Phytocannabinoids Biosynthesis in Angiosperms, Fungi, and Liverworts and Their Versatile Role
Abstract
1. Introduction
2. Phytocannabinoid Biosynthesis Sites
3. Biosynthesis of Phytocannabinoids
3.1. Cannabis sativa
3.2. Rhododendron
3.3. Liverworts
3.4. Application of Biotechnological Approaches to Phytocannabinoids Production in Heterologous Hosts
3.5. Production of Phytocannabinoids through Synthetic Approaches
4. Phytocannabinoid Storage and Maintenance of Cell Homeostasis
5. Biotechnology and In Vitro Propagation of Cannabis
6. Phytocannabinoids and Their Derivative Role and Bioactivity in Humans and Animals
6.1. Cannabinoid Receptors in Humans and Their Role
6.2. Bioactivity of Phytocannabinoids
6.2.1. Neutral Cannabinoids
6.2.2. Cannabinoid Acid
6.2.3. Bibenzyl Cannabinoids
6.2.4. Rhododendron Cannabinoids
7. Antibacterial and Antimicrobial Property of Phytocannabinoids
8. Phytocannabinoids in Stress Tolerance
9. Phytocannabinoid Foe
10. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAE1 | acyl-activated enzyme 1 |
| ABA | abscisic acid |
| ADME | absorption, distribution, metabolism, excretion |
| BBS | bibenzyl synthase |
| C4H | cinnamate 4-hydroxylase |
| 4CL | 4-coumarate:CoA ligase |
| CBR | cannabinoid receptor |
| CB1R | cannabinoid receptor type 1 |
| CB2R | cannabinoid receptor type 2 |
| CBC | cannabichromene |
| CBCA | cannabichromenic acid |
| CBCAS | cannabichromenic acid synthase |
| CBCV | cannabichromevarine |
| CBCVA | cannabichromevarinic acid |
| CBD | cannabidiol |
| CBDA | cannabidiolic acid |
| CBDAS | cannabidiolic acid synthase |
| CBDV | cannabidivarine |
| CBDVA | cannabidivarinic acid |
| CBE | cannabielsoin |
| CBG | cannabigerol |
| CBGA | cannabigerolic acid |
| CBGAS | cannabigerolic acid synthase |
| CBL | cannabicyclol |
| CBN | cannabinol |
| CBND | cannabidiol |
| CBT | cannabitriol |
| CHIL | chalcone isomerases-like protein |
| CNS | central nervous system |
| CYP | cytochrome P450 |
| DBR | double-bond reductase |
| DCA | daurichromenic acid |
| DCAS | daurichromenic acid synthase |
| DHAC | dihydrostilbene acid cyclase |
| Dicamba | 3,6-dichloro-2-methoxybenzoic acid |
| EC50 | effective concentration |
| GFA | grifolic acid |
| GPP | geranyl pyrophosphate |
| GPR | G-protein-coupled receptor |
| GPCR | G-protein-coupled cannabinoid receptor |
| IAA | indole-3-acetic acid |
| KR | ketoreductase |
| LOX | lipoxygenase |
| MEP | methylerythritol-4-phosphate |
| NAA | 1-naphthaleneacetic acid |
| NADES | natural deep eutectic solvent |
| OA | olivetolic acid |
| OAC | olivetolic acid cyclase |
| OLS | olivetol synthase |
| ORS | orcinol synthase |
| OSA | orsellinic acid |
| PA | perrottetinenic acid |
| PAL | phenylalanine ammonia-lyase |
| PAS | perrottetinenic acid synthase |
| PET | perrottetinene |
| PGR | plant growth regulator |
| PKS | polyketide synthase |
| PPARγ | peroxisome proliferator-activated receptor γ |
| PT | prenyltransferase |
| STS | stilbene synthase |
| TAL | tyrosine ammonia-lyase |
| TF | transcription factor |
| TI | therapeutic index |
| TRP | transient receptor potential |
| TRPA1 | TRP type ankyrin 1 |
| TRPM8 | TRP type melastanin 8 |
| TRPV1 | TRP type vanilloid 1 |
| Δ8-THC | Δ8-tetrahydrocannabinol |
| Δ9-THC | Δ9-tetrahydrocannabinol |
| Δ9-THCA | Δ9-tetrahydrocannabinolic acid |
| Δ9-THCAS | Δ9-tetrahydrocannabinolic acid synthase |
| Δ9-THCV | Δ9-tetrahydrocannabivarinic acid |
| Δ9-THCVA | Δ9-tetrahydrocannabidivarinic acid |
References
- Gülck, T.; Møller, B.L. Phytocannabinoids: Origins and biosynthesis. Trends Plant Sci. 2020, 25, 985–1004. [Google Scholar] [CrossRef] [PubMed]
- Hanuš, L.O.; Meyer, S.M.; Muñoz, E.; Taglialatela-Scafati, O.; Appendino, G. Phytocannabinoids: A unified critical inventory. Nat. Prod. Rep. 2016, 33, 1357–1392. [Google Scholar] [CrossRef]
- ElSohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Berman, P.; Futoran, K.; Lewitus, G.M.; Mukha, D.; Benami, M.; Shlomi, T.; Meiri, D. A new ESI-LC/MS approach for comprehensive metabolic profiling of phytocannabinoids in Cannabis. Sci. Rep. 2018, 8, 14280. [Google Scholar] [CrossRef] [PubMed]
- Pollastro, F.; Caprioglio, D.; Del Prete, D.; Rogati, F.; Minassi, A.; Taglialatela-Scafati, O.; Munoz, E.; Appendino, G. Cannabichromene. Nat. Prod. Commun. 2018, 13, 1189–1194. [Google Scholar] [CrossRef]
- Happyana, N.; Kayser, O. Monitoring metabolite profiles of Cannabis sativa L. trichomes during flowering period using 1H NMR-based metabolomics and real-time PCR. Planta Med. 2016, 82, 1217–1223. [Google Scholar] [CrossRef] [PubMed]
- Chanda, D.; Neumann, D.; Glatz, J.F.C. The endocannabinoid system: Overview of an emerging multi-faceted therapeutic target. Prostaglandins Leukot. Essent. Fatty Acids 2019, 140, 51–56. [Google Scholar] [CrossRef]
- Flores-Sanchez, I.J.; Verpoorte, R. Secondary metabolism in Cannabis. Phytochem. Rev. 2008, 7, 615–639. [Google Scholar] [CrossRef]
- Dei Cas, M.; Casagni, E.; Casiraghi, A.; Minghetti, P.; Fornasari, D.M.M.; Ferri, F.; Arnoldi, S.; Gambaro, V.; Roda, G. Phytocannabinoids profile in medicinal cannabis oils: The impact of plant varieties and preparation methods. Front. Pharmacol. 2020, 11, 570616. [Google Scholar] [CrossRef]
- Pollastro, F.; De Petrocellis, L.; Schiano-Moriello, A.; Chianese, G.; Heyman, H.; Appendino, G.; Taglialatela-Scafati, O. Amorfrutin-type phytocannabinoids from Helichrysum umbraculigerum. Fitoterapia 2017, 123, 13–17. [Google Scholar] [CrossRef]
- Taura, F.; Iijima, M.; Kurosaki, F. Daurichromenic acid and grifolic acid: Phytotoxic meroterpenoids that induce cell death in cell culture of their producer Rhododendron dauricum. Plant Signal. Behav. 2018, 13, e1422463. [Google Scholar] [CrossRef] [PubMed]
- Taura, F.; Iijima, M.; Lee, J.-B.; Hashimoto, T.; Asakawa, Y.; Kurosaki, F. Daurichromenic acid-producing oxidocyclase in the young leaves of Rhododendron dauricum. Nat. Prod. Commun. 2014, 9, 1329–1332. [Google Scholar] [CrossRef]
- Kashiwada, Y.; Yamazaki, K.; Ikeshiro, Y.; Yamagishi, T.; Fujioka, T.; Mihashi, K.; Mizuki, K.; Cosentino, L.M.; Fowke, K.; Morris-Natschke, S.L.; et al. Isolation of rhododaurichromanic acid B and the anti-HIV principles rhododaurichromanic acid A and rhododaurichromenic acid from Rhododendron dauricum. Tetrahedron 2001, 57, 1559–1563. [Google Scholar] [CrossRef]
- Rogachev, A.; Komarova, N.; Korchagina, D.; Dolgikh, M.; Sorokina, I.; Baev, D.; Tolstikova, T.; Fomenko, V.; Salakhutdinov, N. Some prenylated phenols of Rhododendron adamsii: Isolation, modification and pharmacological tests. Chem. Sustain. Dev. 2009, 17, 185–193. [Google Scholar]
- Yang, Y.-X.; Wang, J.-X.; Wang, Q.; Li, H.-L.; Tao, M.; Luo, Q.; Liu, H. New chromane and chromene meroterpenoids from flowers of Rhododendron rubiginosum Franch. var. rubiginosum. Fitoterapia 2018, 127, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Iwata, N.; Kitanaka, S. New cannabinoid-like chromane and chromene derivatives from Rhododendron anthopogonoides. Chem. Pharm. Bull. 2011, 59, 1409–1412. [Google Scholar] [CrossRef] [PubMed]
- Chicca, A.; Schafroth, M.A.; Reynoso-Moreno, I.; Erni, R.; Petrucci, V.; Carreira, E.M.; Gertsch, J. Uncovering the psychoactivity of a cannabinoid from liverworts associated with a legal high. Sci. Adv. 2018, 4, eaat2166. [Google Scholar] [CrossRef]
- Hussain, T.; Espley, R.V.; Gertsch, J.; Whare, T.; Stehle, F.; Kayser, O. Demystifying the liverwort Radula marginata, a critical review on its taxonomy, genetics, cannabinoid phytochemistry and pharmacology. Phytochem. Rev. 2019, 18, 953–965. [Google Scholar] [CrossRef]
- Métoyer, B.; Lebouvier, N.; Hnawia, E.; Herbette, G.; Thouvenot, L.; Asakawa, Y.; Nour, M.; Raharivelomanana, P. Chemotypes and biomarkers of seven species of new Caledonian liverworts from the Bazzanioideae subfamily. Molecules 2018, 23, 1353. [Google Scholar] [CrossRef] [PubMed]
- Pryce, R.J. Lunularic acid, a common endogenous growth inhibitor of liverworts. Planta 1971, 97, 354–357. [Google Scholar] [CrossRef]
- Quaghebeur, K.; Coosemans, J.; Toppet, S.; Compernolle, F. Cannabiorci- and 8-chlorocannabiorcichromenic acid as fungal antagonists from Cylindrocarpon olidum. Phytochemistry 1994, 37, 159–161. [Google Scholar] [CrossRef]
- Elhendawy, M.A.; Wanas, A.S.; Radwan, M.M.; Azzaz, N.A.; Toson, E.S.; ElSohly, M.A. Chemical and biological studies of Cannabis sativa roots. Med. Cannabis Cannabinoids 2019, 1, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Happyana, N.; Agnolet, S.; Muntendam, R.; Van Dam, A.; Schneider, B.; Kayser, O. Analysis of cannabinoids in laser-microdissected trichomes of medicinal Cannabis sativa using LCMS and cryogenic NMR. Phytochemistry 2013, 87, 51–59. [Google Scholar] [CrossRef]
- Livingston, S.J.; Quilichini, T.D.; Booth, J.K.; Wong, D.C.J.; Rensing, K.H.; Laflamme-Yonkman, J.; Castellarin, S.D.; Bohlmann, J.; Page, J.E.; Samuels, A.L. Cannabis glandular trichomes alter morphology and metabolite content during flower maturation. Plant J. 2019, 101, 37–56. [Google Scholar] [CrossRef]
- Mahlberg, P.G.; Kim, E.-S. Cuticle development on glandular trichomes of Cannabis sativa (Cannabaceae). Am. J. Bot. 1991, 78, 1113–1122. [Google Scholar] [CrossRef]
- Eichhorn Bilodeau, S.; Wu, B.-S.; Rufyikiri, A.-S.; MacPherson, S.; Lefsrud, M. An update on plant photobiology and implications for cannabis production. Front. Plant Sci. 2019, 10, 296. [Google Scholar] [CrossRef]
- Iijima, M.; Munakata, R.; Takahashi, H.; Kenmoku, H.; Nakagawa, R.; Kodama, T.; Asakawa, Y.; Abe, I.; Yazaki, K.; Kurosaki, F.; et al. Identification and characterization of daurichromenic acid synthase active in anti-HIV biosynthesis. Plant Physiol. 2017, 174, 2213–2230. [Google Scholar] [CrossRef] [PubMed]
- Asakawa, Y.; Ludwiczuk, A. Chemical constituents of bryophytes: Structures and biological activity. J. Nat. Prod. 2017, 81, 641–660. [Google Scholar] [CrossRef]
- He, X.; Sun, Y.; Zhu, R.-L. The oil bodies of liverworts: Unique and important organelles in land plants. Crit. Rev. Plant Sci. 2013, 32, 293–302. [Google Scholar] [CrossRef]
- Geissler, M.; Volk, J.; Stehle, F.; Kayser, O.; Warzecha, H. Subcellular localization defines modification and production of Δ9-tetrahydrocannabinolic acid synthase in transiently transformed Nicotiana benthamiana. Biotechnol. Lett. 2018, 40, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Reiter, M.A.; d’Espaux, L.; Wong, J.; Denby, C.M.; Lechner, A.; Zhang, Y.; Grzybowski, A.T.; Harth, S.; Lin, W.; et al. Complete biosynthesis of cannabinoids and their unnatural analogues in yeast. Nature 2019, 567, 123–126. [Google Scholar] [CrossRef]
- Rodziewicz, P.; Loroch, S.; Marczak, Ł.; Sickmann, A.; Kayser, O. Cannabinoid synthases and osmoprotective metabolites accumulate in the exudates of Cannabis sativa L. glandular trichomes. Plant Sci. 2019, 284, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Gagne, S.J.; Stout, J.M.; Liu, E.; Boubakir, Z.; Clark, S.M.; Page, J.E. Identification of olivetolic acid cyclase from Cannabis sativa reveals a unique catalytic route to plant polyketides. Proc. Natl. Acad. Sci. USA 2012, 109, 12811–12816. [Google Scholar] [CrossRef] [PubMed]
- Raharjo, T.J.; Chang, W.-T.; Choi, Y.H.; Peltenburg-Looman, A.M.G.; Verpoorte, R. Olivetol as product of a polyketide synthase in Cannabis sativa L. Plant Sci. 2004, 166, 381–385. [Google Scholar] [CrossRef]
- Laverty, K.U.; Stout, J.M.; Sullivan, M.J.; Shah, H.; Gill, N.; Holbrook, L.; Deikus, G.; Sebra, R.; Hughes, T.R.; Page, J.E.; et al. A physical and genetic map of Cannabis sativa identifies extensive rearrangements at the THC/CBD acid synthase loci. Genome Res. 2019, 29, 146–156. [Google Scholar] [CrossRef] [PubMed]
- Saeki, H.; Hara, R.; Takahashi, H.; Iijima, M.; Munakata, R.; Kenmoku, H.; Fuku, K.; Sekihara, A.; Yasuno, Y.; Shinada, T.; et al. An aromatic farnesyltransferase functions in biosynthesis of the anti-HIV meroterpenoid daurichromenic acid. Plant Physiol. 2018, 178, 535–551. [Google Scholar] [CrossRef]
- Taura, F.; Iijima, M.; Yamanaka, E.; Takahashi, H.; Kenmoku, H.; Saeki, H.; Morimoto, S.; Asakawa, Y.; Kurosaki, F.; Morita, H. A novel class of plant type III polyketide synthase involved in orsellinic acid biosynthesis from Rhododendron dauricum. Front. Plant Sci. 2016, 7, 1452. [Google Scholar] [CrossRef] [PubMed]
- Katsuyama, Y.; Kita, T.; Funa, N.; Horinouchi, S. Curcuminoid biosynthesis by two type III polyketide synthases in the herb Curcuma longa. J. Biol. Chem. 2009, 284, 11160–11170. [Google Scholar] [CrossRef]
- Hussain, T.; Plunkett, B.; Ejaz, M.; Espley, R.V.; Kayser, O. Identification of putative precursor genes for the biosynthesis of cannabinoid-like compound in Radula marginata. Front. Plant Sci. 2018, 9, 537. [Google Scholar] [CrossRef]
- Akiyama, T.; Shibuya, M.; Liu, H.-M.; Ebizuka, Y. p-Coumaroyltriacetic acid synthase, a new homologue of chalcone synthase, from Hydrangea macrophylla var. thunbergii. Eur. J. Biochem. 1999, 263, 834–839. [Google Scholar] [CrossRef]
- Austin, M.B.; Noel, J.P. The chalcone synthase superfamily of type III polyketide synthases. Nat. Prod. Rep. 2002, 20, 79–110. [Google Scholar] [CrossRef]
- Imoto, S.A.; Ohta, Y. Intracellular localization of lunularic acid and prelunularic acid in suspension cultured cells of Marchantia polymorpha. Plant Physiol. 1985, 79, 751–755. [Google Scholar] [CrossRef] [PubMed]
- Toyota, M.; Shimamura, T.; Ishii, H.; Renner, M.; Braggins, J.; Asakawa, Y. New bibenzyl cannabinoid from the New Zealand liverwort Radula marginata. Chem. Pharm. Bull. 2002, 50, 1390–1392. [Google Scholar] [CrossRef]
- Yang, X.; Matsui, T.; Kodama, T.; Mori, T.; Zhou, X.; Taura, F.; Noguchi, H.; Abe, I.; Morita, H. Structural basis for olivetolic acid formation by a polyketide cyclase from Cannabis sativa. FEBS J. 2016, 283, 1088–1106. [Google Scholar] [CrossRef]
- Toyota, M.; Kinugawa, T.; Asakawa, Y. Bibenzyl cannabinoid and bisbibenzyl derivative from the liverwort Radula perrottetii. Phytochemistry 1994, 37, 859–862. [Google Scholar] [CrossRef]
- Fuentes, P.; Armarego-Marriott, T.; Bock, R. Plastid transformation and its application in metabolic engineering. Curr. Opin. Biotechnol. 2018, 49, 10–15. [Google Scholar] [CrossRef]
- Kodym, A.; Leeb, C.J. Back to the roots: Protocol for the photoautotrophic micropropagation of medicinal Cannabis. Plant Cell Tissue Organ Cult. 2019, 138, 399–402. [Google Scholar] [CrossRef]
- Lange, K.; Schmid, A.; Julsing, M.K. Δ9-Tetrahydrocannabinolic acid synthase: The application of a plant secondary metabolite enzyme in biocatalytic chemical synthesis. J. Biotechnol. 2016, 233, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Lata, H.; Chandra, S.; Khan, I.A.; ElSohly, M.A. Micropropagation of Cannabis sativa L. -An update. In Cannabis sativa L. Botany and Biotechnology; Chandra, S., Lata, H., ElSohly, M.A., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 285–297. [Google Scholar] [CrossRef]
- Wróbel, T.; Dreger, M.; Wielgus, K.; Słomski, R. The application of plant in vitro cultures in cannabinoid production. Biotechnol. Lett. 2017, 40, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Andersen-Ranberg, J.; Kongstad, K.T.; Nielsen, M.T.; Jensen, N.B.; Pateraki, I.; Bach, S.S.; Hamberger, B.; Zerbe, P.; Staerk, D.; Bohlmann, J.; et al. Expanding the landscape of diterpene structural diversity through stereochemically controlled combinatorial biosynthesis. Angew. Chem. Int. Ed. 2016, 55, 2142–2146. [Google Scholar] [CrossRef]
- Chen, R.; Gao, B.; Liu, X.; Ruan, F.; Zhang, Y.; Lou, J.; Feng, K.; Wunsch, C.; Li, S.-M.; Dai, J.; et al. Molecular insights into the enzyme promiscuity of an aromatic prenyltransferase. Nat. Chem. Biol. 2017, 13, 226–234. [Google Scholar] [CrossRef]
- Kannangara, R.; Siukstaite, L.; Borch-Jensen, J.; Madsen, B.; Kongstad, K.T.; Staerk, D.; Bennedsen, M.; Okkels, F.T.; Rasmussen, S.A.; Larsen, T.O.; et al. Characterization of a membrane-bound C-glucosyltransferase responsible for carminic acid biosynthesis in Dactylopius coccus Costa. Nat. Commun. 2017, 8, 1987. [Google Scholar] [CrossRef]
- Yazaki, K.; Sasaki, K.; Tsurumaru, Y. Prenylation of aromatic compounds, a key diversification of plant secondary metabolites. Phytochemistry 2009, 70, 1739–1745. [Google Scholar] [CrossRef] [PubMed]
- Ban, Z.; Qin, H.; Mitchell, A.J.; Liu, B.; Zhang, F.; Weng, J.-K.; Dixon, R.A.; Wang, G. Noncatalytic chalcone isomerase-fold proteins in Humulus lupulus are auxiliary components in prenylated flavonoid biosynthesis. Proc. Natl. Acad. Sci. USA 2018, 115, E5223–E5232. [Google Scholar] [CrossRef] [PubMed]
- Gribble, G.W. Biological activity of recently discovered halogenated marine natural products. Mar. Drugs 2015, 13, 4044–4136. [Google Scholar] [CrossRef] [PubMed]
- Hansen, E.H.; Osmani, S.A.; Kristensen, C.; Møller, B.L.; Hansen, J. Substrate specificities of family 1 UGTs gained by domain swapping. Phytochemistry 2009, 70, 473–482. [Google Scholar] [CrossRef]
- Khan, B.A.; Warner, P.; Wang, H. Antibacterial properties of hemp and other natural fibre plants: A review. BioResources 2014, 9, 3642–3659. [Google Scholar] [CrossRef]
- Choi, Y.H.; van Spronsen, J.; Dai, Y.; Verberne, M.; Hollmann, F.; Arends, I.W.C.E.; Witkamp, G.-J.; Verpoorte, R. Are natural deep eutectic solvents the missing link in understanding cellular metabolism and physiology? Plant Physiol. 2011, 156, 1701–1705. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Tailoring properties of natural deep eutectic solvents with water to facilitate their applications. Food Chem. 2015, 187, 14–19. [Google Scholar] [CrossRef]
- Courchaine, E.M.; Lu, A.; Neugebauer, K.M. Droplet organelles? EMBO J. 2016, 35, 1603–1612. [Google Scholar] [CrossRef]
- Hammond, O.S.; Bowron, D.T.; Edler, K.J. The effect of water upon deep eutectic solvent nanostructure: An unusual transition from ionic mixture to aqueous solution. Angew. Chem. Int. Ed. 2017, 56, 9782–9785. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, C.; Bavishi, K.; Viborg, K.M.; Drew, D.P.; Simonsen, H.T.; Motawia, M.S.; Møller, B.L.; Laursen, T. Stabilization of dhurrin biosynthetic enzymes from Sorghum bicolor using a natural deep eutectic solvent. Phytochemistry 2020, 170, 112214. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, C.; Gallage, N.J.; Hansen, C.C.; Møller, B.L.; Laursen, T. Dynamic metabolic solutions to the sessile life style of plants. Nat. Prod. Rep. 2018, 35, 1140–1155. [Google Scholar] [CrossRef] [PubMed]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 19. [Google Scholar] [CrossRef] [PubMed]
- Lata, H.; Chandra, S.; Techen, N.; Khan, I.A.; ElSohly, M.A. Molecular analysis of genetic fidelity in Cannabis sativa L. plants grown from synthetic (encapsulated) seeds following in vitro storage. Biotechnol. Lett. 2011, 33, 2503–2508. [Google Scholar] [CrossRef] [PubMed]
- Gai, Q.-Y.; Jiao, J.; Luo, M.; Wei, Z.-F.; Zu, Y.-G.; Ma, W.; Fu, Y.-J. Establishment of hairy root cultures by Agrobacterium rhizogenes mediated transformation of Isatis tinctoria L. For the efficient production of flavonoids and evaluation of antioxidant activities. PLoS ONE 2015, 10, e0119022. [Google Scholar] [CrossRef]
- Feeney, M.; Punja, Z.K. Tissue culture and Agrobacterium-mediated transformation of hemp (Cannabis sativa L.). In Vitro Cell. Dev. Biol. Plant 2003, 39, 578–585. [Google Scholar] [CrossRef]
- Lata, H.; Chandra, S.; Khan, I.; ElSohly, M. High frequency plant regeneration from leaf derived callus of high Δ9-tetrahydrocannabinol yielding Cannabis sativa L. Planta Med. 2010, 76, 1629–1633. [Google Scholar] [CrossRef]
- Hazekamp, A.; Verpoorte, R. Structure elucidation of the tetrahydrocannabinol complex with randomly methylated β-cyclodextrin. Eur. J. Pharm. Sci. 2006, 29, 340–347. [Google Scholar] [CrossRef]
- Pellechia, T. Legal Cannabis Industry Poised for Big Growth, in North America and around the World. Forbes 2018. Available online: https://www.forbes.com/sites/thomaspellechia/2018/03/01/double-digit-billions-puts-north-america-in-the-worldwide-cannabis-market-lead/?sh=356dc9565109 (accessed on 15 April 2021).
- Booth, J.K.; Bohlmann, J. Terpenes in Cannabis sativa—From plant genome to humans. Plant Sci. 2019, 284, 67–72. [Google Scholar] [CrossRef]
- Laprairie, R.B.; Bagher, A.M.; Kelly, M.E.M.; Dupré, D.J.; Denovan-Wright, E.M. Type 1 cannabinoid receptor ligands display functional selectivity in a cell culture model of striatal medium spiny projection neurons. J. Biol. Chem. 2014, 289, 24845–24862. [Google Scholar] [CrossRef]
- Kendall, D.A.; Yudowski, G.A. Cannabinoid receptors in the central nervous system: Their signaling and roles in disease. Front. Cell. Neurosci. 2017, 10, 294. [Google Scholar] [CrossRef]
- Pryce, G.; Riddall, D.R.; Selwood, D.L.; Giovannoni, G.; Baker, D. Neuroprotection in experimental autoimmune encephalomyelitis and progressive multiple sclerosis by cannabis-based cannabinoids. J. Neuroimmune Pharmacol. 2014, 10, 281–292. [Google Scholar] [CrossRef]
- Hippalgaonkar, K.; Gul, W.; ElSohly, M.A.; Repka, M.A.; Majumdar, S. Enhanced solubility, stability, and transcorneal permeability of δ-8-tetrahydrocannabinol in the presence of cyclodextrins. AAPS PharmSciTech 2011, 12, 723–731. [Google Scholar] [CrossRef]
- Nadal, X.; Del Río, C.; Casano, S.; Palomares, B.; Ferreiro-Vera, C.; Navarrete, C.; Sánchez-Carnerero, C.; Cantarero, I.; Bellido, M.L.; Meyer, S.; et al. Tetrahydrocannabinolic acid is a potent PPARγ agonist with neuroprotective activity. Br. J. Pharmacol. 2017, 174, 4263–4276. [Google Scholar] [CrossRef]
- Turner, S.E.; Williams, C.M.; Iversen, L.; Whalley, B.J. Molecular pharmacology of phytocannabinoids. In Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa; Kinghorn, A.D., Falk, H., Gibbons, S., Kobayashi, J.I., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 61–101. [Google Scholar] [CrossRef]
- Huestis, M.A.; Solimini, R.; Pichini, S.; Pacifici, R.; Carlier, J.; Busardò, F.P. Cannabidiol adverse effects and yoxicity. Curr. Neuropharmacol. 2019, 17, 974–989. [Google Scholar] [CrossRef]
- Devinsky, O.; Verducci, C.; Thiele, E.A.; Laux, L.C.; Patel, A.D.; Filloux, F.; Szaflarski, J.P.; Wilfong, A.; Clark, G.D.; Park, Y.D.; et al. Open-label use of highly purified CBD (Epidiolex®) in patients with CDKL5 deficiency disorder and Aicardi, Dup15q, and Doose syndromes. Epilepsy Behav. 2018, 86, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Rok Lee, Y.; Wang, X. A short synthetic route to biologically active (±)-daurichromenic acid as highly potent anti-HIV agent. Org. Biomol. Chem. 2005, 3, 3955. [Google Scholar] [CrossRef] [PubMed]
- Verhoeckx, K.C.M.; Korthout, H.A.A.J.; van Meeteren-Kreikamp, A.P.; Ehlert, K.A.; Wang, M.; van der Greef, J.; Rodenburg, R.J.T.; Witkamp, R.F. Unheated Cannabis sativa extracts and its major compound THC-acid have potential immuno-modulating properties not mediated by CB1 and CB2 receptor coupled pathways. Int. Immunopharmacol. 2006, 6, 656–665. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Dias, T.A.; Brito, A.; Proença, F. Biological importance of structurally diversified chromenes. Eur. J. Med. Chem. 2016, 123, 487–507. [Google Scholar] [CrossRef]
- Weidner, C.; Wowro, S.J.; Freiwald, A.; Kawamoto, K.; Witzke, A.; Kliem, M.; Siems, K.; Müller-Kuhrt, L.; Schroeder, F.C.; Sauer, S. Amorfrutin B is an efficient natural peroxisome proliferator-activated receptor gamma (PPARγ) agonist with potent glucose-lowering properties. Diabetologia 2013, 56, 1802–1812. [Google Scholar] [CrossRef]
- Ali, E.M.M.; Almagboul, A.Z.I.; Khogali, S.M.E.; Gergeir, U.M.A. Antimicrobial activity of Cannabis sativa L. Chin. Med. 2012, 3, 61–64. [Google Scholar] [CrossRef]
- Solymosi, K.; Kofalvi, A. Cannabis: A treasure trove or Pandora’s Box? Mini Rev. Med. Chem. 2017, 17, 1223–1291. [Google Scholar] [CrossRef] [PubMed]
- Morales, P.; Hurst, D.P.; Reggio, P.H. Molecular targets of the phytocannabinoids: A complex picture. In Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa; Kinghorn, A.D., Falk, H., Gibbons, S., Kobayashi, J.i., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 103–131. [Google Scholar] [CrossRef]
- Lenkey, N.; Kirizs, T.; Holderith, N.; Máté, Z.; Szabó, G.; Vizi, E.S.; Hájos, N.; Nusser, Z. Tonic endocannabinoid-mediated modulation of GABA release is independent of the CB1 content of axon terminals. Nat. Commun. 2015, 6, 6557. [Google Scholar] [CrossRef] [PubMed]
- Herkenham, M.; Lynn, A.B.; Johnson, M.R.; Melvin, L.S.; de Costa, B.R.; Rice, K.C. Characterization and localization of cannabinoid receptors in rat brain: A quantitative in vitro autoradiographic study. J. Neurosci. 1991, 11, 563–583. [Google Scholar] [CrossRef] [PubMed]
- Fezza, F.; Bari, M.; Florio, R.; Talamonti, E.; Feole, M.; Maccarrone, M. Endocannabinoids, related compounds and their metabolic routes. Molecules 2014, 19, 17078–17106. [Google Scholar] [CrossRef] [PubMed]
- Mukhopadhyay, P.; Baggelaar, M.; Erdelyi, K.; Cao, Z.; Cinar, R.; Fezza, F.; Ignatowska-Janlowska, B.; Wilkerson, J.; van Gils, N.; Hansen, T.; et al. The novel, orally available and peripherally restricted selective cannabinoid CB2 receptor agonist LEI-101 prevents cisplatin-induced nephrotoxicity. Br. J. Pharmacol. 2016, 173, 446–458. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.P.H. So what do we call GPR18 now? Br. J. Pharmacol. 2012, 165, 2411–2413. [Google Scholar] [CrossRef]
- Caldwell, M.D.; Hu, S.S.-J.; Viswanathan, S.; Bradshaw, H.; Kelly, M.E.M.; Straiker, A. A GPR18-based signalling system regulates IOP in murine eye. Br. J. Pharmacol. 2013, 169, 834–843. [Google Scholar] [CrossRef]
- McHugh, D.; Page, J.; Dunn, E.; Bradshaw, H.B. Δ9-Tetrahydrocannabinol and N-arachidonyl glycine are full agonists at GPR18 receptors and induce migration in human endometrial HEC-1B cells. Br. J. Pharmacol. 2012, 165, 2414–2424. [Google Scholar] [CrossRef] [PubMed]
- Console-Bram, L.; Brailoiu, E.; Brailoiu, G.C.; Sharir, H.; Abood, M.E. Activation of GPR18 by cannabinoid compounds: A tale of biased agonism. Br. J. Pharmacol. 2014, 171, 3908–3917. [Google Scholar] [CrossRef] [PubMed]
- Lu, V.B.; Puhl, H.L., 3rd; Ikeda, S.R. N-Arachidonyl glycine does not activate G protein-coupled receptor 18 signaling via canonical pathways. Mol. Pharmacol. 2013, 83, 267–282. [Google Scholar] [CrossRef]
- Offertáler, L.; Mo, F.-M.; Bátkai, S.; Liu, J.; Begg, M.; Razdan, R.K.; Martin, B.R.; Bukoski, R.D.; Kunos, G. Selective ligands and cellular effectors of a G protein-coupled endothelial cannabinoid receptor. Mol. Pharmacol. 2003, 63, 699–705. [Google Scholar] [CrossRef]
- Baker, D.; Pryce, G.; Davies, W.L.; Hiley, C.R. In silico patent searching reveals a new cannabinoid receptor. Trends Pharmacol. Sci. 2006, 27, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Sawzdargo, M.; Nguyen, T.; Lee, D.K.; Lynch, K.R.; Cheng, R.; Heng, H.H.Q.; George, S.R.; O’Dowd, B.F. Identification and cloning of three novel human G protein-coupled receptor genes GPR52, ΨGPR53 and GPR55: GPR55 is extensively expressed in human brain. Mol. Brain Res. 1999, 64, 193–198. [Google Scholar] [CrossRef]
- Shore, D.M.; Reggio, P.H. The therapeutic potential of orphan GPCRs, GPR35 and GPR55. Front. Pharmacol. 2015, 6, 69. [Google Scholar] [CrossRef] [PubMed]
- Nilius, B.; Szallasi, A. Transient receptor potential channels as drug targets: From the science of basic research to the art of medicine. Pharmacol. Rev. 2014, 66, 676–814. [Google Scholar] [CrossRef]
- Köfalvi, A. Alternative interacting sites and novel receptors for cannabinoid ligands. In Cannabinoids and the Brain; Köfalvi, A., Ed.; Springer: Boston, MA, USA, 2008; pp. 131–160. [Google Scholar] [CrossRef]
- Pertwee, R.G.; Howlett, A.C.; Abood, M.E.; Alexander, S.P.H.; Di Marzo, V.; Elphick, M.R.; Greasley, P.J.; Hansen, H.S.; Kunos, G.; Mackie, K.; et al. International union of basic and clinical pharmacology. LXXIX. Cannabinoid receptors and their ligands: Beyond CB1 and CB2. Pharmacol. Rev. 2010, 62, 588–631. [Google Scholar] [CrossRef] [PubMed]
- Storozhuk, M.V.; Zholos, A.V. TRP channels as novel targets for endogenous ligands: Focus on endocannabinoids and nociceptive signalling. Curr. Neuropharmacol. 2018, 16, 137–150. [Google Scholar] [CrossRef]
- Whiting, P.F.; Wolff, R.F.; Deshpande, S.; Di Nisio, M.; Duffy, S.; Hernandez, A.V.; Keurentjes, J.C.; Lang, S.; Misso, K.; Ryder, S.; et al. Cannabinoids for medical use. JAMA 2015, 313, 2456. [Google Scholar] [CrossRef]
- Owens, B. Drug development: The treasure chest. Nature 2015, 525, S6–S8. [Google Scholar] [CrossRef]
- Tzadok, M.; Uliel-Siboni, S.; Linder, I.; Kramer, U.; Epstein, O.; Menascu, S.; Nissenkorn, A.; Yosef, O.B.; Hyman, E.; Granot, D.; et al. CBD-enriched medical cannabis for intractable pediatric epilepsy. Seizure 2016, 35, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.P.H. Therapeutic potential of cannabis-related drugs. Prog. Neuropsychopharmacol. Biol. Psychiatry 2016, 64, 157–166. [Google Scholar] [CrossRef] [PubMed]
- Eisenstein, M. Medical marijuana: Showdown at the cannabis corral. Nature 2015, 525, S15–S17. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A.; Capasso, R.; Aviello, G.; Borrelli, F.; Romano, B.; Piscitelli, F.; Gallo, L.; Capasso, F.; Orlando, P.; Di Marzo, V. Inhibitory effect of cannabichromene, a major non-psychotropic cannabinoid extracted from Cannabis sativa, on inflammation-induced hypermotility in mice. Br. J. Pharmacol. 2012, 166, 1444–1460. [Google Scholar] [CrossRef]
- Jadoon, K.A.; Ratcliffe, S.H.; Barrett, D.A.; Thomas, E.L.; Stott, C.; Bell, J.D.; O’Sullivan, S.E.; Tan, G.D. Efficacy and safety of cannabidiol and tetrahydrocannabivarin on glycemic and lipid parameters in patients with type 2 diabetes: A randomized, double-blind, placebo-controlled, parallel group pilot study. Diabetes Care 2016, 39, 1777–1786. [Google Scholar] [CrossRef]
- Valliere, M.A.; Korman, T.P.; Woodall, N.B.; Khitrov, G.A.; Taylor, R.E.; Baker, D.; Bowie, J.U. A cell-free platform for the prenylation of natural products and application to cannabinoid production. Nat. Commun. 2019, 10, 565. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Sanz, G. Can you pass the acid test? Critical review and novel therapeutic perspectives of Δ9-tetrahydrocannabinolic acid A. Cannabis Cannabinoid Res. 2016, 1, 124–130. [Google Scholar] [CrossRef]
- Rock, E.M.; Limebeer, C.L.; Parker, L.A. Effect of cannabidiolic acid and ∆9-tetrahydrocannabinol on carrageenan-induced hyperalgesia and edema in a rodent model of inflammatory pain. Psychopharmacology 2018, 235, 3259–3271. [Google Scholar] [CrossRef]
- McPartland, J.M.; MacDonald, C.; Young, M.; Grant, P.S.; Furkert, D.P.; Glass, M. Affinity and efficacy studies of tetrahydrocannabinolic acid A at cannabinoid receptor types one and two. Cannabis Cannabinoid Res. 2017, 2, 87–95. [Google Scholar] [CrossRef]
- Fuhr, L.; Rousseau, M.; Plauth, A.; Schroeder, F.C.; Sauer, S. Amorfrutins are natural PPARγ agonists with potent anti-inflammatory properties. J. Nat. Prod. 2015, 78, 1160–1164. [Google Scholar] [CrossRef]
- Iwata, N.; Kitanaka, S. Tetracyclic chromane derivatives from Rhododendron anthopogonoides. J. Nat. Prod. 2010, 73, 1203–1206. [Google Scholar] [CrossRef]
- Khan, B.A.; Wang, J.; Warner, P.; Wang, H. Antibacterial properties of hemp hurd powder against E. coli. J. Appl. Polym. Sci. 2014, 132, 41588. [Google Scholar] [CrossRef]
- Wahby, I.; Caba, J.M.; Ligero, F. Agrobacteriuminfection of hemp (Cannabis sativa L.): Establishment of hairy root cultures. J. Plant Interact. 2013, 8, 312–320. [Google Scholar] [CrossRef]
- Appendino, G.; Gibbons, S.; Giana, A.; Pagani, A.; Grassi, G.; Stavri, M.; Smith, E.; Rahman, M.M. Antibacterial cannabinoids from Cannabis sativa: A structure-activity study. J. Nat. Prod. 2008, 71, 1427–1430. [Google Scholar] [CrossRef]
- Degenhardt, L.; Chiu, W.-T.; Sampson, N.; Kessler, R.C.; Anthony, J.C.; Angermeyer, M.; Bruffaerts, R.; de Girolamo, G.; Gureje, O.; Huang, Y.; et al. Toward a global view of alcohol, tobacco, cannabis, and cocaine use: Findings from the WHO World Mental Health Surveys. PLoS Med. 2008, 5, e141. [Google Scholar] [CrossRef]
- Volkow, N.D.; Baler, R.D.; Compton, W.M.; Weiss, S.R.B. Adverse health effects of marijuana use. N. Engl. J. Med. 2014, 370, 2219–2227. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, A.; Lynskey, M.T.; Bucholz, K.K.; Kapoor, M.; Almasy, L.; Dick, D.M.; Edenberg, H.J.; Foroud, T.; Goate, A.; Hancock, D.B.; et al. DSM-5 cannabis use disorder: A phenotypic and genomic perspective. Drug Alcohol Depend. 2014, 134, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Hasin, D.S.; O’Brien, C.P.; Auriacombe, M.; Borges, G.; Bucholz, K.; Budney, A.; Compton, W.M.; Crowley, T.; Ling, W.; Petry, N.M.; et al. DSM-5 criteria for substance use disorders: Recommendations and rationale. Am. J. Psychiatry 2013, 170, 834–851. [Google Scholar] [CrossRef] [PubMed]
- Pierre, J.M.; Gandal, M.; Son, M. Cannabis-induced psychosis associated with high potency “wax dabs”. Schizophr. Res. 2016, 172, 211–212. [Google Scholar] [CrossRef] [PubMed]
- Ashton, C.H. Pharmacology and effects of cannabis: A brief review. Br. J. Psychiatry 2001, 178, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Rodondi, N.; Pletcher, M.J.; Liu, K.; Hulley, S.B.; Sidney, S. Marijuana use, diet, body mass index, and cardiovascular risk factors (from the CARDIA Study). Am. J. Cardiol. 2006, 98, 478–484. [Google Scholar] [CrossRef]
- Moosmann, B.; Roth, N.; Auwärter, V. Finding cannabinoids in hair does not prove cannabis consumption. Sci. Rep. 2015, 5, 14906. [Google Scholar] [CrossRef] [PubMed]
- Sewell, R.A.; Schnakenberg, A.; Elander, J.; Radhakrishnan, R.; Williams, A.; Skosnik, P.D.; Pittman, B.; Ranganathan, M.; D’Souza, D.C. Acute effects of THC on time perception in frequent and infrequent cannabis users. Psychopharmacology 2013, 226, 401–413. [Google Scholar] [CrossRef] [PubMed]
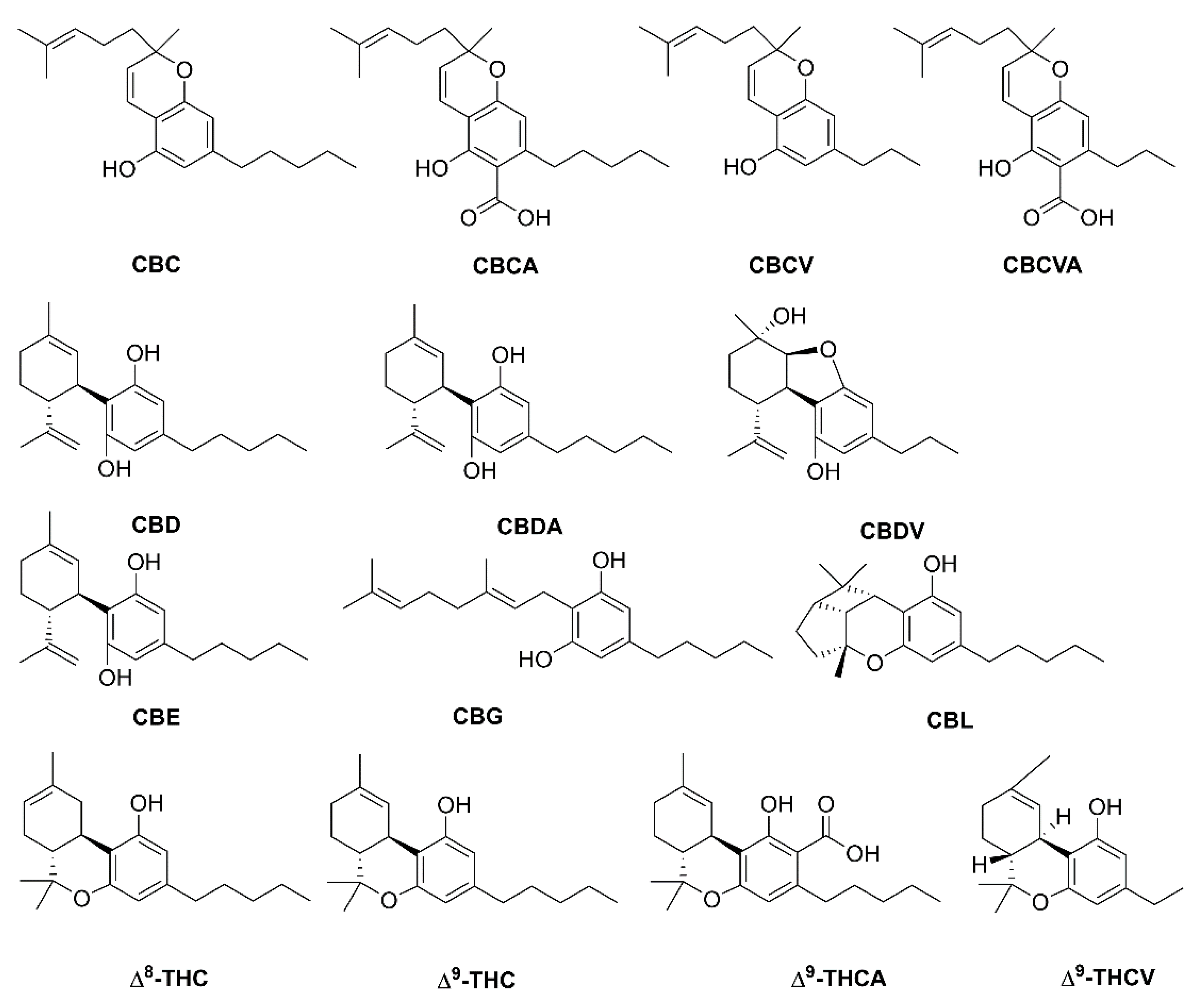
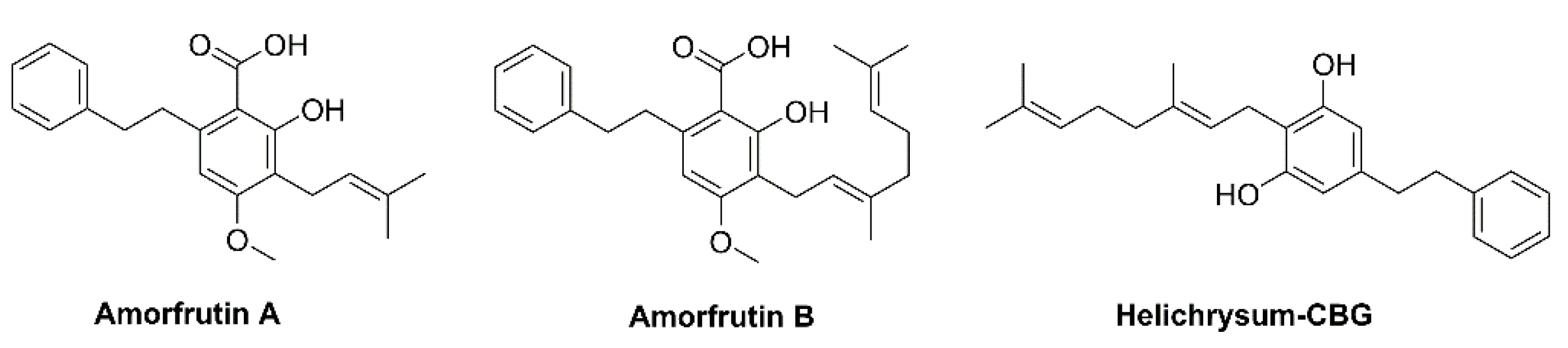
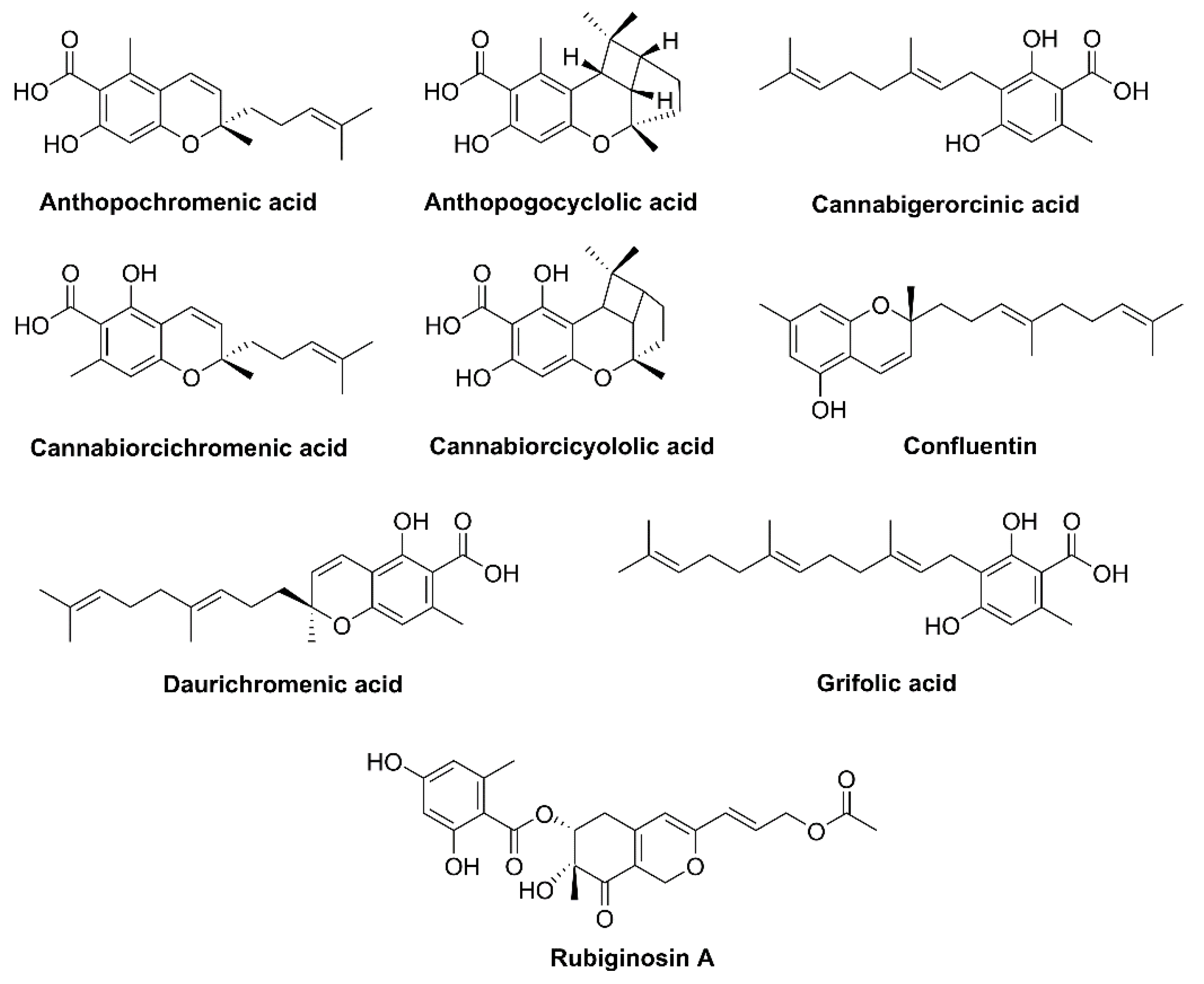
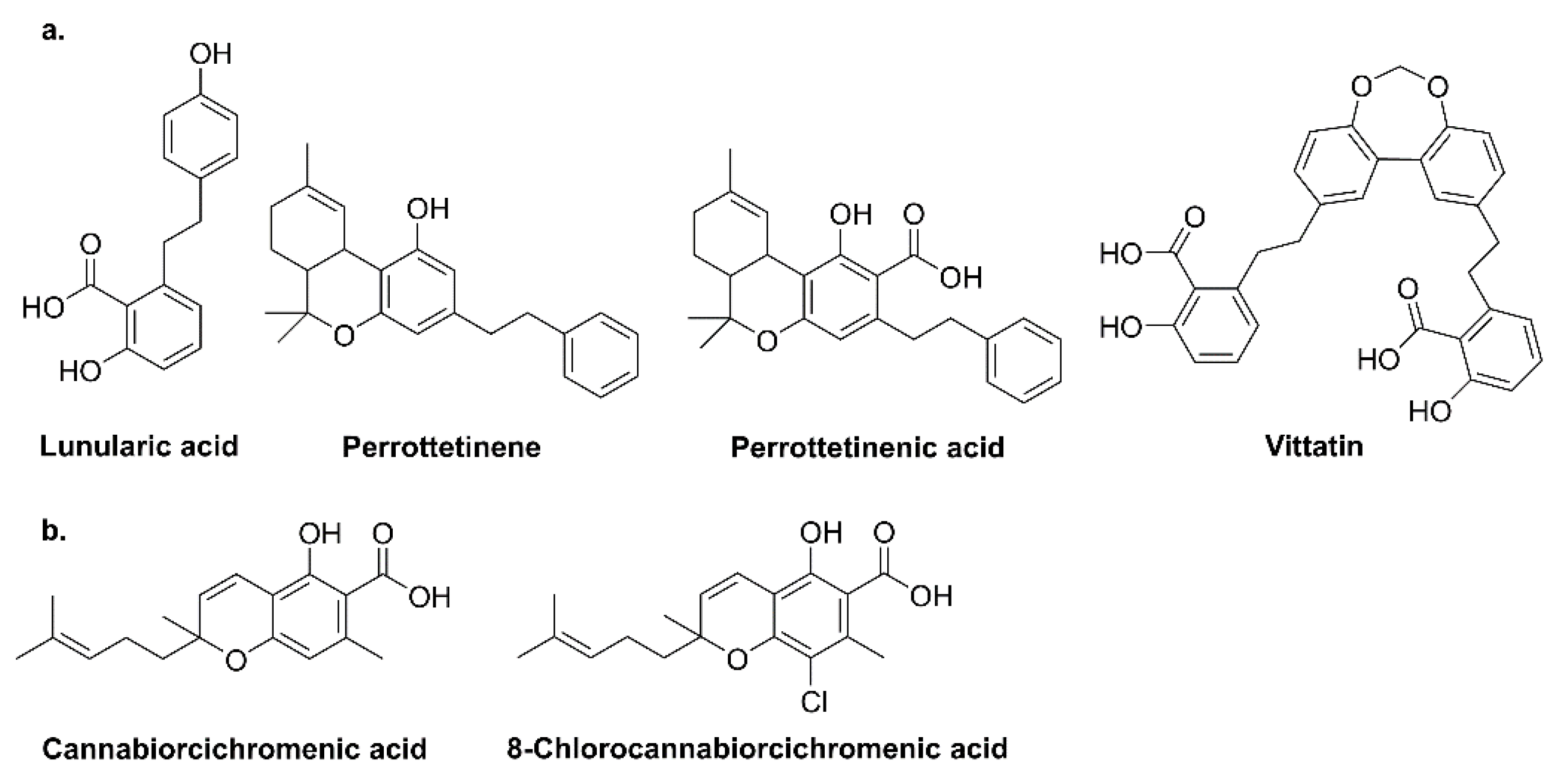
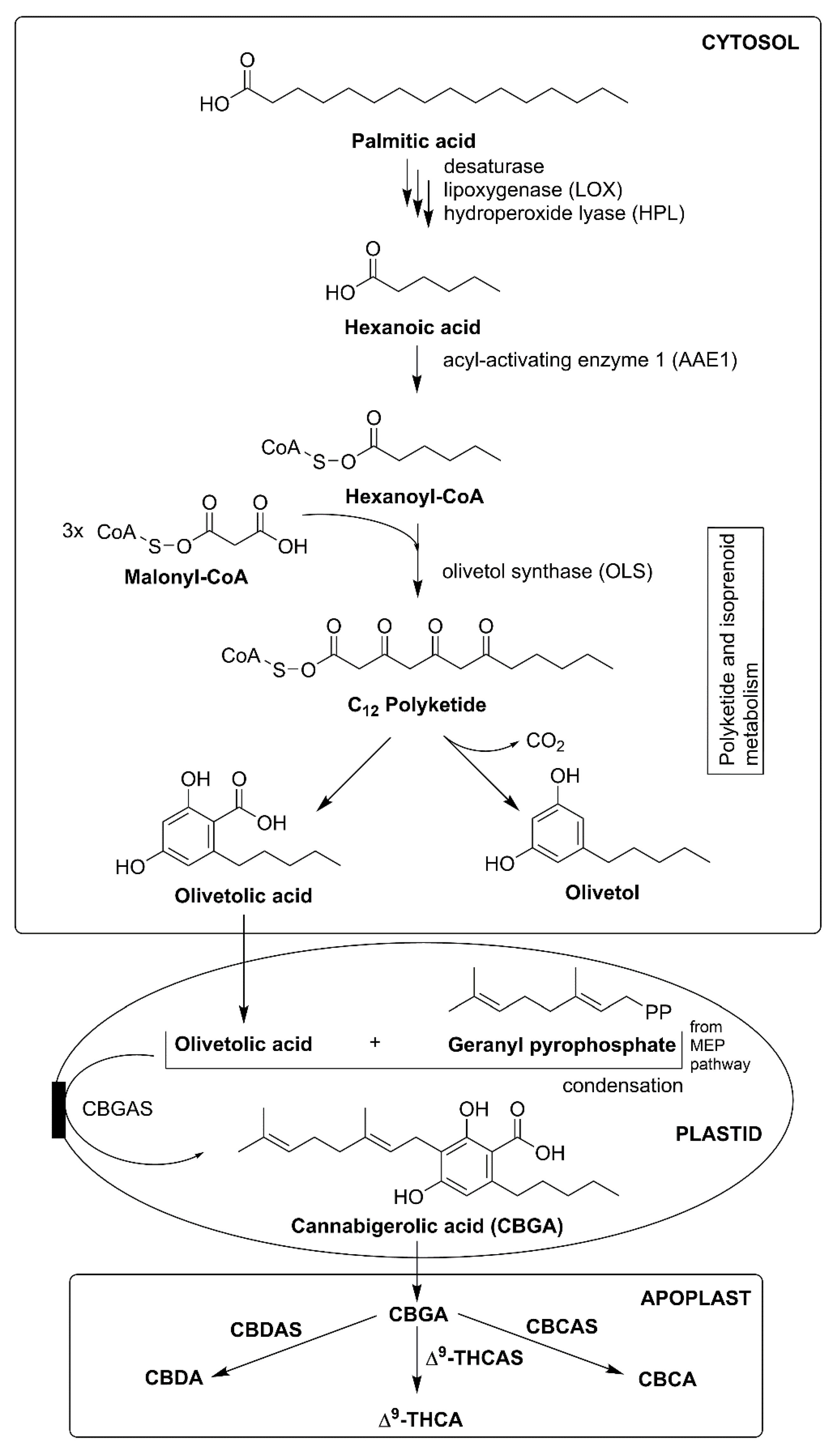
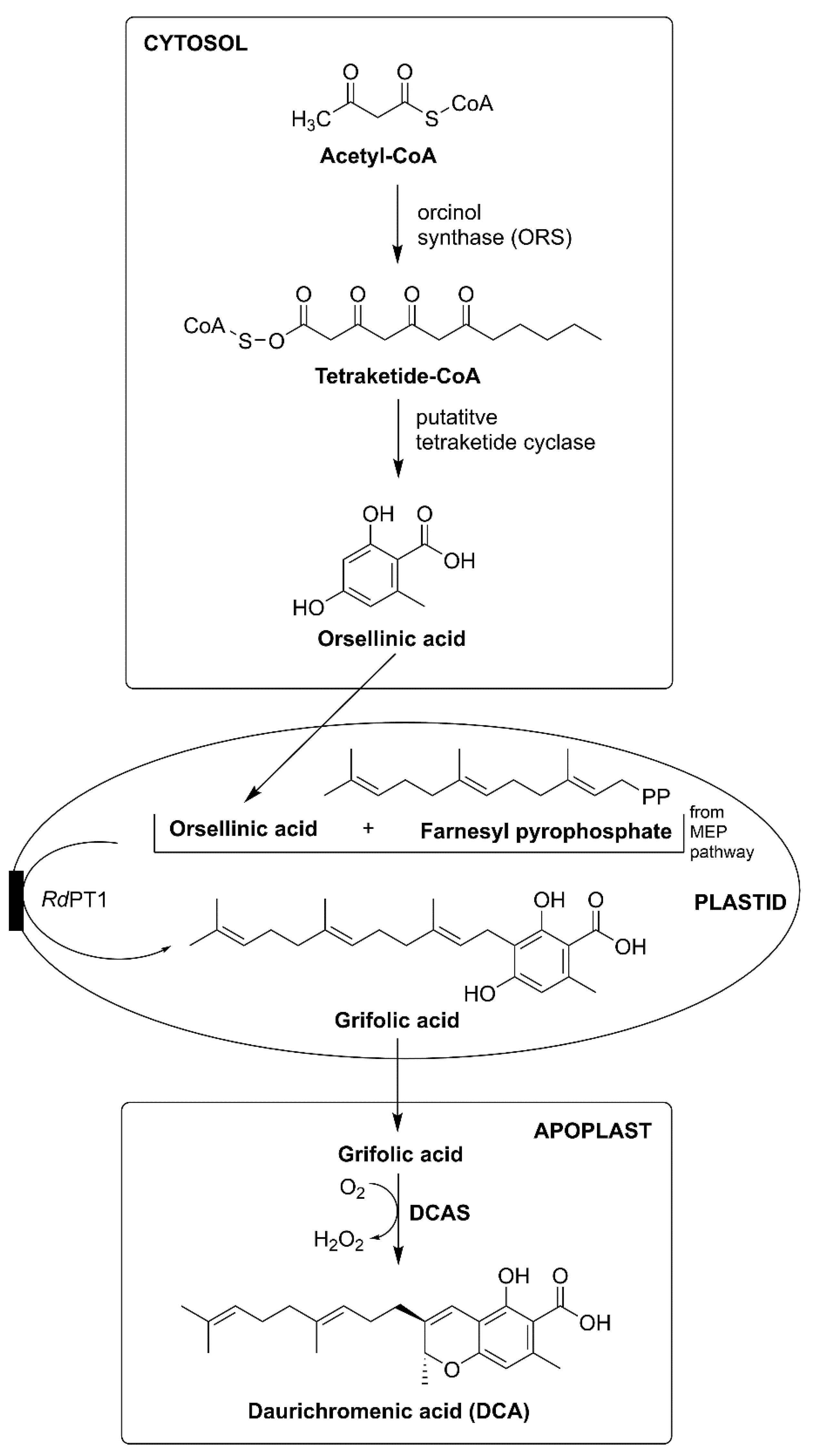
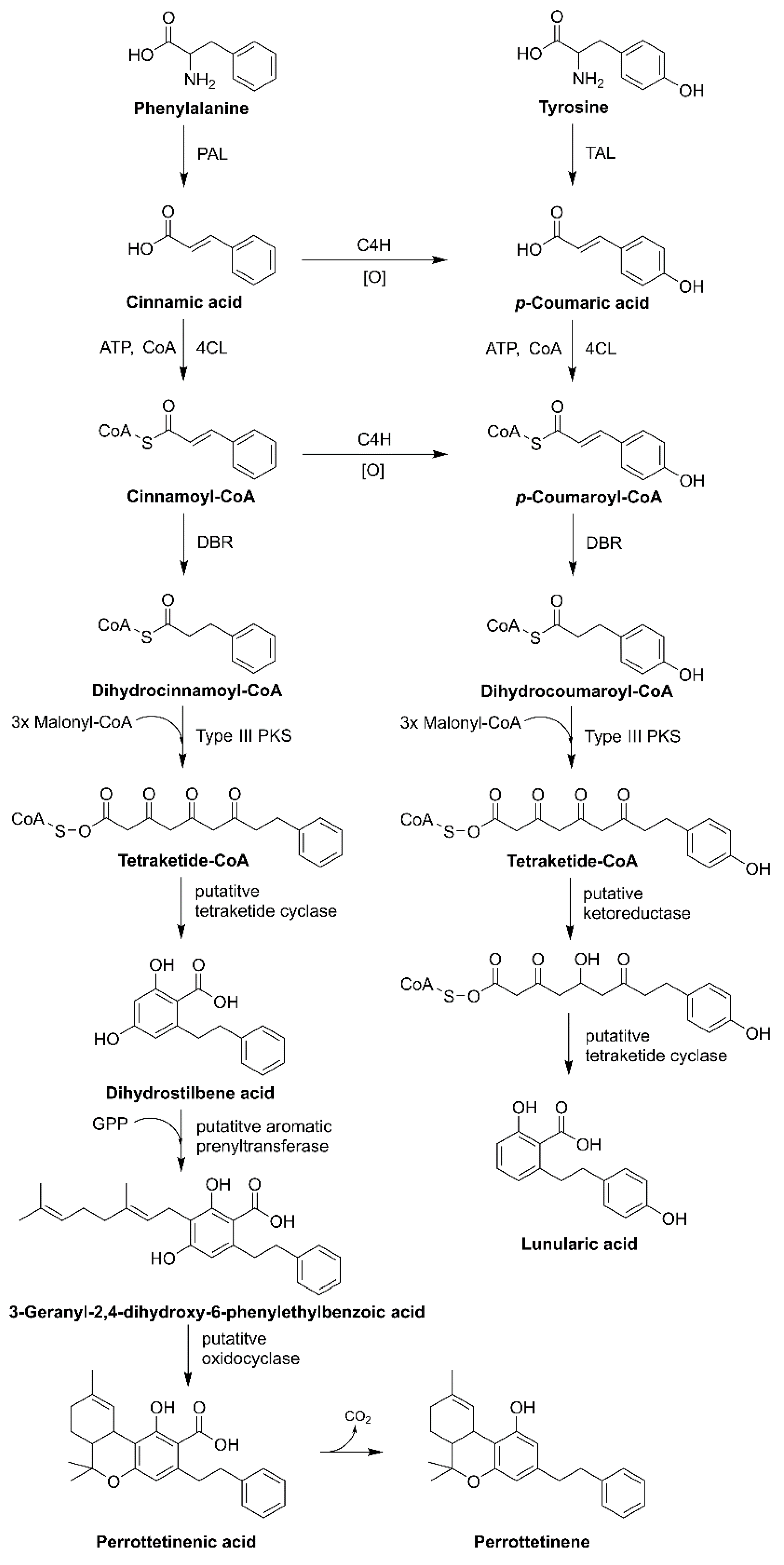
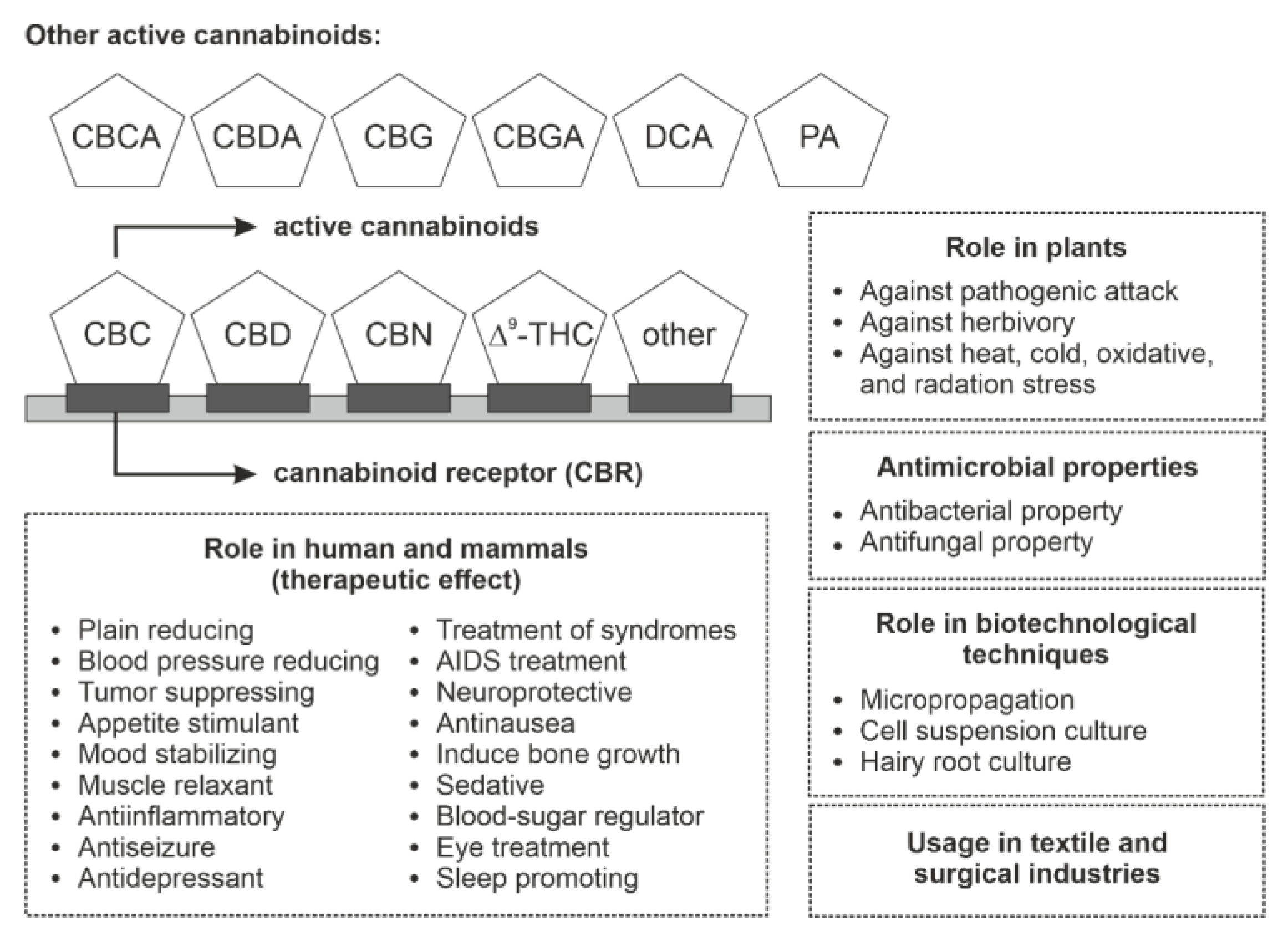
| Phytocannabinoids | Bioactivity in Animals and Human | References |
|---|---|---|
| Δ9-THC | pleiotropic effects such as analgesic, muscle relaxation, and pain tolerance | [1] |
| increases weight and appetite; improves sleep and depression; alters mood, behavior, feeling, and thoughts | [72,73] | |
| cures neuropathic pain, spasticity, dysphoria, and anxiety disorders | [74,75] | |
| antiemetic activity, i.e., prevents vomiting in cancer patient during chemotherapy | [76] | |
| displays antiglaucoma activity and reduces intraocular pressure | [76] | |
| Δ9-THCA | neuroprotective and antitumor activity | [77] |
| Δ9-THCV | non-psychoactive effect; cures obesity | [75] |
| effective against metabolic disorders, pancreatic disease, and hepatosteatosis syndrome | [1] | |
| CBC | non-psychotropic and anti-inflammatory activity | [77,78] |
| CBD | cures memory loss, obesity, convulsive disorder, and rheumatoid arthritis | [79] |
| cures epilepsy; exhibits antipsychotic, antinausea, and antianxiety activity | [80] | |
| CBDA | anti-inflammatory and antihyperalgesia effect | [81,82] |
| CBG | non-psychotic activity | [75] |
| CBC, CBL, and DCA | cures HIV and cancer; possess immune boosting activity | [83] |
| CBG and amofrutin | anti-inflammatory activity | [84] |
| CBL | anti-inflammatory, anti-microbial, antipsychotic, and antiallergic activity | [5] |
| GFA and DCA | anti-microbial activity | [85] |
| PET | increases analgesia, catalepsy, hypolocomotion and hypothermia | [28] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arif, Y.; Singh, P.; Bajguz, A.; Hayat, S. Phytocannabinoids Biosynthesis in Angiosperms, Fungi, and Liverworts and Their Versatile Role. Plants 2021, 10, 1307. https://doi.org/10.3390/plants10071307
Arif Y, Singh P, Bajguz A, Hayat S. Phytocannabinoids Biosynthesis in Angiosperms, Fungi, and Liverworts and Their Versatile Role. Plants. 2021; 10(7):1307. https://doi.org/10.3390/plants10071307
Chicago/Turabian StyleArif, Yamshi, Priyanka Singh, Andrzej Bajguz, and Shamsul Hayat. 2021. "Phytocannabinoids Biosynthesis in Angiosperms, Fungi, and Liverworts and Their Versatile Role" Plants 10, no. 7: 1307. https://doi.org/10.3390/plants10071307
APA StyleArif, Y., Singh, P., Bajguz, A., & Hayat, S. (2021). Phytocannabinoids Biosynthesis in Angiosperms, Fungi, and Liverworts and Their Versatile Role. Plants, 10(7), 1307. https://doi.org/10.3390/plants10071307








