The Unique Substance, Lidocaine and Biological Activity of the Dioscorea Species for Potential Application as a Cancer Treatment, Natural Pesticide and Product
Abstract
:1. Introduction
2. Results
2.1. Phytochemicals Constituent
2.1.1. Gas Chromatography-Mass Spectrometry (GC-MS)
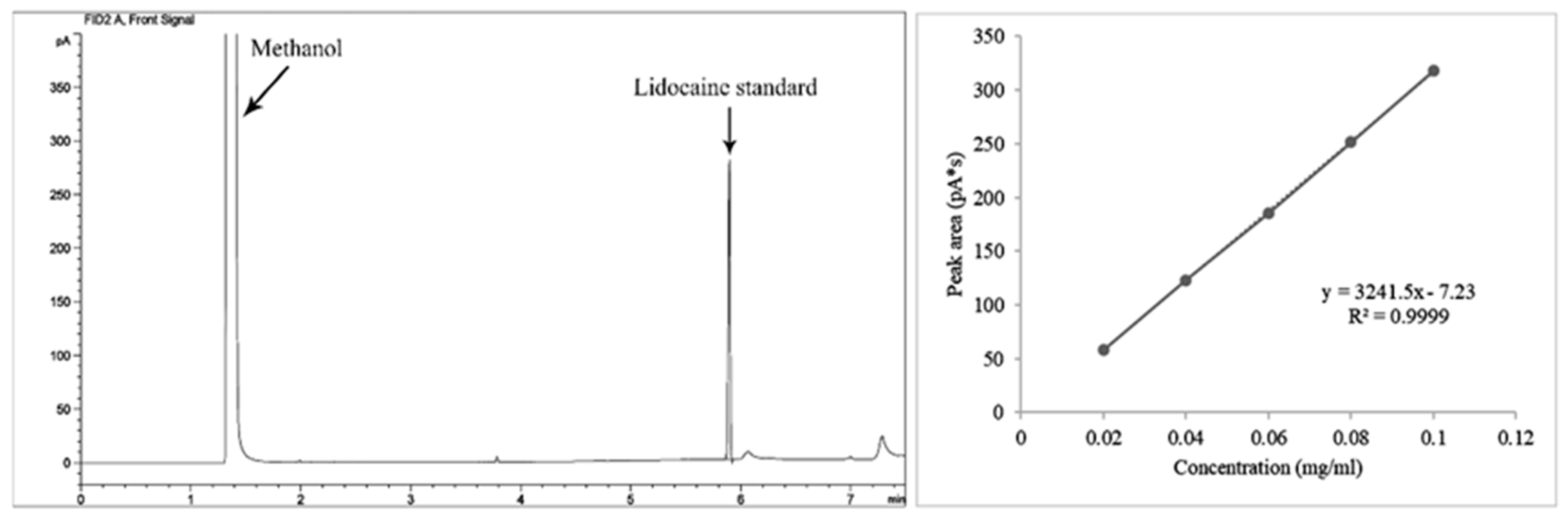
2.1.2. Gas Chromatography (GC) with the Lidocaine Standard
2.2. Toxicity
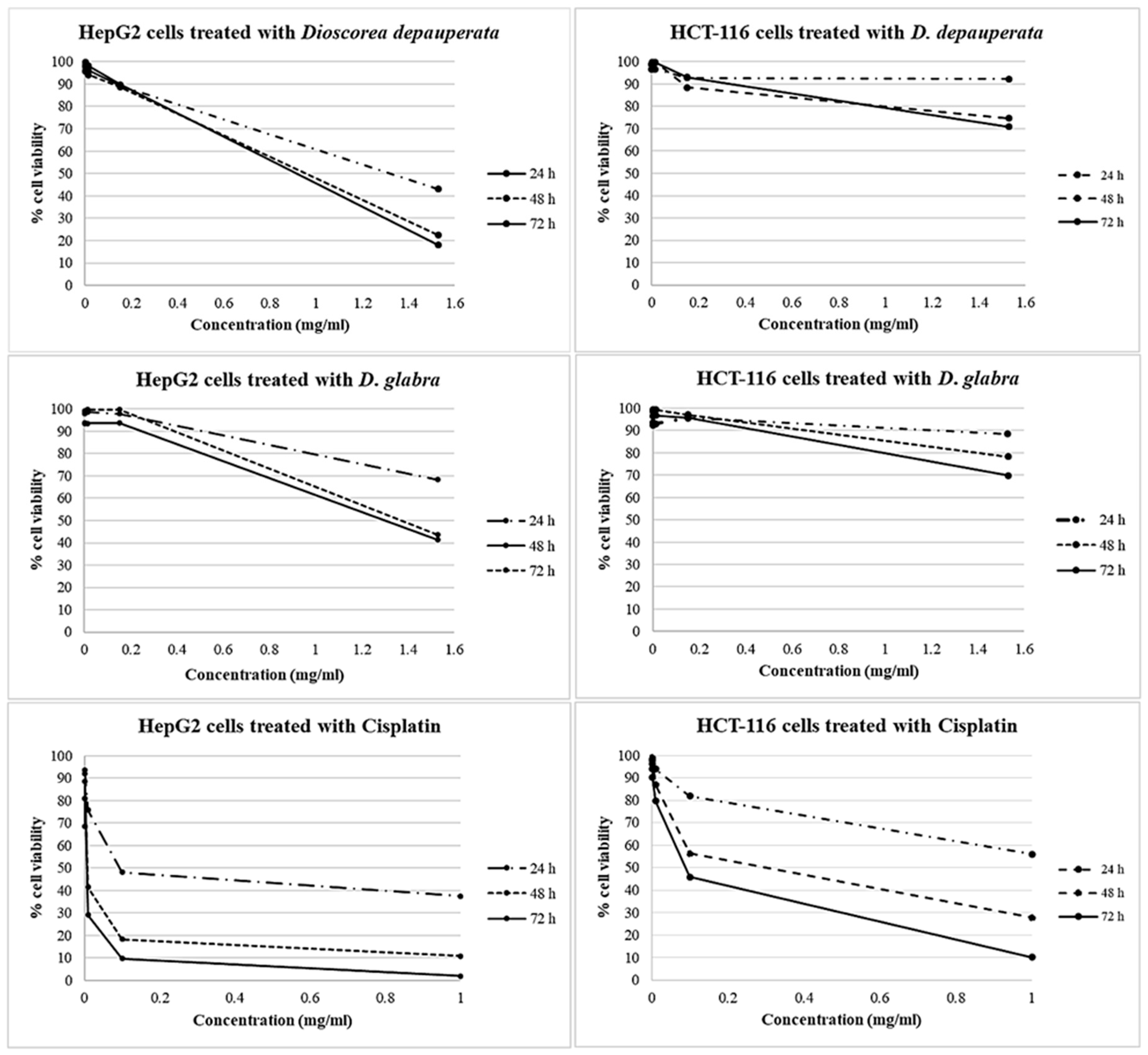
2.2.1. Cytotoxicity
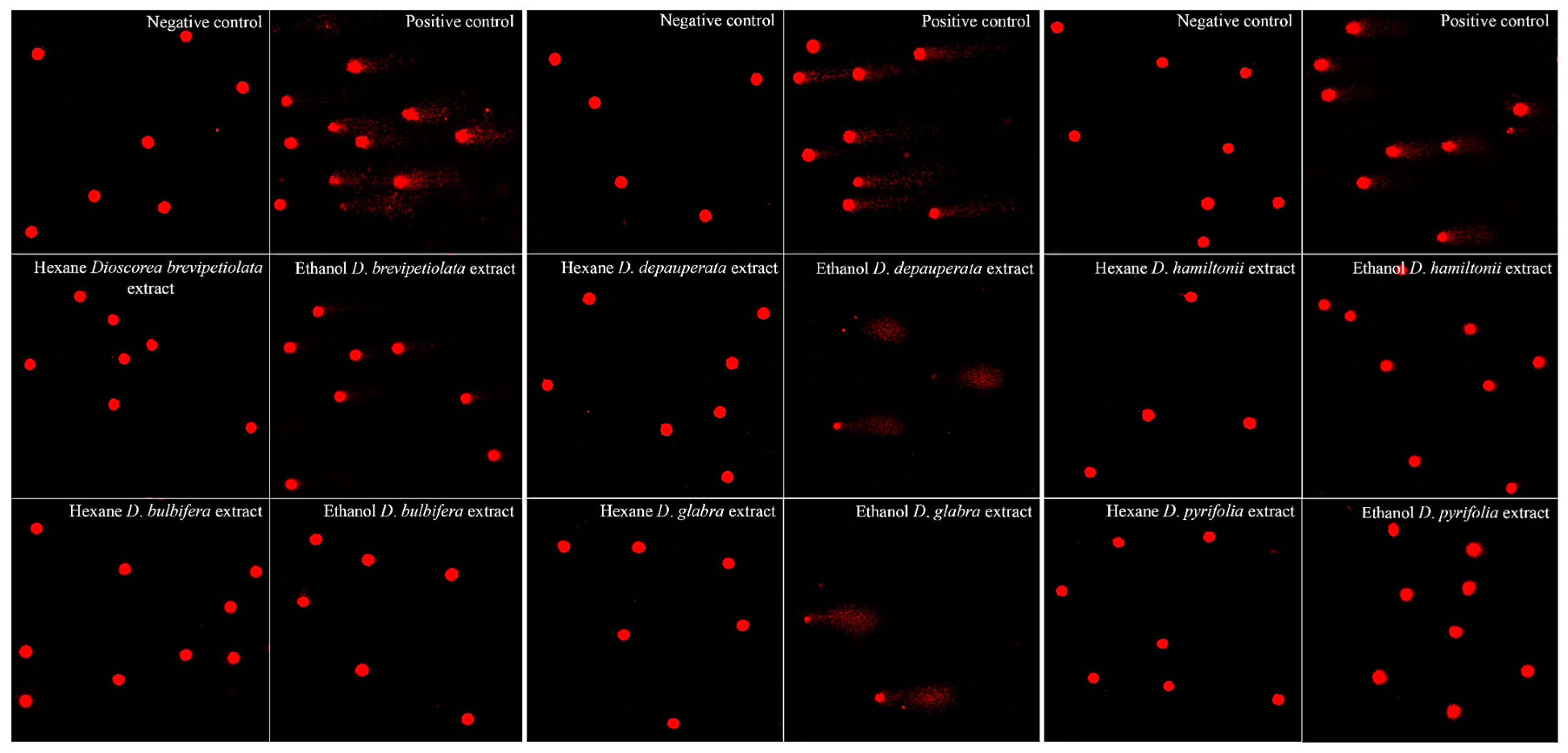
2.2.2. Genotoxicity
2.3. Biological Activity
2.4. Pesticidal Efficiency
3. Discussion
4. Materials and Methods
4.1. Chemicals and Cell Lines
4.2. Plant Materials and Extract Preparation
4.3. Gas Chromatography-Mass Spectrometry (GC-MS)
4.4. Lidocaine Detection by Gas-Chromatography Compared to the Lidocaine Standard
4.5. Cytotoxicity and Genotoxicity Testing via MTT and Comet Assays
- Stock Extract PreparationThe solvents of the filtrates (from plant extract preparations) were removed with a rotary evaporator (Rotavapor R-210, Buchi, Switzerland) at 800–1000 mbar, 15 °C, and 600 rpm for 2 h. Then, dimethyl sulfoxide (DMSO) was added to the extracts until being completely dissolved and maintained as stock extracts at −20 °C conducting for the cytotoxicity and genotoxicity experiments.
- Human Peripheral Blood Mononuclear Cells (PBMCs) PreparationPBMCs were isolated from sodium heparin anticoagulated venous blood from a blood bank using Ficoll-Paque Plus (GE Healthcare). Freshly isolated PBMCs with viability of at least 98% were used for the toxicity testing. The cells were suspended at a concentration of 1 × 106 cells/mL for MTT and 0.4–0.6 × 106 cells/mL for the comet assay in modified RPMI-1640 medium supplemented with 10% FBS, 1% antibiotic (streptomycin and penicillin).
- MTT AssayThe stock extract concentrations were serially 10-fold diluted with water, five times for the working concentrations. The prepared cells were seeded in 96-well plates, 125 μL per well, and 12.5 μL of the extract working concentrations were added to the corresponding wells, incubated for 24 h for PBMCs and 24, 48 and 72 h for cancer cell lines in a humidified CO2 incubator at 37 °C and 5% CO2. Corresponding DMSO concentrations were similarly prepared as vehicle controls, untreated cells and hydrogen peroxide-treated cells were the negative and positive controls, respectively. When the time was over, the plates were centrifuged at 1500 rpm for 10 min and the medium was removed, the MTT (Sigma, USA) was added to a final concentration of 0.5 mg/mL, the plates were wrapped with aluminum foil and incubated for 4 h at 37 °C. The formazan crystals were solubilized by adding 100 μL DMSO to each well, and the plates were left in the dark for 2–4 h. The absorbance was read at 570 nm with a microtiter plate spectrophotometer (Multifunction microplate reader; Varioskan Flash, Thermo fisher, Waltham, MA, USA). Wells containing medium and MTT without cells were used as blanks. Each concentration treatment was performed in triplicate. All values were expressed as the mean ± SD. Cellular reduction of MTT formed a violet crystal formazan through mitochondrial succinate dehydrogenase activity of the viable cells, and the violet crystal formazan was quantified following the methods of Freshney [13]. Percentage of cell viability was calculated using the equation cell viability (%) = average viable treated cells/average viable negative control cells × 100), to reveal the cytotoxicity of the plant extracts. Doses inducing 50% inhibition of cell viability (IC50 value) were determined by plotting a graph of the extract concentration against the cell viability. The IC50 value was used for the LD50 calculation Walum [14] to infer hazardous levels, according to the World Health Organization [15].
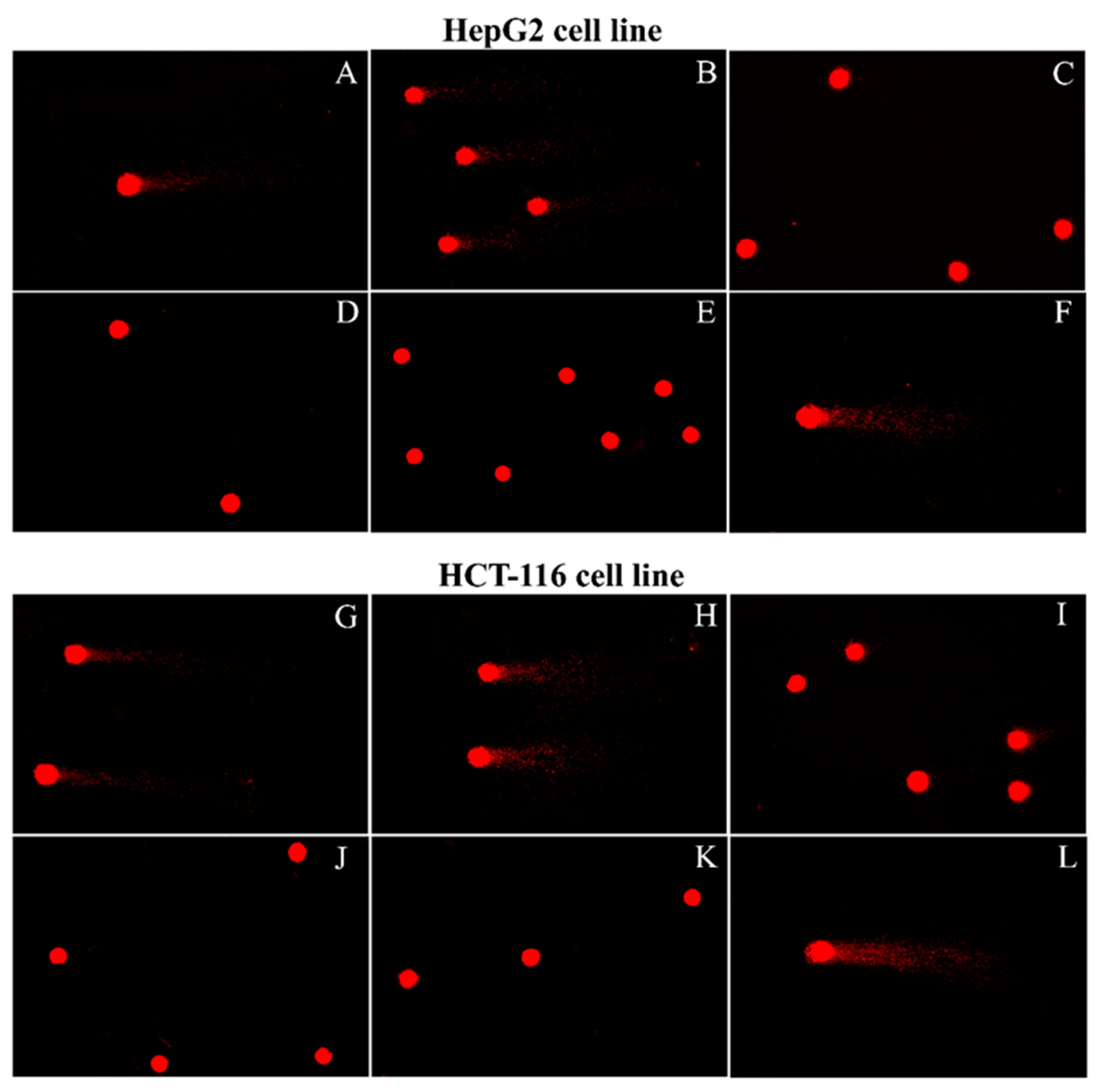
- Comet AssayThe concentration at IC50 value or the maximum-treated concentration, in the case of no IC50 value, was used in the comet assay to assess the genotoxicity of plant extracts, according to Singh et al. [16]. Shortly, 500 µL of cells in media was added with 50 µL extracts in a 1.5 mL microtube, incubated for 24 h for PBMCs and 24, 48 and 72 h for cancer cell lines in a humidified CO2 incubator at 37 °C and 5% CO2, then the DNA was checked by electrophoresis. The electrophoresis buffer consisted of 0.3 M NaOH and 1 mM EDTA (pH = 10). The power was supplied at a constant of 3.4 v/cm with an adjustment to 300 mA, for 25 min. To quantify the level of DNA damage, the extent of DNA migration was defined using the “Olive Tail Moment” (OTM), which is the relative amount of DNA in the tail of the comet multiplied by the median migration distance. The comets were observed at 200× magnifications and images were obtained using an image analysis system (Isis) attached to a fluorescence microscope (Nikon, Tokyo, Japan), equipped with a 560 nm excitation filter, 590 nm barrier filter, and a CCD video camera PCO (Germany). At least 150 cells (50 cells for each of triplicate slides) were examined for each experiment. The CASP software (Wroclaw, Poland) was used to analyze the OTM. The negative control was untreated cells, and the positive control was UV-treated cells. All experiments were in triplicate. The triplicate cultures were scored for the experiment. All values were expressed as the median ± S.D. The nonparametric Mann–Whitney U test was used for statistical analysis of the comet assay results; statistical significance was set at p < 0.05.
4.6. Biological Activity Testing of the Plant Extracts on HepG2 and HCT-116 Compared to Cisplatin
4.7. Biological Activity Testing for Pesticidal Efficiency
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| PBMCs | human peripheral blood mononuclear cells |
| MTT | 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| GC-MS | gas chromatography-mass-spectrometry |
| HepG2 | hepatocellular carcinoma cell line |
| HCT-116 | colorectal carcinoma cell line |
References
- Wilkin, P.; Thapyai, C. Dioscoreaceae. In Flora of Thailand Vol. 10 Part 1; Forest Herbarium, Royal Forest Department: Bangkok, Thailand, 2009; pp. 1–140. ISBN 978-974-286-614-3. [Google Scholar]
- Maneenoon, K. Medicinal Plants of the Genus Dioscorea L. Used in Traditional Thai Medicine Prescriptions. KKU Sci. J. 2013, 41, 797–807. [Google Scholar]
- Jesus, M.; Martins, A.P.J.; Gallardo, E.; Silvestre, S. Diosgenin: Recent Highlights on Pharmacology and Analytical Methodology. J. Anal. Methods Chem. 2016, 2016, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Xu, J.; Luo, L.; Hu, H.; Meng, X.; Li, X.; Chen, S. Predicting the Potential Global Distribution of Diosgenin-Contained Dioscorea Species. Chin. Med. 2018, 13, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Hazrati, K.; Sapuan, S.; Zuhri, M.; Jumaidin, R. Extraction and Characterization of Potential Biodegradable Materials Based on Dioscorea hispida Tubers. Polymers 2021, 13, 584. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Das, G.; Shin, H.-S.; Patra, J.K. Dioscorea spp. (A Wild Edible Tuber): A Study on Its Ethnopharmacological Potential and Traditional Use by the Local People of Similipal Biosphere Reserve, India. Front. Pharmacol. 2017, 8, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ikiriza, H.; Ogwang, P.E.; Peter, E.L.; Hedmon, O.; Tolo, C.U.; Abubaker, M.; Abdalla, A.A.M. Dioscorea bulbifera, a Highly Threatened African Medicinal Plant, A Review. Cogent Biol. 2019, 5. [Google Scholar] [CrossRef]
- Padhan, B.; Panda, D. Potential of Neglected and Underutilized Yams (Dioscorea spp.) for Improving Nutritional Security and Health Benefits. Front. Pharmacol. 2020, 11, 496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gordh, T.; Torsten, E.; Gordh, M.D.; Lindqvist, K. Lidocaine: The Origin of a Modern Local Anesthetic. J. Am. Soc. Anesthesiol. 2010, 113, 1433–1437. [Google Scholar] [CrossRef] [PubMed]
- Golzari, S.; Soleimanpour, H.; Mahmoodpoor, A.; Safari, S.; Ala, A. Lidocaine and Pain Management in the Emergency Department: A Review Article. Anesthesiol. Pain Med. 2014, 3, e15444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panyakhamlerd, Y.; Sirivararom, P.; Chaisen, N.; Turnprakiat, S. A Comparative Study of the Effectiveness of 10% Lidocaine Spray at Oropharyngolarynx Plus Tracheal Tube Cuff Versus Oropharyngolarynx Alone in Attenuation of the Hemodynamic Response During Direct Laryngoscopy and Intubation in Controlled Hypertensive Patients. Thai. J. Anesthesiol. 2013, 39, 292–301. [Google Scholar]
- Prapanbandit, N. Trigger Point Injection with 1% Versus 2% Lidocaine for Treatment of Myofascial Pain Syndrome at Neck and Upper Back: A Randomized Controlled and Double-blinded Clinical Trial. ASEAN J. Rehabil. Med. 2019, 29, 99–106. [Google Scholar]
- Freshney, R.I. Culture of Animal Cells: A Manual of Basic Technique and Specialized Applications, 6th ed.; John Wiley & Sons: Hoboken, NJ, USA, 2010; pp. 1–796. ISBN 978-047-052-812-9. [Google Scholar]
- Walum, E. Acute Oral Toxicity. Environ. Health Perspect. 1998, 106, 497. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification 2009. Available online: https://apps.who.int/iris/handle/10665/44271 (accessed on 21 January 2021).
- Singh, N.P.; McCoy, M.T.; Tice, R.R.; Schneider, E.L. A Simple Technique for Quantitation of Low Levels of DNA Damage in Individual Cells. Exp. Cell Res. 1988, 175, 184–191. [Google Scholar] [CrossRef] [Green Version]
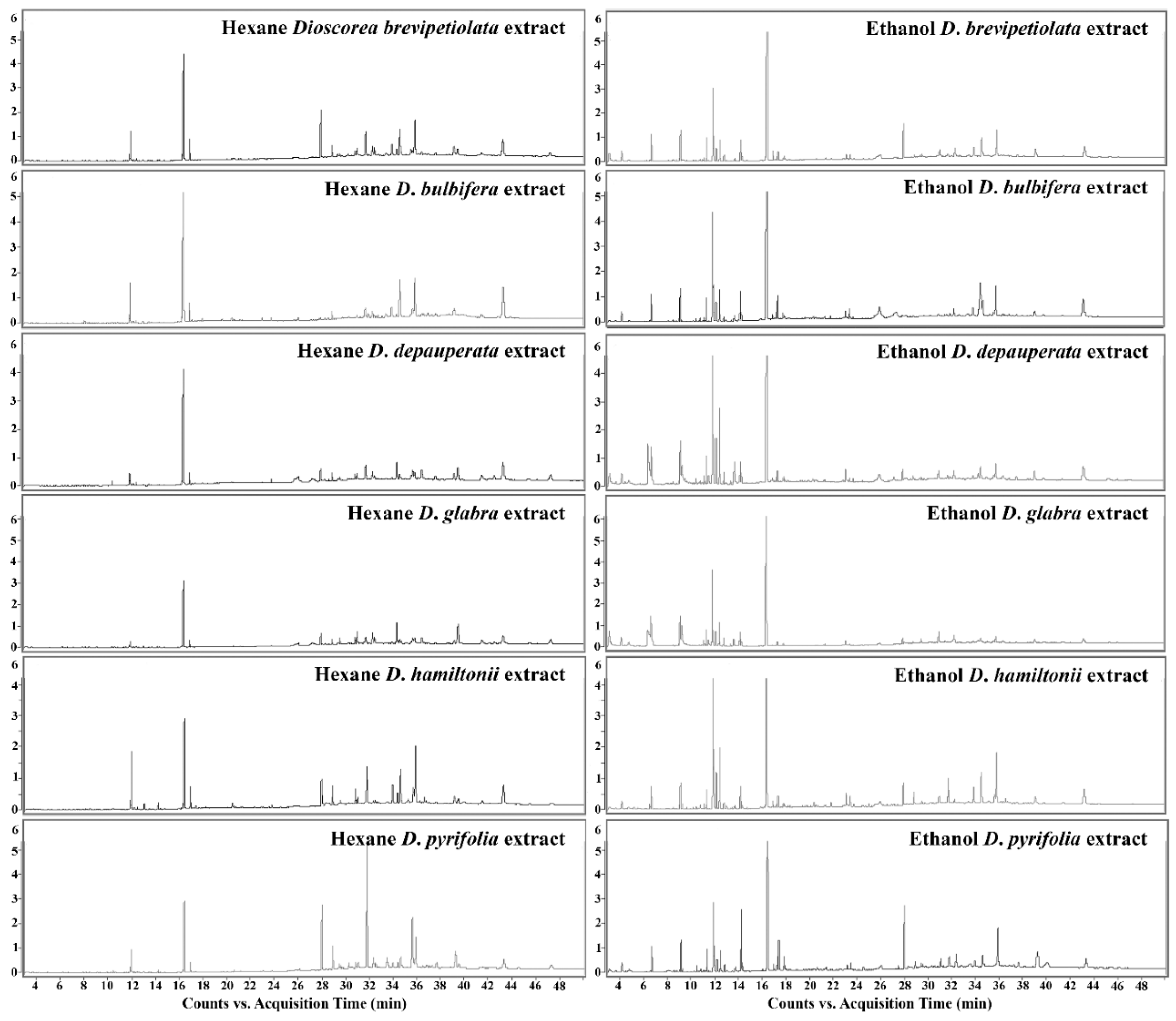





| Compound | Formula | Relative Content (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| D. brevipetiolata | D. bulbifera | D. depauperata | D. glabra | D. hamiltonii | D. pyrifolia | ||||||||
| H | E | H | E | H | E | H | E | H | E | H | E | ||
| Phytol | C20H40O | 24.15 | 56.90 | 47.47 | 48.23 | 46.06 | 50.22 | 31.81 | 47.56 | 16.23 | 41.95 | 10.78 | 44.35 |
| Phytol, acetate | C22H42O2 | 0.97 | - | - | - | 4.62 | - | 1.31 | 10.48 | - | - | 0.63 | - |
| γ-Sitosterol | C28H50O | 15.76 | 5.83 | 15.08 | 5.25 | - | 2.52 | 4.26 | 1.68 | 21.36 | 9.23 | 9.03 | 7.54 |
| Stigmasterol | C29H48O | 9.97 | 3.69 | 12.72 | 10.44 | 2.96 | 1.53 | 2.40 | 0.81 | 11.06 | 4.81 | 2.71 | 2.18 |
| Squalene | C30H50 | 10.80 | 3.97 | - | - | 4.51 | 0.87 | 4.87 | 0.86 | 4.89 | 2.04 | 10.20 | 6.39 |
| Phenol, 2-propyl- | C11H17NO3 | - | - | - | - | - | 8.35 | - | 10.17 | - | - | - | - |
| Vitamin E | C29H50O2 | 6.09 | 2.31 | 4.21 | 1.51 | 5.76 | - | 3.79 | 1.45 | 4.69 | 2.23 | - | 2.06 |
| Triacontanoic acid, methyl ester | C31H62O2 | - | - | - | - | 7.82 | - | - | - | - | - | - | - |
| dl-α-Tocopherol | C29H50O2 | 3.07 | 1.17 | 1.75 | 0.85 | 3.81 | 0.95 | 6.61 | 1.39 | - | - | 2.04 | 1.68 |
| 2-Pentadecanone, 6,10,14-trimethyl- | C18H36O | 4.27 | 2.15 | 5.46 | 2.75 | 3.16 | 1.32 | 2.00 | 1.10 | 6.75 | 4.19 | 2.18 | 1.91 |
| Hexadecanoic acid, ethyl ester | C18H36O2 | - | 2.19 | - | 2.71 | - | 1.73 | - | 2.07 | 1.23 | 1.99 | 0.43 | 6.14 |
| γ-Tocopherol | C28H48O2 | 1.60 | 0.81 | - | - | 2.39 | 0.72 | 5.84 | 1.65 | 1.27 | 0.73 | 0.99 | 0.91 |
| Campesterol | C28H48O | 4.46 | 1.82 | 3.82 | 1.34 | - | - | - | - | 6.18 | 2.67 | 1.39 | 1.22 |
| 1,3-Benzenediol, 4-propyl- | C9H12O2 | - | - | - | - | - | - | - | 4.90 | - | - | - | - |
| δ-Tocopherol | C27H46O2 | - | - | - | - | - | - | 2.93 | 0.63 | - | - | - | - |
| n-Hexadecanoic acid | C16H32O2 | - | - | - | 0.42 | - | 2.60 | - | 1.25 | - | - | - | - |
| Dodecane | C12H26 | - | 1.67 | - | 1.57 | - | 1.47 | - | 2.23 | - | - | - | 1.58 |
| Glycerol β-palmitate | C19H38O4 | - | 0.56 | - | 0.71 | - | 0.95 | - | 0.61 | - | 1.19 | - | - |
| Octadecanoic acid, ethyl ester | C20H40O2 | - | - | - | 0.53 | - | - | - | - | - | - | - | 1.35 |
| Tetradecane | C14H30 | - | 4.45 | - | - | - | 2.07 | - | 2.80 | - | - | - | 2.13 |
| Benzyldiethyl-(2,6-xylylcarbamoylmethyl)- ammonium benzoate | C28H34N2O3 | - | 0.56 | - | 0.93 | - | - | - | - | - | 0.93 | - | 0.62 |
| Lidocaine | C14H22N2O | - | - | - | - | - | 0.81 | - | 1.03 | - | - | - | - |
| Diisooctyl phthalate | C24H38O4 | - | - | - | - | 1.22 | - | - | - | - | - | - | - |
| 2-Hydroxy-5-methylisophthalaldehyde | C9H8O3 | - | - | - | - | - | - | - | 0.80 | - | - | - | - |
| Total of identified compounds | - | 81.14 | 88.08 | 90.51 | 77.24 | 82.31 | 76.11 | 65.82 | 93.47 | 73.66 | 71.96 | 40.38 | 80.06 |
| Unknown | - | 18.86 | 11.92 | 9.49 | 22.76 | 17.69 | 23.89 | 34.18 | 6.53 | 26.34 | 28.04 | 59.62 | 19.94 |
| Plant Extract | Retention Time (min) | Peak Area (pA*s) | Lidocaine | |||
|---|---|---|---|---|---|---|
| Concentration | Amount | |||||
| μg/mL | mg/mL | mg/g | mg/100 g | |||
| Dioscorea alata | 5.87 | 47.55 | 16.90 | 16.90 × 10−3 | 5.91 × 10−2 | 5.92 |
| D. arachidna | 5.87 | 5.18 | 3.83 | 3.83 × 10−3 | 1.05 × 10−2 | 1.05 |
| D. brevipetiolata | 5.88 | 16.18 | 7.83 | 7.83 × 10−3 | 2.03 × 10−2 | 2.03 |
| D. bulbifera | 5.87 | 14.58 | 6.73 | 6.73 × 10−3 | 1.85 × 10−2 | 1.85 |
| D. decipiens | 5.87 | 21.69 | 8.92 | 8.92 × 10−3 | 2.68 × 10−2 | 2.68 |
| D. depauperata | 5.88 | 29.73 | 11.40 | 11.40 × 10−3 | 3.71 × 10−2 | 3.71 |
| D. esculenta | 5.87 | 20.59 | 8.58 | 8.58 × 10−3 | 2.79 × 10−2 | 2.79 |
| D. glabra | 5.88 | 15.59 | 7.04 | 7.04 × 10−3 | 2.46 × 10−2 | 2.46 |
| D. hamiltonii | 5.88 | 68.03 | 23.22 | 23.22 × 10−3 | 8.13 × 10−2 | 8.13 |
| D. hispida | 5.88 | 14.75 | 6.78 | 6.78 × 10−3 | 2.03 × 10−2 | 2.03 |
| D. pentaphylla | 5.88 | 11.94 | 5.91 | 5.91 × 10−3 | 1.92 × 10−2 | 1.92 |
| Plant Extract | Solvent | Maximum Concentration (mg/mL) | Working Concentration (mg/mL) | Cell Viability (%) ± S.D. |
|---|---|---|---|---|
| D. brevipetiolata | hexane | 1.81 | 0.18–0.18 × 10−4 | 95.09 ± 0.04–98.37 ± 0.03 |
| ethanol | 6.15 | 0.61–0.61 × 10−4 | 60.48 ± 0.07–98.21 ± 0.04 | |
| D. bulbifera | hexane | 1.46 | 0.14–0.14 × 10−4 | 84.62 ± 0.13–92.18 ± 0.16 |
| ethanol | 4.00 | 0.40–0.40 × 10−4 | 84.01 ± 0.10–91.65 ± 0.14 | |
| D. depauperata | hexane | 3.53 | 0.35–0.35 × 10−4 | 89.87 ± 0.07–96.40 ± 0.10 |
| ethanol | 14.21 | 1.42–1.42 × 10−4 | 79.86 ± 0.10–97.23 ± 0.08 | |
| D. glabra | hexane | 2.00 | 0.20–0.20 × 10−4 | 94.03 ± 0.07–99.49 ± 0.14 |
| ethanol | 15.30 | 1.53–1.53 × 10−4 | 76.52 ± 0.09–97.30 ± 0.10 | |
| D. hamiltonii | hexane | 0.73 | 0.07–0.07 × 10−4 | 91.03 ± 0.06–96.45 ± 0.12 |
| ethanol | 5.00 | 0.50–0.50 × 10−4 | 89.90 ± 0.09–98.93 ± 0.09 | |
| D. pyrifolia | hexane | 1.66 | 0.16–0.16 × 10−4 | 90.36 ± 0.10–95.02 ± 0.12 |
| ethanol | 5.00 | 0.50–0.50 × 10−4 | 83.77 ± 0.08–96.04 ± 0.13 |
| Plant | Solvent | Median ± S.D. of Negative Control | Median ± S.D. of Treated Cell | p Value |
|---|---|---|---|---|
| D. brevipetiolata | hexane | 0.17 ± 0.02 × 10−2 | 0.49 ± 0.01 × 10−2 | >0.05 |
| ethanol | 131.83 ± 0.19 × 10−2 | <0.05 | ||
| D. bulbifera | hexane | 0.18 ± 0.07 × 10−2 | >0.05 | |
| ethanol | 0.55 ± 0.02 × 10−2 | >0.05 | ||
| D. depauperata | hexane | 0.14 ± 0.02 × 10−2 | 0.14 ± 0.02 × 10−2 | >0.05 |
| ethanol | N/A * | N/A * | ||
| D. glabra | hexane | 0.14 ± 0.01 × 10−2 | >0.05 | |
| ethanol | N/A * | N/A * | ||
| D. hamiltonii | hexane | 0.07 ± 0.02 × 10−2 | 0.07 ± 0.01 × 10−2 | >0.05 |
| ethanol | 69.07 ± 0.18 × 10−2 | <0.05 | ||
| D. pyrifolia | hexane | 0.07 ± 0.08 × 10−2 | >0.05 | |
| ethanol | 0.17 ± 0.02 × 10−2 | <0.05 |
| Sample | Time of Treated (Hours) | Working Concentration (mg/mL) | Cell Viability (% ± S.D.) | IC50 Value (mg/mL)/Time (Hours) | ||
|---|---|---|---|---|---|---|
| HepG2 Cell Line | HCT-116 Cell Line | HepG2 Cell Line | HCT-116 Cell Line | |||
| D. depauperata (Ethanol extract) | 24 | 1.53 × 10−4–1.53 | 95.91–42.92 ± 0.07–0.07 | 96.74–92.36 ± 0.08–0.06 | 1.32/24 | - |
| 48 | 99.70–22.36 ± 0.06–0.06 | 98.97–74.60 ± 0.08–0.10 | ||||
| 72 | 97.97–18.01 ± 0.05–0.04 | 98.77–70.88 ± 0.07–0.08 | ||||
| D. glabra (Ethanol extract) | 24 | 1.53 × 10−4–1.53 | 98.38–68.27 ± 0.07–0.01 | 92.51–88.47 ± 0.08–0.07 | 1.30/48 | - |
| 48 | 93.93–41.35 ± 0.07–0.08 | 96.56–78.28 ± 0.06–0.06 | ||||
| 72 | 98.03–43.58 ± 0.06–0.06 | 98.42–69.79 ± 0.05–0.09 | ||||
| Cisplatin | 24 | 1.00 × 10−4–1.00 | 96.32–36.75 ± 0.10–0.06 | 97.81–55.92 ± 0.07–0.04 | 0.09/24 | 0.29/48 |
| 48 | 84.07–10.20 ± 0.08–0.01 | 98.96–27.67 ± 0.08–0.02 | ||||
| 72 | 81.81–2.17 ± 0.07–0.01 | 93.89–10.05 ± 0.07–0.02 | ||||
| Sample | Treated Cell Type | Concentration of Samples (mg/mL) | Time of Treated (Hours) | OTM (Median ± S.D.) | p Value of Treated Cells | |
|---|---|---|---|---|---|---|
| Negative Control | Treated Cell | |||||
| D. depauperata | HepG2 | 1.32 | 24 | 370.00 ± 3.60 × 10−4 | 4.021 ± 1.57 | <0.01 |
| HCT-116 | 1.53 | 72 | 6.53 ± 0.22 × 10−4 | 9.050 ± 2.55 | <0.01 | |
| D. glabra | HepG2 | 1.30 | 48 | 230.00 ± 3.20 × 10−4 | 12.743 ± 2.39 | <0.01 |
| HCT-116 | 1.53 | 72 | 6.53 ± 0.22 × 10−4 | 11.942 ± 3.05 | <0.01 | |
| cisplatin | HepG2 | 0.09 | 24 | 370.00 ± 3.60 × 10−4 | 0.703 ± 0.19 | <0.01 |
| HCT-116 | 0.29 | 48 | 6.28 ± 0.54 × 10−4 | 0.447 ± 0.21 | <0.01 | |
| Experiment | Number of Destroyed Brassica chinensis Individuals in a Pot | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pot 1 | Pot 2 | Pot 3 | Pot 4 | Pot 5 | Pot 6 | Pot 7 | Pot 8 | Pot 9 | Pot 10 | Total | |
| Control A | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 3 | 30 |
| Control B | 1 | 3 | 3 | 3 | 3 | 3 | 3 | 2 | 3 | 2 | 26 |
| Sample C | 1 | 1 | 0 | 1 | 1 | 2 | 1 | 1 | 1 | 1 | 10 |
| Sample D | 1 | 0 | 0 | 1 | 1 | 0 | 0 | 0 | 0 | 1 | 4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wonok, W.; Chaveerach, A.; Siripiyasing, P.; Sudmoon, R.; Tanee, T. The Unique Substance, Lidocaine and Biological Activity of the Dioscorea Species for Potential Application as a Cancer Treatment, Natural Pesticide and Product. Plants 2021, 10, 1551. https://doi.org/10.3390/plants10081551
Wonok W, Chaveerach A, Siripiyasing P, Sudmoon R, Tanee T. The Unique Substance, Lidocaine and Biological Activity of the Dioscorea Species for Potential Application as a Cancer Treatment, Natural Pesticide and Product. Plants. 2021; 10(8):1551. https://doi.org/10.3390/plants10081551
Chicago/Turabian StyleWonok, Warin, Arunrat Chaveerach, Pornnarong Siripiyasing, Runglawan Sudmoon, and Tawatchai Tanee. 2021. "The Unique Substance, Lidocaine and Biological Activity of the Dioscorea Species for Potential Application as a Cancer Treatment, Natural Pesticide and Product" Plants 10, no. 8: 1551. https://doi.org/10.3390/plants10081551
APA StyleWonok, W., Chaveerach, A., Siripiyasing, P., Sudmoon, R., & Tanee, T. (2021). The Unique Substance, Lidocaine and Biological Activity of the Dioscorea Species for Potential Application as a Cancer Treatment, Natural Pesticide and Product. Plants, 10(8), 1551. https://doi.org/10.3390/plants10081551








