Diverse Biosynthetic Pathways and Protective Functions against Environmental Stress of Antioxidants in Microalgae
Abstract
1. Introduction
2. Carotenoids
2.1. Carotenoid Compounds
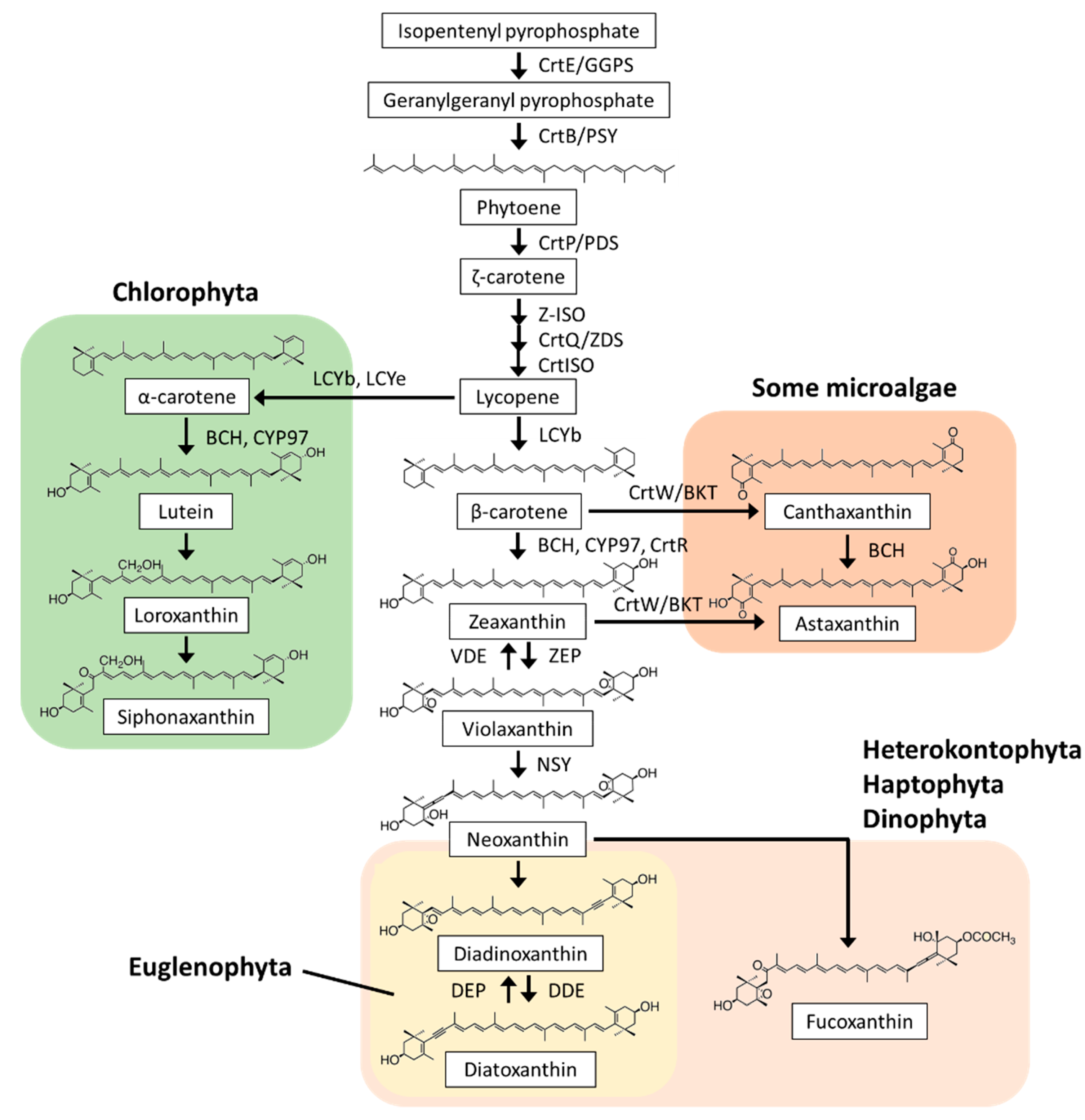
2.2. Carotenoid Biosynthesis
2.2.1. Lycopene Synthesis
2.2.2. α-Carotene and Derivatives Synthesis
2.2.3. β-Carotene and Derivatives Synthesis
2.3. Carotenoid Functions
2.3.1. Light Harvesting
2.3.2. Photoprotection
2.3.3. Xanthophyll Cycles
2.3.4. Stabilization of Lipid Membranes
2.3.5. Eyespot Formation for Phototaxis
3. Ascorbate
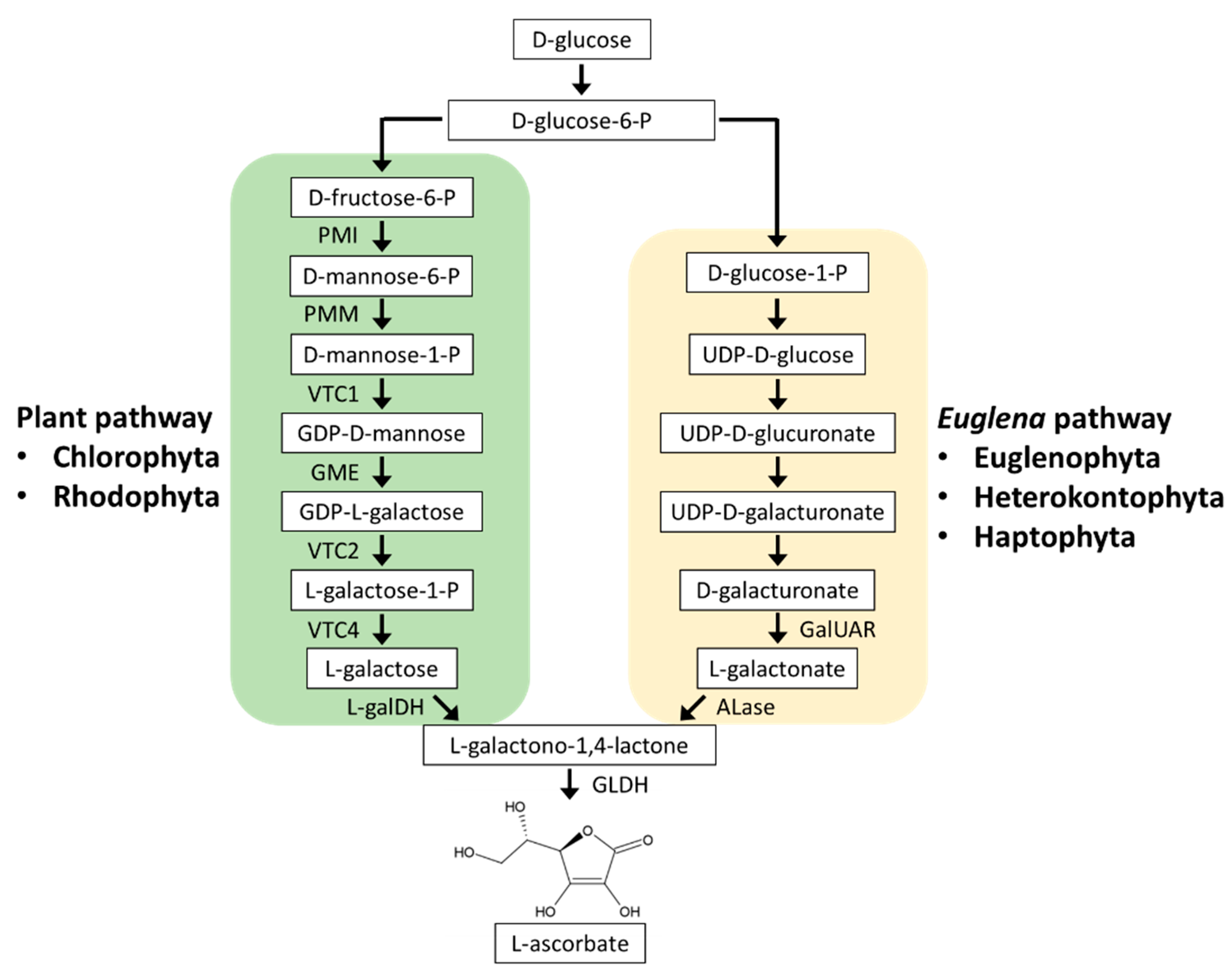
3.1. Ascorbate Biosynthesis
3.1.1. Plant Pathway
3.1.2. Euglena Pathway
3.2. Ascorbate Functions
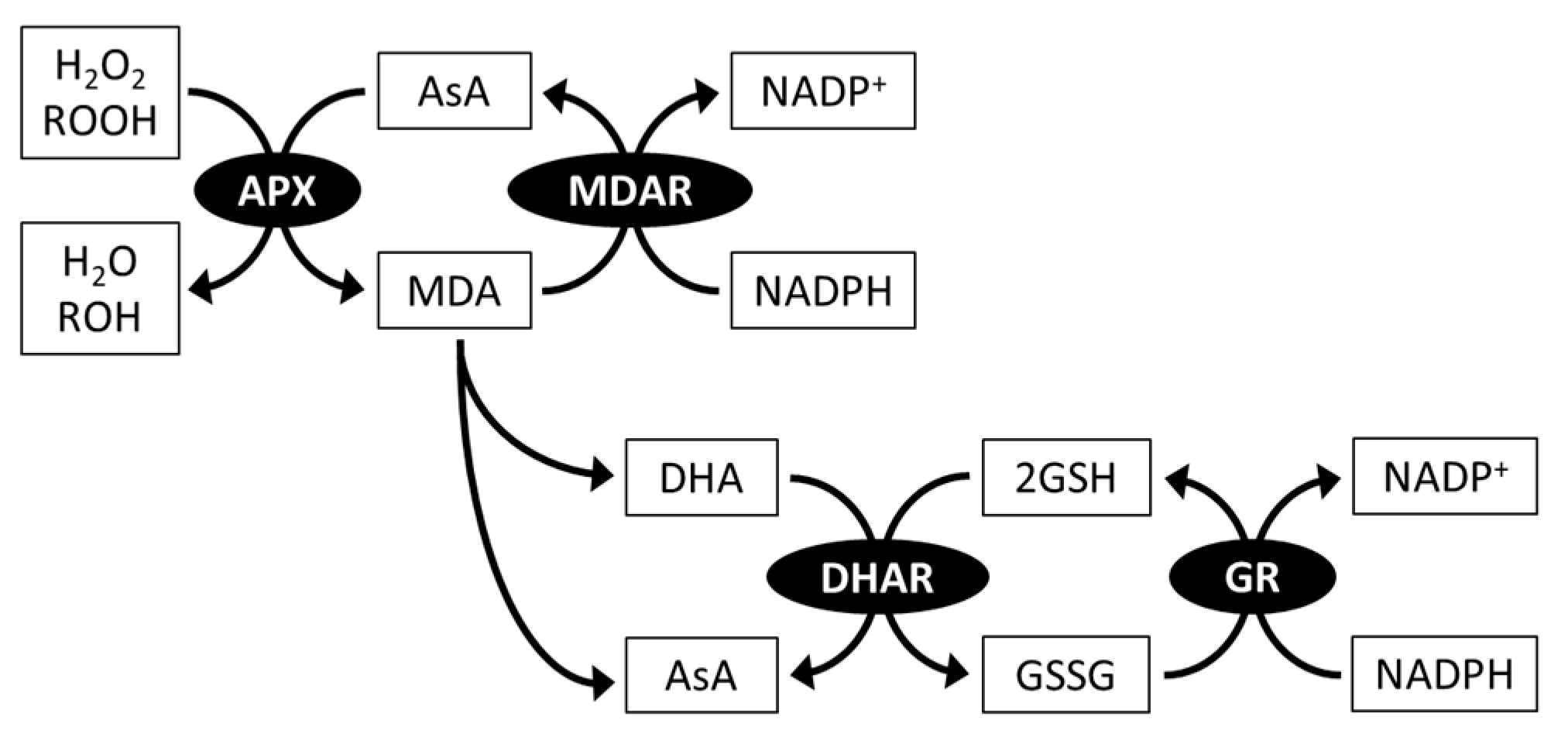
3.2.1. Ascorbate Peroxidase and the Ascorbate-regenerating System
3.2.2. Reductant for Xanthophyll Cycles
4. Glutathione
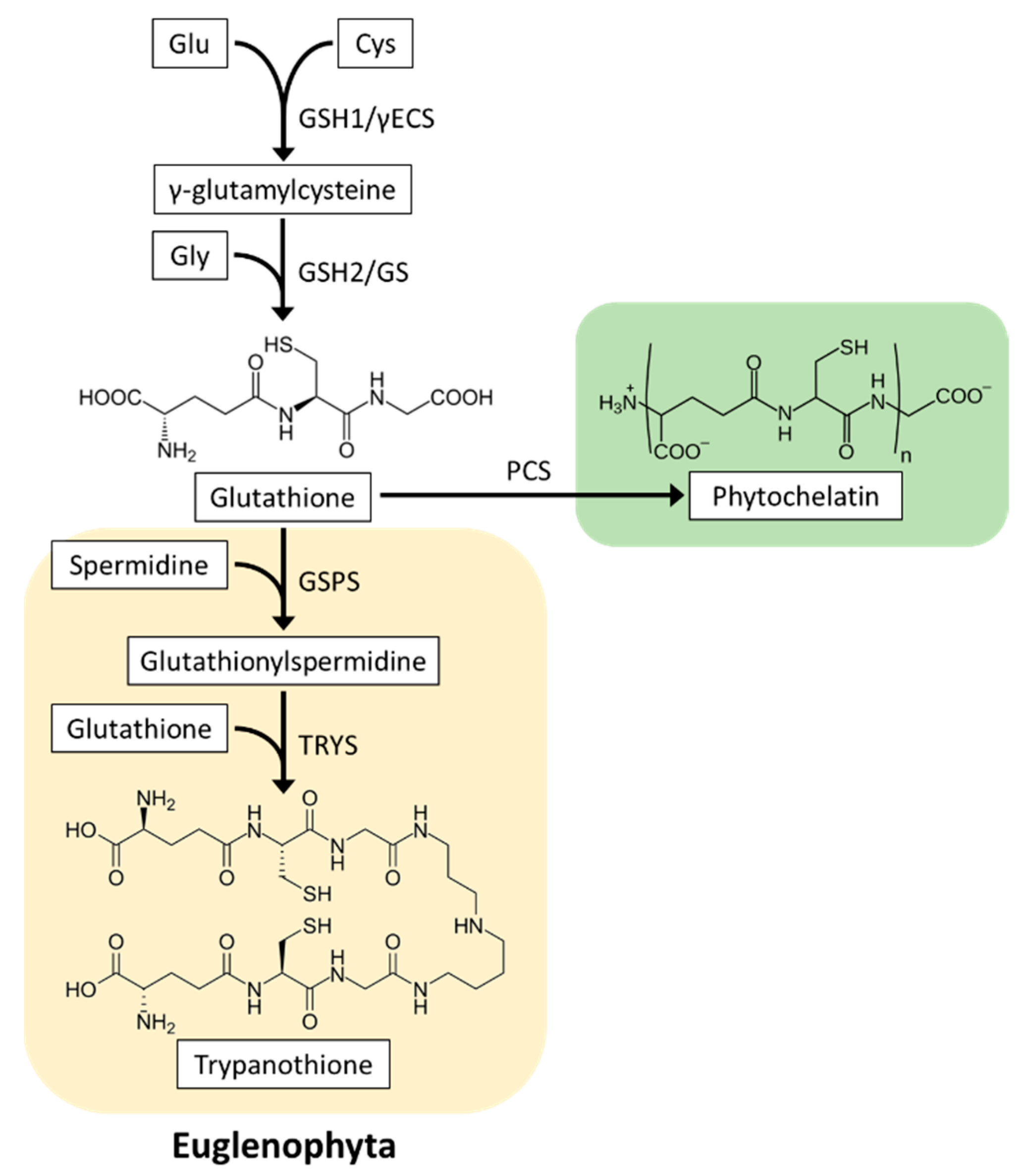
4.1. Glutathione Biosynthesis
4.2. Glutathione Functions
4.2.1. Glutathione Peroxidase
4.2.2. Ascorbate Regeneration
4.2.3. Heavy Metal Detoxification
4.2.4. Glutathione Derivative Trypanothione
4.2.5. Glutathione-Mediated Redox Regulations
5. Conclusion and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Adl, S.M.; Bass, D.; Lane, C.E.; Lukeš, J.; Schoch, C.L.; Smirnov, A.; Agatha, S.; Berney, C.; Brown, M.W.; Burki, F.; et al. Revisions to the classification, nomenclature, and diversity of eukaryotes. J. Eukaryot. Microbiol. 2019, 66, 4–119. [Google Scholar] [CrossRef] [PubMed]
- Merchant, S.S.; Prochnik, S.E.; Vallon, O.; Harris, E.H.; Karpowicz, S.J.; Witman, G.B.; Terry, A.; Salamov, A.; Fritz-Laylin, L.K.; Maréchal-Drouard, L.; et al. The Chlamydomonas genome reveals the evolution of key animal and plant functions. Science 2007, 318, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Misumi, O.; Matsuzaki, M.; Nozaki, H.; Miyagishima, S.Y.; Mori, T.; Nishida, K.; Yagisawa, F.; Yoshida, Y.; Kuroiwa, H.; Kuroiwa, T. Cyanidioschyzon merolae genome. A tool for facilitating comparable studies on organelle biogenesis in photosynthetic eukaryotes. Plant Physiol. 2005, 137, 567–585. [Google Scholar] [CrossRef]
- Camacho, F.; Macedo, A.; Malcata, F. Potential industrial applications and commercialization of microalgae in the functional food and feed industries: A short review. Mar. Drugs 2019, 17, 312. [Google Scholar] [CrossRef]
- Jerney, J.; Spilling, K. Large Scale Cultivation of Microalgae: Open and Closed Systems. In Biofuels from Algae; Spilling, K., Ed.; Springer: Cham, Switzerland, 2020; pp. 1–8. [Google Scholar]
- Harada, R.; Nomura, T.; Yamada, K.; Mochida, K.; Suzuki, K. Genetic engineering strategies for Euglena gracilis and its industrial contribution to sustainable development goals: A review. Front. Bioeng. Biotechnol. 2020, 8, 790. [Google Scholar] [CrossRef]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef]
- Smirnoff, N.; Arnaud, D. Hydrogen peroxide metabolism and functions in plants. New Phytol. 2019, 221, 1197–1214. [Google Scholar] [CrossRef] [PubMed]
- Rezayian, M.; Niknam, V.; Ebrahimzadeh, H. Oxidative damage and antioxidative system in algae. Toxicol. Rep. 2019, 6, 1309–1313. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.J.; Lin, S.; Xu, W.; Cheung, P.C.K. Occurrence and biosynthesis of carotenoids in phytoplankton. Biotechnol. Adv. 2017, 35, 597–618. [Google Scholar] [CrossRef] [PubMed]
- Takaichi, S. Carotenoids in Phototrophic Microalgae: Distributions and Biosynthesis. In Pigments from Microalgae Handbook; Jacob-Lopes, E., Queiroz, M.I., Zepka, L.Q., Eds.; Springer: Cham, Switzerland, 2020; pp. 19–42. [Google Scholar]
- Grossman, A.R.; Lohr, M.; Im, C.S. Chlamydomonas reinhardtii in the landscape of pigments. Annu. Rev. Genet. 2004, 38, 119–173. [Google Scholar] [CrossRef] [PubMed]
- Lamers, P.P.; Janssen, M.; De Vos, R.C.; Bino, R.J.; Wijffels, R.H. Exploring and exploiting carotenoid accumulation in Dunaliella salina for cell-factory applications. Trends Biotechnol. 2008, 26, 631–638. [Google Scholar] [CrossRef]
- Yang, L.E.; Huang, X.Q.; Hang, Y.; Deng, Y.Y.; Lu, Q.Q.; Lu, S. The P450-type carotene hydroxylase PuCHY1 from Porphyra suggests the evolution of carotenoid metabolism in red algae. J. Integr. Plant Biol. 2014, 56, 902–915. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, F.X., Jr.; Lee, H.; Gantt, E. Carotenoid biosynthesis in the primitive red alga Cyanidioschyzon merolae. Eukaryot. Cell 2007, 6, 533–545. [Google Scholar] [CrossRef]
- Dambek, M.; Eilers, U.; Breitenbach, J.; Steiger, S.; Büchel, C.; Sandmann, G. Biosynthesis of fucoxanthin and diadinoxanthin and function of initial pathway genes in Phaeodactylum tricornutum. J. Exp. Bot. 2012, 63, 5607–5612. [Google Scholar] [CrossRef] [PubMed]
- Garrido, J.L.; Brunet, C.; Rodríguez, F. Pigment variations in Emiliania huxleyi (CCMP370) as a response to changes in light intensity or quality. Environ. Microbiol. 2016, 18, 4412–4425. [Google Scholar] [CrossRef]
- Di Lena, G.; Casini, I.; Lucarini, M.; Lombardi-Boccia, G. Carotenoid profiling of five microalgae species from large-scale production. Food Res. Int. 2019, 120, 810–818. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Soshino, M.; Takaichi, S.; Ishikawa, T.; Nagata, N.; Asahina, M.; Shinomura, T. Suppression of the phytoene synthase gene (EgcrtB) alters carotenoid content and intracellular structure of Euglena gracilis. BMC Plant Biol. 2017, 17, 125. [Google Scholar] [CrossRef]
- Lemoine, Y.; Schoefs, B. Secondary ketocarotenoid astaxanthin biosynthesis in algae: A multifunctional response to stress. Photosynth. Res. 2010, 106, 155–177. [Google Scholar] [CrossRef]
- Tamaki, S.; Tanno, Y.; Kato, S.; Ozasa, K.; Wakazaki, M.; Sato, M.; Toyooka, K.; Maoka, T.; Ishikawa, T.; Maeda, M.; et al. Carotenoid accumulation in the eyespot apparatus required for phototaxis is independent of chloroplast development in Euglena gracilis. Plant Sci. 2020, 298, 110564. [Google Scholar] [CrossRef]
- Bertrand, M. Carotenoid biosynthesis in diatoms. Photosynth. Res. 2010, 106, 89–102. [Google Scholar] [CrossRef]
- Rodríguez-Villalón, A.; Gas, E.; Rodríguez-Concepción, M. Phytoene synthase activity controls the biosynthesis of carotenoids and the supply of their metabolic precursors in dark-grown Arabidopsis seedlings. Plant J. 2009, 60, 424–435. [Google Scholar] [CrossRef]
- Inwood, W.; Yoshihara, C.; Zalpuri, R.; Kim, K.; Kustu, S. The ultrastructure of a Chlamydomonas reinhardtii mutant strain lacking phytoene synthase resembles that of a colorless alga. Mol. Plant 2008, 1, 925–937. [Google Scholar] [CrossRef]
- Pérez-Pérez, M.E.; Couso, I.; Crespo, J.L. Carotenoid deficiency triggers autophagy in the model green alga Chlamydomonas reinhardtii. Autophagy 2012, 8, 376–388. [Google Scholar] [CrossRef] [PubMed]
- Breitenbach, J.; Sandmann, G. ζ-Carotene cis isomers as products and substrates in the plant poly-cis carotenoid biosynthetic pathway to lycopene. Planta 2005, 220, 785–793. [Google Scholar] [CrossRef]
- Cunningham, F.X., Jr.; Pogson, B.; Sun, Z.; McDonald, K.A.; DellaPenna, D.; Gantt, E. Functional analysis of the β and ε lycopene cyclase enzymes of Arabidopsis reveals a mechanism for control of cyclic carotenoid formation. Plant Cell 1996, 8, 1613–1626. [Google Scholar]
- Liang, M.; Liang, Z.; Chen, H.; Jiang, J. The bifunctional identification of both lycopene β- and ε-cyclases from the lutein-rich Dunaliella bardawil. Enzyme Microb. Technol. 2019, 131, 109426. [Google Scholar] [CrossRef]
- Blatt, A.; Bauch, M.E.; Pörschke, Y.; Lohr, M. A lycopene β-cyclase/lycopene ε-cyclase/light-harvesting complex-fusion protein from the green alga Ostreococcus lucimarinus can be modified to produce α-carotene and β-carotene at different ratios. Plant J. 2015, 82, 582–595. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Smith, J.J.; Tian, L.; Dellapenna, D. The evolution and function of carotenoid hydroxylases in Arabidopsis. Plant Cell Physiol. 2009, 50, 463–479. [Google Scholar] [CrossRef]
- Liang, M.; Xie, H.; Chen, H.; Liang, Z.; Jiang, J. Functional identification of two types of carotene hydroxylases from the green alga Dunaliella bardawil rich in lutein. ACS Synth. Biol. 2020, 9, 1246–1253. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; Yu, X.; Wang, Y.; Cui, Y.; Li, X.; Liu, Z.; Qin, S. Evolutionary origins, molecular cloning and expression of carotenoid hydroxylases in eukaryotic photosynthetic algae. BMC Genomics. 2013, 14, 457. [Google Scholar] [CrossRef]
- Cunningham, F.X., Jr.; Sun, Z.; Chamovitz, D.; Hirschberg, J.; Gantt, E. Molecular structure and enzymatic function of lycopene cyclase from the cyanobacterium (Synechococcus sp.) strain PCC7942. Plant Cell 1994, 6, 1107–1121. [Google Scholar] [PubMed]
- Linden, H. Carotenoid hydroxylase from Haematococcus pluvialis: cDNA sequence, regulation and functional complementation. Biochim. Biophys. Acta 1999, 1446, 203–212. [Google Scholar] [CrossRef]
- Cui, H.; Ma, H.; Cui, Y.; Zhu, X.; Qin, S.; Li, R. Cloning, identification and functional characterization of two cytochrome P450 carotenoids hydroxylases from the diatom Phaeodactylum tricornutum. J. Biosci. Bioeng. 2019, 128, 755–765. [Google Scholar] [CrossRef]
- Tamaki, S.; Kato, S.; Shinomura, T.; Ishikawa, T.; Imaishi, H. Physiological role of β-carotene monohydroxylase (CYP97H1) in carotenoid biosynthesis in Euglena gracilis. Plant Sci. 2019, 278, 80–87. [Google Scholar] [CrossRef]
- Ramos, A.; Coesel, S.; Marques, A.; Rodrigues, M.; Baumgartner, A.; Noronha, J.; Rauter, A.; Brenig, B.; Varela, J. Isolation and characterization of a stress-inducible Dunaliella salina Lcy-β gene encoding a functional lycopene β-cyclase. Appl. Microbiol. Biotechnol. 2008, 79, 819–828. [Google Scholar] [CrossRef]
- Dautermann, O.; Lohr, M. A functional zeaxanthin epoxidase from red algae shedding light on the evolution of light-harvesting carotenoids and the xanthophyll cycle in photosynthetic eukaryotes. Plant J. 2017, 92, 879–891. [Google Scholar] [CrossRef]
- Niyogi, K.K.; Bjorkman, O.; Grossman, A.R. Chlamydomonas xanthophyll cycle mutants identified by video imaging of chlorophyll fluorescence quenching. Plant Cell 1997, 9, 1369–1380. [Google Scholar] [CrossRef] [PubMed]
- Couso, I.; Cordero, B.F.; Vargas, M.A.; Rodríguez, H. Efficient heterologous transformation of Chlamydomonas reinhardtii npq2 mutant with the zeaxanthin epoxidase gene isolated and characterized from Chlorella zofingiensis. Mar. Drugs 2012, 10, 1955–1976. [Google Scholar] [CrossRef] [PubMed]
- Eilers, U.; Dietzel, L.; Breitenbach, J.; Büchel, C.; Sandmann, G. Identification of genes coding for functional zeaxanthin epoxidases in the diatom Phaeodactylum tricornutum. J. Plant Physiol. 2016, 192, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Leonelli, L.; Erickson, E.; Lyska, D.; Niyogi, K.K. Transient expression in Nicotiana benthamiana for rapid functional analysis of genes involved in non-photochemical quenching and carotenoid biosynthesis. Plant J. 2016, 88, 375–386. [Google Scholar] [CrossRef]
- Bouvier, F.; D’harlingue, A.; Backhaus, R.A.; Kumagai, M.H.; Camara, B. Identification of neoxanthin synthase as a carotenoid cyclase paralog. Eur. J. Biochem. 2000, 267, 6346–6352. [Google Scholar] [CrossRef]
- Kajiwara, S.; Kakizono, T.; Saito, T.; Kondo, K.; Ohtani, T.; Nishio, N.; Nagai, S.; Misawa, N. Isolation and functional identification of a novel cDNA for astaxanthin biosynthesis from Haematococcus pluvialis, and astaxanthin synthesis in Escherichia coli. Plant Mol. Biol. 1995, 29, 343–352. [Google Scholar] [CrossRef]
- Huang, J.C.; Wang, Y.; Sandmann, G.; Chen, F. Isolation and characterization of a carotenoid oxygenase gene from Chlorella zofingiensis (Chlorophyta). Appl. Microbiol. Biotechnol. 2006, 71, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Cordero, B.F.; Couso, I.; Leon, R.; Rodriguez, H.; Vargas, M.A. Isolation and characterization of a lycopene ε-cyclase gene of Chlorella (Chromochloris) zofingiensis. Regulation of the carotenogenic pathway by nitrogen and light. Mar. Drugs 2012, 10, 2069–2088. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, M.; Mao, X.; Kou, Y.; Liu, J. Time-resolved carotenoid profiling and transcriptomic analysis reveal mechanism of carotenogenesis for astaxanthin synthesis in the oleaginous green alga Chromochloris zofingiensis. Biotechnol. Biofuels 2019, 12, 287. [Google Scholar] [CrossRef]
- Mao, X.; Zhang, Y.; Wang, X.; Liu, J. Novel insights into salinity-induced lipogenesis and carotenogenesis in the oleaginous astaxanthin-producing alga Chromochloris zofingiensis: A multi-omics study. Biotechnol. Biofuels 2020, 13, 73. [Google Scholar] [CrossRef] [PubMed]
- Green, B.R.; Durnford, D.G. The chlorophyll-carotenoid proteins of oxygenic photosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1996, 47, 685–714. [Google Scholar] [CrossRef] [PubMed]
- Nagao, R.; Yokono, M.; Teshigahara, A.; Akimoto, S.; Tomo, T. Light-harvesting ability of the fucoxanthin chlorophyll a/c-binding protein associated with photosystem II from the Diatom Chaetoceros gracilis as revealed by picosecond time-resolved fluorescence spectroscopy. J. Phys. Chem. B 2014, 118, 5093–5100. [Google Scholar] [CrossRef]
- Wang, W.; Zhao, S.; Pi, X.; Kuang, T.; Sui, S.; Shen, J. Structural features of the diatom photosystem II-light-harvesting antenna complex. FEBS J. 2020, 287, 2191–2200. [Google Scholar] [CrossRef] [PubMed]
- Ogata, T.; Kodama, M.; Nomura, S.; Kobayashi, M.; Nozawa, T.; Katoh, T.; Mimuro, M. A novel peridinin-chlorophyll a protein (PCP) from the marine dinoflagellate Alexandrium cohorticula: A high pigment content and plural spectral forms of peridinin and chlorophyll a. FEBS Lett. 1994, 356, 367–371. [Google Scholar] [CrossRef]
- Schulte, T.; Johanning, S.; Hofmann, E. Structure and function of native and refolded peridinin-chlorophyll-proteins from dinoflagellates. Eur. J. Cell Biol. 2010, 89, 990–997. [Google Scholar] [CrossRef]
- Murata, N.; Nishiyama, Y. ATP is a driving force in the repair of photosystem II during photoinhibition. Plant Cell Environ. 2018, 41, 285–299. [Google Scholar] [CrossRef]
- Li, L.; Aro, E.; Millar, A.H. Mechanisms of photodamage and protein turnover in photoinhibition. Trends Plant Sci. 2018, 23, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Niyogi, K.K. Safety valves for photosynthesis. Curr. Opin. Plant Biol. 2000, 3, 455–460. [Google Scholar] [CrossRef]
- Santabarbara, S.; Casazza, A.P.; Ali, K.; Economou, C.K.; Wannathong, T.; Zito, F.; Redding, K.E.; Rappaport, F.; Purton, S. The requirement for carotenoids in the assembly and function of the photosynthetic complexes in Chlamydomonas reinhardtii. Plant Physiol. 2013, 161, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Girolomoni, L.; Bellamoli, F.; de la Cruz Valbuena, G.; Perozeni, F.; D’Andrea, C.; Cerullo, G.; Cazzaniga, S.; Ballottari, M. Evolutionary divergence of photoprotection in the green algal lineage: A plant-like violaxanthin de-epoxidase enzyme activates the xanthophyll cycle in the green alga Chlorella vulgaris modulating photoprotection. New Phytol. 2020, 228, 136–150. [Google Scholar] [CrossRef]
- Lavaud, J.; Materna, A.C.; Sturm, S.; Vugrinec, S.; Kroth, P.G. Silencing of the violaxanthin de-epoxidase gene in the diatom Phaeodactylum tricornutum reduces diatoxanthin synthesis and non-photochemical quenching. PLoS ONE 2012, 7, e36806. [Google Scholar] [CrossRef]
- Kato, S.; Tanno, Y.; Takaichi, S.; Shinomura, T. Low temperature stress alters the expression of phytoene desaturase genes (crtP1 and crtP2) and the ζ-carotene desaturase gene (crtQ) together with the cellular carotenoid content of Euglena gracilis. Plant Cell Physiol. 2019, 60, 274–284. [Google Scholar] [CrossRef] [PubMed]
- Havaux, M. Carotenoids as membrane stabilizers in chloroplasts. Trends Plant Sci. 1998, 3, 147–151. [Google Scholar] [CrossRef]
- Gruszecki, W.I.; Strzałka, K. Carotenoids as modulators of lipid membrane physical properties. Biochim. Biophys. Acta 2005, 1740, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Tardy, F.; Havaux, M. Thylakoid membrane fluidity and thermostability during the operation of the xanthophyll cycle in higher-plant chloroplasts. Biochim. Biophys. Acta 1997, 1330, 179–193. [Google Scholar] [CrossRef]
- Bojko, M.; Olchawa-Pajor, M.; Goss, R.; Schaller-Laudel, S.; Strzałka, K.; Latowski, D. Diadinoxanthin de-epoxidation as important factor in the short-term stabilization of diatom photosynthetic membranes exposed to different temperatures. Plant Cell Environ. 2019, 42, 1270–1286. [Google Scholar] [CrossRef] [PubMed]
- Kreimer, G. The green algal eyespot apparatus: A primordial visual system and more? Curr. Genet. 2009, 55, 19–43. [Google Scholar] [CrossRef] [PubMed]
- Colley, N.J.; Nilsson, D. Photoreception in phytoplankton. Integr. Comp. Biol. 2016, 56, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Ueki, N.; Ide, T.; Mochiji, S.; Kobayashi, Y.; Tokutsu, R.; Ohnishi, N.; Yamaguchi, K.; Shigenobu, S.; Tanaka, K.; Minagawa, J.; et al. Eyespot-dependent determination of the phototactic sign in Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 2016, 113, 5299–5304. [Google Scholar] [CrossRef] [PubMed]
- Kato, S.; Ozasa, K.; Maeda, M.; Tanno, Y.; Tamaki, S.; Higuchi-Takeuchi, M.; Numata, K.; Kodama, Y.; Sato, M.; Toyooka, K.; et al. Carotenoids in the eyespot apparatus are required for triggering phototaxis in Euglena gracilis. Plant J. 2020, 101, 1091–1102. [Google Scholar] [CrossRef]
- Wheeler, G.L.; Jones, M.A.; Smirnoff, N. The biosynthetic pathway of vitamin C in higher plants. Nature 1998, 393, 365–369. [Google Scholar] [CrossRef]
- Wheeler, G.; Ishikawa, T.; Pornsaksit, V.; Smirnoff, N. Evolution of alternative biosynthetic pathways for vitamin C following plastid acquisition in photosynthetic eukaryotes. Elife 2015, 4, e06369. [Google Scholar] [CrossRef]
- Bartoli, C.G.; Pastori, G.M.; Foyer, C.H. Ascorbate biosynthesis in mitochondria is linked to the electron transport chain between complexes III and IV. Plant Physiol. 2000, 123, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Yabuta, Y.; Yoshimura, K.; Takeda, T.; Shigeoka, S. Molecular characterization of tobacco mitochondrial L-galactono-γ-lactone dehydrogenase and its expression in Escherichia coli. Plant Cell Physiol. 2000, 41, 666–675. [Google Scholar] [CrossRef]
- Miyaji, T.; Kuromori, T.; Takeuchi, Y.; Yamaji, N.; Yokosho, K.; Shimazawa, A.; Sugimoto, E.; Omote, H.; Ma, J.F.; Shinozaki, K.; et al. AtPHT4;4 is a chloroplast-localized ascorbate transporter in Arabidopsis. Nat. Commun. 2015, 6, 5928. [Google Scholar] [CrossRef] [PubMed]
- Urzica, E.I.; Adler, L.N.; Page, M.D.; Linster, C.L.; Arbing, M.A.; Casero, D.; Pellegrini, M.; Merchant, S.S.; Clarke, S.G. Impact of oxidative stress on ascorbate biosynthesis in Chlamydomonas via regulation of the VTC2 gene encoding a GDP-L-galactose phosphorylase. J. Biol. Chem. 2012, 287, 14234–14245. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Meireles, A.; Neupert, J.; Zsigmond, L.; Rosado-Souza, L.; Kovács, L.; Nagy, V.; Galambos, A.; Fernie, A.R.; Bock, R.; Tóth, S.Z. Regulation of ascorbate biosynthesis in green algae has evolved to enable rapid stress-induced response via the VTC2 gene encoding GDP-l-galactose phosphorylase. New Phytol. 2017, 214, 668–681. [Google Scholar] [CrossRef] [PubMed]
- Shigeoka, S.; Nakano, Y.; Kitaoka, S. The biosynthetic pathway of L-ascorbic acid in Euglena gracilis Z. J. Nutr. Sci. Vitaminol. 1979, 25, 299–307. [Google Scholar] [CrossRef]
- Ishikawa, T.; Masumoto, I.; Iwasa, N.; Nishikawa, H.; Sawa, Y.; Shibata, H.; Nakamura, A.; Yabuta, Y.; Shigeoka, S. Functional characterization of D-galacturonic acid reductase, a key enzyme of the ascorbate biosynthesis pathway, from Euglena gracilis. Biosci. Biotechnol. Biochem. 2006, 70, 2720–2726. [Google Scholar] [CrossRef][Green Version]
- Ishikawa, T.; Nishikawa, H.; Gao, Y.; Sawa, Y.; Shibata, H.; Yabuta, Y.; Maruta, T.; Shigeoka, S. The pathway via D-galacturonate/L-galactonate is significant for ascorbate biosynthesis in Euglena gracilis: Identification and functional characterization of aldonolactonase. J. Biol. Chem. 2008, 283, 31133–31141. [Google Scholar] [CrossRef] [PubMed]
- Helsper, J.P.; Kagan, L.; Hilby, C.L.; Maynard, T.M.; Loewus, F.A. L-Ascorbic acid biosynthesis in Ochromonas danica. Plant Physiol. 1982, 69, 465–468. [Google Scholar] [CrossRef]
- Grün, M.; Loewus, F.A. L-Ascorbic-acid biosynthesis in the euryhaline diatom Cyclotella cryptica. Planta 1984, 160, 6–11. [Google Scholar] [CrossRef]
- Shigeoka, S.; Yokota, A.; Nakano, Y.; Kitaoka, S. The effect of illumination on the L-ascorbic acid content in Euglena gracilis z. Agric. Biol. Chem. 1979, 43, 2053–2058. [Google Scholar] [CrossRef]
- Smerilli, A.; Orefice, I.; Corato, F.; Olea, A.G.; Ruban, A.V.; Brunet, C. Photoprotective and antioxidant responses to light spectrum and intensity variations in the coastal diatom Skeletonema marinoi. Environ. Microbiol. 2017, 19, 611–627. [Google Scholar] [CrossRef] [PubMed]
- Shigeoka, S.; Nakano, Y.; Kitaoka, S. Purification and some properties of L-ascorbic-acid-specific peroxidase in Euglena gracilis Z. Arch. Biochem. Biophys. 1980, 201, 121–127. [Google Scholar] [CrossRef]
- Shigeoka, S.; Ishikawa, T.; Tamoi, M.; Miyagawa, Y.; Takeda, T.; Yabuta, Y.; Yoshimura, K. Regulation and function of ascorbate peroxidase isoenzymes. J. Exp. Bot. 2002, 53, 1305–1319. [Google Scholar] [CrossRef]
- Ishikawa, T.; Takeda, T.; Kohno, H.; Shigeoka, S. Molecular characterization of Euglena ascorbate peroxidase using monoclonal antibody. Biochim. Biophys. Acta 1996, 1290, 69–75. [Google Scholar] [CrossRef]
- Asada, K. The Role of Ascorbate Peroxidase and Monodehydroascorbate Reductase in H2O2 Scavenging in Plants. In Oxidative Stress and the Molecular Biology of Antioxidant Defenses; Scandalios, J.G., Ed.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 1997; pp. 715–735. [Google Scholar]
- Kuo, E.Y.H.; Cai, M.S.; Lee, T.M. Ascorbate peroxidase 4 plays a role in the tolerance of Chlamydomonas reinhardtii to photo-oxidative stress. Sci. Rep. 2020, 10, 13287. [Google Scholar] [CrossRef] [PubMed]
- Yeh, H.L.; Lin, T.H.; Chen, C.C.; Cheng, T.X.; Chang, H.Y.; Lee, T.M. Monodehydroascorbate reductase plays a role in the tolerance of Chlamydomonas reinhardtii to photooxidative stress. Plant Cell Physiol. 2019, 60, 2167–2179. [Google Scholar] [CrossRef]
- Lin, S.T.; Chiou, C.W.; Chu, Y.L.; Hsiao, Y.; Tseng, Y.F.; Chen, Y.C.; Chen, H.J.; Chang, H.Y.; Lee, T.M. Enhanced ascorbate regeneration via dehydroascorbate reductase confers tolerance to photo-oxidative stress in Chlamydomonas reinhardtii. Plant Cell Physiol. 2016, 57, 2104–2121. [Google Scholar] [CrossRef]
- Lin, T.H.; Rao, M.Y.; Lu, H.W.; Chiou, C.W.; Lin, S.T.; Chao, H.W.; Zheng, Z.L.; Cheng, H.C.; Lee, T.M. A role for glutathione reductase and glutathione in the tolerance of Chlamydomonas reinhardtii to photo-oxidative stress. Physiol. Plant. 2018, 162, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, T.; Tajima, N.; Nishikawa, H.; Gao, Y.; Madhusudhan, R.; Shibata, H.; Sawa, Y.; Shigeoka, S. Euglena gracilis ascorbate peroxidase forms an intramolecular dimeric structure: Its unique molecular characterization. Biochem. J. 2010, 426, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Shigeoka, S.; Yasumoto, R.; Onishi, T.; Nakano, Y.; Kitaoka, S. Properties of monodehydroascirbate reductase and dehydroascorbate reductase and their participation in the regeneration of ascorbate in Euglena gracilis. J. Gen. Microbiol. 1987, 133, 227–232. [Google Scholar]
- Shigeoka, S.; Onishi, T.; Nakano, Y.; Kitaoka, S. Characterization and physiological function of glutathione reductase in Euglena gracilis z. Biochem. J. 1987, 242, 511–515. [Google Scholar] [CrossRef]
- Hirooka, S.; Misumi, O.; Yoshida, M.; Mori, T.; Nishida, K.; Yagisawa, F.; Yoshida, Y.; Fujiwara, T.; Kuroiwa, H.; Kuroiwa, T. Expression of the Cyanidioschyzon merolae stromal ascorbate peroxidase in Arabidopsis thaliana enhances thermotolerance. Plant Cell Rep. 2009, 28, 188193. [Google Scholar] [CrossRef]
- Noctor, G.; Mhamdi, A.; Foyer, C.H. Oxidative stress and antioxidative systems: Recipes for successful data collection and interpretation. Plant Cell Environ. 2016, 39, 1140–1160. [Google Scholar] [CrossRef] [PubMed]
- Müller-Moulé, P.; Conklin, P.L.; Niyogi, K.K. Ascorbate deficiency can limit violaxanthin de-epoxidase activity in vivo. Plant Physiol. 2002, 128, 970–977. [Google Scholar] [CrossRef]
- Bojko, M.; Olchawa-Pajor, M.; Tuleja, U.; Kuczyńska, P.; Strzałka, W.; Latowski, D.; Strzałka, K. Expression of three diadinoxanthin de-epoxidase genes of Phaeodacylum tricornutum in Escherichia coli Origami b and BL21 strain. Acta Biochim. Pol. 2013, 60, 857–860. [Google Scholar] [CrossRef] [PubMed]
- Vidal-Meireles, A.; Tóth, D.; Kovács, L.; Neupert, J.; Tóth, S.Z. Ascorbate deficiency does not limit nonphotochemical quenching in Chlamydomonas reinhardtii. Plant Physiol. 2020, 182, 597–611. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.S.; Anjum, N.A.; Hasanuzzaman, M.; Gill, R.; Trivedi, D.K.; Ahmad, I.; Pereira, E.; Tuteja, N. Glutathione and glutathione reductase: A boon in disguise for plant abiotic stress defense operations. Plant Physiol. Biochem. 2013, 70, 204–212. [Google Scholar] [CrossRef] [PubMed]
- Vernoux, T.; Wilson, R.C.; Seeley, K.A.; Reichheld, J.P.; Muroy, S.; Brown, S.; Maughan, S.C.; Cobbett, C.S.; Van Montagu, M.; Inzé, D.; et al. The root meristemless1/cadmium sensitive2 gene defines a glutathione-dependent pathway involved in initiation and maintenance of cell division during postembryonic root development. Plant Cell 2000, 12, 97–110. [Google Scholar] [CrossRef]
- Pasternak, M.; Lim, B.; Wirtz, M.; Hell, R.; Cobbett, C.S.; Meyer, A.J. Restricting glutathione biosynthesis to the cytosol is sufficient for normal plant development. Plant J. 2008, 53, 999–1012. [Google Scholar] [CrossRef]
- Shigeoka, S.; Onishi, T.; Nakano, Y.; Kitaoka, S. Photoinduced biosynthesis of glutathione in Euglena gracilis. Agric. Biol. Chem. 1987, 51, 2257–2258. [Google Scholar] [CrossRef]
- Haghjou, M.M.; Colville, L.; Smirnoff, N. The induction of menadione stress tolerance in the marine microalga, Dunaliella viridis, through cold pretreatment and modulation of the ascorbate and glutathione pools. Plant Physiol. Biochem. 2014, 84, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Maiorino, M.; Gregolin, C.; Ursini, F. Phospholipid hydroperoxide glutathione peroxidase. Methods Enzymol. 1990, 186, 448–457. [Google Scholar] [CrossRef]
- Navrot, N.; Collin, V.; Gualberto, J.; Gelhaye, E.; Hirasawa, M.; Rey, P.; Knaff, D.B.; Issakidis, E.; Jacquot, J.P.; Rouhier, N. Plant glutathione peroxidases are functional peroxiredoxins distributed in several subcellular compartments and regulated during biotic and abiotic stresses. Plant Physiol. 2006, 142, 1364–1379. [Google Scholar] [CrossRef] [PubMed]
- Shigeoka, S.; Takeda, T.; Hanaoka, T. Characterization and immunological properties of selenium-containing glutathione peroxidase induced by selenite in Chlamydomonas reinhardtii. Biochem J. 1991, 275, 623–627. [Google Scholar] [CrossRef] [PubMed]
- Dayer, R.; Fischer, B.B.; Eggen, R.I.L.; Lemaire, S.D. The peroxiredoxin and glutathione peroxidase families in Chlamydomonas reinhardtii. Genetics 2008, 179, 41–57. [Google Scholar] [CrossRef]
- Fischer, B.B.; Dayer, R.; Schwarzenbach, Y.; Lemaire, S.D.; Behra, R.; Liedtke, A.; Eggen, R.I.L. Function and regulation of the glutathione peroxidase homologous gene GPXH/GPX5 in Chlamydomonas reinhardtii. Plant Mol. Biol. 2009, 71, 569–583. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, B.; Miao, R.; Deng, X.; Duan, Y.; Cheng, Y.; Zhang, W.; Shi, M.; Huang, K.; Xia, X.Q. Transcriptomic and physiological responses to oxidative stress in a Chlamydomonas reinhardtii glutathione peroxidase mutant. Genes 2020, 11, 463. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Xu, X. Molecular cloning and functional analyses of glutathione peroxidase homologous genes from (Chlorella sp.) NJ-18. Gene 2012, 501, 17–23. [Google Scholar] [CrossRef]
- Overbaugh, J.M.; Fall, R. Characterization of a selenium-independent glutathione peroxidase from Euglena gracilis. Plant Physiol. 1985, 77, 437–442. [Google Scholar] [CrossRef]
- Ishikawa, T.; Tamaki, S.; Maruta, T.; Shigeoka, S. Biochemistry and Physiology of Reactive Oxygen Species in Euglena. In Euglena: Biochemistry, Cell and Molecular Biology; Schwartzbach, S.D., Shigeoka, S., Eds.; Springer: Cham, Switzerland, 2017; pp. 47–64. [Google Scholar]
- Terai, Y.; Ueno, H.; Ogawa, T.; Sawa, Y.; Miyagi, A.; Kawai-Yamada, M.; Ishikawa, T.; Maruta, T. Dehydroascorbate reductases and glutathione set a threshold for high-light-induced ascorbate accumulation. Plant Physiol. 2020, 183, 112–122. [Google Scholar] [CrossRef]
- Cheng, J.; Qiu, H.; Chang, Z.; Jiang, Z.; Yin, W. The effect of cadmium on the growth and antioxidant response for freshwater algae Chlorella vulgaris. SpringerPlus 2016, 5, 1290. [Google Scholar] [CrossRef]
- Moreno-Sánchez, R.; Rodríguez-Enríquez, S.; Jasso-Chávez, R.; Saavedra, E.; García-García, J.D. Biochemistry and Physiology of Heavy Metal Resistance and Accumulation in Euglena. In Euglena: Biochemistry, Cell and Molecular Biology; Schwartzbach, S.D., Shigeoka, S., Eds.; Springer: Cham, Switzerland, 2017; pp. 91–121. [Google Scholar]
- Bräutigam, A.; Schaumlöffel, D.; Preud’Homme, H.; Thondorf, I.; Wesenberg, D. Physiological characterization of cadmium-exposed Chlamydomonas reinhardtii. Plant Cell Environ. 2011, 34, 2071–2082. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, N.; Hirayanagi, N.; Iwabe, O.; Namba, T.; Tagawa, M.; Miyamoto, S.; Miyasaka, H.; Takagi, M.; Hirata, K.; Miyamoto, K. Regulation of phytochelatin synthesis by zinc and cadmium in marine green alga, Dunaliella tertiolecta. Phytochemistry 2003, 62, 453–459. [Google Scholar] [CrossRef]
- Osaki, Y.; Shirabe, T.; Tamura, S.; Yoshimura, E. A functional putative phytochelatin synthase from the primitive red alga Cyanidioschyzon merolae. Biosci. Biotechnol. Biochem. 2008, 72, 3306–3309. [Google Scholar] [CrossRef][Green Version]
- Kawakami, S.K.; Gledhill, M.; Achterberg, E.P. Effects of metal combinations on the production of phytochelatins and glutathione by the marine diatom Phaeodactylum tricornutum. Biometals 2006, 19, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Mendoza-Cózatl, D.G.; Moreno-Sánchez, R. Cd2+ transport and storage in the chloroplast of Euglena gracilis. Biochim. Biophys. Acta 2005, 1706, 88–97. [Google Scholar] [CrossRef] [PubMed]
- Osaki, Y.; Shirabe, T.; Nakanishi, H.; Wakagi, T.; Yoshimura, E. Characterization of phytochelatin synthase produced by the primitive red alga Cyanidioschyzon merolae. Metallomics 2009, 1, 353–358. [Google Scholar] [CrossRef]
- Stoiber, T.L.; Shafer, M.M.; Armstrong, D.E. Differential effects of copper and cadmium exposure on toxicity endpoints and gene expression in Chlamydomonas reinhardtii. Environ. Toxicol. Chem. 2010, 29, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Ahner, B.A.; Wei, L.P.; Oleson, J.R.; Ogura, N. Glutathione and other low molecular weight thiols in marine phytoplankton under metal stress. Mar. Ecol. Prog. Ser. 2002, 232, 93–103. [Google Scholar] [CrossRef]
- Wu, Y.; Guo, Z.; Zhang, W.; Tan, Q.; Zhang, L.; Ge, X.; Chen, M. Quantitative relationship between cadmium uptake and the kinetics of phytochelatin induction by cadmium in a marine diatom. Sci. Rep. 2016, 6, 35935. [Google Scholar] [CrossRef]
- Mendoza-Cozatl, D.; Devars, S.; Loza-Tavera, H.; Moreno-Sánchez, R. Cadmium accumulation in the chloroplast of Euglena gracilis. Physiol. Plant. 2002, 115, 276–283. [Google Scholar] [CrossRef]
- García-García, J.D.; Girard, L.; Hernández, G.; Saavedra, E.; Pardo, J.P.; Rodríguez-Zavala, J.S.; Encalada, R.; Reyes-Prieto, A.; Mendoza-Cózatl, D.G.; Moreno-Sánchez, R. Zn-bis-glutathionate is the best co-substrate of the monomeric phytochelatin synthase from the photosynthetic heavy metal-hyperaccumulator Euglena gracilis. Metallomics 2014, 6, 604–616. [Google Scholar] [CrossRef]
- O’Neill, E.C.; Trick, M.; Hill, L.; Rejzek, M.; Dusi, R.G.; Hamilton, C.J.; Zimba, P.V.; Henrissat, B.; Field, R.A. The transcriptome of Euglena gracilis reveals unexpected metabolic capabilities for carbohydrate and natural product biochemistry. Mol. Biosyst. 2015, 11, 2808–2820. [Google Scholar] [CrossRef]
- Manta, B.; Comini, M.; Medeiros, A.; Hugo, M.; Trujillo, M.; Radi, R. Trypanothione: A unique bis-glutathionyl derivative in trypanosomatids. Biochim. Biophys. Acta 2013, 1830, 3199–3216. [Google Scholar] [CrossRef] [PubMed]
- Krauth-Siegel, R.L.; Lüdemann, H. Reduction of dehydroascorbate by trypanothione. Mol. Biochem. Parasitol. 1996, 80, 203–208. [Google Scholar] [CrossRef]
- Dumas, C.; Ouellette, M.; Tovar, J.; Cunningham, M.L.; Fairlamb, A.H.; Tamar, S.; Olivier, M.; Papadopoulou, B. Disruption of the trypanothione reductase gene of Leishmania decreases its ability to survive oxidative stress in macrophages. EMBO J. 1997, 16, 2590–2598. [Google Scholar] [CrossRef]
- Wyllie, S.; Oza, S.L.; Patterson, S.; Spinks, D.; Thompson, S.; Fairlamb, A.H. Dissecting the essentiality of the bifunctional trypanothione synthetase-amidase in Trypanosoma brucei using chemical and genetic methods. Mol. Microbiol. 2009, 74, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Montrichard, F.; Le Guen, F.; Laval-Martin, D.L.; Davioud-Charvet, E. Evidence for the co-existence of glutathione reductase and trypanothione reductase in the non-trypanosomatid Euglenozoa: Euglena gracilis Z. FEBS Lett. 1999, 442, 29–33. [Google Scholar] [CrossRef]
- Tamaki, S. Functional analysis of enzymes involved in thiol based redox metabolism in Euglena gracilis. Ph.D. Thesis, Tottori University, Tottori, Japan, 11 September 2015. [Google Scholar]
- Zaffagnini, M.; Bedhomme, M.; Groni, H.; Marchand, C.H.; Puppo, C.; Gontero, B.; Cassier-Chauvat, C.; Decottignies, P.; Lemaire, S.D. Glutathionylation in the photosynthetic model organism Chlamydomonas reinhardtii: A proteomic survey. Mol. Cell. Proteomics 2012, 11, M111.014142. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H.; Pellny, T.K.; Locato, V.; Hull, J.; De Gara, L. Analysis of Redox Relationships in the Plant Cell Cycle: Determination of Ascorbate, Glutathione, and Poly(ADPribose)Polymerase (PARP) in Plant Cell Cultures. In Redox-Mediated Signal Transduction; Hancock, J.T., Conway, M.T., Eds.; Springer: Cham, Switzerland, 2019; pp. 165–181. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tamaki, S.; Mochida, K.; Suzuki, K. Diverse Biosynthetic Pathways and Protective Functions against Environmental Stress of Antioxidants in Microalgae. Plants 2021, 10, 1250. https://doi.org/10.3390/plants10061250
Tamaki S, Mochida K, Suzuki K. Diverse Biosynthetic Pathways and Protective Functions against Environmental Stress of Antioxidants in Microalgae. Plants. 2021; 10(6):1250. https://doi.org/10.3390/plants10061250
Chicago/Turabian StyleTamaki, Shun, Keiichi Mochida, and Kengo Suzuki. 2021. "Diverse Biosynthetic Pathways and Protective Functions against Environmental Stress of Antioxidants in Microalgae" Plants 10, no. 6: 1250. https://doi.org/10.3390/plants10061250
APA StyleTamaki, S., Mochida, K., & Suzuki, K. (2021). Diverse Biosynthetic Pathways and Protective Functions against Environmental Stress of Antioxidants in Microalgae. Plants, 10(6), 1250. https://doi.org/10.3390/plants10061250







