Foliar Nourishment with Nano-Selenium Dioxide Promotes Physiology, Biochemistry, Antioxidant Defenses, and Salt Tolerance in Phaseolus vulgaris
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Layout and Growing Conditions
2.2. Foliar Application of Nano-Selenium (Se-NPs)
2.3. Sampling Date and Sample Preparation for Different Determinations
2.4. Assessment of Growth and Green Yield Traits
2.5. Evaluation of Indicators of Plant Physiology and Biochemistry
2.6. Determination of Enzyme Activities
2.7. Leaf Anatomy
2.8. Analysis of the Resulting Data
3. Results
3.1. Growth, Productivity, and Photosynthetic Efficiency Responses of Salt-Stressed Common Bean to Foliar Nourishment with Se-NPs
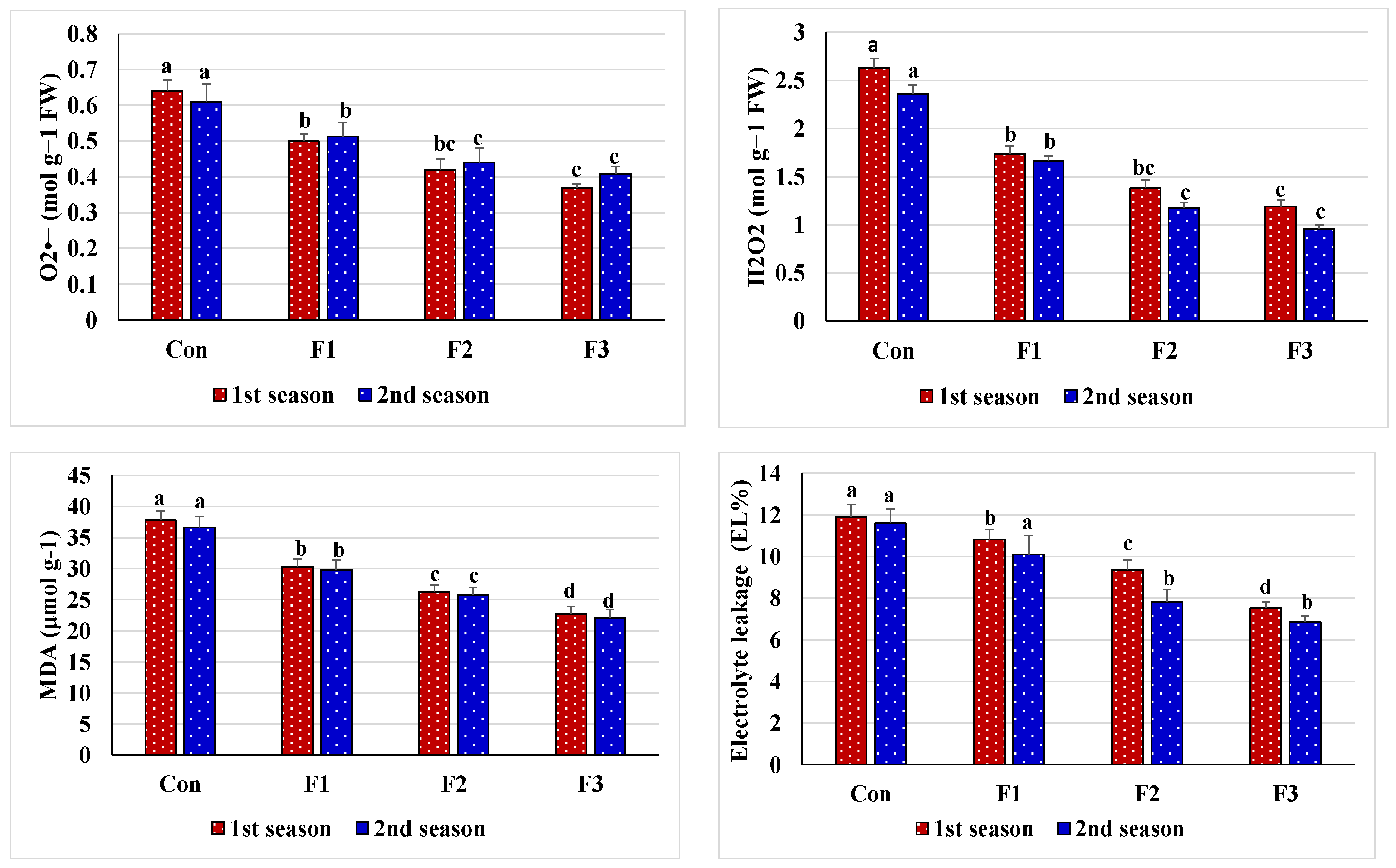
3.2. Plant Tissue Cell Integrity Response of Salt-Stressed Common Bean to Foliar Nourishment with Se-NPs
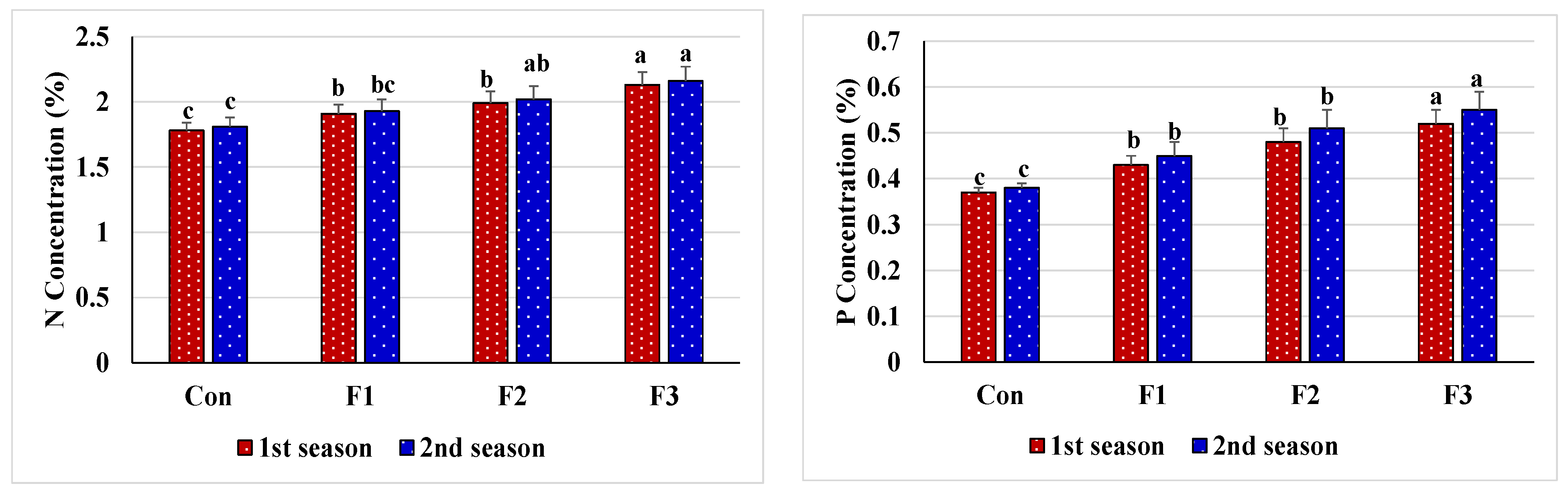
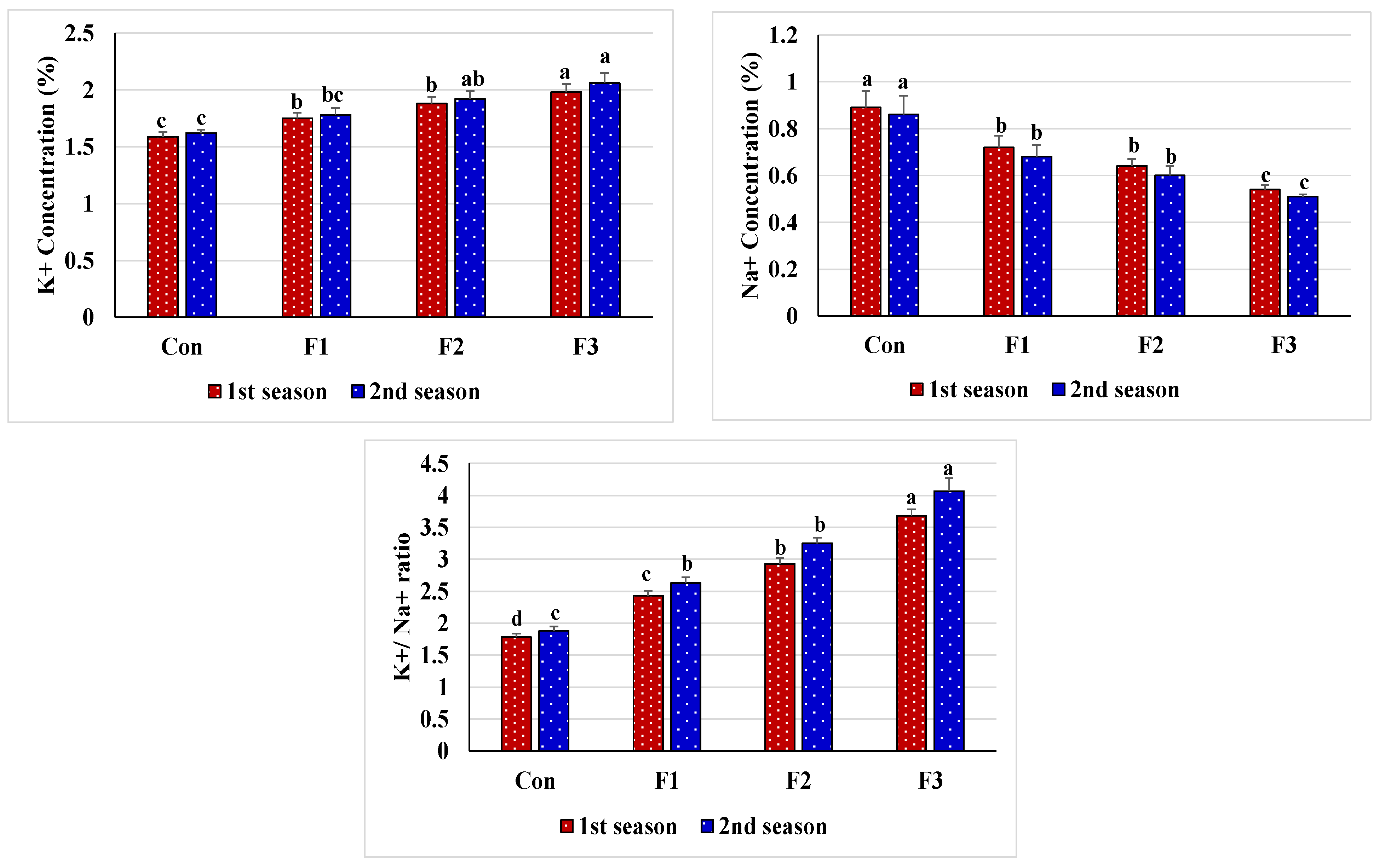
3.3. Enzyme Activity and Nutrient Content Responses of Salt-Stressed Common Bean to Foliar Nourishment with Se-NPs
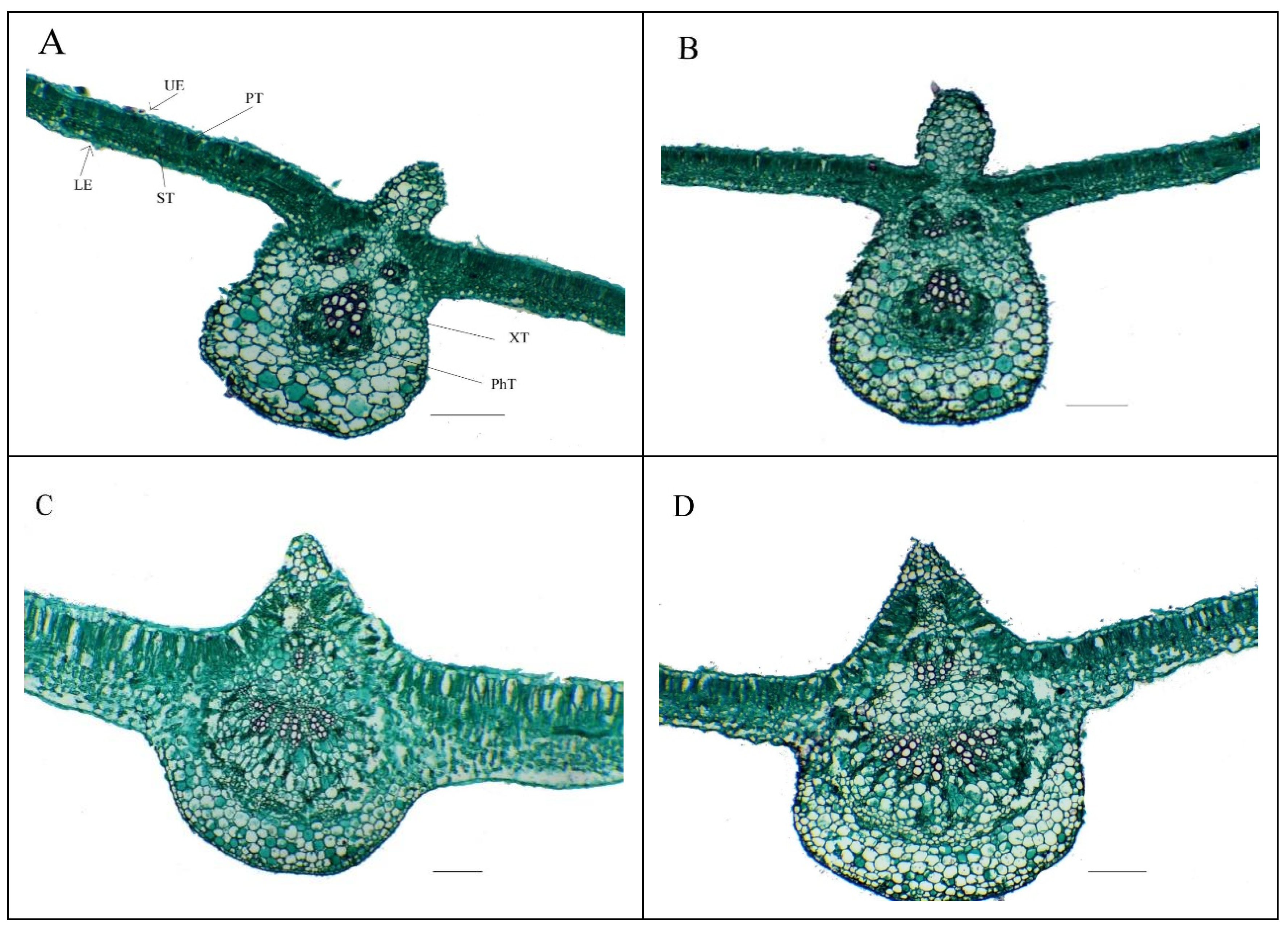
3.4. Anatomical Features Responses of Salt-Stressed Common Bean Leaf to Foliar Nourishment with Se-NPs
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Broughton, W.J.; Hernandez, G.; Blair, M.; Beebe, S.; Gepts, P.; Vanderleyden, J. Beans (Phaseolus spp.)—Model food legumes. Plant Soil 2003, 252, 55–128. [Google Scholar] [CrossRef]
- Rady, M.M.; Bhavya Varma, C.; Howladar, S.M. Common bean (Phaseolus vulgaris L.) seedlings overcome NaCl stress as a result of presoaking in Moringa oleifera leaf extract. Sci. Hortic. 2013, 162, 63–70. [Google Scholar] [CrossRef]
- Sitohy, M.Z.; Desoky, E.M.; Osman, A.; Rady, M.M. Pumpkin seed protein hydrolysate treatment alleviates salt stress effects on Phaseolus vulgaris by elevating antioxidant capacity and recovering ion homeostasis. Sci. Hortic. 2020, 271, 109495. [Google Scholar] [CrossRef]
- Mass, E.V.; Hoffman, G.J. Crop salt tolerance. Current assessment. J. Irrig. Drain. 1977, 103, 115–134. [Google Scholar] [CrossRef]
- Centro Internacional de Agricultura Tropical (CIAT). Contraints to and opportunities for improving bean production. In A Planting Document 1993–98 and Achieving Document 1987–92; CIAT: Cali, Colombia, 1992. [Google Scholar]
- Seleiman, M.F.; Semida, W.M.; Rady, M.M.; Mohamed, G.F.; Hemida, K.A.; Alhammad, B.A.; Hassan, M.M.; Shami, A. Sequential Application of Antioxidants Rectifies Ion Imbalance and Strengthens Antioxidant Systems in Salt-Stressed Cucumber. Plants 2020, 9, 1783. [Google Scholar] [CrossRef]
- ElSayed, A.I.; Boulila, M.; Rafudeen, M.S.; Mohamed, A.H.; Sengupta, S.; Rady, M.; Omar, A.A. Melatonin Regulatory Mechanisms and Phylogenetic Analyses of Melatonin Biosynthesis Related Genes Extracted from Peanut under Salinity Stress. Plants 2020, 9, 854. [Google Scholar] [CrossRef]
- Desoky, E.M.; Mansour, E.-S.; Yasin, M.A.T.; El-Sobky, E.E.A.; Rady, M.M. Improvement of drought tolerance in five different cultivars of Vicia faba with foliar application of ascorbic acid or silicon. Span. J. Agric. Res. 2020, 18, e0802. [Google Scholar] [CrossRef]
- Rady, M.M. Effect of 24-epibrassinolide on growth, yield, antioxidant system and cadmium content of bean (Phaseolus vulgaris L.) plants under salinity and cadmium stress. Sci. Hortic. 2011, 129, 232–237. [Google Scholar] [CrossRef]
- Taha, R.S.; Seleiman, M.F.; Alotaibi, M.; Alhammad, B.A.; Rady, M.M.; Mahdi, A.H.A. Exogenous Potassium Treatments Elevate Salt Tolerance and Performances of Glycine max L. by Boosting Antioxidant Defense System under Actual Saline Field Conditions. Agronomy 2020, 10, 1741. [Google Scholar] [CrossRef]
- Marschner, H. Mineral Nutrition of Higher Plants, 2nd ed.; Academic Press: London, UK, 1995; ISBN 012-473-5-436. [Google Scholar]
- Sudhir, P.; Murthy, S.D.S. Effect of salt stress on basic process of photosynthesis. Photosynthetica 2004, 42, 481–486. [Google Scholar] [CrossRef]
- Desoky, E.M.; EL-Maghraby Lamiaa, M.M.; Awad, A.E.; Abdo, A.I.; Rady, M.M.; Semida, W.M. Fennel and ammi seed extracts modulate antioxidant defence system and alleviate salinity stress in cowpea (Vigna unguiculata). Sci. Hortic. 2020, 272, 109576. [Google Scholar] [CrossRef]
- Rady, M.M.; Elrys, A.S.; Abo El-Maati, M.F.; Desoky, E.M. Interplaying roles of silicon and proline effectively improve salt and cadmium stress tolerance in Phaseolus vulgaris plant. Plant Physiol. Biochem. 2019, 139, 558–568. [Google Scholar] [CrossRef]
- Rady, M.M.; Desoky, E.M.; Elrys, A.S.; Boghdady, M.S. Can licorice root extract be used as an effective natural biostimulant for salt-stressed common bean plants? S. Afr. J. Bot. 2019, 121, 294–305. [Google Scholar] [CrossRef]
- Shardendu, S.N.; Boulyga, S.F.; Stengel, E. Phytoremediation of selenium by two helophyte species in subsurface flow constructed wetland. Chemosphere 2003, 50, 967–973. [Google Scholar] [CrossRef]
- Kamran, M.; Parveen, A.; Ahmar, S.; Hussain, S.; Chattha, M.S.; Saleem, M.H.; Adil, M.; Heidari, P.; Chen, J. An Overview of Hazardous Impacts of Soil Salinity in Crops, Tolerance Mechanisms, and Amelioration through Selenium Supplementation. Int. J. Mol. Sci. 2020, 21, 148. [Google Scholar] [CrossRef] [PubMed]
- Pennanen, A.; Xue, T.; Hartikainen, H. Protective role of selenium in plant subjected to severe UV irradiation stress. J. Appl. Bot. 2002, 76, 66–76. [Google Scholar]
- Sharma, C.S.; Nema, R.K.; Sharma, V.K. Synthesis, anticonvulsant activity and in silico study of some novel amino acids incorporated bicyclo compounds. Stamford J. Pharm. Sci. 2009, 2, 42–47. [Google Scholar] [CrossRef]
- Babajani, A.; Iranbakhsh, A.; Ardebili, Z.O.; Eslami, B. Differential growth, nutrition, physiology, and gene expression in Melissa officinalis mediated by zinc oxide and elemental selenium nanoparticles. Environ. Sci. Pollut. Res. 2019, 26, 24430–24444. [Google Scholar] [CrossRef]
- Djanaguiraman, M.; Belliraj, N.; Bossmann, S.H.; Prasad, P.V. High-temperature stress alleviation by selenium nanoparticle treatment in grain sorghum. ACS Omega 2018, 3, 2479–2491. [Google Scholar] [CrossRef]
- Zsiros, O.; Nagy, V.; Párducz, Á.; Nagy, G.; Ünnep, R.; El-Ramady, H.; Prokisch, J.; Lisztes-Szabó, Z.; Fári, M.; Csajbók, J.; et al. Effects of selenate and red Se-nanoparticles on the photosynthetic apparatus of Nicotiana tabacum. Photosynth. Res. 2019, 139, 449–460. [Google Scholar] [CrossRef] [PubMed]
- Black, C.A. Soil Plant Relationships, 2nd ed.; John Wiley and Sons: Hoboken, NJ, USA, 1968. [Google Scholar]
- Jackson, M.L. Soil Chemical Analysis; Prentice Hall, Inc.: Englewood Califfs, NJ, USA, 1973. [Google Scholar]
- Dahnke, W.C.; Whitney, D.A. Measurement of soil salinity. In Recommended Chemical Soil Test Procedures for the North Central Region, 499; North Central Regional Publication 221; Dahnke, W.C., Ed.; North Dakota Agricultural Experiment Station: Fargo, ND, USA, 1988; pp. 32–34. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts, polyphenoxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Irigoyen, J.J.; Emerich, D.W.; Sanchez-Diaz, M. Water stress induced changes in the concentrations of proline and total soluble sugars in nodulated alfalfa (Medicago sativa) plants. Plant Physiol. 1992, 8, 455–460. [Google Scholar] [CrossRef]
- Velikova, V.; Yordanov, I.; Edreva, A. Oxidative stress and some antioxidant systems in acid rain-treated bean plants. Plant Sci. 2000, 151, 59–66. [Google Scholar] [CrossRef]
- Kubis, J. Exogenous spermidine differentially alters activities of some scavenging system enzymes, H2O2 and superoxide radical levels in water-stressed cucumber leaves. J. Plant Physiol. 2008, 165, 397–406. [Google Scholar] [CrossRef] [PubMed]
- Madhava Rao, K.V.; Sresty, T.V.S. Antioxidative parameters in the seedlings of pigeonpea (Cajanus cajan (L.) Millspaugh) in response to Zn and Ni stresses. Plant Sci. 2000, 157, 113–128. [Google Scholar] [CrossRef]
- Dionisio-Sese, M.L.; Tobita, S. Antioxidant responses of rice seedlings to salinity stress. Plant Sci. 1998, 135, 1–9. [Google Scholar] [CrossRef]
- Osman, A.S.; Rady, M.M. Effect of humic acid as an additive to growing media to enhance the production of eggplant and tomato transplants. J. Hortic. Sci. Biotechnol. 2014, 89, 237–244. [Google Scholar]
- Chapman, H.D.; Pratt, F.P. Determination of Minerals by Titration Method: Methods of Analysis for Soils, Plants and Water, 2nd ed.; Agriculture Division, California University: San Diego, CA, USA, 1982; pp. 169–170. [Google Scholar]
- Watanabe, F.S.; Olsen, S.R. Test of ascorbic acid method for determine phosphorus in water and NaHCO3 extracts from soil. Soil Sci. Soc. Am. Proc. 1965, 29, 677–678. [Google Scholar] [CrossRef]
- Emilio, A.C.; Francisco, P.A.; Vicente, M.; Manuel, C.; Maria, C.B. Evaluation of salt tolerance in cultivated and wild tomato species through in vitro shoot apex culture. Plant Cell Tissue Organ Cult. 1998, 53, 19–26. [Google Scholar]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
- Thomas, R.L.; Jen, J.J.; Morr, C.V. Changes in soluble and bound peroxidase-IAA oxidase during tomato fruit development. J. Food Sci. 1982, 47, 158–161. [Google Scholar] [CrossRef]
- Nakano, Y.; Asada, K. Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 1981, 22, 867–880. [Google Scholar]
- Foster, J.G.; Hess, J.L. Responses of Superoxide Dismutase and Glutathione Reductase Activities in Cotton Leaf Tissue Exposed to an Atmosphere Enriched in Oxygen. Plant Physiol. 1980, 66, 482–487. [Google Scholar] [CrossRef]
- Bradford, M.N. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 12, 248–254. [Google Scholar] [CrossRef]
- Yu, Q.; Rengel, Z. Waterlogging influences plant growth and activities of superoxide dismutases in narrow-leafed lupin and transgenic tobacco plants. J. Plant Physiol. 1999, 155, 431–438. [Google Scholar] [CrossRef]
- Sass, J.A. Botanical Microtechnique, 3rd ed.; The Iowa State University Press: Ames, IA, USA, 1961. [Google Scholar]
- Nassar, M.A.; El-Sahhar, K.F. Botanical Preparations and Microscopy (Microtechnique); Academic Bookshop: Giza, Egypt, 1998; p. 219. (In Arabic) [Google Scholar]
- Kaydan, D.; Okut, M.Y. Effects of salicylic acid on the growth and some physiological characters in salt stressed wheat (Triticum aestivum L.). Tarim Bİlimleri Dergisi. 2007, 13, 114–119. [Google Scholar]
- Abdul Qados, A.M.S. Effects of salicylic acid on growth, yield and chemical contents of pepper (Capsicum annuum L.) plants grown under salt stress conditions. Int. J. Agric. Crop Sci. 2015, 8, 107–113. [Google Scholar]
- Tsai, Y.C.; Hong, C.Y.; Liu, L.F.; Kao, C.H. Relative importance of Na+ and Cl− in NaCl-induced antioxidant systems in roots of rice seedlings. Physiol. Plant. 2004, 122, 86–94. [Google Scholar] [CrossRef]
- Cheeseman, J. Hydrogen peroxide and plant stress: A challenging relationship. Plant Stress 2007, 1, 4–15. [Google Scholar]
- Zaki, S.S.; Rady, M.M. Moringa oleifera leaf extract improves growth, physio-chemical attributes, antioxidant defence system and yields of salt-stressed Phaseolus vulgaris L. plants. Int. J. ChemTech Res. 2015, 8, 120–134. [Google Scholar]
- Desoky, E.M.; Elrys, A.S.; Rady, M.M. Integrative moringa and licorice extracts application improves performance and reduces fruit contamination content of pepper plants grown on heavy metals-contaminated saline soil. Ecotoxicol. Environ. Saf. 2019, 169, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B.; Gutteridge, M.J.C. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Schutzendubel, A.; Polle, A. Plant responses to abiotic stresses: Heavy metal induced oxidative stress and protection by mycorrhization. J. Exp. Bot. 2002, 53, 1351–1365. [Google Scholar] [CrossRef] [PubMed]
- Semida, W.M.; Rady, M.M. Presoaking application of propolis and maize grain extracts alleviates salinity stress in common bean (Phaseolus vulgaris L.). Sci. Hortic. 2014, 68, 210–217. [Google Scholar] [CrossRef]
- Desoky, E.-S.M.; Mansour, E.; Ali, M.M.A.; Yasin, M.A.T.; Abdul-Hamid, M.I.E.; Rady, M.M.; Ali, E.F. Exogenously Used 24-Epibrassinolide Promotes Drought Tolerance in Maize Hybrids by Improving Plant and Water Productivity in an Arid Environment. Plants 2021, 10, 354. [Google Scholar] [CrossRef]
- Semida, W.M.; Abdelkhalik, A.; Mohamed, G.F.; Abd El-Mageed, T.A.; Abd El-Mageed, S.A.; Rady, M.M.; Ali, E.F. Foliar Application of Zinc Oxide Nanoparticles Promotes Drought Stress Tolerance in Eggplant (Solanum melongena L.). Plants 2021, 10, 421. [Google Scholar] [CrossRef]
- Alharby, H.F.; Al-Zahrani, H.S.; Hakeem, K.R.; Alsamadany, H.; Desoky, E.-S.M.; Rady, M.M. Silymarin-Enriched Biostimulant Foliar Application Minimizes the Toxicity of Cadmium in Maize by Suppressing Oxidative Stress and Elevating Antioxidant Gene Expression. Biomolecules 2021, 11, 465. [Google Scholar] [CrossRef]
- Alharby, H.F.; Al-Zahrani, H.S.; Alzahrani, Y.M.; Alsamadany, H.; Hakeem, K.R.; Rady, M.M. Maize Grain Extract Enriched with Polyamines Alleviates Drought Stress in Triticum aestivum through Up-Regulation of the Ascorbate-Glutathione Cycle, Glyoxalase System, and Polyamine Gene Expression. Agronomy 2021, 11, 949. [Google Scholar] [CrossRef]
- Rady, M.M.; Boriek, S.H.K.; Abd El-Mageed, T.A.; Seif El-Yazal, M.A.; Ali, E.F.; Hassan, F.A.S.; Abdelkhalik, A. Exogenous Gibberellic Acid or Dilute Bee Honey Boosts Drought Stress Tolerance in Vicia faba by Rebalancing Osmoprotectants, Antioxidants, Nutrients, and Phytohormones. Plants 2021, 10, 748. [Google Scholar] [CrossRef]
- Mekdad, A.A.A.; El-Enin, M.M.A.; Rady, M.M.; Hassan, F.A.S.; Ali, E.F.; Shaaban, A. Impact of Level of Nitrogen Fertilization and Critical Period for Weed Control in Peanut (Arachis hypogaea L.). Agronomy 2021, 11, 909. [Google Scholar] [CrossRef]
- Azzam, C.R.; Al-Taweel, S.K.; Abdel-Aziz, R.M.; Rabea, K.M.; Abou-Sreea, A.I.B.; Rady, M.M.; Ali, E.F. Salinity Effects on Gene Expression, Morphological, and Physio-Biochemical Responses of Stevia rebaudiana Bertoni In Vitro. Plants 2021, 10, 820. [Google Scholar] [CrossRef]
- Mekdad, A.A.A.; Rady, M.M.; Ali, E.F.; Hassan, F.A.S. Early Sowing Combined with Adequate Potassium and Sulfur Fertilization: Promoting Beta vulgaris (L.) Yield, Yield Quality, and K- and S-Use Efficiency in a Dry Saline Environment. Agronomy 2021, 11, 806. [Google Scholar] [CrossRef]
- Mekdad, A.A.A.; Shaaban, A.; Rady, M.M.; Ali, E.F.; Hassan, F.A.S. Integrated Application of K and Zn as an Avenue to Promote Sugar Beet Yield, Industrial Sugar Quality, and K-Use Efficiency in a Salty Semi-Arid Agro-Ecosystem. Agronomy 2021, 11, 780. [Google Scholar] [CrossRef]
- Djanaguiraman, M.; Devi, D.D.; Shanker, A.K.; Sheeba, J.A.; Bangarusamy, U. Selenium—An antioxidative protectant in soybean during senescence. Plant Soil 2005, 272, 77–86. [Google Scholar] [CrossRef]
- Iqbal, M.; Hussain, I.; Liaqat, H.; Ashraf, M.A.; Rasheed, R.; Rehman, A.U. Exogenously applied selenium reduces oxidative stress and induces heat tolerance in spring wheat. Plant Physiol. Biochem. 2015, 94, 95–103. [Google Scholar] [CrossRef]
- Jiang, C.; Zu, C.; Lu, D.; Zheng, Q.; Shen, J.; Wang, H.; Li, D. Effect of exogenous selenium supply on photosynthesis, Na+ accumulation and antioxidative capacity of maize (Zea mays L.) under salinity stress. Sci. Rep. 2017, 7, 42039. [Google Scholar] [CrossRef] [PubMed]
- Kahakachchi, C.; Boakye, H.T.; Uden, P.C.; Tyson, J.F. Chromatographic speciation of anionic and neutral selenium compounds in Se-accumulating Brassica juncea (Indian mustard) and in selenized yeast. J. Chromatogr. A. 2004, 1054, 303–312. [Google Scholar] [CrossRef]
- Jia, H.; Song, Z.; Wu, F.; Ma, M.; Li, Y.; Han, D.; Yang, Y.; Zhang, S.; Cui, H. Low selenium increases the auxin concentration and enhances tolerance to low phosphorous stress in tobacco. Environ. Exp. Bot. 2018, 153, 127–134. [Google Scholar] [CrossRef]
- Haghighi, M.; Sheibanirad, A.; Pessarakli, M. Effects of selenium as a beneficial element on growth and photosynthetic attributes of greenhouse cucumber. J. Plant Nutr. 2016, 39, 1493–1498. [Google Scholar] [CrossRef]
- Rezende, R.A.L.S.; Rodrigues, F.A.; Soares, J.D.R.; Silveira, H.R.D.O.; Pasqual, M.; Dias, G.D.M.G. Salt stress and exogenous silicon influence physiological and anatomical features of in vitro-grown cape gooseberry. Cienc. Rural. 2018, 48, e20170176. [Google Scholar] [CrossRef]
- Zahedi, S.M.; Abdelrahman, M.; Hosseini, M.S.; Hoveizeh, N.F.; Tran, L.P. Alleviation of the effect of salinity on growth and yield of strawberry by foliar spray of selenium-nanoparticles. Environ. Pollut. 2019, 253, 246–258. [Google Scholar] [CrossRef]
- Djanaguiraman, M.; Prasad, P.V.V.; Seppanen, M. Selenium protects sorghum leaves from oxidative damage under high temperature stress by enhancing antioxidant defense system. Plant Physiol. Biochem. 2010, 48, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Diao, M.; Ma, L.; Wang, J.; Cui, J.; Fu, A.; Liu, H. Selenium promotes the growth and photosynthesis of tomato seedlings under salt stress by enhancing chloroplast antioxidant defense system. J. Plant Growth Regul. 2014, 33, 671–682. [Google Scholar] [CrossRef]
- Nawaz, F.; Ashraf, M.Y.; Ahmad, R.; Waraich, E.A.; Shabbir, R.N. Selenium (Se) regulates seedling growth in wheat under drought stress. Adv. Chem. 2014, 2014, 143567. [Google Scholar] [CrossRef]
- Hawrylak-Nowak, B. Effect of selenium on selected macronutrients in maize plants. J. Elem. 2008, 13, 513–519. [Google Scholar]
- Levitt, J. Responses of Plants to Environmental Stresses, Volume 2. Water, Radiation, Salt, and Other Stresses; Academic Press: New York, NY, USA, 1980; pp. 365–380. [Google Scholar]
- Pattanayak, G.K.; Tripathy, B.C. Overexpression of protochlorophyllide oxidoreductase C regulates oxidative stress in Arabidopsis. PLoS ONE 2011, 6, e26532. [Google Scholar] [CrossRef]
- Elkelish, A.A.; Soliman, M.H.; Alhaithloul, H.A.; El-Esawi, M.A. Selenium protects wheat seedlings against salt stress-mediated oxidative damage by up-regulating antioxidants and osmolytes metabolism. Plant Physiol. Biochem. 2019, 137, 144–153. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Tomar, N.S.; Tittal, M.; Argal, S.; Agarwal, R.M. Plant growth under water/salt stress: ROS production; antioxidants and significance of added potassium under such conditions. Physiol. Mol. Biol. Plants 2017, 23, 731–744. [Google Scholar] [CrossRef]
- Khan, M.I.R.; Asgher, M.; Khan, N.A. Alleviation of salt-induced photosynthesis and growth inhibition by salicylic acid involves glycine betaine and ethylene in mung bean (Vigna radiata L.). Plant Physiol. Biochem. 2014, 80, 67–74. [Google Scholar] [CrossRef]
- Alyemeni, M.N.; Ahanger, M.A.; Wijaya, L.; Alam, P.; Bhardwaj, R.; Ahmad, P. Selenium mitigates cadmium-induced oxidative stress in tomato (Solanum lycopersicum L.) plants by modulating chlorophyll fluorescence, osmolyte accumulation, and antioxidant system. Protoplasma 2018, 255, 459–469. [Google Scholar] [CrossRef]
- Kong, L.; Wang, M.; Bi, D. Selenium modulates the activities of antioxidant enzymes, osmotic homeostasis and promotes the growth of sorrel seedlings under salt stress. Plant Growth Regul. 2005, 45, 155–163. [Google Scholar] [CrossRef]
- Nawaz, F.; Naeem, M.; Ashraf, M.Y.; Tahir, M.N.; Zulfiqar, B.; Salahuddin, M.; Shabbir, R.N.; Aslam, M. Selenium supplementation affects physiological and biochemical processes to improve fodder yield and quality of maize (Zea mays L.) under water deficit conditions. Front. Plant Sci. 2016, 7, 1438. [Google Scholar] [CrossRef]
- Saffaryazdi, A.; Lahouti, M.; Ganjeali, A.; Bayat, H. Impact of selenium supplementation on growth and selenium accumulation on spinach (Spinacia oleracea L.) plants. Not. Sci. Biol. 2012, 4, 95–102. [Google Scholar] [CrossRef]
- Rios, J.J.; Martínez-Ballesta, M.C.; Ruiz, J.M.; Blasco, B.; Carvajal, M. Silicon mediated improvement in plant salinity tolerance: The role of aquaporins. Front. Plant Sci. 2017, 8, 948. [Google Scholar] [CrossRef]
- Desoky, E.M.; ElSayed, A.I.; Merwad, A.M.A.; Rady, M.M. Stimulating antioxidant defenses, antioxidant gene expression, and salt tolerance in Pisum sativum seedling by pretreatment using licorice root extract (LRE) as an organic biostimulant. Plant Physiol. Biochem. 2019, 142, 292–302. [Google Scholar] [CrossRef]
- Stępień, P.; Kłobus, G. Water relations and photosynthesis in Cucumis sativus L. leaves under salt stress. Biol. Plant. 2006, 50, 610–616. [Google Scholar] [CrossRef]
- Trapp, S.; Feificova, D.; Rasmussen, N.F.; Bauer-Gottwein, P. Plant uptake of NaCl in relation to enzyme kinetics and toxic effects. Environ. Exp. Bot. 2008, 64, 1–7. [Google Scholar] [CrossRef]
- Ma, X.; Zhang, J.; Huang, B. Cytokinin-mitigation of salt-induced leaf senescence in perennial ryegrass involving the activation of antioxidant systems and ionic balance. Environ. Exp. Bot. 2016, 125, 1–11. [Google Scholar] [CrossRef]
- Ahanger, M.A.; Agarwal, R.M. Salinity stress induced alterations in antioxidant metabolism and nitrogen assimilation in wheat (Triticum aestivum L) as influenced by potassium supplementation. Plant Physiol. Biochem. 2017, 115, 449–460. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.Y.; Wang, X.Z.; Chen, L.; Ma, L.N.; Wang, R.Z. Physiological and biochemical responses to saline-alkaline stress in two halophytic grass species with different photosynthetic pathways. Photosynthetica 2015, 53, 128–135. [Google Scholar] [CrossRef]
- Zhu, J.K. Plant salt tolerance. Trends Plant Sci. 2001, 6, 66–71. [Google Scholar] [CrossRef]
- Howladar, S.M. A novel Moringa oleifera leaf extract can mitigate the stress effect of salinity and cadmium in bean (Phaseolus vulgaris L.) plants. Ecotoxicol. Environ. Saf. 2014, 100, 69–75. [Google Scholar] [CrossRef]
- Farhangi-Abriz, S.; Torabian, S. Antioxidant enzyme and osmotic adjustment changes in bean seedlings as affected by biochar under salt stress. Ecotoxicol. Environ. Saf. 2017, 137, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekhar, K.R.; Sandhyarani, S. Salinity induced chemical changes in Crotalaria striata DC plants. Indian J. Plant Physiol. 1996, 1, 44–48. [Google Scholar]
- Sairam, R.K.; Rao, K.V.; Srivastava, G.C. Differential response of wheat genotypes to long term salinity stress in relation to oxidative stress, antioxidant activity and osmolyte concentration. Plant Sci. 2002, 163, 1037–1046. [Google Scholar] [CrossRef]
- Hernández, J.A.; Almansa, M.S. Short-term effects of salt stress on antioxidant systems and leaf water relations of pea leaves. Physiol. Plant. 2002, 115, 251–257. [Google Scholar] [CrossRef]
- Allen, D.J.; Kee, I.F.M.; Farage, P.K.; Baker, N.R. Analysis of the limitation to CO2 assimilation to exposure of leaves of two Brassica napus cultivars to UV-B. Plant Cell Environ. 1997, 20, 633–640. [Google Scholar] [CrossRef]
- Jiang, Y.; Huang, N. Drought and Heat stress injury to two cool season turf grasses in relation to antioxidant metabolism and lipid peroxidation. Crop Sci. 2001, 41, 436–442. [Google Scholar] [CrossRef]
- Alireza, Y.; Aboueshaghi, R.S.; Dehnavi, M.M.; Balouchi, H. Effect of micronutrients foliar application on grain qualitative characteristics and some physiological traits of bean (Phaseolus vulgaris L.) under drought stress. Ind. J. Fund. Appl. Life Sci. 2014, 4, 124–131. [Google Scholar]
- Hernández, J.A.; Francisco, F.J.; Corpas, G.M.; Gómez, L.A.; Del Río, F.S. Salt induced oxidative stresses mediated by activated oxygen species in pea leaves mitochondria. Plant Physiol. 1993, 89, 103–110. [Google Scholar] [CrossRef]
- Alscher, R.G.; Erturk, N.; Heath, L.S. Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J. Exp. Biol. 2002, 53, 1331–1341. [Google Scholar]
- Feierabend, J. Catalases in plants: Molecular and functional properties and role in stress defense. In Antioxidants and Reactives Oxygen Species in Plants; Smirnoff, N., Ed.; Blackwell Publishing: London, UK, 2005; pp. 101–140. [Google Scholar]
- Foyer, C.H. Free radical processes in plants. Biochem. Soc. Transact. 1996, 24, 427–434. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Redox Regulation in Photosynthetic Organisms: Signaling: Acclimation and Practical Implications. Trends Plant Sci. 2009, 6, 486–492. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, L.; Liu, Y.; Zhang, Q.; Wei, Q.; Zhang, W. Nitric oxide enhances salt tolerance in maize seedlings through increasing activities of proton-pump and Na+/H+ antiport in the tonoplast. Planta 2006, 224, 545–555. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, P.; Sarwat, M.; Bhat, N.A.; Wani, M.R.; Kazi, A.G.; Tran, L.S. Alleviation of cadmium toxicity in Brassica juncea L. (Czern. & Coss.) by calcium application involves various physiological and biochemical strategies. PLoS ONE 2015, 10, e0114571. [Google Scholar]
- Drahonovsky, J.; Szákova, J.; Mestek, O.; Tremlova, J.; Kanab, A.; Najmanova, J.; Tlustoa, P. Selenium uptake, transformation and inter-element interactions by selected wildlife plant species after foliar selenate application. Environ. Exp. Bot. 2016, 125, 12–19. [Google Scholar] [CrossRef]
- Astaneh, R.K.; Bolandnazar, S.; Nahandi, F.Z.; Oustan, F. The effects of selenium on some physiological traits and K, Na concentration of garlic (Allium sativum L.) under NaCl stress. Inf. Process Agric. 2018, 5, 156–161. [Google Scholar] [CrossRef]
- Shabala, S.; Pottosin, I. Regulation of potassium transport in plants under hostile conditions: Implications for abiotic and biotic stress tolerance. Physiol. Plant. 2014, 151, 257–279. [Google Scholar] [CrossRef]
- Gupta, M.; Gupta, S. An overview of selenium uptake, metabolism, and toxicity in plants. Front. Plant Sci. 2016, 7, 2074. [Google Scholar]
- Jiang, C.; Zheng, Q.; Liu, Z.; Xu, W.; Liu, L.; Zhao, G.; Long, X. Overexpression of Arabidopsis thaliana Na+/H+ antiporter gene enhanced salt resistance in transgenic poplar (Populus × euramericana “Neva”). Trees 2012, 26, 685–694. [Google Scholar] [CrossRef]
- Almeida, D.M.; Gregorio, G.B.; Oliveira, M.M.; Saibo, N.J.M. Five novel transcription factors as potential regulators of OsNHX1 gene expression in a salt tolerant rice genotype. Plant Mol. Biol. 2017, 93, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Feng, R.; Wei, C.; Tu, S. The roles of selenium in protecting plants against abiotic stresses. Environ. Exp. Bot. 2013, 87, 58–68. [Google Scholar] [CrossRef]
- Rady, M.O.A.; Semida, W.M.; Howladar, S.M.; Abd El-Mageed, T.A. Raised beds modulate physiological responses, yield and water use efficiency of wheat (Triticum aestivum L) under deficit irrigation. Agric. Water Manag. 2021, 245, 106629. [Google Scholar] [CrossRef]
- Khattab, H.I.; Emam, M.A.; Emam, M.M.; Helal, N.M.; Mohamed, M.R. Effect of selenium and silicon on transcription factors NAC5 and DREB2A involved in drought-responsive gene expression in rice. Biol. Plant. 2014, 58, 265–273. [Google Scholar] [CrossRef]
- Al-Elwany, O.A.; Mohamed, G.F.; Abdehrahman, H.A.; Rady, M.M.; Abdel Latef, A.H. Exogenous glutathi-one-mediated tolerance to deficit irrigation stress in salt-affected Capsicum frutescence (L.) plants is connected with higher antioxidant content and proper ion homeostasis. Not. Bot. Horti Agrobot. Cluj Napoca 2020, 48, 1957–1979. [Google Scholar] [CrossRef]
- Hussein, H.A.A.; Darwesh, O.M.; Mekki, B.B.; El-Hallouty, S.M. Evaluation of cytotoxicity, biochemical profile and yield components of groundnut plants treated with nano-selenium. Biotechnol. Rep. 2019, 24, e00377. [Google Scholar] [CrossRef]

| Soil Characteristics | Unit | Values | |
|---|---|---|---|
| First Season | Second Season | ||
| Sand | % | 44.2 | 44.7 |
| Silt | 31.7 | 30.8 | |
| Clay | 24.1 | 24.5 | |
| Texture class | Loam | ||
| Field capacity | % | 16.2 | 16.3 |
| CaCO3 | g kg−1 | 60.1 | 62.3 |
| Organic matter | 7.55 | 8.25 | |
| pH (in soil paste) | 7.52 | 7.61 | |
| EC (in soil paste extract) | dS m−1 | 7.55 | 7.61 |
| Soluble ions (anions and cations) ** | |||
| Mg2+ | mmolc L−1 | 19.9 | 20.1 |
| Ca2+ | 15.6 | 15.7 | |
| SO42– | 8.19 | 8.4 | |
| K+ | 6.72 | 6.53 | |
| HCO3− | 20.9 | 20.7 | |
| CO32– | - | - | |
| Na+ | 19.1 | 19.5 | |
| Cl− | 31.7 | 32.7 | |
| Available nutrient | |||
| N | mg kg−1 soil | 59.0 | 59.4 |
| P | 9.80 | 9.90 | |
| K | 98.7 | 99.5 | |
| Se | 0.06 | 0.08 | |
| The Property | The Unit | The Value |
|---|---|---|
| Diameter | nm | 80 |
| Surface area | m2 g−1 | 30−50 |
| Density | g cm−3 | 3.89 |
| Purity | % | 99.5 |
| Morphology | Spherical | |
| Foliar Spray | ShL (cm) | NoL-P | LeA-P (cm2) | DW-Sh (g) | NoP-P | GPY-H (ton ha−1) |
|---|---|---|---|---|---|---|
| The first season (2019) | ||||||
| Distilled water | 16.1 ± 1.1 d | 6.87 ± 0.4 b | 7.26 ± 0.3 c | 3.61 ± 0.1 d | 10.6 ± 0.9 d | 2.17 ± 0.19 c |
| Se-NPs (0.5 mM) | 21.5 ± 1.4 c | 8.24 ± 0.6 b | 7.55 ± 0.4 c | 6.17 ± 0.3 c | 12.6 ± 1.1 c | 4.33 ± 0.21 bc |
| Se-NPs (1 mM) | 32.3 ± 2.1 a | 10.7 ± 0.8 a | 9.25 ± 0.6 a | 9.31 ± 0.6 a | 18.8 ± 1.5 a | 7.88 ± 0.26 a |
| Se-NPs (1.5 mM) | 26.1 ± 1.6 b | 9.82 ± 0.4 a | 8.65 ± 0.5 b | 7.64 ± 0.4 b | 14.9 ± 1.3 b | 6.74 ± 0.19 ab |
| The second season (2020) | ||||||
| Distilled water | 16.9 ± 1.3 d | 7.77 ± 0.3 c | 7.76 ± 0.2 d | 4.11 ± 0.2 c | 11.1 ± 0.8 d | 2.21 ± 0.12 c |
| Se-NPs (0.5 mM) | 22.4 ± 1.5 c | 9.21 ± 0.5 b | 8.29 ± 0.4 c | 6.39 ± 0.3 b | 13.7 ± 1.2 c | 4.40 ± 0.14 bc |
| Se-NPs (1 mM) | 33.6 ± 2.5 a | 12.0 ± 0.7 a | 10.0 ± 0.8 a | 9.39 ± 0.6 a | 19.9 ± 1.2 a | 7.95 ± 0.31 a |
| Se-NPs (1.5 mM) | 27.2 ± 2.3 b | 10.9 ± 0.6 a | 9.35 ± 0.6 b | 7.71 ± 0.5 b | 15.8 ± 1.3 b | 6.81 ± 0.21 ab |
| Foliar Spray | Chlorophyll “a” | Chlorophyll “b” | Carotenoids | Pn (µmol CO2 m−2 s−1) | Tr (mmol H2 O m−2 s−1) |
|---|---|---|---|---|---|
| (mg g−1 FW) | |||||
| The first season (2019) | |||||
| Distilled water | 0.94 ± 0.07 d | 0.53 ± 0.02 c | 0.85 ± 0.03 c | 5.56 ± 0.2 d | 3.18 ± 0.1 d |
| Se-NPs (0.5 mM) | 1.11 ± 0.08 c | 0.62 ± 0.03 b | 0.98 ± 0.04 bc | 9.77 ± 0.4 c | 4.76 ± 0.2 c |
| Se-NPs (1 mM) | 1.33 ± .0.06 a | 0.71 ± 0.04 a | 1.20 ± 0.06 a | 12.9 ± 0.5 a | 7.01 ± 0.5 a |
| Se-NPs (1.5 mM) | 1.20 ± 0.08 b | 0.65 ± 0.02 ab | 1.07 ± 0.07 ab | 11.2 ± 0.6 b | 5.97 ± 0.3 b |
| The second season (2020) | |||||
| Distilled water | 0.97 ± 0.04 c | 0.55 ± 0.01 b | 0.90 ± 0.02 b | 5.86 ± 0.3 d | 3.44 ± 0.1 d |
| Se-NPs (0.5 mM) | 1.14 ± 0.06 b | 0.65 ± 0.02 a | 1.01 ± 0.04 b | 9.88 ± 0.6 c | 4.94 ± 0.1 c |
| Se-NPs (1 mM) | 1.36 ± 0.07 a | 0.74 ± 0.02 a | 1.24 ± 0.04 a | 13.0 ± 0.8 a | 7.26 ± 0.4 a |
| Se-NPs (1.5 mM) | 1.23 ± 0.05 b | 0.68 ± 0.03 a | 1.10 ± 0.03 ab | 11.5 ± 0.7 b | 6.18 ± 0.3 b |
| Foliar Spray | RWC (%) | MSI (%) | Free proline (µmol g−1 DW) | Soluble sugars (mg g−1 DW) | Se content (mg kg−1 DW) |
|---|---|---|---|---|---|
| The first season (2019) | |||||
| Distilled water | 77.1 ± 2.2 c | 36.9 ± 1.2 d | 27.5 ± 1.3 d | 18.6 ± 1.1 d | 10.4 ± 0.2 d |
| Se-NPs (0.5 mM) | 79.6 ± 2.6 c | 48.9 ± 1.5 c | 29.5 ± 1.2 c | 21.3 ± 1.3 c | 21.6 ± 0.4 c |
| Se-NPs (1 mM) | 90.6 ± 3.5 a | 81.4 ± 2.5 a | 34.1 ± 1.3 a | 26.2 ± 1.4 a | 28.2 ± 0.5 b |
| Se-NPs (1.5 mM) | 86.3 ± 3.3 b | 61.0 ± 1.8 b | 32.4 ± 1.5 b | 24.1 ± 1.5 b | 36.4 ± 0.7 a |
| The second season (2020) | |||||
| Distilled water | 79.1 ± 2.5 b | 38.2 ± 1.1 d | 28.1 ± 1.4 c | 19.1 ± 1.2 d | 12.0 ± 0.2 d |
| Se-NPs (0.5 mM) | 80.9 ± 3.2 b | 50.6 ± 1.9 c | 30.1 ± 1.5 b | 21.8 ± 1.3 c | 24.2 ± 0.3 c |
| Se-NPs (1 mM) | 92.5 ± 3.4 a | 83.5 ± 2.9 a | 34.5 ± 1.4 a | 26.6 ± 1.4 a | 29.4 ± 0.6 b |
| Se-NPs (1.5 mM) | 88.1 ± 3.5 a | 63.1 ± 2.3 b | 33.1 ± 1.6 a | 24.5 ± 1.5 b | 37.0 ± 0.8 a |
| Foliar Spray | Blade Thic. (μm) | Palisade Thick. (μm) | Spongy Thick. (μm) | Length of Midvein (μm) | Width of Midvein (μm) | Phloem Thick. (μm) | Xylem Thick. (μm) | Diameter of Vessel (μm) |
|---|---|---|---|---|---|---|---|---|
| Distilled water | 130.6 ± 1.1d | 40.5 ± 0.9d | 54.3 ± 1.3d | 750.4 ± 2.6d | 619.1 ± 4.1d | 101.6 ± 1.1d | 105.4 ± 1.2d | 15.5 ± 0.4d |
| Se-NPs (0.5 mM) | 165.5 ± 1.5c | 49.2 ± 1.2c | 60.6 ± 1.2c | 904.5 ± 3.5c | 722.2 ± 2.5c | 117.3 ± 1.4c | 113.32 ± 1.4c | 20.3 ± 0.5c |
| Se-NPs (1 mM) | 250.8 ± 2.2a | 99.5 ± 1.3a | 114.8 ± 2.2a | 1389.9 ± 2.9a | 1283.6 ± 4.3a | 211.8 ± 1.5a | 211.2 ± 1.9a | 33.9 ± 0.8a |
| Se-NPs (1.5 mM) | 230.3 ± 1.9b | 85.4 ± 1.1b | 100.6 ± 2.5b | 1270.6 ± 3.5b | 1172.7 ± 3.6b | 181.2 ± 1.6b | 145.6 ± 1.7b | 29.2 ± 0.7b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rady, M.M.; Desoky, E.-S.M.; Ahmed, S.M.; Majrashi, A.A.; Ali, E.F.; Arnaout, S.M.A.I.; Selem, E. Foliar Nourishment with Nano-Selenium Dioxide Promotes Physiology, Biochemistry, Antioxidant Defenses, and Salt Tolerance in Phaseolus vulgaris. Plants 2021, 10, 1189. https://doi.org/10.3390/plants10061189
Rady MM, Desoky E-SM, Ahmed SM, Majrashi AA, Ali EF, Arnaout SMAI, Selem E. Foliar Nourishment with Nano-Selenium Dioxide Promotes Physiology, Biochemistry, Antioxidant Defenses, and Salt Tolerance in Phaseolus vulgaris. Plants. 2021; 10(6):1189. https://doi.org/10.3390/plants10061189
Chicago/Turabian StyleRady, Mostafa M., El-Sayed M. Desoky, Safia M. Ahmed, Ali A. Majrashi, Esmat F. Ali, Safaa M. A. I. Arnaout, and Eman Selem. 2021. "Foliar Nourishment with Nano-Selenium Dioxide Promotes Physiology, Biochemistry, Antioxidant Defenses, and Salt Tolerance in Phaseolus vulgaris" Plants 10, no. 6: 1189. https://doi.org/10.3390/plants10061189
APA StyleRady, M. M., Desoky, E.-S. M., Ahmed, S. M., Majrashi, A. A., Ali, E. F., Arnaout, S. M. A. I., & Selem, E. (2021). Foliar Nourishment with Nano-Selenium Dioxide Promotes Physiology, Biochemistry, Antioxidant Defenses, and Salt Tolerance in Phaseolus vulgaris. Plants, 10(6), 1189. https://doi.org/10.3390/plants10061189









