] [CrossRef]
- Bajpai, S.; Rao, A.V.B. Quantitative Determination of 1-Deoxynojirimycin in Different Mulberry Varieties of India. J. Pharmacogn. Phytochem. 2014, 3, 17–22. [Google Scholar]
- Kimura, T.; Nakagawa, K.; Kubota, H.; Kojima, Y.; Goto, Y.; Yamagishi, K.; Oita, S.; Oikawa, S.; Miyazawa, T. Food-Grade Mulberry Powder Enriched with 1-Deoxynojirimycin Suppresses the Elevation of Postprandial Blood Glucose in Humans. J. Agric. Food Chem. 2007, 55, 5869–5874. [Google Scholar] [CrossRef] [PubMed]
- Yatsunami, K.; Ichida, M.; Onodera, S. The Relationship between 1-Deoxynojirimycin Content and α-Glucosidase Inhibitory Activity in Leaves of 276 Mulberry Cultivars (Morus Spp.) in Kyoto, Japan. J. Nat. Med. 2008, 62, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Wang, H.-J.; Bucheli, P.; Zhang, P.-F.; Wei, D.-Z.; Lu, Y.-H. Phytochemical Profiles of Different Mulberry (Morus sp.) Species from China. J. Agric. Food Chem. 2009, 57, 9133–9140. [Google Scholar] [CrossRef]
- Vichasilp, C.; Nakagawa, K.; Sookwong, P.; Higuchi, O.; Luemunkong, S.; Miyazawa, T. Development of High 1-Deoxynojirimycin (DNJ) Content Mulberry Tea and Use of Response Surface Methodology to Optimize Tea-Making Conditions for Highest DNJ Extraction. LWT—Food Sci. Technol. 2012, 45, 226–232. [Google Scholar] [CrossRef]
- Esti Wulandari, Y.R.; Prasasty, V.D.; Rio, A.; Geniola, C. Determination of 1-Deoxynojirimycin Content and Phytochemical Profiles from Young and Mature Mulberry Leaves of Morus Spp. OnLine J. Biol. Sci. 2019, 19, 124–131. [Google Scholar] [CrossRef][Green Version]
- Hao, J.-Y.; Wan, Y.; Yao, X.-H.; Zhao, W.-G.; Hu, R.-Z.; Chen, C.; Li, L.; Zhang, D.-Y.; Wu, G.-H. Effect of Different Planting Areas on the Chemical Compositions and Hypoglycemic and Antioxidant Activities of Mulberry Leaf Extracts in Southern China. PLoS ONE 2018, 13, e0198072. [Google Scholar] [CrossRef]
- Przygoński, K.; Wojtowicz, E. The Optimization of Extraction Process of White Mulberry Leaves and the Characteristic Bioactive Properties Its Powder Extract. Herba Pol. 2019, 65, 12–19. [Google Scholar] [CrossRef]
- Tomotake, H.; Katagiri, M.; Yamato, M. Silkworm Pupae (Bombyx Mori) Are New Sources of High Quality Protein and Lipid. J. Nutr. Sci. Vitaminol. 2010, 56, 446–448. [Google Scholar] [CrossRef]
- Tong, T.-T.; Zhao, E.-H.; Gao, H.-L.; Xu, Y.-H.; Zhao, Y.-J.; Fu, G.; Cui, H. Recent Research Advances of 1-Deoxynojirimycin and Its Derivatives. China J. Chin. Mater. Med. 2018, 43, 1990–1997. [Google Scholar] [CrossRef]
- Ryu, K.S.; Lee, H.S.; Kim, K.Y.; Kim, M.J.; Kang, P.D. Heat Stability and Glucose-Lowering Effect of 1-Deoxynojirimycin from Silkworm (Bombyx Mori) Extract Powder. Int. J. Ind. Entomol. 2013, 27, 277–281. [Google Scholar] [CrossRef][Green Version]
- Rodríguez-Sánchez, S.; Hernández-Hernández, O.; Ruiz-Matute, A.I.; Sanz, M.L. A Derivatization Procedure for the Simultaneous Analysis of Iminosugars and Other Low Molecular Weight Carbohydrates by GC–MS in Mulberry (Morus Sp.). Food Chem. 2011, 126, 353–359. [Google Scholar] [CrossRef]
- Kimura, T.; Nakagawa, K.; Saito, Y.; Yamagishi, K.; Suzuki, M.; Yamaki, K.; Shinmoto, H.; Miyazawa, T. Determination of 1-Deoxynojirimycin in Mulberry Leaves Using Hydrophilic Interaction Chromatography with Evaporative Light Scattering Detection. J. Agric. Food Chem. 2004, 52, 1415–1418. [Google Scholar] [CrossRef]
- Nakagawa, K.; Ogawa, K.; Higuchi, O.; Kimura, T.; Miyazawa, T.; Hori, M. Determination of Iminosugars in Mulberry Leaves and Silkworms Using Hydrophilic Interaction Chromatography—Tandem Mass Spectrometry. Anal. Biochem. 2010, 404, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Tolstikov, V.V.; Fiehn, O. Analysis of Highly Polar Compounds of Plant Origin: Combination of Hydrophilic Interaction Chromatography and Electrospray Ion Trap Mass Spectrometry. Anal. Biochem. 2002, 301, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Burgess, K.S.; Morgan, M.; Deverno, L.; Husband, B.C. Asymmetrical Introgression between Two Morus Species (M. Alba, M. Rubra) That Differ in Abundance. Mol. Ecol. 2005, 14, 3471–3483. [Google Scholar] [CrossRef]
- Nepal, M.P.; Ferguson, C.J. Phylogenetics of Morus (Moraceae) Inferred from ITS and TrnL-TrnF Sequence Data. Syst. Bot. 2012, 37, 442–450. [Google Scholar] [CrossRef]
- Zeng, Q.; Chen, H.; Zhang, C.; Han, M.; Li, T.; Qi, X.; Xiang, Z.; He, N. Definition of Eight Mulberry Species in the Genus Morus by Internal Transcribed Spacer-Based Phylogeny. PLoS ONE 2015, 10, e0135411. [Google Scholar] [CrossRef] [PubMed]
- Gąsecka, M.; Siwulski, M.; Magdziak, Z.; Budzyńska, S.; Stuper-Szablewska, K.; Niedzielski, P.; Mleczek, M. The Effect of Drying Temperature on Bioactive Compounds and Antioxidant Activity of Leccinum Scabrum (Bull.) Gray and Hericium Erinaceus (Bull.) Pers. J. Food Sci. Technol. 2020, 57, 513–525. [Google Scholar] [CrossRef]
- Chlumská, Z.; Janeček, S.; Doležal, J. How to Preserve Plant Samples for Carbohydrate Analysis? Test of Suitable Methods Applicable in Remote Areas. Folia Geobot. 2013, 49, 1–15. [Google Scholar] [CrossRef]
- Hu, X.-Q.; Jiang, L.; Zhang, J.-G.; Deng, W.; Wang, H.-L.; Wei, Z.-J. Quantitative Determination of 1-Deoxynojirimycin in Mulberry Leaves from 132 Varieties. Ind. Crops Prod. 2013, 49, 782–784. [Google Scholar] [CrossRef]
- Han, W.; Chen, X.; Yu, H.; Chen, L.; Shen, M. Seasonal Variations of Iminosugars in Mulberry Leaves Detected by Hydrophilic Interaction Chromatography Coupled with Tandem Mass Spectrometry. Food Chem. 2018, 251, 110–114. [Google Scholar] [CrossRef]
- Asano, N.; Oseki, K.; Tomioka, E.; Kizu, H.; Matsui, K. N-Containing Sugars from Morus Alba and Their Glycosidase Inhibitory Activities. Carbohydr. Res. 1994, 259, 243–255. [Google Scholar] [CrossRef]
- Sugiyama, M.; Katsube, T.; Koyama, A.; Itamura, H. Seasonal Changes in Functional Component Contents in Mulberry (Morus Alba L.) Leaves. Hortic. J. 2017, 86, 534–542. [Google Scholar] [CrossRef]
- Nakanishi, H.; Onose, S.; Kitahara, E.; Chumchuen, S.; Takasaki, M.; Konishi, H.; Kanekatsu, R. Effect of Environmental Conditions on the α-Glucosidase Inhibitory Activity of Mulberry Leaves. Biosci. Biotechnol. Biochem. 2011, 75, 2293–2296. [Google Scholar] [CrossRef] [PubMed]
- Shibano, M.; Fujimoto, Y.; Kushino, K.; Kusano, G.; Baba, K. Biosynthesis of 1-Deoxynojirimycin in Commelina Communis: A Difference between the Microorganisms and Plants. Phytochemistry 2004, 65, 2661–2665. [Google Scholar] [CrossRef]
- Prediction of Worldwide Energy Resource. Available online: https://power.larc.nasa.gov/data-access-viewer/ (accessed on 24 May 2021).
- Power Data Methodology. Available online: https://power.larc.nasa.gov/docs/methodology/ (accessed on 24 May 2021).
- Vichasilp, C.; Nakagawa, K.; Sookwong, P.; Suzuki, Y.; Kimura, F.; Higuchi, O.; Miyazawa, T. Optimization of 1-Deoxynojirimycin Extraction from Mulberry Leaves by Using Response Surface Methodology. Biosci. Biotechnol. Biochem. 2009, 73, 2684–2689. [Google Scholar] [CrossRef] [PubMed]
| Cultivar | End-May 2017 | ||
|---|---|---|---|
| Humidity (%) | Fresh Samples (mg/g of Dry Matter) a | Dry Samples (mg/g of Dry Matter) a | |
| Aobanezumi (M. alba L.) | 69.4 | 0.55 ± 0.02 m,n | 0.44 ± 0.06 z |
| Cattaneo female (M. alba L.) | 66.3 | 0.66 ± 0.02 m,n | 0.59 ± 0.03 z,v |
| Cattaneo male (M. alba L.) | 70.5 | 1.87 ± 0.02 g,h,i | 1.50 ± 0.03 o,p,q |
| Egyptienne (M. alba L.) | 52.9 | 1.78 ± 0.04 g,h,i | 1.66 ± 0.08 o,n |
| Florio (M. alba L.) | 70.7 | 0.74 ± 0.02 n,m | 0.55 ± 0.02 v,z |
| Giazzola (M. alba L.) | 67.7 | 1.33 ± 0.05 i,l | 1.20 ± 0.05 s,t |
| Kayriounezumigaeshi (M. alba L.) | 68.1 | 1.99 ± 0.04 g,h | 1.86 ± 0.08 l,m |
| Limoncina (M. alba L.) | 64.2 | 1.01 ± 0.02 l,m | 0.93 ± 0.05 u |
| Morettiana (M. alba L.) | 72.5 | 1.36 ± 0.02 i,l | 1.26 ± 0.04 r,s |
| Nervosa (M. alba L.) | 74.4 | 1.70 ± 0.04 h,i | 1.62 ± 0.06 n,o,p |
| Pendula (M. alba L.) | 72.6 | 1.49 ± 0.05 h,i,l | 1.43 ± 0.04 q,r |
| Pyramidalis (M. alba L.) | 70.2 | 2.19 ± 0.06 g | 2.07 ± 0.08 i |
| Spain Black fruit (M. alba L.) | 70.3 | 1.81 ± 0.02 g,h,i | 1.76 ± 0.07 m,n |
| Ukraina (M. alba L.) | 70.5 | 2.99 ± 0.03 f | 2.94 ± 0.06 f |
| Filippine (M. alba L.) | 65.9 | 2.26 ± 0.11 n | 2.22 ± 0.11 h |
| Kokosou 21 (M. alba L.) | 70.4 | 3.17 ± 0.11 f | 2.50 ± 0.04 g |
| Restelli (M. alba L.) | 72.3 | 4.56 ± 0.11 d | 3.45 ± 0.05 e |
| Gorgeous (M. alba L.) | 70.6 | 5.65 ± 0.11 c | 4.24 ± 0.01 d |
| Queensland black (M. alba L.) | 64.4 | 3.56 ± 0.11 f,e | 2.96 ± 0.02 f |
| Romana lhou (M. alba L.) | 70.7 | 8.55 ± 0.11 b | 6.86 ± 0.22 b |
| Wildtype (M. nigra L.) | 71.9 | 5.99 ± 0.11 c | 4.46 ± 0.04 c |
| Chirtut (M. nigra L.) | 55.2 | 0.56 ± 0.11 m,n | 0.66 ± 0.03 v |
| Date-Akagi (M. alba L.) | 68.7 | 1.61 ± 0.11 h,i | 1.45 ± 0.21 p,q |
| Akagi (M. alba L.) | 61.2 | 1.57 ± 0.11 h,i | 1.22 ± 0.03 s |
| Illinois everbearing (M. rubra L.) | 59.8 | 1.74 ± 0.11 g,h,i | 1.96 ± 0.02 i,l |
| Muki (M. alba L.) | 69.5 | 5.66 ± 0.11 c | 4.49 ± 0.03 c |
| Korin (M. alba L.) | 72.2 | 9.55 ± 0.11 a | 7.07 ± 0.14 a |
| Miura (M. alba L.) | 70.7 | 4.98 ± 0.1 d | 4.32 ± 0.03 c.d |
| Platanoide (M. alba.) | 69.0 | 3.14 ± 0.11 f | 2.57 ± 0.02 g |
| Sinuense (M. alba L.) | 67.5 | 1.44 ± 0.11 h,i,l | 1.04 ± 0.01 u,t |
| Nagazaki (M. alba L.) | 70.2 | 3.75 ± 0.11 e | 2.94 ± 0.02 f |
| End-May 2018 | End-July 2018 | |||
|---|---|---|---|---|
| M. alba Cultivars | Humidity (%) | Dry Samples (mg/g of Dry Matter) a | Humidity (%) | Dry Samples (mg/g of Dry Matter) a |
| Aobanezumi | 67.0 | 0.38 ± 0.02 l | 65.3 | 0.77 ± 0.02 h,i |
| Cattaneo female | 65.2 | 0.84 ± 0.02 b | 64.0 | 3.88 ± 0.05 c |
| Cattaneo male | 69.4 | 0.74 ± 0.03 c | 69.0 | 2.60 ± 0.03 f |
| Egyptienne | 62.2 | 0.42 ± 0.02 l | 59.2 | 0.74 ± 0.02 h,i |
| Florio | 52.0 | 0.36 ± 0.01 l | 50.3 | 0.88 ± 0.02 h |
| Giazzola | 68.2 | 0.53 ± 0.02 i | - | - |
| Kayriounezumigaeshi | 66.1 | 0.97 ± 0.03 a | 65.3 | 3.48 ± 0.05 d |
| Limoncina | 59.2 | 0.42 ± 0.02 l | 58.8 | 0.44 ± 0.02 l |
| Morettiana | 74.1 | 0.74 ± 0.04 c | 73.3 | 2.75 ± 0.09 e |
| Nervosa | 66.0 | 0.92 ± 0.03 a | 65.2 | 5.01 ± 0.08 b |
| Pendula | 70.2 | 0.99 ± 0.05 a | 68.9 | 5.48 ± 0.10 a |
| Pyramidalis | 69.3 | 0.63 ± 0.05 d | 68.4 | 2.20 ± 0.05 g |
| Spain Black Fruit | 74.4 | 0.97 ± 0.03 a | 74.0 | 2.13 ± 0.05 g |
| Ukraina | 63.8 | 0.53 ± 0.02 i | 62.1 | 0.65 ± 0.02 i |
| End-May 2017 | End-May 2018 | |||
|---|---|---|---|---|
| M. alba Cultivars | Humidity (%) | Dry Samples (mg/g of Dry Matter) a | Humidity (%) | Dry Samples (mg/g of Dry Matter) a |
| Aobanezumi | 69.4 | 0.44 ± 0.06 i | 67.0 | 0.38 ± 0.02 f |
| Cattaneo Female | 66.3 | 0.59 ± 0.03 h | 65.2 | 0.84 ± 0.02 b |
| Cattaneo Male | 70.5 | 1.50 ± 0.03 e | 69.4 | 0.74 ± 0.03 c |
| Egyptienne | 52.9 | 1.66 ± 0.08 d | 62.2 | 0.42 ± 0.02 f |
| Florio | 70.7 | 0.55 ± 0.02 h,i | 52.0 | 0.36 ± 0.01 f |
| Giazzola | 67.7 | 1.20 ± 0.05 f | 68.2 | 0.53 ± 0.02 e |
| Kayriounezumigaeshi | 68.1 | 1.86 ± 0.08 c | 66.1 | 0.97 ± 0.03 a |
| Limoncina | 64.2 | 0.93 ± 0.05 g | 59.2 | 0.42 ± 0.02 f |
| Morettiana | 72.5 | 1.26 ± 0.04 f | 74.1 | 0.74 ± 0.04 c |
| Nervosa | 74.4 | 1.62 ± 0.06 d,e | 66.0 | 0.92 ± 0.03 a |
| Pendula | 72.6 | 1.43 ± 0.04 e | 70.2 | 0.99 ± 0.05 a |
| Pyramidalis | 70.2 | 2.07 ± 0.08 b | 69.3 | 0.63 ± 0.05 d |
| Spain Black Fruit | 70.3 | 1.76 ± 0.07 c,d | 74.4 | 0.97 ± 0.03 a |
| Ukraina | 70.5 | 2.94 ± 0.06 a | 63.8 | 0.53 ± 0.02 e |
| Year | Average Radiation (kWh/m2/day) | Monthly Total Rainfall (mm) | Average Temperature (°C) |
|---|---|---|---|
| 2017 | 5.70 (20.52 MJ/m2/day) | 79 mm | 18.6 |
| 2018 | 5.16 (18.57 MJ/m2/day) | 81.8 mm | 19.4 |
| μg/mL | Intra—Day n = 3 | ||||||
|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | Mean | SD | RSD (%) | Recovery (%) | |
| 0.06 | 0.056 | 0.058 | 0.064 | 0.059 | 0.004 | 7.02 | 98.89 |
| 0.60 | 0.630 | 0.550 | 0.570 | 0.583 | 0.042 | 7.14 | 97.22 |
| 6.00 | 5.980 | 6.720 | 6.400 | 6.367 | 0.371 | 5.83 | 106.11 |
| Inter—Day n = 3 | |||||||
| 1 | 2 | 3 | mean | SD | RSD (%) | Recovery (%) | |
| 0.06 | 0.062 | 0.058 | 0.054 | 0.058 | 0.004 | 6.90 | 96.67 |
| 0.60 | 0.570 | 0.620 | 0.580 | 0.590 | 0.026 | 4.48 | 98.33 |
| 6.00 | 6.120 | 5.940 | 6.200 | 6.087 | 0.133 | 2.19 | 101.44 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marchetti, L.; Saviane, A.; Montà, A.d.; Paglia, G.; Pellati, F.; Benvenuti, S.; Bertelli, D.; Cappellozza, S. Determination of 1-Deoxynojirimycin (1-DNJ) in Leaves of Italian or Italy-Adapted Cultivars of Mulberry (Morus sp.pl.) by HPLC-MS. Plants 2021, 10, 1553. https://doi.org/10.3390/plants10081553
Marchetti L, Saviane A, Montà Ad, Paglia G, Pellati F, Benvenuti S, Bertelli D, Cappellozza S. Determination of 1-Deoxynojirimycin (1-DNJ) in Leaves of Italian or Italy-Adapted Cultivars of Mulberry (Morus sp.pl.) by HPLC-MS. Plants. 2021; 10(8):1553. https://doi.org/10.3390/plants10081553
Chicago/Turabian StyleMarchetti, Lucia, Alessio Saviane, Antonella dalla Montà, Graziella Paglia, Federica Pellati, Stefania Benvenuti, Davide Bertelli, and Silvia Cappellozza. 2021. "Determination of 1-Deoxynojirimycin (1-DNJ) in Leaves of Italian or Italy-Adapted Cultivars of Mulberry (Morus sp.pl.) by HPLC-MS" Plants 10, no. 8: 1553. https://doi.org/10.3390/plants10081553
APA StyleMarchetti, L., Saviane, A., Montà, A. d., Paglia, G., Pellati, F., Benvenuti, S., Bertelli, D., & Cappellozza, S. (2021). Determination of 1-Deoxynojirimycin (1-DNJ) in Leaves of Italian or Italy-Adapted Cultivars of Mulberry (Morus sp.pl.) by HPLC-MS. Plants, 10(8), 1553. https://doi.org/10.3390/plants10081553









