Anti-Inflammatory Activity of a Medicinal Herb Extract Mixture, HM-V, on an Animal Model of DNCB-Induced Chronic Skin Inflammation
Abstract
:1. Introduction
2. Results
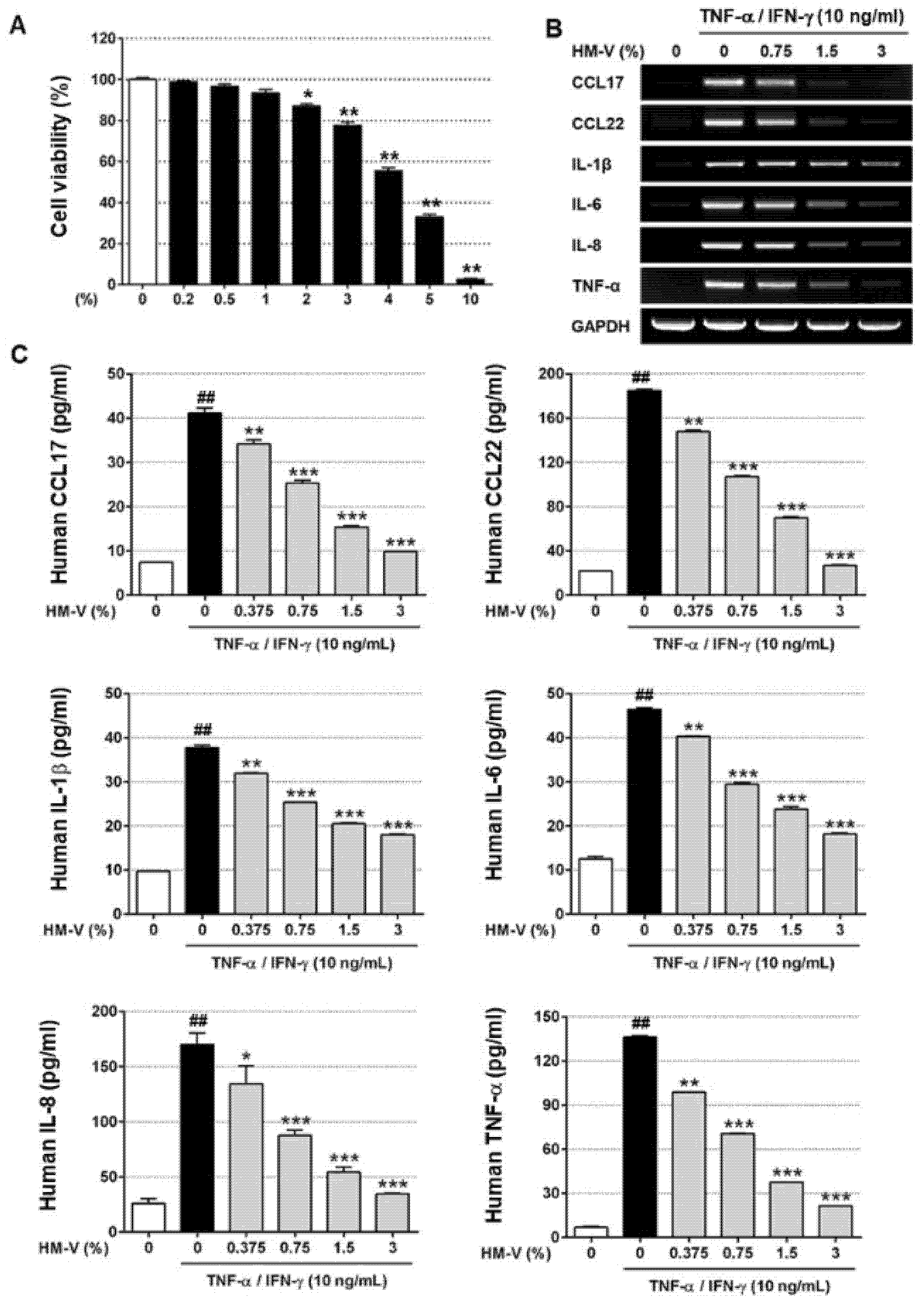
2.1. HM-V Inhibited TNF-α/IFN-γ-Induced Expression of Cytokines and Chemokines in HaCaTs
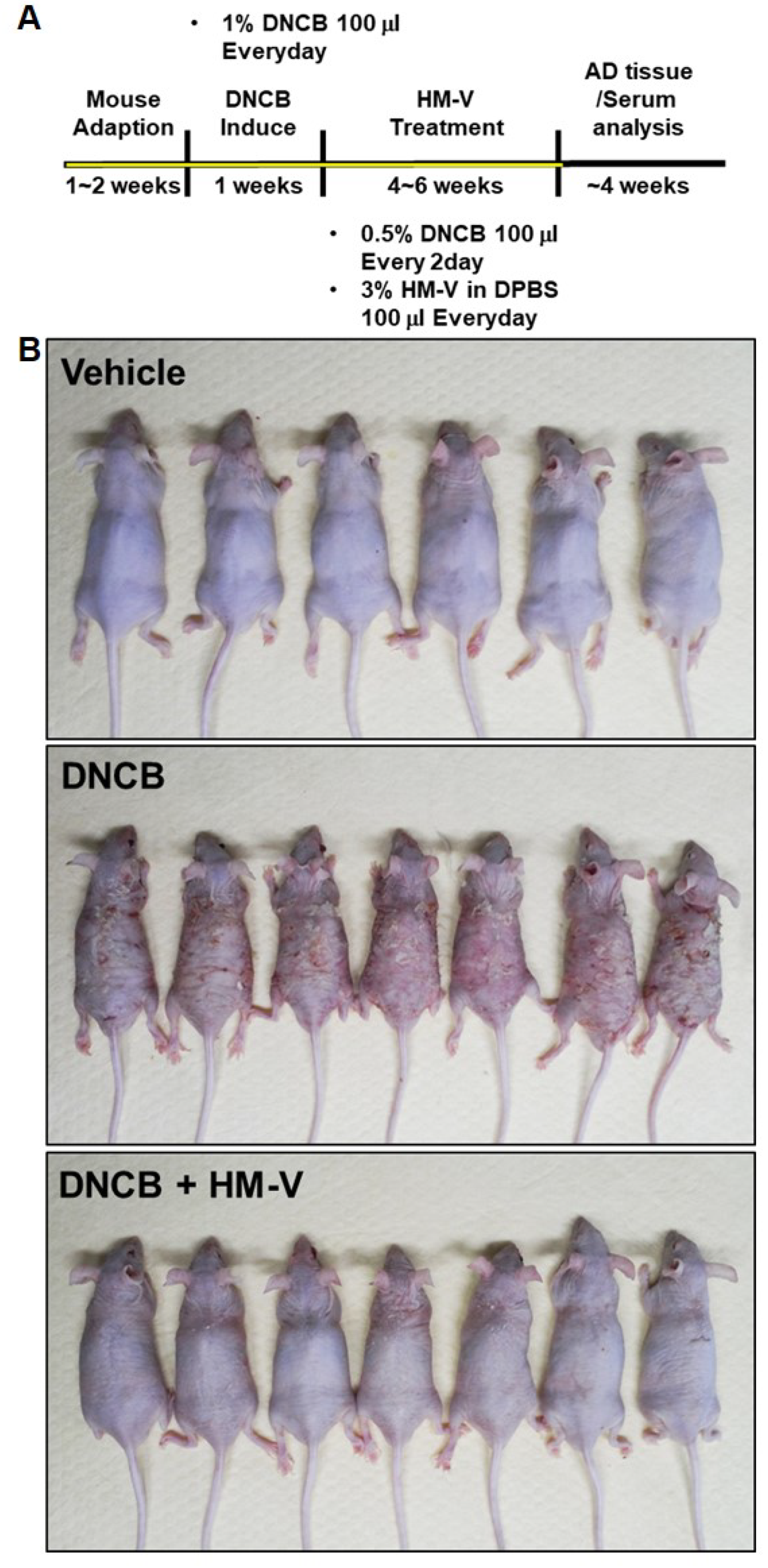
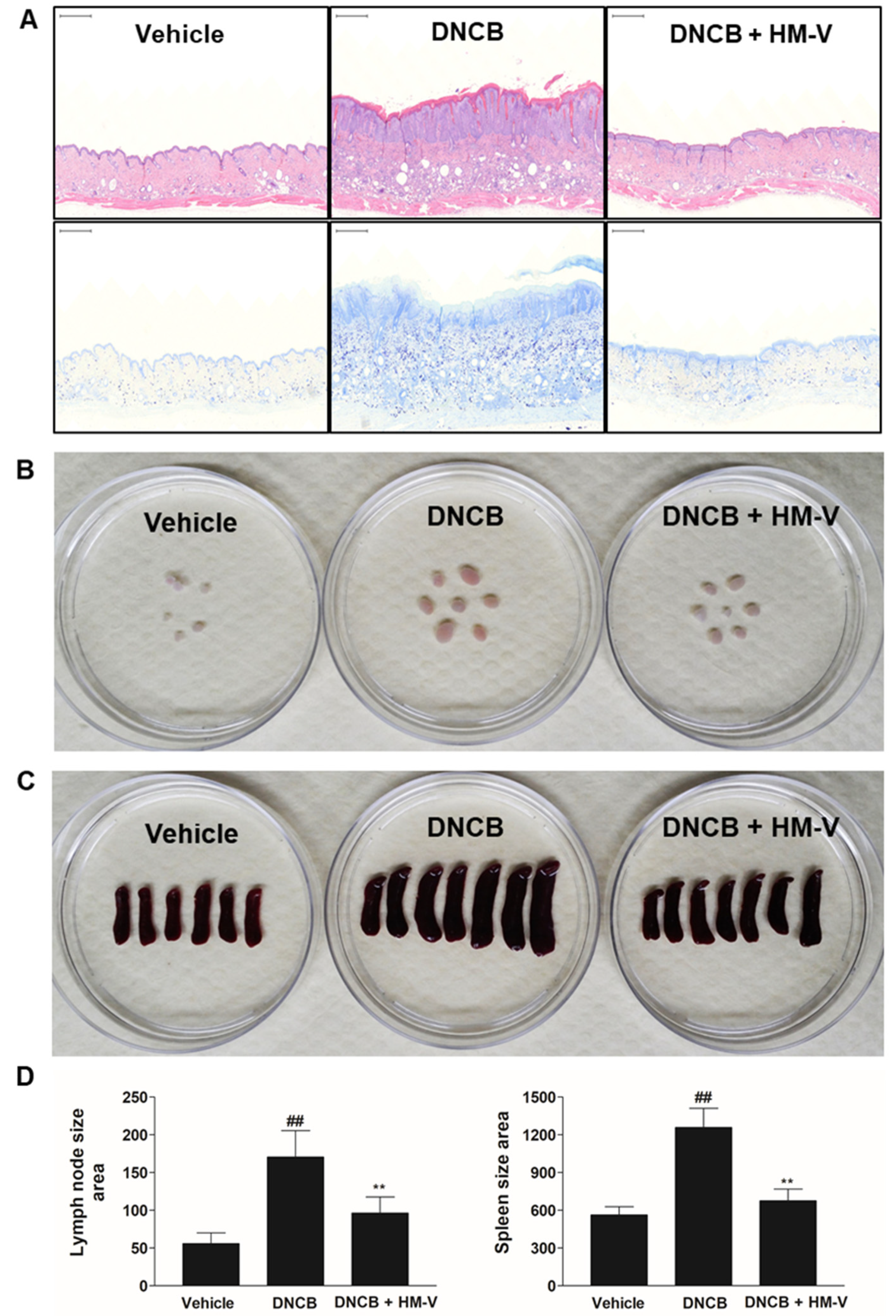
2.2. HM-V Attenuated DNCB-Induced Skin Inflammation
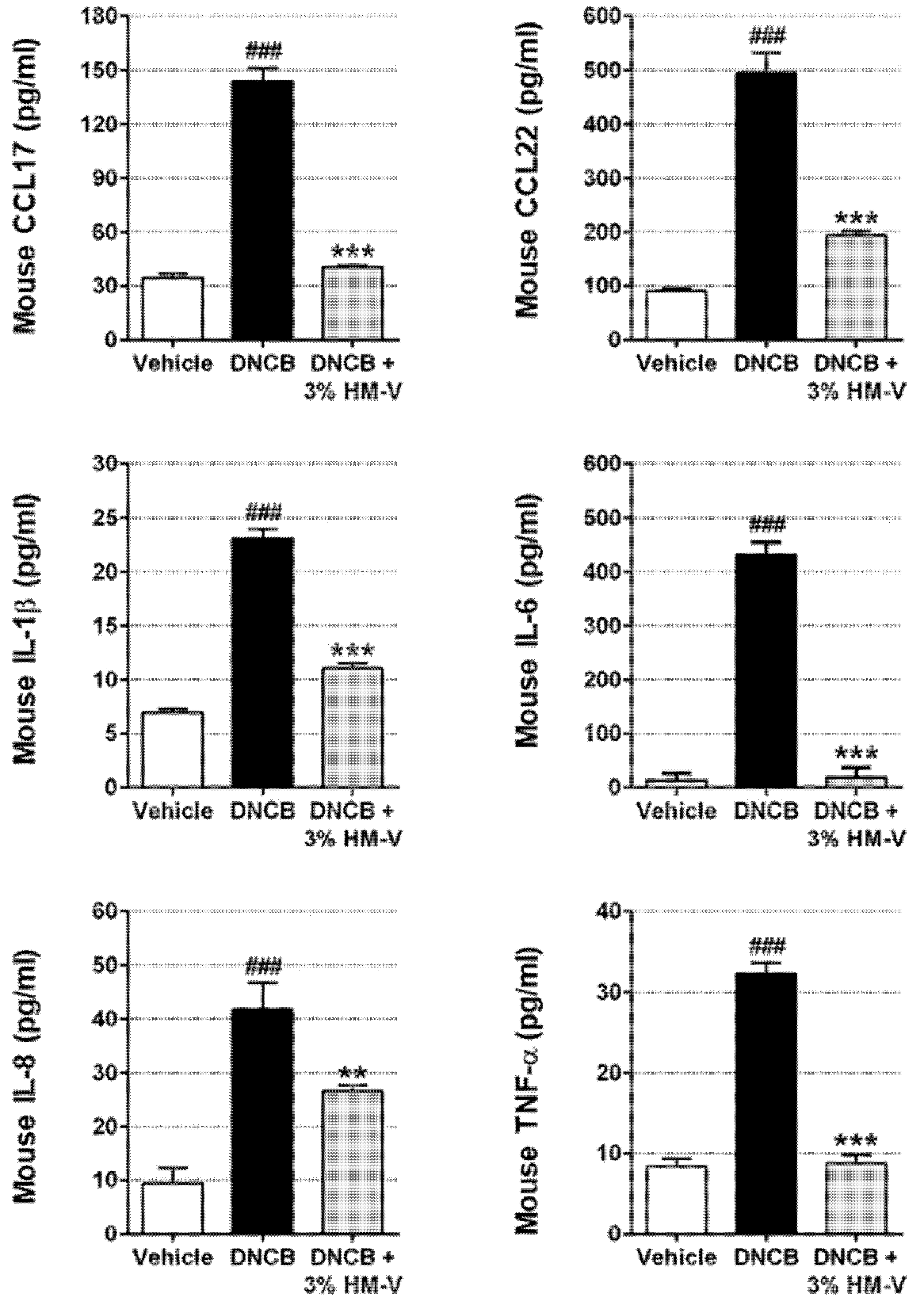
2.3. HM-V Reduced Serum Levels of Pro-Inflammatory Cytokines and Chemokines
3. Discussion
4. Materials and Methods
4.1. Medicinal Herb Extract Formulation
4.2. Cell Culture and Viability Assay
4.3. Animals and Maintenance
4.4. RT-PCR
4.5. Enzyme-Linked Immunosorbent Assay (ELISA)
4.6. Histological Analysis
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kim, J.E.; Kim, J.S.; Cho, D.H.; Park, H.J. Molecular Mechanisms of Cutaneous Inflammatory Disorder: Atopic Dermatitis. Int. J. Mol. Sci. 2016, 17, 1234. [Google Scholar] [CrossRef] [Green Version]
- Mu, Z.; Zhao, Y.; Liu, X.; Chang, C.; Zhang, J. Molecular biology of atopic dermatitis. Clin. Rev. Allergy Immunol. 2014, 47, 193–218. [Google Scholar] [CrossRef] [PubMed]
- Homey, B.; Steinhoff, M.; Ruzicka, T.; Leung, D.Y. Cytokines and chemokines orchestrate atopic skin inflammation. J. Allergy Clin. Immunol. 2006, 118, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Lorz, L.R.; Kim, M.Y.; Cho, J.Y. Medicinal potential of Panax ginseng and its ginsenosides in atopic dermatitis treatment. J. Ginseng Res. 2020, 44, 8–13. [Google Scholar] [CrossRef]
- Giustizieri, M.L.; Mascia, F.; Frezzolini, A.; De Pità, O.; Chinni, L.M.; Giannetti, A.; Girolomoni, G.; Pastore, S. Keratinocytes from patients with atopic dermatitis and psoriasis show a distinct chemokine production profile in response to T cell-derived cytokines. J. Allergy Clin. Immunol. 2001, 107, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Xie, J.; Ma, S.; Liao, W.; Zhang, J.; Luo, R. Targeted knock down of CCL22 and CCL17 by siRNA during DC differentiation and maturation affects the recruitment of T subsets. Immunobiology 2010, 215, 153–162. [Google Scholar] [CrossRef]
- Pavel, A.B.; Song, T.; Kim, H.J.; Del Duca, E.; Krueger, J.G.; Dubin, C.; Peng, X.; Xu, H.; Zhang, N.; Estrada, Y.D.; et al. Oral Janus kinase/SYK inhibition (ASN002) suppresses inflammation and improves epidermal barrier markers in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2019, 144, 1011–1024. [Google Scholar] [CrossRef] [Green Version]
- Miyano, K.; Matsushita, S.; Tsuchida, T.; Nakamura, K. Inhibitory effect of a histamine 4 receptor antagonist on CCL17 and CCL22 production by monocyte-derived Langerhans cells in patients with atopic dermatitis. J. Dermatol. 2016, 43, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Soumelis, V.; Reche, P.A.; Kanzler, H.; Yuan, W.; Edward, G.; Homey, B.; Gilliet, M.; Ho, S.; Antonenko, S.; Lauerma, A.; et al. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nat. Immunol. 2002, 3, 673–680. [Google Scholar] [CrossRef]
- Biedermann, T.; Schwärzler, C.; Lametschwandtner, G.; Thoma, G.; Carballido-Perrig, N.; Kund, J.; de Vries, J.E.; Rot, A.; Carballido, J.M. Targeting CLA/E-selectin interactions prevents CCR4-mediated recruitment of human Th2 memory cells to human skin in vivo. Eur. J. Immunol. 2002, 32, 3171–3180. [Google Scholar] [CrossRef]
- Abi-Younes, S.; Si-Tahar, M.; Luster, A.D. The CC chemokines MDC and TARC induce platelet activation via CCR4. Thromb. Res. 2001, 101, 279–289. [Google Scholar] [CrossRef]
- Nakazato, J.; Kishida, M.; Kuroiwa, R.; Fujiwara, J.; Shimoda, M.; Shinomiya, N. Serum levels of Th2 chemokines, CCL17, CCL22, and CCL27, were the important markers of severity in infantile atopic dermatitis. Pediatr. Allergy Immunol. 2008, 19, 605–613. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Hatanaka, S.; Kimura, Y.; Hara, Y.; Nishiwaki, K.; Quan, Y.S.; Kamiyama, F.; Oiso, N.; Kawada, A.; Kabashima, K.; et al. A CCR4 antagonist ameliorates atopic dermatitis-like skin lesions induced by dibutyl phthalate and a hydrogel patch containing ovalbumin. Biomed. Pharmacother. 2019, 109, 1437–1444. [Google Scholar] [CrossRef] [PubMed]
- Alves, N.S.F.; Setzer, W.N.; da Silva, J.K.R. The chemistry and biological activities of Peperomia pellucida (Piperaceae): A critical review. J. Ethnopharmacol. 2019, 232, 90–102. [Google Scholar] [CrossRef]
- Sukkasem, K.; Panthong, S.; Itharat, A. Antimicrobial Activities of Thai Traditional Remedy “Kheaw-Hom” and Its Plant Ingredients for Skin Infection Treatment in Chickenpox. J. Med. Assoc. Thai. 2016, 99, 116–123. [Google Scholar]
- Sajadimajd, S.; Bahramsoltani, R.; Iranpanah, A.; Kumar Patra, J.; Das, G.; Gouda, S.; Rahimi, R.; Rezaeiamiri, E.; Cao, H.; Giampieri, F.; et al. Advances on Natural Polyphenols as Anticancer Agents for Skin Cancer. Pharmacol. Res. 2020, 151, 104584. [Google Scholar] [CrossRef]
- Zielińska, S.; Czerwińska, M.E.; Dziągwa-Becker, M.; Dryś, A.; Kucharski, M.; Jezierska-Domaradzka, A.; Płachno, B.J.; Matkowski, A. Modulatory Effect of Chelidonium majus Extract and Its Alkaloids on LPS-Stimulated Cytokine Secretion in Human Neutrophils. Molecules 2020, 25, 842. [Google Scholar] [CrossRef] [Green Version]
- Kang, H.; Koppula, S. Hepatoprotective Effect of Houttuynia cordata Thunb Extract against Carbon Tetrachloride-induced Hepatic Damage in Mice. Indian J. Pharm. Sci. 2014, 76, 267–273. [Google Scholar]
- Jung, M.; Lee, T.H.; Oh, H.J.; Kim, H.; Son, Y.; Lee, E.H.; Kim, J. Inhibitory effect of 5,6-dihydroergosteol-glucoside on atopic dermatitis-like skin lesions via suppression of NF-κB and STAT activation. J. Dermatol. Sci. 2015, 79, 252–261. [Google Scholar] [CrossRef]
- Maruyama, T.; Kono, K.; Izawa, S.; Mizukami, Y.; Kawaguchi, Y.; Mimura, K.; Watanabe, M.; Fujii, H. CCL17 and CCL22 chemokines within tumor microenvironment are related to infiltration of regulatory T cells in esophageal squamous cell carcinoma. Dis. Esophagus. 2010, 23, 422–429. [Google Scholar] [CrossRef]
- Vranova, M.; Friess, M.C.; Haghayegh Jahromi, N.; Collado-Diaz, V.; Vallone, A.; Hagedorn, O.; Jadhav, M.; Willrodt, A.H.; Polomska, A.; Leroux, J.C.; et al. Opposing roles of endothelial and leukocyte-expressed IL-7Rα in the regulation of psoriasis-like skin inflammation. Sci. Rep. 2019, 9, 11714. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weidinger, S.; Beck, L.A.; Bieber, T.; Kabashima, K.; Irvine, A.D. Atopic dermatitis. Nat. Rev. Dis. Primers. 2018, 4, 1. [Google Scholar] [CrossRef]
- El Azhary, K.; Tahiri Jouti, N.; El Khachibi, M.; Moutia, M.; Tabyaoui, I.; El Hou, A.; Achtak, H.; Nadifi, S.; Habti, N.; Badou, A. Anti-inflammatory potential of Capparis spinosa L. in vivo in mice through inhibition of cell infiltration and cytokine gene expression. BMC Complement. Altern. Med. 2017, 17, 81. [Google Scholar] [CrossRef] [Green Version]
- Liu, B.; Tai, Y.; Liu, B.; Caceres, A.I.; Yin, C.; Jordt, S.E. Transcriptome profiling reveals Th2 bias and identifies endogenous itch mediators in poison ivy contact dermatitis. JCI Insight 2019, 5, 124497. [Google Scholar] [CrossRef]
- Kumar, S.; Jeong, Y.; Ashraf, M.U.; Bae, Y.S. Dendritic Cell-Mediated Th2 Immunity and Immune Disorders. Int. J. Mol. Sci. 2019, 20, 2159. [Google Scholar] [CrossRef] [Green Version]
- Tajima, H.; Tajiki-Nishino, R.; Watanabe, Y.; Fukuyama, T. Direct activation of aryl hydrocarbon receptor by benzo[a]pyrene elicits T-helper 2-driven proinflammatory responses in a mouse model of allergic dermatitis. J. Appl. Toxicol. 2019, 39, 936–944. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Hieshima, K.; Nagakubo, D.; Sato, E.; Nakayama, M.; Kawa, K.; Yoshie, O. Selective induction of Th2-attracting chemokines CCL17 and CCL22 in human B cells by latent membrane protein 1 of Epstein-Barr virus. J. Virol. 2004, 78, 1665–1674. [Google Scholar] [CrossRef] [Green Version]
- Fujii, Y.; Takeuchi, H.; Sakuma, S.; Sengoku, T.; Takakura, S. Characterization of a 2,4-dinitrochlorobenzene-induced chronic dermatitis model in rats. Skin Pharmacol. Physiol. 2009, 22, 240–247. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; Shi, T.; Hou, J.; Li, M. Ferulic acid alleviates atopic dermatitis-like symptoms in mice via its potent anti-inflammatory effect. Immunopharmacol. Immunotoxicol. 2020, 42, 156–164. [Google Scholar] [CrossRef]
- Cha, K.J.; Song, C.S.; Lee, J.S.; Kashif, A.; Hong, M.H.; Kim, G.; Kim, I.S. Chaenomeles sinensis Koehne extract suppresses the development of atopic dermatitis-like lesions by regulating cytokine and filaggrin expression in NC/Nga mice. Int. J. Med. Sci. 2019, 16, 1604–1613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- An, E.J.; Kim, Y.; Lee, S.H.; Choi, S.H.; Chung, W.S.; Jang, H.J. Ophiopogonin D ameliorates DNCB-induced atopic dermatitis-like lesions in BALB/c mice and TNF-α- inflamed HaCaT cell. Biochem. Biophys. Res. Commun. 2020, 522, 40–46. [Google Scholar] [CrossRef]
- Miao, L.; Tao, H.; Peng, Y.; Wang, S.; Zhong, Z.; El-Seedi, H.; Dragan, S.; Zengin, G.; Cheang, W.S.; Wang, Y.; et al. The anti-inflammatory potential of Portulaca oleracea L. (purslane) extract by partial suppression on NF-κB and MAPK activation. Food Chem. 2019, 290, 239–245. [Google Scholar] [CrossRef]
- Rahimi, V.B.; Ajam, F.; Rakhshandeh, H.; Askari, V.R. A Pharmacological Review on Portulaca oleracea L.: Focusing on Anti-Inflammatory, Anti-Oxidant, Immuno-Modulatory and Antitumor Activities. J. Pharmacopunct. 2019, 22, 7–15. [Google Scholar]
- Yang, X.; Ying, Z.; Liu, H.; Ying, X.; Yang, G. A new homoisoflavone from Portulaca oleracea L. and its antioxidant activity. Nat. Prod. Res. 2019, 33, 3500–3506. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Xin, H.; Rahman, K.; Wang, S.; Peng, C.; Zhang, H. Portulaca oleracea L.: A review of phytochemistry and pharmacological effects. Biomed. Res. Int. 2015, 2015, 925631. [Google Scholar] [CrossRef] [Green Version]
- Lee, H.S.; Jeong, G.S. Therapeutic effect of kaempferol on atopic dermatitis by attenuation of T cell activity via interaction with multidrug resistance-associated protein 1. Br. J. Pharmacol. 2021, 178, 1772–1788. [Google Scholar] [CrossRef] [PubMed]
- Hong, M.H.; Lee, J.Y.; Jung, H.; Jin, D.H.; Go, H.Y.; Kim, J.H.; Jang, B.H.; Shin, Y.C.; Ko, S.G. Sophora flavescens Aiton inhibits the production of pro-inflammatory cytokines through inhibition of the NF kappaB/IkappaB signal pathway in human mast cell line (HMC-1). Toxicol. Vitro 2009, 23, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Hillerns, P.I.; Wink, M. Binding of flavonoids from Sophora flavescens to the rat uterine estrogrn recptor. Planta. Med. 2005, 71, 1065–1068. [Google Scholar] [CrossRef]
- Liao, W.; He, X.; Yi, Z.; Xiang, W.; Ding, Y. Chelidonine suppresses LPS-induced production of inflammatory mediators through the inhibitory of TLR4/NF-κB signaling pathway in RAW264.7 macrophages. Biomed. Pharmacother. 2018, 107, 1151–1159. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, S.; Lee, S.; Weon, Y.; Kim, T.; Kim, H.; Lee, T. Anti-Inflammatory Activity of a Medicinal Herb Extract Mixture, HM-V, on an Animal Model of DNCB-Induced Chronic Skin Inflammation. Plants 2021, 10, 1546. https://doi.org/10.3390/plants10081546
Park S, Lee S, Weon Y, Kim T, Kim H, Lee T. Anti-Inflammatory Activity of a Medicinal Herb Extract Mixture, HM-V, on an Animal Model of DNCB-Induced Chronic Skin Inflammation. Plants. 2021; 10(8):1546. https://doi.org/10.3390/plants10081546
Chicago/Turabian StylePark, Sungbae, Sangmin Lee, Youngho Weon, Taewook Kim, Hakwon Kim, and Taehoon Lee. 2021. "Anti-Inflammatory Activity of a Medicinal Herb Extract Mixture, HM-V, on an Animal Model of DNCB-Induced Chronic Skin Inflammation" Plants 10, no. 8: 1546. https://doi.org/10.3390/plants10081546
APA StylePark, S., Lee, S., Weon, Y., Kim, T., Kim, H., & Lee, T. (2021). Anti-Inflammatory Activity of a Medicinal Herb Extract Mixture, HM-V, on an Animal Model of DNCB-Induced Chronic Skin Inflammation. Plants, 10(8), 1546. https://doi.org/10.3390/plants10081546





